The Total Antioxidant Status, Serum Selenium Concentrations and the Ultrasound Assessment Carotid Intima Media Thickness in Patients with Arterial Hypertension
Abstract
1. Introduction
2. Material and Methods
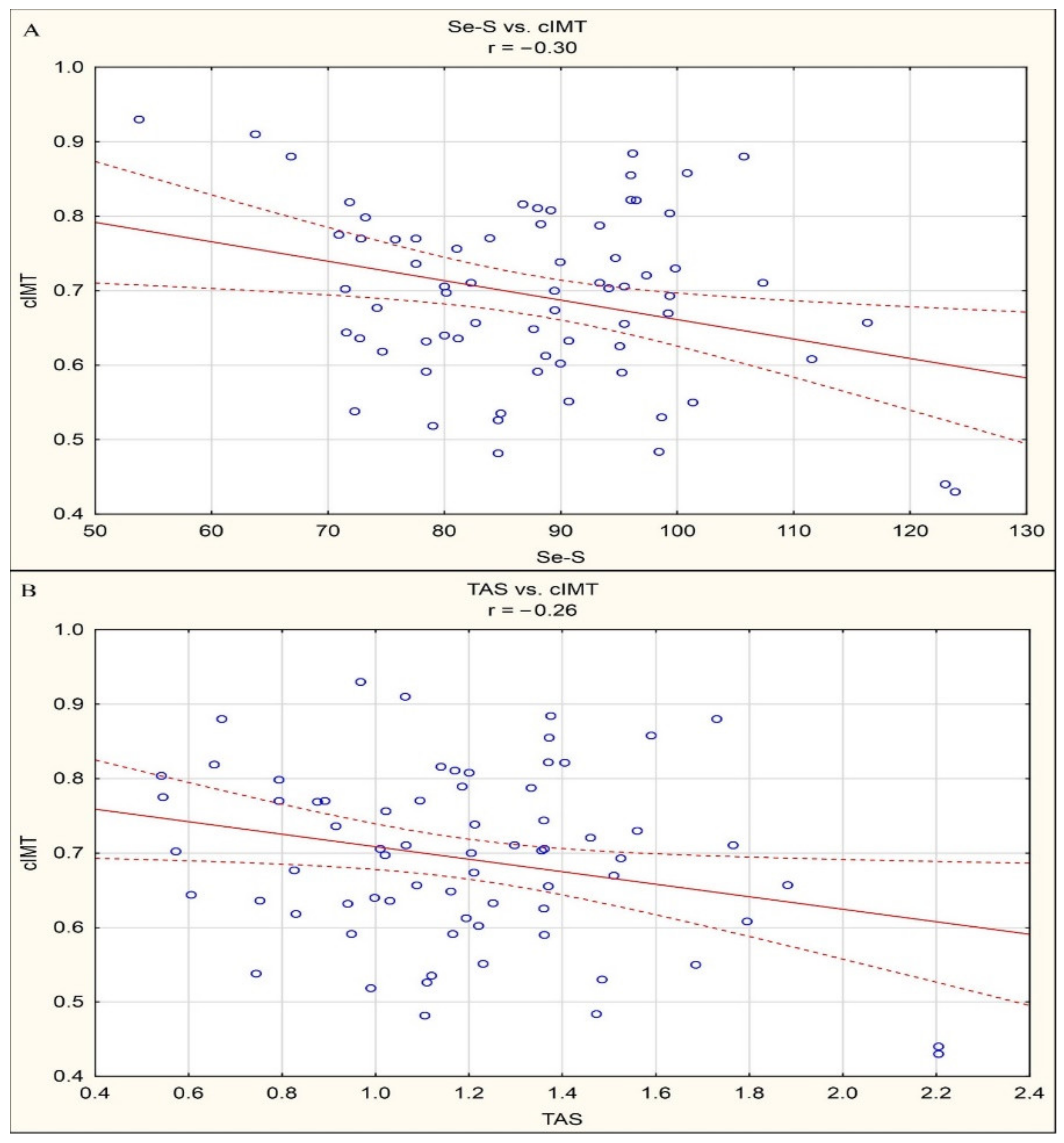
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wang, N.; Tan, H.Y.; Li, S.; Xu, Y.; Guo, W.; Feng, Y. Supplementation of Micronutrient Selenium in Metabolic Diseases: Its Role as an Antioxidant. Oxid. Med. Cell. Longev. 2017, 2017, 7478523. [Google Scholar] [CrossRef] [PubMed]
- Mangiapane, E.; Pessione, A.; Pessione, E. Selenium and selenoproteins: An overview on different biological systems. Curr. Protein Pept. Sci. 2014, 15, 598–607. [Google Scholar] [CrossRef] [PubMed]
- Gać, P.; Pawlas, N.; Poręba, R.; Poręba, M.; Markiewicz-Górka, I.; Januszewska, L.; Olszowy, Z.; Pawlas, K. Interaction between blood selenium concentration and a level of oxidative stress and antioxidative capacity in healthy children. Environ. Toxicol. Pharmacol. 2015, 39, 137–144. [Google Scholar] [CrossRef]
- Rayman, M.P. Selenium and human health. Lancet 2012, 379, 1256–1268. [Google Scholar] [CrossRef]
- Fernández-Lázaro, D.; Fernandez-Lazaro, C.I.; Mielgo-Ayuso, J.; Navascués, L.J.; Córdova Martínez, A.; Seco-Calvo, J. The Role of Selenium Mineral Trace Element in Exercise: Antioxidant Defense System, Muscle Performance, Hormone Response, and Athletic Performance. A Systematic Review. Nutrients 2020, 12, 1790. [Google Scholar] [CrossRef]
- Cai, Z.; Zhang, J.; Li, H. Selenium, aging and aging-related diseases. Aging Clin. Exp. Res. 2019, 31, 1035–1047. [Google Scholar] [CrossRef]
- Vinceti, M.; Filippini, T.; Wise, L.A. Environmental Selenium and Human Health: An Update. Curr. Environ. Health Rep. 2018, 5, 464–485. [Google Scholar] [CrossRef]
- Sun, D.; Sun, S.; Feng, H.; Hou, J. Keshan Disease. Public Health in China. In Endemic Disease in China; Sun, D., Ed.; Springer: Singapore, 2019; Volume 2. [Google Scholar] [CrossRef]
- Kuruppu, D.; Hendrie, H.C.; Yang, L.; Gao, S. Selenium levels and hypertension: A systematic review of the literature. Public Health Nutr. 2014, 17, 1342–1352. [Google Scholar] [CrossRef]
- Vinceti, M.; Filippini, T.; Cilloni, S.; Bargellini, A.; Chawla, R.; Loomba, R.; Bargellini, A.; Orsini, N.; Dhillon, K.S.; Whelton, P. Blood pressure levels and hypertension prevalence in a high selenium environment: Results from a cross-sectional study. Nutr. Metab. Cardiovasc. Dis. 2019, 29, 398–408. [Google Scholar] [CrossRef]
