Melatonin Modulates the Antioxidant Defenses and the Expression of Proinflammatory Mediators in Pancreatic Stellate Cells Subjected to Hypoxia
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Culture of Pancreatic Stellate Cells
2.3. Induction of Hypoxia
2.4. Experimental Conditions for Melatonin Treatment
2.5. Determination of Reactive Oxygen Species Generation
2.6. Determination of Protein Carbonyls (Allysine)
2.7. Analysis of Thiobarbituric-Reactive Substances
2.8. Determination of Glutathione Levels
2.9. Determination of Total Antioxidant Capacity
2.10. Western Blotting Analysis
2.11. Quantitative Reverse Transcription-Polymerase Chain Reaction (RT-qPCR) Analysis
2.12. Determination of Cell Viability
2.13. Statistical Analysis
3. Results
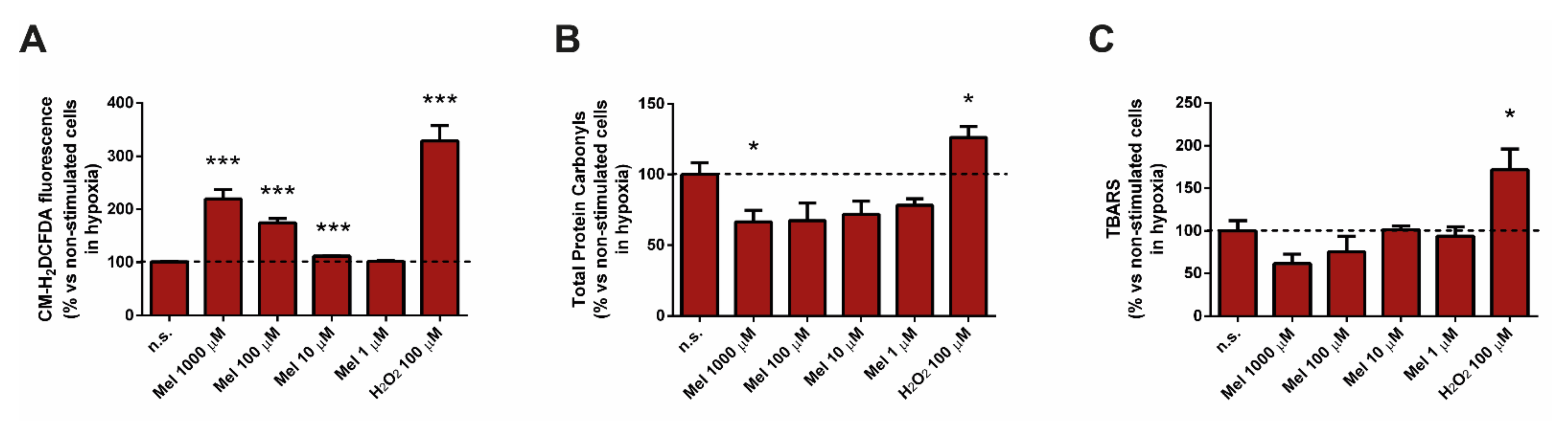
3.1. Effect of Melatonin on the Oxidative State of PSC
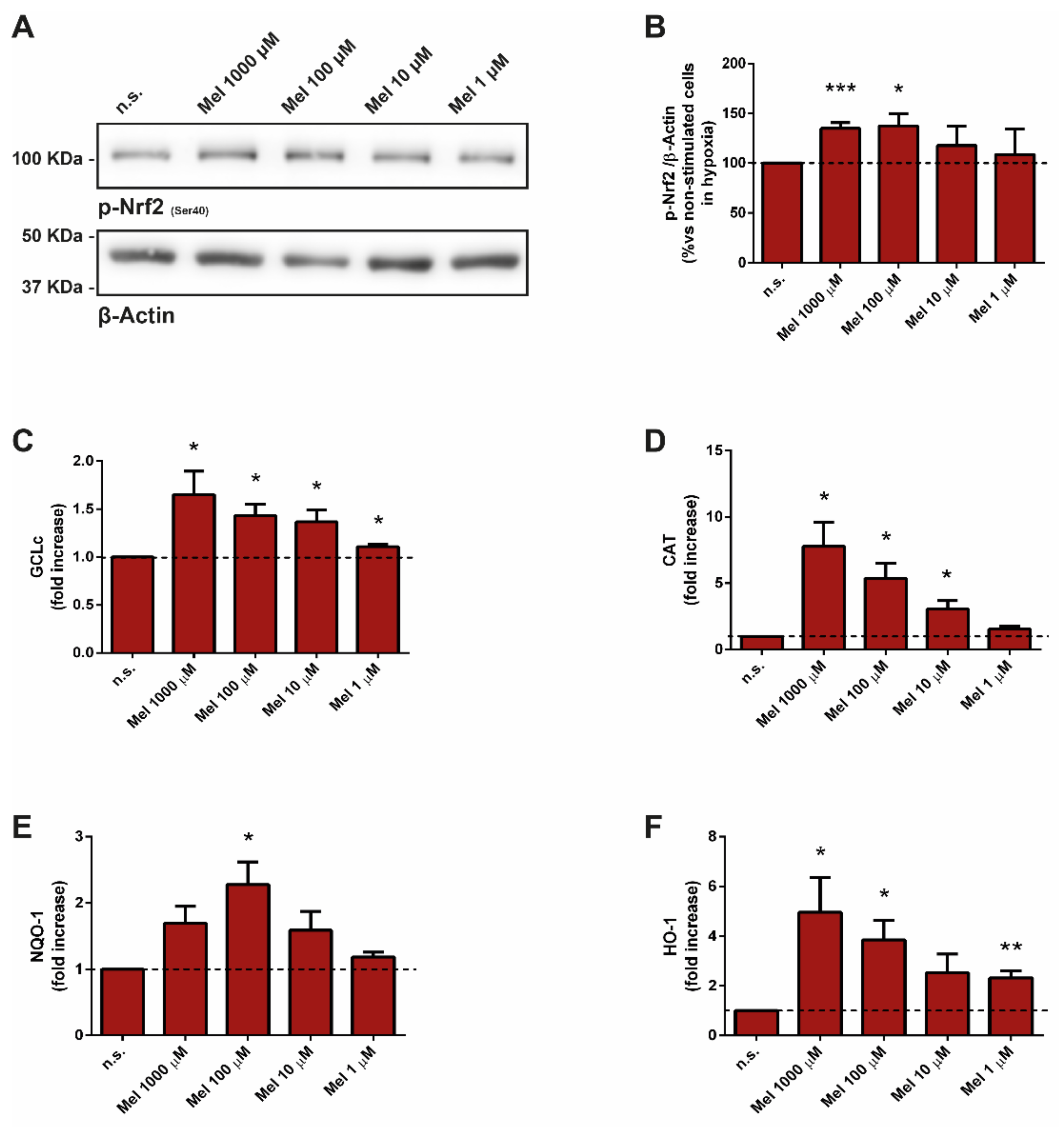
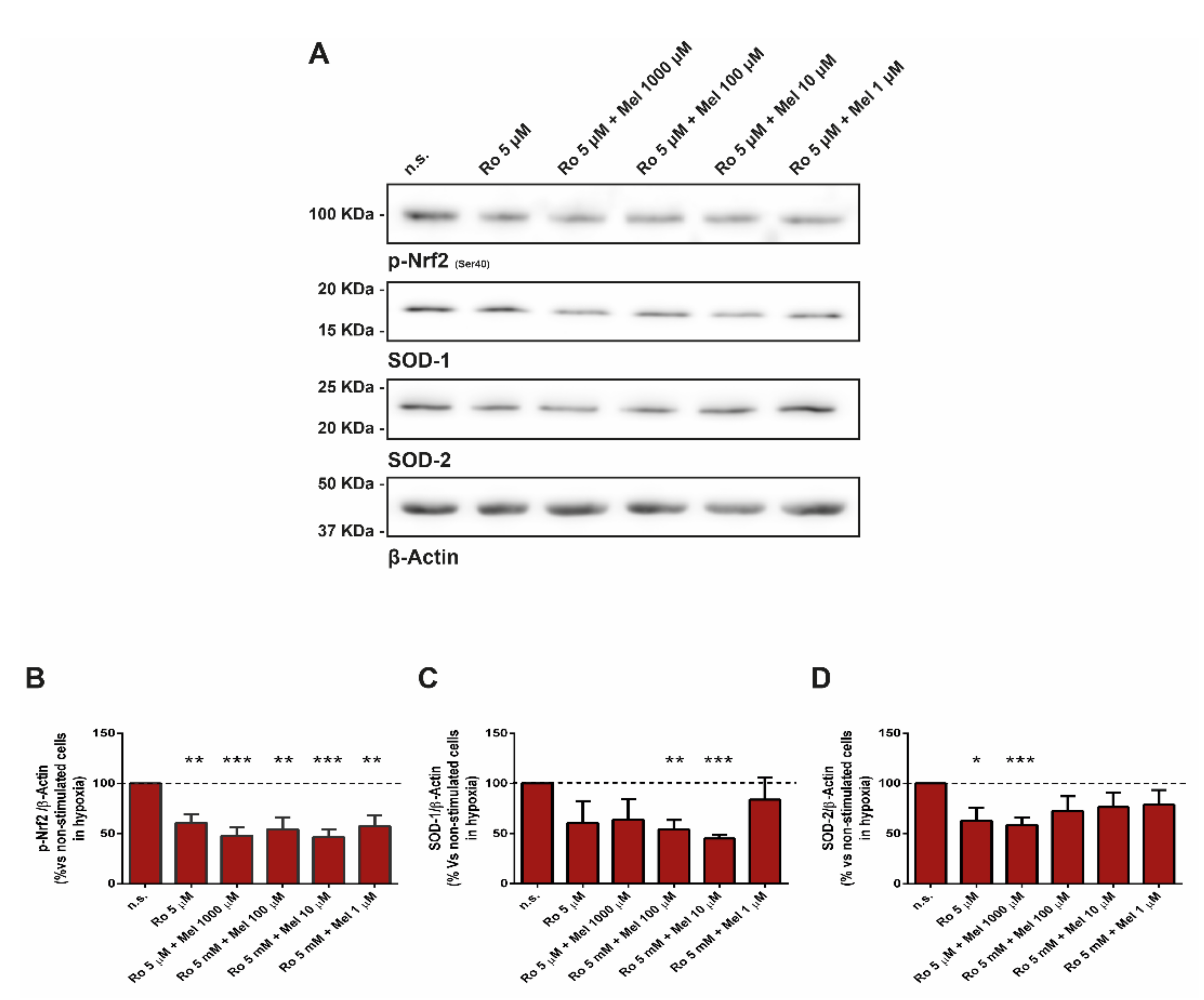
3.2. Effect of Melatonin on Nuclear Factor Erythroid 2-Related Factor and Related Antioxidant Enzymes
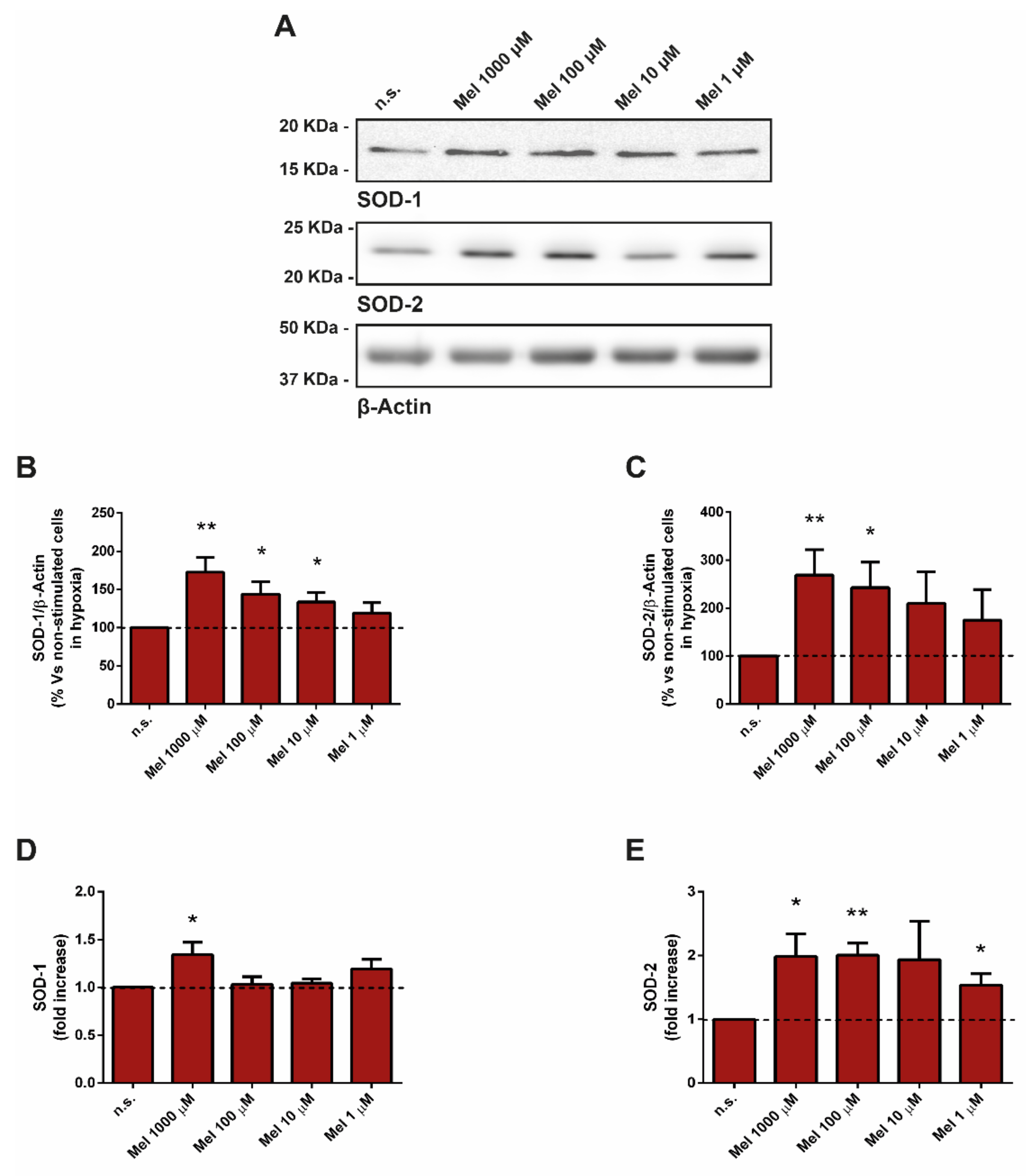
3.3. Effect of Melatonin on Superoxide Dismutase
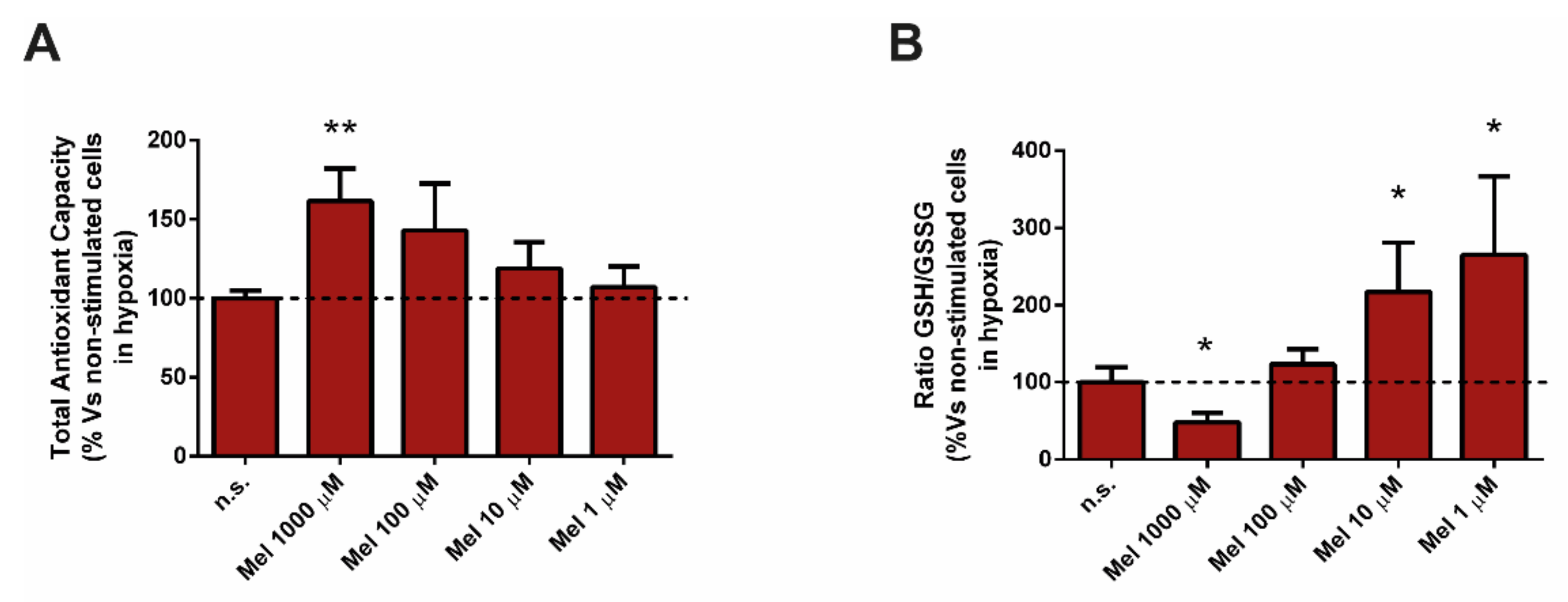
3.4. Effect of Melatonin on Glutathione and on Total Antioxidant Capacity (TAC)
3.5. Involvement of Protein Kinase C in Melatonin-Induced Changes in Nrf2-Related Antioxidant Enzymes and in SOD
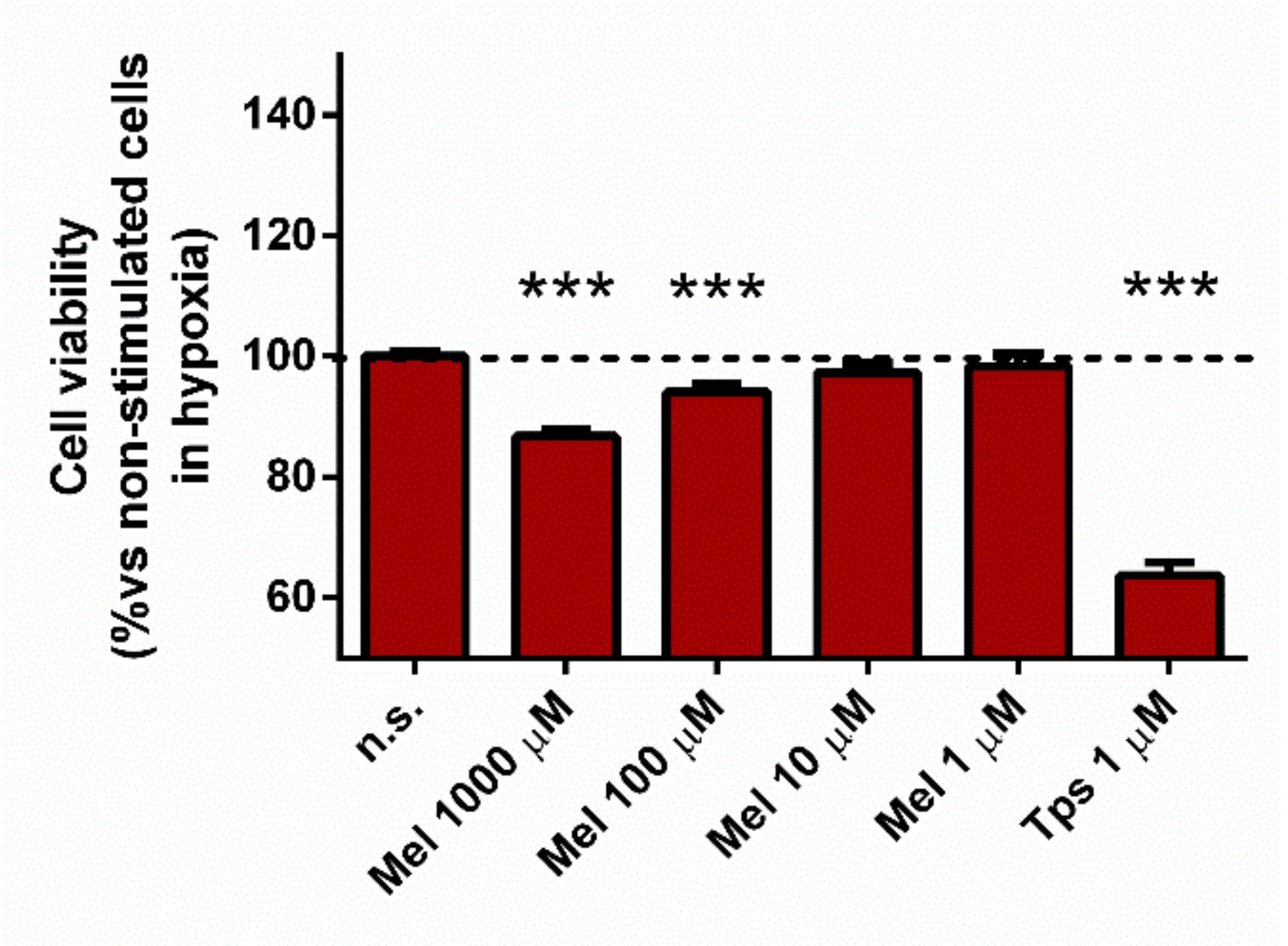
3.6. Effect of Melatonin on Cell Viability
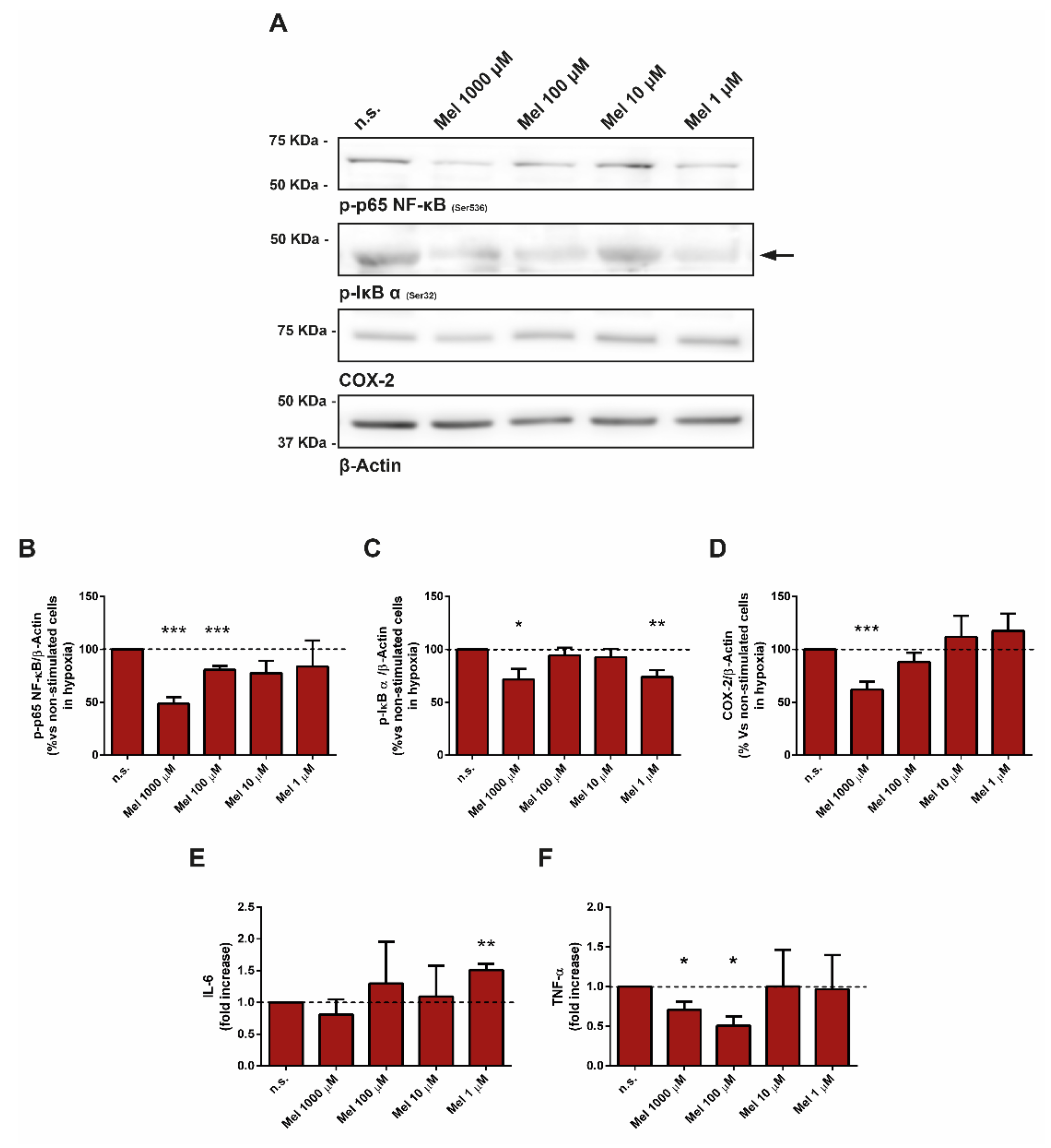
3.7. Effect of Melatonin on Pivotal Members of Inflammation
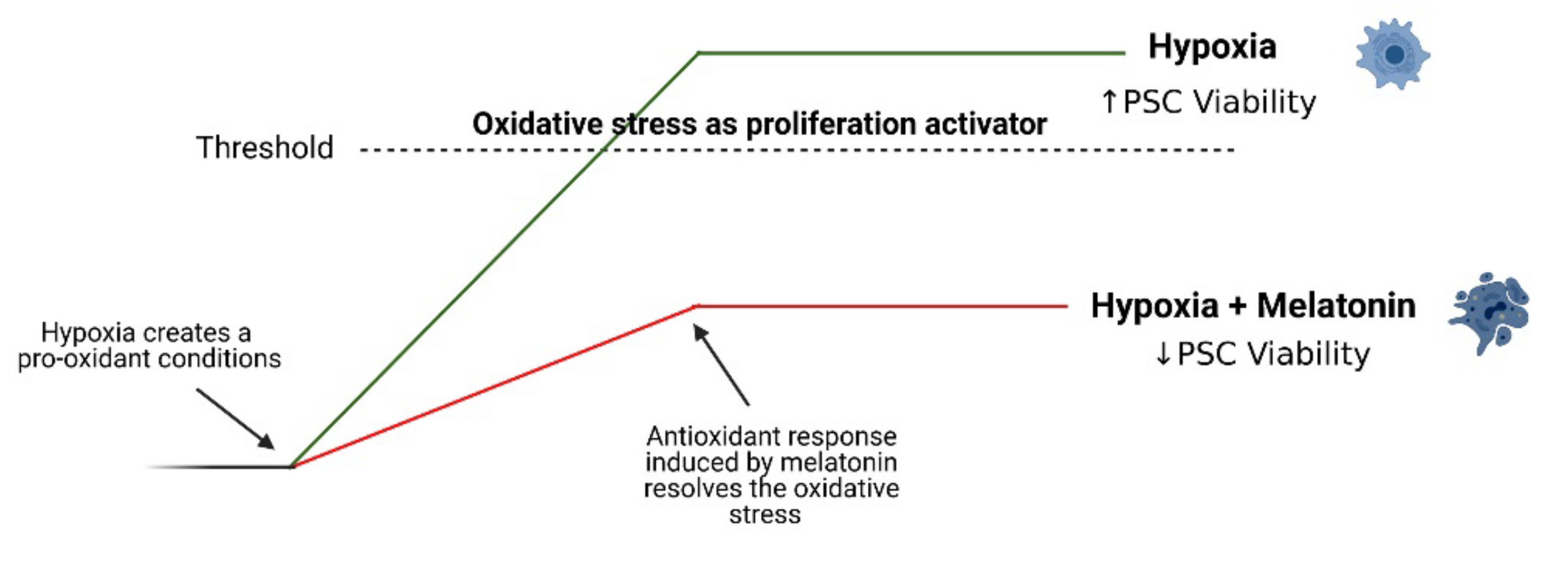
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sherman, M.H. Stellate Cells in Tissue Repair, Inflammation, and Cancer. Annu. Rev. Cell Dev. Biol. 2018, 34, 333–355. [Google Scholar] [CrossRef] [PubMed]
- Zinger, A.; Koren, L.; Adir, O.; Poley, M.; Alyan, M.; Yaari, Z.; Noor, N.; Krinsky, N.; Simon, A.; Gibori, H.; et al. Collagenase Nanoparticles Enhance the Penetration of Drugs into Pancreatic Tumors. ACS Nano 2019, 13, 11008–11021. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Zhang, H.; Ma, Q.; Huang, R.; Lu, J.; Liang, X.; Liu, X.; Zhang, Z.; Yu, L.; Pang, J.; et al. YAP1-mediated pancreatic stellate cell activation inhibits pancreatic cancer cell proliferation. Cancer Lett. 2019, 462, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Li, J.J.; Liang, X.L.; Wu, H.; Liang, Z. PSME3 Promotes TGFB1 Secretion by Pancreatic Cancer Cells to Induce Pancreatic Stellate Cell Proliferation. J. Cancer 2019, 10, 2128–2138. [Google Scholar] [CrossRef]
- Flores-Martinez, A.; Garcia-Nunez, A.; Rojas, A.; Cano, D.A. Stabilization of HIF-2alpha impacts pancreas growth. Sci. Rep. 2018, 8, 13713–13715. [Google Scholar] [CrossRef]
- Nan, X.; Wang, J.; Cheng, H.; Yin, Z.; Sheng, J.; Qiu, B.; Lau, C.C.; Yustein, J.T.; Zhao, H.; Wong, S.T.C. Imatinib revives the therapeutic potential of metformin on ewing sarcoma by attenuating tumor hypoxic response and inhibiting convergent signaling pathways. Cancer Lett. 2020, 469, 195–206. [Google Scholar] [CrossRef]
