High Cervical Spinal Cord Stimulation: A One Year Follow-Up Study on Motor and Non-Motor Functions in Parkinson’s Disease
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Approval
2.2. Patients
2.3. Surgical Procedure
2.4. Stimulation Parameters and Clinical Evaluations
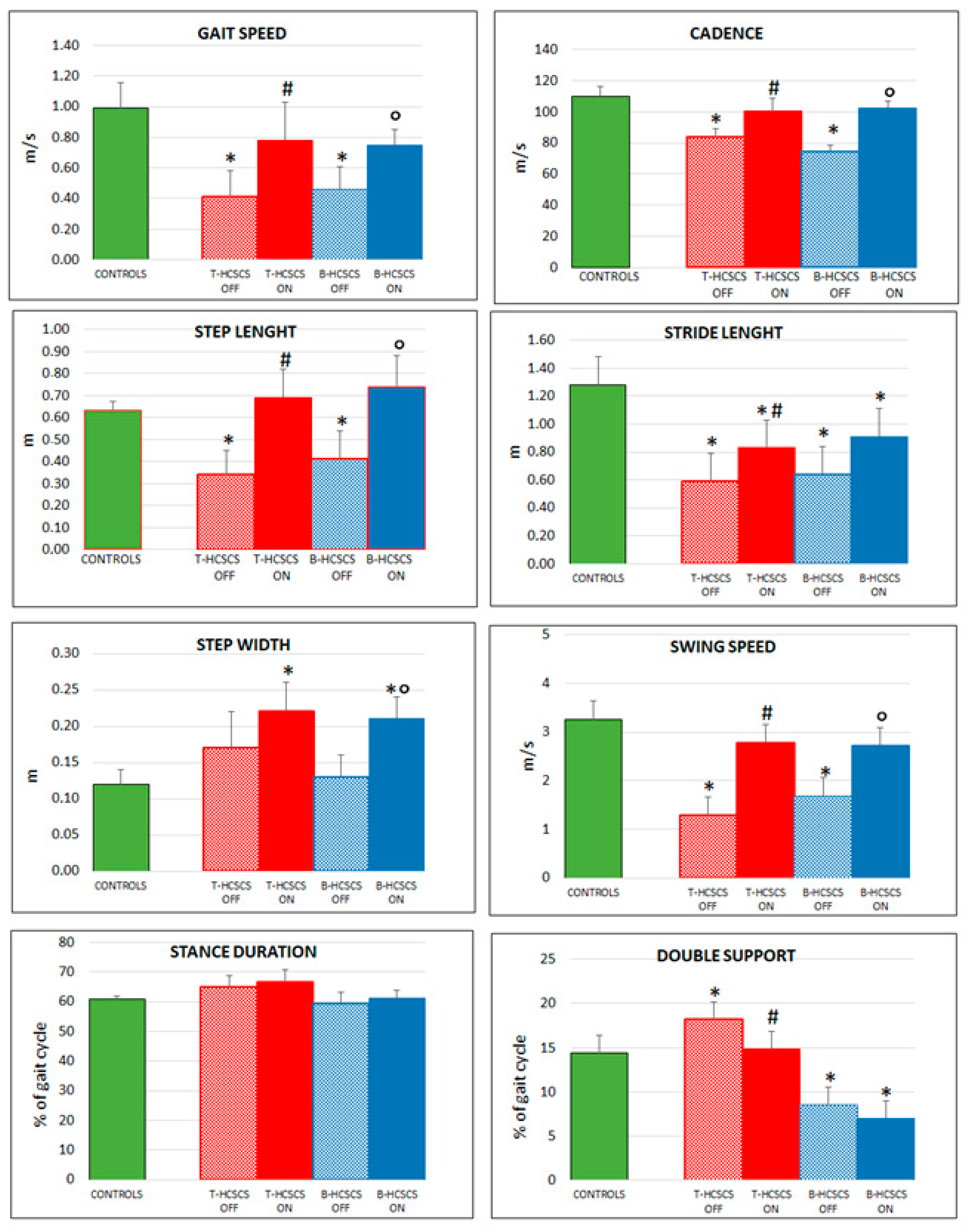
2.5. Instrumental Evaluation of Gait
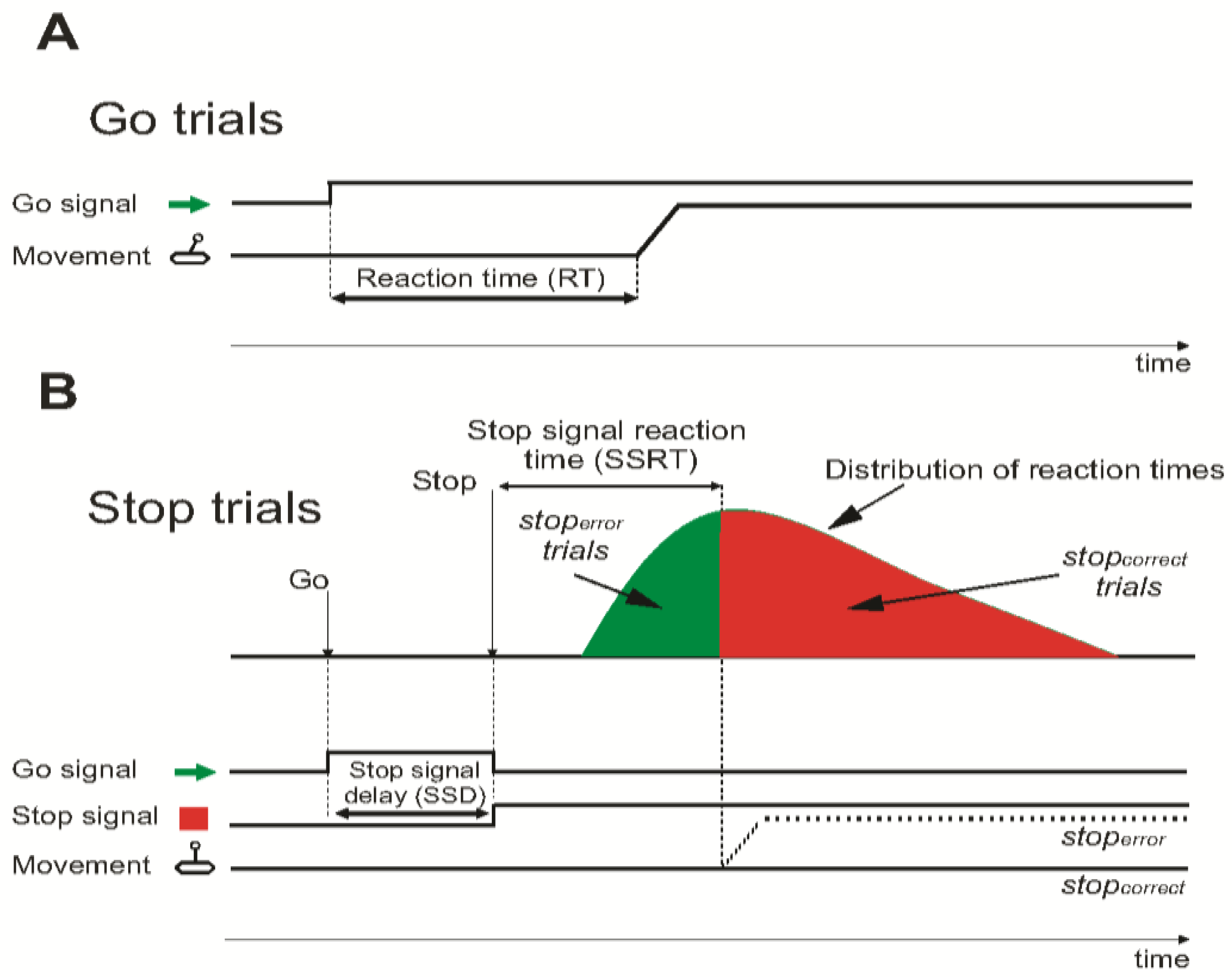
2.6. Cognitive Motor Task
2.7. Autonomic Function Evaluation
2.8. Statistic Analysis
3. Results
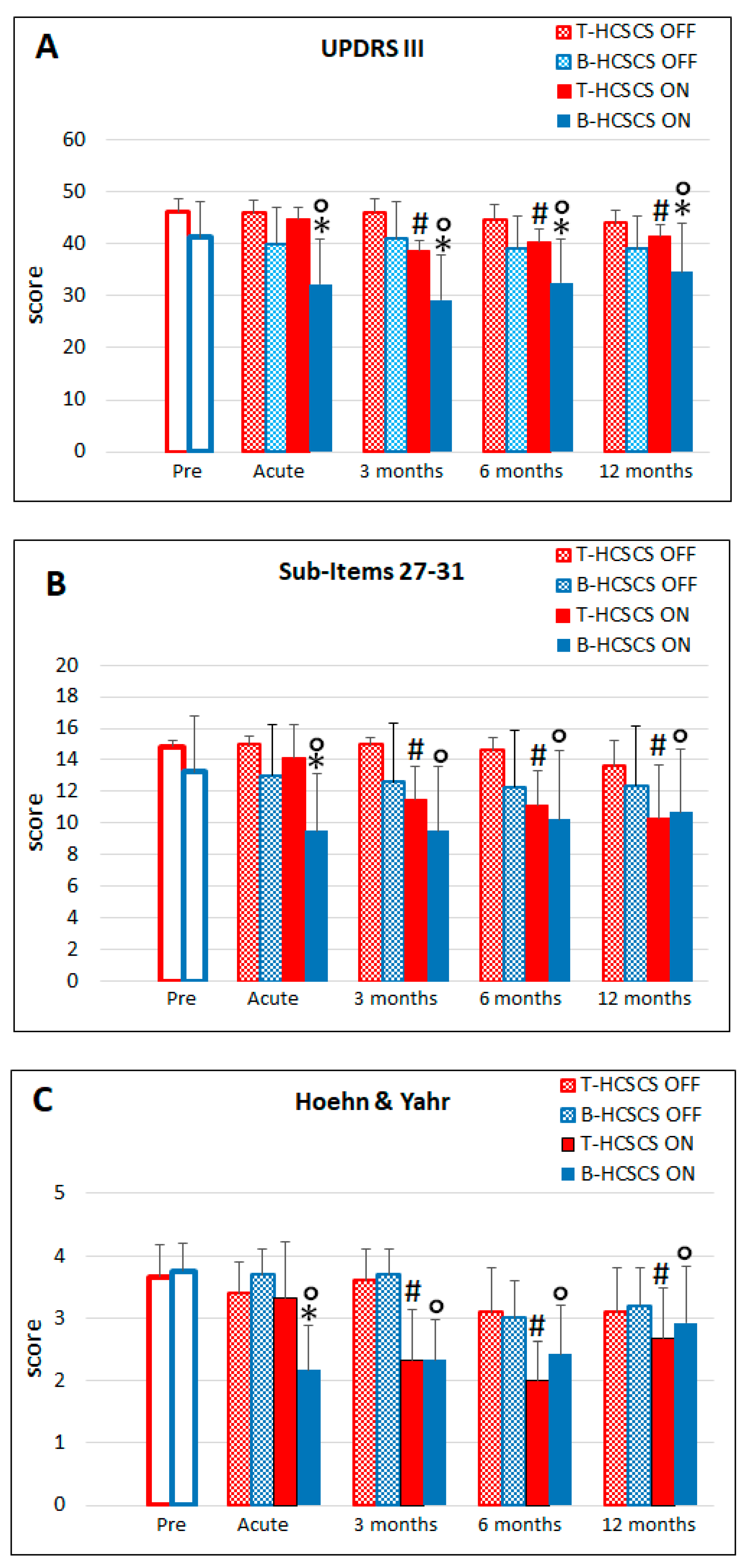
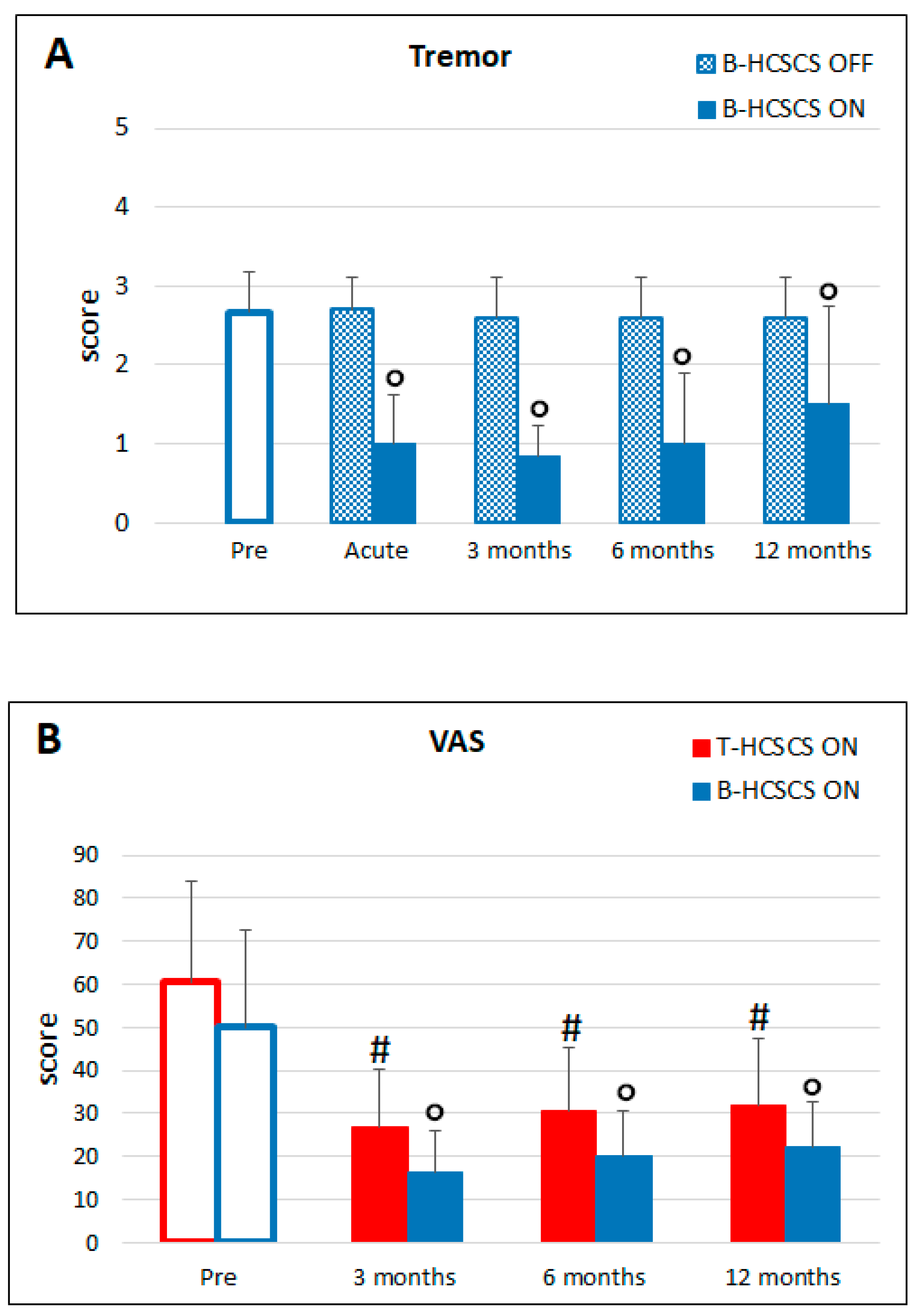
3.1. T-HCSCS (Group I Patients)
3.2. B-HCSCS (Group II Patients)
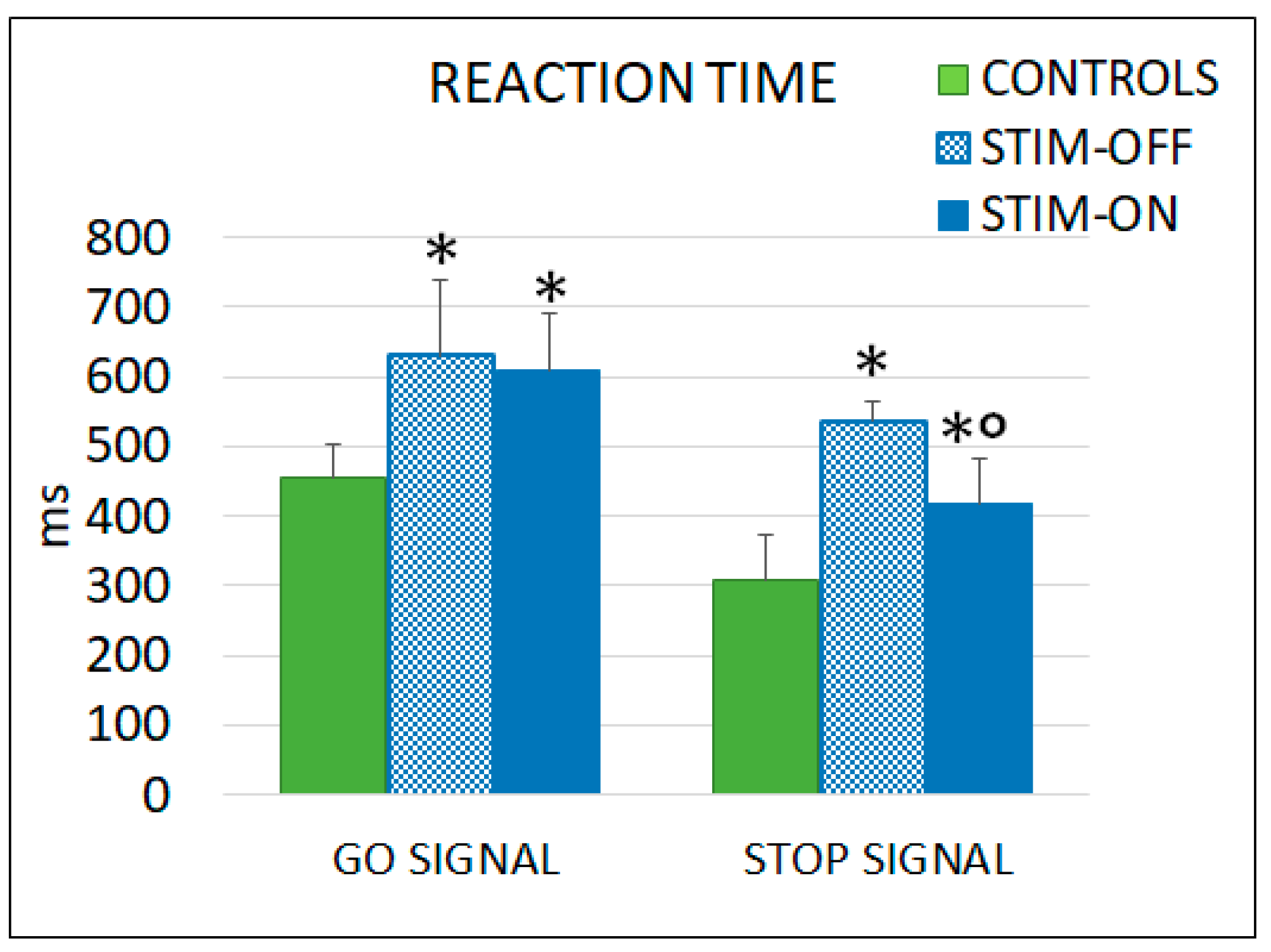
3.3. Cognitive–Motor Task
3.4. Effects on Vegetative Functions
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Shealy, C.N.; Mortimer, J.T.; Reswick, J.B. Electrical inhibition of pain by stimulation of the dorsal columns: Preliminary clinical report. Anesth. Analg. 1967, 46, 489–491. [Google Scholar] [CrossRef] [PubMed]
- Gildenberg, P.L. History of neuroaugmentative procedures. Neurosurg. Clin. N. Am. 2003, 14, 327–337. [Google Scholar] [CrossRef]
- Melzack, R.; Wall, P.D. Pain mechanisms: A new theory. Science 1965, 150, 971–979. [Google Scholar] [CrossRef] [PubMed]
- Melzack, R.; Wall, P.D. Evolution of pain theories. Int. Anesthesiol. Clin. 1970, 8, 3–34. [Google Scholar] [CrossRef]
- Ropero Pelaez, F.J.; Taniguchi, S. The Gate Theory of Pain Revisited: Modeling Different Pain Conditions with a Parsimonious Neurocomputational Model. Neural Plast. 2016, 2016, 4131395. [Google Scholar] [CrossRef] [PubMed]
- Grill, W.M.; Craggs, M.D.; Foreman, R.D.; Ludlow, C.L.; Buller, J.L. Emerging clinical applications of electrical stimulation: Opportunities for restoration of function. J. Rehabil. Res. Dev. 2001, 38, 641–653. [Google Scholar] [PubMed]
- Kowalski, K.E.; Romaniuk, J.R.; Kowalski, T.; DiMarco, A.F. Effects of expiratory muscle activation via high-frequency spinal cord stimulation. J. Appl. Physiol (1985) 2017, 123, 1525–1531. [Google Scholar] [CrossRef]
- Meier, K. Spinal cord stimulation: Background and clinical application. Scand. J. Pain 2014, 5, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, R.; Petersson, P.; Siesser, W.B.; Caron, M.G.; Nicolelis, M.A. Spinal cord stimulation restores locomotion in animal models of Parkinson’s disease. Science 2009, 323, 1578–1582. [Google Scholar] [CrossRef]
- Fuentes, R.; Petersson, P.; Nicolelis, M.A. Restoration of locomotive function in Parkinson’s disease by spinal cord stimulation: Mechanistic approach. Eur. J. Neurosci. 2010, 32, 1100–1108. [Google Scholar] [CrossRef]
