Protein Lipoxidation: Basic Concepts and Emerging Roles
Abstract
1. Introduction
- Lipid Oxidation: an overall term encompassing both radical and non-radical (electrophilic) reactions and leading to an increase in the number of oxygens and other heteroatoms (such as nitrogen or chlorine) or a decrease in the hydrogen content of the lipid.
- Lipid Peroxidation: a specific form of radical attack, usually at bis-allylic sites in an unsaturated hydrocarbon chain, that leads first to a carbon-centred radical and then the addition of molecular oxygen to form a peroxyl radical (-O-O•) on that carbon. The peroxyl radical remains reactive and can abstract hydrogens from adjacent molecules, resulting in a chain reaction and propagation of damage.
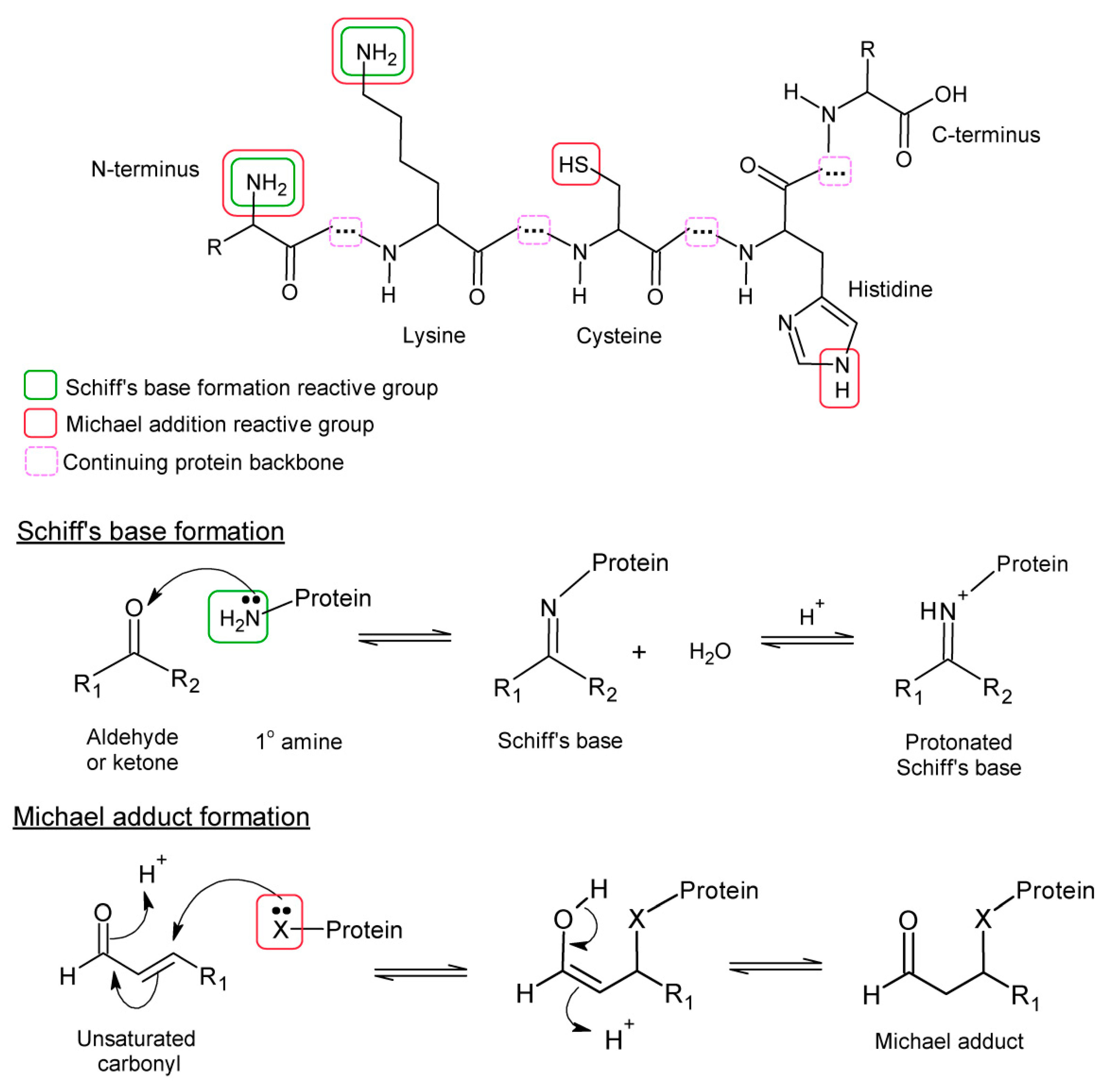
- Lipoxidation: covalent reaction of reactive and electrophilic lipid products, mostly arising from lipid oxidation, for example, aldehydes or α,β-unsaturated breakdown products such as acrolein and 4-hydrononenal, or cyclopentenone-containing lipids (e.g., 15-deoxy-Δ12,14-prostaglandin J2) with macromolecules. The targets of lipoxidation include proteins, DNA or head groups of phospholipids.
- Advanced Lipoxidation End-products (ALEs): the covalent adducts formed by the process of lipoxidation.
- Protein lipoxidation: the modification of proteins by electrophilic lipids. Although is not an oxidative modification per se, it frequently contributes to the damage to proteins under oxidative stress conditions.
- Protein lipidation: enzymatically-catalysed covalent modification of proteins by lipids, which usually enable the proteins to associate with membranes. Typical examples include N-myristoylation, S-palmitoylation, or S-prenylation, as well as the addition of a glycosylphosphatidylinositol anchor.
- Lipoproteins: particles formed by amphipathic proteins embedded in a phospholipid monolayer and surrounding an inner core of cholesterol, cholesterol esters and triacylglycerols. They function as lipid transporters and are commonly found in plasma.
2. Lipid Oxidation and Protein Lipoxidation
| Reactive Lipid Product | Type | Source | Reactions Reported With | Cross-Linking |
|---|---|---|---|---|
| Malondialdehyde | bis-aldehyde, isomerizes to β-hydroxy-acrolein | Polyunsaturated chains with ≥3 double bonds | Lys (Michael and Schiff’s) His, Arg, Cys (Michael) | √ |
| Acrolein | Alkenal (3 carbons) (α-β-unsaturated aldehyde) | Polyunsaturated lipids but also other environmental sources | Lys (Michael and Schiff’s) His, Cys (Michael) | √ |
| Crotonaldehyde | Alkenal (4 carbons) (α-β-unsaturated aldehyde) | ω-3 unsaturated lipids (α-linolenic, eicosapentaenoic or docosahexaenoic acid) | Lys (Michael and Schiff’s) His, Cys (Michael) | √ |
| 4-hydroxy-2- hexenal (HHE) | 4-hydroxy-alkenal (α-β-unsaturated aldehyde) | ω-3 polyunsaturated lipids (α-linolenic, eicosapentaenoic or docosahexaenoic acid) | Lys (Michael and Schiff’s) His, Cys (Michael) | √ |
| 4-hydroxy-2-nonenal (HNE) | 4-hydroxy-alkenal (α-β-unsaturated aldehyde) | ω-6 polyunsaturated lipids (γ-linolenic or arachidonic acid) | Lys (Michael and Schiff’s) His, Cys (Michael) | √ |
| 4-oxo-2-nonenal (ONE) | 4-oxo-alkenal (α-β-unsaturated aldehyde) | ω-6 polyunsaturated lipids (γ-linolenic or arachidonic acid) | Lys (Michael and Schiff’s) His, Cys (Michael) | √ |
| 15-deoxy-Δ12,14-prosta-glandin J2 (15d-PGJ2) | Cyclopentenone prostaglandin (cyPG) | Arachidonic acid | His, Cys (Michael) | √ |
| 15-keto-prostaglandin E2 | Prostaglandin | Arachidonic acid | Cys (Michael) | X |
| Palmitoyl-oxovaleroyl phosphatidylcholine (POVPC) | Esterified alkenal | ω-6 polyunsaturated lipids (γ-linolenic or arachidonic acid) | Lys (Michael and Schiff’s) His, Cys (Michael) | X |
| Palmitoyl-oxononanoyl phosphatidylcholine (PONPC) | Esterified alkenal | ω-6 polyunsaturated lipids (γ-linolenic or arachidonic acid) | Lys (Schiff’s base only) | X |
| Isolevuglandins (isoLGs) and Isoketals | γ-keto-aldehydes | Arachidonic acid and docosahexenoic acid | Lys (Schiff’s base only) | √ |
| Nitro-oleate and nitro-linoleate | Nitro-fatty acids (NO2-FAs) (can be esterified in PLs) | Unsaturated fatty acyl chains (e.g., oleoyl or linoleoyl) | Lys, His, Cys (Michael) (nitro-alkylation) | X |
| Chloro-hexadecanal or chloro-octadecanal | Chloro-fatty aldehydes | Plasmenyl phospholipids (palmitate or stearate attached by vinyl ether bond) | Lys (Schiff’s base only) | X |
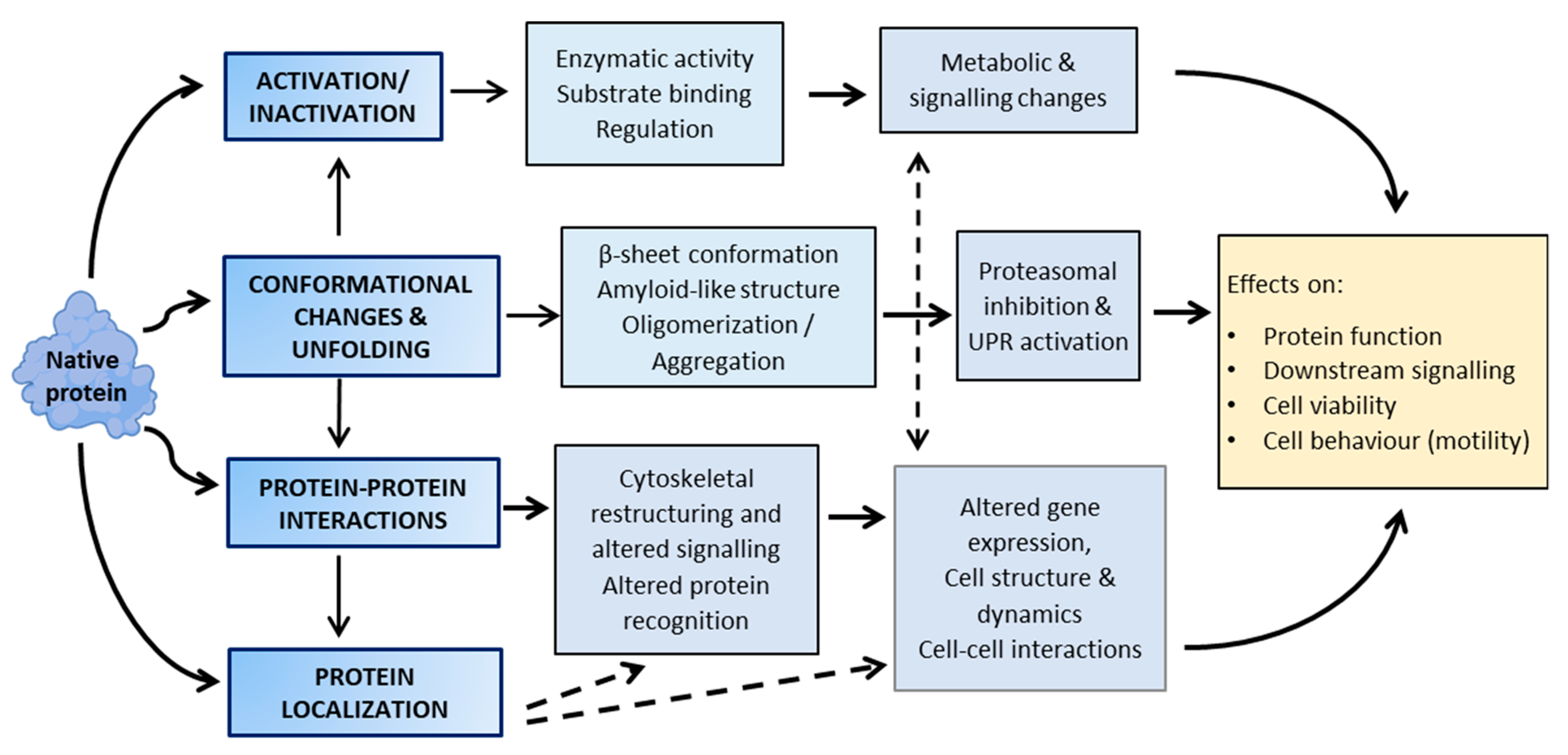
3. Functional Consequences of Lipoxidation
| Category | Protein | Lipid | Residue | Implication | Reference |
|---|---|---|---|---|---|
| Cytoskeletal protein | Vimentin | 15d-PGJ2, PGA1 HNE | Cys328 | Filament reorganisation | [73,74] [75,76] |
| GFAP | 15d-PGJ2, PGA1 | Cys294 | Filament reorganisation | [77] | |
| Actin | HNE PGA1 15d-PGJ2 Acrolein | Cys374 Cys374 Cys374, His87, His173 | Electrophilic scavenger, filament disruption | [78] [79] [80] [81] | |
| Tubulin | HNE | Cys295 | Filament reorganisation | [75] | |
| Metabolic enzymes | AKR1B1 AKR1B AKR1B10 | Acrolein HNE PGA1 | Cys298 Cys298 Cys299 | Activation Inhibition Inhibition | [56] [57] [82] |
| α-Enolase | HNE 15d-PGJ2 | ? * | Inhibition | [83] [80] | |
| Soluble epoxide hydrolase | 15d-PGJ2 | Cys521 | Inhibition | [84] | |
| Pyruvate kinase | 15d-PGJ2 Acrolein, HHE, MDA HNE, ONE | ? Cys152, Cys358, Cys423, Cys474 Cys424, His439 | ? Inhibition Inhibition | [74] [33] [85] | |
| Pin1 | HNE | Cys113 | Inhibition | [86] | |
| Chaperones | Hsp 90 | 15d-PGJ2 PGA1 HNE, ONE | ? Cys572 | Inhibition | [74] [73,87] [88] |
| Hsp 70 | cyPG HNE | ? Cys267 | Inhibition | [84,87] [89] | |
| Transcription factor | PPARγ | NO2-FAs 15d-PGJ2 | Cys285 Cys285 | Activation | [90] [91,92] |
| P53 | 15d-PGJ2 | Cys277 | Inhibition | [93] | |
| NF-κB | 15d-PGJ2, PGA1 | Cys38 (p65) and Cys62 (p50) | Inhibition | [94,95] | |
| STAT3 | 15-keto-PGE2 | Cys259 | Inhibition | [96] | |
| AP-1 | 15d-PGJ2 | Cys269 (c-Jun) | Inhibition | [97] | |
| Membrane receptor | Estrogen receptor α | 15d-PGJ2 | Cys227, Cys240 | Inhibition | [98] |
| EGFR | HNE | ? | Activation (low levels); inhibition (high levels) | [99] | |
| TRPA | 15d-PGJ2 | Cys421, Cys621 | Activation | [100] | |
| Regulatory proteins | Keap1 | NO2-FAs HNE 15d-PGJ2 | Cys151, 273, 288 Cys 273, Cys288 | Inhibition | [101,102] [103] [101,102,103,104] |
| IKK | HNE cyPG | ? Cys179 | Inhibition | [105] [106] | |
| H-Ras | 15d-PGJ2, PGA1 | Cys118, Cys181, Cys184 | Activation | [107,108] | |
| Signalling protein | PTEN | Acrolein, HNE, PGA2, 15d-PGJ2 | Cys71, Lys327 | Inhibition | [58,109] |
| Akt | HNE | His196, His267, Cys311 | Inhibition | [110] | |
| PP2A | HNE | ? | Inhibition | [60] | |
| Epigenetic regulation | Sirt2 | Acrolein, HNE | Cys482 | Inhibition | [111] |
| HDACs | Acrolein HNE, 15d-PGJ2 | Cys274 Cys 274 | Inhibition Inhibition | [112] [61] | |
| Mitochondrial proteins | DRP1 | 15d-PGJ2 | At least Cys644 | Fission inhibition | [113] |
| Cytochrome c | HNE | His196, His267, Lys87 | In vitro modification | [114] | |
| Aconitase | HNE | Cys99, Cys358, Cys421, Cys424, Cys565 | Inhibition | [115] | |
| Others | Albumin | Δ12-PGJ2 HNE, acrolein | His146 Cys34 | ? | [116] [117] |
4. Selectivity and Protein Targets of Lipoxidation
5. The Emerging Role of Lipoxidation in Cellular Regulation
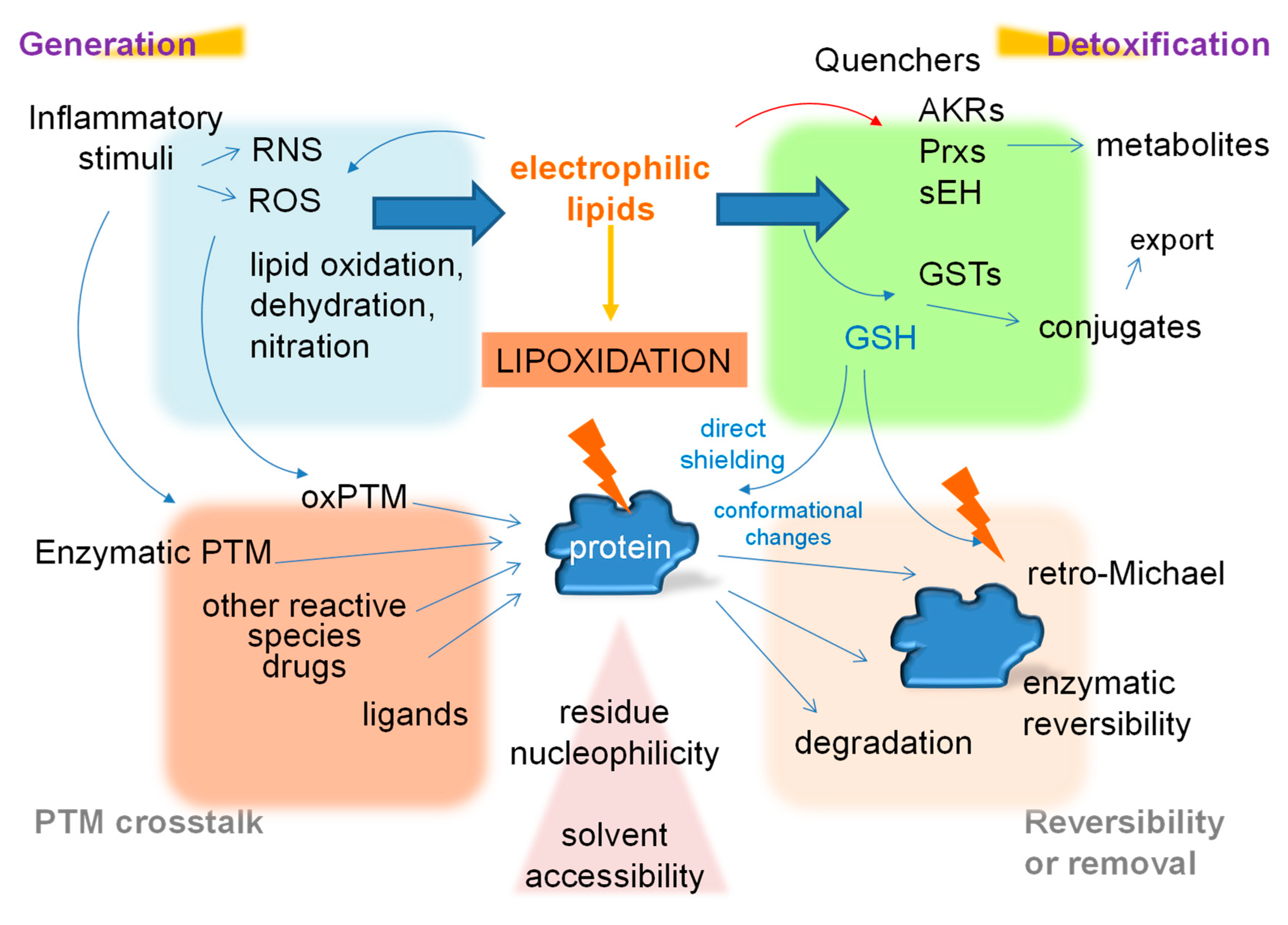
6. The Dependence of Lipoxidation on the Cellular Environment
7. Interplay among Post-Translational Modifications
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ACR | Alkenal/one reductase |
| ADH | Alcohol dehydrogenase |
| AER | Alkenal reductase |
| AKR | Aldo-keto reductase |
| AKR1B1 | Aldo-ketoreductase B1 |
| ALDH | Aldehyde dehydrogenase |
| ALDH2 | Aldehyde dehydrogenase 2 |
| ALEs | Advanced lipoxidation end products |
| ALH | Alkenal hydrogenase |
| ALR | Aldose/aldehyde reductase |
| CaMKII | Ca2+/calmodulin-dependent protein kinase II |
| COX | Cyclooxygenases |
| CRM1 | Chromosomal maintenance 1 |
| cyPG | Cyclopentenone prostaglandin(s) |
| CYP450 | Cytochrome P450 |
| FDP | Nε-(3-formyl-3,4-dehydropiperidino) |
| GAPDH | Glyceraldehyde-3-phosphate dehydrogenase |
