A Single Dose of Marine Chlorella vulgaris Increases Plasma Concentrations of Lutein, β-Carotene and Zeaxanthin in Healthy Male Volunteers
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Standards
2.2. Participants
2.3. Test Product
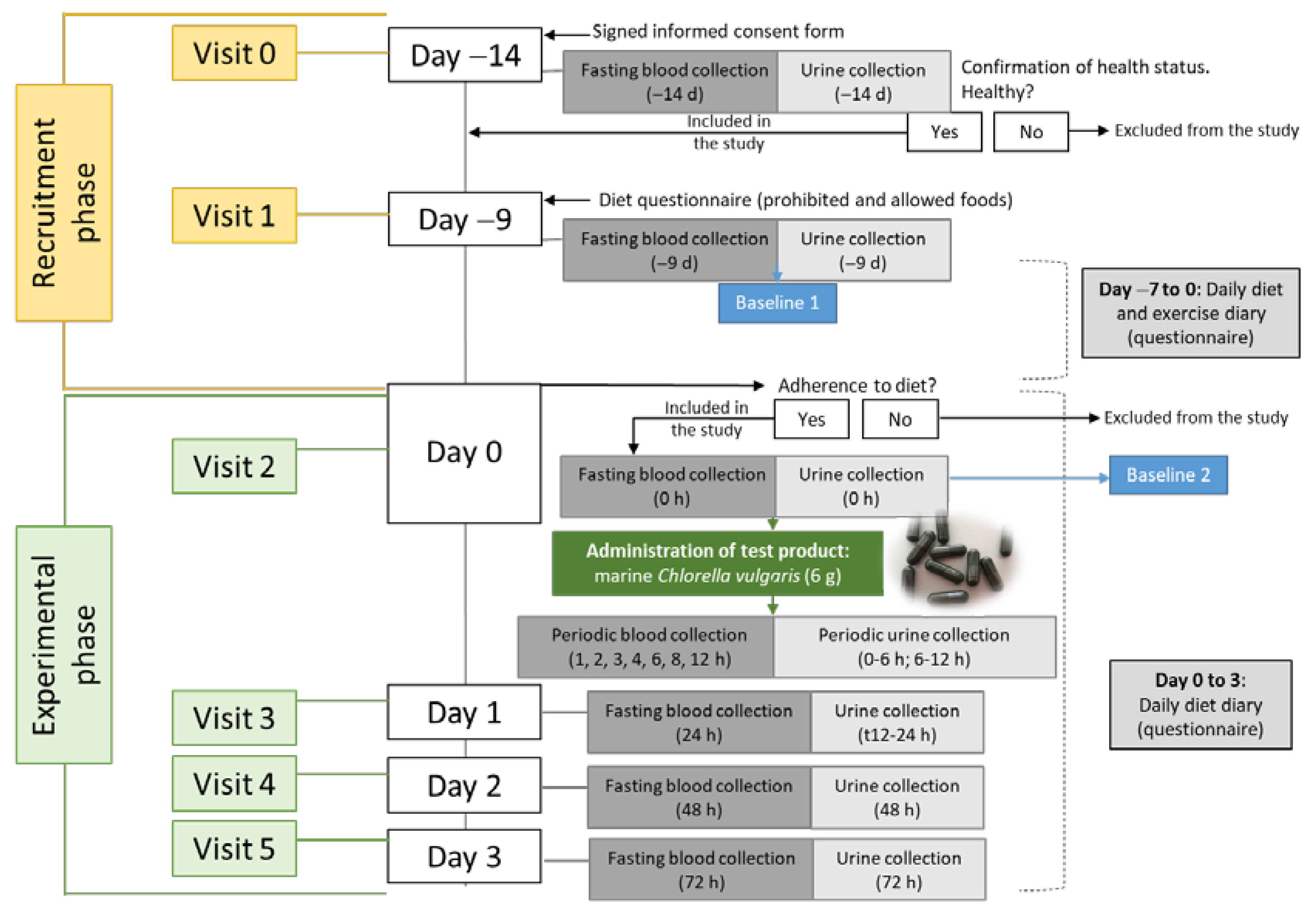
2.4. Protocol
2.5. Analysis of Biological Samples
2.5.1. Carotenoids Analysis in Blood Samples
2.5.2. PUFA Analysis in Blood Samples
2.5.3. Se and I Analysis in Blood and Urine
2.6. Statistical Analysis
3. Results and Discussion
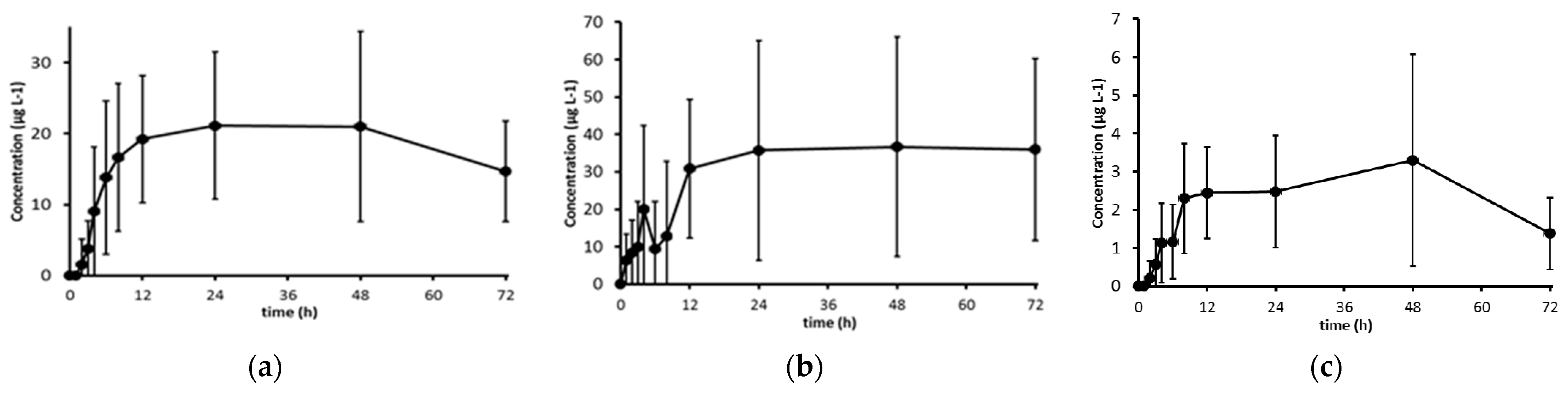
3.1. Bioavailability of Carotenoids
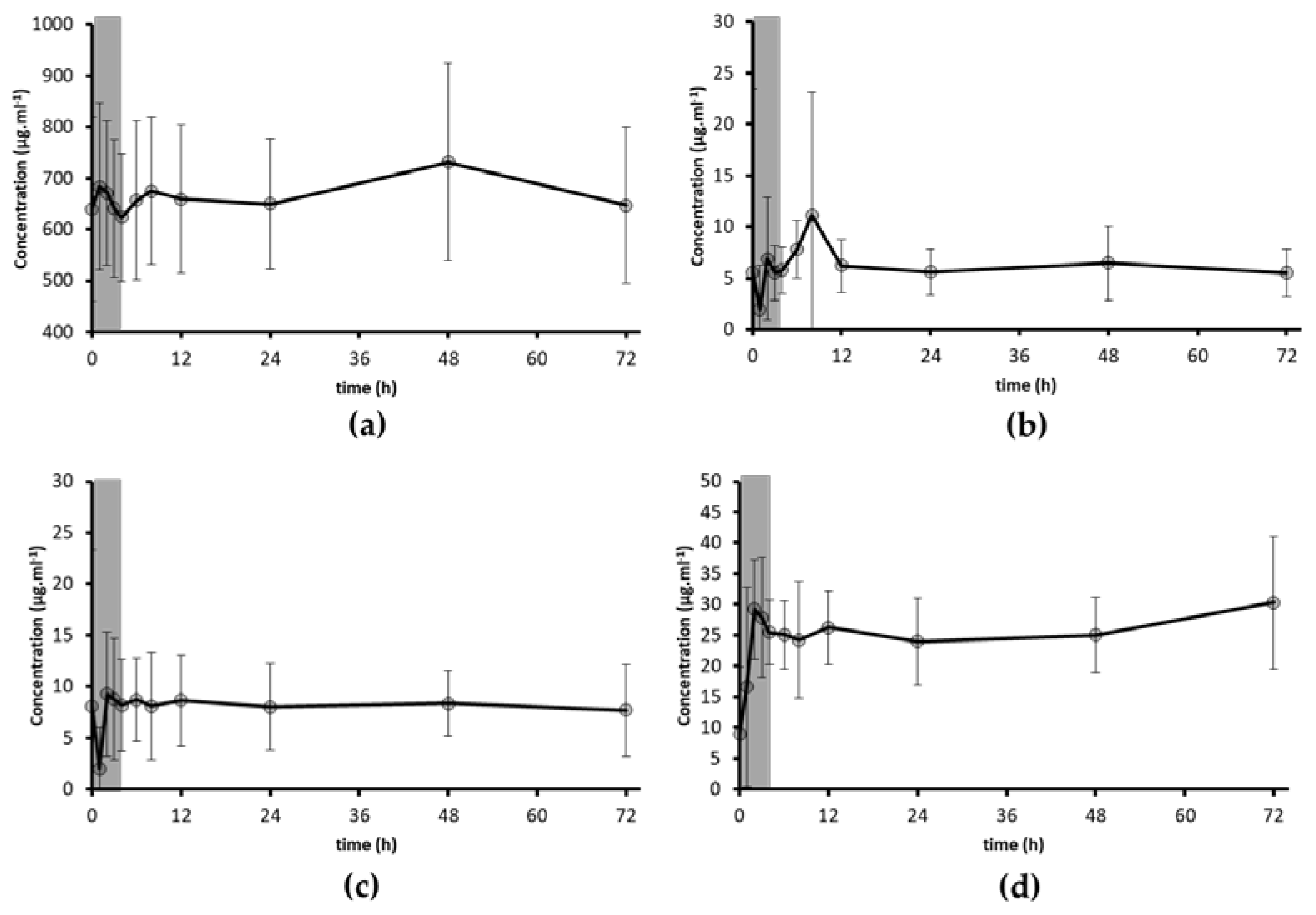
3.2. Bioavailability of PUFA
3.3. Bioavailability of Trace Elements: Se and I
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wells, M.L.; Potin, P.; Craigie, J.S.; Raven, J.A.; Merchant, S.S.; Helliwell, K.E.; Smith, A.G.; Camire, M.E.; Brawley, S.H. Algae as nutritional and functional food sources: Revisiting our understanding. J. Appl. Phycol. 2017, 29, 949–982. [Google Scholar] [CrossRef] [PubMed]
- Safi, C.; Zebib, B.; Merah, O.; Pontalier, P.; Vaca-Carcia, C. Morphology, composition, production, processing and applications of Chlorella vulgaris: A review. Renew. Sustain. Energy Rev. 2014, 35, 265–278. [Google Scholar] [CrossRef]
- Kim, S.; Kim, J.; Lim, Y.; Kim, Y.J.; Kim, J.Y.; Kwon, O. A dietary cholesterol challenge study to assess Chlorella supplementation in maintaining healthy lipid levels in adults: A double-blinded, randomized, placebo-controlled study. Nutr. J. 2015, 15, 1–7. [Google Scholar] [CrossRef]
