Antioxidant Therapy in Pancreatitis
Abstract
1. Introduction
2. Inflammation and Oxidative Stress in the Pathogenesis of Pancreatitis
2.1. Acute Pancreatitis
2.2. Chronic Pancreatitis
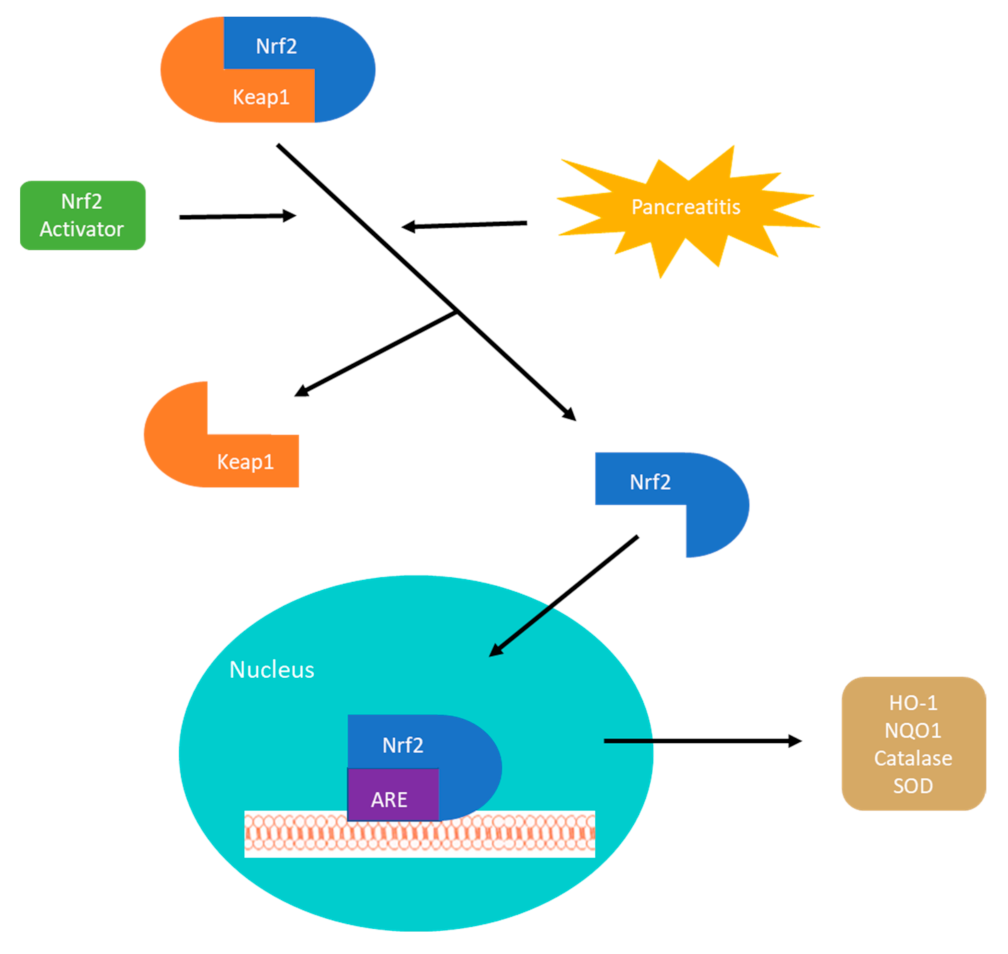
3. Nrf2-Keap1 Signaling Pathway
4. Roles of Antioxidant Therapy in Pancreatitis
4.1. Pharmacological Intervention
4.2. Pain Control
4.3. Surgical Approach
5. Discussion
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hoffmeister, A.; Mayerle, J.; Beglinger, C.; Buchler, M.W.; Bufler, P.; Dathe, K.; Fölsch, U.R.; Friess, H.; Izbicki, J.R.; Kahl, S.; et al. English language version of the S3-consensus guidelines on chronic pancreatitis: Definition, aetiology, diagnostic examinations, medical, endoscopic and surgical management of chronic pancreatitis. Z. Gastroenterol. 2015, 53, 1447–1495. [Google Scholar] [CrossRef]
- Xiao, A.Y.; Tan, M.L.Y.; Wu, L.M.; Asrani, V.M.; A Windsor, J.; Yadav, D.; Petrov, M.S. Global incidence and mortality of pancreatic diseases: A systematic review, meta-analysis, and meta-regression of population-based cohort studies. Lancet Gastroenterol. Hepatol. 2016, 1, 45–55. [Google Scholar] [CrossRef]
- Lankisch, P.G.; Apte, M.; Banks, P.A. Acute pancreatitis. Lancet 2015, 386, 85–96. [Google Scholar] [CrossRef]
- Banks, P.A.; Bollen, T.L.; Dervenis, C.; Gooszen, H.G.; Johnson, C.D.; Sarr, M.G.; Tsiotos, G.G.; Vege, S.S. Classification of acute pancreatitis—2012: Revision of the Atlanta classification and definitions by international consensus. Gut 2012, 62, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Buter, A.; Imrie, C.W.; Carter, C.R.; Evans, S.; McKay, C.J. Dynamic nature of early organ dysfunction determines outcome in acute pancreatitis. Br. J. Surg. 2002, 89, 298–302. [Google Scholar] [CrossRef] [PubMed]
- Frey, C.F.; Zhou, H.; Harvey, D.J.; White, R.H. The Incidence and Case-fatality Rates of Acute Biliary, Alcoholic, and Idiopathic Pancreatitis in California, 1994–2001. Pancreas 2006, 33, 336–344. [Google Scholar] [CrossRef] [PubMed]
- Yadav, D.; Timmons, L.; Benson, J.T.; A Dierkhising, R.; Chari, S.T. Incidence, Prevalence, and Survival of Chronic Pancreatitis: A Population-Based Study. Am. J. Gastroenterol. 2011, 106, 2192–2199. [Google Scholar] [CrossRef]
- Peery, A.F.; Crockett, S.D.; Barritt, A.S.; Dellon, E.S.; Eluri, S.; Gangarosa, L.M.; Jensen, E.T.; Lund, J.L.; Pasricha, S.; Runge, T.; et al. Burden of Gastrointestinal, Liver, and Pancreatic Diseases in the United States. Gastroenterology 2015, 149, 1731–1741.e3. [Google Scholar] [CrossRef]
- Testoni, P.A. Acute recurrent pancreatitis: Etiopathogenesis, diagnosis and treatment. World J. Gastroenterol. 2014, 20, 16891–16901. [Google Scholar] [CrossRef]
- Raimondi, S.; Lowenfels, A.B.; Morselli-Labate, A.M.; Maisonneuve, P.; Pezzilli, R. Pancreatic cancer in chronic pancreatitis; aetiology, incidence, and early detection. Best Pr. Res. Clin. Gastroenterol. 2010, 24, 349–358. [Google Scholar] [CrossRef]
- Ammann, R.W.; Akovbiantz, A.; Largiader, F.; Schueler, G. Course and outcome of chronic pancreatitis. Longitudinal study of a mixed medical-surgical series of 245 patients. Gastroenterology 1984, 86, 820–828. [Google Scholar]
- Bassi, C.; Falconi, M.; Butturini, G.; Pederzoli, P. Early complications of severe acute pancreatitis. In Surgical Treatment: Evidence-Based and Problem-Oriented; Holzheimer, R., Mannick, J., Eds.; Zuckschwerdt: Munich, Germany, 2001. [Google Scholar]
- Beyer, G.; Habtezion, A.; Werner, J.; Lerch, M.M.; Mayerle, J. Chronic pancreatitis. Lancet 2020, 396, 499–512. [Google Scholar] [CrossRef]
- Dunbar, E.; Greer, P.J.; Melhem, N.; Alkaade, S.; Amann, S.T.; Brand, R.; Coté, G.A.; Forsmark, C.E.; Gardner, T.B.; Gelrud, A.; et al. Constant-severe pain in chronic pancreatitis is associated with genetic loci for major depression in the NAPS2 cohort. J. Gastroenterol. 2020, 55, 1000–1009. [Google Scholar] [CrossRef]
- APA Acute Pancreatitis Guidelines. Working Group IAP/APA Acute Pancreatitis Guidelines IAP/APA evidence-based guidelines for the management of acute pancreatitis. Pancreatology 2013, 13, e1–e15. [Google Scholar] [CrossRef]
- Barreto, S.G.; Habtezion, A.; Gukovskaya, A.; Lugea, A.; Jeon, C.; Yadav, D.; Hegyi, P.; Venglovecz, V.; Sutton, R.; Pandol, S.J. Critical thresholds: Key to unlocking the door to the prevention and specific treatments for acute pancreatitis. Gut 2021, 70, 194–203. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Kudo, M.; Strober, W. Immunopathogenesis of pancreatitis. Mucosal Immunol. 2017, 10, 283–298. [Google Scholar] [CrossRef] [PubMed]
- Muller, W.A. Getting Leukocytes to the Site of Inflammation. Vet. Pathol. 2013, 50, 7–22. [Google Scholar] [CrossRef]