- Kunutsor, S.K.; Laukkanen, J.A. Serum zinc concentrations and incident hypertension: New findings from a population-based cohort study. J. Hypertens. 2016, 34, 1055–1061. [Google Scholar] [CrossRef]
- Li, Z.; Wang, W.; Liu, H.; Li, S.; Zhang, D. The association of serum zinc and copper with hypertension: A meta-analysis. J. Trace Elem. Med. Biol. 2019, 53, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.F.; Eccles, K.M.; Chan, H.M. High selenium exposure lowers the odds ratios for hypertension, stroke, and myocardial infarction associated with mercury exposure among Inuit in Canada. Environ. Int. 2017, 102, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Verdecchia, P.; Reboldi, G.; Angeli, F. The 2020 International Society of Hypertension global hypertension practice guidelines—Key messages and clinical considerations. Eur. J. Intern Med. 2020. [Google Scholar] [CrossRef]
- Van den Born, B.H.; Lip, G.Y.H.; Brguljan-Hitij, J.; Cremer, A.; Segura, J.; Morales, E.; Mahfoud, F.; Amraoui, F.; Persu, A.; Kahan, T.; et al. ESC Council on hypertension position document on the management of hypertensive emergencies. Eur. Heart J. Cardiovasc. Pharmacother. 2019, 5, 37–46. [Google Scholar] [CrossRef]
- Piskorz, D. Hypertensive Mediated Organ Damage and Hypertension Management. How to Assess Beneficial Effects of Antihypertensive Treatments? High Blood Press. Cardiovasc. Prev. 2020, 27, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Katakami, N.; Matsuoka, T.A.; Shimomura, I. Clinical utility of carotid ultrasonography: Application for the management of patients with diabetes. J. Diabetes Investig. 2019, 10, 883–898. [Google Scholar] [CrossRef] [PubMed]
- Naqvi, T.Z.; Lee, M.S. Carotid intima-media thickness and plaque in cardiovascular risk assessment. JACC Cardiovasc. Imaging. 2014, 7, 1025–1038. [Google Scholar] [CrossRef]
- Nezu, T.; Hosomi, N.; Aoki, S.; Matsumoto, M. Carotid Intima-Media Thickness for Atherosclerosis. J. Atheroscler. Thromb. 2016, 23, 18–31. [Google Scholar] [CrossRef]
- Gać, P.; Urbanik, D.; Pawlas, N.; Poręba, M.; Martynowicz, H.; Prokopowicz, A.; Januszewska, L.; Markiewicz-Górka, I.; Jaremków, A.; Mazur, G.; et al. Total antioxidant status reduction conditioned by a serum selenium concentration decrease as a mechanism of the ultrasonographically measured brachial artery dilatation impairment in patients with arterial hypertension. Environ. Toxicol. Pharmacol. 2020, 75, 103332. [Google Scholar] [CrossRef]