- Mendell, L.M. Constructing and deconstructing the gate theory of pain. Pain 2014, 155, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Shinko, A.; Agari, T.; Kameda, M.; Yasuhara, T.; Kondo, A.; Tayra, J.T.; Sato, K.; Sasaki, T.; Sasada, S.; Takeuchi, H.; et al. Spinal cord stimulation exerts neuroprotective effects against experimental Parkinson’s disease. PLoS ONE 2014, 9, e101468. [Google Scholar] [CrossRef]
- Yadav, A.P.; Nicolelis, M.A.L. Electrical stimulation of the dorsal columns of the spinal cord for Parkinson’s disease. Mov Disord. 2017, 32, 820–832. [Google Scholar] [CrossRef] [PubMed]
- Yadav, A.P.; Fuentes, R.; Zhang, H.; Vinholo, T.; Wang, C.H.; Freire, M.A.; Nicolelis, M.A. Chronic spinal cord electrical stimulation protects against 6-hydroxydopamine lesions. Sci. Rep. 2014, 4, 3839. [Google Scholar] [CrossRef] [PubMed]
- Agari, T.; Date, I. Spinal cord stimulation for the treatment of abnormal posture and gait disorder in patients with Parkinson’s disease. Neurol. Med. Chir (Tokyo) 2012, 52, 470–474. [Google Scholar] [CrossRef]
- Hassan, S.; Amer, S.; Alwaki, A.; Elborno, A. A patient with Parkinson’s disease benefits from spinal cord stimulation. J. Clin. Neurosci. 2013, 20, 1155–1156. [Google Scholar] [CrossRef] [PubMed]
- de Lima-Pardini, A.C.; Coelho, D.B.; Souza, C.P.; Souza, C.O.; Ghilardi, M.G.D.S.; Garcia, T.; Voos, M.; Milosevic, M.; Hamani, C.; Teixeira, L.A.; et al. Effects of spinal cord stimulation on postural control in Parkinson’s disease patients with freezing of gait. Elife 2018, 7, e37727. [Google Scholar] [CrossRef]
- Pinto de, S.C.; Hamani, C.; Oliveira, S.C.; Lopez Contreras, W.O.; Dos Santos Ghilardi, M.G.; Cury, R.G.; Reis, B.E.; Jacobsen, T.M.; Talamoni, F.E. Spinal cord stimulation improves gait in patients with Parkinson’s disease previously treated with deep brain stimulation. Mov. Disord. 2017, 32, 278–282. [Google Scholar] [CrossRef] [PubMed]
- Fenelon, G.; Goujon, C.; Gurruchaga, J.M.; Cesaro, P.; Jarraya, B.; Palfi, S.; Lefaucheur, J.P. Spinal cord stimulation for chronic pain improved motor function in a patient with Parkinson’s disease. Parkinsonism Relat. Disord. 2012, 18, 213–214. [Google Scholar] [CrossRef]
- Thiriez, C.; Gurruchaga, J.M.; Goujon, C.; Fenelon, G.; Palfi, S. Spinal stimulation for movement disorders. Neurotherapeutics 2014, 11, 543–552. [Google Scholar] [CrossRef]
- Thevathasan, W.; Mazzone, P.; Jha, A.; Djamshidian, A.; Dileone, M.; Di Lazzaro, V.; Brown, P. Spinal cord stimulation failed to relieve akinesia or restore locomotion in Parkinson disease. Neurology 2010, 74, 1325–1327. [Google Scholar] [CrossRef]
- de Andrade, E.M.; Ghilardi, M.G.; Cury, R.G.; Barbosa, E.R.; Fuentes, R.; Teixeira, M.J.; Fonoff, E.T. Spinal cord stimulation for Parkinson’s disease: A systematic review. Neurosurg. Rev. 2016, 39, 27–35. [Google Scholar] [CrossRef]
