Redox Balance in Male Infertility: Excellence through Moderation—“Μέτρον ἄριστον”
Abstract
1. Introduction
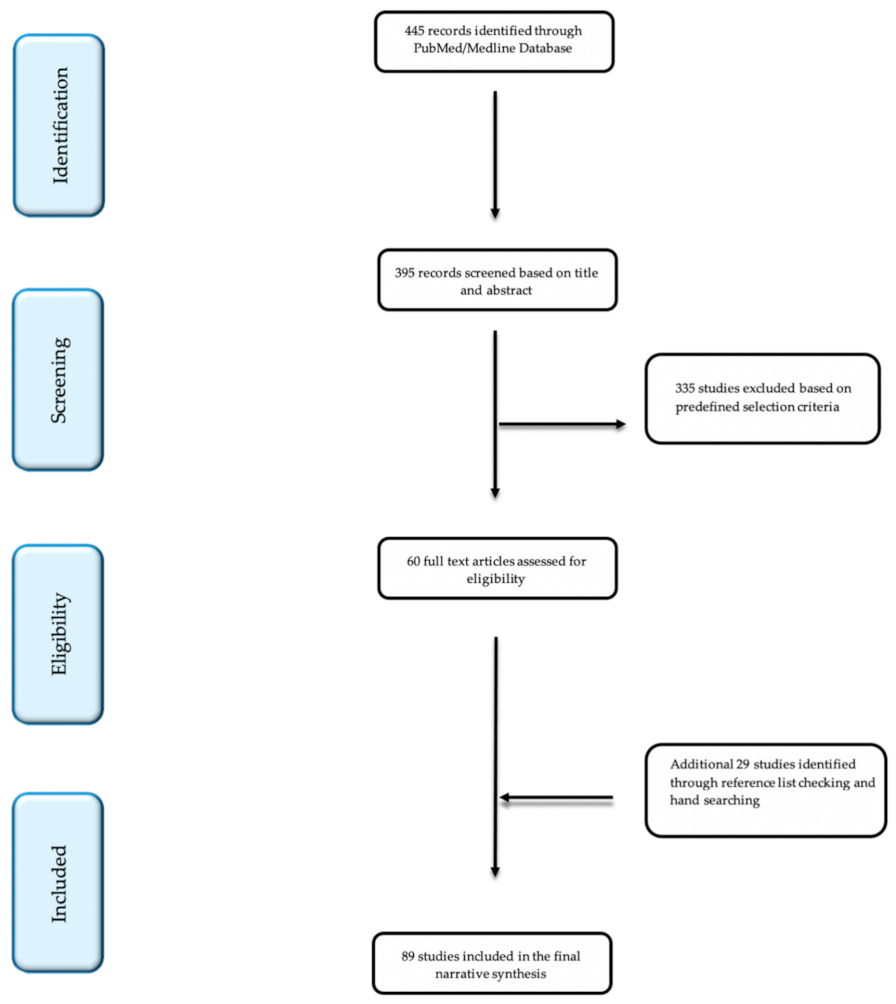
2. Search Strategy
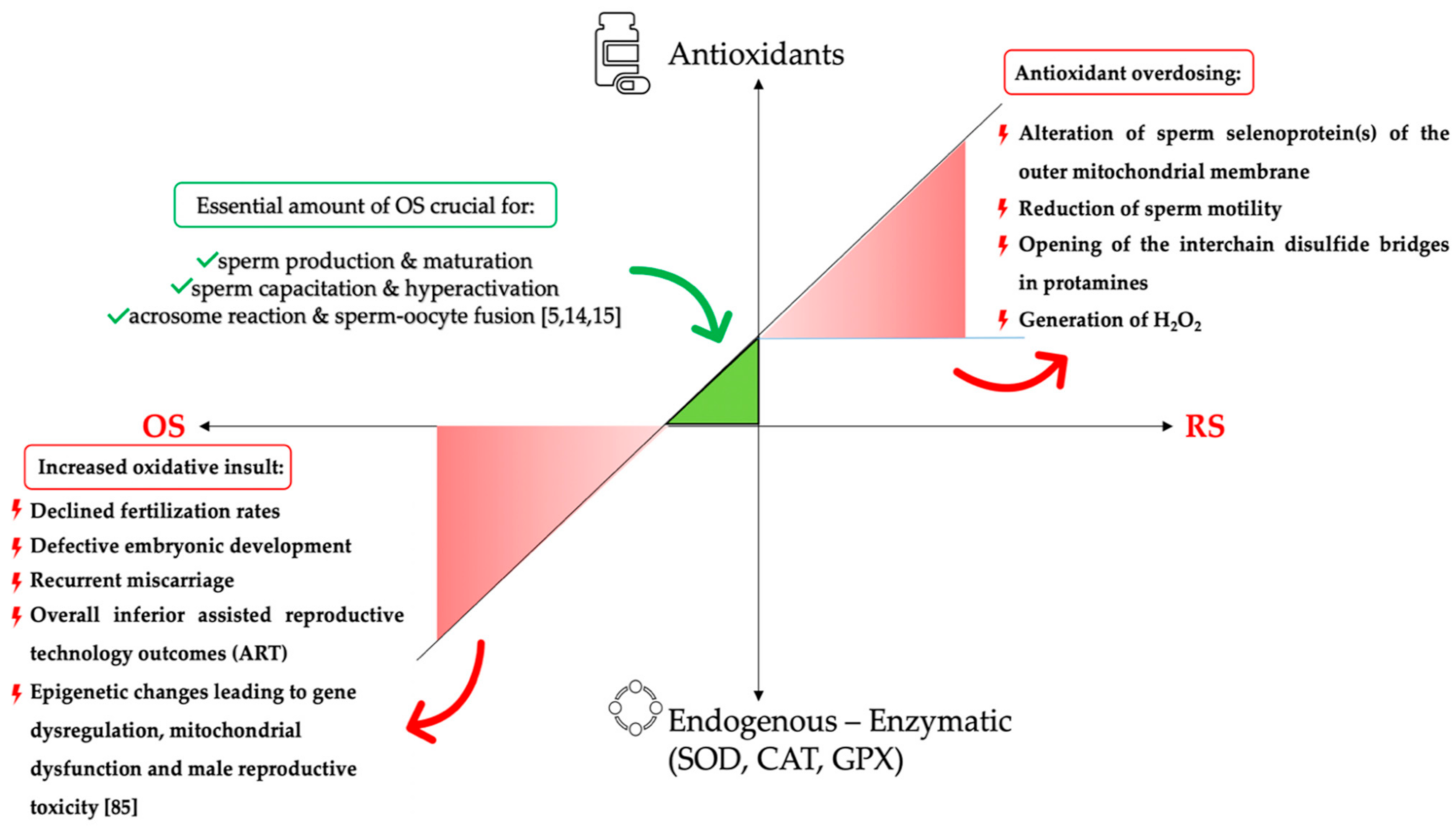
3. Redox Balance Implies Moderation-“μέτρον”
4. Antioxidant Treatment for Male Infertility—Friend or Foe?
5. Oxidation-Reduction Potential (ORP)—A Reliable Guarantor for Redox Balance?
6. Conclusions, Future Directions, and Perspectives
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Type | Number | References |
|---|---|---|
| Reviews | 43 | [1,2,3,4,5,6,7,8,13,15,17,19,21,22,23,25,26,27,28,29,30,31,32,33,35,36,37,38,39,40,41,42,43,44,46,47,48,49,68,84,86,87,88] |
| Original Research | 33 | [9,10,11,12,14,18,20,24,34,45,50,51,52,53,54,55,56,57,58,59,60,61,63,64,65,66,67,69,70,71,72,79,85] |
| Systematic Review | 7 | [62,73,75,77,78,81,82] |
| Systematic Review and Meta-analyses | 4 | [74,76,80,83] |
| Letter to the editor | 1 | [16] |
| Editorial | 1 | [89] |
| Total | 89 | |
| Hand-searched/Reference list checked | 29 | [9,10,27,28,31,34,42,47,49,50,51,52,53,54,55,58,59,60,61,63,64,65,66,67,69,70,81,82,87] |
References
- Kopa, Z.; Keszthelyi, M.; Sofikitis, N. Administration of Antioxidants in the Infertile Male: When it may have a Beneficial Effect? Curr. Pharm. Des. 2021, 27, 2665–2668. [Google Scholar] [CrossRef] [PubMed]
- Dimitriadis, F.; Tsounapi, P.; Zachariou, A.; Kaltsas, A.; Sokolakis, I.; Hatzichristodoulou, G.; Symeonidis, E.N.; Kotsiris, D.; Gabales, M.R.; Vlachopoulou, E.; et al. Therapeutic effects of micronutrient supplements on sperm parameters: Fact or Fiction? Curr. Pharm. Des. 2021, 27, 2757–2769. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Mulgund, A.; Hamada, A.; Chyatte, M.R. A unique view on male infertility around the globe. Reprod. Biol. Endocrinol. 2015, 13, 37. [Google Scholar] [CrossRef] [PubMed]
- Evans, E.P.P.; Scholten, J.T.M.; Mzyk, A.; Reyes-San-Martin, C.; Llumbet, A.E.; Hamoh, T.; Arts, E.G.J.M.; Schirhagl, R.; Cantineau, A.E.P. Male subfertility and oxidative stress. Redox Biol. 2021, 46, 102071. [Google Scholar] [CrossRef]
- Agarwal, A.; Bui, A.D. Oxidation-reduction potential as a new marker for oxidative stress: Correlation to male infertility. Investig. Clin. Urol. 2017, 58, 385–399. [Google Scholar] [CrossRef]
- Dimitriadis, F.; Symeonidis, E.N.; Tsounapi, P.; Kaltsas, A.; Hatzichristodoulou, G.; Sokolakis, I.; Zachariou, A.; Takenaka, A.; Sofikitis, N. Administration of antioxidants in the infertile male: When may it have a detrimental effect? Curr. Pharm. Des. 2021, 27, 2796–2801. [Google Scholar] [CrossRef]
- Agarwal, A.; Parekh, N.; Panner Selvam, M.K.; Henkel, R.; Shah, R.; Homa, S.T.; Ramasamy, R.; Ko, E.; Tremellen, K.; Esteves, S.; et al. Male Oxidative Stress Infertility (MOSI): Proposed Terminology and Clinical Practice Guidelines for Management of Idiopathic Male Infertility. World J. Mens Health 2019, 37, 296–312. [Google Scholar] [CrossRef]
- Baskaran, S.; Finelli, R.; Agarwal, A.; Henkel, R. Reactive oxygen species in male reproduction: A boon or a bane? Andrologia 2021, 53, e13577. [Google Scholar] [CrossRef]
- Tsounapi, P.; Honda, M.; Dimitriadis, F.; Kawamoto, B.; Hikita, K.; Muraoka, K.; Saito, M.; Sofikitis, N.; Takenaka, A. Impact of antioxidants on seminal vesicles function and fertilizing potential in diabetic rats. Asian J. Androl. 2017, 19, 639–646. [Google Scholar]
- Tsounapi, P.; Saito, M.; Dimitriadis, F.; Koukos, S.; Shimizu, S.; Satoh, K.; Takenaka, A.; Sofikitis, N. Antioxidant treatment with edaravone or taurine ameliorates diabetes-induced testicular dysfunction in the rat. Mol. Cell Biochem. 2012, 369, 195–204. [Google Scholar] [CrossRef]
