Polymorphism of Transferrin Gene Impacts the Mediating Effects of Psychotic Symptoms on the Relationship between Oxidative Stress and Cognition in Patients with Chronic Schizophrenia
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Clinical Examination and Cognitive Assessments
2.3. Measurement of Antioxidant Enzymes Activity
2.4. DNA Extraction and SNP Detection
2.5. Statistical Analysis
3. Results
3.1. Subject Characteristics and Genotypic Effects on Susceptibility to Schizophrenia
3.2. TF Gene Polymorphisms and Clinical Symptoms of Schizophrenia
3.3. Correlation among Psychotic Symptoms, Cognitive Performance, and Oxidative Stress in Schizophrenia Patients
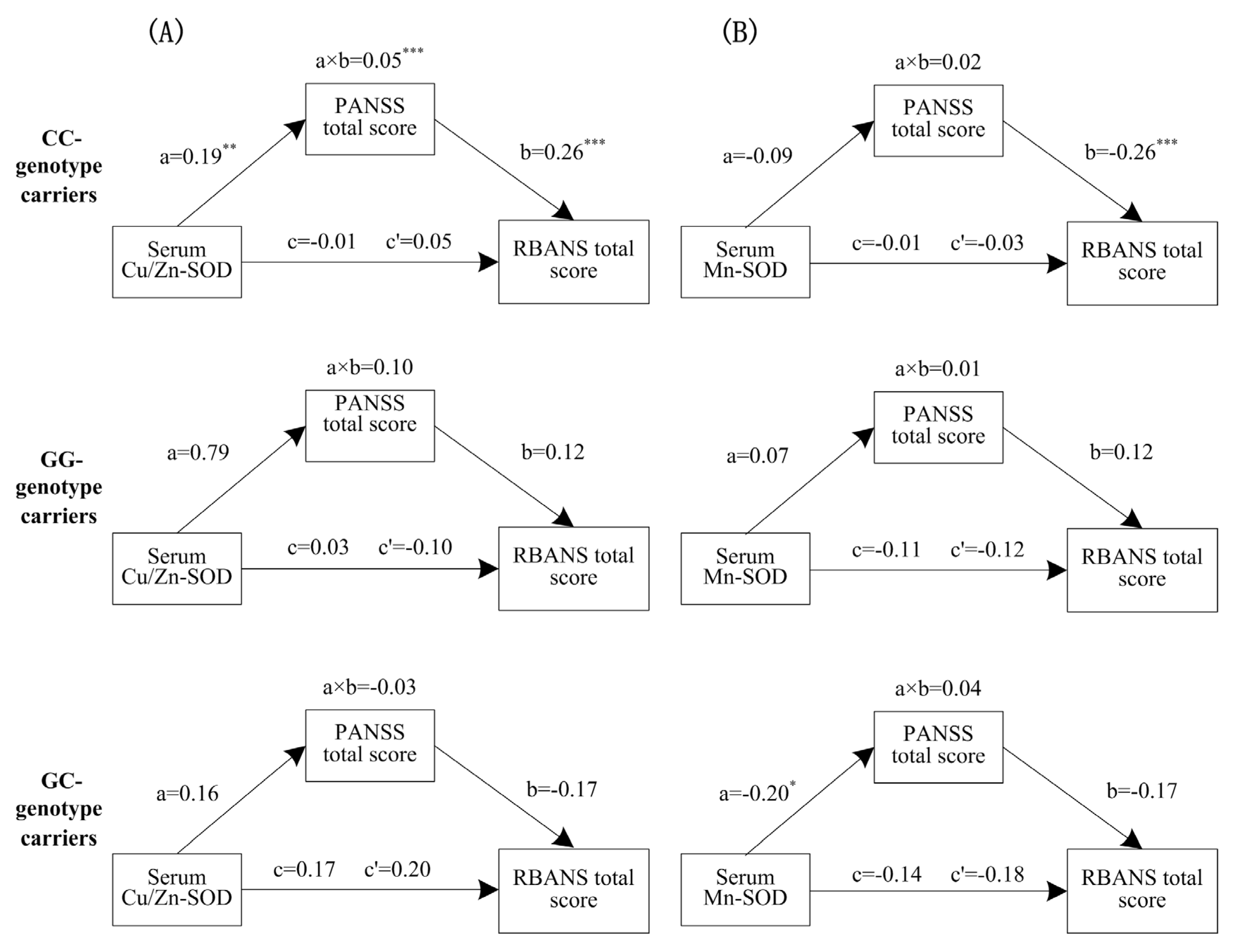
3.4. Mediation Effects of Psychotic Symptoms on the Relationship between Oxidative Stress and Cognitive Performance
3.5. The Mediating Effects Varied with Transferrin rs3811655 Polymorphism
4. Discussion
4.1. More Severe Symptoms, Worse Cognition, and Higher SOD Activity in Schizophrenia Patients Are Associated with the rs3811655-G Variant
4.2. The Mediating Role of Psychotic Symptoms between SOD and Cognition
4.3. The Mediation Model of Psychotic Symptoms Moderated by Transferrin Gene Polymorphism
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- O’Leary, D.S.; Flaum, M.; Kesler, M.L.; Flashman, L.A.; Arndt, S.; Andreasen, N.C. Cognitive correlates of the negative, disorganized, and psychotic symptom dimensions of schizophrenia. J. Neuropsychiatry Clin. Neurosci. 2000, 12, 4–15. [Google Scholar] [CrossRef]
- Walton, E.; Hibar, D.P.; van Erp, T.G.; Potkin, S.G.; Roiz-Santiañez, R.; Crespo-Facorro, B.; Suarez-Pinilla, P.; Van Haren, N.E.M.; de Zwarte, S.M.C.; Kahn, R.S.; et al. Left medial orbitofrontal cortical thinning is associated with negative symptom severity in schizophrenia: A meta-analysis by the ENIGMA-Schizophrenia consortium. Psychol. Med. 2018, 48, 82. [Google Scholar] [CrossRef]
- Reichenberg, A.; Velthorst, E.; Davidson, M. Cognitive impairment and psychosis in schizophrenia: Independent or linked conditions? World Psychiatry 2019, 18, 162. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.Y.; Xiu, M.H.; Wang, F.; Chen, D.C.; Qi, L.Y.; Sun, H.Q.; Chen, S.; He, S.C.; Wu, G.Y.; Haile, C.N.; et al. The novel oxidative stress marker thioredoxin is increased in first-episode schizophrenic patients. Schizophr. Res. 2009, 113, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Lang, X.; Wang, D.M.; Du, X.D.; Fang, Q.J.; Chen, D.C.; Xiu, M.; Wang, L.; Zhang, X.Y. Elevated activity of plasma superoxide dismutase in never-treated first-episode schizophrenia patients: Associated with depressive symptoms. Schizophr. Res. 2020, 222, 291–296. [Google Scholar] [CrossRef]