- Ahmed, S.; Yearwood, T.; De, R.D.; Vanneste, S. Burst and high frequency stimulation: Underlying mechanism of action. Expert Rev. Med. Devices 2018, 15, 61–70. [Google Scholar] [CrossRef]
- Courtney, P.; Espinet, A.; Mitchell, B.; Russo, M.; Muir, A.; Verrills, P.; Davis, K. Improved Pain Relief with Burst Spinal Cord Stimulation for Two Weeks in Patients Using Tonic Stimulation: Results From a Small Clinical Study. Neuromodulation 2015, 18, 361–366. [Google Scholar] [CrossRef]
- De Vos, C.C.; Bom, M.J.; Vanneste, S.; Lenders, M.W.; De, R.D. Burst spinal cord stimulation evaluated in patients with failed back surgery syndrome and painful diabetic neuropathy. Neuromodulation 2014, 17, 152–159. [Google Scholar] [CrossRef]
- De Ridder, D.; van der Loo, E.; Van der Kelen, K.; Menovsky, T.; Van de Heyning, P.; Moller, A. Do tonic and burst TMS modulate the lemniscal and extralemniscal system differentially? Int. J. Med. Sci. 2007, 4, 242–246. [Google Scholar] [CrossRef]
- De Ridder, D.; Vanneste, S.; Plazier, M.; van der Loo, E.; Menovsky, T. Burst spinal cord stimulation: Toward paresthesia-free pain suppression. Neurosurgery 2010, 66, 986–990. [Google Scholar] [CrossRef] [PubMed]
- De Ridder, D.; Plazier, M.; Kamerling, N.; Menovsky, T.; Vanneste, S. Burst spinal cord stimulation for limb and back pain. World Neurosurg. 2013, 80, 642–649. [Google Scholar] [CrossRef] [PubMed]
- De Ridder, D.; Lenders, M.W.; De Vos, C.C.; Dijkstra-Scholten, C.; Wolters, R.; Vancamp, T.; Van, L.P.; Van, H.T.; Vanneste, S. A 2-center comparative study on tonic versus burst spinal cord stimulation: Amount of responders and amount of pain suppression. Clin. J. Pain 2015, 31, 433–437. [Google Scholar] [CrossRef] [PubMed]
- De Ridder, D.; Perera, S.; Vanneste, S. Are 10 kHz Stimulation and Burst Stimulation Fundamentally the Same? Neuromodulation 2017, 20, 650–653. [Google Scholar] [CrossRef] [PubMed]
- Deer, T.R.; Campos, L.W.; Pope, J.E. Evaluation of Abbott’s BurstDR stimulation device for the treatment of chronic pain. Expert Rev. Med. Devices 2017, 14, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Meuwissen, K.P.V.; Gu, J.W.; Zhang, T.C.; Joosten, E.A.J. Conventional-SCS vs. Burst-SCS and the Behavioral Effect on Mechanical Hypersensitivity in a Rat Model of Chronic Neuropathic Pain: Effect of Amplitude. Neuromodulation 2018, 21, 19–30. [Google Scholar] [CrossRef]
- De Ridder, D.; Vancamp, T.; Lenders, M.W.; De Vos, C.C.; Vanneste, S. Is preoperative pain duration important in spinal cord stimulation? A comparison between tonic and burst stimulation. Neuromodulation 2015, 18, 13–17. [Google Scholar] [CrossRef]