- Majzoub, A.; Arafa, M.; Mahdi, M.; Agarwal, A.; Al Said, S.; Al-Emadi, I.; El Ansari, W.; Alattar, A.; Al Rumaihi, K.; Elbardisi, H. Oxidation-reduction potential and sperm DNA fragmentation, and their associations with sperm morphological anomalies amongst fertile and infertile men. Arab. J. Urol. 2018, 16, 87–95. [Google Scholar] [CrossRef]
- Agarwal, A.; Roychoudhury, S.; Sharma, R.; Gupta, S.; Majzoub, A.; Sabanegh, E. Diagnostic application of oxidation-reduction potential assay for measurement of oxidative stress: Clinical utility in male factor infertility. Reprod. Biomed. Online 2017, 34, 48–57. [Google Scholar] [CrossRef]
- Wagner, H.; Cheng, J.W.; Ko, E.Y. Role of reactive oxygen species in male infertility: An updated review of literature. Arab. J. Urol. 2018, 16, 35–43. [Google Scholar] [CrossRef]
- Homa, S.T.; Vassiliou, A.M.; Stone, J.; Killeen, A.P.; Dawkins, A.; Xie, J.; Gould, F.; Ramsay, J.W.A. A Comparison Between Two Assays for Measuring Seminal Oxidative Stress and their Relationship with Sperm DNA Fragmentation and Semen Parameters. Genes 2019, 10, 236. [Google Scholar] [CrossRef]
- De Lamirande, E.; Jiang, H.; Zini, A.; Kodama, H.; Gagnon, C. Reactive oxygen species and sperm physiology. Rev. Reprod. 1997, 2, 48–54. [Google Scholar] [CrossRef]
- Aitken, R.J. Antioxidant trials-the need to test for stress. Hum. Reprod. Open 2021, 2021, hoab007. [Google Scholar] [CrossRef]
- Amorini, A.M.; Listorti, I.; Bilotta, G.; Pallisco, R.; Saab, M.W.; Mangione, R.; Manca, B.; Lazzarino, G.; Tavazzi, B.; Lazzarino, G.; et al. Antioxidant-Based Therapies in Male Infertility: Do We Have Sufficient Evidence Supporting Their Effectiveness? Antioxidants 2021, 10, 220. [Google Scholar] [CrossRef]
- Agarwal, A.; Panner Selvam, M.K.; Arafa, M.; Okada, H.; Homa, S.; Killeen, A.; Balaban, B.; Saleh, R.; Armagan, A.; Roychoudhury, S.; et al. Multi-center evaluation of oxidation-reduction potential by the MiOXSYS in males with abnormal semen. Asian J. Androl. 2019, 21, 565–569. [Google Scholar] [CrossRef]
- Agarwal, A.; Roychoudhury, S.; Bjugstad, K.B.; Cho, C.-L. Oxidation-reduction potential of semen: What is its role in the treatment of male infertility? Ther. Adv. Urol. 2016, 8, 302–318. [Google Scholar] [CrossRef]
- Agarwal, A.; Sharma, R.; Roychoudhury, S.; Du Plessis, S.; Sabanegh, E. MiOXSYS: A novel method of measuring oxidation reduction potential in semen and seminal plasma. Fertil. Steril. 2016, 106, 566–573. [Google Scholar] [CrossRef]
- Agarwal, A.; Saleh, R.A. Role of oxidants in male infertility: Rationale, significance, and treatment. Urol. Clin. N. Am. 2002, 29, 817–827. [Google Scholar] [CrossRef]
- Torres-Arce, E.; Vizmanos, B.; Babio, N.; Márquez-Sandoval, F.; Salas-Huetos, A. Dietary Antioxidants in the Treatment of Male Infertility: Counteracting Oxidative Stress. Biology 2021, 10, 241. [Google Scholar] [CrossRef]
- Tremellen, K. Oxidative stress and male infertility—A clinical perspective. Hum. Reprod. Update 2008, 14, 243–258. [Google Scholar] [CrossRef]
- Pasqualotto, F.F.; Sundaram, A.; Sharma, R.K.; Borges, E.; Pasqualotto, E.B.; Agarwal, A. Semen quality and oxidative stress scores in fertile and infertile patients with varicocele. Fertil. Steril. 2008, 89, 602–607. [Google Scholar] [CrossRef]
- Agarwal, A.; Makker, K.; Sharma, R. Clinical relevance of oxidative stress in male factor infertility: An update. Am. J. Reprod Immunol. 2008, 59, 2–11. [Google Scholar] [CrossRef]