- Bukowczan, J.; Warzecha, Z.; Ceranowicz, P.; Kusnierz-Cabala, B.; Tomaszewska, R.; Dembinski, A. Therapeutic Effect of Ghrelin in the Course of Ischemia/Reperfusion-Induced Acute Pancreatitis. Curr. Pharm. Des. 2015, 21, 2284–2290. [Google Scholar] [CrossRef] [PubMed]
- Ceranowicz, D.; Warzecha, Z.; Dembinski, A.; Ceranowicz, P.; Cieszkowski, J.; Kusnierz-Cabala, B.; Tomaszewska, R.; Kuwahara, A.; Kato, I. Role of hormonal axis, growth hormone—IGF-1, in the therapeutic effect of ghrelin in the course of cerulein-induced acute pancreatitis. J. Physiol. Pharmacol. Off. J. Pol. Physiol. Soc. 2010, 61, 599–606. [Google Scholar]
- Dumnicka, P.; Maduzia, D.; Ceranowicz, P.; Olszanecki, R.; Drożdż, R.; Kuśnierz-Cabala, B. The Interplay between Inflammation, Coagulation and Endothelial Injury in the Early Phase of Acute Pancreatitis: Clinical Implications. Int. J. Mol. Sci. 2017, 18, 354. [Google Scholar] [CrossRef]
- Mole, D.J.; Olabi, B.; Robinson, V.; Garden, O.J.; Parks, R.W. Incidence of individual organ dysfunction in fatal acute pancreatitis: Analysis of 1024 death records. HPB 2009, 11, 166–170. [Google Scholar] [CrossRef] [PubMed]
- Esmon, C.T. Crosstalk between inflammation and thrombosis. Maturitas 2004, 47, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Finkel, T.; Holbrook, N.J. Oxidants, oxidative stress and the biology of ageing. Nature 2000, 408, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Booth, D.M.; Mukherjee, R.; Sutton, R.; Criddle, D.N. Calcium and Reactive Oxygen Species in Acute Pancreatitis: Friend or Foe? Antioxid. Redox Signal. 2011, 15, 2683–2698. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, J.A.; Cash, N.; Soares, P.M.G.; Souza, M.H.L.P.; Sutton, R.; Criddle, D.N. Oxidative stress in acute pancreatitis: Lost in translation? Free. Radic. Res. 2013, 47, 917–933. [Google Scholar] [CrossRef] [PubMed]
- Brookes, P.S.; Yoon, Y.; Robotham, J.L.; Anders, M.W.; Sheu, S.-S. Calcium, ATP, and ROS: A mitochondrial love-hate triangle. Am. J. Physiol. Physiol. 2004, 287, C817–C833. [Google Scholar] [CrossRef]
- Schrader, H.; Menge, B.A.; Schneider, S.; Belyaev, O.; Tannapfel, A.; Uhl, W.; Schmidt, W.E.; Meier, J.J. Reduced Pancreatic Volume and β-Cell Area in Patients with Chronic Pancreatitis. Gastroenterology 2009, 136, 513–522. [Google Scholar] [CrossRef]
- Bachem, M.G.; Schneider, E.; Gross, H.; Weidenbach, H.; Schmid, R.M.; Menke, A.; Siech, M.; Beger, H.; Grünert, A.; Adler, G. Identification, culture, and characterization of pancreatic stellate cells in rats and humans. Gastroenterology 1998, 115, 421–432. [Google Scholar] [CrossRef]
- Shimosegawa, T. A New Insight into Chronic Pancreatitis. Tohoku J. Exp. Med. 2019, 248, 225–238. [Google Scholar] [CrossRef]
- Lardon, J.; Rooman, I.; Bouwens, L. Nestin expression in pancreatic stellate cells and angiogenic endothelial cells. Histochem. Cell Biol. 2002, 117, 535–540. [Google Scholar] [CrossRef]
- Dodson, M.; De La Vega, M.R.; Cholanians, A.B.; Schmidlin, C.J.; Chapman, E.; Zhang, D.D. Modulating NRF2 in Disease: Timing Is Everything. Annu. Rev. Pharmacol. Toxicol. 2019, 59, 555–575. [Google Scholar] [CrossRef]
- Itoh, K.; Wakabayashi, N.; Katoh, Y.; Ishii, T.; Igarashi, K.; Engel, J.D.; Yamamoto, M. Keap1 represses nuclear activation of antioxidant responsive elements by Nrf2 through binding to the amino-terminal Neh2 domain. Genes Dev. 1999, 13, 76–86. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Khor, T.O.; Xu, C.; Shen, G.; Jeong, W.-S.; Yu, S.; Kong, A.-N. Activation of Nrf2-antioxidant signaling attenuates NFκB-inflammatory response and elicits apoptosis. Biochem. Pharmacol. 2008, 76, 1485–1489. [Google Scholar] [CrossRef] [PubMed]
- Van Gossum, A.; Closset, P.; Noel, E.; Cremer, M.; Neve, J. Deficiency in antioxidant factors in patients with alcohol-related chronic pancreatitis. Dig. Dis. Sci. 1996, 41, 1225–1231. [Google Scholar] [CrossRef]
- Muhammad, Y.; Kani, Y.A.; Iliya, S.; Muhammad, J.B.; Binji, A.; Ahmad, A.E.-F.; Kabir, M.B.; Bindawa, K.U.; Ahmed, A. Deficiency of antioxidants and increased oxidative stress in COVID-19 patients: A cross-sectional comparative study in Jigawa, Northwestern Nigeria. SAGE Open Med. 2021, 9, 2050312121991246. [Google Scholar] [CrossRef] [PubMed]
- Magesh, S.; Chen, Y.; Hu, L. Small Molecule Modulators of Keap1-Nrf2-ARE Pathway as Potential Preventive and Therapeutic Agents. Med. Res. Rev. 2012, 32, 687–726. [Google Scholar] [CrossRef]
