Paternal—but Not Maternal—Autistic Traits Predict Frontal EEG Alpha Asymmetry in Infants with Later Symptoms of Autism
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample
2.2. Frontal EEG Alpha Asymmetry
2.2.1. EEG Data Acquisition
2.2.2. EEG Data Processing and Analysis
2.2.3. Autistic Traits in Parents: The Autism Spectrum Quotient
2.2.4. Autistic Traits in Infants: The CBCL 1½–5 Pervasive Developmental Problems Scale
2.3. Statistical Analysis
3. Results
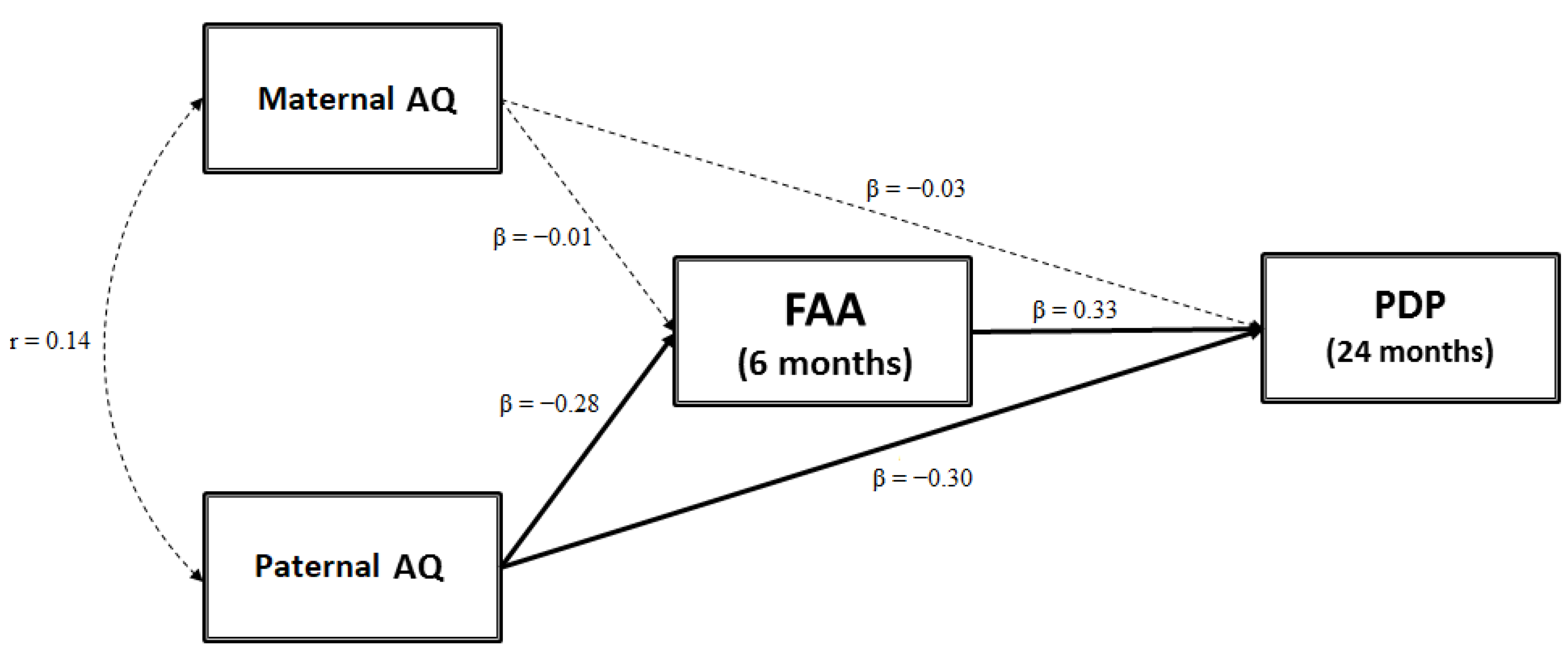
3.1. Testing a Mediation Model: FAA as a Mediator between Parental AQ Scores and Child ASD-Related Traits
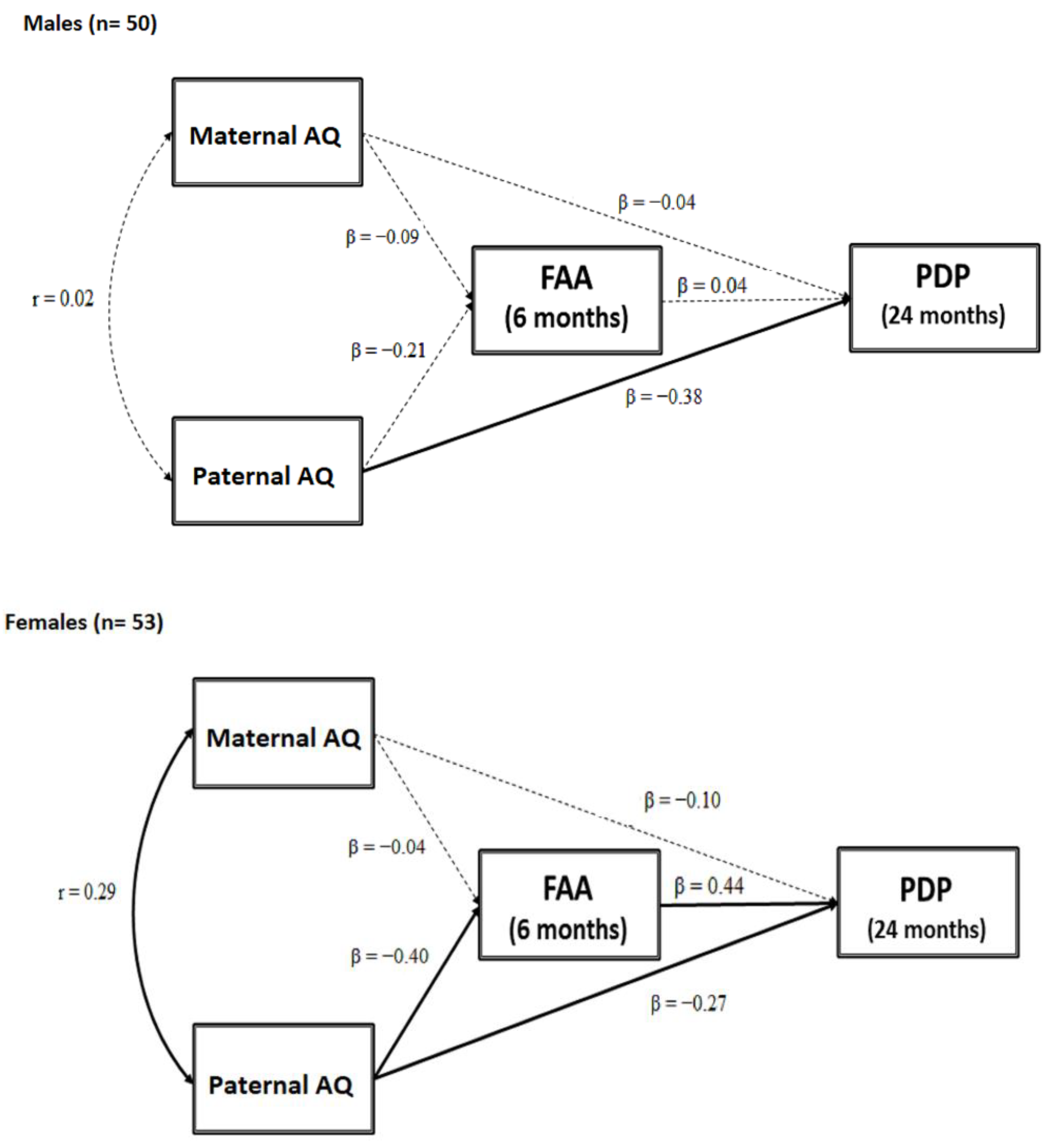
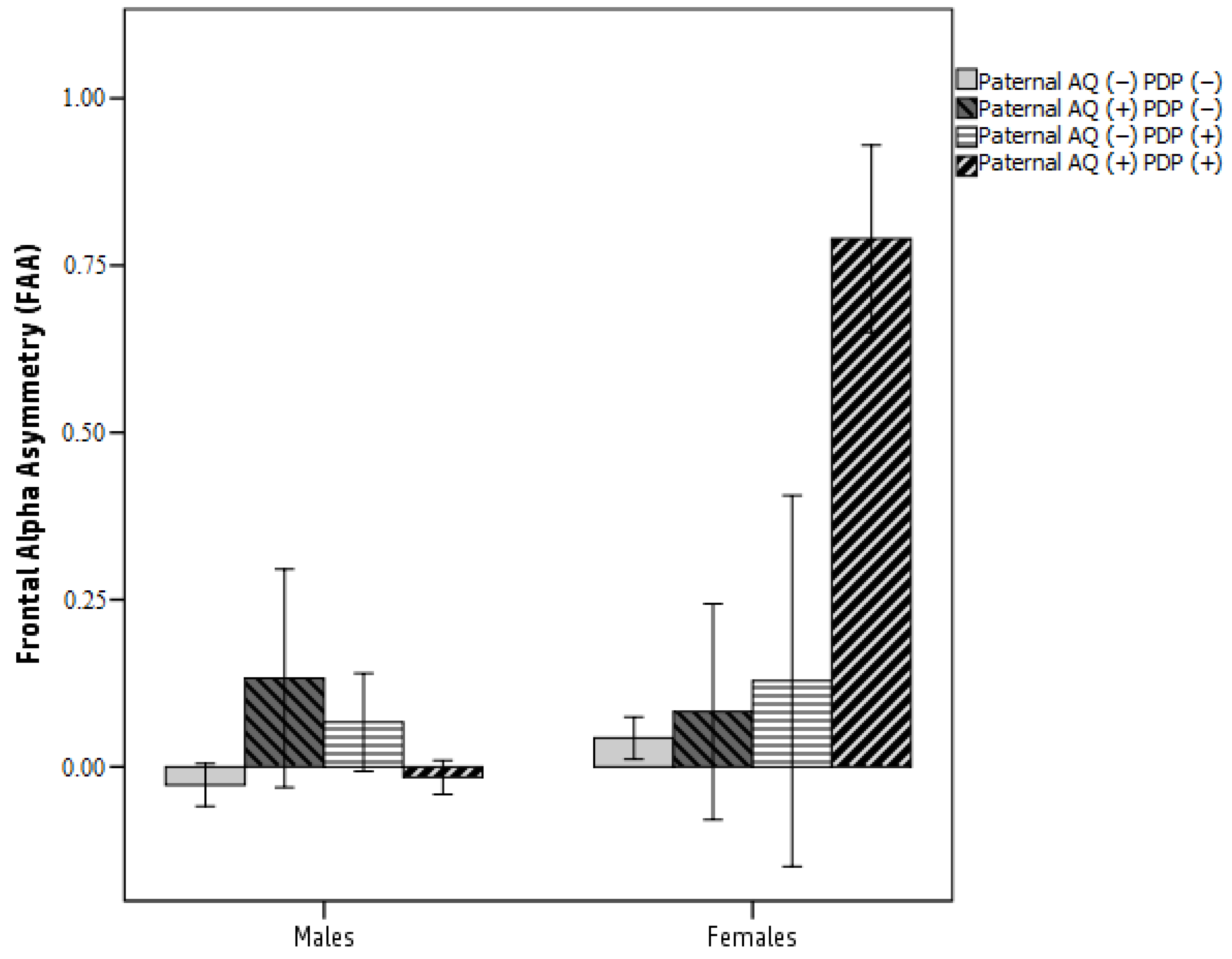
3.2. Testing a Moderated Mediation Model: FAA as a Mediator between Parental AQ and Child ASD-Related Traits Moderated by Sex
4. Discussion
4.1. Parental Autistic Traits and Child ASD Symptoms
4.2. Parental Autistic Traits and Frontal Asymmetry in Alpha Oscillation
4.3. Frontal Asymmetry in Alpha Oscillation and Child ASD Symptoms
4.4. Frontal Asymmetry in Alpha Oscillation as a Mediator between Paternal Autistic Traits and Child ASD Symptoms
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- American Psychiatric Association. Manuale Diagnostico e Statistico dei Disturbi Mentali-Quarta Edizione-Text Revision (DSM-IV-TR), 2000th ed.; American Psychiatric Association: Milano, Italy, 2002. [Google Scholar]
