Taxifolin as a Promising Ingredient of Cosmetics for Adult Skin
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Study Groups
2.3. Methods
2.3.1. Tape-Stripping Procedure with Electrospray Ionization Mass Spectrometry
2.3.2. Confocal Raman Spectroscopy
2.3.3. Patch Test
2.3.4. Biophysical and Biomechanical Skin Properties
2.3.5. Statistical Analysis
3. Results
3.1. Tape-Stripping Procedure with Electrospray Ionization Mass Spectrometry
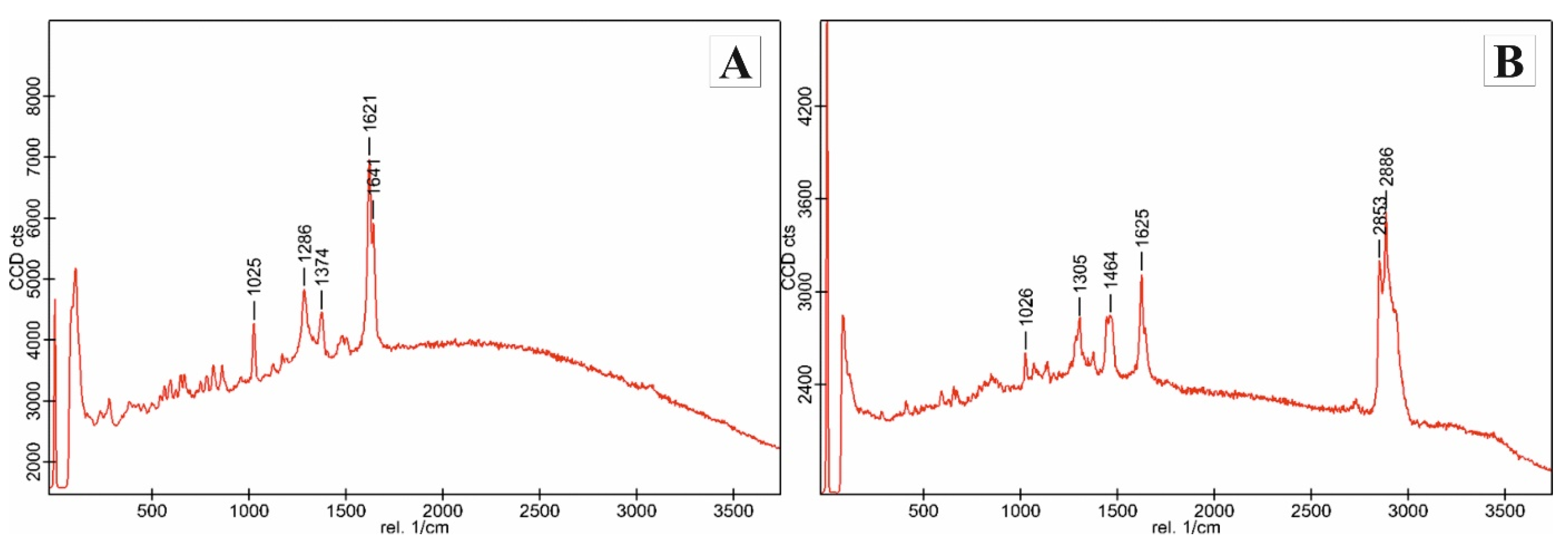
3.2. Confocal Raman Spectroscopy
3.3. Patch Testing
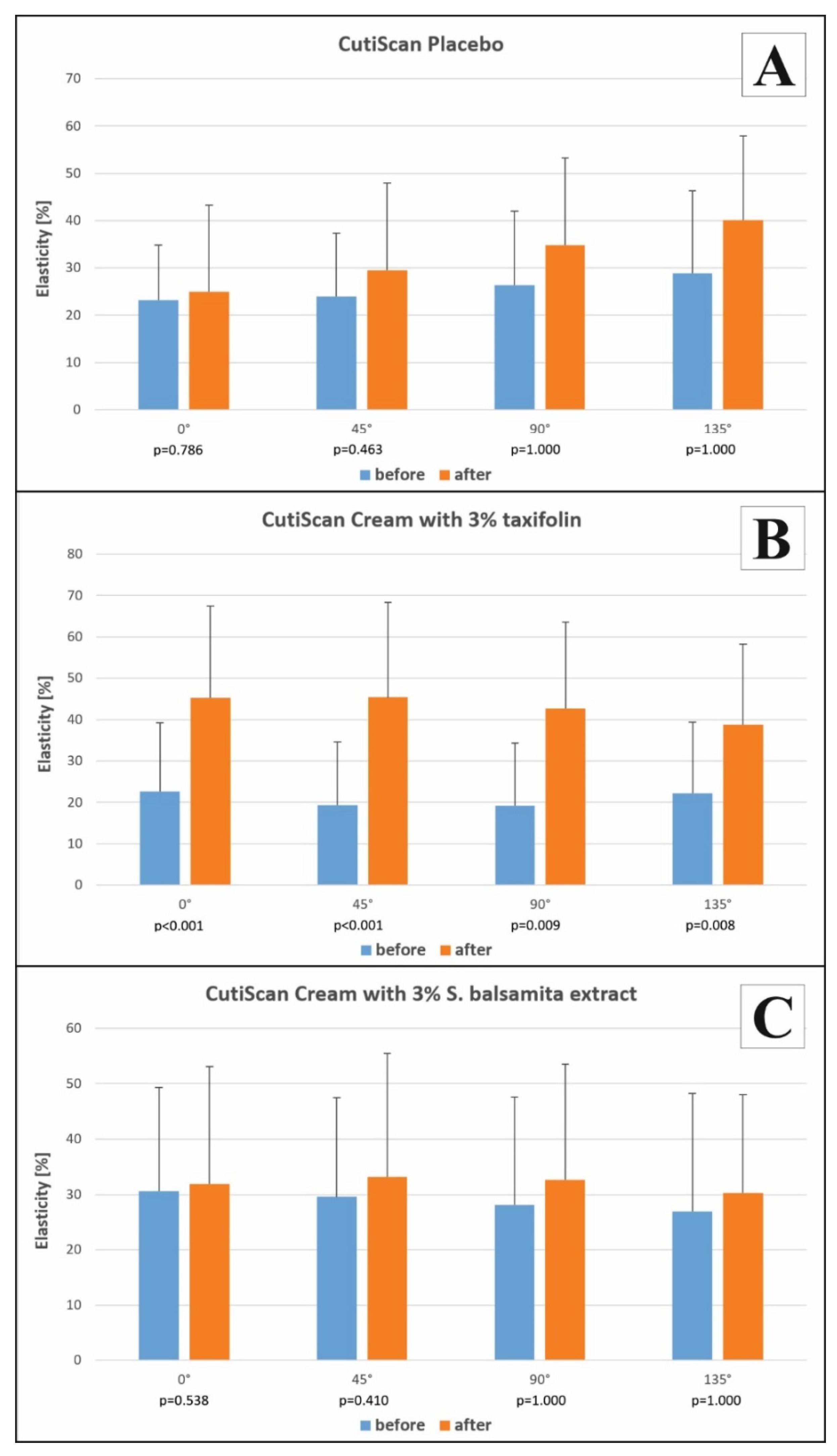
3.4. Biophysical and Biomechanical Skin Properties
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Boer, M.; Duchnik, E.; Maleszka, R.; Marchlewicz, M. Structural and biophysical characteristics of human skin in maintaining proper epidermal barrier function. Post. Dermatol. Alergol. 2016, 33, 1–5. [Google Scholar] [CrossRef]
- Kalia, Y.N.; Pirot, F.; Guy, R.H. Homogeneous transport in a heterogeneous membrane: Water diffusion across human stratum corneum in vivo. Biophys. J. 1996, 71, 2692–2700. [Google Scholar] [CrossRef]
- Parrado, C.; Mercado-Saenz, S.; Perez-Davo, A.; Gilaberte, Y.; Gonzalez, S.; Juarranz, A. Environmental Stressors on Skin Aging. Mechanistic Insights. Front. Pharmacol. 2019, 10, 759. [Google Scholar] [CrossRef]
- Darlenski, R.; Sassning, S.; Tsankov, N.; Fluhr, J.W. Non-invasive in vivo methods for investigation of the skin barrier physical properties. Eur. J. Pharm. Biopharm. 2009, 72, 295–303. [Google Scholar] [CrossRef]
- Rittié, L.; Fisher, G.J. Natural and sun-induced aging of human skin. Cold Spring Harb. Perspect. Med. 2015, 5, a015370. [Google Scholar] [CrossRef]
- Lu, Y.; Tonissen, K.F.; Di Trapani, G. Modulating skin colour: Role of the thioredoxin and glutathione systems in regulating melanogenesis. Biosci. Rep. 2021, 41, BSR20210427. [Google Scholar] [CrossRef] [PubMed]
- Vertuani, S.; Ziosi, P.; Solaroli, N.; Buzzoni, V.; Carli, M.; Lucchi, E.; Valgimigli, L.; Baratto, G.; Manfredini, S. Determination of antioxidant efficacy of cosmetic formulations by non-invasive measurements. Skin Res. Technol. 2003, 9, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Poljsak, B.; Dahmane, R.G.; Godic, A. Intrinsic skin aging: The role of oxidative stress. Acta Dermatovenerol. Alp. Panon. Adriat. 2012, 21, 33–36. [Google Scholar]
- Kammeyer, A.; Luiten, R.M. Oxidation events and skin aging. Aging Res. Rev. 2015, 21, 16–29. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.I.; Choi, S.; Roh, W.S.; Lee, J.H.; Kim, T.G. Cellular senescence and inflammaging in the skin microenvironment. Int. J. Mol. Sci. 2021, 22, 3849. [Google Scholar] [CrossRef]
- Hanasaki, Y.; Ogawa, S.; Fukui, S. The correlation between active oxygens scavenging and antioxidative effects of flavonoids. Free Radic. Biol. Med. 1994, 16, 845–850. [Google Scholar] [CrossRef]