- Tu, W.; Wang, H.; Li, S.; Liu, Q.; Sha, H. The Anti-Inflammatory and Anti-Oxidant Mechanisms of the Keap1/Nrf2/ARE Signaling Pathway in Chronic Diseases. Aging Dis. 2019, 10, 637–651. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-E.; You, D.-J.; Lee, C.; Ahn, C.; Seong, J.Y.; Hwang, J.-I. Suppression of NF-κB signaling by KEAP1 regulation of IKKβ activity through autophagic degradation and inhibition of phosphorylation. Cell. Signal. 2010, 22, 1645–1654. [Google Scholar] [CrossRef]
- Hayes, J.D.; Ashford, M.L. Nrf2 Orchestrates Fuel Partitioning for Cell Proliferation. Cell Metab. 2012, 16, 139–141. [Google Scholar] [CrossRef]
- Pellegrini, N.; Visioli, F.; Buratti, S.; Brighenti, F. Direct Analysis of Total Antioxidant Activity of Olive Oil and Studies on the Influence of Heating. J. Agric. Food Chem. 2001, 49, 2532–2538. [Google Scholar] [CrossRef]
- Leri, M.; Scuto, M.; Ontario, M.L.; Calabrese, V.; Calabrese, E.J.; Bucciantini, M.; Stefani, M. Healthy Effects of Plant Polyphenols: Molecular Mechanisms. Int. J. Mol. Sci. 2020, 21, 1250. Available online:https://pubmed.ncbi.nlm.nih.gov/32070025 (accessed on 21 April 2021). [CrossRef]
- Ortega-García, F.; Blanco, S.; Peinado, M.Á.; Peragón, J. Polyphenol oxidase and its relationship with oleuropein concentration in fruits and leaves of olive (Olea europaea) cv. ‘Picual’ trees during fruit ripening. Tree Physiol. 2008, 28, 45–54. [Google Scholar] [CrossRef]
- Peng, S.; Zhang, B.; Yao, J.; Duan, D.; Fang, J. Dual protection of hydroxytyrosol, an olive oil polyphenol, against oxidative damage in PC12 cells. Food Funct. 2015, 6, 2091–2100. [Google Scholar] [CrossRef] [PubMed]
- Fusco, R.; Cordaro, M.; Siracusa, R.; D’Amico, R.; Genovese, T.; Gugliandolo, E.; Peritore, A.F.; Crupi, R.; Impellizzeri, D.; Cuzzocrea, S.; et al. Biochemical Evaluation of the Antioxidant Effects of Hydroxytyrosol on Pancreatitis-Associated Gut Injury. Antioxidants 2020, 9, 781. [Google Scholar] [CrossRef] [PubMed]
- Schoenberg, M.H.; Büchler, M.; Younes, M.; Kirchmayr, R.; Brückner, U.B.; Beger, H.G. Effect of antioxidant treatment in rats with acute hemorrhagic pancreatitis. Dig. Dis. Sci. 1994, 39, 1034–1040. [Google Scholar] [CrossRef] [PubMed]
- Özgül, H.; Tatar, C.; Özer, B.; Aydın, H.; Sarı, S.; Özer, S.P. The effects of alpha tocopherol on acute pancreatitis in rats. Turk. J. Trauma Emerg. Surg. 2018, 25, 1–6. [Google Scholar] [CrossRef]
- Choi, S.; Kim, H. The Remedial Potential of Lycopene in Pancreatitis through Regulation of Autophagy. Int. J. Mol. Sci. 2020, 21, 5775. [Google Scholar] [CrossRef]
- Li, X.; Lu, X.; Chen, H. α-Tocopherol Treatment Ameliorates Chronic Pancreatitis in an Experimental Rat Model Induced by Trinitrobenzene Sulfonic Acid. Pancreatology 2011, 11, 5–11. [Google Scholar] [CrossRef]
- Bhardwaj, P.; Garg, P.K.; Maulik, S.K.; Saraya, A.; Tandon, R.K.; Acharya, S.K. A Randomized Controlled Trial of Antioxidant Supplementation for Pain Relief in Patients with Chronic Pancreatitis. Gastroenterology 2009, 136, 149–159.e2. [Google Scholar] [CrossRef]
- Siriwardena, A.K.; Mason, J.M.; Sheen, A.J.; Makin, A.J.; Shah, N.S. Antioxidant Therapy Does Not Reduce Pain in Patients with Chronic Pancreatitis: The ANTICIPATE Study. Gastroenterology 2012, 143, 655–663.e1. [Google Scholar] [CrossRef]
- Gu, W.-J.; Wei, C.-Y.; Yin, R.-X. Antioxidant supplementation for the prevention of post-endoscopic retrograde cholangiopancreatography pancreatitis: A meta-analysis of randomized controlled trials. Nutr. J. 2013, 12, 23. [Google Scholar] [CrossRef]
- Rustagi, T.; Njei, B. Antioxidant therapy for pain reduction in patients with chronic pancreatitis: A systematic review and meta-analysis. Pancreas 2015, 44, 812–818. [Google Scholar] [CrossRef] [PubMed]
- Robles, L. Role of Oxidative Stress in the Pathogenesis of Pancreatitis: Effect of Antioxidant Therapy. Pancreat. Disord. Ther. 2013, 3, 112. [Google Scholar] [CrossRef]
- Jovic, T.H.; Ali, S.R.; Ibrahim, N.; Jessop, Z.M.; Tarassoli, S.P.; Dobbs, T.D.; Holford, P.; A Thornton, C.; Whitaker, I.S. Could Vitamins Help in the Fight Against COVID-19? Nutrients 2020, 12, 2550. [Google Scholar] [CrossRef]
- Lai, F.; Zhang, Y.; Xie, D.-P.; Mai, S.-T.; Weng, Y.-N.; Du, J.-D.; Wu, G.-P.; Zheng, J.-X.; Han, Y. A Systematic Review of Rhubarb (a Traditional Chinese Medicine) Used for the Treatment of Experimental Sepsis. Evid. Based Complement. Altern. Med. 2015, 2015, 131283. [Google Scholar] [CrossRef]
- Li, J.; Zhang, C.-X.; Liu, Y.-M.; Chen, K.-L.; Chen, G. A comparative study of anti-aging properties and mechanism: Resveratrol and caloric restriction. Oncotarget 2017, 8, 65717–65729. [Google Scholar] [CrossRef] [PubMed]
