Melatonin as an Antioxidant and Immunomodulator in Atopic Dermatitis—A New Look on an Old Story: A Review
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. MT Secretion and Blood Level in Patients with AD
3.2. Oxidative Stress Markers, Melatonin and Severity of Atopic Dermatitis
3.3. Melatonin and Sleep Quality in Patients with Atopic Dermatitis
3.4. Effect of MT Supplementation on Atopic Dermatitis
3.5. Results of Experimental Studies on the Effect of MT on AD
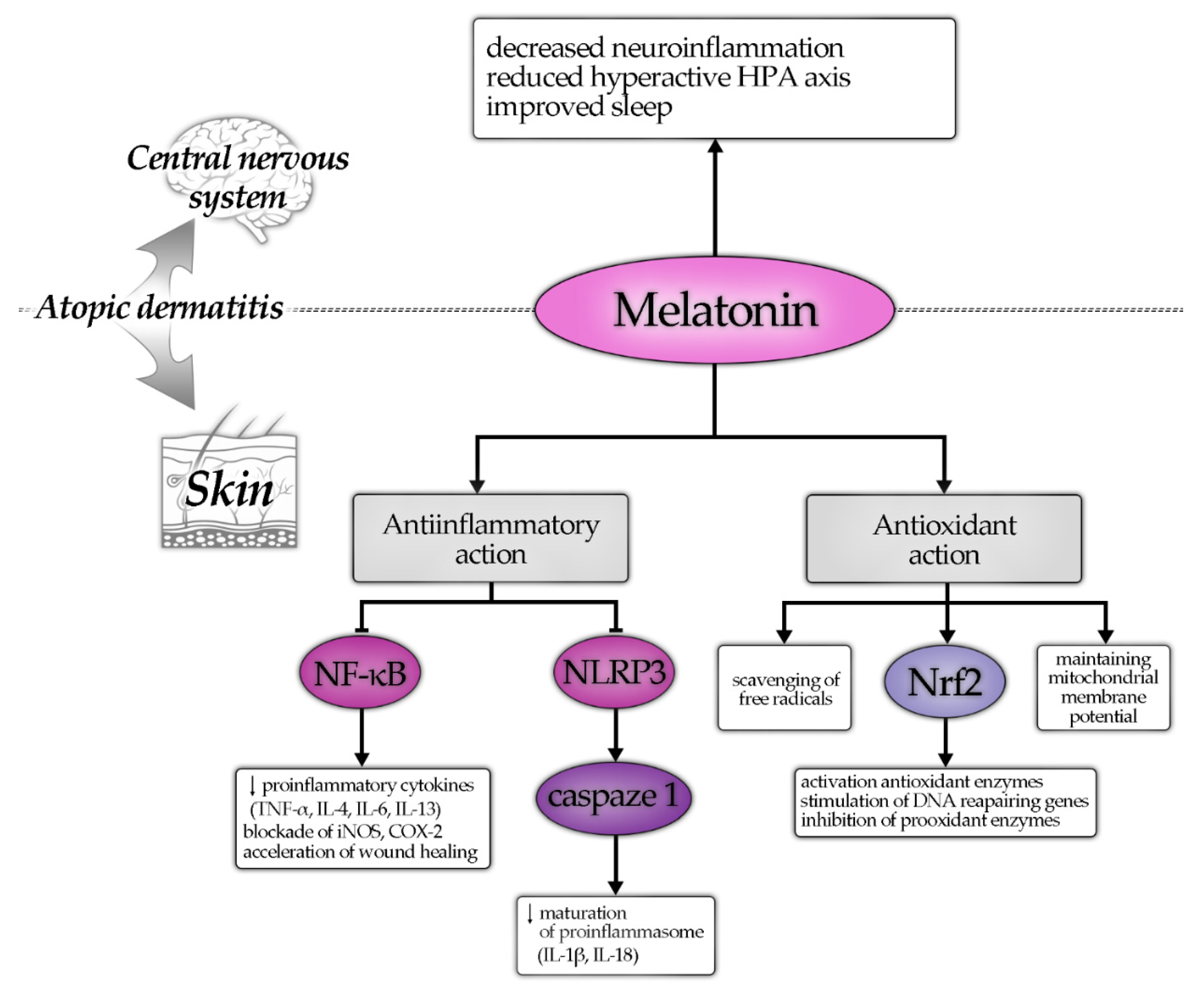
4. Discussion
5. Conclusions
- Based on previous and recent publications [103], it could be stated that AD is related to the increase in oxidative stress, and since MT is powerful antioxidant, its implication in the defense of inflammatory reaction of AD patients is very likely.
- Lower serum MT is correlated with more severe inflammation in AD patients.
- MT supplementation improves sleep and, with application of adequate dose, reduces SCORAD and serum IgE in AD patients.