- Magalhães, P.V.; Dean, O.; Andreazza, A.C.; Berk, M.; Kapczinski, F. Antioxidant treatments for schizophrenia. Cochrane Database Syst. Rev. 2016, 2, CD008919. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maas, D.; Vallès, A.; Martens, G. Oxidative stress, prefrontal cortex hypomyelination and cognitive symptoms in schizophrenia. Transl. Psychiatry 2017, 7, e1171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Flatow, J.; Buckley, P.; Miller, B.J. Meta-analysis of oxidative stress in schizophrenia. Biol. Psychiatry 2013, 74, 400–409. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, M.; Zhao, Z.; He, L.; Wan, C. A meta-analysis of oxidative stress markers in schizophrenia. Sci. China Life Sci. 2010, 53, 112–124. [Google Scholar] [CrossRef]
- Murray, A.J.; Rogers, J.C.; Katshu, M.Z.U.H.; Liddle, P.F.; Upthegrove, R. Oxidative stress and the pathophysiology and symptom profile of Schizophrenia Spectrum Disorders. Front. Psychiatry 2021, 1235, 703452. [Google Scholar] [CrossRef]
- Wei, C.; Sun, Y.; Chen, N.; Chen, S.; Xiu, M.H.; Zhang, X.Y. Interaction of oxidative stress and BDNF on executive dysfunction in patients with chronic schizophrenia. Psychoneuroendocrinology 2020, 111, 104473. [Google Scholar] [CrossRef]
- Martínez-Cengotitabengoa, M.; Mac-Dowell, K.S.; Leza, J.C.; Micó, J.A.; Fernandez, M.; Echevarria, E.; Sanjuan, J.; Elorza, J.; González-Pinto, A. Cognitive impairment is related to oxidative stress and chemokine levels in first psychotic episodes. Schizophr. Res. 2012, 137, 66–72. [Google Scholar] [CrossRef]
- Schrag, M.; Mueller, C.; Zabel, M.; Crofton, A.; Kitsch, W.M.; Ghribi, O.; Squitti, R.; Perry, G. Oxidative stress in blood in Alzheimer’s disease and mild cognitive impairment: A meta-analysis. Neurobiol. Dis. 2013, 59, 100–110. [Google Scholar] [CrossRef]
- Evola, M.; Hall, A.; Wall, T.; Young, A.; Grammas, P. Oxidative stress impairs learning and memory in apoE knockout mice. Pharmacol. Biochem. Behav. 2010, 96, 181–186. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahluwalia, N. Diagnostic utility of serum transferrin receptors measurement in assessing iron status. Nutr. Rev. 1998, 56, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-W.; Stewart, R.; Park, W.-Y.; Jhon, M.; Lee, J.-Y.; Kim, S.-Y.; Kim, J.-M.; Amminger, P.; Chung, Y.-C.; Yoon, J.-S. Latent iron deficiency as a marker of negative symptoms in patients with first-episode schizophrenia spectrum disorder. Nutrients 2018, 10, 1707. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davis, K.L.; Stewart, D.G.; Friedman, J.I.; Buchsbaum, M.; Harvey, P.D.; Hof, P.R.; Buxbaum, J.; Har4outunian, V. White matter changes in schizophrenia: Evidence for myelin-related dysfunction. Arch. Gen. Psychiatry 2003, 60, 443–456. [Google Scholar] [CrossRef] [PubMed]
- Maeno, N.; Takahashi, N.; Saito, S.; Ji, X.; Branko, A.; Ishihara, R.; Yoshida, K.; Inada, T.; Iidaka, T.; Ozaki, N. Association study between the transferrin gene and schizophrenia in the Japanese population. Neuro Rep. 2007, 18, 517–520. [Google Scholar] [CrossRef]
- Qu, M.; Yue, W.; Tang, F.; Wang, L.; Han, Y.; Zhang, D. Polymorphisms of Transferrin gene are associated with schizophrenia in Chinese Han population. J. Psychiatr. Res. 2008, 42, 877–883. [Google Scholar] [CrossRef]
- Buretić-Tomljanović, A.; Vraneković, J.; Rubeša, G.; Jonovska, S.; Tomljanovic, D.; Sendula-Jengic, V.; Kapovic, M.; Ristic, S. HFE mutations and transferrin C1/C2 polymorphism among Croatian patients with schizophrenia and schizoaffective disorder. Mol. Biol. Rep. 2012, 39, 2253–2258. [Google Scholar] [CrossRef] [PubMed]
- Bitanihirwe, B.K.; Woo, T.-U.W. Oxidative stress in schizophrenia: An integrated approach. Neurosci. Biobehav. Rev. 2011, 35, 878–893. [Google Scholar] [CrossRef] [Green Version]
- Jahanshad, N.; Kohannim, O.; Hibar, D.P.; Stein, J.L.; McMahon, K.L.; de Zubicaray, G.I.; Medland, S.E.; Montgomery, G.W.; Whitfield, J.B.; Martin, N.G.; et al. Brain structure in healthy adults is related to serum transferrin and the H63D polymorphism in the HFE gene. Proc. Natl. Acad. Sci. USA 2012, 109, E851–E859. [Google Scholar] [CrossRef] [Green Version]
- Yamaji, Y.; Nakazato, Y.; Oshima, N.; Hayashi, M.; Saruta, T. Oxidative stress induced by iron released from transferrin in low pH peritoneal dialysis solution. Nephrol. Dial. Transplant. 2004, 19, 2592–2597. [Google Scholar] [CrossRef] [Green Version]
- Kay, S.R.; Fiszbein, A.; Opler, L.A. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr. Bull. 1987, 13, 261–276. [Google Scholar] [CrossRef] [PubMed]
- Randolph, C.; Tierney, M.C.; Mohr, E.; Chase, T.N. The Repeatable Battery for the Assessment of Neuropsychological Status (RBANS): Preliminary clinical validity. J. Clin. Exp. Neuropsychol. 1998, 20, 310–319. [Google Scholar] [CrossRef]