| GFAP | Glial fibrillary acidic protein |
| GO | Gene ontology |
| GSH | Reduced glutathione |
| GST | Glutathione S-transferase |
| HDACs | Histone deacetylases |
| HNE | 4-hydroxynonenal |
| Hsp70 | Heat shock protein 70 |
| Hsp90 | Heat shock protein 90 |
| iNOS | Nitric oxide synthase |
| LOX | Lipoxygenases |
| MDA | Malondialdehyde |
| NF-κB | Nuclear factor-κB |
| NO2-FA | Nitrate fatty acid |
| NO2-POPC | Nitrated phospholipid 1-palmitoyl-2-oleyl-phosphatidylcholine |
| Nrf2 | Nuclear factor erythroid 2–related factor 2 |
| ONE | 4-oxononenal |
| oxo-ODE | Oxooctadecadienoic acid |
| PG | Prostaglandin(s) |
| PGA1 | Prostaglandin A1 |
| PGA2 | Prostaglandin A2 |
| PGD | Prostaglandin D |
| PGE2 | Prostaglandin E2 |
| PI3K | Phosphoinositide 3-kinase |
| PKA | Protein kinase A |
| PKC | Protein kinase C |
| PKG | Protein kinase G |
| PP1 | Protein phosphatase 1 |
| PP2A | Protein phosphatase 2 |
| PP2B | Protein phosphatase 2B |
| PPARγ | Peroxisome proliferator-activated receptor γ |
| Prx | Peroxiredoxin |
| PTEN | Phosphatidylinositol 3,4,5-trisphosphate 3-phosphatase |
| PTM | Post-translational modification |
| PUFAs | Polyunsaturated fatty acids |
| RAGE | Receptor of advanced glycation end products |
| RNS | Reactive nitrogen species |
| ROS | Reactive oxygen species |
| sEH | soluble epoxide hydrolase |
| SOD | Superoxide dismutase |
| STAT3 | Signal transducer and activator of transcription 3 |
| TCA | Tricarboxylic acid cycle |
| Trx | Thioredoxins |
| UPR | Unfolded protein response |
| 15d-PGJ2 | 15-deoxy-Δ12,14-prostaglandin J2 |
References
- Burdge, G.C.; Calder, P.C. Introduction to fatty acids and lipids. World Rev. Nutr. Diet. 2015, 112, 1–16. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, K.; Yang, L.; Liu, R.; Chu, Y.; Qin, X.; Yang, P.; Yu, H. Lipid metabolism in inflammation-related diseases. Analyst 2018, 143, 4526–4536. [Google Scholar] [CrossRef] [PubMed]
- Spickett, C.M. Formation of Oxidatively Modified Lipids as the Basis for a Cellular Epilipidome. Front. Endocrinol. 2020, 11, 602771. [Google Scholar] [CrossRef]
- Que, X.; Hung, M.Y.; Yeang, C.; Gonen, A.; Prohaska, T.A.; Sun, X.; Diehl, C.; Maatta, A.; Gaddis, D.E.; Bowden, K.; et al. Oxidized phospholipids are proinflammatory and proatherogenic in hypercholesterolaemic mice. Nature 2018, 558, 301–306. [Google Scholar] [CrossRef]
- Tomita, K.; Takashi, Y.; Ouchi, Y.; Kuwahara, Y.; Igarashi, K.; Nagasawa, T.; Nabika, H.; Kurimasa, A.; Fukumoto, M.; Nishitani, Y.; et al. Lipid peroxidation increases hydrogen peroxide permeability leading to cell death in cancer cell lines that lack mtDNA. Cancer Sci. 2019, 110, 2856–2866. [Google Scholar] [CrossRef] [PubMed]
- Hochstein, P.; Jain, S.K. Association of lipid peroxidation and polymerization of membrane proteins with erythrocyte aging. Fed. Proc. 1981, 40, 183–188. [Google Scholar] [PubMed]
- Fam, S.S.; Murphey, L.J.; Terry, E.S.; Zackert, W.E.; Chen, Y.; Gao, L.; Pandalai, S.; Milne, G.L.; Roberts, L.J.; Porter, N.A.; et al. Formation of highly reactive A-ring and J-ring isoprostane-like compounds (A4/J4-neuroprostanes) in vivo from docosahexaenoic acid. J. Biol. Chem. 2002, 277, 36076–36084. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, V.B.; Eiserich, J.P.; Chumley, P.H.; Jablonsky, M.J.; Krishna, N.R.; Kirk, M.; Barnes, S.; Darley-Usmar, V.M.; Freeman, B.A. Nitration of unsaturated fatty acids by nitric oxide-derived reactive nitrogen species peroxynitrite, nitrous acid, nitrogen dioxide, and nitronium ion. Chem. Res. Toxicol. 1999, 12, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Sun, Y.; Niu, J.; Jarugumilli, G.K.; Wu, X. Protein Lipidation in Cell Signaling and Diseases: Function, Regulation, and Therapeutic Opportunities. Cell Chem. Biol. 2018, 25, 817–831. [Google Scholar] [CrossRef] [PubMed]
- Hentschel, A.; Zahedi, R.P.; Ahrends, R. Protein lipid modifications--More than just a greasy ballast. Proteomics 2016, 16, 759–782. [Google Scholar] [CrossRef]
- Feingold, K.R. Introduction to Lipids and Lipoproteins. In Endotext; Feingold, K.R., Anawalt, B., Boyce, A., Chrousos, G., de Herder, W.W., Dungan, K., Grossman, A., Hershman, J.M., Hofland, J., Kaltsas, G., et al., Eds.; MDText.com. Inc.: South Dartmouth, MA, USA, 2000. [Google Scholar]
- Afonso, C.B.; Spickett, C.M. Lipoproteins as targets and markers of lipoxidation. Redox Biol. 2018, 101066. [Google Scholar] [CrossRef] [PubMed]
- De Bus, I.; Witkamp, R.; Zuilhof, H.; Albada, B.; Balvers, M. The role of n-3 PUFA-derived fatty acid derivatives and their oxygenated metabolites in the modulation of inflammation. Prostaglandins Lipid Mediat. 2019, 144, 106351. [Google Scholar] [CrossRef] [PubMed]
- Vigor, C.; Bertrand-Michel, J.; Pinot, E.; Oger, C.; Vercauteren, J.; Le Faouder, P.; Galano, J.M.; Lee, J.C.; Durand, T. Non-enzymatic lipid oxidation products in biological systems: Assessment of the metabolites from polyunsaturated fatty acids. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2014, 964, 65–78. [Google Scholar] [CrossRef]
- Malmsten, C.L. Prostaglandins, thromboxanes, and leukotrienes in inflammation. Am. J. Med. 1986, 80, 11–17. [Google Scholar] [CrossRef]
- Zhong, S.; Li, L.; Shen, X.; Li, Q.; Xu, W.; Wang, X.; Tao, Y.; Yin, H. An update on lipid oxidation and inflammation in cardiovascular diseases. Free Radic. Biol. Med. 2019, 144, 266–278. [Google Scholar] [CrossRef] [PubMed]
- Reis, A.; Spickett, C.M. Chemistry of phospholipid oxidation. Biochim. Biophys. Acta 2012, 1818, 2374–2387. [Google Scholar] [CrossRef]
- Galano, J.M.; Lee, J.C.; Gladine, C.; Comte, B.; Le Guennec, J.Y.; Oger, C.; Durand, T. Non-enzymatic cyclic oxygenated metabolites of adrenic, docosahexaenoic, eicosapentaenoic and alpha-linolenic acids; bioactivities and potential use as biomarkers. Biochim. Biophys. Acta 2015, 1851, 446–455. [Google Scholar] [CrossRef]
- Domingues, M.R.; Domingues, P.; Melo, T.; Pérez-Sala, D.; Reis, A.; Spickett, C. Lipoxidation adducts with peptides and proteins: Deleterious modifications or signalling mechanisms? J. Proteom. 2013, 92, 110–131. [Google Scholar] [CrossRef]
- Schneider, C.; Boeglin, W.E.; Yin, H.; Porter, N.A.; Brash, A.R. Intermolecular peroxyl radical reactions during autoxidation of hydroxy and hydroperoxy arachidonic acids generate a novel series of epoxidized products. Chem. Res. Toxicol. 2008, 21, 895–903. [Google Scholar] [CrossRef]
- Salomon, R.G.; Batyreva, E.; Kaur, K.; Sprecher, D.L.; Schreiber, M.J.; Crabb, J.W.; Penn, M.S.; DiCorletoe, A.M.; Hazen, S.L.; Podrez, E.A. Isolevuglandin-protein adducts in humans: Products of free radical-induced lipid oxidation through the isoprostane pathway. Biochim. Biophys. Acta 2000, 1485, 225–235. [Google Scholar] [CrossRef]
- Esterbauer, H.; Schaur, R.J.; Zollner, H. Chemistry and biochemistry of 4-hydroxynonenal, malonaldehyde and related aldehydes. Free Radic. Biol. Med. 1991, 11, 81–128. [Google Scholar] [CrossRef]
- Sousa, B.C.; Pitt, A.R.; Spickett, C.M. Chemistry and analysis of HNE and other prominent carbonyl-containing lipid oxidation compounds. Free Radic. Biol. Med. 2017, 111, 294–308. [Google Scholar] [CrossRef]
- Gueraud, F.; Atalay, M.; Bresgen, N.; Cipak, A.; Eckl, P.M.; Huc, L.; Jouanin, I.; Siems, W.; Uchida, K. Chemistry and biochemistry of lipid peroxidation products. Free Radic. Res. 2010, 44, 1098–1124. [Google Scholar] [CrossRef]
- Shibata, T.; Kondo, M.; Osawa, T.; Shibata, N.; Kobayashi, M.; Uchida, K. 15-Deoxy-Delta 12,14-prostaglandin J2. A PROSTAGLANDIN D2 METABOLITE GENERATED DURING INFLAMMATORY PROCESSES. J. Biol. Chem. 2002, 277, 10459–10466. [Google Scholar] [CrossRef]
- Uchida, K.; Shibata, T. 15-Deoxy-Delta(12,14)-prostaglandin J2: An electrophilic trigger of cellular responses. Chem. Res. Toxicol. 2008, 21, 138–144. [Google Scholar] [CrossRef]
- Stamatakis, K.; Pérez-Sala, D. Prostanoids with cyclopentenone structure as tools for the characterization of electrophilic eicosanoid-protein interactomes. Ann. N. Y. Acad. Sci. 2006, 1091, 548–570. [Google Scholar] [CrossRef]