- Sikka, S.C. Oxidative stress and role of antioxidants in normal and abnormal sperm function. Front. Biosci. 1996, 1, e78–e86. [Google Scholar] [CrossRef]
- Otasevic, V.; Stancic, A.; Korac, A.; Jankovic, A.; Korac, B. Reactive oxygen, nitrogen, and sulfur species in human male fertility. A crossroad of cellular signaling and pathology. Biofactors 2020, 46, 206–219. [Google Scholar] [CrossRef]
- Kadlec, M.; Ros-Santaella, J.L.; Pintus, E. The Roles of NO and H2S in Sperm Biology: Recent Advances and New Perspectives. Int. J. Mol. Sci. 2020, 21, 2174. [Google Scholar] [CrossRef]
- Saleh, R.A.; Agarwal, A. Oxidative stress and male infertility: From research bench to clinical practice. J. Androl. 2002, 23, 737–752. [Google Scholar]
- Agarwal, A.; Gupta, S.; Sharma, R.K. Role of oxidative stress in female reproduction. Reprod. Biol. Endocrinol. 2005, 3, 28. [Google Scholar] [CrossRef]
- Zini, A.; Libman, J. Sperm DNA damage: Importance in the era of assisted reproduction. Curr. Opin. Urol. 2006, 16, 428–434. [Google Scholar] [CrossRef] [PubMed]
- Deepinder, F.; Chowdary, H.T.; Agarwal, A. Role of metabolomic analysis of biomarkers in the management of male infertility. Expert Rev. Mol. Diagn. 2007, 7, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Deepinder, F.; Cocuzza, M.; Agarwal, A. Should seminal oxidative stress measurement be offered routinely to men presenting for infertility evaluation? Endocr. Pract. 2008, 14, 484–491. [Google Scholar] [CrossRef] [PubMed]
- Cocuzza, M.; Athayde, K.S.; Agarwal, A.; Sharma, R.; Pagani, R.; Lucon, A.M.; Srougi, M.; Hallak, J. Age-related increase of reactive oxygen species in neat semen in healthy fertile men. Urology 2008, 71, 490–494. [Google Scholar] [CrossRef]
- Cocuzza, M.; Sikka, S.C.; Athayde, K.S.; Agarwal, A. Clinical relevance of oxidative stress and sperm chromatin damage in male infertility: An evidence based analysis. Int. Braz. J. Urol. 2007, 33, 603–621. [Google Scholar] [CrossRef]
- Fraczek, M.; Kurpisz, M. Inflammatory mediators exert toxic effects of oxidative stress on human spermatozoa. J. Androl. 2007, 28, 325–333. [Google Scholar] [CrossRef]
- Villaverde, A.I.S.B.; Netherton, J.; Baker, M.A. From Past to Present: The Link Between Reactive Oxygen Species in Sperm and Male Infertility. Antioxidants 2019, 8, 616. [Google Scholar] [CrossRef]
- Agarwal, A.; Prabakaran, S.A.; Said, T.M. Prevention of oxidative stress injury to sperm. J. Androl. 2005, 26, 654–660. [Google Scholar] [CrossRef]
- Ebisch, I.M.W.; Thomas, C.M.G.; Peters, W.H.M.; Braat, D.D.M.; Steegers-Theunissen, R.P.M. The importance of folate, zinc and antioxidants in the pathogenesis and prevention of subfertility. Hum. Reprod. Update 2007, 13, 163–174. [Google Scholar] [CrossRef]
- Agarwal, A.; Gupta, S.; Sikka, S. The role of free radicals and antioxidants in reproduction. Curr. Opin. Obstet. Gynecol. 2006, 18, 325–332. [Google Scholar] [CrossRef]
- Rashki Ghaleno, L.; Alizadeh, A.; Drevet, J.R.; Shahverdi, A.; Valojerdi, M.R. Oxidation of Sperm DNA and Male Infertility. Antioxidants 2021, 10, 97. [Google Scholar] [CrossRef]
- Park, Y.-J.; Pang, M.-G. Mitochondrial Functionality in Male Fertility: From Spermatogenesis to Fertilization. Antioxidants 2021, 10, 98. [Google Scholar] [CrossRef]
- Castellini, C.; D’Andrea, S.; Cordeschi, G.; Totaro, M.; Parisi, A.; Di Emidio, G.; Tatone, C.; Francavilla, S.; Barbonetti, A. Pathophysiology of Mitochondrial Dysfunction in Human Spermatozoa: Focus on Energetic Metabolism, Oxidative Stress and Apoptosis. Antioxidants 2021, 10, 695. [Google Scholar] [CrossRef]
