Exploring Early Detection of Frailty Syndrome in Older Adults: Evaluation of Oxi-Immune Markers, Clinical Parameters and Modifiable Risk Factors
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Frailty Status Evaluation
2.3. Biological Sample Collection
2.4. CRP Test and IL-6 Immunoassay
2.5. Neopterin, Tryptophan, Kynurenine, Phenylalanine, Tyrosine and Nitrite Analyses
2.6. Vitamin A and E Determinations
2.7. Enzyme-Modified Comet Assay
2.8. Statistical Analysis
3. Results
3.1. Study Population
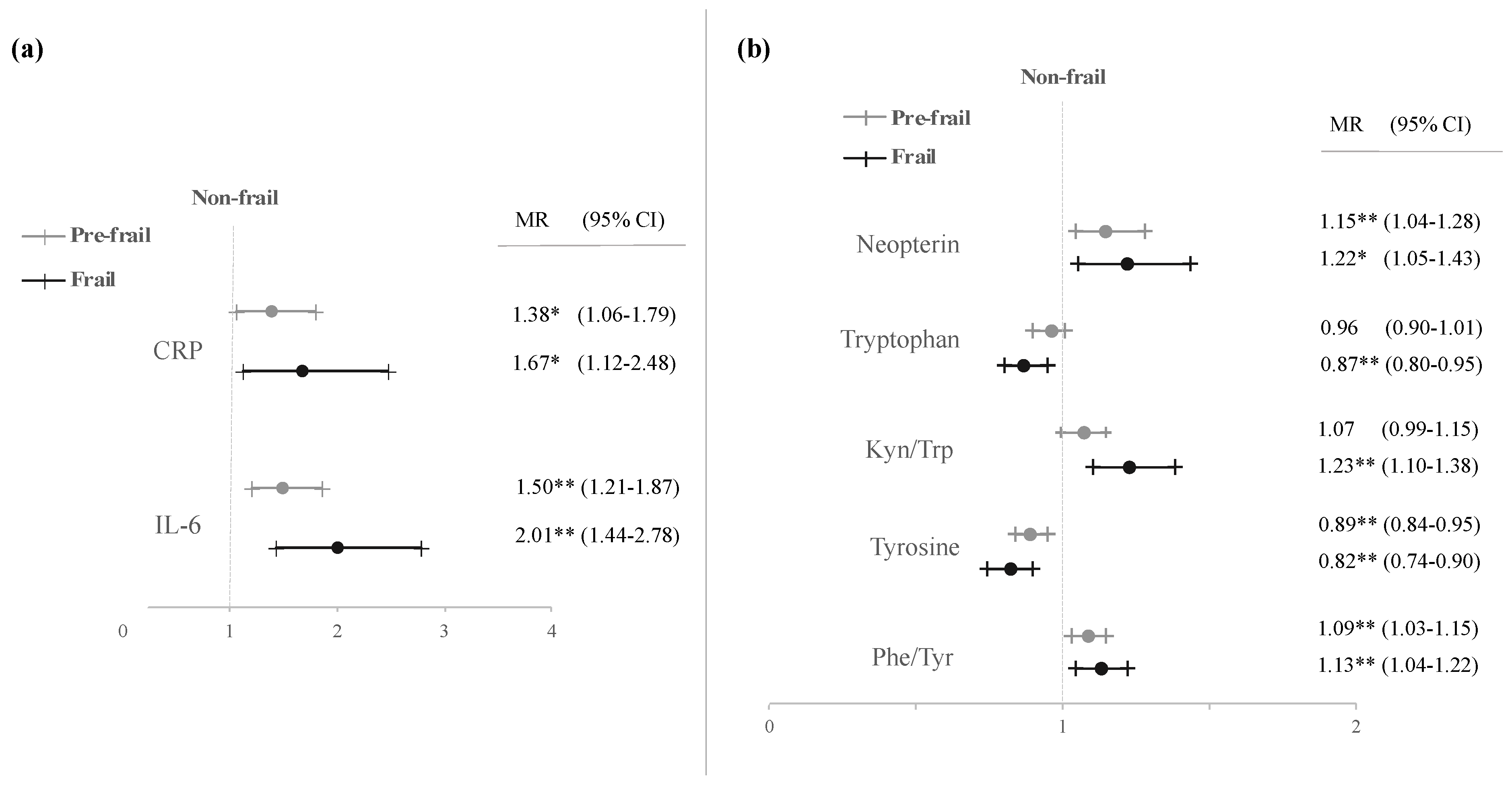
3.2. Analysis of the Frailty Phenotype vs. Biomarkers Studied
Single Contribution of Each of the 5-Frailty Criteria to the Biomarkers Studied
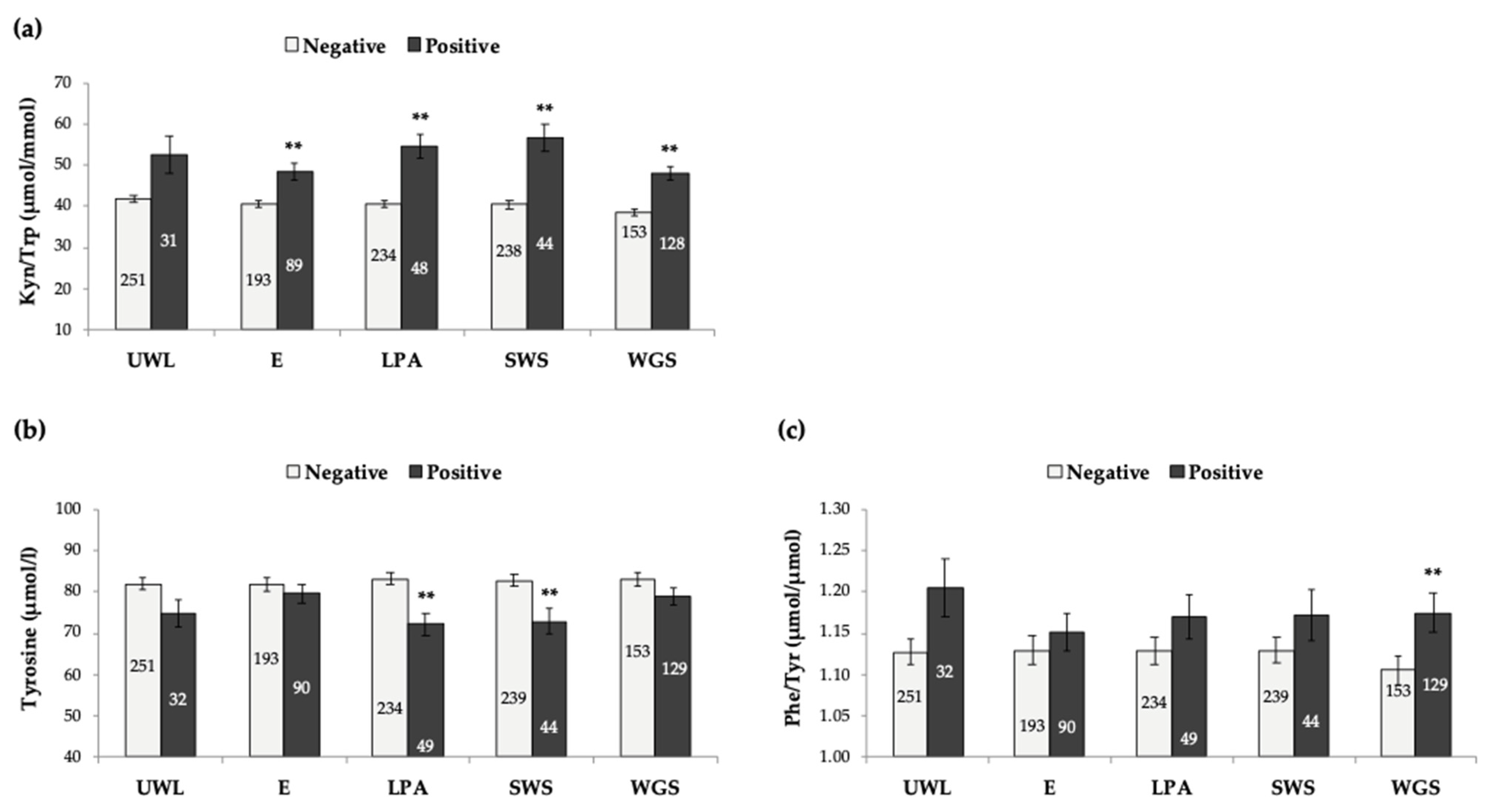
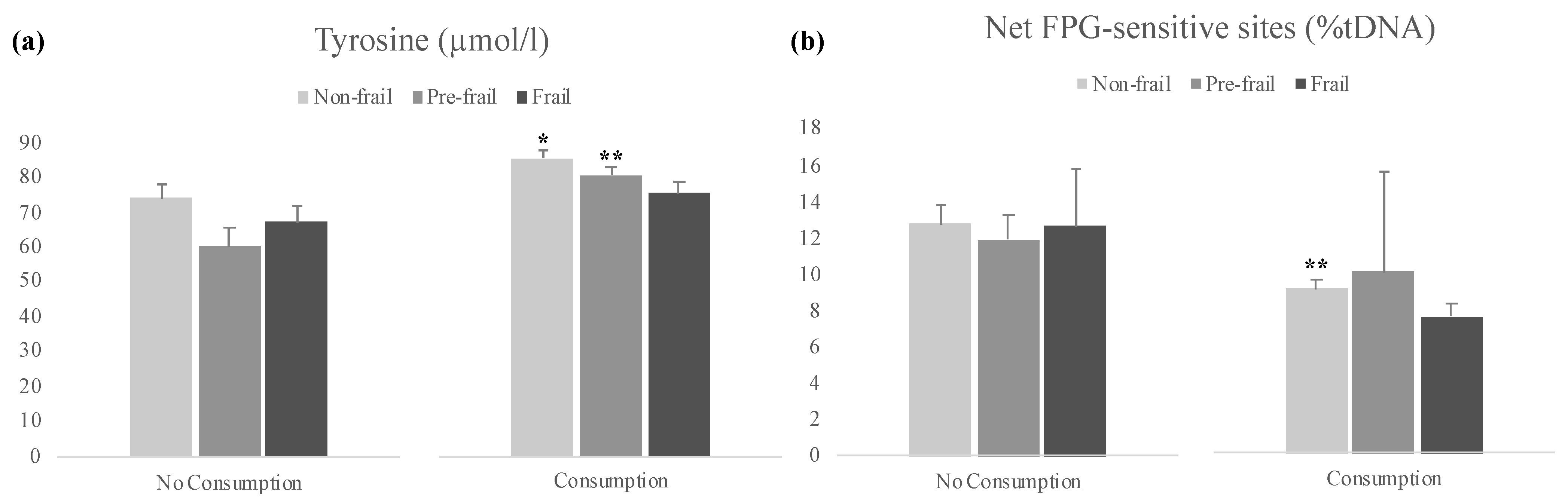
3.3. Analysis of Host and Modifiable Lifestyle Factors vs. Biomarkers Studied
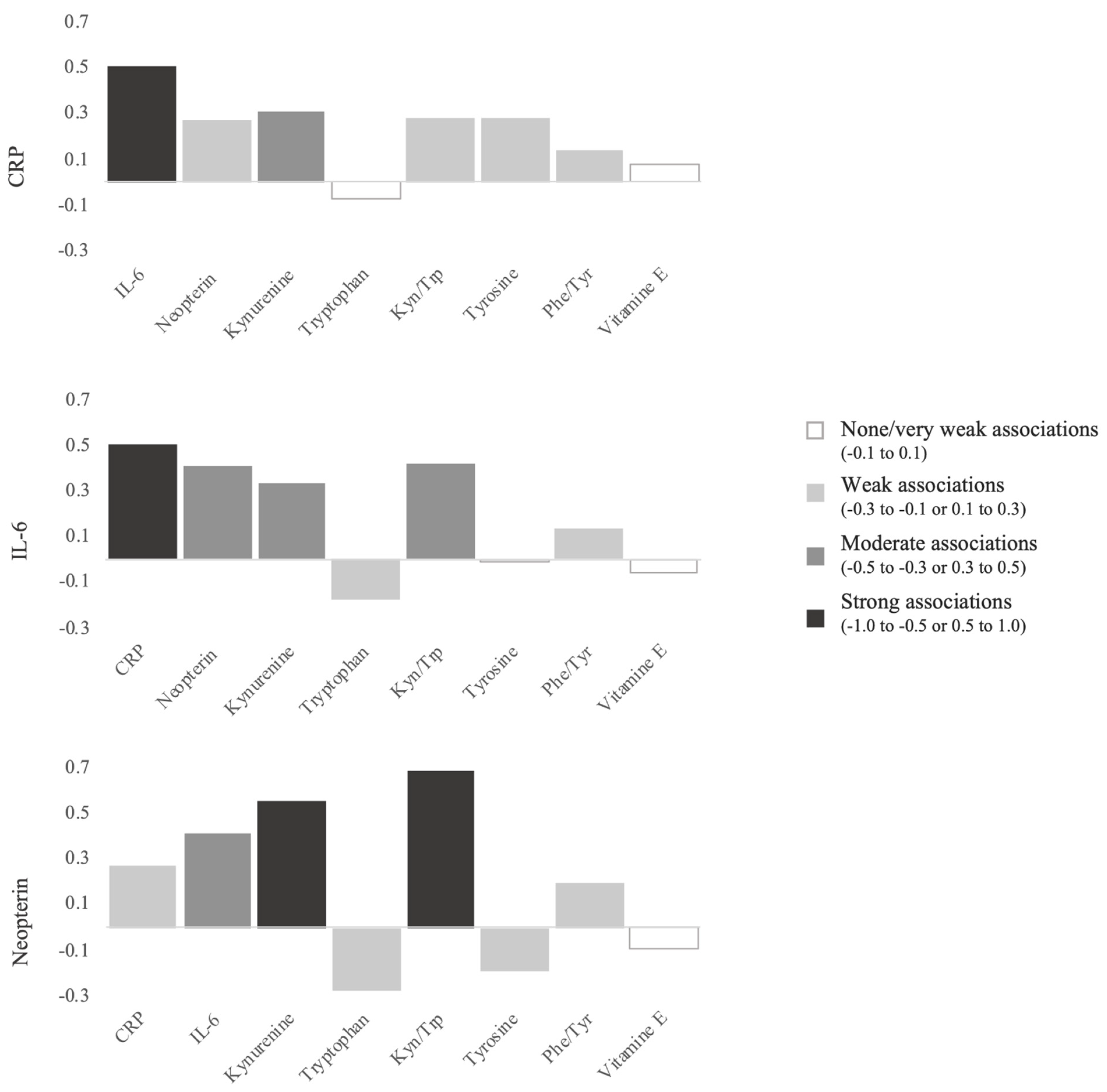
3.4. Correlations between the Different Biomarkers Studied
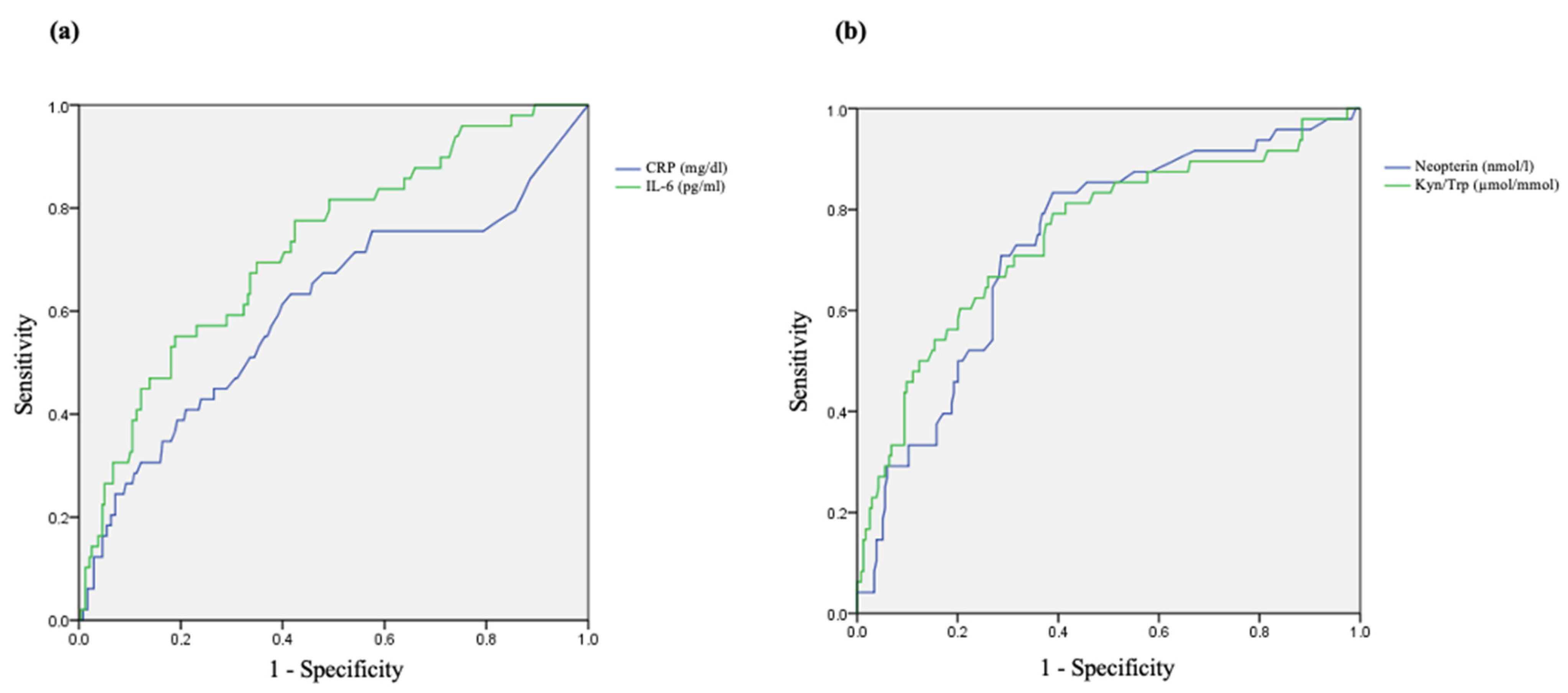
3.5. Predictive Value of Studied Biomarkers
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Available online: https://www.who.int/news-room/fact-sheets/detail/ageing-and-health (accessed on 9 May 2021).
- Ribeiro, A.R.; Howlett, S.E.; Fernandes, A. Frailty-A promising concept to evaluate disease vulnerability. Mech. Ageing Dev. 2020, 187, 111217. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, M.; Cefalu, C.; Reske, T. Frailty syndrome in geriatric medicine. Am. J. Med. Sci. 2012, 344, 395–398. [Google Scholar] [CrossRef]
- Mulero, J.; Zafrilla, P.; Martinez-Cacha, A. Oxidative stress, frailty and cognitive decline. J. Nutr. Health Aging 2011, 15, 756–760. [Google Scholar] [CrossRef]
- Dent, E.; Kowal, P.; Hoogendijk, E.O. Frailty measurement in research and clinical practice: A review. Eur. J. Intern. Med. 2016, 31, 3–10. [Google Scholar] [CrossRef]
- Rockwood, K.; Stadnyk, K.; MacKnight, C.; McDowell, I.; Hébert, R.; Hogan, D.B. A brief clinical instrument to classify frailty in elderly people. Lancet 1999, 353, 205–206. [Google Scholar] [CrossRef]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G. Frailty in older adults evidence for a phenotype. J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, M146–M157. [Google Scholar] [CrossRef]
- Villacampa-Fernandez, P.; Navarro-Pardo, E.; Tarin, J.J.; Cano, A. Frailty and multimorbidity: Two related yet different concepts. Maturitas 2017, 95, 31–35. [Google Scholar] [CrossRef]
- Rodriguez-Artalejo, F.; Rodriguez-Manas, L. The frailty syndrome in the public health agenda. J. Epidemiol. Community Health 2014, 68, 703–704. [Google Scholar] [CrossRef]
- Lage, B.; Teixeira-Gomes, A.; Mendes, A.; Valdiglesias, V.; Teixeira, J.P.; Costa, S. Frailty syndrome: An emergent concern of unknown causes. In Elderly Care: Options, Challenges and Trends; Nielsen, D.L., Ed.; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2018; pp. 105–142. [Google Scholar]
- Bandeen-Roche, K.; Xue, Q.L.; Ferrucci, L.; Walston, J.; Guralnik, J.M.; Chaves, P.; Zeger, S.L.; Fried, L.P. Phenotype of frailty: Characterization in the women’s health and aging studies. J. Gerontol. A Biol. Sci. Med. Sci. 2006, 61, 262–266. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.S.; Weiss, C.O.; Xue, Q.L.; Fried, L.P. Patterns of comorbid inflammatory diseases in frail older women: The Women’s Health and Aging Studies I and II. J. Gerontol. A Biol. Sci. Med. Sci. 2010, 65, 407–413. [Google Scholar] [CrossRef]
- Fried, L.P.; Ferrucci, L.; Darer, J.; Williamson, J.D.; Anderson, G. Untangling the concepts of disability, frailty, and comorbidity: Implications for improved targeting and care. J. Gerontol. A Biol. Sci. Med. Sci. 2004, 59, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Walston, J.; McBurnie, M.A.; Newman, A.; Tracy, R.P.; Kop, W.J.; Hirsch, C.H.; Gottdiener, J.; Fried, L.P. Frailty and activation of the inflammation and coagulation systems with and without clinical comorbidities: Results from the Cardiovascular Health Study. Arch. Intern. Med. 2002, 162, 2333–2341. [Google Scholar] [CrossRef] [PubMed]