- Panche, A.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e475. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Obaid, R.J.; Mughal, E.U.; Naeem, N.; Sadiq, A.; Alsantali, R.I.; Jassas, R.S.; Moussa, Z.; Saleh, A. Natural and synthetic flavonoid derivatives as new potential tyrosinase inhibitors: A systematic review. RSC Adv. 2021, 11, 22159–22198. [Google Scholar] [CrossRef]
- Nunes, C.; Arantes, M.B.; Pereira, S.M.; Cruz, L.L.; Passos, M.; Moraes, L.P.; Vieira, I.J.C.; Oliveira, D.B. Plants as sources of anti-inflammatory agents. Molecules 2020, 25, 3726. [Google Scholar] [CrossRef] [PubMed]
- Ullah, A.; Munir, S.; Badshah, S.L.; Khan, N.; Ghani, L.; Poulson, B.G.; Emwas, A.-H.; Jaremko, M. Important flavonoids and their role as therapeutic agent. Molecules 2020, 25, 5243. [Google Scholar] [CrossRef]
- Bito, T.; Roy, S.; Sen, C.K.; Shirakawa, T.; Gotoh, A.; Ueda, M.; Ichihashi, M.; Packer, L. Flavonoids differentially regulate IFNgamma-induced ICAM-1 expression in human keratinocytes: Molecular mechanism of action. FEBS Lett. 2002, 520, 145–152. [Google Scholar] [CrossRef] [Green Version]
- Topal, F.; Nar, M.; Gocer, H.; Kalin, P.; Kocyigit, U.M.; Gülçin, İ.; Alwasel, S.H. Antioxidant activity of taxifolin: An activity-structure relationship. J. Enzyme Inhib. Med. Chem. 2016, 31, 674–683. [Google Scholar] [CrossRef]
- Drouet, S.; Leclerc, E.A.; Garros, L.; Tungmunnithum, D.; Kabra, A.; Abbasi, B.H.; Lainé, É.; Hano, C. A green ultrasound-assisted extraction optimization of the natural antioxidant and anti-aging flavonolignans from milk thistle Silybum marianum (L.) Gaertn. Fruits for cosmetic applications. Antioxidants 2019, 8, 304. [Google Scholar] [CrossRef] [Green Version]
- Lee, C.W.; Park, N.H.; Kim, J.W.; Um, B.H.; Shpatov, A.V.; Shults, E.E.; Sorokina, I.V.; Popov, S.A. Study of skin anti-aging and anti-inflammatory effects of dihydroquercetin, natural terpenoids and their synthetic derivatives. Russ. J. Bioorg. Chem. 2012, 38, 328–3334. [Google Scholar] [CrossRef]
- An, S.M.; Kim, H.J.; Kim, J.-E.; Boo, Y.C. Flavonoooids, taxifolin and luteolin attenuate cellular melanogenesis despite increasing tyrosinase protein levels. Phytother. Res. 2008, 22, 1200–1207. [Google Scholar] [CrossRef]
- Pantouris, G.; Mowat, C.G. Antitumour agents as inhibitors of tryptophan 2,3-dioxygenase. Biooochem. Biophys. Res. Commun. 2014, 443, 28–31. [Google Scholar] [CrossRef]
- Wang, Y.H.; Wang, W.Y.; Chang, C.C.; Liou, K.T.; Sung, Y.J.; Liao, J.F.; Chen, C.F.; Chang, S.; Hou, Y.C.; Chou, Y.C.; et al. Taxifolin ameliorates cerebral ischemia-reperfusion injury in rats through its anti-oxidative effect and modulation of NF-kappa B activation. J. Biomed. Sci. 2006, 13, 127–141. [Google Scholar] [CrossRef] [Green Version]
