A Pilot Quantitative Evaluation of Early Life Language Development in Fragile X Syndrome
Abstract
1. Introduction
2. Method
2.1. Participants
2.2. Measures
2.2.1. LENA
2.2.2. LENA Developmental Snapshot (LDS)
2.3. Procedures
2.4. Data Analysis
3. Results
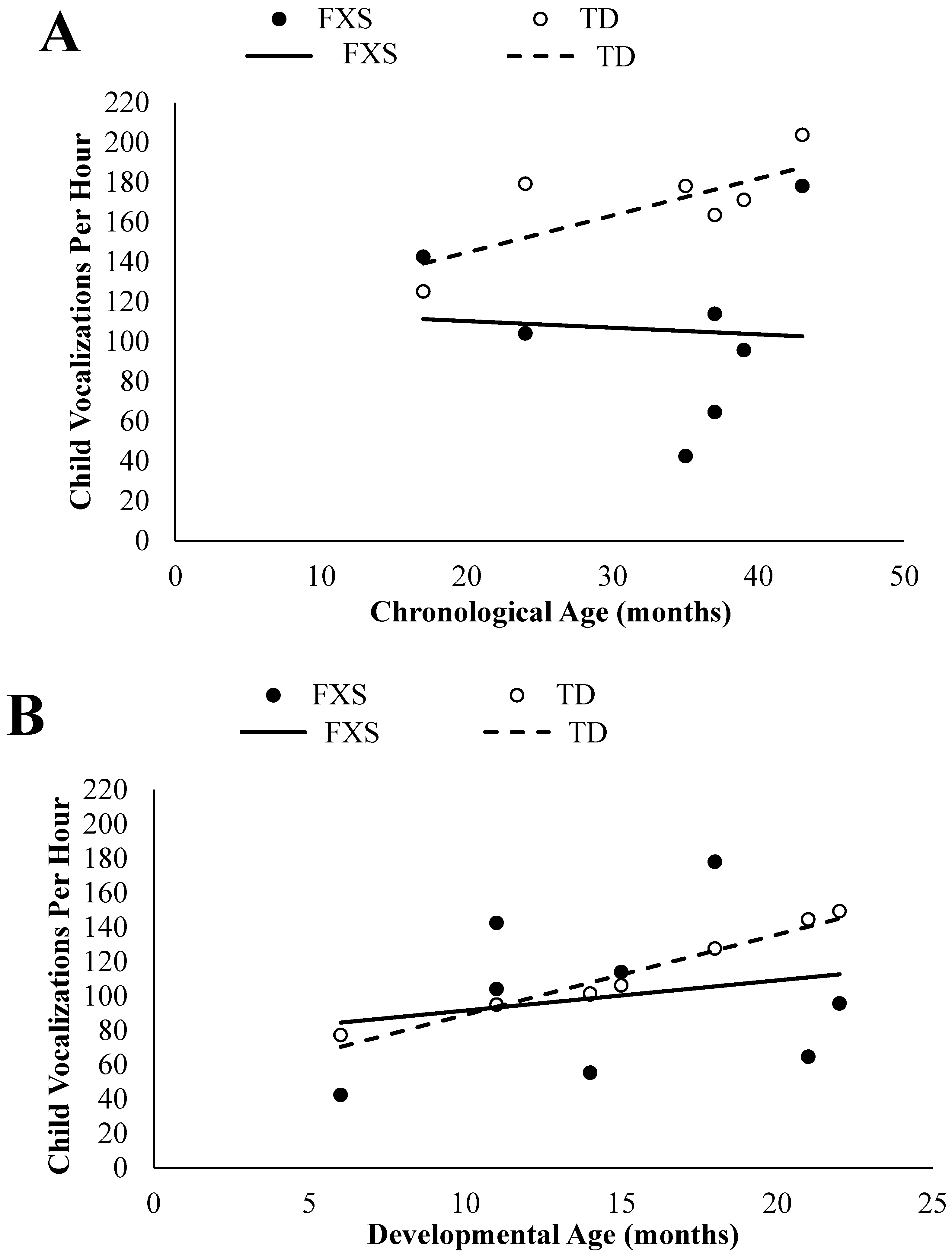
3.1. Child Vocalizations
3.1.1. Chronological Age Comparisons
3.1.2. Developmental Age Comparisons
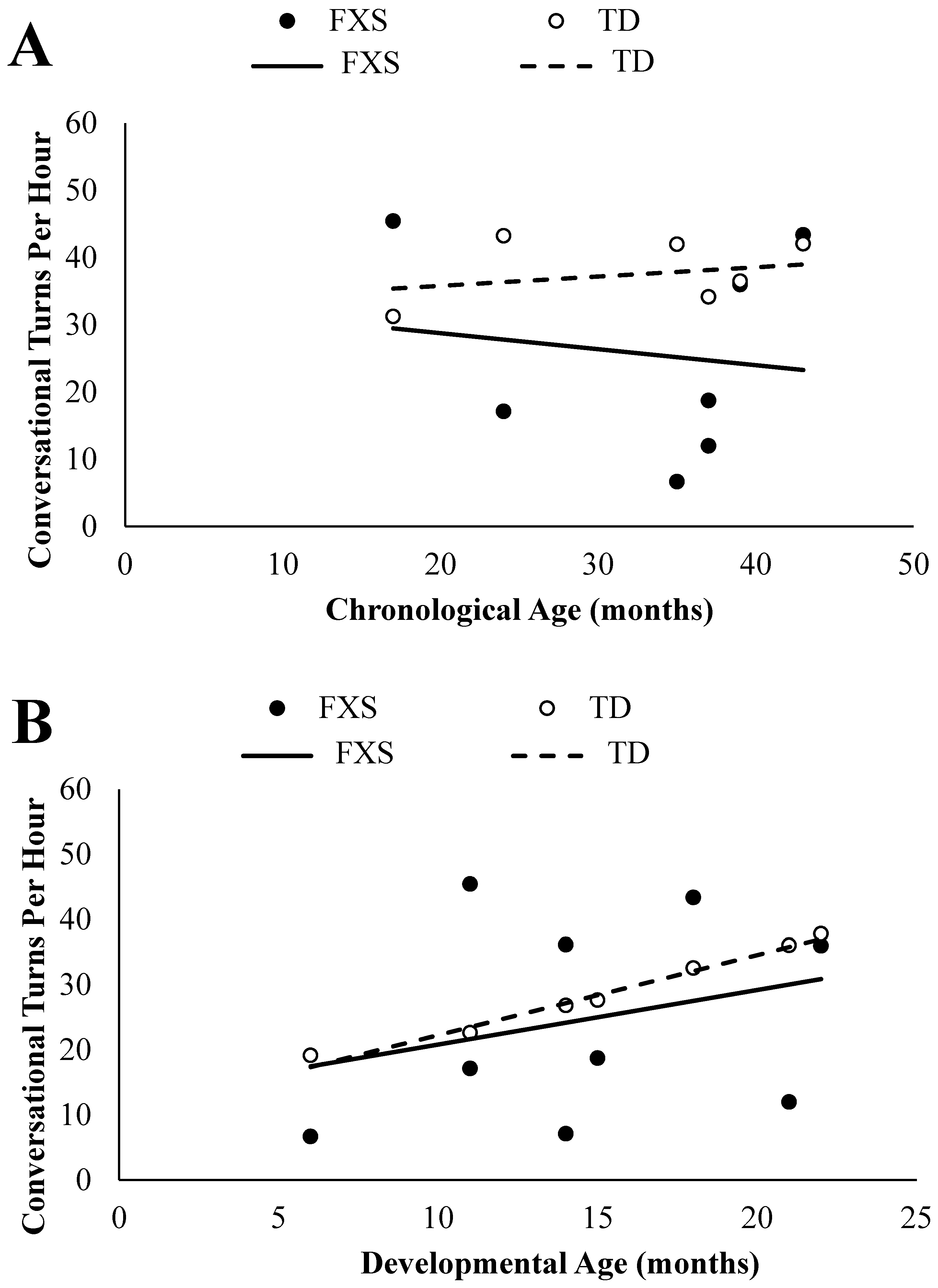
3.2. Conversational Turns
3.2.1. Chronological Age Comparisons
3.2.2. Developmental Age Comparisons
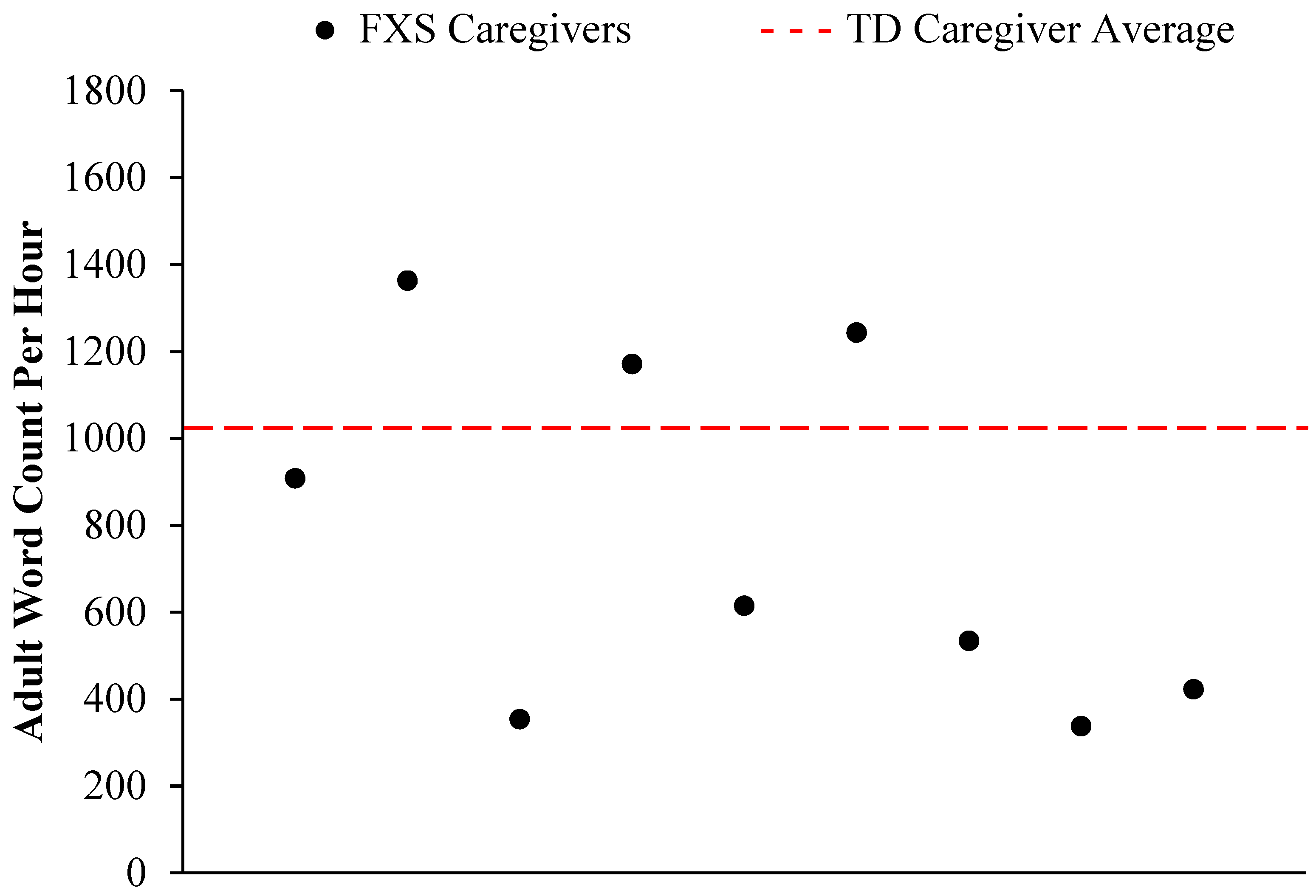
3.3. Adult Word Count
4. Discussion
4.1. Summary of Findings
4.2. Limitations
5. Conclusions and Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hagerman, R.J.; Hagerman, P.J. (Eds.) Fragile X Syndrome: Diagnosis, Treatment, and Research, 3rd ed.; Johns Hopkins Univ. Press: Baltimore, MD, USA, 2002; p. 540. [Google Scholar]
- Cordeiro, L.; Ballinger, E.; Hagerman, R.; Hessl, D. Clinical assessment of DSM-IV anxiety disorders in fragile X syndrome: Prevalence and characterization. J. Neurodev. Disord. 2011, 3, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Martin, G.E.; Losh, M.; Estigarribia, B.; Sideris, J.; Roberts, J. Longitudinal profiles of expressive vocabulary, syntax and pragmatic language in boys with fragile X syndrome or Down syndrome. Int. J. Lang. Commun. Disord. 2013, 48, 432–443. [Google Scholar] [CrossRef]
- Roberts, J.E.; Mirrett, P.; Burchinal, M. Receptive and Expressive Communication Development of Young Males with Fragile X Syndrome. Am. J. Ment. Retard. 2001, 106, 216–230. [Google Scholar] [CrossRef]