- Henkel, R.; Sandhu, I.S.; Agarwal, A. The excessive use of antioxidant therapy: A possible cause of male infertility? Andrologia 2019, 51, e13162. [Google Scholar] [CrossRef]
- Panner Selvam, M.K.; Henkel, R.; Sharma, R.; Agarwal, A. Calibration of redox potential in sperm wash media and evaluation of oxidation-reduction potential values in various assisted reproductive technology culture media using MiOXSYS system. Andrology 2018, 6, 293–300. [Google Scholar] [CrossRef]
- Leisegang, K.; Henkel, R.; Agarwal, A. Redox Regulation of Fertility in Aging Male and the Role of Antioxidants: A Savior or Stressor. Curr. Pharm. Des. 2017, 23, 4438–4450. [Google Scholar] [CrossRef]
- Panner Selvam, M.K.; Agarwal, A.; Henkel, R.; Finelli, R.; Robert, K.A.; Iovine, C.; Baskaran, S. The effect of oxidative and reductive stress on semen parameters and functions of physiologically normal human spermatozoa. Free Radic. Biol. Med. 2020, 152, 375–385. [Google Scholar] [CrossRef]
- Chehab, M.; Madala, A.; Trussell, J.C. On-label and off-label drugs used in the treatment of male infertility. Fertil. Steril. 2015, 103, 595–604. [Google Scholar] [CrossRef]
- Martin-Hidalgo, D.; Bragado, M.J.; Batista, A.R.; Oliveira, P.F.; Alves, M.G. Antioxidants and Male Fertility: From Molecular Studies to Clinical Evidence. Antioxidants 2019, 8, 89. [Google Scholar] [CrossRef]
- Balercia, G.; Regoli, F.; Armeni, T.; Koverech, A.; Mantero, F.; Boscaro, M. Placebo-controlled double-blind randomized trial on the use of L-carnitine, L-acetylcarnitine, or combined L-carnitine and L-acetylcarnitine in men with idiopathic asthenozoospermia. Fertil. Steril. 2005, 84, 662–671. [Google Scholar] [CrossRef]
- Balercia, G.; Buldreghini, E.; Vignini, A.; Tiano, L.; Paggi, F.; Amoroso, S.; Ricciardo-Lamonica, G.; Boscaro, M.; Lenzi, A.; Littarru, G. Coenzyme Q10 treatment in infertile men with idiopathic asthenozoospermia: A placebo-controlled, double-blind randomized trial. Fertil. Steril. 2009, 91, 1785–1792. [Google Scholar] [CrossRef]
- Tremellen, K.; Miari, G.; Froiland, D.; Thompson, J. A randomised control trial examining the effect of an antioxidant (Menevit) on pregnancy outcome during IVF-ICSI treatment. Aust. N. Z. J. Obstet. Gynaecol. 2007, 47, 216–221. [Google Scholar] [CrossRef]
- Busetto, G.M.; Agarwal, A.; Virmani, A.; Antonini, G.; Ragonesi, G.; Del Giudice, F.; Micic, S.; Gentile, V.; De Berardinis, E. Effect of metabolic and antioxidant supplementation on sperm parameters in oligo-astheno-teratozoospermia, with and without varicocele: A double-blind placebo-controlled study. Andrologia 2018, 50, e12927. [Google Scholar] [CrossRef]
- Micic, S.; Lalic, N.; Djordjevic, D.; Bojanic, N.; Bogavac-Stanojevic, N.; Busetto, G.M.; Virmani, A.; Agarwal, A. Double-blind, randomised, placebo-controlled trial on the effect of L-carnitine and L-acetylcarnitine on sperm parameters in men with idiopathic oligoasthenozoospermia. Andrologia 2019, 51, e13267. [Google Scholar] [CrossRef]
- Safarinejad, M.R.; Safarinejad, S.; Shafiei, N.; Safarinejad, S. Effects of the reduced form of coenzyme Q10 (ubiquinol) on semen parameters in men with idiopathic infertility: A double-blind, placebo controlled, randomized study. J. Urol. 2012, 188, 526–531. [Google Scholar] [CrossRef]
- Haghighian, H.K.; Haidari, F.; Mohammadi-Asl, J.; Dadfar, M. Randomized, triple-blind, placebo-controlled clinical trial examining the effects of alpha-lipoic acid supplement on the spermatogram and seminal oxidative stress in infertile men. Fertil. Steril. 2015, 104, 318–324. [Google Scholar] [CrossRef]