- Ryu, N.H.; Lim, Y.; Park, J.E.; Kim, J.; Kim, J.Y.; Kwon, S.W.; Kwon, O. Impact of daily Chlorella consumption on serum lipid and carotenoid profiles in mildly hypercholesterolemic adults: A double-blinded, randomized, placebo-controlled study. Nutr. J. 2014, 13, 1–8. [Google Scholar] [CrossRef]
- Panahi, Y.; Badeli, R.; Karami, G.; Badeli, Z.; Sahebkar, A. A randomized controlled trial of 6-week Chlorella vulgaris supplementation in patients with major depressive disorder. Complement. Ther. Med. 2015, 23, 598–602. [Google Scholar] [CrossRef] [PubMed]
- Otsuki, T.; Shimizu, K.; Iemitsu, M.; Kono, I. Salivary secretory immunoglobulin a secretion increases after 4-weeks ingestion of chlorella-derived multicomponent supplement in humans: A randomized cross over study. Nutr. J. 2011, 10, 91. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Haeng, S.J.; Phan, M.G.; Jin, X.; Lee, S.; Phan, T.S.; Lee, D.; Hong, Y.S.; Lee, K.; Jung, J.L. Blockade of nuclear factor-κB signaling pathway and anti-inflammatory activity of cardamomin, a chalcone analog from Alpinia conchigera. J. Pharmacol. Exp. Ther. 2006, 316, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Fallah, A.A.; Sarmast, E.; Dehkordi, S.H.; Engardeh, J.; Mahmoodnia, L.; Khaledifar, A.; Jafari, T. Effect of Chlorella supplementation on cardiovascular risk factors: A meta-analysis of randomized controlled trials. Clin. Nutr. 2018, 37, 1892–1901. [Google Scholar] [CrossRef]