- Pena, M.; Maki, A.; Kovacic, D.; Dehaene-Lambertz, G.; Koizumi, H.; Bouquet, F.; Mehler, J. Sounds and silence: An optical topography study of language recognition at birth. Proc. Natl. Acad. Sci. USA 2003, 100, 11702–11705. [Google Scholar] [CrossRef] [PubMed]
- Vouloumanos, A.; Werker, J.F. Listening to language at birth: Evidence for a bias for speech in neonates. Dev. Sci. 2007, 10, 159–164. [Google Scholar] [CrossRef]
- Ferguson, C.A.; Menn, L.; Stoel-Gammon, C. Phonological Development: Models, Research, Implications; York Press: Timonium, MD, USA, 1992; ISBN 9780912752242. [Google Scholar]
- Tamis-LeMonda, C.S.; Bornstein, M.H.; Baumwell, L. Maternal responsiveness and children’s achievement of language milestones. Child Dev. 2001, 72, 748–767. [Google Scholar] [CrossRef] [PubMed]
- Kuhl, P.K. Early language acquisition: Cracking the speech code. Nat. Rev. Neurosci. 2004, 5, 831–843. [Google Scholar] [CrossRef] [PubMed]
- Hart, B.; Risley, T.R. Meaningful Differences in the Everyday Experience of Young American Children; Paul H Brookes Publishing: Baltimore, MD, USA, 1995; ISBN 1-55766-197-9. [Google Scholar]
- Tamis-LeMonda, C.S.; Shannon, J.D.; Cabrera, N.J.; Lamb, M.E. Fathers and Mothers at Play with Their 2- and 3-Year-Olds: Contributions to Language and Cognitive Developmen. Child Dev. 2004, 75, 1806–1820. [Google Scholar] [CrossRef] [PubMed]
- Patten, E.; Belardi, K.; Baranek, G.T.; Watson, L.R.; Labban, J.D.; Oller, D.K. Vocal patterns in infants with autism spectrum disorder: Canonical babbling status and vocalization frequency. J. Autism Dev. Disord. 2014, 44, 2413–2428. [Google Scholar] [CrossRef]
- Longard, J.; Brian, J.; Zwaigenbaum, L.; Duku, E.; Moore, C.; Smith, I.M.; Garon, N.; Szatmari, P.; Vaillancourt, T.; Bryson, S. Early expressive and receptive language trajectories in high-risk infant siblings of children with autism spectrum disorder. Autism Dev. Lang. Impair. 2017, 2, 1–11. [Google Scholar] [CrossRef]