- Estaras, M.; Martinez-Morcillo, S.; Garcia, A.; Martinez, R.; Estevez, M.; Perez-Lopez, M.; Miguez, M.P.; Fernandez-Bermejo, M.; Mateos, J.M.; Vara, D.; et al. Pancreatic stellate cells exhibit adaptation to oxidative stress evoked by hypoxia. Biol. Cell 2020, 112, 280–299. [Google Scholar] [CrossRef]
- Erkan, M.; Reiser-Erkan, C.; Michalski, C.W.; Deucker, S.; Sauliunaite, D.; Streit, S.; Esposito, I.; Friess, H.; Kleeff, J. Cancer-stellate cell interactions perpetuate the hypoxia-fibrosis cycle in pancreatic ductal adenocarcinoma. Neoplasia 2009, 11, 497–508. [Google Scholar] [CrossRef]
- Patel, S.; Rahmani, B.; Gandhi, J.; Seyam, O.; Joshi, G.; Reid, I.; Smith, N.L.; Waltzer, W.C.; Khan, S.A. Revisiting the pineal gland: A review of calcification, masses, precocious puberty, and melatonin functions. Int. J. Neurosci. 2020, 130, 464–475. [Google Scholar] [CrossRef]
- Jaworek, J.; Leja-Szpak, A.; Bonior, J.; Nawrot, K.; Tomaszewska, R.; Stachura, J.; Sendur, R.; Pawlik, W.; Brzozowski, T.; Konturek, S.J. Protective effect of melatonin and its precursor L-tryptophan on acute pancreatitis induced by caerulein overstimulation or ischemia/reperfusion. J. Pineal Res. 2003, 34, 40–52. [Google Scholar] [CrossRef]
- Tordjman, S.; Chokron, S.; Delorme, R.; Charrier, A.; Bellissant, E.; Jaafari, N.; Fougerou, C. Melatonin: Pharmacology, Functions and Therapeutic Benefits. Curr. Neuropharmacol. 2017, 15, 434–443. [Google Scholar] [CrossRef]
- Carrasco, C.; Rodriguez, A.B.; Pariente, J.A. Effects of melatonin on the oxidative damage and pancreatic antioxidant defenses in cerulein-induced acute pancreatitis in rats. Hepatobiliary Pancreat. Dis. Int. 2014, 13, 442–446. [Google Scholar] [CrossRef]
- Brazao, V.; Santello, F.H.; Colato, R.P.; Mazotti, T.T.; Tazinafo, L.F.; Toldo, M.P.A.; do Vale, G.T.; Tirapelli, C.R.; do Prado, J.C. Melatonin: Antioxidant and modulatory properties in age-related changes during Trypanosoma cruzi infection. J. Pineal Res. 2017, 63. [Google Scholar] [CrossRef]
- Ramadan, H.M.; Taha, N.A.; Ahmed, H.H. Melatonin enhances antioxidant defenses but could not ameliorate the reproductive disorders in induced hyperthyroidism model in male rats. Environ. Sci. Pollut. Res. Int. 2021, 28, 4790–4804. [Google Scholar] [CrossRef]
- Santofimia-Castano, P.; Clea Ruy, D.; Garcia-Sanchez, L.; Jimenez-Blasco, D.; Fernandez-Bermejo, M.; Bolanos, J.P.; Salido, G.M.; Gonzalez, A. Melatonin induces the expression of Nrf2-regulated antioxidant enzymes via PKC and Ca2+ influx activation in mouse pancreatic acinar cells. Free Radic. Biol. Med. 2015, 87, 226–236. [Google Scholar] [CrossRef]
- Yang, Y.C.; Chiou, P.C.; Chen, P.C.; Liu, P.Y.; Huang, W.C.; Chao, C.C.; Tang, C.H. Melatonin reduces lung cancer stemness through inhibiting of PLC, ERK, p38, beta-catenin, and Twist pathways. Environ. Toxicol. 2019, 34, 203–209. [Google Scholar] [CrossRef]