Author Contributions
Funding
Conflicts of Interest
References
- Ständer, S. Atopic Dermatitis. N. Engl. J. Med. 2021, 384, 1136–1143. [Google Scholar] [CrossRef] [PubMed]
- Silverberg, J.I.; Garg, N.K.; Paller, A.S.; Fishbein, A.B.; Zee, P.C. Sleep disturbances in adults with eczema are associated with impaired overall health: A US population-based study. J. Investig. Dermatol. 2015, 135, 56–66. [Google Scholar] [CrossRef] [Green Version]
- Weidinger, S.; Beck, L.A.; Bieber, T.; Kabashima, K.; Irvine, A.D. Atopic dermatitis. Nat. Rev. Dis. Primers 2018, 4. [Google Scholar] [CrossRef]
- Laughter, M.R.; Maymone, M.B.C.; Mashayekhi, S.; Arents, B.W.M.; Karimkhani, C.; Langan, S.M.; Dellavalle, R.P.; Flohr, C. The global burden of atopic dermatitis: Lessons from the Global Burden of Disease Study 1990–2017. Br. J. Dermatol. 2020, 184, 304–309. [Google Scholar] [CrossRef]
- Jaworek, A.K.; Szafraniec, K.; Jaworek, M.; Hałubiec, P.; Wojas-Pelc, A. Is the presence of a fur animal an exacerbating factor of atopic dermatitis in adults? Pol. Merkur. Lek. 2020, 48, 19–22. [Google Scholar]
- Weidinger, S.; Novak, N. Atopic dermatitis. Lancet 2016, 387, 1109–1122. [Google Scholar] [CrossRef]
- Langan, S.M.; Irvine, A.D.; Weidinger, S. Atopic dermatitis. Lancet 2020, 396, 345–360. [Google Scholar] [CrossRef]
- Andersen, R.M.; Thyssen, J.P.; Maibach, H.I. Qualitative vs. quantitative atopic dermatitis criteria–in historical and present perspectives. J. Eur. Acad. Dermatol. Venereol. 2016, 30, 604–618. [Google Scholar] [CrossRef] [PubMed]
- Yew, Y.W.; Thyssen, J.P.; Silverberg, J.I. A systematic review and meta-analysis of the regional and age-related differences in atopic dermatitis clinical characteristics. J. Am. Acad. Dermatol. 2019, 80, 390–401. [Google Scholar] [CrossRef]
- Mack, M.R.; Kim, B.S. The itch-scratch cycle: A neuroimmune perspective. Trends. Immunol. 2018, 39, 980–991. [Google Scholar] [CrossRef] [PubMed]
- Jaworek, A.K.; Szafraniec, K.; Zuber, Z.; Wojas-Pelc, A.; Jaworek, J. Interleukin 25, thymic stromal lymphopoietin and house dust mites in pathogenesis of atopic dermatitis. J. Physiol. Pharmacol. 2020, 71. [Google Scholar] [CrossRef]
- Martin, M.J.; Estravís, M.; García-Sánchez, A.; Dávila, I.; Isidoro-García, M.; Sanz, C. Genetics and epigenetics of atopic dermatitis: An updated systematic review. Genes 2020, 11, 442. [Google Scholar] [CrossRef] [Green Version]
- Tokura, Y. Extrinsic and intrinsic types of atopic dermatitis. J. Derm. Sci. 2010, 58, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Fu, J.; Zhou, Y. Research progress in atopic march. Front. Immunol. 2020, 11, 1907. [Google Scholar] [CrossRef]
- Eichenfield, L.F.; Tom, W.L.; Chamlin, S.L.; Feldman, S.R.; Hanifin, J.M.; Simpson, E.L.; Bergman, J.N.; Cohen, D.E.; Cooper, K.D.; Cordoro, K.M. Guidelines of care for the management of atopic dermatitis: Section 1. Diagnosis and assessment of atopic dermatitis. J. Am. Acad. Dermatol. 2014, 70, 338–351. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Severity Scoring of Atopic Dermatitis: The SCORAD Index. Dermatology 1993, 186, 23–31. [CrossRef] [PubMed]
- Chang, Y.-S.; Chiang, B.-L. Sleep disorders and atopic dermatitis: A 2-way street? J. Allergy Clin. Immunol. 2018, 142, 1033–1040. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dogan, D.G.; Canaloglu, S.K.; Kivilcim, M.; Kum, Y.E.; Topal, E.; Catal, F. Sleep patterns of young children with newly diagnosed atopic dermatitis. Adv. Derm. Allergol. 2017, 34, 143–147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hon, K.L.; Leung, T.F.; Ma, K.C.; Li, A.M.; Wong, Y.; Yin, J.A.; Fok, T.F. Resting energy expenditure, oxygen consumption and carbon dioxide production during sleep in children with atopic dermatitis. J. Dermatolog. Treat. 2005, 16, 22–25. [Google Scholar] [CrossRef] [PubMed]
- Meltzer, L.J.; Moore, M. Sleep disruptions in parents of children and adolescents with chronic illnesses: Prevalence, causes, and consequences. J. Pediatr. Psychol. 2008, 33, 279–291. [Google Scholar] [CrossRef] [Green Version]
- Claustrat, B.; Brum, J.; Chazot, G. The basic physiology and pathophysiology of melatonin. Sleep Med. Rev. 2005, 9, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Liu, X.; Rosales-Corral, S.A.; Acuna-Castroviejo, D.; Reiter, R.J. Mitochondria and chloroplasts as the original sites of melatonin synthesis: A hypothesis related to melatonin’s primary function and evolution in eukariotes. J. Pineal Res. 2013, 54, 127–138. [Google Scholar] [CrossRef]
- Back, K.; Tan, D.X.; Reiter, R.J. Melatonin biosynthesis in plants: Multiple pathways catalyze tryptophan to melatonin in the cytoplasm of chloroplasts. J. Pineal Res. 2016, 61, 426–437. [Google Scholar] [CrossRef] [PubMed]