- Masataka, N. Why early linguistic milestones are delayed in children with Williams syndrome: Late onset of hand banging as a possible rate-limiting constraint on the emergence of canonical babbling. Dev. Sci. 2001, 4, 158. [Google Scholar] [CrossRef]
- Belardi, K.; Watson, L.R.; Faldowski, R.A.; Hazlett, H.; Crais, E.; Baranek, G.T.; McComish, C.; Patten, E.; Oller, D.K. A Retrospective Video Analysis of Canonical Babbling and Volubility in Infants with Fragile X Syndrome at 9–12 Months of Age. J. Autism Dev. Disord. 2017, 47, 1193–1206. [Google Scholar] [CrossRef] [PubMed]
- Ellis Weismer, S.; Lord, C.; Esler, A. Early language patterns of toddlers on the autism spectrum compared to toddlers with developmental delay. J. Autism Dev. Disord. 2010, 40, 1259–1273. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.E.; Hennon, E.A.; Price, J.R.; Dear, E.; Anderson, K.; Vandergrift, N.A. Expressive Language During Conversational Speech in Boys with Fragile X Syndrome. Am. J. Ment. Retard. 2007, 112, 1–17. [Google Scholar] [CrossRef]
- Haebig, E.; Sterling, A.; Hoover, J. Examining the Language Phenotype in Children with Typical Development, Specific Language Impairment, and Fragile X Syndrome. J. Speech Lang. Hear. Res. 2016, 59, 1046–1058. [Google Scholar] [CrossRef] [PubMed]
- Finestack, L.H.; Abbeduto, L. Expressive Language Profiles of Verbally Expressive Adolescents and Young Adults with Down Syndrome or Fragile X Syndrome. J. Speech Lang. Hear. Res. 2010, 53, 1334–1348. [Google Scholar] [CrossRef]
- Roberts, J.E.; Hatton, D.D.; Bailey, D.B. Development and Behavior of Male Toddlers with Fragile X Syndrome. J. Early Interv. 2001, 24, 207–223. [Google Scholar] [CrossRef]