- Calogero, A.E.; Gullo, G.; La Vignera, S.; Condorelli, R.A.; Vaiarelli, A. Myoinositol improves sperm parameters and serum reproductive hormones in patients with idiopathic infertility: A prospective double-blind randomized placebo-controlled study. Andrology 2015, 3, 491–495. [Google Scholar] [CrossRef]
- Mohammadi, F.; Varanloo, N.; Nasrabadi, M.H.; Vatannejad, A.; Amjadi, F.S.; Masroor, M.J.; Bajelan, L.; Mehdizadeh, M.; Aflatoonian, R.; Zandieh, Z. Supplementation of sperm freezing medium with myoinositol improve human sperm parameters and protects it against DNA fragmentation and apoptosis. Cell Tissue Bank. 2019, 20, 77–86. [Google Scholar] [CrossRef]
- Condorelli, R.A.; La Vignera, S.; Di Bari, F.; Unfer, V.; Calogero, A.E. Effects of myoinositol on sperm mitochondrial function in-vitro. Eur. Rev. Med. Pharmacol. Sci. 2011, 15, 129–134. [Google Scholar]
- Gulino, F.A.; Leonardi, E.; Marilli, I.; Musmeci, G.; Vitale, S.G.; Leanza, V.; Palumbo, M.A. Effect of treatment with myo-inositol on semen parameters of patients undergoing an IVF cycle: In vivo study. Gynecol. Endocrinol. 2016, 32, 65–68. [Google Scholar] [CrossRef]
- Rubino, P.; Palini, S.; Chigioni, S.; Carlomagno, G.; Quagliariello, A.; De Stefani, S.; Baglioni, A.; Bulletti, C. Improving fertilization rate in ICSI cycles by adding myoinositol to the semen preparation procedures: A prospective, bicentric, randomized trial on sibling oocytes. J. Assist. Reprod. Genet. 2015, 32, 387–394. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Agarwal, A.; Leisegang, K.; Majzoub, A.; Henkel, R.; Finelli, R.; Panner Selvam, M.K.; Tadros, N.; Parekh, N.; Ko, E.Y.; Cho, C.L.; et al. Utility of Antioxidants in the Treatment of Male Infertility: Clinical Guidelines Based on a Systematic Review and Analysis of Evidence. World J. Mens Health 2021, 39, 233–290. [Google Scholar] [CrossRef] [PubMed]
- Rolf, C.; Cooper, T.G.; Yeung, C.H.; Nieschlag, E. Antioxidant treatment of patients with asthenozoospermia or moderate oligoasthenozoospermia with high-dose vitamin C and vitamin E: A randomized, placebo-controlled, double-blind study. Hum. Reprod. 1999, 14, 1028–1033. [Google Scholar] [CrossRef] [PubMed]
- Greco, E.; Iacobelli, M.; Rienzi, L.; Ubaldi, F.; Ferrero, S.; Tesarik, J. Reduction of the incidence of sperm DNA fragmentation by oral antioxidant treatment. J. Androl. 2005, 26, 349–353. [Google Scholar] [CrossRef]
- Silver, E.W.; Eskenazi, B.; Evenson, D.P.; Block, G.; Young, S.; Wyrobek, A.J. Effect of antioxidant intake on sperm chromatin stability in healthy nonsmoking men. J. Androl. 2005, 26, 550–556. [Google Scholar] [CrossRef]
- Sigman, M.; Glass, S.; Campagnone, J.; Pryor, J.L. Carnitine for the treatment of idiopathic asthenospermia: A randomized, double-blind, placebo-controlled trial. Fertil. Steril. 2006, 85, 1409–1414. [Google Scholar] [CrossRef]
- Ménézo, Y.J.R.; Hazout, A.; Panteix, G.; Robert, F.; Rollet, J.; Cohen-Bacrie, P.; Chapuis, F.; Clément, P.; Benkhalifa, M. Antioxidants to reduce sperm DNA fragmentation: An unexpected adverse effect. Reprod. Biomed. Online 2007, 14, 418–421. [Google Scholar] [CrossRef]
- Ménézo, Y.; Entezami, F.; Lichtblau, I.; Belloc, S.; Cohen, M.; Dale, B. Oxidative stress and fertility: Incorrect assumptions and ineffective solutions? Zygote 2014, 22, 80–90. [Google Scholar] [CrossRef][Green Version]
- Bleau, G.; Lemarbre, J.; Faucher, G.; Roberts, K.D.; Chapdelaine, A. Semen selenium and human fertility. Fertil. Steril. 1984, 42, 890–894. [Google Scholar] [CrossRef]