- Yao, W.-Y.; Zhou, Y.-F.; Qian, A.; Zhang, Y.-P.; Qiao, M.-M.; Zhai, Z.; Yuan, Y.-Z.; Yang, S.-L. Emodin has a protective effect in cases of severe acute pancreatitis via inhibition of nuclear factor-κB activation resulting in antioxidation. Mol. Med. Rep. 2014, 11, 1416–1420. [Google Scholar] [CrossRef]
- Zhao, Z.; Xie, J.; Liu, B.; Ge, X.; Song, C.; Ren, M.; Zhou, Q.; Miao, L.; Zhang, H.; Shan, F.; et al. The effects of emodin on cell viability, respiratory burst and gene expression of Nrf2-Keap1 signaling molecules in the peripheral blood leukocytes of blunt snout bream (Megalobrama amblycephala). Fish Shellfish. Immunol. 2017, 62, 75–85. [Google Scholar] [CrossRef]
- Ning, J.-W.; Zhang, Y.; Yu, M.-S.; Gu, M.-L.; Xu, J.; Usman, A.; Ji, F. Emodin alleviates intestinal mucosal injury in rats with severe acute pancreatitis via the caspase-1 inhibition. Hepatobiliary Pancreat. Dis. Int. 2017, 16, 431–436. [Google Scholar] [CrossRef]
- Xiang, H.; Zhang, Q.; Wang, D.; Xia, S.; Wang, G.; Zhang, G.; Chen, H.; Wu, Y.; Shang, D. iTRAQ-based quantitative proteomic analysis for identification of biomarkers associated with emodin against severe acute pancreatitis in rats. RSC Adv. 2016, 6, 72447–72457. [Google Scholar] [CrossRef]
- Ni, Q.; Sun, K.; Chen, G.; Shang, D. In vitro effects of emodin on peritoneal macrophages that express membrane-bound CD14 protein in a rat model of severe acute pancreatitis/systemic inflammatory response syndrome. Mol. Med. Rep. 2014, 9, 355–359. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.-T.; Wan, B.; Liu, D.-D.; Wan, S.-X.; Fu, H.-Y.; Wan, Y.; Zhang, H.; Chen, Y. Emodin alleviates lung injury in rats with sepsis. J. Surg. Res. 2016, 202, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Cai, B.; Liu, X.; Cai, H. Emodin attenuates calcium overload and endoplasmic reticulum stress in AR42J rat pancreatic acinar cells. Mol. Med. Rep. 2013, 9, 267–272. [Google Scholar] [CrossRef]
- Wu, L.; Cai, B.; Zheng, S.; Liu, X.; Cai, H.; Li, H. Effect of Emodin on Endoplasmic Reticulum Stress in Rats with Severe Acute Pancreatitis. Inflammation 2013, 36, 1020–1029. [Google Scholar] [CrossRef]
- Schmitt, M.; Klonowski-Stumpe, H.; Eckert, M.; Lüthen, R.; Häussinger, D. Disruption of Paracellular Sealing is an Early Event in Acute Caerulein-Pancreatitis. Pancreas 2004, 28, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Liu, L.; Chen, B.; Bai, Y.; Zhang, F.; Li, Q.; Lv, C.; Sun, H.; Li, J.; Rubby, S.; et al. Involvement of the PI3K/Akt/NF-κB Signaling Pathway in the Attenuation of Severe Acute Pancreatitis-Associated Acute Lung Injury by Sedum sarmentosum Bunge Extract. BioMed Res. Int. 2017, 2017, 9698140. [Google Scholar] [CrossRef] [PubMed]
- Cha, T.-L.; Qiu, L.; Chen, C.-T.; Wen, Y.; Hung, M.-C. Emodin Down-Regulates Androgen Receptor and Inhibits Prostate Cancer Cell Growth. Cancer Res. 2005, 65, 2287–2295. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.-T.; Lin, S.-Z.; Liu, D.-L.; Wang, Z.-H. The distinct mechanisms of the antitumor activity of emodin in different types of cancer (Review). Oncol. Rep. 2013, 30, 2555–2562. [Google Scholar] [CrossRef] [PubMed]
- Sanders, B.; Ray, A.M.; Goldberg, S.; Clark, T.; McDaniel, H.R.; Atlas, S.E.; Farooqi, A.; Konefal, J.; Lages, L.C.; Lopez, J.; et al. Anti-cancer effects of aloe-emodin: A systematic review. J. Clin. Transl. Res. 2017, 3, 283–296. [Google Scholar]
- Shafik, N.M.; El Batsh, M.M. Protective Effects of Combined Selenium and Punica granatum Treatment on Some Inflammatory and Oxidative Stress Markers in Arsenic-Induced Hepatotoxicity in Rats. Biol. Trace Element Res. 2016, 169, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Jurenka, J.S. Anti-inflammatory properties of curcumin, a major constituent of Curcuma longa: A review of preclinical and clinical research. Altern. Med. Rev. 2009, 14, 141–153. [Google Scholar] [PubMed]
- Wang, Y.; Bu, C.; Wu, K.; Wang, R.; Wang, J. Curcumin protects the pancreas from acute pancreatitis via the mitogen-activated protein kinase signaling pathway. Mol. Med. Rep. 2019, 20, 3027–3034. [Google Scholar] [CrossRef] [PubMed]
- Sandur, S.K.; Pandey, M.K.; Sung, B.; Ahn, K.S.; Murakami, A.; Sethi, G.; Limtrakul, P.; Badmaev, V.; Aggarwal, B.B. Curcumin, demethoxycurcumin, bisdemethoxycurcumin, tetrahydrocurcumin and turmerones differentially regulate anti-inflammatory and anti-proliferative responses through a ROS-independent mechanism. Carcinogenesis 2007, 28, 1765–1773. [Google Scholar] [CrossRef] [PubMed]
- Sandur, S.K.; Ichikawa, H.; Pandey, M.K.; Kunnumakkara, A.B.; Sung, B.; Sethi, G.; Aggarwal, B.B. Role of pro-oxidants and antioxidants in the anti-inflammatory and apoptotic effects of curcumin (diferuloylmethane). Free Radic. Biol. Med. 2007, 43, 568–580. [Google Scholar] [CrossRef]