- Chen, C.C.; Chen, C.Y.; Wang, S.H.; Yeh, C.T.; Su, S.C.; Ueng, S.H.; Chuang, W.Y.; Hsueh, C.; Wang, T.H. Melatonin Sensitizes Hepatocellular Carcinoma Cells to Chemotherapy Through Long Non-Coding RNA RAD51-AS1-Mediated Suppression of DNA Repair. Cancers 2018, 10, 320. [Google Scholar] [CrossRef]
- Mirza-Aghazadeh-Attari, M.; Mohammadzadeh, A.; Mostavafi, S.; Mihanfar, A.; Ghazizadeh, S.; Sadighparvar, S.; Gholamzadeh, S.; Majidinia, M.; Yousefi, B. Melatonin: An important anticancer agent in colorectal cancer. J. Cell Physiol. 2020, 235, 804–817. [Google Scholar] [CrossRef]
- Li, W.; Wu, J.; Li, Z.; Zhou, Z.; Zheng, C.; Lin, L.; Tan, B.; Huang, M.; Fan, M. Melatonin induces cell apoptosis in Mia PaCa-2 cells via the suppression of nuclear factor-kappaB and activation of ERK and JNK: A novel therapeutic implication for pancreatic cancer. Oncol. Rep. 2016, 36, 2861–2867. [Google Scholar] [CrossRef]
- Jaworek, J.; Leja-Szpak, A.; Nawrot-Porabka, K.; Szklarczyk, J.; Kot, M.; Pierzchalski, P.; Goralska, M.; Ceranowicz, P.; Warzecha, Z.; Dembinski, A.; et al. Effects of Melatonin and Its Analogues on Pancreatic Inflammation, Enzyme Secretion, and Tumorigenesis. Int. J. Mol. Sci. 2017, 18, 1014. [Google Scholar] [CrossRef]
- Estaras, M.; Moreno, N.; Santofimia-Castano, P.; Martinez-Morcillo, S.; Roncero, V.; Blanco, G.; Lopez, D.; Fernandez-Bermejo, M.; Mateos, J.M.; Iovanna, J.L.; et al. Melatonin induces reactive oxygen species generation and changes in glutathione levels and reduces viability in human pancreatic stellate cells. J. Physiol. Biochem. 2019, 75, 185–197. [Google Scholar] [CrossRef]
- Santofimia-Castano, P.; Garcia-Sanchez, L.; Ruy, D.C.; Sanchez-Correa, B.; Fernandez-Bermejo, M.; Tarazona, R.; Salido, G.M.; Gonzalez, A. Melatonin induces calcium mobilization and influences cell proliferation independently of MT1/MT2 receptor activation in rat pancreatic stellate cells. Cell Biol. Toxicol. 2015, 31, 95–110. [Google Scholar] [CrossRef]
- Muhlbauer, E.; Gross, E.; Labucay, K.; Wolgast, S.; Peschke, E. Loss of melatonin signalling and its impact on circadian rhythms in mouse organs regulating blood glucose. Eur. J. Pharmacol. 2009, 606, 61–71. [Google Scholar] [CrossRef]
- Slominski, R.M.; Reiter, R.J.; Schlabritz-Loutsevitch, N.; Ostrom, R.S.; Slominski, A.T. Melatonin membrane receptors in peripheral tissues: Distribution and functions. Mol. Cell Endocrinol. 2012, 351, 152–166. [Google Scholar] [CrossRef] [PubMed]
- McCarroll, J.A.; Naim, S.; Sharbeen, G.; Russia, N.; Lee, J.; Kavallaris, M.; Goldstein, D.; Phillips, P.A. Role of pancreatic stellate cells in chemoresistance in pancreatic cancer. Front. Physiol. 2014, 5, 141. [Google Scholar] [CrossRef]
- Zha, M.; Li, F.; Xu, W.; Chen, B.; Sun, Z. Isolation and characterization of islet stellate cells in rat. Islets 2014, 6, e28701. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gonzalez, A.; Salido, G.M. Determination of reactive oxygen species production in pancreatic acinar cells. Pancreapedia Exocrine Pancreas Knowl. Base 2016. Available online: https://pancreapedia.org/tools/methods/determination-of-reactive-oxygen-species-production-in-pancreatic-acinar-cells (accessed on 6 December 2016). [CrossRef]