- Schu, S.; Slotty, P.J.; Bara, G.; von, K.M.; Edgar, D.; Vesper, J. A prospective, randomised, double-blind, placebo-controlled study to examine the effectiveness of burst spinal cord stimulation patterns for the treatment of failed back surgery syndrome. Neuromodulation 2014, 17, 443–450. [Google Scholar] [CrossRef]
- Mazzone, P.; Paoloni, M.; Mangone, M.; Santilli, V.; Insola, A.; Fini, M.; Scarnati, E. Unilateral deep brain stimulation of the pedunculopontine tegmental nucleus in idiopathic Parkinson’s disease: Effects on gait initiation and performance. Gait Posture 2014, 40, 357–362. [Google Scholar] [CrossRef]
- Mazzone, P.; Vitale, F.; Capozzo, A.; Viselli, F.; Scarnati, E. Deep brain stimulation of the pedunculopontine tegmental nucleus improves static balance in Parkinson’s disease. In Neuromodulation 2nd; Krames, E., Hunter Peckham, P., Rezai, A., Eds.; Academic Press Elsevier: New York, NY, USA, 2018; pp. 967–976. [Google Scholar]
- Benis, D.; David, O.; Piallat, B.; Kibleur, A.; Goetz, L.; Bhattacharjee, M.; Fraix, V.; Seigneuret, E.; Krack, P.; Chabardes, S.; et al. Response inhibition rapidly increases single-neuron responses in the subthalamic nucleus of patients with Parkinson’s disease. Cortex 2016, 84, 111–123. [Google Scholar] [CrossRef] [PubMed]
- Brunamonti, E.; Ferraina, S.; Pare, M. Controlled movement processing: Evidence for a common inhibitory control of finger, wrist, and arm movements. Neuroscience 2012, 215, 69–78. [Google Scholar] [CrossRef]
- Mione, V.; Canterini, S.; Brunamonti, E.; Pani, P.; Donno, F.; Fiorenza, M.T.; Ferraina, S. Both the COMT Val158Met single-nucleotide polymorphism and sex-dependent differences influence response inhibition. Front. Behav. Neurosci. 2015, 9, 127. [Google Scholar] [CrossRef]
- Olivito, G.; Brunamonti, E.; Clausi, S.; Pani, P.; Chiricozzi, F.R.; Giamundo, M.; Molinari, M.; Leggio, M.; Ferraina, S. Atrophic degeneration of cerebellum impairs both the reactive and the proactive control of movement in the stop signal paradigm. Exp. Brain Res. 2017, 235, 2971–2981. [Google Scholar] [CrossRef] [PubMed]
- Battaglia-Mayer, A.; Buiatti, T.; Caminiti, R.; Ferraina, S.; Lacquaniti, F.; Shallice, T. Correction and suppression of reaching movements in the cerebral cortex: Physiological and neuropsychological aspects. Neurosci. Biobehav. Rev. 2014, 42, 232–251. [Google Scholar] [CrossRef]
- Rocchi, C.; Pierantozzi, M.; Galati, S.; Chiaravalloti, A.; Pisani, V.; Prosperetti, C.; Lauretti, B.; Stampanoni, B.M.; Olivola, E.; Schillaci, O.; et al. Autonomic Function Tests and MIBG in Parkinson’s Disease: Correlation to Disease Duration and Motor Symptoms. CNS Neurosci. Ther. 2015, 21, 727–732. [Google Scholar] [CrossRef] [PubMed]