- Oi, N.; Chen, H.; Kim, M.O.; Lubet, R.A.; Bode, A.M.; Dong, Z. Taxifolin suppresses UV-induced skin carcinogenesis by targeting EGFR and PI3K. Cancer Prev. Res. 2012, 5, 1103–1114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, S.B.; Cha, K.H.; Selenge, D.; Solongo, A.; Nho, C.W. The chemopreventive effect of taxifolins exerted through ARE-dependent gene regulation. Biol. Pharm. Bull. 2007, 30, 1074–1079. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, J.Y.; Lee, O.S.; Ha, S.; Kim, J.H.; Park, G.; Kim, J.K.; Oh, C.H. In vivo assessment of the effect of taxifolin glycoside on atopic dermatitis-like skin lesions using biomedical tools in NC/Nga mice. Clin. Exp. Dermatol. 2015, 40, 547–555. [Google Scholar] [CrossRef] [PubMed]
- Shubina, V.S.; Shatalin, Y.V. Skin regeneration after chemical burn under the effect of taxifolin-based preparations. Bull. Exp. Biol. Med. 2012, 154, 152–157. [Google Scholar] [CrossRef] [PubMed]
- Shubina, V.S.; Shatalin, Y.V. Effect of the liposomal form of flavonoid-metal complexes on skin regeneration after chemical burn. Tsitologiya 2012, 54, 251–260. [Google Scholar] [CrossRef]
- Slimestad, R.; Fossen, T.; Vagen, I.M. Onions: A source of unique dietary flavonoids. J. Agric. Food Chem. 2007, 55, 10067–10080. [Google Scholar] [CrossRef]
- Wallace, S.N.; Carrier, D.J.; Clausen, E.C. Batch slvent extraction of flavanolignans from milk thistle (Silybummarianum L. Gaertner). Phytochem. Anal. 2005, 16, 7–16. [Google Scholar] [CrossRef]
- Rohdewald, P. A review of the french maritime pine bark extract (Pycnogenol), a herbal medication with diverse clinical pharmacology. Int. J. Clin. Pharmacol. Ther. 2002, 40, 158–168. [Google Scholar] [CrossRef] [PubMed]
- Kiehlmann, E.; Li, E.P.M. Isomerization of dihydroquercetin. J. Nat. Prod. 1995, 58, 450–455. [Google Scholar] [CrossRef]
- Nawrot, J.; Budzianowski, J.; Nowak, G. Phytochemical profiles of the leaves of Stizolophus balsamita and Psephellu ssibiricus and their chemotaxonomic implications. Phytochemistry 2019, 159, 172–178. [Google Scholar] [CrossRef]
- Nawrot, J.; Gornowicz-Porowska, J.; Nowak, G. Phytotherapy perspectives for treating fungal infections, migraine, sebhorreic dermatitis and hyperpigmentations with the plants of the Centaureinae subtribe (Asteraceae). Molecules 2020, 25, 5329. [Google Scholar] [CrossRef] [PubMed]
- Nawrot, J.; Budzianowski, J.; Nowak, G.; Micek, I.; Budzianowska, A.; Gornowicz-Porowska, J. Biologically active compounds in Stizolophus balsamita Inflorescences: Isolation, phytochemical characterization and effects on the skin biophysical parameters. Int. J. Mol. Sci. 2021, 22, 4428. [Google Scholar] [CrossRef] [PubMed]