- Hammond, V.J.; Morgan, A.H.; Lauder, S.; Thomas, C.P.; Brown, S.; Freeman, B.A.; Lloyd, C.M.; Davies, J.; Bush, A.; Levonen, A.L.; et al. Novel keto-phospholipids are generated by monocytes and macrophages, detected in cystic fibrosis, and activate peroxisome proliferator-activated receptor-gamma. J. Biol. Chem. 2012, 287, 41651–41666. [Google Scholar] [CrossRef]
- Schopfer, F.J.; Cipollina, C.; Freeman, B.A. Formation and signaling actions of electrophilic lipids. Chem. Rev. 2011, 111, 5997–6021. [Google Scholar] [CrossRef]
- Zhao, J.; Chen, J.; Zhu, H.; Xiong, Y.L. Mass spectrometric evidence of malonaldehyde and 4-hydroxynonenal adductions to radical-scavenging soy peptides. J. Agric. Food Chem. 2012, 60, 9727–9736. [Google Scholar] [CrossRef]
- Spickett, C.M.; Pitt, A.R. Modification of proteins by reactive lipid oxidation products and biochemical effects of lipoxidation. Essays Biochem. 2020, 64, 19–31. [Google Scholar] [CrossRef]
- Ayala, A.; Munoz, M.F.; Arguelles, S. Lipid peroxidation: Production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxid. Med. Cell. Longev. 2014, 2014, 360438. [Google Scholar] [CrossRef]
- Sousa, B.C.; Ahmed, T.; Dann, W.L.; Ashman, J.; Guy, A.; Durand, T.; Pitt, A.R.; Spickett, C.M. Short-chain lipid peroxidation products form covalent adducts with pyruvate kinase and inhibit its activity in vitro and in breast cancer cells. Free Radic. Biol. Med. 2019, 144, 223–233. [Google Scholar] [CrossRef]
- Liu-Snyder, P.; Borgens, R.B.; Shi, R. Hydralazine rescues PC12 cells from acrolein-mediated death. J. Neurosci. Res. 2006, 84, 219–227. [Google Scholar] [CrossRef]
- Niedernhofer, L.J.; Daniels, J.S.; Rouzer, C.A.; Greene, R.E.; Marnett, L.J. Malondialdehyde, a product of lipid peroxidation, is mutagenic in human cells. J. Biol. Chem. 2003, 278, 31426–31433. [Google Scholar] [CrossRef]
- Esterbauer, H.; Eckl, P.; Ortner, A. Possible mutagens derived from lipids and lipid precursors. Mutat Res. 1990, 238, 223–233. [Google Scholar] [CrossRef]
- Roberts, L.J., 2nd; Morrow, J.D. Products of the isoprostane pathway: Unique bioactive compounds and markers of lipid peroxidation. Cell. Mol. Life Sci. 2002, 59, 808–820. [Google Scholar] [CrossRef]
- Pérez-Sala, D. Electrophilic eicosanoids: Signaling and targets. Chem. Biol. Interact. 2011, 192, 96–100. [Google Scholar] [CrossRef]
- Schopfer, F.J.; Vitturi, D.A.; Jorkasky, D.K.; Freeman, B.A. Nitro-fatty acids: New drug candidates for chronic inflammatory and fibrotic diseases. Nitric Oxide 2018, 79, 31–37. [Google Scholar] [CrossRef]
- Salomon, R.G. Levuglandins and isolevuglandins: Stealthy toxins of oxidative injury. Antioxid. Redox Signal 2005, 7, 185–201. [Google Scholar] [CrossRef]
- Montuschi, P.; Barnes, P.J.; Roberts, L.J., 2nd. Isoprostanes: Markers and mediators of oxidative stress. FASEB J. 2004, 18, 1791–1800. [Google Scholar] [CrossRef]
- Martyniuk, C.J.; Fang, B.; Koomen, J.M.; Gavin, T.; Zhang, L.; Barber, D.S.; Lopachin, R.M. Molecular mechanism of glyceraldehyde-3-phosphate dehydrogenase inactivation by alpha,beta-unsaturated carbonyl derivatives. Chem. Res. Toxicol. 2011, 24, 2302–2311. [Google Scholar] [CrossRef]
- Sayre, L.M.; Lin, D.; Yuan, Q.; Zhu, X.; Tang, X. Protein adducts generated from products of lipid oxidation: Focus on HNE and one. Drug Metab. Rev. 2006, 38, 651–675. [Google Scholar] [CrossRef]
- Yamauchi, Y.; Sugimoto, Y. Effect of protein modification by malondialdehyde on the interaction between the oxygen-evolving complex 33 kDa protein and photosystem II core proteins. Planta 2010, 231, 1077–1088. [Google Scholar] [CrossRef]
- Mano, J.; Nagata, M.; Okamura, S.; Shiraya, T.; Mitsui, T. Identification of oxidatively modified proteins in salt-stressed Arabidopsis: A carbonyl-targeted proteomics approach. Plant Cell Physiol. 2014, 55, 1233–1244. [Google Scholar] [CrossRef]
- Alche, J.D. A concise appraisal of lipid oxidation and lipoxidation in higher plants. Redox Biol. 2019, 101136. [Google Scholar] [CrossRef]
- Mano, J.; Biswas, M.S.; Sugimoto, K. Reactive Carbonyl Species: A Missing Link in ROS Signaling. Plants 2019, 8, 391. [Google Scholar] [CrossRef]
- Belenky, P.; Ye, J.D.; Porter, C.B.; Cohen, N.R.; Lobritz, M.A.; Ferrante, T.; Jain, S.; Korry, B.J.; Schwarz, E.G.; Walker, G.C.; et al. Bactericidal Antibiotics Induce Toxic Metabolic Perturbations that Lead to Cellular Damage. Cell Rep. 2015, 13, 968–980. [Google Scholar] [CrossRef]
- Vistoli, G.; De Maddis, D.; Cipak, A.; Zarkovic, N.; Carini, M.; Aldini, G. Advanced glycoxidation and lipoxidation end products (AGEs and ALEs): An overview of their mechanisms of formation. Free Radic Res. 2013, 47, 3–27. [Google Scholar] [CrossRef]
- Pizzimenti, S.; Ciamporcero, E.; Daga, M.; Pettazzoni, P.; Arcaro, A.; Cetrangolo, G.; Minelli, R.; Dianzani, C.; Lepore, A.; Gentile, F.; et al. Interaction of aldehydes derived from lipid peroxidation and membrane proteins. Front. Physiol. 2013, 4, 242. [Google Scholar] [CrossRef] [PubMed]
- Oeste, C.L.; Pérez-Sala, D. Modification of cysteine residues by cyclopentenone prostaglandins: Interplay with redox regulation of protein function. Mass Spectrom. Rev. 2014, 33, 110–125. [Google Scholar] [CrossRef] [PubMed]
- Elguero, B.; Gonilski Pacin, D.; Cardenas Figueroa, C.; Fuertes, M.; Arzt, E. Modifications in the cellular proteome and their clinical application. Medicina 2019, 79, 570–575. [Google Scholar]
- Perez-Sala, D.; Domingues, R. Lipoxidation targets: From basic mechanisms to pathophysiology. Redox Biol. 2019, 23, 101208. [Google Scholar] [CrossRef]
- Aluise, C.D.; Camarillo, J.M.; Shimozu, Y.; Galligan, J.J.; Rose, K.L.; Tallman, K.A.; Marnett, L.J. Site-Specific, Intramolecular Cross-Linking of Pin1 Active Site Residues by the Lipid Electrophile 4-Oxo-2-nonenal. Chem. Res. Toxicol. 2015, 28, 817–827. [Google Scholar] [CrossRef]
- Doorn, J.A.; Hurley, T.D.; Petersen, D.R. Inhibition of human mitochondrial aldehyde dehydrogenase by 4-hydroxynon-2-enal and 4-oxonon-2-enal. Chem. Res. Toxicol. 2006, 19, 102–110. [Google Scholar] [CrossRef]
- Srivastava, S.K.; Ramana, K.V.; Chandra, D.; Srivastava, S.; Bhatnagar, A. Regulation of aldose reductase and the polyol pathway activity by nitric oxide. Chem. Biol. Interact. 2003, 143–144, 333–340. [Google Scholar] [CrossRef]
- Del Corso, A.; Dal Monte, M.; Vilardo, P.G.; Cecconi, I.; Moschini, R.; Banditelli, S.; Cappiello, M.; Tsai, L.; Mura, U. Site-specific inactivation of aldose reductase by 4-hydroxynonenal. Arch Biochem. Biophys. 1998, 350, 245–248. [Google Scholar] [CrossRef]
- Covey, T.M.; Edes, K.; Coombs, G.S.; Virshup, D.M.; Fitzpatrick, F.A. Alkylation of the tumor suppressor PTEN activates Akt and beta-catenin signaling: A mechanism linking inflammation and oxidative stress with cancer. PLoS ONE 2010, 5, e13545. [Google Scholar] [CrossRef] [PubMed]
- Shearn, C.T.; Smathers, R.L.; Stewart, B.J.; Fritz, K.S.; Galligan, J.J.; Hail, N., Jr.; Petersen, D.R. Phosphatase and tensin homolog deleted on chromosome 10 (PTEN) inhibition by 4-hydroxynonenal leads to increased Akt activation in hepatocytes. Mol. Pharm. 2011, 79, 941–952. [Google Scholar] [CrossRef] [PubMed]
- Di Domenico, F.; Tramutola, A.; Barone, E.; Lanzillotta, C.; Defever, O.; Arena, A.; Zuliani, I.; Foppoli, C.; Iavarone, F.; Vincenzoni, F.; et al. Restoration of aberrant mTOR signaling by intranasal rapamycin reduces oxidative damage: Focus on HNE-modified proteins in a mouse model of down syndrome. Redox Biol. 2019, 23, 101162. [Google Scholar] [CrossRef]
- Doyle, K.; Fitzpatrick, F.A. Redox signaling, alkylation (carbonylation) of conserved cysteines inactivates class I histone deacetylases 1, 2, and 3 and antagonizes their transcriptional repressor function. J. Biol. Chem. 2010, 285, 17417–17424. [Google Scholar] [CrossRef]
- Igarashi, K.; Uemura, T.; Kashiwagi, K. Acrolein toxicity at advanced age: Present and future. Amino. Acids 2018, 50, 217–228. [Google Scholar] [CrossRef]
- Moldogazieva, N.T.; Mokhosoev, I.M.; Mel’nikova, T.I.; Porozov, Y.B.; Terentiev, A.A. Oxidative Stress and Advanced Lipoxidation and Glycation End Products (ALEs and AGEs) in Aging and Age-Related Diseases. Oxid. Med. Cell. Longev. 2019, 2019, 3085756. [Google Scholar] [CrossRef]