- Miller, S.A.; Dykes, D.; Polesky, H. A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res. 1988, 16, 1215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jahanshad, N.; Rajagopalan, P.; Thompson, P.M. Neuroimaging, nutrition, and iron-related genes. Cell. Mol. Life Sci. 2013, 70, 4449–4461. [Google Scholar] [CrossRef] [Green Version]
- Benyamin, B.; McRae, A.F.; Zhu, G.; Gordon, S.; Henders, A.K.; Palotie, A.; Peltonen, L.; Martin, N.G.; Montgomery, G.W.; Whitfield, J.B.; et al. Variants in TF and HFE explain ~40% of genetic variation in serum-transferrin levels. Am. J. Hum. Genet. 2009, 84, 60–65. [Google Scholar] [CrossRef] [Green Version]
- Wysokinski, D.; Danisz, K.; Blasiak, J.; Dorecka, M.; Romaniuk, D.; Szaflik, J.; Szaflik, J.P. An association of transferrin gene polymorphism and serum transferrin levels with age-related macular degeneration. Exp. Eye Res. 2013, 106, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.; Xia, T.; Hu, W.; Chen, S.; Chi, S.; Lei, Y.; Liu, Z. Visualizing the regulation of hydroxyl radical level by superoxide dismutase via a specific molecular probe. Anal. Chem. 2018, 90, 1317–1324. [Google Scholar] [CrossRef]
- Raffa, M.; Mechri, A.; Othman, L.B.; Fendri, C.; Gaha, L.; Kerkeni, A. Decreased glutathione levels and antioxidant enzyme activities in untreated and treated schizophrenic patients. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2009, 33, 1178–1183. [Google Scholar] [CrossRef]
- Zhang, X.Y.; Zhou, D.F.; Cao, L.Y.; Zhang, P.Y.; Wu, G.Y.; Shen, Y.C. The effect of risperidone treatment on superoxide dismutase in schizophrenia. J. Clin. Psychopharmacol. 2003, 23, 128–131. [Google Scholar] [CrossRef] [PubMed]
- Popa-Wagner, A.; Mitran, S.; Sivanesan, S.; Chang, E.; Buga, A.-M. ROS and brain diseases: The good, the bad, and the ugly. Oxid. Med. Cell. Longev. 2013, 2013, 963520. [Google Scholar] [CrossRef] [PubMed]
- Xiu, M.H.; Li, Z.; Chen, D.C.; Chen, S.; Curbo, M.E.; Wu, H.E.; Tong, Y.S.; Tan, S.P.; Zhang, X.Y. Interrelationships between BDNF, superoxide dismutase, and cognitive impairment in drug-naive first-episode patients with schizophrenia. Schizophr. Bull. 2020, 46, 1498–1510. [Google Scholar] [CrossRef] [PubMed]
- Hendouei, N.; Farnia, S.; Mohseni, F.; Salehi, A.; Bagheri, M.; Shadfar, F.; Barzegar, F.; Hoseini, S.D.; Charati, J.Y.; Shaki, F. Alterations in oxidative stress markers and its correlation with clinical findings in schizophrenic patients consuming perphenazine, clozapine and risperidone. Biomed. Pharmacother. 2018, 103, 965–972. [Google Scholar] [CrossRef] [PubMed]