- Villaverde, A.; Parra, V.; Estevez, M. Oxidative and nitrosative stress induced in myofibrillar proteins by a hydroxyl-radical-generating system: Impact of nitrite and ascorbate. J. Agric. Food Chem. 2014, 62, 2158–2164. [Google Scholar] [CrossRef]
- Gonzalez, A.; Estaras, M.; Martinez-Morcillo, S.; Martinez, R.; Garcia, A.; Estevez, M.; Santofimia-Castano, P.; Tapia, J.A.; Moreno, N.; Perez-Lopez, M.; et al. Melatonin modulates red-ox state and decreases viability of rat pancreatic stellate cells. Sci. Rep. 2020, 10, 6352–6356. [Google Scholar] [CrossRef]
- Gonzalez, A.; del Castillo-Vaquero, A.; Miro-Moran, A.; Tapia, J.A.; Salido, G.M. Melatonin reduces pancreatic tumor cell viability by altering mitochondrial physiology. J. Pineal Res. 2011, 50, 250–260. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Nath, R.; Raser, K.J.; Hajimohammadreza, I.; Wang, K.K. Thapsigargin induces apoptosis in SH-SY5Y neuroblastoma cells and cerebrocortical cultures. Biochem. Mol. Biol. Int. 1997, 43, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Algul, H.; Treiber, M.; Lesina, M.; Schmid, R.M. Mechanisms of disease: Chronic inflammation and cancer in the pancreas—A potential role for pancreatic stellate cells? Nat. Clin. Pract. Gastroenterol. Hepatol. 2007, 4, 454–462. [Google Scholar] [CrossRef] [PubMed]
- Birtolo, C.; Pham, H.; Morvaridi, S.; Chheda, C.; Go, V.L.; Ptasznik, A.; Edderkaoui, M.; Weisman, M.H.; Noss, E.; Brenner, M.B.; et al. Cadherin-11 Is a Cell Surface Marker Up-Regulated in Activated Pancreatic Stellate Cells and Is Involved in Pancreatic Cancer Cell Migration. Am. J. Pathol. 2017, 187, 146–155. [Google Scholar] [CrossRef]
- Li, Y.; Song, T.; Chen, Z.; Wang, Y.; Zhang, J.; Wang, X. Pancreatic Stellate Cells Activation and Matrix Metallopeptidase 2 Expression Correlate with Lymph Node Metastasis in Pancreatic Carcinoma. Am. J. Med. Sci. 2019, 357, 16–22. [Google Scholar] [CrossRef]
- Manohar, M.; Verma, A.K.; Venkateshaiah, S.U.; Sanders, N.L.; Mishra, A. Pathogenic mechanisms of pancreatitis. World J. Gastrointest. Pharmacol. Ther. 2017, 8, 10–25. [Google Scholar] [CrossRef]
- Chao, C.C.; Chen, P.C.; Chiou, P.C.; Hsu, C.J.; Liu, P.I.; Yang, Y.C.; Reiter, R.J.; Yang, S.F.; Tang, C.H. Melatonin suppresses lung cancer metastasis by inhibition of epithelial-mesenchymal transition through targeting to Twist. Clin. Sci. 2019, 133, 709–722. [Google Scholar] [CrossRef]
- Odeh, L.H.; Talib, W.H.; Basheti, I.A. Synergistic effect of thymoquinone and melatonin against breast cancer implanted in mice. J. Cancer Res. Ther. 2018, 14, S324–S330. [Google Scholar] [CrossRef]
- Wang, T.H.; Hsueh, C.; Chen, C.C.; Li, W.S.; Yeh, C.T.; Lian, J.H.; Chang, J.L.; Chen, C.Y. Melatonin Inhibits the Progression of Hepatocellular Carcinoma through MicroRNA Let7i-3p Mediated RAF1 Reduction. Int. J. Mol. Sci. 2018, 19, 2687. [Google Scholar] [CrossRef]