- Fedarko, N.S. The biology of aging and frailty. Clin. Geriatr. Med. 2011, 27, 27–37. [Google Scholar] [CrossRef]
- Franceschi, C.; Capri, M.; Monti, D.; Giunta, S.; Olivieri, F.; Sevini, F.; Panourgia, M.P.; Invidia, L.; Celani, L.; Scurti, M.; et al. Inflammaging and anti-inflammaging: A systemic perspective on aging and longevity emerged from studies in humans. Mech. Ageing Dev. 2007, 128, 92–105. [Google Scholar] [CrossRef]
- Franceschi, C.; Bonafè, M.; Valensin, S. Human immunosenescence: The prevailing of innate immunity, the failing of clonotypic immunity, and the filling of immunological space. Vaccine 2000, 18, 1717–1720. [Google Scholar] [CrossRef]
- De la Fuente, M.; Miquel, J. An update of the oxidation-inflammation theory of aging: The involvement of the immune system in oxi-inflamm-aging. Curr. Pharm. Des. 2009, 15, 3003–3026. [Google Scholar] [CrossRef] [PubMed]
- Ferrucci, L.; Fabbri, E. Inflammageing: Chronic inflammation in ageing, cardiovascular disease, and frailty. Nat. Rev. Cardiol. 2018, 15, 505–522. [Google Scholar] [CrossRef]
- Lencel, P.; Magne, D. Inflammaging: The driving force in osteoporosis? Med. Hypotheses 2011, 76, 317–321. [Google Scholar] [CrossRef]
- Morales, I.; Farías, G.; Maccioni, R.B. Neuroimmunomodulation in the pathogenesis of Alzheimer’s disease. Neuroimmunomodulation 2010, 17, 202–204. [Google Scholar] [CrossRef]
- Pradhan, A.D.; Manson, J.E.; Rifai, N.; Buring, J.E.; Ridker, P.M. C-reactive protein, interleukin 6, and risk of developing type 2 diabetes mellitus. Jama 2001, 286, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, D.B.; Firth, C.M.; Phillips, A.C.; Moss, P.; Baylis, D.; Syddall, H.; Sayer, A.A.; Cooper, C.; Lord, J.M. The age-related increase in low-grade systemic inflammation (Inflammaging) is not driven by cytomegalovirus infection. Aging Cell 2012, 11, 912–915. [Google Scholar] [CrossRef]
- Marcos-Perez, D.; Sanchez-Flores, M.; Maseda, A.; Lorenzo-Lopez, L.; Millan-Calenti, J.C.; Gostner, J.M.; Fuchs, D.; Pasaro, E.; Laffon, B.; Valdiglesias, V. Frailty in Older Adults Is Associated With Plasma Concentrations of Inflammatory Mediators but Not With Lymphocyte Subpopulations. Front. Immunol. 2018, 9, 1056. [Google Scholar] [CrossRef]
- Collerton, J.; Martin-Ruiz, C.; Davies, K.; Hilkens, C.M.; Isaacs, J.; Kolenda, C.; Parker, C.; Dunn, M.; Catt, M.; Jagger, C.; et al. Frailty and the role of inflammation, immunosenescence and cellular ageing in the very old: Cross-sectional findings from the Newcastle 85+ Study. Mech. Ageing Dev. 2012, 133, 456–466. [Google Scholar] [CrossRef] [PubMed]
- Marcos-Pérez, D.; Sánchez-Flores, M.; Proietti, S.; Bonassi, S.; Costa, S.; Teixeira, J.P.; Fernández-Tajes, J.; Pásaro, E.; Laffon, B.; Valdiglesias, V. Association of inflammatory mediators with frailty status in older adults: Results from a systematic review and meta-analysis. Geroscience 2020, 42, 1451–1473. [Google Scholar] [CrossRef]
- Soysal, P.; Stubbs, B.; Lucato, P.; Luchini, C.; Solmi, M.; Peluso, R.; Sergi, G.; Isik, A.T.; Manzato, E.; Maggi, S.; et al. Inflammation and frailty in the elderly: A systematic review and meta-analysis. Ageing Res. Rev. 2016, 31, 1–8. [Google Scholar] [CrossRef]
- Hamrick, M.W.; Isales, C.M. Special issue: The kynurenine pathway in aging. Exp. Gerontol. 2020, 134, 110895. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, D.; Möller, A.A.; Reibnegger, G.; Werner, E.R.; Werner-Felmayer, G.; Dierich, M.P.; Wachter, H. Increased endogenous interferon-gamma and neopterin correlate with increased degradation of tryptophan in human immunodeficiency virus type 1 infection. Immunol. Lett. 1991, 28, 207–211. [Google Scholar] [CrossRef]
- Gostner, J.M.; Geisler, S.; Stonig, M.; Mair, L.; Sperner-Unterweger, B.; Fuchs, D. Tryptophan Metabolism and Related Pathways in Psychoneuroimmunology: The Impact of Nutrition and Lifestyle. Neuropsychobiology 2020, 79, 89–99. [Google Scholar] [CrossRef]