- Rocchi, C.; Pierantozzi, M.; Pisani, V.; Marfia, G.A.; Di, G.A.; Stanzione, P.; Bernardi, G.; Stefani, A. The impact of rotigotine on cardiovascular autonomic function in early Parkinson’s disease. Eur. Neurol. 2012, 68, 187–192. [Google Scholar] [CrossRef]
- Pstras, L.; Thomaseth, K.; Waniewski, J.; Balzani, I.; Bellavere, F. The Valsalva manoeuvre: Physiology and clinical examples. Acta Physiol. (Oxf.) 2016, 217, 103–119. [Google Scholar] [CrossRef]
- Verbruggen, F.; Logan, G.D. Models of response inhibition in the stop-signal and stop-change paradigms. Neurosci. Biobehav. Rev. 2009, 33, 647–661. [Google Scholar] [CrossRef]
- Nishioka, K.; Nakajima, M. Beneficial therapeutic effects of spinal cord stimulation in advanced cases of Parkinson’s disease with intractable chronic pain: A case series. Neuromodulation 2015, 18, 751–753. [Google Scholar] [CrossRef]
- Santana, M.B.; Halje, P.; Simplicio, H.; Richter, U.; Freire, M.A.M.; Petersson, P.; Fuentes, R.; Nicolelis, M.A.L. Spinal cord stimulation alleviates motor deficits in a primate model of Parkinson disease. Neuron 2014, 84, 716–722. [Google Scholar] [CrossRef]
- Crosby, N.D.; Weisshaar, C.L.; Smith, J.R.; Zeeman, M.E.; Goodman-Keiser, M.D.; Winkelstein, B.A. Burst and Tonic Spinal Cord Stimulation Differentially Activate GABAergic Mechanisms to Attenuate Pain in a Rat Model of Cervical Radiculopathy. IEEE Trans. Biomed. Eng. 2015, 62, 1604–1613. [Google Scholar] [CrossRef]
- Ahmed, Z.; Wieraszko, A. Trans-spinal direct current enhances corticospinal output and stimulation-evoked release of glutamate analog, D-2,3-(3)H-aspartic acid. J. Appl. Physiol. (1985) 2012, 112, 1576–1592. [Google Scholar] [CrossRef]
- Garcia-Rill, E.; Luster, B.; D’Onofrio, S.; Mahaffey, S.; Bisagno, V.; Urbano, F.J. Pedunculopontine arousal system physiology—Deep brain stimulation (DBS). Sleep Sci. 2015, 8, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Rill, E.; Luster, B.; D’Onofrio, S.; Mahaffey, S.; Bisagno, V.; Urbano, F.J. Implications of gamma band activity in the pedunculopontine nucleus. J. Neural Transm. (Vienna) 2016, 123, 655–665. [Google Scholar] [CrossRef] [PubMed]
- Mazzone, P.; Vilela, F.O.; Viselli, F.; Insola, A.; Sposato, S.; Vitale, F.; Scarnati, E. Our first decade of experience in deep brain stimulation of the brainstem: Elucidating the mechanism of action of stimulation of the ventrolateral pontine tegmentum. J. Neural Transm. (Vienna) 2016, 123, 751–767. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Rill, E.; Mahaffey, S.; Hyde, J.R.; Urbano, F.J. Bottom-up gamma maintenance in various disorders. Neurobiol. Dis. 2018. [Google Scholar] [CrossRef] [PubMed]