- Tamtaji, O.R.; Mirhosseini, N.; Reiter, R.J.; Behnamfar, M.; Asemi, Z. Melatonin and pancreatic cancer: Current knowledge and future perspectives. J. Cell Physiol. 2019, 234, 5372–5378. [Google Scholar] [CrossRef]
- Estaras, M.; Pena, F.J.; Tapia, J.A.; Fernandez-Bermejo, M.; Mateos, J.M.; Vara, D.; Roncero, V.; Blanco, G.; Lopez, D.; Salido, G.M.; et al. Melatonin modulates proliferation of pancreatic stellate cells through caspase-3 activation and changes in cyclin A and D expression. J. Physiol. Biochem. 2020, 76, 345–355. [Google Scholar] [CrossRef]
- Cuesta, S.; Kireev, R.; Garcia, C.; Forman, K.; Escames, G.; Vara, E.; Tresguerres, J.A. Beneficial effect of melatonin treatment on inflammation, apoptosis and oxidative stress on pancreas of a senescence accelerated mice model. Mech. Ageing Dev. 2011, 132, 573–582. [Google Scholar] [CrossRef]
- Quail, D.F.; Joyce, J.A. Microenvironmental regulation of tumor progression and metastasis. Nat. Med. 2013, 19, 1423–1437. [Google Scholar] [CrossRef]
- Cristofanon, S.; Uguccioni, F.; Cerella, C.; Radogna, F.; Dicato, M.; Ghibelli, L.; Diederich, M. Intracellular prooxidant activity of melatonin induces a survival pathway involving NF-kappaB activation. Ann. N. Y. Acad. Sci. 2009, 1171, 472–478. [Google Scholar] [CrossRef]
- Gurer-Orhan, H.; Suzen, S. Melatonin, its metabolites and its synthetic analogs as multi-faceted compounds: Antioxidant, prooxidant and inhibitor of bioactivation reactions. Curr. Med. Chem. 2015, 22, 490–499. [Google Scholar] [CrossRef]
- Ozdemir, F.; Deniz, O.; Kaynar, K.; Arslan, M.; Kavgaci, H.; Yildiz, B.; Aydin, F. The effects of melatonin on human hepatoma (Hep G2) cell line. Bratisl. Lek. Listy 2009, 110, 276–279. [Google Scholar]
- Zhang, H.M.; Zhang, Y. Melatonin: A well-documented antioxidant with conditional pro-oxidant actions. J. Pineal Res. 2014, 57, 131–146. [Google Scholar] [CrossRef]
- Osseni, R.A.; Rat, P.; Bogdan, A.; Warnet, J.M.; Touitou, Y. Evidence of prooxidant and antioxidant action of melatonin on human liver cell line HepG2. Life Sci. 2000, 68, 387–399. [Google Scholar] [CrossRef]
- Wolfler, A.; Caluba, H.C.; Abuja, P.M.; Dohr, G.; Schauenstein, K.; Liebmann, P.M. Prooxidant activity of melatonin promotes fas-induced cell death in human leukemic Jurkat cells. FEBS Lett. 2001, 502, 127–131. [Google Scholar] [CrossRef]
- Albertini, M.C.; Radogna, F.; Accorsi, A.; Uguccioni, F.; Paternoster, L.; Cerella, C.; De Nicola, M.; D’Alessio, M.; Bergamaschi, A.; Magrini, A.; et al. Intracellular pro-oxidant activity of melatonin deprives U937 cells of reduced glutathione without affecting glutathione peroxidase activity. Ann. N. Y. Acad. Sci. 2006, 1091, 10–16. [Google Scholar] [CrossRef]
- Bejarano, I.; Espino, J.; Barriga, C.; Reiter, R.J.; Pariente, J.A.; Rodriguez, A.B. Pro-oxidant effect of melatonin in tumour leucocytes: Relation with its cytotoxic and pro-apoptotic effects. Basic Clin. Pharmacol. Toxicol. 2011, 108, 14–20. [Google Scholar] [CrossRef]
- Zhang, H.M.; Zhang, Y.; Zhang, B.X. The role of mitochondrial complex III in melatonin-induced ROS production in cultured mesangial cells. J. Pineal Res. 2011, 50, 78–82. [Google Scholar] [CrossRef]