- Gupta, S.C.; Patchva, S.; Koh, W.; Aggarwal, B.B. Discovery of curcumin, a component of golden spice, and its miraculous biological activities. Clin. Exp. Pharmacol. Physiol. 2011, 39, 283–299. [Google Scholar] [CrossRef]
- Kunnumakkara, A.B.; Harsha, C.; Banik, K.; Vikkurthi, R.; Sailo, B.L.; Bordoloi, D.; Gupta, S.C.; Aggarwal, B.B. Is curcumin bioavailability a problem in humans: Lessons from clinical trials. Expert Opin. Drug Metab. Toxicol. 2019, 15, 705–733. [Google Scholar] [CrossRef] [PubMed]
- Durgaprasad, S.; Pai, C.G.; Alvres, J.F.; Namitha, S. Vasanthkumar A pilot study of the antioxidant effect of curcumin in tropical pancreatitis. Ind. J. Med. Res. 2005, 122, 315–318. [Google Scholar]
- Robles, L.; Vaziri, N.D.; Li, S.; Takasu, C.; Masuda, Y.; Vo, K.; Farzaneh, S.H.; Stamos, M.J.; Ichii, H. Dimethyl Fumarate Ameliorates Acute Pancreatitis in Rodent. Pancreas 2014, 44, 441–447. [Google Scholar] [CrossRef]
- Robles, L.; Vaziri, N.D.; Li, S.; Masuda, Y.; Takasu, C.; Takasu, M.; Vo, K.; Farzaneh, S.H.; Stamos, M.J.; Ichii, H. Dimethyl Fumarate Protects Pancreatic Islet Cells and Non-Endocrine Tissue in L-Arginine-Induced Chronic Pancreatitis. PLoS ONE 2014, 9, e107111. [Google Scholar] [CrossRef]
- Robles, L.; Vaziri, N.D.; Li, S.; Masuda, Y.; Takasu, C.; Takasu, M.; Vo, K.; Farzaneh, S.H.; Stamos, M.J.; Ichii, H. Synthetic Triterpenoid RTA dh404 (CDDO-dhTFEA) Ameliorates Acute Pancreatitis. Pancreas 2016, 45, 720–729. [Google Scholar] [CrossRef] [PubMed]
- Kojayan, G.G.; Alizadeh, R.F.; Li, S.; Ichii, H. Reducing Pancreatic Fibrosis Using Antioxidant Therapy Targeting Nrf2 Antioxidant Pathway. Pancreas 2019, 48, 1259–1262. [Google Scholar] [CrossRef]
- Zhang, W.-X.; Zhao, J.-H.; Ping, F.-M.; Liu, Z.-J.; Gu, J.-X.; Lu, X.-Q. Effect of dimethyl fumarate on rats with chronic pancreatitis. Asian Pac. J. Trop. Med. 2016, 9, 261–264. [Google Scholar] [CrossRef]
- Mrowietz, U.; Altmeyer, P.; Bieber, T.; Röcken, M.; Schopf, R.E.; Sterry, W. Treatment of psoriasis with fumaric acid esters (Fumaderm®). J. Dtsch. Dermatol. Ges. 2007, 5, 716–717. [Google Scholar] [CrossRef]
- TECFIDERA Dimethyl Fumarate Capsule. Available online: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=665d7e74-036c-5f68-5b67-ab84b9b49151 (accessed on 27 February 2021).
- Demir, I.E.; Friess, H.; Ceyhan, G.O. Neural plasticity in pancreatitis and pancreatic cancer. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 649–659. [Google Scholar] [CrossRef] [PubMed]
- Olesen, S.S.; Frøkjær, J.B.; Lelic, D.; Valeriani, M.; Drewes, A.M. Pain-Associated Adaptive Cortical Reorganisation in Chronic Pancreatitis. Pancreatology 2011, 10, 742–751. [Google Scholar] [CrossRef]
- Buscher, H.C.; Goor, H.; Wilder-Smith, O.H. Chronic pancreatitis patients show hyperalgesia of central origin: A pilot study. Eur. J. Pain 2006, 10, 363. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, M.; Singh, G.; Das, S.; Concha-Parra, R.; Erber, J.; Micames, C.; Gress, F. Efficacy of Endoscopic Ultrasound-guided Celiac Plexus Block and Celiac Plexus Neurolysis for Managing Abdominal Pain Associated with Chronic Pancreatitis and Pancreatic Cancer. J. Clin. Gastroenterol. 2010, 44, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Fusaroli, P.; Caletti, G. Is there a role for celiac plexus block for chronic pancreatitis? Endosc. Int. Open 2015, 3, E60–E62. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.-Q.; Liu, D.-Q.; Chen, S.-P.; Chen, N.; Sun, J.; Wang, X.-M.; Cao, F.; Tian, Y.-K.; Ye, D.-W. Nrf2 activation ameliorates mechanical allodynia in paclitaxel-induced neuropathic pain. Acta Pharmacol. Sin. 2020, 41, 1041–1048. [Google Scholar] [CrossRef]
- Singh, V.K.; Yadav, D.; Garg, P.K. Diagnosis and Management of Chronic Pancreatitis: A Review. JAMA 2019, 322, 2422–2434. [Google Scholar] [CrossRef]
- Narayanan, S.; Bhutiani, N.; Adamson, D.T.; Jones, C.M. Pancreatectomy, Islet Cell Transplantation, and Nutrition Considerations. Nutr. Clin. Pract. 2020, 6. [Google Scholar] [CrossRef]
- Kesseli, S.J.; Smith, K.A.; Gardner, T.B. Total pancreatectomy with islet autologous transplantation: The cure for chronic pancreatitis? Clin. Transl. Gastroenterol. 2015, 6, e73. [Google Scholar] [CrossRef] [PubMed]
- Forsmark, C.E. Management of chronic pancreatitis. Gastroenterology 2013, 144, 1282–1291.e3. [Google Scholar] [CrossRef] [PubMed]
- Parekh, D.; Natarajan, S. Surgical Management of Chronic Pancreatitis. Ind. J. Surg. 2015, 77, 453–469. [Google Scholar] [CrossRef] [PubMed]
- Baron, T.H. Endoscopic Pancreatic Necrosectomy. Gastroenterol. Hepatol. 2008, 4, 617–620. Available online: https://pubmed.ncbi.nlm.nih.gov/22798744 (accessed on 21 April 2021).