- Hardeland, R.; Poeggeler, B. Melatonin and synthetic melatonergic agonists: Actions and metabolism in the central nervous system. Cent. Nerv. Syst. Agents Med. Chem. 2012, 12, 189–216. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Zheng, X.; Kong, J.; Manchester, L.C.; Haderland, R.; Kim, S.J.; Xu, X.; Reiter, R.J. Fundamental issue related to the origin of melatonin and melatonin isomers during evolution: Relation to their biological functions. Int. J. Mol. Sci. 2014, 15, 15858–15890. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tan, D.X.; Haderland, R.; Manchester, L.C.; Korkmas, A.; Ma, S.; Rosales–Corral, S.; Reiter, R.J. Functional roles of melatonin in plants, and perspectives in nutritional and agricultural science. J. Exp. Bot. 2012, 63, 577–597. [Google Scholar] [CrossRef]
- Haderland, R.; Balzer, I.; Poeggler, B.; Fuhrberg, B.; Uria, H.; Behrmann, G.; Wolf, R.; Meyer, T.J.; Reiter, R.J. On the primary functions of melatonin in evolution: Mediation of photoperiodic signals in a unicell, photooxidation, and scavenging of free radicals. J. Pineal Res. 1995, 18, 104–111. [Google Scholar]
- Reiter, R.J.; Rosales–Corral, S.; Tan, D.X.; Jou, M.J.; Galano, A.; Xu, B. Melatonin as a mitochondria-targeted antioxidant: One of evolution’s best ideas. Cell. Mol. Life. Sci. 2017, 74, 3863–3881. [Google Scholar] [CrossRef] [PubMed]
- Kurowska, A.; Bodys-Cupak, I.; Staszkiewicz, M.; Szklarczyk, J.; Zalewska-Puchala, J.; Klis-Kalinowska, A.; Makara-Studzińska, M.; Majda, A. Interleukin 6 and melatonin as predictors of cognitive, emotional and functional ageing in older people. Int. J. Environ. Res. Pub. Health 2020, 17, 3623. [Google Scholar] [CrossRef] [PubMed]
- Cardinali, D.P. Melatonin and healthy aging. Vitam. Horm. 2021, 115, 167–188. [Google Scholar]
- Chen, C.Q.; Fichna, J.; Bashashati, M.; Li, Y.Y.; Storr, M. Distribution, function and physiological role of melatonin in the lower gut. World J. Gastroenterol. 2011, 17, 3888–3898. [Google Scholar] [CrossRef]
- Messner, M.; Huether, G.; Lorf, T. Presence of melatonin in the human hepatobiliary tract. Life Sci. 2001, 69, 543–551. [Google Scholar] [CrossRef]
- Bubenik, G.A. Gastrointestinal melatonin: Localization, function, and clinical relevance. Dig. Dis. Sci. 2002, 47, 2336–2348. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhu, T.; Ma, X.; Wang, Y.; Liu, J.; Li, G.; Liu, Y.; Ji, P.; Liu, G. Melatonergic systems of AANAT, melatonin, and its receptor MT2 in the corpus luteum are essential for reproductive success in mammals. Biol. Reprod. 2021, 104, 430–444. [Google Scholar] [CrossRef]
- Acuna-Castroviejo, D.; Escames, G.; Venegas, C.; Diaz-Casado, M.E.; Lima-Cabello, R.; Lopez, L.C.; Rosales-Corral, S.; Tan, D.X.; Reiter, R.J. Extrapineal melatonin: Sources, regulation and potential functions. Cell. Mol. Life Sci. 2014, 71, 2997–3025. [Google Scholar] [CrossRef] [PubMed]
- Muxel, S.M.; Pires-Lapa, M.A.; Monteiro, A.W.A.; Cecon, E.; Tamura, E.K.; Floeter-Winter, L.M.; Markus, R.P. NF-κB drives the synthesis of melatonin in RAW 264.7 macrophages by inducing the transcription of the arylalkylamine-N-acetyltransferase (AA-NAT) gene. PLoS ONE, 2012; 7, e52010. [Google Scholar]
- Carrillo-Vico, A.; Calvo, J.R.; Abreu, P.; Lardone, P.J.; Garcia-Maurino, S.; Reiter, R.J.; Guerrero, J. Evidence of melatonin synthesis by human lymphocytes and its physiological significance: Possible role as intracrine, autocrine, and/or paracrine substance. FASEB J. 2004, 18, 537–539. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.K.; Lin, Z.; Tidwell, W.J.; Li, W.; Slominski, A. Melatonin and its metabolites accumulate in the human epidermis in vivo and inhibit proliferation and tyrosinase activity in epidermal melanocytes in vitro. Mol. Cell. Endocrinol. 2013, 404, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Fischer, T.W.; Scholz, G.; Knoll, B.; Hipler, U.C.; Elsner, P. Melatonin suppress reactive oxygen species inUV-irradiated leukocytes more than vitamin C and trolox. Skin Pharmacol. Sppl. Skin Physiol. 2002, 15, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Fischer, T.W.; Slominski, A.; Zmijewski, M.A.; Reiter, R.J.; Paus, R. Melatonin as a major skin protectant from free radical scavengivg to DNA damage repair. Exp. Dermatol. 2008, 17, 713–730. [Google Scholar] [CrossRef]
- Fischer, T.W.; Trueb, R.M.; Hanggi, G.; Innocenti, M.; Elsner, P. Topical melatonin for treatment of androgenetic alopecia. Int. J. Trichology 2012, 4, 236–245. [Google Scholar] [CrossRef] [Green Version]
- Skobowiat, C.; Brozyna, A.; Janetovic, Z.; Jeang, S.; Oak, A.S.W.; Kim, T.K.; Paruch, U.; Reiter, R.J.; Slominski, A. Melatonin and its derivatives counteracts the ultraviolet B radiation-induced damage in human and porcine skin ex vivo. J. Pineal Res. 2018, 65, 1–16. [Google Scholar] [CrossRef]