- Mazzone, P.; Pisani, R.; Pizio, N.; Arrigo, A.; Nobili, F. Cerebral blood flow and somatosensory evoked response changes induced by spinal cord stimulation: Preliminary follow-up observations. Stereotact. Funct. Neurosurg. 1994, 62, 179–185. [Google Scholar] [CrossRef]
- Mazzone, P.; Pisani, R.; Nobili, F.; Arrigo, A.; Gambaro, M.; Rodriguez, G. Assessment of regional cerebral blood flow during spinal cord stimulation in humans. Stereotact. Funct. Neurosurg. 1995, 64, 197–201. [Google Scholar] [PubMed]
- Mazzone, P.; Rodriguez, G.; Arrigo, A.; Nobili, F.; Pisani, R.; Rosadini, G. Cerebral haemodynamic changes induced by spinal cord stimulation in man. Ital. J. Neurol. Sci. 1996, 17, 55–57. [Google Scholar] [CrossRef] [PubMed]
- Rasche, D.; Siebert, S.; Stippich, C.; Kress, B.; Nennig, E.; Sartor, K.; Tronnier, V.M. Spinal cord stimulation in Failed-Back-Surgery-Syndrome. Preliminary study for the evaluation of therapy by functional magnetic resonance imaging (fMRI). Schmerz 2005, 19, 497–500. [Google Scholar] [CrossRef] [PubMed]
- Si, J.; Dang, Y.; Zhang, Y.; Li, Y.; Zhang, W.; Yang, Y.; Cui, Y.; Lou, X.; He, J.; Jiang, T. Spinal Cord Stimulation Frequency Influences the Hemodynamic Response in Patients with Disorders of Consciousness. Neurosci. Bull. 2018, 34, 659–667. [Google Scholar] [CrossRef]
- Nambu, A.; Tokuno, H.; Takada, M. Functional significance of the cortico-subthalamo-pallidal ‘hyperdirect’ pathway. Neurosci. Res. 2002, 43, 111–117. [Google Scholar] [CrossRef]
- Nambu, A. A new approach to understand the pathophysiology of Parkinson’s disease. J. Neurol. 2005, 252 (Suppl. 4), IV1–IV4. [Google Scholar] [CrossRef]
- Canolty, R.T.; Edwards, E.; Dalal, S.S.; Soltani, M.; Nagarajan, S.S.; Kirsch, H.E.; Berger, M.S.; Barbaro, N.M.; Knight, R.T. High gamma power is phase-locked to theta oscillations in human neocortex. Science 2006, 313, 1626–1628. [Google Scholar] [CrossRef]
- Liu, C.C.; Chien, J.H.; Kim, J.H.; Chuang, Y.F.; Cheng, D.T.; Anderson, W.S.; Lenz, F.A. Cross-frequency coupling in deep brain structures upon processing the painful sensory inputs. Neuroscience 2015, 303, 412–421. [Google Scholar] [CrossRef]
- Voloh, B.; Valiante, T.A.; Everling, S.; Womelsdorf, T. Theta-gamma coordination between anterior cingulate and prefrontal cortex indexes correct attention shifts. Proc. Natl. Acad. Sci. USA 2015, 112, 8457–8462. [Google Scholar] [CrossRef]
- De Ridder, D.; van der Loo, E.; Van der Kelen, K.; Menovsky, T.; Van de Heyning, P.; Moller, A. Theta, alpha and beta burst transcranial magnetic stimulation: Brain modulation in tinnitus. Int. J. Med. Sci. 2007, 4, 237–241. [Google Scholar] [CrossRef]