- Philips, N.; Conte, J.; Chen, Y.J.; Natrajan, P.; Taw, M.; Keller, T.; Givant, J.; Tuason, M.; Dulaj, L.; Leonardi, D.; et al. Beneficial regulation of matrix metalloproteinases and their inhibitors, fibrillar collagens and transforming growth factor-β by Polypodium leucotomos, directly or in dermal fibroblasts, ultraviolet radiated fibroblasts, and melanoma cells. Arch. Dermatol. Res. 2009, 301, 487–495. [Google Scholar] [CrossRef] [PubMed]
- Katiyar, S.K.; Ahmad, N.; Mukhtar, H. Green tea and skin. Arch. Dermatol. 2000, 136, 989–994. [Google Scholar] [CrossRef]
- Wen, K.C.; Fan, P.C.; Tsai, S.Y.; Shih, I.C.; Chiang, H.M. Ixora parviflora protects against UVB-induced photoaging by inhibiting the expression of mmps, map kinases, and cox-2 and by promoting type I procollagen synthesis. Evid. -Based Complement. Altern. Med. 2012, 2012, 417346. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chiang, H.M.; Lin, T.J.; Chiu, C.Y.; Chang, C.W.; Hsu, K.C.; Fan, P.C.; Wen, K.C. Coffea arabica extract and its constituents prevent photoaging by suppressing MMPs expression and MAP kinase pathway. Food Chem. Toxicol. 2011, 49, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.K.; Kim, D.H.; Kim, J.W.; Ngadiran, S.; Sarmidi, M.R.; Park, C.S. Labisia pumila extract protects skin cells from photoaging caused by UVB irradiation. J. Biosci. Bioeng. 2010, 109, 291–296. [Google Scholar] [CrossRef]
- Ahmed, I.A.; Mikail, M.A.; Zamakshshari, N.; Abdullah, A.H. Natural anti-aging skincare: Role and potential. Biogerontology 2020, 21, 293–310. [Google Scholar] [CrossRef]
- Wagenitz, G. Flora Iranica: Compositae III-Cynarae. In Akademische Druck Und Verlagsanstalt; Dittrich, M., Rechinger, K.H., Petrak, F., Wagenitz, G., Eds.; Akademische Druck Und Verlagsanstalt: Graz, Austria, 1980; p. 332. [Google Scholar]
- Tutin, T.G.; Heywood, V.H.; Burges, N.A.; Moore, D.M.; Valentine, D.H.; Walters, S.M.; Weeb, D.A. Flora Europaea; Cambridge University Press: Cambridge, UK; London, UK; New York, NY, USA; Melbourne, Australia, 1964; Volume 4, p. 297. [Google Scholar]
- Fitzpatrick, T.B. Soleil et peau [Sun and skin]. J. Méd. Esthét. 1975, 2, 33–34. [Google Scholar]
- Escobar-Chávez, J.J.; Merino-Sanjuán, V.; López-Cervantes, M.; Urban-Morlan, Z.; Piñón-Segundo, E.; Quintanar-Guerrero, D.; Ganem-Quintanar, A. The tape-stripping technique as a method for drug quantification in skin. J. Pharm. Pharm. Sci. 2008, 11, 104–130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lademann, J.; Jacobi, U.; Surber, C.; Weigmann, H.J.; Fluhr, J.W. The tape stripping procedure--evaluation of some critical parameters. Eur. J. Pharm. Biopharm. 2009, 72, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Ono, S.; Eda, N.; Mori, T.; Otsuka, A.; Nakamura, N.; Inai, Y.; Ota, N.; Akama, T. Tape stripping method is useful for the quantification of antimicrobial peptides on the human skin surface including the stratum corneum. Sci. Rep. 2020, 10, 15259. [Google Scholar] [CrossRef] [PubMed]