- Tick, B.; Bolton, P.; Happé, F.; Rutter, M.; Rijsdijk, F. Heritability of autism spectrum disorders: a meta-analysis of twin studies. J. Child Psychol. Psychiatry 2016, 57, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Levin-Decanini, T.; Maltman, N.; Francis, S.M.; Guter, S.; Anderson, G.M.; Cook, E.H.; Jacob, S. Parental Broader Autism Subphenotypes in ASD Affected Families: Relationship to Gender, Child’s Symptoms, SSRI Treatment, and Platelet Serotonin. Autism Res. 2013, 6, 621–630. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Losh, M.; Esserman, D.; Piven, J. Rapid automatized naming as an index of genetic liability to autism. J. Neurodev. Disord. 2010, 2, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Schwichtenberg, A.J.; Young, G.S.; Sigman, M.; Hutman, T.; Ozonoff, S. Can family affectedness inform infant sibling outcomes of autism spectrum disorders? J. Child Psychol. Psychiatry 2010, 51, 1021–1030. [Google Scholar] [CrossRef] [PubMed]
- Wilson, C.E.; Freeman, P.; Brock, J.; Burton, A.M.; Palermo, R. Facial Identity Recognition in the Broader Autism Phenotype. PLoS ONE 2010, 5, e12876. [Google Scholar] [CrossRef]
- Klusek, J.; Losh, M.; Martin, G.E. Sex differences and within-family associations in the broad autism phenotype. Autism 2014, 18, 106–116. [Google Scholar] [CrossRef] [PubMed]