- Bito, T.; Okumura, E.; Fujishima, M.; Watanabe, F. Potential of chlorella as a dietary supplement to promote human health. Nutrients 2020, 12, 2524. [Google Scholar] [CrossRef]
- Miyazawa, T.; Nakagawa, K.; Kimura, F.; Nakashima, Y.; Maruyama, I.; Higuchi, O.; Miyazawa, T. Chlorella is an effective dietary source of lutein for human erythrocytes. J. Oleo Sci. 2013, 62, 773–779. [Google Scholar] [CrossRef]
- Chung, H.Y.; Rasmussen, H.M.; Johnson, E.J. Lutein bioavailability is higher from lutein-enriched eggs than from supplements and spinach in men. J. Nutr. 2004, 134, 1887–1893. [Google Scholar] [CrossRef] [PubMed]
- Desmarchelier, C.; Borel, P. Overview of carotenoid bioavailability determinants: From dietary factors to host genetic variations. Trends Food Sci. Technol. 2017, 69, 270–280. [Google Scholar] [CrossRef]
- Bohn, T.; Desmarchelier, C.; Dragsted, L.O.; Nielsen, C.S.; Stahl, W.; Rühl, R.; Keyer, J.; Borel, P. Host-related factors explaining interindividual variability of carotenoid bioavailability and tissue concentrations in humans. Mol. Nutr. Food Res. 2017, 61, 1–37. [Google Scholar] [CrossRef] [PubMed]
- Forman, M.R.; Johnson, E.J.; Lanza, E.; Graubard, B.I.; Beecher, G.R.; Muesing, R. Effect of menstrual cycle phase on the concentration of individual carotenoids in lipoproteins of premenopausal women: A controlled dietary study. Am. J. Clin. Nutr. 1998, 67, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Mumford, S.L.; Browne, R.W.; Schliep, K.C.; Schmelzer, J.; Plowden, T.C.; Michels, K.A.; Sjaarda, L.A.; Zarek, S.M.; Perkins, N.J.; Messer, L.C.; et al. Serum antioxidants are associated with serum reproductive hormones and ovulation among healthy women. J. Nutr. 2016, 146, 98–106. [Google Scholar] [CrossRef]
- Gama, J.J.T.; Sylos, C.M. Major carotenoid composition of Brazilian Valencia orange juice: Identification and quantification by HPLC. Food Res. Int. 2005, 38, 899–903. [Google Scholar] [CrossRef]
- Ajana, S.; Weber, D.; Helmer, C.; Merle, B.M.; Stuetz, W.; Dartigues, J.F.; Rougier, M.B.; Korobelnik, J.F.; Grune, T.; Delcourt, C.; et al. Plasma concentrations of lutein and zeaxanthin, macular pigment optical density, and their associations with cognitive performances among older adults. Investig. Ophthalmol. Vis. Sci. 2018, 59, 1828–1835. [Google Scholar] [CrossRef]
- Stuetz, W.; Weber, D.; Dollé, M.E.T.; Jansen, E.; Grubeck-Loebenstein, B.; Fiegl, S.; Toussaint, O.; Bernhardt, J.; Gonos, E.S.; Franceschi, C.; et al. Plasma carotenoids, tocopherols, and retinol in the age-stratified (35–74 years) general population: A cross-sectional study in six European countries. Nutrients 2016, 8, 614. [Google Scholar] [CrossRef] [PubMed]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef]
- ISO 5509:2000 Animal and Vegetable Fats and Oils, Preparation of Methyl Esters of Fatty Acids. Available online: http://www.iso.org/iso/home/store/catalogue_ics/catalogue_detail_ics.htm?csnumber=11560 (accessed on 15 May 2021).