- Llinas, R.R.; Ribary, U.; Jeanmonod, D.; Kronberg, E.; Mitra, P.P. Thalamocortical dysrhythmia: A neurological and neuropsychiatric syndrome characterized by magnetoencephalography. Proc. Natl. Acad. Sci. USA 1999, 96, 15222–15227. [Google Scholar] [CrossRef] [PubMed]
- Von Stein, A.; Sarnthein, J. Different frequencies for different scales of cortical integration: From local gamma to long range alpha/theta synchronization. Int. J. Psychophysiol. 2000, 38, 301–313. [Google Scholar] [CrossRef]
- Ding, X.; Mountain, D.J.; Subramanian, V.; Singh, K.; Williams, C.A. The effect of high cervical spinal cord stimulation on the expression of SP, NK-1 and TRPV1 mRNAs during cardiac ischemia in rat. Neurosci. Lett. 2007, 424, 139–144. [Google Scholar] [CrossRef]
- Simpson, R.K., Jr.; Robertson, C.S.; Goodman, J.C.; Halter, J.A. Recovery of amino acid neurotransmitters from the spinal cord during posterior epidural stimulation: A preliminary study. J. Am. Paraplegia Soc. 1991, 14, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Ultenius, C.; Song, Z.; Lin, P.; Meyerson, B.A.; Linderoth, B. Spinal GABAergic mechanisms in the effects of spinal cord stimulation in a rodent model of neuropathic pain: Is GABA synthesis involved? Neuromodulation 2013, 16, 114–120. [Google Scholar] [CrossRef]

| Patients | Age | Gender | Disease | Duration (years) | L-Dopa Pre-Op (mg/day) | L-Dopa Post-Op (mg/day) | HCSCS Early Indication | |
|---|---|---|---|---|---|---|---|---|
| Group I | 1 | 76 | M | LBP + IPD | 18 | 1800 | 1300 | Pain |
| 2 | 79 | M | VP + AP * | 20 | 600 ° | 600 | Pain | |
| 3 | 58 | M | VPD | 16 | 1000 | 1000 | PD | |
| 4 | 74 | M | VP + VPD | 27 | 1500 | 1200 | Pain | |
| 5 | 71 | M | FBSS + IPD * | 9 | 1300 | 1100 | Pain | |
| 6 | 69 | M | LBP + IPD | 13 | 1800 | 1300 | Pain | |
| Mean ± SD | 71.1 ± 7.3 | 17.1 ± 6.1 | 1334 ± 472 | 1083.3 ± 263.9 | ||||
| Group II | 7 | 72 | M | AP | 5 | (-) | (-) | AP |
| 8 | 48 | M | VPD | 11 | 850 | 850 | PD | |
| 9 | 65 | F | IPD | 5 | 500 | 350 | PD | |
| 10 | 61 | M | VPD | 11 | 1200 | 750 | PD | |
| 11 | 73 | M | VPD | 16 | 800 | 800 | PD | |
| 12 | 73 | M | VPD | 14 | 1000 | 750 | PD | |
| 13 | 76 | M | VPD | 9 | 650 | 650 | PD | |
| 14 | 81 | M | VP + AP * | 20 | 600 | 600 | PD/Pain | |
| 15 | 64 | F | VPD | 8 | 350 | 350 | PD | |
| 16 | 63 | M | VPD | 20 | 1200 | 1200 | PD | |
| 17 | 43 | M | VPD | 9 | 1200 | 1000 | PD | |
| 18 | 69 | M | AP | 5 | (-) | (-) | AP | |
| Mean ± SD | 65.6 ± 11.1 | 11.1 ± 5.3 | 835 ± 310 (§) | 730 ± 263.7 |
| Patients | Hz ON | Hz OFF | Pulse Width (µs) | Intensity | Electrical Changes Every 3 Months | Improvement |
|---|---|---|---|---|---|---|
| Group I | ||||||