- Hinton, R.; Budimirovic, D.B.; Marschik, P.B.; Talisa, V.B.; Einspieler, C.; Gipson, T.; Johnston, M.V. Parental reports on early language and motor milestones in fragile X syndrome with and without autism spectrum disorders. Dev. Neurorehabil. 2013, 16, 58–66. [Google Scholar] [CrossRef]
- Marschik, P.B.; Bartl-Pokorny, K.D.; Sigafoos, J.; Urlesberger, L.; Pokorny, F.; Didden, R.; Einspieler, C.; Kaufmann, W.E. Development of socio-communicative skills in 9- to 12-month-old individuals with fragile X syndrome. Res. Dev. Disabil. 2014, 35, 597–602. [Google Scholar] [CrossRef]
- Sigafoos, J.; Woodyatt, G.; Keen, D.; Tait, K.; Tucker, M.; Roberts-Pennell, D.; Pittendreigh, N. Identifying Potential Communicative Acts in Children with Developmental and Physical Disabilities. Commun. Disord. Q. 2000, 21, 77–86. [Google Scholar] [CrossRef]
- Kover, S.T.; McCary, L.M.; Ingram, A.M.; Hatton, D.D.; Roberts, J.E. Language development in infants and toddlers with fragile X syndrome: Change over time and the role of attention. Am. J. Intellect. Dev. Disabil. 2015, 120, 125–144. [Google Scholar] [CrossRef] [PubMed]
- Warren, S.F.; Gilkerson, J.; Richards, J.A.; Oller, D.K.; Xu, D.; Yapanel, U.; Gray, S. What automated vocal analysis reveals about the vocal production and language learning environment of young children with autism. J. Autism Dev. Disord. 2010, 40, 555–569. [Google Scholar] [CrossRef] [PubMed]
- Warren, S.F.; Brady, N.; Sterling, A.; Fleming, K.; Marquis, J. Maternal responsivity predicts language development in young children with fragile X syndrome. Am. J. Intellect. Dev. Disabil. 2010, 115, 54–75. [Google Scholar] [CrossRef] [PubMed]