- Miles, C.B.; Gardner, T.B. Expanding indications for pancreatic islet cell transplantation. Curr. Opin. Gastroenterol. 2020, 36, 452–455. [Google Scholar] [CrossRef]
- Bellin, M.D.; Freeman, M.L.; Gelrud, A.; Slivka, A.; Clavel, A.; Humar, A.; Schwarzenberg, S.J.; Lowe, M.E.; Rickels, M.R.; Whitcomb, D.C.; et al. Total pancreatectomy and islet autotransplantation in chronic pancreatitis: Recommendations from Pancreas Fest. Pancreatology 2014, 14, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, D.E.; Matas, A.J.; Najarian, J.S. Pancreatic Islet Cell Transplantation. Surg. Clin. N. Am. 1978, 58, 365–382. [Google Scholar] [CrossRef]
- Shapiro, A.M.J.; Ricordi, C.; Hering, B.J.; Auchincloss, H.; Lindblad, R.; Robertson, R.P.; Secchi, A.; Brendel, M.D.; Berney, T.; Brennan, D.C.; et al. International Trial of the Edmonton Protocol for Islet Transplantation. N. Engl. J. Med. 2006, 355, 1318–1330. [Google Scholar] [CrossRef] [PubMed]
- Garcea, G.; Weaver, J.; Phillips, J.; Pollard, C.A.; Ilouz, S.C.; Webb, M.A.; Berry, D.P.; Dennison, A.R. Total Pancreatectomy with and Without Islet Cell Transplantation for Chronic Pancreatitis. Pancreas 2009, 38, 1–7. [Google Scholar] [CrossRef]
- Balzano, G.; Maffi, P.; Nano, R.; Mercalli, A.; Melzi, R.; Aleotti, F.; De Cobelli, F.; Magistretti, P.; Scavini, M.; Secchi, A.; et al. Diabetes-free survival after extended distal pancreatectomy and islet auto transplantation for benign or borderline/malignant lesions of the pancreas. Am. J. Transplant. 2019, 19, 920–928. [Google Scholar] [CrossRef]
- Robertson, R.P. Total Pancreatectomy and Islet Autotransplantation for Chronic Pancreatitis: Breaking Down Barriers. J. Clin. Endocrinol. Metab. 2015, 100, 1762–1763. [Google Scholar] [CrossRef]
- Kotagal, M.; Slusher, J.; Ahmad, S.; Aronson, L.A.; Brunner, J.; Chima, R.; Elder, D.A.; Goldschneider, K.R.; Hornung, L.; Lin, T.K.; et al. In-hospital and 90-day outcomes after total pancreatectomy with islet autotransplantation for pediatric chronic and acute recurrent pancreatitis. Am. J. Transplant. 2019, 19, 1187–1194. [Google Scholar] [CrossRef]
- Bellin, M.D.; Beilman, G.J.; Sutherland, D.E.; Ali, H.; Petersen, A.; Mongin, S.; Kirchner, V.; Schwarzenberg, S.J.; Trikudanathan, G.; Freeman, M.L.; et al. How Durable Is Total Pancreatectomy and Intraportal Islet Cell Transplantation for Treatment of Chronic Pancreatitis? J. Am. Coll. Surg. 2019, 228, 329–339. [Google Scholar] [CrossRef]
- Georgiev, G.; Del Rio, M.B.; Gruessner, A.; Tiwari, M.; Cercone, R.; Delbridge, M.; Grigsby, B.; Rilo, H. Patient quality of life and pain improve after autologous islet transplantation (AIT) for treatment of chronic pancreatitis: 53 patient series at the University of Arizona. Pancreatology 2015, 15, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, D.E.; Radosevich, D.M.; Bellin, M.D.; Hering, B.J.; Beilman, G.J.; Dunn, T.B.; Chinnakotla, S.; Vickers, S.M.; Bland, B.; Balamurugan, A.; et al. Total Pancreatectomy and Islet Autotransplantation for Chronic Pancreatitis. J. Am. Coll. Surg. 2012, 214, 409–424. [Google Scholar] [CrossRef] [PubMed]
- Bellin, M.D.; Kerdsirichairat, T.; Beilman, G.J.; Dunn, T.B.; Chinnakotla, S.; Pruett, T.L.; Radosevich, D.R.; Schwarzenberg, S.J.; Sutherland, D.E.; Arain, M.A.; et al. Total Pancreatectomy with Islet Autotransplantation Improves Quality of Life in Patients with Refractory Recurrent Acute Pancreatitis. Clin. Gastroenterol. Hepatol. 2016, 14, 1317–1323. [Google Scholar] [CrossRef] [PubMed]
- Wilson, G.C.; Sutton, J.M.; Abbott, D.E.; Smith, M.T.; Lowy, A.M.; Matthews, J.B.; Rilo, H.L.R.; Schmulewitz, N.; Salehi, M.; Choe, K.; et al. Long-Term Outcomes After Total Pancreatectomy and Islet Cell Autotransplantation. Ann. Surg. 2014, 260, 659–667. [Google Scholar] [CrossRef] [PubMed]
- Wilson, G.C.; Sutton, J.M.; Smith, M.T.; Schmulewitz, N.; Salehi, M.; Choe, K.A.; Levinsky, N.C., Jr.; Brunner, J.E.; Abbott, D.E.; Sussman, J.J.; et al. Completion pancreatectomy and islet cell autotransplantation as salvage therapy for patients failing previous operative interventions for chronic pancreatitis. Surgery 2015, 158, 872–879. [Google Scholar] [CrossRef] [PubMed]
- Johnston, P.C.; Lin, Y.K.; Walsh, R.M.; Bottino, R.; Stevens, T.K.; Trucco, M.; Bena, J.; Faiman, C.; Hatipoglu, B.A. Factors Associated with Islet Yield and Insulin Independence After Total Pancreatectomy and Islet Cell Autotransplantation in Patients with Chronic Pancreatitis Utilizing Off-site Islet Isolation: Cleveland Clinic Experience. J. Clin. Endocrinol. Metab. 2015, 100, 1765–1770. [Google Scholar] [CrossRef]
- Walsh, R.M.; Saavedra, J.R.A.; Lentz, G.; Guerron, A.D.; Scheman, J.; Stevens, T.; Trucco, M.; Bottino, R.; Hatipoglu, B. Improved Quality of Life Following Total Pancreatectomy and Auto-islet Transplantation for Chronic Pancreatitis. J. Gastrointest. Surg. 2012, 16, 1469–1477. [Google Scholar] [CrossRef]
- Wilson, G.C.; Sutton, J.M.; Salehi, M.; Schmulewitz, N.; Smith, M.T.; Kucera, S.; Choe, K.A.; Brunner, J.E.; Abbott, D.E.; Sussman, J.J.; et al. Surgical outcomes after total pancreatectomy and islet cell autotransplantation in pediatric patients. Surgery 2013, 154, 777–783. [Google Scholar] [CrossRef] [PubMed]