- Girish, K.S.; Paul, M.; Thushara, R.M.; Hemshekhar, M.; Shanmuga Sundaram, M.; Rangappa, K.S.; Kemparaju, K. Melatonin elevates apoptosis in human platelets via ROS mediated mitochondrial damage. Biochem. Biophys. Res. Commun. 2013, 438, 198–204. [Google Scholar] [CrossRef]
- Hanko, M.; Svorc, L.; Plankova, A.; Mikus, P. Overview and recent advances in electrochemical sensing of glutathione—A review. Anal. Chim. Acta 2019, 1062, 1–27. [Google Scholar] [CrossRef]
- Garcia-Santos, G.; Antolin, I.; Herrera, F.; Martin, V.; Rodriguez-Blanco, J.; del Pilar Carrera, M.; Rodriguez, C. Melatonin induces apoptosis in human neuroblastoma cancer cells. J. Pineal Res. 2006, 41, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Munik, M.S.; Ekmekçioglu, C. Prooxidant effects of melatonin: A brief review. Turk. J. Biol. 2015, 39, 832–839. [Google Scholar] [CrossRef]
- Orhan, H.; Gurer-Orhan, H.; Vriese, E.; Vermeulen, N.P.; Meerman, J.H. Application of lipid peroxidation and protein oxidation biomarkers for oxidative damage in mammalian cells. A comparison with two fluorescent probes. Toxicol. In Vitro 2006, 20, 1005–1013. [Google Scholar] [CrossRef]
- Robledinos-Anton, N.; Fernandez-Gines, R.; Manda, G.; Cuadrado, A. Activators and Inhibitors of NRF2: A Review of Their Potential for Clinical Development. Oxid. Med. Cell Longev. 2019, 2019, 9372182. [Google Scholar] [CrossRef]
- Masuda, Y.; Vaziri, N.D.; Li, S.; Le, A.; Hajighasemi-Ossareh, M.; Robles, L.; Foster, C.E.; Stamos, M.J.; Al-Abodullah, I.; Ricordi, C.; et al. The effect of Nrf2 pathway activation on human pancreatic islet cells. PLoS ONE 2015, 10, e0131012. [Google Scholar] [CrossRef]
- Lin, Z.; Deng, A. Antioxidative and Free Radical Scavenging Activity of Ganoderma (Lingzhi). Adv. Exp. Med. Biol. 2019, 1182, 271–297. [Google Scholar] [CrossRef] [PubMed]
- Milani, P.; Gagliardi, S.; Cova, E.; Cereda, C. SOD1 Transcriptional and Posttranscriptional Regulation and Its Potential Implications in ALS. Neurol. Res. Int. 2011, 2011, 458427. [Google Scholar] [CrossRef] [PubMed]
- Zou, X.; Ratti, B.A.; O’Brien, J.G.; Lautenschlager, S.O.; Gius, D.R.; Bonini, M.G.; Zhu, Y. Manganese superoxide dismutase (SOD2): Is there a center in the universe of mitochondrial redox signaling? J. Bioenerg. Biomembr. 2017, 49, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Hoshino, H.; Takaku, K.; Nakajima, O.; Muto, A.; Suzuki, H.; Tashiro, S.; Takahashi, S.; Shibahara, S.; Alam, J.; et al. Hemoprotein Bach1 regulates enhancer availability of heme oxygenase-1 gene. EMBO J. 2002, 21, 5216–5224. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, A.A.; Zuvanich, E.G.; Khan, D.A.; Mushtaq, S.; Silswal, N.; Qureshi, N. Proteasome inhibitors modulate anticancer and anti-proliferative properties via NF-kB signaling, and ubiquitin-proteasome pathways in cancer cell lines of different organs. Lipids Health Dis. 2018, 17, 62–65. [Google Scholar] [CrossRef]