- Salonen, J.T.; Salonen, R.; Seppänen, K.; Kantola, M.; Suntioinen, S.; Korpela, H. Interactions of serum copper, selenium, and low density lipoprotein cholesterol in atherogenesis. BMJ 1991, 302, 756–760. [Google Scholar] [CrossRef]
- Salonen, J.T.; Salonen, R. Ultrasound B-mode imaging in observational studies of atherosclerotic progression. Circulation 1993, 87 (Suppl. S3), II56–II65. [Google Scholar] [PubMed]
- Bonithon-Kopp, C.; Coudray, C.; Berr, C.; Touboul, P.J.; Fève, J.M.; Favier, A.; Ducimetière, P. Combined effects of lipid peroxidation and antioxidant status on carotid atherosclerosis in a population aged 59–71 y: The EVA Study. Etude sur le Vieillisement Artériel. Am. J. Clin. Nutr. 1997, 65, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Falcone, E.L.; Mangili, A.; Tang, A.M.; Jones, C.Y.; Woods, M.N.; Polak, J.F.; Wanke, C.A. Micronutrient concentrations and subclinical atherosclerosis in adults with HIV. Am. J. Clin. Nutr. 2010, 91, 1213–1219. [Google Scholar] [CrossRef] [PubMed]
- Xun, P.; Liu, K.; Morris, J.S.; Daviglus, M.L.; He, K. Longitudinal association between toenail selenium levels and measures of subclinical atherosclerosis: The CARDIA trace element study. Atherosclerosis 2010, 210, 662–667. [Google Scholar] [CrossRef]
- Swart, R.; Schutte, A.E.; Van Rooyen, J.M.; Mels, C.M.C. Selenium and large artery structure and function: A 10-year prospective study. Eur. J. Nutr. 2019, 58, 3313–3323. [Google Scholar] [CrossRef]
- Ferreira, L.L.; Silva, T.R.; Maturana, M.A.; Spritzer, P.M. Dietary intake of isoflavones is associated with a lower prevalence of subclinical cardiovascular disease in postmenopausal women: Cross-sectional study. J. Hum. Nutr. Diet. 2019, 32, 810–818. [Google Scholar] [CrossRef]
- Maugeri, A.; Hruskova, J.; Jakubik, J.; Kunzova, S.; Sochor, O.; Barchitta, M.; Agodi, A.; Bauerova, H.; Medina-Inojosa, J.R.; Vinciguerra, M. Dietary antioxidant intake decreases carotid intima media thickness in women but not in men: A cross-sectional assessment in the Kardiovize study. Free Radic. Biol. Med. 2019, 131, 274–281. [Google Scholar] [CrossRef]
- Zureik, M.; Galan, P.; Bertrais, S.; Mennen, L.; Czernichow, S.; Blacher, J.; Ducimetière, P.; Hercberg, S. Effects of long-term daily low-dose supplementation with antioxidant vitamins and minerals on structure and function of large arteries. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 1485–1491. [Google Scholar] [CrossRef]
- Liberda, E.N.; Zuk, A.M.; Tsuji, L.J.S. Complex contaminant mixtures and their associations with intima-media thickness. BMC Cardiovasc. Disord. 2019, 19, 289. [Google Scholar] [CrossRef]