- Koharudin, L.M.; Liu, H.; Di Maio, R.; Kodali, R.B.; Graham, S.H.; Gronenborn, A.M. Cyclopentenone prostaglandin-induced unfolding and aggregation of the Parkinson disease-associated UCH-L1. Proc. Natl. Acad. Sci. USA 2010, 107, 6835–6840. [Google Scholar] [CrossRef]
- Sánchez-Gómez, F.J.; Díez-Dacal, B.; Pajares, M.A.; Llorca, O.; Pérez-Sala, D. Cyclopentenone prostaglandins with dienone structure promote cross-linking of the chemoresistance-inducing enzyme Glutathione Transferase P1-1. Mol. Pharm. 2010, 78, 723–733. [Google Scholar] [CrossRef]
- Miyata, T.; Kurokawa, K.; Van Ypersele De Strihou, C. Advanced glycation and lipoxidation end products: Role of reactive carbonyl compounds generated during carbohydrate and lipid metabolism. J. Am. Soc. Nephrol. 2000, 11, 1744–1752. [Google Scholar]
- Takasugi, N.; Hiraoka, H.; Nakahara, K.; Akiyama, S.; Fujikawa, K.; Nomura, R.; Furuichi, M.; Uehara, T. The Emerging Role of Electrophiles as a Key Regulator for Endoplasmic Reticulum (ER) Stress. Int. J. Mol. Sci. 2019, 20, 1783. [Google Scholar] [CrossRef] [PubMed]
- West, J.D.; Marnett, L.J. Alterations in gene expression induced by the lipid peroxidation product, 4-hydroxy-2-nonenal. Chem. Res. Toxicol. 2005, 18, 1642–1653. [Google Scholar] [CrossRef] [PubMed]
- Vladykovskaya, E.; Sithu, S.D.; Haberzettl, P.; Wickramasinghe, N.S.; Merchant, M.L.; Hill, B.G.; McCracken, J.; Agarwal, A.; Dougherty, S.; Gordon, S.A.; et al. Lipid peroxidation product 4-hydroxy-trans-2-nonenal causes endothelial activation by inducing endoplasmic reticulum stress. J. Biol. Chem. 2012, 287, 11398–11409. [Google Scholar] [CrossRef]
- Haberzettl, P.; Hill, B.G. Oxidized lipids activate autophagy in a JNK-dependent manner by stimulating the endoplasmic reticulum stress response. Redox Biol. 2013, 1, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Shringarpure, R.; Grune, T.; Sitte, N.; Davies, K.J. 4-Hydroxynonenal-modified amyloid-beta peptide inhibits the proteasome: Possible importance in Alzheimer’s disease. Cell. Mol. Life Sci. 2000, 57, 1802–1809. [Google Scholar] [CrossRef]
- Santra, M.; Dill, K.A.; de Graff, A.M.R. Proteostasis collapse is a driver of cell aging and death. Proc. Natl. Acad. Sci. USA 2019, 116, 22173–22178. [Google Scholar] [CrossRef]
- Gharbi, S.; Garzón, B.; Gayarre, J.; Timms, J.; Pérez-Sala, D. Study of protein targets for covalent modification by the antitumoral and anti-inflammatory prostaglandin PGA1: Focus on vimentin. J. Mass Spectrom. 2007, 42, 1474–1484. [Google Scholar] [CrossRef] [PubMed]
- Stamatakis, K.; Sánchez-Gómez, F.J.; Pérez-Sala, D. Identification of novel protein targets for modification by 15-deoxy-Δ12,14-prostaglandin J2 in mesangial cells reveals multiple interactions with the cytoskeleton. J. Am. Soc. Nephrol. 2006, 17, 89–98. [Google Scholar] [CrossRef]
- Chavez, J.; Chung, W.G.; Miranda, C.L.; Singhal, M.; Stevens, J.F.; Maier, C.S. Site-specific protein adducts of 4-hydroxy-2(E)-nonenal in human THP-1 monocytic cells: Protein carbonylation is diminished by ascorbic acid. Chem. Res. Toxicol. 2010, 23, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Griesser, E.; Vemula, V.; Mónico, A.; Pérez-Sala, D.; Fedorova, M. Dynamic posttranslational modifications of cytoskeletal proteins unveil hot spots under nitroxidative stress. BioRxiv 2021, 427971. [Google Scholar] [CrossRef]
- Viedma-Poyatos, Á.; Pablo, Y.d.; Pekny, M.; Pérez-Sala, D. The cysteine residue of glial fibrillary acidic protein is a critical target for lipoxidation and required for efficient network organization. Free Rad. Biol. Med. 2018, 120, 380–394. [Google Scholar] [CrossRef] [PubMed]
- Aldini, G.; Dalle-Donne, I.; Vistoli, G.; Maffei Facino, R.; Carini, M. Covalent modification of actin by 4-hydroxy-trans-2-nonenal (HNE): LC-ESI-MS/MS evidence for Cys374 Michael adduction. J. Mass Spectrom. 2005, 40, 946–954. [Google Scholar] [CrossRef]
- Gayarre, J.; Sánchez, D.; Sánchez-Gómez, F.J.; Terrón, C.; Llorca, O.; Pérez-Sala, D. Addition of electrophilic lipids to actin alters filament structure. Biochem. Biophys. Res. Commun. 2006, 349, 1387–1393. [Google Scholar] [CrossRef]
- Aldini, G.; Carini, M.; Vistoli, G.; Shibata, T.; Kusano, Y.; Gamberoni, L.; Dalle-Donne, I.; Milzani, A.; Uchida, K. Identification of Actin as a 15-Deoxy-Delta(12,14)-prostaglandin J(2) Target in Neuroblastoma Cells: Mass Spectrometric, Computational, and Functional Approaches To Investigate the Effect on Cytoskeletal Derangement. Biochemistry 2007, 46, 2707–2718. [Google Scholar] [CrossRef]
- Dalle-Donne, I.; Carini, M.; Vistoli, G.; Gamberoni, L.; Giustarini, D.; Colombo, R.; Maffei Facino, R.; Rossi, R.; Milzani, A.; Aldini, G. Actin Cys374 as a nucleophilic target of alpha,beta-unsaturated aldehydes. Free Radic. Biol. Med. 2007, 42, 583–598. [Google Scholar] [CrossRef]
- Díez-Dacal, B.; Gayarre, J.; Gharbi, S.; Timms, J.F.; Coderch, C.; Gago, F.; Pérez-Sala, D. Identification of aldo-keto reductase AKR1B10 as a selective target for modification and inhibition by PGA1: Implications for anti-tumoral activity. Cancer Res. 2011, 71, 4161–4171. [Google Scholar] [CrossRef]
- Reed, T.; Perluigi, M.; Sultana, R.; Pierce, W.M.; Klein, J.B.; Turner, D.M.; Coccia, R.; Markesbery, W.R.; Butterfield, D.A. Redox proteomic identification of 4-hydroxy-2-nonenal-modified brain proteins in amnestic mild cognitive impairment: Insight into the role of lipid peroxidation in the progression and pathogenesis of Alzheimer’s disease. Neurobiol Dis. 2008, 30, 107–120. [Google Scholar] [CrossRef] [PubMed]
- Charles, R.L.; Burgoyne, J.R.; Mayr, M.; Weldon, S.M.; Hubner, N.; Dong, H.; Morisseau, C.; Hammock, B.D.; Landar, A.; Eaton, P. Redox regulation of soluble epoxide hydrolase by 15-deoxy-delta-prostaglandin J2 controls coronary hypoxic vasodilation. Circ. Res. 2011, 108, 324–334. [Google Scholar] [CrossRef] [PubMed]
- Camarillo, J.M.; Ullery, J.C.; Rose, K.L.; Marnett, L.J. Electrophilic Modification of PKM2 by 4-Hydroxynonenal and 4-Oxononenal Results in Protein Cross-Linking and Kinase Inhibition. Chem. Res. Toxicol. 2017, 30, 635–641. [Google Scholar] [CrossRef]
- Aluise, C.D.; Rose, K.; Boiani, M.; Reyzer, M.L.; Manna, J.D.; Tallman, K.; Porter, N.A.; Marnett, L.J. Peptidyl-prolyl cis/trans-isomerase A1 (Pin1) is a target for modification by lipid electrophiles. Chem. Res. Toxicol. 2013, 26, 270–279. [Google Scholar] [CrossRef]
- Garzón, B.; Gayarre, J.; Gharbi, S.; Díez-Dacal, B.; Sánchez-Gómez, F.J.; Timms, J.F.; Pérez-Sala, D. A biotinylated analog of the anti-proliferative prostaglandin A1 allows assessment of PPAR-independent effects and identification of novel cellular targets for covalent modification. Chem. Biol. Interact. 2010, 183, 212–221. [Google Scholar] [CrossRef]
- Carbone, D.L.; Doorn, J.A.; Kiebler, Z.; Ickes, B.R.; Petersen, D.R. Modification of heat shock protein 90 by 4-hydroxynonenal in a rat model of chronic alcoholic liver disease. J. Pharm. Exp. 2005, 315, 8–15. [Google Scholar] [CrossRef]
- Carbone, D.L.; Doorn, J.A.; Kiebler, Z.; Sampey, B.P.; Petersen, D.R. Inhibition of Hsp72-mediated protein refolding by 4-hydroxy-2-nonenal. Chem. Res. Toxicol. 2004, 17, 1459–1467. [Google Scholar] [CrossRef]
- Schopfer, F.J.; Cole, M.P.; Groeger, A.L.; Chen, C.S.; Khoo, N.K.; Woodcock, S.R.; Golin-Bisello, F.; Motanya, U.N.; Li, Y.; Zhang, J.; et al. Covalent peroxisome proliferator-activated receptor gamma adduction by nitro-fatty acids: Selective ligand activity and anti-diabetic signaling actions. J. Biol. Chem. 2010, 285, 12321–12333. [Google Scholar] [CrossRef]
- Shiraki, T.; Kamiya, N.; Shiki, S.; Kodama, T.S.; Kakizuka, A.; Jingami, H. a,ß-Unsaturated Ketone is a Core Moiety of Natural Ligands for Covalent Binding to Peroxisome proliferator-activated receptor. J. Biol. Chem. 2005, 280, 14145–14153. [Google Scholar] [CrossRef]
- Itoh, T.; Fairall, L.; Amin, K.; Inaba, Y.; Szanto, A.; Balint, B.L.; Nagy, L.; Yamamoto, K.; Schwabe, J.W. Structural basis for the activation of PPARgamma by oxidized fatty acids. Nat. Struct. Mol. Biol. 2008, 15, 924–931. [Google Scholar] [CrossRef]