- Ozler, M.; Simsek, K.; Ozkan, C.; Akgul, E.O.; Topal, T.; Oter, S. Comparison of the effect of topical and systemic melatonin administration on delayed wound healing in rats that underwent pinealectomy. Scand. J. Clin. Lab. Investig. 2010, 70, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Song, R.; Ren, L.; Ma, H.; Hu, R.; Gao, H.; Wang, L.; Hen, X.; Zhao, Z.; Liu, J. Melatonin promotes diabetic wound healing in vitro by regulating keratinocyte activity. Am. J. Transl. Res. 2016, 8, 4682–4693. [Google Scholar] [PubMed]
- Park, E.K.; Lee, H.J.; Lee, H.; Kim, J.H.; Hvang, J.; Koo, J.I.; Kim, S.H. The anti-wrinkle mechanism of melatonin in UVB treated HaCAT keratinocytes and hairless mice via inhibition of ROS and sonic hedgehog mediated inflammatory proteins. Int. J. Mol. Sci. 2018, 19, 1995. [Google Scholar] [CrossRef] [Green Version]
- Slominski, A.; Pisarchik, A.; Semak, I.; Sweatman, T.; Wortsman, J.; Szczesniewski, A.; Slugocki, G.; McNulty, J.; Kauser, S.; Tobin, D.J. Serotonergic and melatonergic systems are fully expressed in human skin. FASEB J. 2002, 16, 896–898. [Google Scholar] [CrossRef] [Green Version]
- Venegas, C.; García, J.A.; Escames, G.; Ortiz, F.; López, A.; Doerrier, C.; García-Corzo, L.; López, L.C.; Reiter, R.J.; Acuña-Castroviejo, D. Extrapineal melatonin: Analysis of its subcellular distribution and daily fluctuations. J. Pineal Res. 2012, 52, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.; Semak, I.; Fischer, T.W.; Kim, T.K.; Kleszczynski, K.; Haderland, R.; Reiter, R.J. Metabolism of melatonin in the skin.: Why is important? Exp. Dermatol. 2017, 26, 563–568. [Google Scholar] [CrossRef]
- Haderland, R.; Tan, D.X.; Reiter, R.J. Kynuramines, metabolites of melatonin and other indoles: The resurrection of an almost forgotten class of biogenic amines. J. Pineal Res. 2009, 42, 109–126. [Google Scholar]
- Liu, L.; Labani, N.; Cecon, E.; Jockers, J. Melatonin target proteins: Too many or not enough. Front. Endocrinol. (Lausanne) 2019, 10, 791. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Slominski, A.; Haderland, R.; Zmijewski, M.A.; Slominski, R.M.; Reiter, R.J.; Paus, R. Melatonin: A cutaneous perspective on its production, metabolism and functions. J. Invest. Dermatol. 2018, 138, 490–499. [Google Scholar] [CrossRef] [Green Version]
- Rusanova, I.; Martinez-Ruiz, L.; Florido, J.; Rodriguez-Santana, C.; Guerra-Librero, A.; Acuna-Castroviejo, D.; Escames, G. Protective effect of melatonin on the skin: Further perspectives. Int. J. Mol. Sci. 2019, 20, 4948. [Google Scholar] [CrossRef] [Green Version]
- Boutin, J.A.; Ferry, G. Is there sufficient evidence that the melatonin binding site MT3 is quinone reductase 2? J. Pharm. Exp. Ther. 2019, 368, 59–65. [Google Scholar] [CrossRef] [Green Version]
- Singh, M.; Jadhav, H.R. Melatonin function and ligands. Drug. Discov. Today. 2014, 19, 1410–1418. [Google Scholar] [CrossRef]
- Xiong, J.; Wang, Z.; Cao, J.; Dong, Y.; Chen, Y. Melatonin mediates monochromatic light-induced proliferation of T/B lymphocytes in the spleen via the membrane receptors or nuclear receptor. Poult. Sci. 2020, 99, 4294–4302. [Google Scholar] [CrossRef]
- Dai, J.; Ho, K.; Park, J.M.; Fischer, D.E. Topical ROR inverse agonists suppress inflammation in mouse models of atopic dermatitis and acute irritant dermatitis. J. Invest. Dermatol. 2017, 137, 2523–2531. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sunyer-Figueres, M.; Vazquez, J.; Mas, A.; Torija, M.J.; Beltran, G. Transcriptomic insight into the effect of melatonin in Saccharomyces cerevisiae in the presence and absence of oxidative stress. Antioxidants (Basel) 2020, 9, 947. [Google Scholar] [CrossRef] [PubMed]
- Acuna-Castroviejo, D.; Lopez, L.C.; Escames, G.; Lopez, A.; Garcia, J.A.; Reiter, R.J. Melatonin-mitochondria interplay in health and disease. Curr. Top. Med. Chem. 2011, 11, 221–240. [Google Scholar] [CrossRef]
- García, J.J.; Lõpez-Pingarrõn, L.; Almeida-Souza, P.; Tres, A.; Escudero, P.; García-Gil, F.A.; Tan, D.X.; Reiter, R.J.; Ramírez, J.M.; Bernal-Pérez, M. Protective effects of melatonin in reducing oxidative stress and in preserving the fluidity of biological membranes: A review. J. Pineal Res. 2014, 56, 225–237. [Google Scholar] [CrossRef]
- Reiter, R.J.; Ma, Q.; Sharma, R. Melatonin in mitochondria: Migrating clear and present dangers. Physiology (Bethesda) 2020, 35, 86–95. [Google Scholar] [CrossRef]
- Beaupre, L.M.M.; Brown, G.M.; Gonçalves, V.F.; Kennedy, J.L. Melatonin’s neuroprotective role in mitochondria and its potential as a biomarker in aging, cognition and psychiatric disorders. Transl. Psychiatry 2021, 11, 339. [Google Scholar] [CrossRef] [PubMed]