- Billeci, L.; Calderoni, S.; Conti, E.; Gesi, C.; Carmassi, C.; Dell’Osso, L.; Cioni, G.; Muratori, F.; Guzzetta, A. The Broad Autism (Endo)Phenotype: Neurostructural and Neurofunctional Correlates in Parents of Individuals with Autism Spectrum Disorders. Front. Neurosci. 2016, 10, 346. [Google Scholar] [CrossRef] [PubMed]
- Ronconi, L.; Facoetti, A.; Bulf, H.; Franchin, L.; Bettoni, R.; Valenza, E. Paternal Autistic Traits are Predictive of Infants Visual Attention. J. Autism Dev. Disord. 2013. [Google Scholar] [CrossRef]
- Wang, J.; Barstein, J.; Ethridge, L.E.; Mosconi, M.W.; Takarae, Y.; Sweeney, J.A. Resting state EEG abnormalities in autism spectrum disorders. J. Neurodev. Disord. 2013, 5, 24. [Google Scholar] [CrossRef]
- Tierney, A.L.; Gabard-Durnam, L.; Vogel-Farley, V.; Tager-Flusberg, H.; Nelson, C.A. Developmental Trajectories of Resting EEG Power: An Endophenotype of Autism Spectrum Disorder. PLoS ONE 2012, 7, e39127. [Google Scholar] [CrossRef]
- Allen, J.B.; Kline, P. Frontal EEG asymmetry, emotion, and psychopathology: the first, and the next 25 years. Biol. Psychol. 2004, 67, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Burnette, C.P.; Henderson, H.A.; Inge, A.P.; Zahka, N.E.; Schwartz, C.B.; Mundy, P.C. Anterior EEG asymmetry and the Modifier Model of Autism. J. Autism Dev. Disord. 2011, 41, 1113–1124. [Google Scholar] [CrossRef] [PubMed]