- Hawkes, W.C.; Turek, P.J. Effects of dietary selenium on sperm motility in healthy men. J. Androl. 2001, 22, 764–772. [Google Scholar]
- Murata, M.; Kawanishi, S. Oxidative DNA damage by vitamin A and its derivative via superoxide generation. J. Biol. Chem. 2000, 275, 2003–2008. [Google Scholar] [CrossRef]
- Steiner, A.Z.; Hansen, K.R.; Barnhart, K.T.; Cedars, M.I.; Legro, R.S.; Diamond, M.P.; Krawetz, S.A.; Usadi, R.; Baker, V.L.; Coward, R.M.; et al. The effect of antioxidants on male factor infertility: The Males, Antioxidants, and Infertility (MOXI) randomized clinical trial. Fertil. Steril. 2020, 113, 552–560. [Google Scholar] [CrossRef]
- Salvio, G.; Cutini, M.; Ciarloni, A.; Giovannini, L.; Perrone, M.; Balercia, G. Coenzyme Q10 and Male Infertility: A Systematic Review. Antioxidants 2021, 10, 874. [Google Scholar] [CrossRef]
- Lafuente, R.; González-Comadrán, M.; Solà, I.; López, G.; Brassesco, M.; Carreras, R.; Checa, M.A. Coenzyme Q10 and male infertility: A meta-analysis. J. Assist. Reprod. Genet. 2013, 30, 1147–1156. [Google Scholar] [CrossRef]
- Di Tucci, C.; Galati, G.; Mattei, G.; Bonanni, V.; Capri, O.; D’Amelio, R.; Muzii, L.; Benedetti Panici, P. The role of alpha lipoic acid in female and male infertility: A systematic review. Gynecol. Endocrinol. 2021, 37, 497–505. [Google Scholar] [CrossRef]
- Smits, R.M.; Mackenzie-Proctor, R.; Yazdani, A.; Stankiewicz, M.T.; Jordan, V.; Showell, M.G. Antioxidants for male subfertility. Cochrane DatabaseSyst. Rev. 2019, 3, CD007411. [Google Scholar] [CrossRef]
- Ross, C.; Morriss, A.; Khairy, M.; Khalaf, Y.; Braude, P.; Coomarasamy, A.; El-Toukhy, T. A systematic review of the effect of oral antioxidants on male infertility. Reprod. Biomed.Online 2010, 20, 711–723. [Google Scholar] [CrossRef]
- Majzoub, A.; Agarwal, A. Systematic review of antioxidant types and doses in male infertility: Benefits on semen parameters, advanced sperm function, assisted reproduction and live-birth rate. Arab. J. Urol. 2018, 16, 113–124. [Google Scholar] [CrossRef]
- Esteves, S.C.; Agarwal, A.; Cho, C.-L.; Majzoub, A. A Strengths-Weaknesses-Opportunities-Threats (SWOT) analysis on the clinical utility of sperm DNA fragmentation testing in specific male infertility scenarios. Transl. Androl. Urol. 2017, 6 (Suppl. S4), S734–S760. [Google Scholar] [CrossRef]
- Zhao, J.; Dong, X.; Hu, X.; Long, Z.; Wang, L.; Liu, Q.; Sun, B.; Wang, Q.; Wu, Q.; Li, L. Zinc levels in seminal plasma and their correlation with male infertility: A systematic review and meta-analysis. Sci. Rep. 2016, 6, 22386. [Google Scholar] [CrossRef]
- Tsampoukas, G.; Khan, M.F.; Katsouri, A.; Akhter, W.; Moussa, M.; Deliveliotis, K.; Papatsoris, A.; Buchholz, N. L-carnitine as primary or adjuvant treatment in infertile patients with varicocele. A systematic review. Arch. Ital. Urol. Androl. 2020, 92, 263–267. [Google Scholar] [CrossRef]
- Tsampoukas, G.; Gkeka, K.; Dellis, A.; Brown, D.; Katsouri, A.; Alneshawy, A.; Moussa, M.; Papatsoris, A.; Buchholz, N. Vitamins as primary or adjunctive treatment in infertile men with varicocele: A systematic review. Arab. J. Urol. 2021, 19, 264–273. [Google Scholar] [CrossRef]
- Pyrgidis, N.; Sokolakis, I.; Palapelas, V.; Tishukov, M.; Mykoniatis, I.; Symeonidis, E.N.; Zachariou, A.; Kaltsas, A.; Sofikitis, N.; Hatzichristodoulou, G.; et al. The Effect of Antioxidant Supplementation on Operated or Non-Operated Varicocele-Associated Infertility: A Systematic Review and Meta-Analysis. Antioxidants 2021, 10, 1067. [Google Scholar] [CrossRef]