- Kim, D.H.; Kim, E.H.; Na, H.K.; Sun, Y.; Surh, Y.J. 15-Deoxy-Delta(12,14)-prostaglandin J(2) stabilizes, but functionally inactivates p53 by binding to the cysteine 277 residue. Oncogene 2010, 29, 2560–2576. [Google Scholar] [CrossRef]
- Cernuda-Morollón, E.; Pineda-Molina, E.; Cañada, F.J.; Pérez-Sala, D. 15-Deoxy-Δ12,14-prostaglandin J2 inhibition of NF-κB DNA binding through covalent modification of the p50 subunit. J. Biol. Chem. 2001, 276, 35530–35536. [Google Scholar] [CrossRef]
- Straus, D.S.; Pascual, G.; Li, M.; Welch, J.S.; Ricote, M.; Hsiang, C.H.; Sengchanthalangsy, L.L.; Ghosh, G.; Glass, C.K. 15-deoxy-delta 12,14-prostaglandin J2 inhibits multiple steps in the NF- kappa B signaling pathway. Proc. Natl. Acad. Sci. USA 2000, 97, 4844–4849. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.J.; Kim, S.-J.; Hahn, Y.-I.; Yoon, H.-J.; Hane, B.; Kim, K.; Lee, S.; Kim, K.P.; Suh, Y.G.; Na, H.-K.; et al. 15-Keto prostaglandin E2 suppresses STAT3 signaling and inhibits breast cancer cell growth and progression. Redox Biol. 2019, 23, 101175. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Sala, D.; Cernuda-Morollón, E.; Cañada, F.J. Molecular basis for the inhibition of AP-1 DNA binding by 15-deoxy-Δ12,14-prostaglandin J2. J. Biol. Chem. 2003, 278, 51251–51260. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Kim, J.Y.; Meng, Z.; Wang, L.H.; Liu, F.; Conrads, T.P.; Burke, T.R.; Veenstra, T.D.; Farrar, W.L. 15-deoxy-Delta12,14-prostaglandin J2 inhibits transcriptional activity of estrogen receptor-alpha via covalent modification of DNA-binding domain. Cancer Res. 2007, 67, 2595–2602. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Akhand, A.A.; Kato, M.; Yokoyama, I.; Miyata, T.; Kurokawa, K.; Uchida, K.; Nakashima, I. 4-hydroxynonenal triggers an epidermal growth factor receptor-linked signal pathway for growth inhibition. J. Cell Sci. 1999, 112 Pt 14, 2409–2417. [Google Scholar]
- Takahashi, N.; Mizuno, Y.; Kozai, D.; Yamamoto, S.; Kiyonaka, S.; Shibata, T.; Uchida, K.; Mori, Y. Molecular characterization of TRPA1 channel activation by cysteine-reactive inflammatory mediators. Channels 2008, 2, 287–298. [Google Scholar] [CrossRef]
- Tsujita, T.; Li, L.; Nakajima, H.; Iwamoto, N.; Nakajima-Takagi, Y.; Ohashi, K.; Kawakami, K.; Kumagai, Y.; Freeman, B.A.; Yamamoto, M.; et al. Nitro-fatty acids and cyclopentenone prostaglandins share strategies to activate the Keap1-Nrf2 system: A study using green fluorescent protein transgenic zebrafish. Genes Cells 2011, 16, 46–57. [Google Scholar] [CrossRef]
- Oh, J.Y.; Giles, N.; Landar, A.; Darley-Usmar, V. Accumulation of 15-deoxy-delta(12,14)-prostaglandin J2 adduct formation with Keap1 over time: Effects on potency for intracellular antioxidant defence induction. Biochem. J. 2008, 411, 297–306. [Google Scholar] [CrossRef]
- Suzuki, T.; Muramatsu, A.; Saito, R.; Iso, T.; Shibata, T.; Kuwata, K.; Kawaguchi, S.I.; Iwawaki, T.; Adachi, S.; Suda, H.; et al. Molecular Mechanism of Cellular Oxidative Stress Sensing by Keap1. Cell Rep. 2019, 28, 746–758. [Google Scholar] [CrossRef]
- Kobayashi, M.; Li, L.; Iwamoto, N.; Nakajima-Takagi, Y.; Kaneko, H.; Nakayama, Y.; Eguchi, M.; Wada, Y.; Kumagai, Y.; Yamamoto, M. The antioxidant defense system Keap1-Nrf2 comprises a multiple sensing mechanism for responding to a wide range of chemical compounds. Mol. Cell. Biol. 2009, 29, 493–502. [Google Scholar] [CrossRef] [PubMed]
- Ji, C.; Kozak, K.R.; Marnett, L.J. IkappaB kinase, a molecular target for inhibition by 4-hydroxy-2-nonenal. J. Biol. Chem. 2001, 276, 18223–18228. [Google Scholar] [CrossRef]
- Rossi, A.; Kapahi, P.; Natoli, G.; Takahashi, T.; Chen, Y.; Karin, M.; Santoro, M.G. Anti-inflammatory cyclopentenone prostaglandins are direct inhibitors of IκB kinase. Nature 2000, 403, 103–108. [Google Scholar] [CrossRef]
- Oliva, J.L.; Pérez-Sala, D.; Castrillo, A.; Martínez, N.; Cañada, F.J.; Boscá, L.; Rojas, J.M. The cyclopentenone 15-deoxy-Δ12,14-prostaglandin J2 binds to and activates H-Ras. Proc. Natl. Acad. Sci. USA 2003, 100, 4772–4777. [Google Scholar] [CrossRef]
- Renedo, M.; Gayarre, J.; García-Domínguez, C.A.; Pérez-Rodríguez, A.; Prieto, A.; Cañada, F.J.; Rojas, J.M.; Pérez-Sala, D. Modification and activation of Ras proteins by electrophilic prostanoids with different structure are site-selective. Biochemistry 2007, 46, 6607–6616. [Google Scholar] [CrossRef] [PubMed]
- Shearn, C.T.; Smathers, R.L.; Backos, D.S.; Reigan, P.; Orlicky, D.J.; Petersen, D.R. Increased carbonylation of the lipid phosphatase PTEN contributes to Akt2 activation in a murine model of early alcohol-induced steatosis. Free Radic. Biol. Med. 2013, 65, 680–692. [Google Scholar] [CrossRef]
- Shearn, C.T.; Fritz, K.S.; Reigan, P.; Petersen, D.R. Modification of Akt2 by 4-hydroxynonenal inhibits insulin-dependent Akt signaling in HepG2 cells. Biochemistry 2011, 50, 3984–3996. [Google Scholar] [CrossRef]
- Caito, S.; Rajendrasozhan, S.; Cook, S.; Chung, S.; Yao, H.; Friedman, A.E.; Brookes, P.S.; Rahman, I. SIRT1 is a redox-sensitive deacetylase that is post-translationally modified by oxidants and carbonyl stress. FASEB J. 2010, 24, 3145–3159. [Google Scholar] [CrossRef] [PubMed]
- Randall, M.J.; Haenen, G.R.; Bouwman, F.G.; van der Vliet, A.; Bast, A. The tobacco smoke component acrolein induces glucocorticoid resistant gene expression via inhibition of histone deacetylase. Toxicol. Lett. 2016, 240, 43–49. [Google Scholar] [CrossRef]
- Mishra, N.; Kar, R.; Singha, P.K.; Venkatachalam, M.A.; McEwen, D.G.; Saikumar, P. Inhibition of mitochondrial division through covalent modification of Drp1 protein by 15 deoxy-Delta(12,14)-prostaglandin J2. Biochem. Biophys. Res. Commun. 2010, 395, 17–24. [Google Scholar] [CrossRef]
- Isom, A.L.; Barnes, S.; Wilson, L.; Kirk, M.; Coward, L.; Darley-Usmar, V. Modification of Cytochrome c by 4-hydroxy- 2-nonenal: Evidence for histidine, lysine, and arginine-aldehyde adducts. J. Am. Soc. Mass Spectrom. 2004, 15, 1136–1147. [Google Scholar] [CrossRef]
- Liu, Q.; Simpson, D.C.; Gronert, S. Carbonylation of mitochondrial aconitase with 4-hydroxy-2-(E)-nonenal: Localization and relative reactivity of addition sites. Biochim. Biophys. Acta 2013, 1834, 1144–1154. [Google Scholar] [CrossRef]
- Yamaguchi, S.; Aldini, G.; Ito, S.; Morishita, N.; Shibata, T.; Vistoli, G.; Carini, M.; Uchida, K. Delta(12)-Prostaglandin J(2) as a Product and Ligand of Human Serum Albumin: Formation of an Unusual Covalent Adduct at His146. J. Am. Chem. Soc. 2010, 132, 824–832. [Google Scholar] [CrossRef] [PubMed]
- Aldini, G.; Regazzoni, L.; Orioli, M.; Rimoldi, I.; Facino, R.M.; Carini, M. A tandem MS precursor-ion scan approach to identify variable covalent modification of albumin Cys34: A new tool for studying vascular carbonylation. J. Mass Spectrom. 2008, 43, 1470–1481. [Google Scholar] [CrossRef]
- Ricote, M.; Li, A.C.; Willson, T.M.; Kelly, C.J.; Glass, C.K. The peroxisome proliferator-activated receptor-γ is a negative regulator of macrophage activation. Nature 1998, 391, 79–82. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Pascual, G.; Glass, C.K. Peroxisome proliferator-activated receptor gamma-dependent repression of the inducible nitric oxide synthase gene. Mol. Cell. Biol. 2000, 20, 4699–4707. [Google Scholar] [CrossRef]
- Waku, T.; Shiraki, T.; Oyama, T.; Fujimoto, Y.; Maebara, K.; Kamiya, N.; Jingami, H.; Morikawa, K. Structural insight into PPARgamma activation through covalent modification with endogenous fatty acids. J. Mol. Biol. 2009, 385, 188–199. [Google Scholar] [CrossRef]
- Zhang, D.D. The Nrf2-Keap1-ARE signaling pathway: The regulation and dual function of Nrf2 in cancer. Antioxid. Redox Signal. 2010, 13, 1623–1626. [Google Scholar] [CrossRef]
- Patinen, T.; Adinolfi, S.; Cortes, C.C.; Harkonen, J.; Jawahar Deen, A.; Levonen, A.L. Regulation of stress signaling pathways by protein lipoxidation. Redox Biol. 2019, 101114. [Google Scholar] [CrossRef]
- Mónico, A.; Duarte, S.; Pajares, M.A.; Pérez-Sala, D. Vimentin disruption by lipoxidation and electrophiles: Role of the cysteine residue and filament dynamics. Redox Biol. 2019, 23, 101098. [Google Scholar] [CrossRef]