- Hughes, A.J.; Tawfik, S.S.; Baruah, K.P.; O’Toole, E.A.; O’Shaughnessy, R.F.L. Tape strips in dermatology research*. Br. J. Dermatol. 2021, 185, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Tippavajhala, V.K.; de Oliveira Mendes, T.; Martin, A.A. In Vivo Human Skin Penetration Study of Sunscreens by Confocal Raman Spectroscopy. AAPS Pharm. Sci. Tech. 2018, 19, 753–760. [Google Scholar] [CrossRef]
- Johansen, J.D.; Aalto-Korte, K.; Agner, T.; Andersen, K.E.; Bircher, A.; Bruze, M.; Cannavó, A.; Giménez-Arnau, A.; Gonçalo, M.; Goossens, A.; et al. European Society of Contact Dermatitis guideline for diagnostic patch testing—recommendations on best practice. Contact Dermat. 2015, 73, 195–221. [Google Scholar] [CrossRef] [PubMed]
- du Plessis, J.; Stefaniak, A.; Eloff, F.; John, S.; Agner, T.; Chou, T.C.; Nixon, R.; Steiner, M.; Franken, A.; Kudla, I.; et al. International guidelines for the in vivo assessment of skin properties in non-clinical settings: Part 2. transepidermal water loss and skin hydration. Skin Res. Technol. 2013, 19, 265–278. [Google Scholar] [CrossRef] [Green Version]
- Berardesca, E.; Loden, M.; Serup, J.; Masson, P.; Rodrigues, L.M. The revised EEMCO guidance for the in vivo measurement of water in the skin. Skin Res. Technol. 2018, 24, 351–358. [Google Scholar] [CrossRef]
- Clarys, P.; Alewaeters, K.; Lambrecht, R.; Barel, A.O. Skin color measurements: Comparison between three instruments: The Chromameter, the DermaSpectrometer and the Mexameter. Skin Res. Technol. 2000, 6, 230–238. [Google Scholar] [CrossRef]
- Ly, B.C.K.; Dyer, E.B.; Feig, J.L.; Chien, A.L.; Del Bino, S. Research techniques made simple: Cutaneous colorimetry: A reliable technique for objective skin color measurement. J. Investig. Dermatol. 2020, 140, 3–12.e1. [Google Scholar] [CrossRef] [Green Version]
- Del Bino, S.; Bernerd, F. Variations in skin colour and the biological consequences of ultraviolet radiation exposure. Br. J. Dermatol. 2013, 169, 33–40. [Google Scholar] [CrossRef] [Green Version]
- Courage+Khazaka Electronic GmbH. Information and Operating Instruction for the CutiScan CS 100. 2014, pp. 1–21. Available online: http://www.courage-khazaka.de (accessed on 12 January 2021).
- Rosado, C.; Antunes, F.; Barbosa, R.; Fernando, R.; Estudante, M.; Silva, H.N.; Rodrigues, L.M. About the in vivo quantitation of skin anisotropy. Skin Res. Technol. 2016, 23, 429–436. [Google Scholar] [CrossRef]
- Choi, E.H. Aging of the skin barrier. Clin. Dermatol. 2019, 37, 336–345. [Google Scholar] [CrossRef]
- Blaak, J.; Wohlfart, R.; Schürer, N.Y. Treatment of aged skin with a pH 4 skin care product normalizes increased skin surface pH and improves barrier function: Results of a pilot study. J. Cosmet. Dermatol. Sci. Appl. 2011, 1, 50–58. [Google Scholar] [CrossRef] [Green Version]