- Chinnakotla, S.; Radosevich, D.M.; Dunn, T.B.; Bellin, M.D.; Freeman, M.L.; Schwarzenberg, S.J.; Balamurugan, A.; Wilhelm, J.; Bland, B.; Vickers, S.M.; et al. Long-Term Outcomes of Total Pancreatectomy and Islet Auto Transplantation for Hereditary/Genetic Pancreatitis. J. Am. Coll. Surg. 2014, 218, 530–543. [Google Scholar] [CrossRef] [PubMed]
- Bellin, M.D.; Freeman, M.L.; Schwarzenberg, S.J.; Dunn, T.B.; Beilman, G.J.; Vickers, S.M.; Chinnakotla, S.; Balamurugan, A.; Hering, B.J.; Radosevich, D.M.; et al. Quality of Life Improves for Pediatric Patients After Total Pancreatectomy and Islet Autotransplant for Chronic Pancreatitis. Clin. Gastroenterol. Hepatol. 2011, 9, 793–799. Available online:https://pubmed.ncbi.nlm.nih.gov/21683160 (accessed on 21 April 2021). [CrossRef]
- Bellin, M.D.; Abu-El-Haija, M.; Morgan, K.; Adams, D.; Beilman, G.J.; Chinnakotla, S.; Conwell, D.L.; Dunn, T.B.; Freeman, M.L.; Gardner, T.; et al. POST study consortium A multicenter study of total pancreatectomy with islet autotransplantation (TPIAT): POST (Prospective Observational Study of TPIAT). Pancreatology 2018, 18, 286–290. [Google Scholar] [CrossRef]
- Balamurugan, A.; Loganathan, G.; Bellin, M.D.; Wilhelm, J.J.; Harmon, J.; Anazawa, T.; Soltani, S.M.; Radosevich, D.M.; Yuasa, T.; Tiwari, M.; et al. A New Enzyme Mixture to Increase the Yield and Transplant Rate of Autologous and Allogeneic Human Islet Products. Transplantation 2012, 93, 693–702. [Google Scholar] [CrossRef]
- Kaddis, J.S.; Danobeitia, J.S.; Niland, J.C.; Stiller, T.; Fernandez, L.A. Multicenter analysis of novel and established variables associated with successful human islet isolation outcomes. Am. J. Transplant. 2010, 10, 646–656. [Google Scholar] [CrossRef] [PubMed]
- Lopez, A.J.; Lau, H.; Li, S.; Ichii, H. Potential Benefits of Nrf2/Keap1 Targeting in Pancreatic Islet Cell Transplantation. Antioxidants 2020, 9, 321. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Gou, W.; Strange, C.; Wang, J.; Nietert, P.J.; Cloud, C.; Owzarski, S.; Shuford, B.; Duke, T.; Luttrell, L.; et al. Islet Harvest in Carbon Monoxide-Saturated Medium for Chronic Pancreatitis Patients Undergoing Islet Autotransplantation. Cell Transplant. 2019, 28, 25S–36S. [Google Scholar] [CrossRef]
- Ozmen, L.; Ekdahl, K.N.; Elgue, G.; Larsson, R.; Korsgren, O.; Nilsson, B. Inhibition of thrombin abrogates the instant blood-mediated inflammatory reaction triggered by isolated human islets: Possible application of the thrombin inhibitor melagatran in clinical islet transplantation. Diabetes 2002, 51, 1779–1784. [Google Scholar] [CrossRef]
- Naziruddin, B.; Iwahashi, S.; Kanak, M.A.; Takita, M.; Itoh, T.; Levy, M.F. Evidence for instant blood-mediated inflammatory reaction in clinical autologous islet transplantation. Am. J. Transplant. 2014, 14, 428–437. [Google Scholar] [CrossRef]
- Miki, A.; Ricordi, C.; Sakuma, Y.; Yamamoto, T.; Misawa, R.; Mita, A.; Molano, R.D.; Vaziri, N.D.; Pileggi, A.; Ichii, H. Divergent antioxidant capacity of human islet cell subsets: A potential cause of beta-cell vulnerability in diabetes and islet transplantation. PLoS ONE 2018, 13, e0196570. [Google Scholar] [CrossRef] [PubMed]
- Hubber, E.L.; Rackham, C.L.; Jones, P.M. Protecting islet functional viability using mesenchymal stromal cells. Stem Cells Transl. Med. 2021, 10. [Google Scholar] [CrossRef]
- Yang, B.; Zhou, Y.; Tian, L.; Lu, Y. Octreotide Reduces Pancreatic Islet Apoptosis and Improves Islet Transplantation Efficiency In Vitro and In Vivo. J. Biomed. Nanotechnol. 2020, 16, 1082–1101. [Google Scholar] [CrossRef]
- Keshtkar, S.; Kaviani, M.; Jabbarpour, Z.; Al-Abdullah, I.H.; Aghdaei, M.H.; Nikeghbalian, S.; Shamsaeefar, A.; Geramizadeh, B.; Azarpira, N.; Ghahremani, M.H. Significant reduction of apoptosis induced via hypoxia and oxidative stress in isolated human islet by resveratrol. Nutr. Metab. Cardiovasc. Dis. 2020, 30, 1216–1226. [Google Scholar] [CrossRef] [PubMed]
- Lebreton, F.; Bellofatto, K.; Wassmer, C.H.; Perez, L.; Lavallard, V.; Parnaud, G.; Cottet-Dumoulin, D.; Kerr-Conte, J.; Pattou, F.; Bosco, D.; et al. Shielding islets with human amniotic epithelial cells enhances islet engraftment and revascularization in a murine diabetes model. Am. J. Transplant. 2020, 20, 1551–1561. [Google Scholar] [CrossRef]
- Keshtkar, S.; Kaviani, M.; Jabbarpour, Z.; Geramizadeh, B.; Motevaseli, E.; Nikeghbalian, S.; Shamsaeefar, A.; Motazedian, N.; Al-Abdullah, I.H.; Ghahremani, M.H.; et al. Protective effect of nobiletin on isolated human islets survival and function against hypoxia and oxidative stress-induced apoptosis. Sci. Rep. 2019, 9, 1–13. [Google Scholar] [CrossRef]
- Masuda, Y.; Vaziri, N.D.; Li, S.; Le, A.; Hajighasemi-Ossareh, M.; Robles, L.; Foster, C.E.; Stamos, M.J.; Al-Abodullah, I.; Ricordi, C.; et al. The Effect of Nrf2 Pathway Activation on Human Pancreatic Islet Cells. PLoS ONE 2015, 10, e0131012. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Vaziri, N.D.; Masuda, Y.; Hajighasemi-Ossareh, M.; Robles, L.; Le, A.; Vo, K.; Chan, J.Y.; Foster, C.E.; Stamos, M.J.; et al. Pharmacological Activation of Nrf2 Pathway Improves Pancreatic Islet Isolation and Transplantation. Cell Transplant. 2015, 24, 2273–2283. [Google Scholar] [CrossRef] [PubMed]