- Mol, M.; Degani, G.; Coppa, C.; Baron, G.; Popolo, L.; Carini, M.; Aldini, G.; Vistoli, G.; Altomare, A. Advanced lipoxidation end products (ALEs) as RAGE binders: Mass spectrometric and computational studies to explain the reasons why. Redox Biol. 2019, 23, 101083. [Google Scholar] [CrossRef]
- Oeste, C.L.; Díez-Dacal, B.; Bray, F.; García de Lacoba, M.; de la Torre, B.G.; Andreu, D.; Ruiz-Sánchez, A.J.; Pérez-Inestrosa, E.; García-Domínguez, C.A.; Rojas, J.M.; et al. The C-terminus of H-Ras as a target for the covalent binding of reactive compounds modulating Ras-dependent pathways. PLoS ONE 2011, 6, e15866. [Google Scholar] [CrossRef]
- Hilliard, M.; Frohnert, C.; Spillner, C.; Marcone, S.; Nath, A.; Lampe, T.; Fitzgerald, D.; Kehlenbach, R.H. The anti-inflammatory prostaglandin 15-Deoxy-{Delta}12,14 PGJ2 inhibits CRM1-dependent nuclear protein export. J. Biol. Chem. 2010, 285, 22202–22210. [Google Scholar] [CrossRef] [PubMed]
- Jove, M.; Mota-Martorell, N.; Pradas, I.; Martin-Gari, M.; Ayala, V.; Pamplona, R. The Advanced Lipoxidation End-Product Malondialdehyde-Lysine in Aging and Longevity. Antioxidants 2020, 9, 1132. [Google Scholar] [CrossRef]
- Gesslbauer, B.; Kuerzl, D.; Valpatic, N.; Bochkov, V.N. Unbiased Identification of Proteins Covalently Modified by Complex Mixtures of Peroxidized Lipids Using a Combination of Electrophoretic Mobility Band Shift with Mass Spectrometry. Antioxidants 2018, 7, 116. [Google Scholar] [CrossRef] [PubMed]
- Aldini, G.; Domingues, M.R.; Spickett, C.M.; Domingues, P.; Altomare, A.; Sánchez-Gómez, F.J.; Oeste, C.L.; Pérez-Sala, D. Protein lipoxidation: Detection strategies and challenges. Redox Biol. 2015, 5, 253–266. [Google Scholar] [CrossRef]
- Kansanen, E.; Jyrkkanen, H.K.; Levonen, A.L. Activation of stress signaling pathways by electrophilic oxidized and nitrated lipids. Free Radic. Biol. Med. 2012, 52, 973–982. [Google Scholar] [CrossRef]
- Chen, Y.; Liu, Y.; Hou, X.; Ye, Z.; Wang, C. Quantitative and Site-Specific Chemoproteomic Profiling of Targets of Acrolein. Chem. Res. Toxicol. 2019, 32, 467–473. [Google Scholar] [CrossRef]
- Pamplona, R.; Borras, C.; Jove, M.; Pradas, I.; Ferrer, I.; Vina, J. Redox lipidomics to better understand brain aging and function. Free Radic. Biol. Med. 2019, 144, 310–321. [Google Scholar] [CrossRef]
- Dong, Y.; Noda, K.; Murata, M.; Yoshida, S.; Saito, W.; Kanda, A.; Ishida, S. Localization of Acrolein-Lysine Adduct in Fibrovascular Tissues of Proliferative Diabetic Retinopathy. Curr. Eye Res. 2017, 42, 111–117. [Google Scholar] [CrossRef]
- Chavez, J.D.; Wu, J.; Bisson, W.; Maier, C.S. Site-specific proteomic analysis of lipoxidation adducts in cardiac mitochondria reveals chemical diversity of 2-alkenal adduction. J. Proteom. 2011, 74, 2417–2429. [Google Scholar] [CrossRef]
- Han, B.; Hare, M.; Wickramasekara, S.; Fang, Y.; Maier, C.S. A comparative ‘bottom up’ proteomics strategy for the site-specific identification and quantification of protein modifications by electrophilic lipids. J. Proteom. 2012, 75, 5724–5733. [Google Scholar] [CrossRef]
- Codreanu, S.G.; Ullery, J.C.; Zhu, J.; Tallman, K.A.; Beavers, W.N.; Porter, N.A.; Marnett, L.J.; Zhang, B.; Liebler, D.C. Alkylation damage by lipid electrophiles targets functional protein systems. Mol Cell Proteom. 2014, 13, 849–859. [Google Scholar] [CrossRef] [PubMed]
- Campos-Pinto, I.; Mendez, L.; Schouten, J.; Wilkins, J.; Fedorova, M.; Pitt, A.R.; Davis, P.; Spickett, C.M. Epitope mapping and characterization of 4-hydroxy-2-nonenal modified-human serum albumin using two different polyclonal antibodies. Free Radic. Biol. Med. 2019, 144, 234–244. [Google Scholar] [CrossRef]
- Ishii, T.; Ito, S.; Kumazawa, S.; Sakurai, T.; Yamaguchi, S.; Mori, T.; Nakayama, T.; Uchida, K. Site-specific modification of positively-charged surfaces on human serum albumin by malondialdehyde. Biochem. Biophys. Res. Commun. 2008, 371, 28–32. [Google Scholar] [CrossRef] [PubMed]
- Tran, T.N.; Kosaraju, M.G.; Tamamizu-Kato, S.; Akintunde, O.; Zheng, Y.; Bielicki, J.K.; Pinkerton, K.; Uchida, K.; Lee, Y.Y.; Narayanaswami, V. Acrolein modification impairs key functional features of rat apolipoprotein E: Identification of modified sites by mass spectrometry. Biochemistry 2014, 53, 361–375. [Google Scholar] [CrossRef] [PubMed]
- Eliuk, S.M.; Renfrow, M.B.; Shonsey, E.M.; Barnes, S.; Kim, H. Active site modifications of the brain isoform of creatine kinase by 4-hydroxy-2-nonenal correlate with reduced enzyme activity: Mapping of modified sites by Fourier transform-ion cyclotron resonance mass spectrometry. Chem. Res. Toxicol. 2007, 20, 1260–1268. [Google Scholar] [CrossRef]
- Vistoli, G.; Mantovani, C.; Gervasoni, S.; Pedretti, A.; Aldini, G. Key factors regulating protein carbonylation by alpha,beta unsaturated carbonyls: A structural study based on a retrospective meta-analysis. Biophys. Chem. 2017, 230, 20–26. [Google Scholar] [CrossRef]
- Chen, Y.; Cong, Y.; Quan, B.; Lan, T.; Chu, X.; Ye, Z.; Hou, X.; Wang, C. Chemoproteomic profiling of targets of lipid-derived electrophiles by bioorthogonal aminooxy probe. Redox Biol. 2017, 12, 712–718. [Google Scholar] [CrossRef] [PubMed]
- Wall, S.B.; Oh, J.Y.; Mitchell, L.; Laube, A.H.; Campbell, S.L.; Renfrow, M.B.; Landar, A. Rac1 modification by an electrophilic 15-deoxy Delta(12,14)-prostaglandin J2 analog. Redox Biol. 2015, 4, 346–354. [Google Scholar] [CrossRef]
- Roos, G.; Foloppe, N.; Messens, J. Understanding the pK(a) of redox cysteines: The key role of hydrogen bonding. Antioxid. Redox Signal. 2013, 18, 94–127. [Google Scholar] [CrossRef]
- Poole, L.B. The basics of thiols and cysteines in redox biology and chemistry. Free Radic. Biol. Med. 2015, 80, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Bhatnagar, A.; Pierce, W.M., Jr. Protein modification by acrolein: Formation and stability of cysteine adducts. Chem. Res. Toxicol. 2009, 22, 708–716. [Google Scholar] [CrossRef] [PubMed]
- Fisher, A.A.; Labenski, M.T.; Malladi, S.; Gokhale, V.; Bowen, M.E.; Milleron, R.S.; Bratton, S.B.; Monks, T.J.; Lau, S.S. Quinone electrophiles selectively adduct “electrophile binding motifs” within cytochrome c. Biochemistry 2007, 46, 11090–11100. [Google Scholar] [CrossRef] [PubMed]
- Stewart, M.D.; Igumenova, T.I. Reactive cysteine in the structural Zn(2+) site of the C1B domain from PKCalpha. Biochemistry 2012, 51, 7263–7277. [Google Scholar] [CrossRef][Green Version]
- Gayarre, J.; Stamatakis, K.; Renedo, M.; Pérez-Sala, D. Differential selectivity of protein modification by the cyclopentenone prostaglandins PGA1 and 15-deoxy-Δ12,14-PGJ2: Role of glutathione. FEBS Lett. 2005, 579, 5803–5808. [Google Scholar] [CrossRef] [PubMed]
- Rajakariar, R.; Hilliard, M.; Lawrence, T.; Trivedi, S.; Colville-Nash, P.; Bellingan, G.; Fitzgerald, D.; Yaqoob, M.M.; Gilroy, D.W. Hematopoietic prostaglandin D2 synthase controls the onset and resolution of acute inflammation through PGD2 and 15-deoxyDelta12 14 PGJ2. Proc. Natl. Acad. Sci. USA 2007, 104, 20979–20984. [Google Scholar] [CrossRef]
- Ong, W.Y.; Farooqui, T.; Kokotos, G.; Farooqui, A.A. Synthetic and natural inhibitors of phospholipases A2: Their importance for understanding and treatment of neurological disorders. ACS Chem. Neurosci. 2015, 6, 814–831. [Google Scholar] [CrossRef] [PubMed]
- Hawkey, C.J. Cyclooxygenase inhibition: Between the devil and the deep blue sea. Gut 2002, 50 (Suppl. 3), III25–III30. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Paumi, C.M.; Smitherman, P.K.; Townsend, A.J.; Morrow, C.S. Glutathione S-Transferases (GSTs) inhibit transcriptional activation by the peroxisomal proliferator-activated receptor γ (PPARγ) ligand, 15-deoxy-Δ12,14prostaglandin J2 (15-d-PGJ2). Biochemistry 2004, 43, 2345–2352. [Google Scholar] [CrossRef]
- Alary, J.; Gueraud, F.; Cravedi, J.P. Fate of 4-hydroxynonenal in vivo: Disposition and metabolic pathways. Mol. Asp. Med. 2003, 24, 177–187. [Google Scholar] [CrossRef]
- Hayes, J.D.; Flanagan, J.U.; Jowsey, I.R. GLUTATHIONE TRANSFERASES. Annu. Rev. Pharm. Toxicol. 2005, 45, 51–88. [Google Scholar] [CrossRef]