- Gharagozloo, P.; Aitken, R.J. The role of sperm oxidative stress in male infertility and the significance of oral antioxidant therapy. Hum. Reprod. 2011, 26, 1628–1640. [Google Scholar] [CrossRef]
- Arafa, M.; Agarwal, A.; Majzoub, A.; Panner Selvam, M.K.; Baskaran, S.; Henkel, R.; Elbardisi, H. Efficacy of Antioxidant Supplementation on Conventional and Advanced Sperm Function Tests in Patients with Idiopathic Male Infertility. Antioxidants 2020, 9, 219. [Google Scholar] [CrossRef]
- Fainberg, J.; Kashanian, J.A. Recent advances in understanding and managing male infertility. F1000Res 2019, 8, 670. [Google Scholar] [CrossRef]
- Han, X.; Huang, Q. Environmental pollutants exposure and male reproductive toxicity: The role of epigenetic modifications. Toxicology 2021, 456, 152780. [Google Scholar] [CrossRef]
- Takeshima, T.; Usui, K.; Mori, K.; Asai, T.; Yasuda, K.; Kuroda, S.; Yumura, Y. Oxidative stress and male infertility. Reprod. Med. Biol. 2021, 20, 41–52. [Google Scholar] [CrossRef]
- O’Flaherty, C. Reactive Oxygen Species and Male Fertility. Antioxidants 2020, 9, 287. [Google Scholar] [CrossRef]

| Author, Year | Examined Antioxidant Compound | Results |
|---|---|---|
| Balercia et al., 2005 [50] | LC, LAC, and a combination of LC and LAC | Sperm kinetic parameters improvement |
| Total seminal antioxidant capacity increase and positive correlation with kinetic features improvement | ||
| Balercia et al., 2009 [51] | CoQ10 | Sperm cell total motility and forward motility improvement |
| Twice as many spontaneous pregnancies during the observation period | ||
| Busetto et al., 2018 [53] | LC, LAC, fructose, fumarate, vitamin C, citric acid, zinc, CoQ10, Se, folic acid, and vitamin B12 | Significant increase in sperm concentration, total sperm count, progressive, and total motility |
| 10 out of 12 pregnancies during the follow-up favored the supplementation arm, although the pregnancy rate was not a study endpoint | ||
| Micic et al., 2019 [54] | LC, LAC, fructose, fumarate, vitamin C, citric acid, zinc, CoQ10, selenium, folic acid, and vitamin B12 | Significant increases in sperm volume, progressive motility, and vitality after six months of therapy. |
| Significant decline in sperm DNA fragmentation index (DFI) compared with baseline and 3-month therapy | ||
| Decreased levels of DFI represented a reliable predictor of progressive sperm motility >10% | ||
| Positive correlation of elevated levels of seminal carnitine and α-glycosidase with improved progressive motility | ||
| Safarinejad et al., 2012 [55] | Ubiquinol—Reduced form of CoQ10 | Significant increases in sperm density, motility, and morphology |
| Haghighian et al., 2015 [56] | Alpha-lipoic acid | Significant increases in total sperm count, sperm concentration, and sperm motility |
| Potential to significantly improve total antioxidant capacity (TAC) and malondialdehyde (MDA) levels | ||
| Calogero et al., 2015 [57] | Myoinositol | Significant increase in the percentage of acrosome-reacted spermatozoa, sperm concentration, total count, and progressive motility |
| Rebalance in serum luteinizing hormone, follicle-stimulating hormone, and inhibin B concentrations | ||
| Tremellen et al., 2007 [52] | Vitamins C and E, Se, garlic, lycopene, zinc, and folic acid | Significant improvement in viable pregnancy rates |
| No significant changes in oocyte fertilisation rate or embryo quality | ||
| Steiner et al., 2020 [72] | Vitamin C, vitamin E, Se, LC, zinc, folic acid, and lycopene | No significant improvements on sperm morphology, motility, or DNA fragmentation |
| No improvement on in vivo pregnancy or live birth rates | ||