- Von Zur-Mühlen, B.; Lundgren, T.; Bayman, L.; Berne, C.; Bridges, N.; Eggerman, T.; Foss, A.; Goldstein, J.; Jenssen, T.; Jorns, C.; et al. Open Randomized Multicenter Study to Evaluate Safety and Efficacy of Low Molecular Weight Sulfated Dextran in Islet Transplantation. Transplantation 2019, 103, 630–637. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, R.; Nunes, Q.; Huang, W.; Sutton, R. Precision medicine for acute pancreatitis: Current status and future opportunities. Precis. Clin. Med. 2019, 2, 81–86. [Google Scholar] [CrossRef]
- Monfared, S.S.M.S.; Vahidi, H.; Abdolghaffari, A.H.; Nikfar, S.; Abdollahi, M. Antioxidant therapy in the management of acute, chronic and post-ERCP pancreatitis: A systematic review. World J. Gastroenterol. 2009, 15, 4481–4490. [Google Scholar] [CrossRef] [PubMed]
- Kirk, G.R.; White, J.S.; McKie, L.; Stevenson, M.; Young, I.; Clements, W.D.B.; Rowlands, B.J. Combined Antioxidant Therapy Reduces Pain and Improves Quality of Life in Chronic Pancreatitis. J. Gastrointest. Surg. 2006, 10, 499–503. [Google Scholar] [CrossRef]
- Xue, P.; Deng, L.-H.; Xiao-Nan, Y.; Zhang, Z.-D.; Hu, W.-M.; Yang, X.-N.; Song, B.; Huang, Z.-W. Impact of alanyl-glutamine dipeptide on severe acute pancreatitis in early stage. World J. Gastroenterol. 2008, 14, 474–478. [Google Scholar] [CrossRef] [PubMed]
- Fuentes-Orozco, C.; Anaya-Prado, R.; González-Ojeda, A.; Arenas-Márquez, H.; Cabrera-Pivaral, C.; Cervantes-Guevara, G.; Barrera-Zepeda, L.M. l-Alanyl-l-glutamine-supplemented parenteral nutrition improves infectious morbidity in secondary peritonitis. Clin. Nutr. 2004, 23, 13–21. [Google Scholar] [CrossRef]
- Du, W.-D.; Yuan, Z.-R.; Sun, J.; Tang, J.-X.; Cheng, A.-Q.; Shen, D.-M.; Huang, C.-J.; Song, X.-H.; Yu, X.-F.; Zheng, S.-B. Therapeutic efficacy of high-dose vitamin C on acute pancreatitis and its potential mechanisms. World J. Gastroenterol. 2003, 9, 2565–2569. [Google Scholar] [CrossRef] [PubMed]
- Salim, A.S. Role of Oxygen-Derived Free Radical Scavengers in the Treatment of Recurrent Pain Produced by Chronic Pancreatitis. Arch. Surg. 1991, 126, 1109–1114. [Google Scholar] [CrossRef]
- Bolourani, S.; Diao, L.; Thompson, D.A.; Wang, P.; Coppa, G.F.; Patel, V.M.; Giangola, M.D. Risk Factors for Early Readmission After Acute Pancreatitis: Importance of Timely Interventions. J. Surg. Res. 2020, 252, 96–106. [Google Scholar] [CrossRef]
- Symon, M.; Trattner, D. Fix It with Food: More Than 125 Recipes to Address Autoimmune Issues and Inflammation: A Cookbook; Crown Publishing Group: New York City, NY, USA, 2019; p. 256. [Google Scholar]
- Micha, R.; Peñalvo, J.L.; Cudhea, F.; Imamura, F.; Rehm, C.D.; Mozaffarian, D. Association Between Dietary Factors and Mortality from Heart Disease, Stroke, and Type 2 Diabetes in the US. JAMA 2017, 317, 912–924. [Google Scholar] [CrossRef]
- Salehi, B.; Mishra, A.P.; Nigam, M.; Sener, B.; Kilic, M.; Sharifi-Rad, M.; Fokou, P.V.T.; Martins, N.; Sharifi-Rad, J. Resveratrol: A Double-Edged Sword in Health Benefits. Biomedicines 2018, 6, 91. [Google Scholar] [CrossRef]
- Najjar, E.; Staun-Ram, E.; Volkowich, A.; Miller, A. Dimethyl fumarate promotes B cell-mediated anti-inflammatory cytokine profile in B and T cells, and inhibits immune cell migration in patients with MS. J. Neuroimmunol. 2020, 343, 577230. [Google Scholar] [CrossRef]
- Lückel, C.; Picard, F.; Raifer, H.; Campos Carrascosa, L.; Guralnik, A.; Zhang, Y.; Klein, M.; Bittner, S.; Steffen, F.; Moos, S.; et al. IL-17+ CD8+ T cell suppression by dimethyl fumarate associates with clinical response in multiple sclerosis. Nat. Commun. 2019, 10, 5722. [Google Scholar] [CrossRef] [PubMed]

| Source | Baseline | Post-TPIAT | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| n | BP | PCS | SF | MCS | n | BP | PCS | SF | MCS | |
| Bellin et al. [106] | 59 | 25 | 27 | NR | 30 | 65 | 53 | 36 | NR | 42 |
| Bellin et al. [116] | 19 | 24 | 30 | 25 | 34 | 19 | 79 | 50 | 80 | 46 |
| Kotagal et al. [105] | 11 | NR | 34 | NR | NR | 11 | NR | 55 | NR | NR |
| Sutherland et al. [108] | 160 | 22 | 29 | 32 | 38 | 66 | 49 | 38 | 60 | 49 |
| Wilson et al. [110] | 23 | 9 | NR | 32 | NR | 34 | 59 | NR | 76 | NR |
| Wilson et al. [111] | 15 | 7 | NR | 42 | NR | 17 | 65 | NR | 81 | NR |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Swentek, L.; Chung, D.; Ichii, H. Antioxidant Therapy in Pancreatitis. Antioxidants 2021, 10, 657. https://doi.org/10.3390/antiox10050657
Swentek L, Chung D, Ichii H. Antioxidant Therapy in Pancreatitis. Antioxidants. 2021; 10(5):657. https://doi.org/10.3390/antiox10050657
Chicago/Turabian StyleSwentek, Lourdes, Dean Chung, and Hirohito Ichii. 2021. "Antioxidant Therapy in Pancreatitis" Antioxidants 10, no. 5: 657. https://doi.org/10.3390/antiox10050657
APA StyleSwentek, L., Chung, D., & Ichii, H. (2021). Antioxidant Therapy in Pancreatitis. Antioxidants, 10(5), 657. https://doi.org/10.3390/antiox10050657