- Valdiglesias, V.; Marcos-Pérez, D.; Lorenzi, M.; Onder, G.; Gostner, J.M.; Strasser, B.; Fuchs, D.; Bonassi, S. Immunological alterations in frail older adults: A cross sectional study. Exp. Gerontol. 2018, 112, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Theofylaktopoulou, D.; Midttun, Ø.; Ulvik, A.; Ueland, P.M.; Tell, G.S.; Vollset, S.E.; Nygård, O.; Eussen, S.J. A community-based study on determinants of circulating markers of cellular immune activation and kynurenines: The Hordaland Health Study. Clin. Exp. Immunol. 2013, 173, 121–130. [Google Scholar] [CrossRef]
- Marcos-Perez, D.; Sanchez-Flores, M.; Maseda, A.; Lorenzo-Lopez, L.; Millan-Calenti, J.C.; Strasser, B.; Gostner, J.M.; Fuchs, D.; Pasaro, E.; Valdiglesias, V.; et al. Frailty Status in Older Adults Is Related to Alterations in Indoleamine 2,3-Dioxygenase 1 and Guanosine Triphosphate Cyclohydrolase I Enzymatic Pathways. J. Am. Med. Dir. Assoc. 2017, 18, 1049–1057. [Google Scholar] [CrossRef]
- Jang, I.Y.; Park, J.H.; Kim, J.H.; Lee, S.; Lee, E.; Lee, J.Y.; Park, S.J.; Kim, D.A.; Hamrick, M.W.; Kim, B.J. The association of circulating kynurenine, a tryptophan metabolite, with frailty in older adults. Aging 2020, 12, 22253–22265. [Google Scholar] [CrossRef] [PubMed]
- Liguori, I.; Russo, G.; Curcio, F.; Bulli, G.; Aran, L.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Cacciatore, F.; Bonaduce, D.; et al. Oxidative stress, aging, and diseases. Clin. Interv. Aging 2018, 13, 757–772. [Google Scholar] [CrossRef] [PubMed]
- Namioka, N.; Hanyu, H.; Hirose, D.; Hatanaka, H.; Sato, T.; Shimizu, S. Oxidative stress and inflammation are associated with physical frailty in patients with Alzheimer’s disease. Geriatr. Gerontol. Int. 2017, 17, 913–918. [Google Scholar] [CrossRef]
- Saum, K.U.; Dieffenbach, A.K.; Jansen, E.H.; Schöttker, B.; Holleczek, B.; Hauer, K.; Brenner, H. Association between Oxidative Stress and Frailty in an Elderly German Population: Results from the ESTHER Cohort Study. Gerontology 2015, 61, 407–415. [Google Scholar] [CrossRef]
- Serviddio, G.; Romano, A.; Greco, A.; Rollo, T.; Bellanti, F.; Altomare, E.; Vendemiale, G. Frailty syndrome is associated with altered circulating redox balance and increased markers of oxidative stress. Int. J. Immunopathol. Pharmacol. 2009, 22, 819–827. [Google Scholar] [CrossRef]
- Wu, I.C.; Shiesh, S.C.; Kuo, P.H.; Lin, X.Z. High oxidative stress is correlated with frailty in elderly chinese. J. Am. Geriatr. Soc. 2009, 57, 1666–1671. [Google Scholar] [CrossRef]
- Ble, A.; Cherubini, A.; Volpato, S.; Bartali, B.; Walston, J.D.; Windham, B.G.; Bandinelli, S.; Lauretani, F.; Guralnik, J.M.; Ferrucci, L. Lower plasma vitamin E levels are associated with the frailty syndrome: The InCHIANTI study. J. Gerontol. A Biol. Sci. Med. Sci. 2006, 61, 278–283. [Google Scholar] [CrossRef] [PubMed]
- Goulet, E.D.; Hassaine, A.; Dionne, I.J.; Gaudreau, P.; Khalil, A.; Fulop, T.; Shatenstein, B.; Tessier, D.; Morais, J.A. Frailty in the elderly is associated with insulin resistance of glucose metabolism in the postabsorptive state only in the presence of increased abdominal fat. Exp. Gerontol. 2009, 44, 740–744. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.R. Vitamins in aging, health, and longevity. Clin. Interv. Aging 2006, 1, 81–91. [Google Scholar] [CrossRef]
- Kaiser, M.J.; Bauer, J.; Ramsch, C.; Uter, W.; Guigoz, Y.; Cederholm, T.; Thomas, D.R.; Anthony, P.; Charlton, K.; Maggio, M. Validation of the Mini Nutritional Assessment Short-Form (MNA-SF): A practical tool for identification of nutritional status. J. Nutr. Health Aging 2009, 13, 782–788. [Google Scholar] [CrossRef]
- Katz, S.; Ford, A.B.; Moskowitz, R.W.; Jackson, B.A.; Jaffe, M.W. Studies of illness in the aged. the index of ADL: A standardized measure of biological and psychosocial function. Jama 1963, 185, 914–919. [Google Scholar] [CrossRef] [PubMed]