| Sigman et al., 2006 [66] | LC and LAC | No statistically or clinically significant increase in sperm motility and total motile sperm counts at sequential treatment time points |
| Rolf et al., 1999 [63] | Vitamin C and E | No specific improvement on conventional sperm parameters, 24 h sperm survival rate, or pregnancy initiation |
| Greco et al. 2005 [64] | Vitamin C and E | No significant improvement on sperm concentration, count, motility, and morphology |
| Significant decline in DNA-fragmented spermatozoa | ||
| Silver et al. 2005 [65] | Vitamin C, vitamin E, and beta-carotene | Moderate, but not high beta-carotene intake increased standard deviation of DNA fragmentation index (SD DFI), as well as the percentage of immature sperm, compared with low intake |
| Hawkes et al., 2001 [70] | Se | Overdosing of Se may alter the sperm selenoprotein(s) of the outer mitochondrial membrane and alter the thyroid hormone metabolism both conditions associated with asthenospermia and impaired fertility |
| Ménézo et al., 2007 [67] | Vitamins C and E, β-carotene, zinc, and Se | Decrease in sperm DNA fragmentation |
| Increase in sperm decondensation | ||
| Bleau et al. 1984 [69] | Se | Se level between approximately 40 and 70 ng/mL was optimal for reproductive performance (high pregnancy rate and low abortion rate) |
| Sperm motility was maximal at semen Se levels ranging between 50 and 69 ng/mL | ||
| Se levels ≤35 ng/mL were associated with male infertility | ||
| Se levels ≥80 ng/mL were associated with a high abortion rate and signs of ovarian dysfunction in the partner |
| Direct and Indirect Techniques to Measure OS in the Seminal Plasma | |
|---|---|
| Direct | Indirect |
| Oxidation-reduction potential (ORP) | Total antioxidant capacity (TAC) |
| ROS by chemiluminescence | ROS-TAC |
| Nitro blue tetrazolium (NBT) | Malondialdehyde (MDA) |
| Cytochrome C reduction test | Myeloperoxidase or Endtz test |
| Fluorescein probe | Lipid peroxidation levels |
| Oxidation-induced fluorochrome probe | Chemokines |
| Electron spin resonance | Ascorbate |
| DNA fragmentation | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Symeonidis, E.N.; Evgeni, E.; Palapelas, V.; Koumasi, D.; Pyrgidis, N.; Sokolakis, I.; Hatzichristodoulou, G.; Tsiampali, C.; Mykoniatis, I.; Zachariou, A.; et al. Redox Balance in Male Infertility: Excellence through Moderation—“Μέτρον ἄριστον”. Antioxidants 2021, 10, 1534. https://doi.org/10.3390/antiox10101534
Symeonidis EN, Evgeni E, Palapelas V, Koumasi D, Pyrgidis N, Sokolakis I, Hatzichristodoulou G, Tsiampali C, Mykoniatis I, Zachariou A, et al. Redox Balance in Male Infertility: Excellence through Moderation—“Μέτρον ἄριστον”. Antioxidants. 2021; 10(10):1534. https://doi.org/10.3390/antiox10101534
Chicago/Turabian StyleSymeonidis, Evangelos N., Evangelini Evgeni, Vasileios Palapelas, Dimitra Koumasi, Nikolaos Pyrgidis, Ioannis Sokolakis, Georgios Hatzichristodoulou, Chara Tsiampali, Ioannis Mykoniatis, Athanasios Zachariou, and et al. 2021. "Redox Balance in Male Infertility: Excellence through Moderation—“Μέτρον ἄριστον”" Antioxidants 10, no. 10: 1534. https://doi.org/10.3390/antiox10101534
APA StyleSymeonidis, E. N., Evgeni, E., Palapelas, V., Koumasi, D., Pyrgidis, N., Sokolakis, I., Hatzichristodoulou, G., Tsiampali, C., Mykoniatis, I., Zachariou, A., Sofikitis, N., Kaltsas, A., & Dimitriadis, F. (2021). Redox Balance in Male Infertility: Excellence through Moderation—“Μέτρον ἄριστον”. Antioxidants, 10(10), 1534. https://doi.org/10.3390/antiox10101534