- Mondola, P.; Damiano, S.; Sasso, A.; Santillo, M. The Cu, Zn superoxide dismutase: Not only a dismutase enzyme. Front. Physiol. 2016, 7, 594. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moya, G.E.; Rivera, P.D.; Dittenhafer-Reed, K.E. Evidence for the role of mitochondrial DNA release in the inflammatory response in neurological disorders. Int. J. Mol. Sci. 2021, 22, 7030. [Google Scholar] [CrossRef]
- Prabakaran, S.; Swatton, J.; Ryan, M.; Huffaker, S.J.; Huang, J.T.-J.; Griffin, J.L.; Wayland, M.; Freeman, T.; Dudbridge, F.; Lilley, K.S.; et al. Mitochondrial dysfunction in schizophrenia: Evidence for compromised brain metabolism and oxidative stress. Mol. Psychiatry 2004, 9, 684–697. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trześniowska-Drukała, B.; Kalinowska, S.; Safranow, K.; Kloda, K.; Misiak, B.; Samochowiec, J. Evaluation of hyperhomocysteinemia prevalence and its influence on the selected cognitive functions in patients with schizophrenia. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2019, 95, 109679. [Google Scholar] [CrossRef]
- Kalinowska, S.; Trześniowska-Drukała, B.; Safranow, K.; Pełka-Wysiecka, J.; Kloda, K.; Misiak, B.; Samochowiec, J. Association between thyroid function and metabolic syndrome in male and female schizophrenia patients. Psychiatry Res. 2019, 274, 167–175. [Google Scholar] [CrossRef]

| Variable | Schizophrenia (n = 564) | Healthy Control (n = 468) | t/χ2 | p |
|---|---|---|---|---|
| Gender (M/F) | 499/65 | 194/274 | 256.38 | 0.00 *** |
| Age (years) | 47.8 ± 9.0 | 44.9 ± 13.6 | −3.97 | 0.00 *** |
| Education (years) | 8.9 ± 4.5 | 9.7 ± 5.3 | 2.40 | 0.02 ** |
| Body mass index (kg/m2) | 24.8 ± 3.9 | 25.1 ± 4.1 | 1.14 | 0.26 |
| Smokers (%) | 396/564 | 161/468 | 132.04 | 0.00 *** |
| Mean antipsychotic dose (as CPZ equivalents) (mg/d) | 433.6 ± 366.6 | |||
| Types of antipsychotics (typical/atypical) | 93/459 | |||
| TF allele frequency (%) | ||||
| C | 78.5% | 78.2% | 0.03 | 0.85 |
| G | 21.5% | 21.8% | ||
| TF genotype distribution [n (%)] | ||||
| CC | 350 (62.0%) | 256 (60.6%) | 0.64 | 0.73 |
| GG | 28 (5.0%) | 18 (4.3%) | ||
| GC | 186 (33.0%) | 148 (35.1%) |
| 5 | Schizophrenia | |||
|---|---|---|---|---|
| C/C (n = 350) | G/G (n = 28) | G/C (n = 186) | F/χ2 (p) | |
| PANSS scores | ||||
| P subscore | 11.1 ± 4.6 | 11.0 ± 4.9 | 11.4 ± 5.0 | 0.47 (0.62) |
| N subscore | 21.7 ± 7.2 | 25.4 ± 7.3 | 23.0 ± 7.3 | 4.37 (<0.05) |
| G subscore | 24.6 ± 5.2 | 24.7 ± 4.8 | 25.2 ± 6.0 | 0.89 (0.41) |
| total score | 57.3 ± 13.0 | 61.2 ± 11.7 | 59.5 ± 14.7 | 2.42 (0.09) |
| RBANS Scores | ||||
| Immediate Memory | 58.6 ± 16.2 | 54.8 ± 13.9 | 59.2 ± 16.5 | 0.51 (0.60) |