- Lawton, M.P.; Brody, E.M. Assessment of older people: Self-maintaining and instrumental activities of daily living. Gerontologist 1969, 9, 179–186. [Google Scholar] [CrossRef]
- Teixeira-Gomes, A.; Lage, B.; Esteves, F.; Sousa, A.C.; Pastorinho, M.R.; Valdiglesias, V.; Costa, S.; Laffon, B.; Teixeira, J.P. Frailty syndrome, biomarkers and environmental factors-A pilot study. Toxicol. Lett. 2020, 330, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Ruiz Comellas, A.; Pera, G.; Baena Diez, J.M.; Mundet Tuduri, X.; Alzamora Sas, T.; Elosua, R.; Toran Monserrat, P.; Heras, A.; Fores Raurell, R.; Fuste Gamisans, M.; et al. Validación de una versión reducida en español del cuestionario de actividad física en el tiempo libre de Minnesota (VREM). Rev. Esp. Salud Publica 2012, 86, 495–508. [Google Scholar] [CrossRef] [PubMed]
- Laich, A.; Neurauter, G.; Widner, B.; Fuchs, D. More rapid method for simultaneous measurement of tryptophan and kynurenine by HPLC. Clin. Chem. 2002, 48, 579–581. [Google Scholar] [CrossRef]
- Neurauter, G.; Scholl-Bürgi, S.; Haara, A.; Geisler, S.; Mayersbach, P.; Schennach, H.; Fuchs, D. Simultaneous measurement of phenylalanine and tyrosine by high performance liquid chromatography (HPLC) with fluorescence detection. Clin. Biochem. 2013, 46, 1848–1851. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, D.; Möller, A.A.; Reibnegger, G.; Stöckle, E.; Werner, E.R.; Wachter, H. Decreased serum tryptophan in patients with HIV-1 infection correlates with increased serum neopterin and with neurologic/psychiatric symptoms. J. Acquir. Immune Defic. Syndr. 1990, 3, 873–876. [Google Scholar] [PubMed]
- Geisler, S.; Mayersbach, P.; Becker, K.; Schennach, H.; Fuchs, D.; Gostner, J.M. Serum tryptophan, kynurenine, phenylalanine, tyrosine and neopterin concentrations in 100 healthy blood donors. Pteridines 2015, 26, 31–36. [Google Scholar] [CrossRef]
- Azqueta, A.; Collins, A.R. The essential comet assay: A comprehensive guide to measuring DNA damage and repair. Arch. Toxicol. 2013, 87, 949–968. [Google Scholar] [CrossRef] [PubMed]
- Costa, S.; Coelho, P.; Costa, C.; Silva, S.; Mayan, O.; Santos, L.S.; Gaspar, J.; Teixeira, J.P. Genotoxic damage in pathology anatomy laboratory workers exposed to formaldehyde. Toxicology 2008, 252, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Søreide, K. Receiver-operating characteristic curve analysis in diagnostic, prognostic and predictive biomarker research. J. Clin. Pathol. 2009, 62, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Shengping, Y.; Gilbert, B. The receiver operating characteristic (ROC) curve. Southwest Respir. Crit. Care Chron. 2017, 5. [Google Scholar] [CrossRef]
- Di Micco, R.; Krizhanovsky, V.; Baker, D.; d’Adda di Fagagna, F. Cellular senescence in ageing: From mechanisms to therapeutic opportunities. Nat. Rev. Mol. Cell Biol. 2021, 22, 75–95. [Google Scholar] [CrossRef]
- Álvarez-Satta, M.; Berna-Erro, A.; Carrasco-Garcia, E.; Alberro, A.; Saenz-Antoñanzas, A.; Vergara, I.; Otaegui, D.; Matheu, A. Relevance of oxidative stress and inflammation in frailty based on human studies and mouse models. Aging 2020, 12, 9982–9999. [Google Scholar] [CrossRef] [PubMed]
- Pansarasa, O.; Pistono, C.; Davin, A.; Bordoni, M.; Mimmi, M.C.; Guaita, A.; Cereda, C. Altered immune system in frailty: Genetics and diet may influence inflammation. Ageing Res. Rev. 2019, 54, 100935. [Google Scholar] [CrossRef] [PubMed]
- Vatic, M.; von Haehling, S.; Ebner, N. Inflammatory biomarkers of frailty. Exp. Gerontol. 2020, 133, 110858. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Flores, M.; Marcos-Perez, D.; Costa, S.; Teixeira, J.P.; Bonassi, S.; Pasaro, E.; Laffon, B.; Valdiglesias, V. Oxidative stress, genomic features and DNA repair in frail elderly: A systematic review. Ageing Res. Rev. 2017, 37, 1–15. [Google Scholar] [CrossRef]