- Martín Giménez, V.M.; de las Heras, N.; Ferder, L.; Lahera, V.; Reiter, R.J.; Manucha, W. Potential Effects of Melatonin and Micronutrients on Mitochondrial Dysfunction during a Cytokine Storm Typical of Oxidative/Inflammatory Diseases. Diseases 2021, 9, 30. [Google Scholar] [CrossRef] [PubMed]
- Kopustinskiene, D.M.; Bernatoniene, J. Molecular Mechanisms of Melatonin-Mediated Cell Protection and Signaling in Health and Disease. Pharmaceutics 2021, 13, 129. [Google Scholar] [CrossRef]
- Jou, M.J. Melatonin preserves the transient mitochondrial permeability transition for protection during mitochondrial Ca2+ stress in astrocyte. J. Pineal Res. 2011, 50, 427–435. [Google Scholar] [CrossRef]
- Fang, Y.; Zhao, C.; Xiang, H.; Jia, G.X.; Zhong, R. Melatonin improves cryopreservation of ram sperm by inhibiting mitochondrial permeability transition pore opening. Reprod. Domest. Anim. 2020, 55, 1240–1249. [Google Scholar] [CrossRef] [PubMed]
- Bejarano, I.; Redondo, P.C.; Espino, J.; Rosado, J.A.; Paredes, S.D.; Barriga, C.; Reiter, R.J.; Pariente, J.A.; Rodriquez, A.B. Melatonin induces mitochondrial-mediated apoptosis in human myeloid hl-60 cells. J. Pineal Res. 2009, 46, 392–400. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Sharma, R.; de Campos Zuccari, D.A.P.; de Almeida Chuffa, L.G.; Manucha, W.; Rodriguez, C. Melatonin synthesis in and uptake by mitochondria: Implications for diseased cells with dysfunctional mitochondria. Future Med. Chem. 2021, 13, 335–339. [Google Scholar] [CrossRef]
- Galano, A.; Tan, D.X.; Reiter, R.J. Melatonin: A versatile protector against oxidative DNA damage. Molecules 2018, 23, 530. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Janetovic, Z.; Jarret, S.G.; Lee, E.F.; Duprey, C.; Reiter, R.J.; Slominski, A.T. Melatonin and its metabolites protect human melanocytes against UVB-induced damage. Involvement of NRF2-mediated pathways. Sci. Rep. 2017, 7, 1274. [Google Scholar] [CrossRef] [Green Version]
- Fischer, T.W.; Kleszczynski, K.; Hardkop, L.H.; Kruse, N.; Zillikens, D. Melatonin enhances antioxidative enzyme gene expression (CAT, GPx, SOD), prevents their UVR-induced depletion, and protects against the formation of DNA damage (8-hydroxy-20-deoxyguanosine) in ex vivo human skin. J. Pineal Res. 2013, 54, 303–312. [Google Scholar] [CrossRef]
- Santofimia-Castano, P.; Clea Ruy, D.; Garcia-Sanchez, L.; Jimenez-Blasco, D.; Fernander-Bezmejo, M.; Bolanos, J.P.; Salido, G.M.; Gonzales, A. Melatonin induces the expression Nrf2-regulated antioxidant enzymes via PKC and Ca2+ influx activation in mouse isolated acinar cells. Free Radic. Biol. Med. 2015, 87, 226–236. [Google Scholar] [CrossRef] [Green Version]
- Sun, T.C.; Liu, X.C.; Yang, S.A.; Song, L.L.; Zhu, S.J.; Deng, S.L.; Tiang, L.; Chen, L.Y. Melatonin inhibits oxidative stress and apoptosis in cryopreserved ovarian tissue via Nrf2/HO-1 signaling pathway. Front. Mol. Biosci. 2020, 7, 163. [Google Scholar] [CrossRef]
- Kleszczynski, K.; Zillikens, D.; Fischer, T.W. Melatonin enhances mitochondrial ATP synthesis, reduces reactive oxygen species formation, and mediates translocation of the nuclear erythroid 2-related factor 2 resulting in activation of phase-2 antioxidant enzymes (GCS, HO-1, NQO1) in ultraviolet radiation-treated normal human epidermal keratinocytes (NHEK). J. Pineal Res. 2016, 61, 187–197. [Google Scholar]
- Bekyarova, G.; Tzaneva, M.; Hristova, M. Melatonin protects against burn-induced hepatic oxidative injury by inducing HO-1 via the Nrf2 pathway. Vet. Med. (Praha) 2015, 60, 621–628. [Google Scholar] [CrossRef] [Green Version]
- Meena, R.; Kumari, K.; Kumar, J.; Rajamani, P.; Verma, H.N.; Kesari, K.K. Therapeutic approaches of melatonin in microwave radiations-induced oxidative stress-mediated toxicity on male fertility pattern of wistar rats. Electromagn. Biol. Med. 2014, 33, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Sokolovic, D.; Djordjevic, B.; Kocic, G.; Stoimenov, T.J.; Stanojkovic, Z.; Sokolovic, D.M.; Veljkovic, A.; Ristic, G.; Despotovic, M.; Milisavljevic, D.; et al. The effects of melatonin on oxidative stress parameters and DNA fragmentation in testicular tissue of rats exposed to microwave radiation. Adv. Clin. Exp. Med. 2015, 24, 429–436. [Google Scholar] [CrossRef] [Green Version]
- Rezapoor, S.; Shirazi, A.; Abbasi, S.; Bazzaz, J.; Izadi, P.; Rezaeejam, H.; Valizadeh, M.; Soleimani-Mohammadi, F.; Najafi, M. Modulation of radiation-induced base excision repair pathway gene expression by melatonin. J. Med. Phys. 2017, 42, 245–250. [Google Scholar] [PubMed]