- Gao, Y.L.; Jia, X.X.; Wang, M.; Hua, Y.; Zheng, H.; Xiang, W.Z.; Song, X.Z. Melanocyte activation and skin barrier disruption induced in melasma patients after 1064 nm Nd:YAG laser treatment. Lasers Med. Sci. 2019, 4, 767–771. [Google Scholar] [CrossRef] [PubMed]
- Man, M.Q.; Lin, T.K.; Santiago, J.L.; Celli, A.; Zhong, L.; Huang, Z.M.; Roelandt, T.; Hupe, M.; Sundberg, J.P.; Silva, K.A.; et al. Basis for enhanced barrier function of pigmented skin. J. Investig. Dermatol. 2014, 134, 2399–2407. [Google Scholar] [CrossRef] [Green Version]
- Lee, D.J.; Lee, J.; Ha, J.; Park, K.C.; Ortonne, J.P.; Kang, H.Y. Defective barrier function in melasma skin. J. Eur. Acad. Dermatol. Venereol. 2012, 26, 1533–1537. [Google Scholar] [CrossRef]
- Landriscina, A.; Rosen, J.; Friedman, A.J. Nanotechnology, inflammation and the skin barrier: Innovative approaches for skin health and cosmesis. Cosmetics 2015, 2, 177–186. [Google Scholar] [CrossRef] [Green Version]
- D’Angelo Costa, G.M.; Maia Campos, P.M.B.G. Efficacy of topical antioxidants in the skin hyperpigmentation control: A clinical study by reflectance confocal microscopy. J. Cosmet. Dermatol. 2021, 20, 538–545. [Google Scholar] [CrossRef] [PubMed]
- Babbush, K.M.; Babbush, R.A.; Khachemoune, A. The therapeutic use of antioxidants for melasma. J. Drugs Dermatol. 2020, 19, 788–792. [Google Scholar] [CrossRef] [PubMed]
- Nahhas, A.F.; Abdel-Malek, Z.A.; Kohli, I.; Braunberger, T.L.; Lim, H.W.; Hamzavi, I.H. The potential role of antioxidants in mitigating skin hyperpigmentation resulting from ultraviolet and visible light-induced oxidative stress. Photodermatol. Photoimmunol. Photomed. 2019, 35, 420–428. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mori, S.; Shiraishi, A.; Epplen, K.; Butcher, D.; Murase, D.; Yasuda, Y.; Murase, T. Characterization of skin function associated with obesity and specific correlation to local/systemic parameters in American women. Lipids Health Dis. 2017, 16, 214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Majewski, S.; Pietrzak, A.; Tworek, D.; Szewczyk, K.; Kumor-Kisielewska, A.; Kurmanowska, Z.; Górski, P.; Zalewska-Janowska, A.; Piotrowski, W.J. Skin condition and its relationship to systemic inflammation in chronic obstructive pulmonary disease. Int. J. Chron. Obstruct. Pulmon. Dis. 2017, 12, 2407–2415. [Google Scholar] [CrossRef] [Green Version]
- Amano, S. Characterization and mechanisms of photoaging-related changes in skin. Damages of basement membrane and dermal structures. Exp. Dermatol. 2016, 25, 14–19. [Google Scholar] [CrossRef]
- Monteiro Rodrigues, L.; Fluhr, J.W.; the EEMCO Group. EEMCO Guidance for the in vivo Assessment of Biomechanical Properties of the Human Skin and Its Annexes: Revisiting Instrumentation and Test Modes. Skin Pharmacol. Physiol. 2020, 33, 44–60. [Google Scholar] [CrossRef]