- Skoczyńska, A.; Poreba, R.; Steinmentz-Beck, A.; Martynowicz, H.; Affelska-Jercha, A.; Turczyn, B.; Wojakowska, A.; Jedrychowska, I. The dependence between urinary mercury concentration and carotid arterial intima-media thickness in workers occupationally exposed to mercury vapour. Int. J. Occup. Med. Environ. Health 2009, 22, 135–142. [Google Scholar] [CrossRef]
- Bogdanski, P.; Miller-Kasprzak, E.; Pupek-Musialik, D.; Jablecka, A.; Lacinski, M.; Jagodzinski, P.P.; Jakubowski, H. Plasma total homocysteine is a determinant of carotid intima-media thickness and circulating endothelial progenitor cells in patients with newly diagnosed hypertension. Clin. Chem. Lab. Med. 2012, 50, 1107–1113. [Google Scholar] [CrossRef] [PubMed]
- Gur, M.; Yilmaz, R.; Demirbag, R.; Yildiz, A.; Kocyigit, A.; Celik, H.; Cayli, M.; Polat, M.; Bas, M.M. Lymphocyte DNA damage is associated with increased aortic intima-media thickness. Mutat. Res. 2007, 617, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Gür, M.; Çaylı, M.; Uçar, H.; Elbasan, Z.; Şahin, D.Y.; Gözükara, M.Y.; Selek, Ş.; Koyunsever, N.Y.; Şeker, T.; Türkoğlu, C.; et al. Paraoxonase (PON1) activity in patients with subclinical thoracic aortic atherosclerosis. Int. J. Cardiovasc. Imaging 2014, 30, 889–895. [Google Scholar] [CrossRef] [PubMed]
- Tabatabaei-Malazy, O.; Fakhrzadeh, H.; Sharifi, F.; Mirarefin, M.; Arzaghi, S.M.; Badamchizadeh, Z.; Alizadeh Khoee, M.; Larijani, B. Effect of metabolic control on oxidative stress, subclinical atherosclerosis and peripheral artery disease in diabetic patients. J. Diabetes Metab. Disord. 2015, 14, 84. [Google Scholar] [CrossRef] [PubMed]
- Cakir, E.; Ozbek, M.; Ozkaya, E.; Colak, N.; Cakal, E.; Sayki, M.; Gungunes, A.; Aliyazicioglu, Y.; Mentese, A.; Delibasi, T. Oxidative stress markers are not valuable markers in lean and early age of polycystic ovary syndrome patients. J. Endocrinol. Investig. 2011, 34, e178–e182. [Google Scholar] [CrossRef]
- Karkucak, M.; Capkin, E.; Alver, A.; Akyuz, A.; Kiris, A.; Ak, E.; Topbas, M.; Tosun, M. The effect of anti-TNF agent on oxidation status in patients with ankylosing spondylitis. Clin. Rheumatol. 2010, 29, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Kipp, A.P.; Strohm, D.; Brigelius-Flohé, R.; Schomburg, L.; Bechthold, A.; Leschik-Bonnet, E.; Heseker, H.; German Nutrition Society (DGE). Revised reference values for selenium intake. J. Trace Elem. Med. Biol. 2015, 32, 195–199. [Google Scholar] [CrossRef]