- Sousa-Santos, A.R.; Afonso, C.; Moreira, P.; Padrão, P.; Santos, A.; Borges, N.; Amaral, T.F. Weakness: The most frequent criterion among pre-frail and frail older Portuguese. Arch. Gerontol. Geriatr. 2018, 74, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Coelho, T.; Santos, R.; Paúl, C.; Gobbens, R.J.; Fernandes, L. Portuguese version of the Tilburg Frailty Indicator: Transcultural adaptation and psychometric validation. Geriatr. Gerontol. Int. 2015, 15, 951–960. [Google Scholar] [CrossRef] [PubMed]
- Duarte, M.; Paúl, C. Prevalence of phenotypic frailty during the aging process in a Portuguese community. Rev. Bras. Geriatr. Gerontol. 2015, 18, 871–880. [Google Scholar] [CrossRef]
- Valdiglesias, V.; Sanchez-Flores, M.; Marcos-Perez, D.; Lorenzo-Lopez, L.; Maseda, A.; Millan-Calenti, J.C.; Pasaro, E.; Laffon, B. Exploring Genetic Outcomes as Frailty Biomarkers. J. Gerontol. A Biol. Sci. Med. Sci. 2019, 74, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Xue, Q.L. The frailty syndrome: Definition and natural history. Clin. Geriatr. Med. 2011, 27, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Collard, R.M.; Boter, H.; Schoevers, R.A.; Oude Voshaar, R.C. Prevalence of Frailty in Community-Dwelling Older Persons: A Systematic Review. J. Am. Geriatr. Soc. 2012, 60, 1487–1492. [Google Scholar] [CrossRef] [PubMed]
- Alves, S.; Teixeira, L.; Ribeiro, O.; Paúl, C. Examining Frailty Phenotype Dimensions in the Oldest Old. Front. Psychol. 2020, 11, 434. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, O.; Duarte, N.; Teixeira, L.; Paúl, C. Frailty and depression in centenarians. Int. Psychogeriatr. 2018, 30, 115–124. [Google Scholar] [CrossRef] [PubMed]
- United Nations. World Population Ageing 2019: Highlights; United Nations, Department of Economic and Social Affairs, Population Division (ST/ESA/SER.A/430): New York, NY, USA, 2019. [Google Scholar]
- Oksuzyan, A.; Juel, K.; Vaupel, J.W.; Christensen, K. Men: Good health and high mortality. Sex differences in health and aging. Aging Clin. Exp. Res. 2008, 20, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Gordon, E.H.; Hubbard, R.E. Differences in frailty in older men and women. Med. J. Aust. 2020, 212, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Fialova, D.; Laffon, B.; Marinkovic, V.; Tasic, L.; Doro, P.; Somicronos, G.; Mota, J.; Dogan, S.; Brkic, J.; Teixeira, J.P.; et al. Medication use in older patients and age-blind approach: Narrative literature review (insufficient evidence on the efficacy and safety of drugs in older age, frequent use of PIMs and polypharmacy, and underuse of highly beneficial nonpharmacological strategies). Eur. J. Clin. Pharmacol. 2019, 75, 451–466. [Google Scholar] [CrossRef] [PubMed]
- Herr, M.; Robine, J.M.; Pinot, J.; Arvieu, J.J.; Ankri, J. Polypharmacy and frailty: Prevalence, relationship, and impact on mortality in a French sample of 2350 old people. Pharmacoepidemiol. Drug Saf. 2015, 24, 637–646. [Google Scholar] [CrossRef]
- Saum, K.U.; Schöttker, B.; Meid, A.D.; Holleczek, B.; Haefeli, W.E.; Hauer, K.; Brenner, H. Is Polypharmacy Associated with Frailty in Older People? Results from the ESTHER Cohort Study. J. Am. Geriatr. Soc. 2017, 65, e27–e32. [Google Scholar] [CrossRef]
- Moulis, F.; Moulis, G.; Balardy, L.; Gérard, S.; Montastruc, F.; Sourdet, S.; Rougé-Bugat, M.E.; Lapeyre-Mestre, M.; Montastruc, J.L.; Rolland, Y.; et al. Exposure to atropinic drugs and frailty status. J. Am. Med. Dir. Assoc. 2015, 16, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Valencia, M.; Izquierdo, M.; Cesari, M.; Casas-Herrero, Á.; Inzitari, M.; Martínez-Velilla, N. The relationship between frailty and polypharmacy in older people: A systematic review. Br. J. Clin. Pharmacol. 2018, 84, 1432–1444. [Google Scholar] [CrossRef]
- Shmuel, S.; Lund, J.L.; Alvarez, C.; Hsu, C.D.; Palta, P.; Kucharska-Newton, A.; Jordan, J.M.; Nelson, A.E.; Golightly, Y.M. Polypharmacy and Incident Frailty in a Longitudinal Community-Based Cohort Study. J. Am. Geriatr. Soc. 2019, 67, 2482–2489. [Google Scholar] [CrossRef]