- Brady, N.; Warren, S.F.; Fleming, K.; Keller, J.; Sterling, A. Effect of Sustained Maternal Responsivity on Later Vocabulary Development in Children with Fragile X Syndrome. J. Speech Lang. Hear. Res. 2014, 57, 212–226. [Google Scholar] [CrossRef]
- Sterling, A.M.; Warren, S.F.; Brady, N.; Fleming, K. Influences on maternal responsivity in mothers of children with fragile X syndrome. Am. J. Intellect. Dev. Disabil. 2013, 118, 310–326. [Google Scholar] [CrossRef] [PubMed]
- Gilkerson, J.; Richards, J. The LENA Natural Language Study (Technical Report LTR-02-2); LENA Foundation: Boulder, CO, USA, 2008; pp. 1–26. [Google Scholar]
- Xu, D.; Yapanel, U.; Gray, S. Reliability of the LENA™ language environment analysis system in young children’s natural home environment. LENA Tech. Rep. 2009, 5, 1–16. [Google Scholar]
- Oetting, J.B.; Hartfield, L.R.; Pruitt, S.L. Exploring LENA as a Tool for Researchers and Clinicians. ASHA Lead. 2009, 14, 20–22. [Google Scholar] [CrossRef]
- Gilkerson, J.; Richards, J.A. The LENA Developmental Snapshot; LENA Foundation: Boulder, CO, USA, 2008; pp. 1–7. [Google Scholar]
- Gilkerson, J.; Richards, J.A.; Greenwood, C.R.; Montgomery, J.K. Language assessment in a snap: Monitoring progress up to 36 months. Child Lang. Teach. Ther. 2017, 33, 99–115. [Google Scholar] [CrossRef]
- Finestack, L.H.; Richmond, E.K.; Abbeduto, L. Language Development in Individuals with Fragile X Syndrome. Top. Lang. Disord. 2009, 29, 133–148. [Google Scholar] [CrossRef]
- Siller, M.; Hutman, T.; Sigman, M. A parent-mediated intervention to increase responsive parental behaviors and child communication in children with ASD: A randomized clinical trial. J. Autism Dev. Disord. 2013, 43, 540–555. [Google Scholar] [CrossRef]