- Valizadeh, M.; Shirazi, A.; Izadi, P.; Tavakkoly Bazzaz, J.; Rezaeejam, H. Expression levels of two dna repair-related genes under 8 gy ionizing radiation and 100 mg/kg melatonin delivery in rat peripheral blood. J. Biomed. Phys. Eng. 2017, 7, 27–36. [Google Scholar]
- Mannino, G.; Caradonna, F.; Cruciata, I.; Lauria, A.; Perrone, A.; Gentile, C. Melatonin reduces inflammatory response in human intestinal epithelial cells stimulated by interleukin-1β. J. Pineal. Res. 2019, 67, e12598. [Google Scholar] [CrossRef]
- Wang, Y.; Zhao, T.T.; Zhao, H.Y.; Wang, H. Melatonin protects methotrexate-induced testicular injury in rats. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 7517–7525. [Google Scholar]
- Shin, I.S.; Park, J.W.; Shin, N.R.; Jeon, C.M.; Kwon, O.K.; Ki, J.S.; Kim, J.C.; Oh, S.R.; Ahn, K.S. Melatonin reduces airway inflammation in ovalbumin-induced asthma. Immunobiology 2014, 219, 901–908. [Google Scholar] [CrossRef]
- Favero, G.; Franceschetti, L.; Bonomi, F.; Rodella, L.R.; Rezzani, R. Melatonin as an anti-inflammatory agent modulating inflammasome activation. Int. J. Endocrinol. 2017, 2017, 1835195. [Google Scholar] [CrossRef] [Green Version]
- Haderland, R. Aging, melatonin and the pro- and anti-inflammatory networks. Int. J. Mol. Sci. 2019, 20, 1223. [Google Scholar]
- Bonomi, F.; Dos Santos, M.; Veronese, F.V.; Rezzani, R. NLRP3 inflammasome modulation by melatonin supplementation in chronic pristane-induced lupus nephritis. Int. J. Mol. Sci. 2019, 20, 3466. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ashrafizadeh, M.; Najafi, M.; Kavyani, M.; Mohammadinejad, R.; Farkhondeh, T.; Samarghandian, S. Anti-inflammatory activity of melatonin: A focus on the role of NLRP3 inflammasome. Inflammation 2021, 44, 1207–1222. [Google Scholar] [CrossRef] [PubMed]
- Carrillo-Vico, A.; Lardone, P.J.; Alvarez-Sanchez, N.; Rodriguez-Rodriguez, A.; Guerrero, J.M. Melatonin. Buffering the immune system. Int. J. Mol. Sci. 2013, 14, 8638–8683. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuklina, E.M.; Glebezdina, N.S.; Nekrasova, I.V. Role of melatonin in the regulation of differentiation of T cells producing interleukin 17 (IL-17). Bull. Exp. Biol. Med. 2016, 160, 656–658. [Google Scholar] [CrossRef]
- PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov (accessed on 8 June 2021).
- Schwarz, W.; Birau, N.; Hornstein, O.P.; Heubeck, B.; Schonberger, A.; Meyer, C.; Gottschalk, J. Alterations of melatonin secretion in atopic dermatitis. Acta Derm. Venerol. 1988, 68, 224–229. [Google Scholar]
- Kimata, H. Laughter elevates the levels of breast-milk melatonin. J. Psychosom. Res. 2007, 62, 699–702. [Google Scholar] [CrossRef]
- Kimata, H. Elevation of salivary melatonin levels by viewing a humorous film in patients with atopic eczema. Horm. Metab. Res. 2007, 39, 310–311. [Google Scholar] [CrossRef]
- Munoz-Hoyos, A.; Espin-Quirantes, C.; Molina-Carballo, A.; Uberos, J.; Contreraz-Chova, F. Neuroendocrine and circadian aspects (meatonin and beta-endorphin) of atopic dermatitis in the child. Pediatr. Allergy Immunol. 2007, 18, 679–686. [Google Scholar] [CrossRef]
- Chang, Y.S.; Chou, Y.T.; Lee, J.H.; Lee, P.L.; Dai, T.S.; Sun, C.; Lin, Y.T.; Wang, L.C.; Yu, H.H.; Yang, Y.H.; et al. Atopic dermatitis, melatonin and sleep disturbances. Pediatrics 2014, 134, e397–e405. [Google Scholar] [CrossRef] [Green Version]
- Uysal, P.; Avcil, S.; Abas, B.I.; Yenisey, C. Evaluation of oxidant-antioxidant balance in children with atopic dermatitis: A case-control study. Am. J. Clin. Dermatol. 2016, 1795, 527–537. [Google Scholar] [CrossRef] [PubMed]
- Devadasan, S.; Sarkar, R.; Barman, A.D.; Kaushik, S. Role of serum melatonin and oxidative stress in childchood atopic dermatitis: A prospective study. Indian. Dermatol. Online J. 2020, 11, 925–929. [Google Scholar] [PubMed]
- Chang, Y.S.; Lin, M.H.; Lee, J.H.; Lee, P.L.; Dai, Y.S.; Chu, K.H.; Sun, C.; Lin, Y.T.; Wang, L.C.; Yu, H.H.; et al. Melatonin supplementation for children with atopic dermatitis and sleep disturbance: A randomized clinical trial. JAMA Pediatr. 2016, 170, 35–42. [Google Scholar] [CrossRef] [Green Version]
- Ardakani, A.T.; Farrehi, M.; Sharif, M.R.; Ostadmohammadi, V.; Mirhosseini, N.; Kheirkhah, D.; Moosavi, S.G.A.; Behnejad, M.; Reter, R.J.; Asemi, Z. The effects of melatonin administration on disease severity and sleep quality in children with atopic dermatitis. A randomized, double-blinded, placebo-controlled trial. Pediatr. Allergy Immunol. 2018, 29, 834–840. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.-H.; Jung, J.-A.; Kim, G.-D.; Jang, A.-H.; Ahn, H.-J.; Park, Y.S.; Park, C.-S. Melatonin inhibits the development of 2,4-dinitrofluorobenzene-induced atopic dermatitis-like skin lesions in NC/Nga mice. J. Pineal Res. 2009, 47, 324–329. [Google Scholar] [CrossRef]