- Sutton, S.K.; Burnette, C.P.; Mundy, P.C.; Meyer, J.; Vaughan, A.; Sanders, C.; Yale, M. Resting cortical brain activity and social behavior in higher functioning children with autism. J. Child Psychol. Psychiatry 2005, 46, 211–222. [Google Scholar] [CrossRef] [PubMed]
- Fox, N.A.; Calkins, S.D.; Bell, M.A. Neural plasticity and development in the first two years of life: Evidence from cognitive and socioemotional domains of research. Dev. Psychopathol. 1994, 6, 677. [Google Scholar] [CrossRef]
- Fox, N.A.; Henderson, H.A.; Rubin, K.H.; Calkins, S.D.; Schmidt, L.A. Continuity and discontinuity of behavioral inhibition and exuberance: psychophysiological and behavioral influences across the first four years of life. Child Dev. 2001, 72, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Gabard-Durnam, L.; Tierney, A.L.; Vogel-Farley, V.; Tager-Flusberg, H.; Nelson, C.A. Alpha Asymmetry in Infants at Risk for Autism Spectrum Disorders. J. Autism Dev. Disord. 2015, 45, 473–480. [Google Scholar] [CrossRef]
- Damiano-Goodwin, C.R.; Woynaroski, T.G.; Simon, D.M.; Ibañez, L.V.; Murias, M.; Kirby, A.; Newsom, C.R.; Wallace, M.T.; Stone, W.L.; Cascio, C.J. Developmental sequelae and neurophysiologic substrates of sensory seeking in infant siblings of children with autism spectrum disorder. Dev. Cogn. Neurosci. 2018, 29, 41–53. [Google Scholar] [CrossRef]
- Gartstein, M.A.; Bell, M.A.; Calkins, S.D. EEG asymmetry at 10 months of age: are temperament trait predictors different for boys and girls? Dev. Psychobiol. 2014, 56, 1327–1340. [Google Scholar] [CrossRef]