- Landry, S.H.; Smith, K.E.; Swank, P.R.; Guttentag, C. A Responsive Parenting Intervention: The Optimal Timing Across Early Childhood for Impacting Maternal Behaviors and Child Outcomes. Dev. Psychol. 2008, 44, 1335–1353. [Google Scholar] [CrossRef] [PubMed]
- Venker, C.E.; McDuffie, A.; Ellis Weismer, S.; Abbeduto, L. Increasing verbal responsiveness in parents of children with autism: A pilot study. Autism 2012, 16, 568–585. [Google Scholar] [CrossRef] [PubMed]
- McDuffie, A.; Oakes, A.; Machalicek, W.; Ma, M.; Bullard, L.; Nelson, S.; Abbeduto, L. Early language intervention using distance video-teleconferencing: A pilot study of young boys with fragile X syndrome and their mothers. Am. J. Speech Lang. Pathol. 2016, 25, 46–66. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reisinger, D.L.; Shaffer, R.C.; Pedapati, E.V.; Dominick, K.C.; Erickson, C.A. A Pilot Quantitative Evaluation of Early Life Language Development in Fragile X Syndrome. Brain Sci. 2019, 9, 27. https://doi.org/10.3390/brainsci9020027
Reisinger DL, Shaffer RC, Pedapati EV, Dominick KC, Erickson CA. A Pilot Quantitative Evaluation of Early Life Language Development in Fragile X Syndrome. Brain Sciences. 2019; 9(2):27. https://doi.org/10.3390/brainsci9020027
Chicago/Turabian StyleReisinger, Debra L., Rebecca C. Shaffer, Ernest V. Pedapati, Kelli C. Dominick, and Craig A. Erickson. 2019. "A Pilot Quantitative Evaluation of Early Life Language Development in Fragile X Syndrome" Brain Sciences 9, no. 2: 27. https://doi.org/10.3390/brainsci9020027
APA StyleReisinger, D. L., Shaffer, R. C., Pedapati, E. V., Dominick, K. C., & Erickson, C. A. (2019). A Pilot Quantitative Evaluation of Early Life Language Development in Fragile X Syndrome. Brain Sciences, 9(2), 27. https://doi.org/10.3390/brainsci9020027