- McMillan, D.C.; Maguire, D.; Talwar, D. Relationship between nutritional status and the systemic inflammatory response: Micronutrients. Proc. Nutr. Soc. 2019, 78, 56–67. [Google Scholar] [CrossRef]
| Age (Years) a | 53.48 ± 12.78 |
|---|---|
| men b | 60.5 (46) |
| women b | 39.5 (30) |
| height (cm) a | 172.76 ± 10.93 |
| body mass (kg) a | 94.99 ± 18.22 |
| BMI (kg/m2) a | 31.70 ± 4.69 |
| normal body mass b | 3.9 (3) |
| overweight b | 38.1 (29) |
| obese b | 57.9 (44) |
| smoking b | 21.0 (16) |
| arterial hypertension b | 100.0 (76) |
| diuretics b | 50.0 (38) |
| β-blockers b | 51.3 (39) |
| ACE inhibitors b | 35.5 (27) |
| angiotensin receptor blockers b | 40.8 (31) |
| calcium channel blockers b | 51.3 (39) |
| total cholesterol (mg/dL) a | 205.36 ± 44.57 |
| HDL cholesterol (mg/dL) a | 48.79 ± 13.44 |
| LDL cholesterol (mg/dL) a | 121.46 ± 36.05 |
| triglicerides (mg/dL) a | 198.95 ± 145.44 |
| glucose (mg/dL) a | 100.05 ± 22.07 |
| Subgroup | Se-S (µg/L) a | TAS (mM) a | Atherosclerotic Plaques b | Non-Significant Stenosis b | cIMT (mm) a |
|---|---|---|---|---|---|
| low-Se | 76.86 ± 7.89 | 0.98 ± 0.22 | 63.2 | 15.8 | 0.73 ± 0.19 |
| high-Se | 102.61 ± 18.11 | 1.39 ± 0.34 | 55.3 | 13.1 | 0.65 ± 0.10 |
| p | <0.05 | <0.05 | Ns | Ns | <0.05 |
| low-TAS | 83.94 ± 0.91 | 0.91 ± 0.19 | 63.2 | 18.4 | 0.79 ± 0.18 |
| high-TAS | 95.53 ± 12.18 | 1.45 ± 0.25 | 55.3 | 10.5 | 0.56 ± 0.13 |
| p | <0.05 | <0.05 | Ns | Ns | <0.05 |
| low-cIMT | 94.98 ± 22.26 | 1.25 ± 0.38 | 21.0 | 0.0 | 0.57 ± 0.05 |
| high-cIMT | 84.49 ± 13.37 | 1.13 ± 0.33 | 97.4 | 28.9 | 0.80 ± 0.12 |
| p | <0.05 | ns | <0.05 | <0.05 | <0.05 |
| Model for: cIMT (mm) | ||||||
|---|---|---|---|---|---|---|
| Univariate Regression | Multivariable Regression | |||||
| Rc | SEM of Rc | p | Rc | SEM of Rc | p | |
| age (years) | 0.003 | 0.001 | <0.05 | 0.003 | 0.001 | <0.05 |
| men | 0.070 | 0.031 | <0.05 | 0.060 | 0.036 | ns |
| BMI (kg/m2) | 0.003 | 0.001 | <0.05 | 0.004 | 0.001 | <0.05 |
| smoking | 0.123 | 0.008 | <0.05 | 0.100 | 0.032 | <0.05 |
| diuretics | −0.034 | 0.031 | Ns | - | - | - |
| β-blockers | −0.022 | 0.003 | <0.05 | −0.009 | 0.011 | ns |
| ACE inhibitors | −0.016 | 0.033 | Ns | - | - | - |
| angiotensin receptor blockers | −0.039 | 0.032 | Ns | - | - | - |
| calcium channel blockers | −0.023 | 0.003 | <0.05 | −0.032 | 0.024 | ns |
| total cholesterol (mg/dL) | 0.001 | 0.001 | Ns | - | - | - |
| HDL cholesterol (mg/dL) | −0.001 | 0.001 | Ns | - | - | - |
| triglicerides (mg/dL) | 0.001 | 0.000 | <0.05 | 0.001 | 0.001 | ns |
| glucose (mg/dL) | 0.001 | 0.001 | Ns | - | - | - |