| 1 | 185 | - | 80 | 2.5 V | 9 | delayed |
| 2 | 130 | - | 60 | 3.6 V | 16 | delayed |
| 3 | 130 | - | 90 | 4.0 V | 15 | acute |
| 4 | 185 | - | 210 | 1.3 V | 26 | delayed |
| 5 | 135 | - | 60 | 3.5 V | 21 | delayed |
| 6 | 135 | - | 60 | 3.5 V | 19 | delayed |
| Mean ± SD | 150.0 ± 27.2 | 93.3 ± 58.5 | 3.0 ± 0.9 | 17.6 ± 5.7 | ||
| Group II | ||||||
| 7 | 500 | 40 | 1000 | 0.9 mAmp | 4 | acute |
| (*) 8 | 500 | 40 | 1000 | 0.6 mAmp | 5 | acute |
| (*) 9 | 500 | 40 | 1000 | 0.7 mAmp | 4 | acute |
| 10 | 500 | 40 | 1000 | 0.9 mAmp | 4 | acute |
| (*) 11 | 500 | 40 | 1000 | 0.7 mAmp | 4 | acute |
| 12 | 250 | 40 | 1000 | 0.6 mAmp | 2 | acute |
| 13 | 500 | 40 | 1000 | 0.7 mAmp | 4 | acute |
| (*) 14 | 500 | 40 | 1000 | 0.3 mAmp | 5 | acute |
| 15 | 500 | 40 | 1000 | 0.2 mAmp | 3 | acute |
| (*) 16 | 500 | 40 | 1000 | 0.5 mAmp | 5 | acute |
| 17 | 500 | 40 | 1000 | 0.4 mAmp | 3 | acute |
| (*) 18 | 500 | 40 | 1000 | 0.2 mAmp | 4 | acute |
| Mean ± SD | 479.2 ± 72.2 | 40 | 1000 | 0.5 ± 0.2 | 3.9 ± 0.9 |
| Stimulation OFF | Stimulation ON | |
|---|---|---|
| Head-up tilt test | ||
| ∆ SBP (mmHg) | −41 | −17 |
| ∆ DBP (mmHg) | −13 | 16 |
| ∆ HR (bpm) | 11 | 22 |
| VALSAVA | ||
| OV mmHg | Absent | 4 |
| VR | 1.41 | 1.21 |
| Hand grip test | ||
| ∆ DBP (mmHg) | 3 | 15 |
| Deep–Breathing test | ||
| I-E difference (bpm) | 11 | 16 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mazzone, P.; Viselli, F.; Ferraina, S.; Giamundo, M.; Marano, M.; Paoloni, M.; Masedu, F.; Capozzo, A.; Scarnati, E. High Cervical Spinal Cord Stimulation: A One Year Follow-Up Study on Motor and Non-Motor Functions in Parkinson’s Disease. Brain Sci. 2019, 9, 78. https://doi.org/10.3390/brainsci9040078
Mazzone P, Viselli F, Ferraina S, Giamundo M, Marano M, Paoloni M, Masedu F, Capozzo A, Scarnati E. High Cervical Spinal Cord Stimulation: A One Year Follow-Up Study on Motor and Non-Motor Functions in Parkinson’s Disease. Brain Sciences. 2019; 9(4):78. https://doi.org/10.3390/brainsci9040078
Chicago/Turabian StyleMazzone, Paolo, Fabio Viselli, Stefano Ferraina, Margherita Giamundo, Massimo Marano, Marco Paoloni, Francesco Masedu, Annamaria Capozzo, and Eugenio Scarnati. 2019. "High Cervical Spinal Cord Stimulation: A One Year Follow-Up Study on Motor and Non-Motor Functions in Parkinson’s Disease" Brain Sciences 9, no. 4: 78. https://doi.org/10.3390/brainsci9040078
APA StyleMazzone, P., Viselli, F., Ferraina, S., Giamundo, M., Marano, M., Paoloni, M., Masedu, F., Capozzo, A., & Scarnati, E. (2019). High Cervical Spinal Cord Stimulation: A One Year Follow-Up Study on Motor and Non-Motor Functions in Parkinson’s Disease. Brain Sciences, 9(4), 78. https://doi.org/10.3390/brainsci9040078