- Cesari, M. How polypharmacy affects frailty. Expert Rev. Clin. Pharmacol. 2020, 13, 1179–1181. [Google Scholar] [CrossRef]
- Dorner, T.E.; Luger, E.; Tschinderle, J.; Stein, K.V.; Haider, S.; Kapan, A.; Lackinger, C.; Schindler, K.E. Association between nutritional status (MNA®-SF) and frailty (SHARE-FI) in acute hospitalised elderly patients. J. Nutr. Health Aging 2014, 18, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Pilleron, S.; Ajana, S.; Jutand, M.A.; Helmer, C.; Dartigues, J.F.; Samieri, C.; Féart, C. Dietary Patterns and 12-Year Risk of Frailty: Results From the Three-City Bordeaux Study. J. Am. Med. Dir. Assoc. 2017, 18, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Nourhashémi, F.; Andrieu, S.; Gillette-Guyonnet, S.; Vellas, B.; Albarède, J.L.; Grandjean, H. Instrumental activities of daily living as a potential marker of frailty: A study of 7364 community-dwelling elderly women (the EPIDOS study). J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, M448–M453. [Google Scholar] [CrossRef] [PubMed]
- Provencher, V.; Béland, F.; Demers, L.; Desrosiers, J.; Bier, N.; Ávila-Funes, J.A.; Galand, C.; Julien, D.; Fletcher, J.D.; Trottier, L.; et al. Are frailty components associated with disability in specific activities of daily living in community-dwelling older adults? A multicenter Canadian study. Arch. Gerontol. Geriatr. 2017, 73, 187–194. [Google Scholar] [CrossRef]
- Vermeulen, J.; Neyens, J.C.; van Rossum, E.; Spreeuwenberg, M.D.; de Witte, L.P. Predicting ADL disability in community-dwelling elderly people using physical frailty indicators: A systematic review. BMC Geriatr. 2011, 11, 33. [Google Scholar] [CrossRef]
- Kojima, G. Frailty as a predictor of disabilities among community-dwelling older people: A systematic review and meta-analysis. Disabil. Rehabil. 2017, 39, 1897–1908. [Google Scholar] [CrossRef] [PubMed]
- Puzianowska-Kuźnicka, M.; Owczarz, M.; Wieczorowska-Tobis, K.; Nadrowski, P.; Chudek, J.; Slusarczyk, P.; Skalska, A.; Jonas, M.; Franek, E.; Mossakowska, M. Interleukin-6 and C-reactive protein, successful aging, and mortality: The PolSenior study. Immun. Ageing 2016, 13, 21. [Google Scholar] [CrossRef] [PubMed]
- Harris, T.B.; Ferrucci, L.; Tracy, R.P.; Corti, M.C.; Wacholder, S.; Ettinger, W.H., Jr.; Heimovitz, H.; Cohen, H.J.; Wallace, R. Associations of elevated interleukin-6 and C-reactive protein levels with mortality in the elderly. Am. J. Med. 1999, 106, 506–512. [Google Scholar] [CrossRef]
- Cardoso, A.L.; Fernandes, A.; Aguilar-Pimentel, J.A.; de Angelis, M.H.; Guedes, J.R.; Brito, M.A.; Ortolano, S.; Pani, G.; Athanasopoulou, S.; Gonos, E.S.; et al. Towards frailty biomarkers: Candidates from genes and pathways regulated in aging and age-related diseases. Ageing Res. Rev. 2018, 47, 214–277. [Google Scholar] [CrossRef] [PubMed]
- Kiecolt-Glaser, J.K.; Preacher, K.J.; MacCallum, R.C.; Atkinson, C.; Malarkey, W.B.; Glaser, R. Chronic stress and age-related increases in the proinflammatory cytokine IL-6. Proc. Natl. Acad. Sci. USA 2003, 100, 9090–9095. [Google Scholar] [CrossRef]
- Klein, S.L.; Flanagan, K.L. Sex differences in immune responses. Nat. Rev. Immunol. 2016, 16, 626–638. [Google Scholar] [CrossRef] [PubMed]
- Lustgarten, M.S.; Fielding, R.A. Metabolites Associated With Circulating Interleukin-6 in Older Adults. J. Gerontol. A Biol. Sci. Med. Sci. 2017, 72, 1277–1283. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Flores, M.; Marcos-Perez, D.; Lorenzo-Lopez, L.; Maseda, A.; Millan-Calenti, J.C.; Bonassi, S.; Pasaro, E.; Laffon, B.; Valdiglesias, V. Frailty Syndrome and Genomic Instability in Older Adults: Suitability of the Cytome Micronucleus Assay As a Diagnostic Tool. J. Gerontol. A Biol. Sci. Med. Sci. 2018, 73, 864–872. [Google Scholar] [CrossRef] [PubMed]