- Wang, Y.R.; Tian, F.L.; Yan, M.X.; Fan, J.H.; Wang, L.Y.; Kuang, R.G.; Li, Y.Q. Sulfasalazine inhibits inflammation and fibrogenesis in pancreas via NF-kappaB signaling pathway in rats with oxidative stress-induced pancreatic injury. Drug Des. Dev. Ther. 2016, 10, 1743–1751. [Google Scholar] [CrossRef][Green Version]
- Dobrovolskaia, M.A.; Kozlov, S.V. Inflammation and cancer: When NF-kappaB amalgamates the perilous partnership. Curr. Cancer Drug Targets 2005, 5, 325–344. [Google Scholar] [CrossRef]
- Lesina, M.; Wormann, S.M.; Neuhofer, P.; Song, L.; Algul, H. Interleukin-6 in inflammatory and malignant diseases of the pancreas. Semin. Immunol. 2014, 26, 80–87. [Google Scholar] [CrossRef]
- Binker, M.G.; Binker-Cosen, A.A.; Richards, D.; Gaisano, H.Y.; de Cosen, R.H.; Cosen-Binker, L.I. Chronic stress sensitizes rats to pancreatitis induced by cerulein: Role of TNF-alpha. World J. Gastroenterol. 2010, 16, 5565–5581. [Google Scholar] [CrossRef]
- Carrascal, L.; Nunez-Abades, P.; Ayala, A.; Cano, M. Role of Melatonin in the Inflammatory Process and its Therapeutic Potential. Curr. Pharm. Des. 2018, 24, 1563–1588. [Google Scholar] [CrossRef]
- Hu, C.; Zhao, L.; Tao, J.; Li, L. Protective role of melatonin in early-stage and end-stage liver cirrhosis. J. Cell Mol. Med. 2019, 23, 7151–7162. [Google Scholar] [CrossRef]
- Jaworek, J.; Konturek, S.J. Hormonal protection in acute pancreatitis by ghrelin, leptin and melatonin. World J. Gastroenterol. 2014, 20, 16902–16912. [Google Scholar] [CrossRef]
- Du, X.; He, S.; Jiang, Y.; Wei, L.; Hu, W. Adiponectin prevents islet ischemia-reperfusion injury through the COX2-TNFalpha-NF-kappaB-dependent signal transduction pathway in mice. J. Endocrinol. 2013, 218, 75–84. [Google Scholar] [CrossRef]
- Ohashi, S.; Nishio, A.; Nakamura, H.; Asada, M.; Tamaki, H.; Kawasaki, K.; Fukui, T.; Yodoi, J.; Chiba, T. Overexpression of redox-active protein thioredoxin-1 prevents development of chronic pancreatitis in mice. Antioxid. Redox Signal. 2006, 8, 1835–1845. [Google Scholar] [CrossRef]
- Masamune, A.; Watanabe, T.; Kikuta, K.; Shimosegawa, T. Roles of pancreatic stellate cells in pancreatic inflammation and fibrosis. Clin. Gastroenterol. Hepatol. 2009, 7, 48. [Google Scholar] [CrossRef]
- Acuna-Castroviejo, D.; Escames, G.; Venegas, C.; Diaz-Casado, M.E.; Lima-Cabello, E.; Lopez, L.C.; Rosales-Corral, S.; Tan, D.X.; Reiter, R.J. Extrapineal melatonin: Sources, regulation, and potential functions. Cell Mol. Life Sci. 2014, 71, 2997–3025. [Google Scholar] [CrossRef]
- Chen, C.Q.; Fichna, J.; Bashashati, M.; Li, Y.Y.; Storr, M. Distribution, function and physiological role of melatonin in the lower gut. World J. Gastroenterol. 2011, 17, 3888–3898. [Google Scholar] [CrossRef]
- Garcia-Marin, R.; de Miguel, M.; Fernandez-Santos, J.M.; Carrillo-Vico, A.; Utrilla, J.C.; Morillo-Bernal, J.; Diaz-Parrado, E.; Rodriguez-Prieto, I.; Guerrero, J.M.; Martin-Lacave, I. Melatonin-synthesizing enzymes and melatonin receptor in rat thyroid cells. Histol. Histopathol. 2012, 27, 1429–1438. [Google Scholar] [CrossRef]