- Atsmon, J.; Freeman, M.L.; Meredith, M.J.; Sweetman, B.J.; Roberts, L.J., II. Conjugation of 9-deoxy-delta 9, delta 12(E)-prostaglandin D2 with intracellular glutathione and enhancement of its antiproliferative activity by glutathione depletion. Cancer Res. 1990, 50, 1879–1885. [Google Scholar] [PubMed]
- Bogaards, J.J.; Venekamp, J.C.; van Bladeren, P.J. Stereoselective conjugation of prostaglandin A2 and prostaglandin J2 with glutathione, catalyzed by the human glutathione S-transferases A1-1, A2-2, M1a-1a, and P1-1. Chem. Res. Toxicol. 1997, 10, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, K. Role of Soluble Epoxide Hydrolase in Metabolism of PUFAs in Psychiatric and Neurological Disorders. Front. Pharmacol. 2019, 10, 36. [Google Scholar] [CrossRef] [PubMed]
- Spite, M.; Baba, S.P.; Ahmed, Y.; Barski, O.A.; Nijhawan, K.; Petrash, J.M.; Bhatnagar, A.; Srivastava, S. Substrate specificity and catalytic efficiency of aldo-keto reductases with phospholipid aldehydes. Biochem. J. 2007, 405, 95–105. [Google Scholar] [CrossRef]
- Shen, Y.; Zhong, L.; Johnson, S.; Cao, D. Human aldo-keto reductases 1B1 and 1B10: A comparative study on their enzyme activity toward electrophilic carbonyl compounds. Chem. Biol. Interact. 2011, 191, 192–198. [Google Scholar] [CrossRef]
- Aldini, G.; Granata, P.; Orioli, M.; Santaniello, E.; Carini, M. Detoxification of 4-hydroxynonenal (HNE) in keratinocytes: Characterization of conjugated metabolites by liquid chromatography/electrospray ionization tandem mass spectrometry. J. Mass Spectrom. 2003, 38, 1160–1168. [Google Scholar] [CrossRef]
- Díez-Dacal, B.; Sánchez-Gómez, F.J.; Sánchez-Murcia, P.A.; Milackova, I.; Zimmerman, T.; Ballekova, J.; García-Martín, E.; Agúndez, J.A.G.; Gharbi, S.; Gago, F.; et al. Molecular interactions and implications of aldose reductase inhibition by PGA1 and clinically used prostaglandins. Mol. Pharm. 2016, 89, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Sun, R.; Fu, L.; Liu, K.; Tian, C.; Yang, Y.; Tallman, K.A.; Porter, N.A.; Liebler, D.C.; Yang, J. Chemoproteomics Reveals Chemical Diversity and Dynamics of 4-Oxo-2-nonenal Modifications in Cells. Mol. Cell. Proteom. 2017, 16, 1789–1800. [Google Scholar] [CrossRef]
- Lin, D.; Lee, H.G.; Liu, Q.; Perry, G.; Smith, M.A.; Sayre, L.M. 4-Oxo-2-nonenal is both more neurotoxic and more protein reactive than 4-hydroxy-2-nonenal. Chem. Res. Toxicol. 2005, 18, 1219–1231. [Google Scholar] [CrossRef] [PubMed]
- Randall, M.J.; Hristova, M.; van der Vliet, A. Protein alkylation by the alpha,beta-unsaturated aldehyde acrolein. A reversible mechanism of electrophile signaling? FEBS Lett. 2013, 587, 3808–3814. [Google Scholar] [CrossRef]
- Cui, Y.; Li, X.; Lin, J.; Hao, Q.; Li, X.D. Histone Ketoamide Adduction by 4-Oxo-2-nonenal Is a Reversible Posttranslational Modification Regulated by Sirt2. ACS Chem. Biol. 2017, 12, 47–51. [Google Scholar] [CrossRef]
- Jin, J.; He, B.; Zhang, X.; Lin, H.; Wang, Y. SIRT2 Reverses 4-Oxononanoyl Lysine Modification on Histones. J. Am. Chem. Soc. 2016, 138, 12304–12307. [Google Scholar] [CrossRef]
- Freeman, B.A.; O’Donnell, V.B.; Schopfer, F.J. The discovery of nitro-fatty acids as products of metabolic and inflammatory reactions and mediators of adaptive cell signaling. Nitric Oxide 2018, 77, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Khoo, N.K.H.; Schopfer, F.J. Nitrated fatty acids: From diet to disease. Curr. Opin. Physiol. 2019, 9, 67–72. [Google Scholar] [CrossRef]
- Aldini, G.; Vistoli, G.; Stefek, M.; Chondrogianni, N.; Grune, T.; Sereikaite, J.; Sadowska-Bartosz, I.; Bartosz, G. Molecular strategies to prevent, inhibit, and degrade advanced glycoxidation and advanced lipoxidation end products. Free Radic. Res. 2013, 47 (Suppl. 1), 93–137. [Google Scholar] [CrossRef] [PubMed]
- Hill, B.G.; Haberzettl, P.; Ahmed, Y.; Srivastava, S.; Bhatnagar, A. Unsaturated lipid peroxidation-derived aldehydes activate autophagy in vascular smooth-muscle cells. Biochem. J. 2008, 410, 525–534. [Google Scholar] [CrossRef]
- Díez-Dacal, B.; Pérez-Sala, D. Anti-inflammatory prostanoids: Focus on the interactions between electrophile signalling and resolution of inflammation. Sci. World J. 2010, 10, 655–675. [Google Scholar] [CrossRef]
- Nimse, S.B.; Pal, D. Free radicals, natural antioxidants, and their reaction mechanisms. RSC Adv. 2015, 5, 27986–28006. [Google Scholar] [CrossRef]
- Suzuki, M.; Mori, M.; Niwa, T.; Hirata, R.; Furuta, K.; Ishikawa, T.; Noyori, R. Chemical implications for antitumor and antiviral prostaglandins: Reaction of Δ7-prostaglandin A1 and prostaglandin A1 methyl esters with thiols. J. Am. Chem. Soc. 1997, 119, 2376–2385. [Google Scholar] [CrossRef]
- Goto, S.; Ihara, Y.; Urata, Y.; Izumi, S.; Abe, K.; Koji, T.; Kondo, T. Doxorubicin-induced DNA intercalation and scavenging by nuclear glutathione S-transferase pi. FASEB J. 2001, 15, 2702–2714. [Google Scholar] [CrossRef] [PubMed]
- Soh, Y.; Goto, S.; Kitajima, M.; Moriyama, S.; Kotera, K.; Nakayama, T.; Nakajima, H.; Kondo, T.; Ishimaru, T. Nuclear localisation of glutathione S-transferase pi is an evaluation factor for drug resistance in gynaecological cancers. Clin. Oncol. 2005, 17, 264–270. [Google Scholar] [CrossRef]
- Kondo, M.; Oya-Ito, T.; Kumagai, T.; Osawa, T.; Uchida, K. Cyclopentenone prostaglandins as potential inducers of intracellular oxidative stress. J. Biol. Chem. 2001, 276, 12076–12083. [Google Scholar] [CrossRef]
- Roede, J.R.; Carbone, D.L.; Doorn, J.A.; Kirichenko, O.V.; Reigan, P.; Petersen, D.R. In vitro and in silico characterization of peroxiredoxin 6 modified by 4-hydroxynonenal and 4-oxononenal. Chem. Res. Toxicol. 2008, 21, 2289–2299. [Google Scholar] [CrossRef] [PubMed]
- Go, Y.M.; Halvey, P.J.; Hansen, J.M.; Reed, M.; Pohl, J.; Jones, D.P. Reactive aldehyde modification of thioredoxin-1 activates early steps of inflammation and cell adhesion. Am. J. Pathol. 2007, 171, 1670–1681. [Google Scholar] [CrossRef]
- Shibata, T.; Yamada, T.; Ishii, T.; Kumazawa, S.; Nakamura, H.; Masutani, H.; Yodoi, J.; Uchida, K. Thioredoxin as a molecular target of cyclopentenone prostaglandins. J. Biol. Chem. 2003, 278, 26046–26054. [Google Scholar] [CrossRef] [PubMed]
- Moos, P.J.; Edes, K.; Cassidy, P.; Massuda, E.; Fitzpatrick, F.A. Electrophilic Prostaglandins and Lipid Aldehydes Repress Redox- sensitive Transcription Factors p53 and Hypoxia-inducible Factor by Impairing the Selenoprotein Thioredoxin Reductase. J. Biol. Chem. 2003, 278, 745–750. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.Y.; Appel, L.J.; Croft, K.D.; Miller, E.R., 3rd; Mori, T.A.; Puddey, I.B. Effects of vitamin C and vitamin E on in vivo lipid peroxidation: Results of a randomized controlled trial. Am. J. Clin. Nutr. 2002, 76, 549–555. [Google Scholar] [CrossRef]
- Kang, Z.; Li, H.; Li, G.; Yin, D. Reaction of pyridoxamine with malondialdehyde: Mechanism of inhibition of formation of advanced lipoxidation end-products. Amino Acids 2006, 30, 55–61. [Google Scholar] [CrossRef]
- Metz, T.O.; Alderson, N.L.; Chachich, M.E.; Thorpe, S.R.; Baynes, J.W. Pyridoxamine traps intermediates in lipid peroxidation reactions in vivo: Evidence on the role of lipids in chemical modification of protein and development of diabetic complications. J. Biol. Chem. 2003, 278, 42012–42019. [Google Scholar] [CrossRef] [PubMed]
- Gianazza, E.; Brioschi, M.; Fernandez, A.M.; Banfi, C. Lipoxidation in cardiovascular diseases. Redox Biol. 2019, 23, 101119. [Google Scholar] [CrossRef]
- Maret, W. The redox biology of redox-inert zinc ions. Free Radic. Biol. Med. 2019, 134, 311–326. [Google Scholar] [CrossRef] [PubMed]
- Pace, N.J.; Weerapana, E. A competitive chemical-proteomic platform to identify zinc-binding cysteines. ACS Chem. Biol. 2014, 9, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Sala, D.; Oeste, C.L.; Martínez, A.E.; Garzón, B.; Carrasco, M.J.; Cañada, F.J. Vimentin filament organization and stress sensing depend on its single cysteine residue and zinc binding. Nat. Commun. 2015, 6, 7287. [Google Scholar] [CrossRef] [PubMed]
- Hao, Q.; Maret, W. Aldehydes release zinc from proteins. A pathway from oxidative stress/lipid peroxidation to cellular functions of zinc. FEBS J. 2006, 273, 4300–4310. [Google Scholar] [CrossRef]