- Park, G.; Lee, S.H.; Oh, D.S.; Kim, Y.U. Melatonin inhibits neuronal dysfunction-associated with neuroinflammation by atopic psychological stress in an NC/Nga atopic-like mouse model. J. Pineal Res. 2017, 63. [Google Scholar] [CrossRef]
- Park, G.; Jung, Y.S.; Park, M.K.; Yang, Y.H.; Kim, Y.U. Melatonin inhibits attention-deficit/hyperactivity disorder caused by atopic dermatitis–induce psychological stress in an NC/Nga atopic-like mouse model. Sci. Rep. 2018, 8, 14981. [Google Scholar] [CrossRef] [Green Version]
- Crowley, S.J.; Acebo, C.; Carskadon, M.A. Human puberty, salivary melatonin profiles in constant conditions. Dev. Psychobiol. 2012, 54, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Esposti, D.; Lissoni, P.; Mauri, R.; Rovelli, F.; Orsenigo, L.; Pescia, S.; Vegetti, G.; Esposti, G.; Fraschini, F. The pineal gland-opioid system relation: Melatonin-naloxone interactions in regulating GH and LH releases in man. J. Endocrinol. Invest. 1988, 11, 103–106. [Google Scholar] [CrossRef]
- Bertino, L.; Guarneri, F.; Cannavò, S.P.; Casciaro, M.; Pioggia, G.; Gangemi, S. Oxidative Stress and Atopic Dermatitis. Antioxidants 2020, 9, 196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gurer-Orhan, H.; Suzen, S. Melatonin, its metabolites and its synthetic analogs as multi-faceted compounds: Antioxidant, prooxidant and inhibitor of bioactivation reactions. Curr. Med. Chem. 2015, 22, 490–499. [Google Scholar] [CrossRef] [PubMed]
- Suzen, S.; Cihaner, S.S.; Coban, T. Synthesis and comparison of antioxidant properties of indole-basedmelatonin analogue indole amino acid derivatives. Chem. Biol. Drug Des. 2012, 79, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Jaworek, J.; Szklarczyk, J.; Bonior, J.; Kot, M.; Pierzchalski, P.; Góralska, M.; Reiter, R.J.; Czech, U.; Tomaszewska, R. Melatonin metabolite N(1)-acetyl-n(1)-formyl-5-methoxykynuramine (AFMK) attenuates acute pancreatitis in the rat in vivo and in vitro. J. Physiol. Pharmacol. 2016, 67, 311–321. [Google Scholar]
- Jaworek, J.; Leja-Szpak, A.; Nawrot-Porabka, K.; Szklarczyk, J.; Kot, M.; Pierzchalski, P.; Góralska, M.; Ceranowicz, P.; Warzecha, Z.; Dembinski, A.; et al. Effects of melatonin and its analogues on pancreatic inflammation, enzyme secretion and tumorigenesis. Int. J. Mol. Sci. 2017, 18, 1014. [Google Scholar] [CrossRef]
| Author and Reference | Year | Patients Enrolled (N) | Treatment | Parameters Tested | Tissue Samples | MT Measurement | Method | Results |
|---|---|---|---|---|---|---|---|---|
| Schwarz et al. [89] | 1988 | adults with AD (18) | n/a | MT | blood | endogenous MT measured every 2 h for 24 h | not given | low serum MT in 6 patients and abolished circadian rhythm of its release; decreased nocturnal MT peak, in additional 8 AD patients; only 4 patients showed a normal pattern of MT secretion |
| Kimata [90] | 2007 | women with AD, mild SCORAD (48) | laughter, viewing a humorous film | MT, IgE | breast milk, serum | endogenous MT measured at 11.00 p.m., at 2.00 a.m., at 0.50 a.m. | ELISA (MT) | low basal MT in milk of AD patients, however stimulated MT in milk was similar to healthy control; decreased nocturnal MT peak in these patients; higher IgE in AD mothers |
| Kimata [91] | 2007 | children with AD, moderate SCORAD (24) | laughter, viewing a humorous film | MT, sleep, SCORAD | saliva | endogenous MT measured every 2 h from 10.0 p.m., to 6.00 a.m. | ELISA (MT), ELISA (INF-γ) sleep question-naire | low basal and stimulated MT in AD patients; decreased nocturnal MT peak; increased INF-γ; better sleep correlated with higher MT |
| Munoz-Hoyoz et al. [92] | 2007 | children with AD (40) -severe (20) -asymptomatic (20) | n/a | MT, β-endorphin | blood | endogenous MT measured at 9.00 a.m. and at 9.00 p.m. | RIA (MT) | low MT at 9.00 p.m.; nocturnal MT at 9.00 p.m. slightly, but non significantly lower than control; MT in asymptomatic group similar to control; β-endorphin decreased |
| Chang et al. [93] | 2014 | children and adolescents with AD, moderate SCORAD (72) | n/a | MT, sleep, IgE, pruritus, scratching movement | urine | endogenous MT measured as 6-sulfatoxymelatonin (in morning urine samples) | ELISA (MT), actigraphy, polysomnography, sleep questionnaire | nocturnal MT higher in AD patients than in controls; lower MT correlated with poor sleep efficiency, longer onset sleep latency, decreased NREM sleep, more sleep fragmentation; IgE in serum increased |
| Uysal et al. [94] | 2016 | children with AD (73) -mild (12) -moderate (22) -severe (39) | n/a | MT, NO, malondialdehyde (MDA), SCORAD | blood | endogenous MT, measured at 9.00 a.m. | ELISA (MT), ELISA (MDA), reduction method (NO) | increased MT in AD patients, comparing to control; MT lower in severe AD, than in mild; NO decreased in all AD patient; MDA similar to control |