- Rodrigues, L.A.; Pereira, C.V.; Partidario, A.M.C.; Gouveia, L.F.; Simoes, P.; Paiva, A.; Matias, A.A. Supercritical CO2 extraction of bioactive lipids from canned sardine waste streams. J. CO2 Util. 2021, 43, 101359. [Google Scholar] [CrossRef]
- Chow, S.-C.; Liu, J. Design and Analysis of Bioavailability and Bioequivalence Studies, 3rd ed.; Chapman and Hall/CRC: Boca Raton, FL, USA, 2008. [Google Scholar]
- Patterson, S.D.; Jones, B. Bioequivalence and Statistics in Clinical Pharmacology, 2nd ed.; Chapman and Hall/CRC: Boca Raton, FL, USA, 2016. [Google Scholar]
- Shibata, S.; Hayakawa, K. Bioavailability of lutein in Chlorella powder: A single ingestion of chlorella powder raises serum lutein concentrations in healthy human volunteers. Food Sci. Technol. Res. 2009, 15, 449–452. [Google Scholar] [CrossRef][Green Version]
- Lakshminarayana, R.; Baskaran, V. Influence of olive oil on the bioavailability of carotenoids. Eur. J. Lipid Sci. Technol. 2013, 115, 1085–1093. [Google Scholar] [CrossRef]
- Nidhi, B.; Mamatha, B.S.; Baskaran, V. Olive oil improves the intestinal absorption and bioavailability of lutein in lutein-deficient mice. Eur. J. Nutr. 2014, 53, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Riso, P.; Brusamolino, A.; Ciappellano, S. Comparison of lutein bioavailability from vegetables and supplement. Int. J. Vitam. Nutr. Res. 2003, 73, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Böhm, V.; Lietz, G.; Olmedilla-Alonso, B.; Phelan, D.; Reboul, E.; Bánati, D.; Borel, P.; Corte-Real, J.; de Lera, A.R.; Desmarchelier, C.; et al. From carotenoid intake to carotenoid blood and tissue concentrations—Implications for dietary intake recommendations. Nutr. Rev. 2021, 79, 544–573. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.-Y.; Ferreira, A.L.A.; Epstein, S.; Paiva, S.A.; Castaneda-Sceppa, C.; Johnson, E.J. Site-specific concentrations of carotenoids in adipose tissue: Relations with dietary and serum carotenoid concentrations in healthy adults. Am. J. Clin. Nutr. 2009, 90, 533–539. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.Y.; Ok, H.M.; Park, M.Y.; Kim, J.Y.; Kwon, O. Bioavailability of carotenoids from chlorella powder in healthy subjects: A comparison with marigold petal extract. J. Funct. Foods 2016, 21, 27–35. [Google Scholar] [CrossRef]
- van Het Hof, K.H.; West, C.E.; Weststrate, J.A.; Hautvast, J.G. Dietary factors that affect the bioavailability of carotenoids. J. Nutr. 2000, 130, 503–506. [Google Scholar] [CrossRef]
- Zdolsek, J.; Metander, A.; Hahn, R. Volume kinetic evaluation of fluid turnover after oral intake of tap water, lemonade and saline in volunteers. BMC Sports Sci. Med. Rehabil. 2016, 8, 1–7. [Google Scholar] [CrossRef]
- Sollanek, K.J.; Kenefick, R.W.; Cheuvront, S.N.; Axtell, R.S. Potential impact of a 500-mL water bolus and body mass on plasma osmolality dilution. Eur. J. Appl. Physiol. 2011, 111, 1999–2004. [Google Scholar] [CrossRef]
- Goyal, R.K.; Guo, Y.; Mashimo, H. Advances in the physiology of gastric emptying. Neurogastroenterol. Motil. 2019, 31, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Vestland, T.L.; Åsberg, A.; Klaveness, A.J.; Klaveness, J. Bioavailability of eicosapentaenoic acid and docosahexaenoic acid from omega-3 tablets. JSM Bioavailab. Bioequivalence 2017, 1001, 1–5. [Google Scholar]
- Cases, J.; Vacchina, V.; Napolitano, A.; Caporiccio, B.; Besançon, P.; Lobinski, R.; Rouanet, J.-M. Selenium from selenium-rich spirulina is less bioavailable than selenium from sodium selenite and selenomethionine in selenium-deficient rats 1. Nutr. Requir. 2001, 131, 2343–2350. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Dietetic Products. Scientific opinion on dietary reference values for iodine. EFSA J. 2014, 12, 1–57. [Google Scholar] [CrossRef]
- Leung, A.M.; Avram, A.M.; Brenner, A.V.; Duntas, L.H.; Ehrenkranz, J.; Hennessey, J.V.; Lee, S.L.; Pearce, E.N.; Roman, S.A.; Stagnaro-Green, A.; et al. Potential risks of excess iodine ingestion and exposure: Statement by the American Thyroid Association Public Health Committee. Thyroid 2015, 25, 145–146. [Google Scholar] [CrossRef]
- EFSA Panel on Dietetic Products. Nutrition and Alergies Summary of tolerable upper intake levels—Version 4. EFSA J. 2018. Available online: https://www.efsa.europa.eu/sites/default/files/assets/UL_Summary_tables.pdf (accessed on 15 May 2021).