- Marcos-Perez, D.; Sanchez-Flores, M.; Maseda, A.; Lorenzo-Lopez, L.; Millan-Calenti, J.C.; Pasaro, E.; Laffon, B.; Valdiglesias, V. Serum cortisol but not oxidative stress biomarkers are related to frailty: Results of a cross-sectional study in Spanish older adults. J. Toxicol. Environ. Health A 2019, 82, 815–825. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.J.; Lee, E.K.; Yu, B.P.; Chung, H.Y. Significance of protein tyrosine kinase/protein tyrosine phosphatase balance in the regulation of NF-kappaB signaling in the inflammatory process and aging. Free Radic. Biol. Med. 2009, 47, 983–991. [Google Scholar] [CrossRef] [PubMed]
- Meyers, S. Use of neurotransmitter precursors for treatment of depression. Altern. Med. Rev. 2000, 5, 64–71. [Google Scholar] [PubMed]
- Carhill, A.A.; Cabanillas, M.E.; Jimenez, C.; Waguespack, S.G.; Habra, M.A.; Hu, M.; Ying, A.; Vassilopoulou-Sellin, R.; Gagel, R.F.; Sherman, S.I.; et al. The noninvestigational use of tyrosine kinase inhibitors in thyroid cancer: Establishing a standard for patient safety and monitoring. J. Clin. Endocrinol. Metab. 2013, 98, 31–42. [Google Scholar] [CrossRef]
- Webster, D.; Wildgoose, J. Tyrosine supplementation for phenylketonuria. Cochrane Database Syst. Rev. 2013, 2013, Cd001507. [Google Scholar] [CrossRef]
- Garcia-Esquinas, E.; Rahi, B.; Peres, K.; Colpo, M.; Dartigues, J.F.; Bandinelli, S.; Feart, C.; Rodriguez-Artalejo, F. Consumption of fruit and vegetables and risk of frailty: A dose-response analysis of 3 prospective cohorts of community-dwelling older adults. Am. J. Clin. Nutr. 2016, 104, 132–142. [Google Scholar] [CrossRef]
- Shivappa, N.; Stubbs, B.; Hébert, J.R.; Cesari, M.; Schofield, P.; Soysal, P.; Maggi, S.; Veronese, N. The Relationship between the Dietary Inflammatory Index and Incident Frailty: A Longitudinal Cohort Study. J. Am. Med. Dir. Assoc. 2018, 19, 77–82. [Google Scholar] [CrossRef]
- Murr, C.; Winklhofer-Roob, B.M.; Schroecksnadel, K.; Maritschnegg, M.; Mangge, H.; Böhm, B.O.; Winkelmann, B.R.; März, W.; Fuchs, D. Inverse association between serum concentrations of neopterin and antioxidants in patients with and without angiographic coronary artery disease. Atherosclerosis 2009, 202, 543–549. [Google Scholar] [CrossRef]
- Feart, C. Nutrition and frailty: Current knowledge. Prog. Neuropsychopharmacol. Biol. Psychiatry 2019, 95, 109703. [Google Scholar] [CrossRef]
- Yannakoulia, M.; Ntanasi, E.; Anastasiou, C.A.; Scarmeas, N. Frailty and nutrition: From epidemiological and clinical evidence to potential mechanisms. Metabolism 2017, 68, 64–76. [Google Scholar] [CrossRef]
| Non-Frail | Pre-Frail | Frail | p-Value | |
|---|---|---|---|---|
| Total individuals n (%) | 111 (38.1) | 130 (44.7) | 50 (17.2) | |
| Age [years-old] a | 73.0 ± 5.9 [65–91] | 75.6 ± 6.7 [65–94] | 83.6 ± 7.0 [67–94] | <0.001 c |
| Gender n (%) | 0.003 b | |||
| Males | 59 (53.2) | 49 (37.7) | 13 (26.0) | |
| Females | 52 (46.8) | 81 (62.3) | 37 (74.0) | |
| BMI [kg/m2] a | 24.9 ± 3.6 [22.2–45.0] | 28.8 ± 4.2 [20.3–42.2] | 28.2 ± 6.3 [16.2–47.9] | 0.249 c |
| Smoking habits n (%) | 0.569 b | |||
| Non-smokers | 75 (67.6) | 95 (73.1) | 37 (74.0) | |
| Ever smokers | 36 (32.4) | 35 (26.9) | 13 (26.0) | |
| Years smoking a | 25.0 ± 15.8 (0.5–57) | 23.7 ± 16.8 (1–67) | 26.9 ± 121.0 (2–66) | 0.836 c |
| Second-hand smokers n (%) | 0.376 b | |||
| No | 65 (58.6) | 82 (63.1) | 35 (70.0) | |
| Yes | 46 (41.4) | 48 (36.9) | 15 (30.0) | |
| Alcohol consumption n (%) | <0.001 b | |||
| No | 24 (21.6) | 62 (47.7) | 27 (54.0) | |
| Yes | 87 (78.4) | 68 (52.3) | 23 (46.0) | |
| Polypharmacy n (%) | <0.001 b | |||
| No | 77 (69.4) | 61 (47.7) | 11 (22.0) | |
| Yes | 34 (30.6) | 67 (52.3) | 39 (78.0) | |
| No. drugs consumed/day a | 3.6 ± 2.5 (0–13) | 5.1 ± 2.6 (1–13) | 7.2 ± 3.4 (1–15) | <0.001 c |
| Vitamin supplementation n (%) | 0.224 b | |||
| No | 109 (98.2) | 122 (93.8) | 47 (94.0) | |
| Yes | 2 (1.8) | 8 (6.2) | 3 (6.0) | |
| Vaccination in the last year n (%) | 0.104 b | |||
| No | 30 (27.0) | 22 (16.9) | 8 (16.0) | |
| Yes | 81 (73.0) | 108 (83.1) | 42 (84.0) | |
| Nutrition n (%) | <0.001 b | |||