| Se-S (μg/L) | −0.004 | 0.001 | <0.05 | −0.005 | 0.001 | <0.05 |
| TAS (mM) | −0.184 | 0.045 | <0.05 | −0.116 | 0.087 | ns |
| Model for: TAS (mM) | ||||||
|---|---|---|---|---|---|---|
| Univariate Regression | Multivariable Regression | |||||
| Rc | SEM of Rc | p | Rc | SEM of Rc | p | |
| age (years) | −0.003 | 0.003 | Ns | - | - | - |
| men | 0.116 | 0.079 | Ns | - | - | - |
| BMI (kg/m2) | −0.012 | 0.002 | <0.05 | −0.016 | 0.007 | <0.05 |
| smoking | −0.227 | 0.092 | <0.05 | −0.170 | 0.085 | <0.05 |
| diuretics | 0.131 | 0.057 | <0.05 | 0.048 | 0.068 | ns |
| β-blockers | 0.117 | 0.008 | <0.05 | 0.138 | 0.026 | <0.05 |
| ACE inhibitors | 0.181 | 0.032 | <0.05 | 0.109 | 0.040 | <0.05 |
| angiotensin receptor blockers | 0.081 | 0.079 | ns | - | - | - |
| calcium channel blockers | 0.019 | 0.078 | ns | - | - | - |
| total cholesterol (mg/dL) | −0.001 | 0.001 | ns | - | - | - |
| HDL cholesterol (mg/dL) | 0.002 | 0.003 | ns | - | - | - |
| triglicerides (mg/dL) | −0.001 | 0.001 | ns | - | - | - |
| glucose (mg/dL) | −0.002 | 0.002 | ns | - | - | - |
| Se-S (μg/L) | 0.010 | 0.002 | <0.05 | 0.012 | 0.002 | <0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gać, P.; Poręba, M.; Januszewska, L.; Prokopowicz, A.; Martynowicz, H.; Mazur, G.; Poręba, R. The Total Antioxidant Status, Serum Selenium Concentrations and the Ultrasound Assessment Carotid Intima Media Thickness in Patients with Arterial Hypertension. Antioxidants 2021, 10, 63. https://doi.org/10.3390/antiox10010063
Gać P, Poręba M, Januszewska L, Prokopowicz A, Martynowicz H, Mazur G, Poręba R. The Total Antioxidant Status, Serum Selenium Concentrations and the Ultrasound Assessment Carotid Intima Media Thickness in Patients with Arterial Hypertension. Antioxidants. 2021; 10(1):63. https://doi.org/10.3390/antiox10010063
Chicago/Turabian StyleGać, Paweł, Małgorzata Poręba, Lidia Januszewska, Adam Prokopowicz, Helena Martynowicz, Grzegorz Mazur, and Rafał Poręba. 2021. "The Total Antioxidant Status, Serum Selenium Concentrations and the Ultrasound Assessment Carotid Intima Media Thickness in Patients with Arterial Hypertension" Antioxidants 10, no. 1: 63. https://doi.org/10.3390/antiox10010063
APA StyleGać, P., Poręba, M., Januszewska, L., Prokopowicz, A., Martynowicz, H., Mazur, G., & Poręba, R. (2021). The Total Antioxidant Status, Serum Selenium Concentrations and the Ultrasound Assessment Carotid Intima Media Thickness in Patients with Arterial Hypertension. Antioxidants, 10(1), 63. https://doi.org/10.3390/antiox10010063