- Aldini, G.; Facino, R.M.; Beretta, G.; Carini, M. Carnosine and related dipeptides as quenchers of reactive carbonyl species: From structural studies to therapeutic perspectives. Biofactors 2005, 24, 77–87. [Google Scholar] [CrossRef]
- Anderson, E.J.; Vistoli, G.; Katunga, L.A.; Funai, K.; Regazzoni, L.; Monroe, T.B.; Gilardoni, E.; Cannizzaro, L.; Colzani, M.; De Maddis, D.; et al. A carnosine analog mitigates metabolic disorders of obesity by reducing carbonyl stress. J. Clin. Investig. 2018, 128, 5280–5293. [Google Scholar] [CrossRef]
- Davies, M.J. The oxidative environment and protein damage. Biochim. Biophys. Acta 2005, 1703, 93–109. [Google Scholar] [CrossRef] [PubMed]
- Hagglund, P.; Mariotti, M.; Davies, M.J. Identification and characterization of protein cross-links induced by oxidative reactions. Expert Rev. Proteom. 2018, 15, 665–681. [Google Scholar] [CrossRef]
- Hawkins, C.L.; Davies, M.J. Detection, identification, and quantification of oxidative protein modifications. J. Biol. Chem. 2019, 294, 19683–19708. [Google Scholar] [CrossRef]
- Lavergne, S.N.; Wang, H.; Callan, H.E.; Park, B.K.; Naisbitt, D.J. “Danger” conditions increase sulfamethoxazole-protein adduct formation in human antigen-presenting cells. J. Pharm. Exp. 2009, 331, 372–381. [Google Scholar] [CrossRef]
- Karimi, M.; Crossett, B.; Cordwell, S.J.; Pattison, D.I.; Davies, M.J. Characterization of disulfide (cystine) oxidation by HOCl in a model peptide: Evidence for oxygen addition, disulfide bond cleavage and adduct formation with thiols. Free Radic. Biol. Med. 2020, 154, 62–74. [Google Scholar] [CrossRef]
- Klatt, P.; Lamas, S. Regulation of protein function by S-glutathiolation in response to oxidative and nitrosative stress. Eur. J. Biochem. 2000, 267, 4928–4944. [Google Scholar] [CrossRef]
- Hill, B.G.; Bhatnagar, A. Role of glutathiolation in preservation, restoration and regulation of protein function. IUBMB Life 2007, 59, 21–26. [Google Scholar] [CrossRef]
- Viedma-Poyatos, A.; Pajares, M.A.; Pérez-Sala, D. Type III intermediate filaments as targets and effectors of electrophiles and oxidants. Redox Biol. 2020, 36, 101582. [Google Scholar] [CrossRef]
- Kaus-Drobek, M.; Mucke, N.; Szczepanowski, R.H.; Wedig, T.; Czarnocki-Cieciura, M.; Polakowska, M.; Herrmann, H.; Wyslouch-Cieszynska, A.; Dadlez, M. Vimentin S-glutathionylation at Cys328 inhibits filament elongation and induces severing of mature filaments in vitro. FEBS J. 2020. [Google Scholar] [CrossRef] [PubMed]
- Duarte, S.; Melo, T.; Domingues, R.; Alché, J.d.D.; Pérez-Sala, D. Insight into the cellular effects of nitrated phospholipids: Evidence for pleiotropic mechanisms of action. Free Rad. Biol. Med. 2019, 144, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Burgoyne, J.R.; Eaton, P. Oxidant sensing by protein kinases a and g enables integration of cell redox state with phosphoregulation. Sensors 2010, 10, 2731–2751. [Google Scholar] [CrossRef] [PubMed]
- Anderson, M.E. Oxidant stress promotes disease by activating CaMKII. J. Mol. Cell. Cardiol. 2015, 89, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Shearn, C.T.; Backos, D.S.; Orlicky, D.J.; Smathers-McCullough, R.L.; Petersen, D.R. Identification of 5’ AMP-activated kinase as a target of reactive aldehydes during chronic ingestion of high concentrations of ethanol. J. Biol. Chem. 2014, 289, 15449–15462. [Google Scholar] [CrossRef] [PubMed]
- Adler, V.; Yin, Z.; Fuchs, S.Y.; Benezra, M.; Rosario, L.; Tew, K.D.; Pincus, M.R.; Sardana, M.; Henderson, C.J.; Wolf, C.R.; et al. Regulation of JNK signaling by GSTp. EMBO J. 1999, 18, 1321–1334. [Google Scholar] [CrossRef]
- Thevenin, A.F.; Zony, C.L.; Bahnson, B.J.; Colman, R.F. GST pi modulates JNK activity through a direct interaction with JNK substrate, ATF2. Protein Sci. A Publ. Protein Soc. 2011, 20, 834–848. [Google Scholar] [CrossRef] [PubMed]
- De Luca, A.; Mei, G.; Rosato, N.; Nicolai, E.; Federici, L.; Palumbo, C.; Pastore, A.; Serra, M.; Caccuri, A.M. The fine-tuning of TRAF2-GSTP1-1 interaction: Effect of ligand binding and in situ detection of the complex. Cell Death Dis. 2014, 5, e1015. [Google Scholar] [CrossRef]
- Millán, O.; Rico, D.; Peinado, H.; Zarich, N.; Stamatakis, K.; Pérez-Sala, D.; Rojas, J.M.; Cano, A.; Boscá, L. Potentiation of tumor formation by topic administration of 15-deoxy-Δ12,14-prostaglandin J2 in a model of skin carcinogenesis. Carcinogenesis 2006, 27, 328–336. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rhee, S.G.; Bae, Y.S.; Lee, S.R.; Kwon, J. Hydrogen peroxide: A key messenger that modulates protein phosphorylation through cysteine oxidation. Sci. Signal. 2000, 2000, pe1. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.B.; Guan, P.P.; Wang, P. Prostaglandin A1 Decreases the Phosphorylation of Tau by Activating Protein Phosphatase 2A via a Michael Addition Mechanism at Cysteine 377. Mol. Neurobiol. 2020. [Google Scholar] [CrossRef]
- Suh, J.; Kim, D.H.; Kim, E.H.; Park, S.A.; Park, J.M.; Jang, J.H.; Kim, S.J.; Na, H.K.; Kim, N.D.; Kim, N.J.; et al. 15-Deoxy-Delta(12,14)-prostaglandin J2 activates PI3K-Akt signaling in human breast cancer cells through covalent modification of the tumor suppressor PTEN at cysteine 136. Cancer Lett. 2018, 424, 30–45. [Google Scholar] [CrossRef]
- Ichikawa, T.; Zhang, J.; Chen, K.; Liu, Y.; Schopfer, F.J.; Baker, P.R.; Freeman, B.A.; Chen, Y.E.; Cui, T. Nitroalkenes suppress lipopolysaccharide-induced signal transducer and activator of transcription signaling in macrophages: A critical role of mitogen-activated protein kinase phosphatase 1. Endocrinology 2008, 149, 4086–4094. [Google Scholar] [CrossRef] [PubMed]
- Steeg, P.S.; Palmieri, D.; Ouatas, T.; Salerno, M. Histidine kinases and histidine phosphorylated proteins in mammalian cell biology, signal transduction and cancer. Cancer Lett. 2003, 190, 1–12. [Google Scholar] [CrossRef]
- Fritz, K.S.; Galligan, J.J.; Smathers, R.L.; Roede, J.R.; Shearn, C.T.; Reigan, P.; Petersen, D.R. 4-Hydroxynonenal inhibits SIRT3 via thiol-specific modification. Chem. Res. Toxicol. 2011, 24, 651–662. [Google Scholar] [CrossRef] [PubMed]
| Protein | Targeted Residue (Position) | Electrophile | Type of Adduction | Reference |
|---|---|---|---|---|
| Pyruvate kinase | Cys 49, 152, 326, 358, 423, 474 | Acrolein, HHE and MDA | Michael, Schiff’s or FDP adduction | [33] |
| Lys 66, 115, 135, 166, 188, 207, 224, 247, 270, 305, 367, 393, 475 | ||||
| His 379, 391, 464 | ||||
| Cyclin-dependent Kinase 2 | Cys 177 | HNE | Michael | [85] |
| Lys 129 | ||||
| His 60, 71, 161, 268, 283, 295 | ||||
| Serum Albumin | Cys 53, 62, 75, 101, 124, 245, 246, 253, 269, 270, 277, 514 | HNE and MDA | Michael and Schiff’s (N-propenal-lysine adduct with MDA) | [137,138] |
| Lys 73, 106, 136, 174, 233, 240, 281, 378, 525, 541, 545 | ||||
| His 67, 105, 128, 242, 247, 510 | ||||
| Apolipoprotein E | Lys 64, 67, 68, 135, 138, 149, 155, 254 | Acrolein | Michael and Schiff’s | [139] |
| Creatine kinase | Cys 141, 145, 254, 283 | HNE | Michael and Schiff’s | [140] |
| Lys 86, 101 | ||||
| His 7, 26, 29, 66, 97, 191, 219, 234, 276, 296, 305 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Viedma-Poyatos, Á.; González-Jiménez, P.; Langlois, O.; Company-Marín, I.; Spickett, C.M.; Pérez-Sala, D. Protein Lipoxidation: Basic Concepts and Emerging Roles. Antioxidants 2021, 10, 295. https://doi.org/10.3390/antiox10020295
Viedma-Poyatos Á, González-Jiménez P, Langlois O, Company-Marín I, Spickett CM, Pérez-Sala D. Protein Lipoxidation: Basic Concepts and Emerging Roles. Antioxidants. 2021; 10(2):295. https://doi.org/10.3390/antiox10020295
Chicago/Turabian StyleViedma-Poyatos, Álvaro, Patricia González-Jiménez, Ophélie Langlois, Idoia Company-Marín, Corinne M. Spickett, and Dolores Pérez-Sala. 2021. "Protein Lipoxidation: Basic Concepts and Emerging Roles" Antioxidants 10, no. 2: 295. https://doi.org/10.3390/antiox10020295
APA StyleViedma-Poyatos, Á., González-Jiménez, P., Langlois, O., Company-Marín, I., Spickett, C. M., & Pérez-Sala, D. (2021). Protein Lipoxidation: Basic Concepts and Emerging Roles. Antioxidants, 10(2), 295. https://doi.org/10.3390/antiox10020295