- Martin, M.; Macias, M.; Leon, J.; Escames, G.; Khaldy, H.; Acuna-Castroviejo, D. Melatonin increases the activity of the oxidative phosphorylation enzymes and the production of ATP in rat brain and liver mitochondria. Int. J. Biochem. Cell Biol. 2002, 34, 348–357. [Google Scholar] [CrossRef]

| Antibody | Dilution | Supplier |
|---|---|---|
| Β-Actin HRP-Conjugated | 1:50,000 | Thermo Fisher |
| COX-2 | 1:2000 | Cell Signaling |
| p-Nrf2 (Ser40) | 1:2000 | Thermo Fisher |
| p-p65 NF-κB (Ser536) | 1:1000 | Cell Signaling |
| p-IκB α (Ser32) | 1:500 | Santa Cruz Biotechnology |
| SOD-1 | 1:1000 | Thermo Fisher |
| SOD-2 | 1:2000 | Santa Cruz Biotechnology |
| Primer | Forward | Reverse |
|---|---|---|
| Cat | 5′-ACTTTGAGGTCACCCACGAT-3′ | 5′-AACGGCAATAGGGGTCCTCTT-3′ |
| Gapdh | 5′-GGGTGTGAACCACGAGAAAT-3′ | 5′-CCTTCCACGATGCCAAAGTT-3′ |
| Gclc | 5′-GGCACAAGGACGTGCTCAAGT-3′ | 5′-TGCAGAGTTTCAAGAACATCG-3′ |
| IL-6 | 5′-GTTTGGAAGCATCCATCATTT-3′ | 5′-TGGAAATGAGAAAAGAGTTGTG-3′ |
| Ho-1 | 5′-AGCACAGGGTGACAGAAGAG-3′ | 5′-GAGGGACTCTGGTCTTTGTG-3′ |
| Nqo-1 | 5′-GGGGACATGAACGTCATTCTCT-3′ | 5′-AAGACCTGGAAGCCACAGAAGC-3′ |
| Sod-1 | 5′-GGGGACAATACACAAGGCTGTA-3′ | 5′-CAGGTCTCCAACATGCCTCT-3′ |
| Sod-2 | 5′-GTGGAGAACCCAAAGGAGAG-3′ | 5′-GAACCTTGGACTCCCACAGA-3′ |
| TNF-α | 5′-CCACCAGTTGGTTGTCTTTG-3′ | 5′-TAGCCCACGTCGTAGCAAAC-3′ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Estaras, M.; Gonzalez-Portillo, M.R.; Martinez, R.; Garcia, A.; Estevez, M.; Fernandez-Bermejo, M.; Mateos, J.M.; Vara, D.; Blanco-Fernández, G.; Lopez-Guerra, D.; et al. Melatonin Modulates the Antioxidant Defenses and the Expression of Proinflammatory Mediators in Pancreatic Stellate Cells Subjected to Hypoxia. Antioxidants 2021, 10, 577. https://doi.org/10.3390/antiox10040577
Estaras M, Gonzalez-Portillo MR, Martinez R, Garcia A, Estevez M, Fernandez-Bermejo M, Mateos JM, Vara D, Blanco-Fernández G, Lopez-Guerra D, et al. Melatonin Modulates the Antioxidant Defenses and the Expression of Proinflammatory Mediators in Pancreatic Stellate Cells Subjected to Hypoxia. Antioxidants. 2021; 10(4):577. https://doi.org/10.3390/antiox10040577
Chicago/Turabian StyleEstaras, Matias, Manuel R. Gonzalez-Portillo, Remigio Martinez, Alfredo Garcia, Mario Estevez, Miguel Fernandez-Bermejo, Jose M. Mateos, Daniel Vara, Gerardo Blanco-Fernández, Diego Lopez-Guerra, and et al. 2021. "Melatonin Modulates the Antioxidant Defenses and the Expression of Proinflammatory Mediators in Pancreatic Stellate Cells Subjected to Hypoxia" Antioxidants 10, no. 4: 577. https://doi.org/10.3390/antiox10040577
APA StyleEstaras, M., Gonzalez-Portillo, M. R., Martinez, R., Garcia, A., Estevez, M., Fernandez-Bermejo, M., Mateos, J. M., Vara, D., Blanco-Fernández, G., Lopez-Guerra, D., Roncero, V., Salido, G. M., & Gonzalez, A. (2021). Melatonin Modulates the Antioxidant Defenses and the Expression of Proinflammatory Mediators in Pancreatic Stellate Cells Subjected to Hypoxia. Antioxidants, 10(4), 577. https://doi.org/10.3390/antiox10040577