| Devadasan et al. [95] | 2020 | children with AD (30) -mild, -moderate -severe | n/a | MT, SCORAD, MDA, SOD, GPx | blood | endogenous MT measured at 9.00 a.m. | ELISA (MT), RANDOX (SOD, GPx), TBA-TCA-HCl (MDA) | increased MT and MDA comparing to control; SOD, GPx increased, but insignificantly; none of parmeters correlated with SCORAD |
| Author and Reference | Year | Patients Enrolled (N) | Experimental Treatment | Parameters Tested | Tissue Samples | Method | Results |
|---|---|---|---|---|---|---|---|
| Chang et al. [96] | 2016 | children and adolescent with AD, moderate SCORAD (38) | exogenous MT given 3 mg/day for 4 weeks before bedtime | MT, sleep, IgE, SCORAD | urine | ELISA (MT), actigraphy | MT application reduced SCORAD, decreased sleep onset latency; improvement of sleep was not correlated with SCORAD; serum IgE was similar to control |
| Ardakani et al. [97] | 2018 | children with AD, moderate and severe SCORAD (35) | exogenous MT given 6 mg/day for 6 weeks before bedtime | SCORAD, sleep, CRP, IgE | blood | SCORAD, sleep questionnaire | MT application reduced SCORAD, and IgE, improved total sleep scores; CRP and pruritus were similar to control |
| Author and Reference | Year | Experimental Models | Experimental Design | Material and Method | Parameters Tested | Results |
|---|---|---|---|---|---|---|
| Kim et al. [98] | 2009 | NC/Nga mice | sensitization with 2,3-dinitrofluorobenzene (DNFB) followed by repeated application of DNFB on the skin + MT 10, or 20 mg/day i.p. for 2 weeks | skin, blood, CD4+ cells isolated from animals, histologic analysis, ELISA kits for interleukins and IgE | skin lesions, IgE, IL-4, INF-γ | MT treatment reduced skin lesions, such as hypertrophy, hyperkeratosis and inflammatory cell infiltration, as well as serum IgE; MT treatment inhibited production of IL-4 and INF-γ by activated CD4+ cells |
| Park et al. [99] | 2017 | NC/Nga mice, mouse HT 22 hippocampal cell culture, rat brain primary hypothalamic neuronal cells (RPHN) | sensitization with 2,3-dinitrochlorobenzene (DNCB) followed by repeated application of DNCB on the skin + MT 20 mg/day orally with or without cortisone 20 mg/day orally for 6 weeks; incubation of cells with various concentrations of cortisol | skin, blood, brain samples, cell cultures, PCR, Western blot, immunochemistry, immunocyto-chemistry, immuno-fluorescence, comercial kits | skin lesions; scratching behavior; neuroinflammation and neuronal cells viability; IgE; MT1R, CRH, POMC, ACTH; COX2, iNOS; TNFα, IL-4, IL-1α | MT treatment reduced skin lesions, scratching behavior and serum IgE; MT treatment reversed atopic stress-induced neuronal damage; increased reduced by stress MT and MT1R in the brain and skin and supressed neuroinflammation |
| Park et al. [100] | 2018 | NC/Nga mice, SH-SY5Y human neuronal cell culture | DNCB model + MT as above, incubation of cells with various concentrations of cortisol or MT | brain tissues, human neuronal cell culture, PCR, Western blot, immunohisto-chemistry, commercial kits | MT, CRH, CRHR 1, ACTH, norepinephrine, dopamine, dopamine β-hydroxylase, tyrosine hydroxylase | MT reversed induced by AD stress-increase in norepinephrine and dopamine, and hyperactivity of hypothalamic-hypophyseal-adrenal (HPA) axis, MT corrected dysregulated dopamine and noradrenline system, which is related to attention-deficit/hyperactivity disorder (ADHD) caused by atopic mouse model |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jaworek, A.K.; Szepietowski, J.C.; Hałubiec, P.; Wojas-Pelc, A.; Jaworek, J. Melatonin as an Antioxidant and Immunomodulator in Atopic Dermatitis—A New Look on an Old Story: A Review. Antioxidants 2021, 10, 1179. https://doi.org/10.3390/antiox10081179
Jaworek AK, Szepietowski JC, Hałubiec P, Wojas-Pelc A, Jaworek J. Melatonin as an Antioxidant and Immunomodulator in Atopic Dermatitis—A New Look on an Old Story: A Review. Antioxidants. 2021; 10(8):1179. https://doi.org/10.3390/antiox10081179
Chicago/Turabian StyleJaworek, Andrzej Kazimierz, Jacek Cezary Szepietowski, Przemysław Hałubiec, Anna Wojas-Pelc, and Jolanta Jaworek. 2021. "Melatonin as an Antioxidant and Immunomodulator in Atopic Dermatitis—A New Look on an Old Story: A Review" Antioxidants 10, no. 8: 1179. https://doi.org/10.3390/antiox10081179
APA StyleJaworek, A. K., Szepietowski, J. C., Hałubiec, P., Wojas-Pelc, A., & Jaworek, J. (2021). Melatonin as an Antioxidant and Immunomodulator in Atopic Dermatitis—A New Look on an Old Story: A Review. Antioxidants, 10(8), 1179. https://doi.org/10.3390/antiox10081179