| Visuospatial/Constructional | 77.6 ± 18.8 | 73.1 ± 20.8 | 77.7 ± 18.2 | 0.95 (0.39) |
| Language | 81.7 ± 15.8 | 76.8 ± 15.0 | 82.1 ± 14.4 | 1.42 (0.24) |
| Attention | 71.4 ± 17.5 | 61.4 ± 14.7 | 70.3 ± 17.4 | 6.18 (<0.001) |
| Delayed Memory | 67.2 ± 19.0 | 63.5 ± 19.1 | 65.6 ± 19.8 | 0.85 (0.43) |
| Total scale | 64.7 ± 15.1 | 60.4 ± 12.7 | 64.5 ± 14.9 | 1.33 (0.27) |
| Biological Markers of Oxidative Stress | C/C (n = 168) | G/G (n = 11) | G/C (n = 96) | |
| Total SOD activity | 77.4 ± 8.3 | 80.2 ± 14.6 | 77.8 ± 11.6 | 0.37 (0.69) |
| Cu/Zn-SOD activity | 57.4 ± 17.1 | 45.4 ± 24.9 | 53.9 ± 19.6 | 2.35 (0.10) |
| Mn-SOD activity | 20.0 ± 14.7 | 34.8 ± 18.0 | 23.9 ± 16.0 | 4.95 (<0.05) |
| MDA content | 2.3 ± 2.0 | 3.0 ± 1.9 | 2.1 ± 8.1 | 0.23 (0.79) |
| PANSS Total Score | RBANS Total Score | Total SOD | Cu/Zn-SOD | Mn-SOD | MDA Content | |
|---|---|---|---|---|---|---|
| PANSS total score | - | |||||
| RBANS total score | −0.23 *** | - | ||||
| Total SOD | 0.12 | 0.01 | - | |||
| Cu/Zn-SOD | 0.16 ** | 0.05 | 0.54 *** | - | ||
| Mn-SOD | −0.12 * | −0.06 | −0.01 | −0.85 *** | - | |
| MDA content | −0.04 | −0.09 | −0.05 | −0.07 | 0.05 | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, P.; Wang, D.; Xiu, M.; Chen, D.; Lackey, B.; Wu, H.E.; Zhou, J.; Zhang, X. Polymorphism of Transferrin Gene Impacts the Mediating Effects of Psychotic Symptoms on the Relationship between Oxidative Stress and Cognition in Patients with Chronic Schizophrenia. Antioxidants 2022, 11, 125. https://doi.org/10.3390/antiox11010125
Chen P, Wang D, Xiu M, Chen D, Lackey B, Wu HE, Zhou J, Zhang X. Polymorphism of Transferrin Gene Impacts the Mediating Effects of Psychotic Symptoms on the Relationship between Oxidative Stress and Cognition in Patients with Chronic Schizophrenia. Antioxidants. 2022; 11(1):125. https://doi.org/10.3390/antiox11010125
Chicago/Turabian StyleChen, Pinhong, Dongmei Wang, Meihong Xiu, Dachun Chen, Blake Lackey, Hanjing E. Wu, Jin Zhou, and Xiangyang Zhang. 2022. "Polymorphism of Transferrin Gene Impacts the Mediating Effects of Psychotic Symptoms on the Relationship between Oxidative Stress and Cognition in Patients with Chronic Schizophrenia" Antioxidants 11, no. 1: 125. https://doi.org/10.3390/antiox11010125
APA StyleChen, P., Wang, D., Xiu, M., Chen, D., Lackey, B., Wu, H. E., Zhou, J., & Zhang, X. (2022). Polymorphism of Transferrin Gene Impacts the Mediating Effects of Psychotic Symptoms on the Relationship between Oxidative Stress and Cognition in Patients with Chronic Schizophrenia. Antioxidants, 11(1), 125. https://doi.org/10.3390/antiox11010125