| Normal nutrition status | 110 (99.1) | 115 (88.5) | 24 (48.0) | |
| At risk or malnourished | 1 (0.9) | 15 (11.5) | 26 (52.0) | |
| Functional status-BADL n (%) | <0.001 b | |||
| No dependence | 98 (88.3) | 91 (70.0) | 13 (26.0) | |
| Dependence | 13 (11.7) | 39 (30.0) | 37 (74.0) | |
| Functional status-IADL n (%) | <0.001 b | |||
| No dependence | 102 (91.9) | 96 (73.8) | 5 (10.0) | |
| Dependence | 9 (8.1) | 34 (26.2) | 45 (90.0) | |
| Falls in the last 6 months a (self-reported) | 0.3 ± 1.2 (0–12) | 0.4 ± 0.9 (0–6) | 0.4 ± 0.7 (0–3) | 0.714 c |
| Non-Frail | Pre-Frail | Frail | p-Value # | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| n | Mean | SE | n | Mean | SE | n | Mean | SE | |||||
| CRP (mg/dL) | 109 | 0.24 | ± | 0.02 a | 130 | 0.61 | ± | 0.13 b | 49 | 0.72 | ± | 0.16 b | 0.002 |
| IL-6 (pg/mL) | 109 | 2.18 | ± | 0.17 a | 130 | 4.10 | ± | 0.54 b | 50 | 6.42 | ± | 1.04 c | <0.001 |
| Neopterin (nmol/L) | 106 | 8.19 | ± | 0.44 a | 128 | 10.27 | ± | 0.51 b | 48 | 14.26 | ± | 1.58 c | <0.001 |
| Tryptophan (μmol/L) | 106 | 62.99 | ± | 1.06 a | 128 | 60.31 | ± | 1.11 a | 48 | 53.92 | ± | 1.85 b | <0.001 |
| Kynurenine (μmol/L) | 106 | 2.33 | ± | 0.05 a | 128 | 2.47 | ± | 0.06 a | 48 | 2.87 | ± | 0.12 b | <0.001 |
| Kyn/Trp (μmol/mmol) | 106 | 37.94 | ± | 1.14 a | 128 | 42.28 | ± | 1.18 b | 48 | 56.20 | ± | 3.06 c | <0.001 |
| Tyrosine (μmol/L) | 106 | 85.36 | ± | 1.99 a | 128 | 79.99 | ± | 2.03 a | 48 | 75.16 | ± | 2.93 b | 0.018 |
| Phenylalanine (μmol/L) | 106 | 89.89 | ± | 1.75 a | 128 | 89.10 | ± | 1.64 a | 48 | 85.68 | ± | 2.86 a | 0.413 |
| Phe/Tyr (μmol/μmol) | 106 | 1.08 | ± | 0.02 a | 128 | 1.17 | ± | 0.02 b | 49 | 1.17 | ± | 0.03 a,b | 0.012 |
| Nitrite (μmol/L) | 102 | 26.33 | ± | 1.96 a | 125 | 24.15 | ± | 1.68 a | 39 | 26.83 | ± | 3.29 a | 0.783 |
| Vitamin A (mg/L) | 109 | 0.52 | ± | 0.01 a | 129 | 0.49 | ± | 0.01 a | 49 | 0.50 | ± | 0.03 a | 0.323 |
| Vitamin E (mg/L) | 109 | 14.11 | ± | 0.27 a | 129 | 13.51 | ± | 0.26 a,b | 49 | 12.86 | ± | 0.48 b | 0.046 |
| Net FPG-sensitive sites (%tDNA) | 107 | 9.37 | ± | 0.47 a,b | 127 | 10.15 | ± | 0.49 a | 48 | 8.11 | ± | 0.71 b | 0.024 |
| Tyrosine | Net FPG-Sensitive Sites | |||
|---|---|---|---|---|
| Mean Ratio | 95% CI | Mean Ratio | 95% CI | |
| Fruits and vegetables consumption >2/day | ||||
| No consumption | 1.00 | 1.00 | ||
| Consumption | 1.15 * | (1.02–1.29) | 0.69 ** | (0.53–0.89) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teixeira-Gomes, A.; Laffon, B.; Valdiglesias, V.; Gostner, J.M.; Felder, T.; Costa, C.; Madureira, J.; Fuchs, D.; Teixeira, J.P.; Costa, S. Exploring Early Detection of Frailty Syndrome in Older Adults: Evaluation of Oxi-Immune Markers, Clinical Parameters and Modifiable Risk Factors. Antioxidants 2021, 10, 1975. https://doi.org/10.3390/antiox10121975
Teixeira-Gomes A, Laffon B, Valdiglesias V, Gostner JM, Felder T, Costa C, Madureira J, Fuchs D, Teixeira JP, Costa S. Exploring Early Detection of Frailty Syndrome in Older Adults: Evaluation of Oxi-Immune Markers, Clinical Parameters and Modifiable Risk Factors. Antioxidants. 2021; 10(12):1975. https://doi.org/10.3390/antiox10121975
Chicago/Turabian StyleTeixeira-Gomes, Armanda, Blanca Laffon, Vanessa Valdiglesias, Johanna M. Gostner, Thomas Felder, Carla Costa, Joana Madureira, Dietmar Fuchs, João Paulo Teixeira, and Solange Costa. 2021. "Exploring Early Detection of Frailty Syndrome in Older Adults: Evaluation of Oxi-Immune Markers, Clinical Parameters and Modifiable Risk Factors" Antioxidants 10, no. 12: 1975. https://doi.org/10.3390/antiox10121975
APA StyleTeixeira-Gomes, A., Laffon, B., Valdiglesias, V., Gostner, J. M., Felder, T., Costa, C., Madureira, J., Fuchs, D., Teixeira, J. P., & Costa, S. (2021). Exploring Early Detection of Frailty Syndrome in Older Adults: Evaluation of Oxi-Immune Markers, Clinical Parameters and Modifiable Risk Factors. Antioxidants, 10(12), 1975. https://doi.org/10.3390/antiox10121975