| TXF Concentration (mg/mL) | Average Value | Signal Intensity m/z 302 | ||
|---|---|---|---|---|
| 2.500 | 2.24 × 107 | 2.22 × 107 | 2.20 × 107 | 2.29 × 107 |
| 1.568 | 1.40 × 107 | 1.40 × 107 | 1.44 × 107 | 1.37 × 107 |
| 0.941 | 8.42 × 106 | 8.09 × 106 | 8.82 × 106 | 8.36 × 106 |
| 0.272 | 2.44 × 106 | 2.35 × 106 | 2.42 × 106 | 2.55 × 106 |
| 0.092 | 8.27 × 105 | 8.28 × 105 | 8.33 × 105 | 8.19 × 105 |
| 0.001 | 1.24 × 104 | 1.10 × 104 | 1.23 × 104 | 1.40 × 104 |
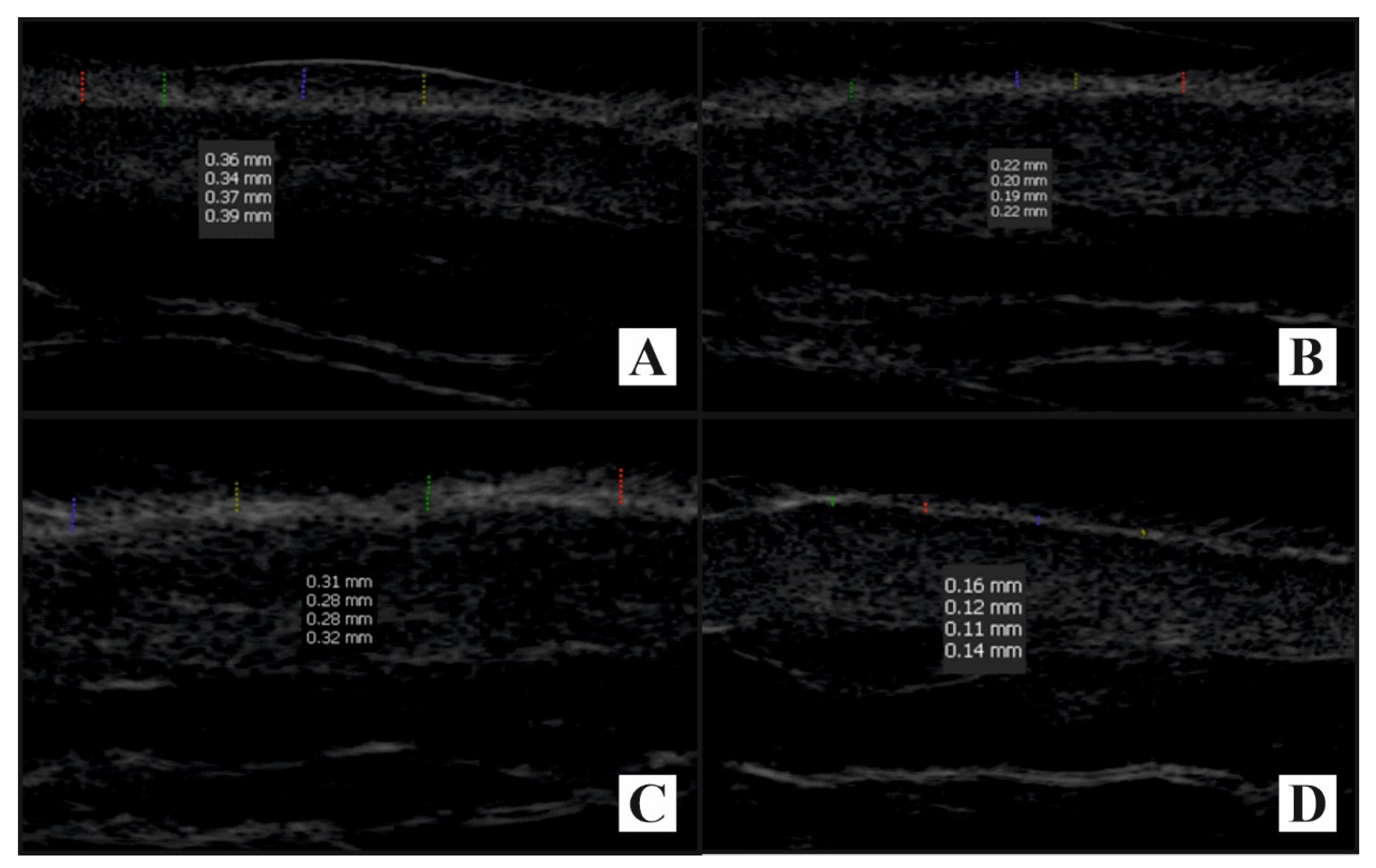
| Cream with 3% of Taxifolin (Quantity Applied: 26.00 mg/cm2) | Cream with 3% of S. balsamita Extract (Quantity Applied: 26.00 mg/cm2) | ||||
|---|---|---|---|---|---|
| No. of Layer | Thickness of Stripped Epidermis [mm] | Subject 1 | Subject 2 | Subject 1 | Subject 2 |
| 1 | 0.017 | 0.323 ± 0.006 | 0.358 ± 0.006 | 0.416 ± 0.006 | 0.422 ± 0.006 |
| 2 | 0.034 | 0.295 ± 0.010 | 0.337 ± 0.027 | 0.016 ± 0.006 | 0.010 ± 0.006 |
| 3 | 0.051 | 0.284 ± 0.003 | 0.285 ± 0.010 | 0.014 ± 0.006 | 0.004 ± 0.006 |
| 4 | 0.068 | 0.270 ± 0.003 | 0.228 ± 0.006 | 0.014 ± 0.006 | 0.002 ± 0.006 |
| 5 | 0.085 | 0.225 ± 0.025 | 0.156 ± 0.003 | 0.005 ± 0.006 | 0.002 ± 0.006 |
| 6 | 0.102 | 0.089 ± 0.022 | 0.089 ± 0.001 | 0.002 ± 0.006 | nd |
| 7 | 0.119 | 0.032 ± 0.002 | 0.022 ± 0.001 | nd | nd |
| 8 | 0.136 | nd | nd | nd | nd |
| 9 | 0.153 | nd | nd | nd | nd |
| 10 | 0.170 | nd | nd | nd | nd |
| Taxifolin quantity determined on the tapes | 1.518 mg | 1.475 mg | 0.467 mg | 0.440 mg | |
| Suspected Cosmetics | Patients Tested | Positivity | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Eczema Group (n) | Healthy Group (n) | Total (n) | Eczema Group (n) | Healthy Group (n) | Total (n) | ||||
| After 48 h | After 72 h | After 48 h | After 72 h | After 48 h | After 72 h | ||||
| 3% S. balsamita extract cream | 18 | 17 | 35 | 0 | 0 | 0 | 0 | 0 | 0 |
| 3% TXF cream | 18 | 17 | 35 | 0 | 0 | 0 | 0 | 0 | 0 |
| Parameter | 3% S. balsamita Extraxt Cream (n = 20) | 3% Taxifolin Cream (n = 20) | Placebo (n = 20) | Placebo Versus 3% S. balsamita Extract Cream | Placebo Versus 3% Taxifolin Cream (p Value) | |
|---|---|---|---|---|---|---|
| Melanin Index (AU) | Before treatment Mean ± SD | 120.0 ± 29.4 | 135.6 ± 22.1 | 125.1 ± 28.5 | ||
| After treatment Mean ± SD | 114.7 ± 30.7 | 129.0 ± 22.0 | 125.8 ± 36.3 | |||
| p value | 0.0618 * | 0.0206 * | 0.8728 | 0.3408 | 0.2012 | |
| Erythema Index (AU) | Before treatment Mean ± SD | 372.9 ± 75.9 | 345.7 ± 75.0 | 349.6 ± 58.2 | ||
| After treatment Mean ± SD | 342.6 ± 72.0 | 337.6 ± 72.4 | 354.6 ± 77.8 | |||
| p value | 0.0069 * | 0.3846 | 0.6407 | 0.0430 * | 0.5117 | |