| Study | Lutein (µg·L−1) | β-Carotene (µg·L−1) | Zeaxanthin (µg·L−1) |
|---|---|---|---|
| This study | |||
| Day −14 | 107.2 ± 51.2 (range: 61.9–205.8) | 199.0 ± 152.0 (range: 55.3–491.6) | 17.3 ± 6.4 (range: 11.5–30.2) |
| Day −9 | 93.2 ± 44.9 (range: 63.1–180.9) | 127.5 ± 64.9 (range: 34.1–401.5) | 15.5 ± 5.2 (range: 8.0–24.9) |
| Day 0 | 74.8 ± 24.2 (range: 56.9–119.2) | 104.6 ± 72.3 (range: 27.8–232.8) | 13.5 ± 4.6 (range: 7.2–22.1) |
| Riso et al., 2002 [27] | 386.7 ± 39.8 | 590.6 ± 64.4 | 39.8 ± 5.7 |
| Stuetz et al., 2016 [18] | 157.0 | 289.9 | 25.6 |
| Carotenoid | Content in 6 g of Chlorella Vulgaris (mg) | AUC 1 (µg·h·L−1) | p-Value | Cmax 1 (µg·L−1) | p-Value | tmax2 (h) |
|---|---|---|---|---|---|---|
| Lutein | 7.08 | 1001.6|141.5 * (235.3–4263.8)|(33.3–602.2) * | p < 0.01 | 20.4|2.9 * (4.8–86.4)|(0.7–12.2)* | p < 0.01 | 19.0 ± 15.7 |
| β- Carotene | 1.88 | 1302.4|692.8 * (338.3–5015.5)|(179.9–2667.8) * | p < 0.01 | 34.9|18.6 * (6.9–178.1)|(3.7–94.7)* | p < 0.01 | 34.9 ± 26.4 |
| Zeaxanthin | 1.47 | 122.2|83.1 * (25.5–585.8)|(17.3–56.5) * | p < 0.01 | 3.4|2.3 * (0.8–14.5)|(0.5–9.9)* | p < 0.01 | 22.5 ± 17.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Serra, A.T.; Silva, S.D.; Pleno de Gouveia, L.; Alexandre, A.M.R.C.; Pereira, C.V.; Pereira, A.B.; Partidário, A.C.; Silva, N.E.; Bohn, T.; Gonçalves, V.S.S.; et al. A Single Dose of Marine Chlorella vulgaris Increases Plasma Concentrations of Lutein, β-Carotene and Zeaxanthin in Healthy Male Volunteers. Antioxidants 2021, 10, 1164. https://doi.org/10.3390/antiox10081164
Serra AT, Silva SD, Pleno de Gouveia L, Alexandre AMRC, Pereira CV, Pereira AB, Partidário AC, Silva NE, Bohn T, Gonçalves VSS, et al. A Single Dose of Marine Chlorella vulgaris Increases Plasma Concentrations of Lutein, β-Carotene and Zeaxanthin in Healthy Male Volunteers. Antioxidants. 2021; 10(8):1164. https://doi.org/10.3390/antiox10081164
Chicago/Turabian StyleSerra, Ana Teresa, Sandra D. Silva, Luís Pleno de Gouveia, Agostinho M. R. C. Alexandre, Carolina V. Pereira, Ana Barbara Pereira, Ana Carvalho Partidário, Nuno Elvas Silva, Torsten Bohn, Vanessa S. S. Gonçalves, and et al. 2021. "A Single Dose of Marine Chlorella vulgaris Increases Plasma Concentrations of Lutein, β-Carotene and Zeaxanthin in Healthy Male Volunteers" Antioxidants 10, no. 8: 1164. https://doi.org/10.3390/antiox10081164
APA StyleSerra, A. T., Silva, S. D., Pleno de Gouveia, L., Alexandre, A. M. R. C., Pereira, C. V., Pereira, A. B., Partidário, A. C., Silva, N. E., Bohn, T., Gonçalves, V. S. S., Real, G., Escudero, P., Fernández, N., Matias, A. A., & Bronze, M. R. (2021). A Single Dose of Marine Chlorella vulgaris Increases Plasma Concentrations of Lutein, β-Carotene and Zeaxanthin in Healthy Male Volunteers. Antioxidants, 10(8), 1164. https://doi.org/10.3390/antiox10081164