| Brightness L* (AU) | Before treatment Mean ± SD | 62.8 ± 4.1 | 63.2 ± 3.3 | 63.9 ± 3.2 | ||
| After treatment Mean ± SD | 64.5 ± 3.3 | 64.0 ± 2.9 | 63.3 ± 3.2 | |||
| p value | 0.0028 * | 0.2712 | 0.1167 | 0.0005 * | 0.0402 * | |
| Red intensity a* (AU) | Before treatment Mean ± SD | 11.9 ± 1.9 | 11.1 ± 2.2 | 10.7 ± 1.5 | ||
| After treatment Mean ± SD | 10.9 ± 1.6 | 10.5 ± 1.9 | 10.9 ± 1.5 | |||
| p value | 0.0706 * | 0.1459 | 0.1261 | 0.0283 * | 0.9893 | |
| Yellow intensity b* (AU) | Before treatment Mean ± SD | 11.1 ± 1.1 | 11.9 ± 1.9 | 11.4 ± 2.4 | ||
| After treatment Mean ± SD | 11.6 ± 1.5 | 11.3 ± 1.4 | 11.1 ± 2.4 | |||
| p value | 0.0034 * | 0.1432 | 0.3898 | 0.0032 * | 0.0375 * | |
| Individual typological angle ITA (°) | Before treatment Mean ± SD | 47.4 ± 11.4 | 46.7 ± 6.6 | 50.2 ± 8.6 | ||
| After treatment Mean ± SD | 50.5 ± 7.2 | 50.6 ± 4.9 | 49.8 ± 8.8 | |||
| p value | 0.0231 * | 0.0111 * | 0.7141 | 0.0167 * | 0.0080 * | |
| pH | Before treatment Mean ± SD | 5.5 ± 0.4 | 5.4 ± 0.4 | 5.2 ± 0.5 | ||
| After treatment Mean ± SD | 5.3 ± 0.5 | 5.6 ± 0.3 | 5.5 ± 0.4 | |||
| p value | 0.0323 * | 0.0352 * | 0.0127 * | 0.0014 * | 0.6017 | |
| Skin temperature in °C | Before treatment Mean ± SDAfter treatment Mean ± SD | 30.6 ± 1.729.6 ± 1.3 | 30.5 ± 1.329.8 ± 1.3 | 30.2 ± 1.230.0 ± 1.5 | ||
| p value | 0.0468 * | 0.0314 * | 0.5511 | 0.2766 | 0.3834 | |
| Diffuse reflected/scattered light (AU) | Before treatment Mean ± SD | 25.3 ± 2.0 | 24.6 ± 3.0 | 26.0 ± 3.4 | ||
| After treatment Mean ± SD | 26.0 ± 2.8 | 26.0 ± 2.8 | 25.8 ± 3.4 | |||
| p value | 0.1432 | 0.0485 * | 0.9019 | 0.4945 | 0.0460 * | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Micek, I.; Nawrot, J.; Seraszek-Jaros, A.; Jenerowicz, D.; Schroeder, G.; Spiżewski, T.; Suchan, A.; Pawlaczyk, M.; Gornowicz-Porowska, J. Taxifolin as a Promising Ingredient of Cosmetics for Adult Skin. Antioxidants 2021, 10, 1625. https://doi.org/10.3390/antiox10101625
Micek I, Nawrot J, Seraszek-Jaros A, Jenerowicz D, Schroeder G, Spiżewski T, Suchan A, Pawlaczyk M, Gornowicz-Porowska J. Taxifolin as a Promising Ingredient of Cosmetics for Adult Skin. Antioxidants. 2021; 10(10):1625. https://doi.org/10.3390/antiox10101625
Chicago/Turabian StyleMicek, Iwona, Joanna Nawrot, Agnieszka Seraszek-Jaros, Dorota Jenerowicz, Grzegorz Schroeder, Tomasz Spiżewski, Adela Suchan, Mariola Pawlaczyk, and Justyna Gornowicz-Porowska. 2021. "Taxifolin as a Promising Ingredient of Cosmetics for Adult Skin" Antioxidants 10, no. 10: 1625. https://doi.org/10.3390/antiox10101625
APA StyleMicek, I., Nawrot, J., Seraszek-Jaros, A., Jenerowicz, D., Schroeder, G., Spiżewski, T., Suchan, A., Pawlaczyk, M., & Gornowicz-Porowska, J. (2021). Taxifolin as a Promising Ingredient of Cosmetics for Adult Skin. Antioxidants, 10(10), 1625. https://doi.org/10.3390/antiox10101625