- Peltola, M.J.; Bakermans-Kranenburg, M.J.; Alink, L.R.A.; Huffmeijer, R.; Biro, S.; van IJzendoorn, M.H. Resting frontal EEG asymmetry in children: Meta-analyses of the effects of psychosocial risk factors and associations with internalizing and externalizing behavior. Dev. Psychobiol. 2014, 56, 1377–1389. [Google Scholar] [CrossRef]
- Riva, V.; Cantiani, C.; Benasich, A.A.; Molteni, M.; Piazza, C.; Giorda, R.; Dionne, G.; Marino, C. From CNTNAP2 to Early Expressive Language in Infancy: The Mediation Role of Rapid Auditory Processing. Cereb. Cortex 2017, 28, 2100–2108. [Google Scholar] [CrossRef]
- Riva, V.; Cantiani, C.; Mornati, G.; Gallo, M.; Villa, L.; Mani, E.; Saviozzi, I.; Marino, C.; Molteni, M. Distinct ERP profiles for auditory processing in infants at-risk for autism and language impairment. Sci. Rep. 2018, 8, 715. [Google Scholar] [CrossRef] [PubMed]
- Bayley, N. Bayley Scales of Infant and Toddler Development, 3rd ed.; Ferri, R., Orsini, A., Stoppa, E., Eds.; Giunti Psychometrics: Firenze, Italy, 2006. [Google Scholar]
- Hollingshead, A.B. Four factor index of social status. 1975; Unpublished paper. [Google Scholar]
- Baron-Cohen, S.; Wheelwright, S.; Skinner, R.; Martin, J.; Clubley, E. The autism–spectrum quotient (AQ): Evidence from asperger syndrome/high-functioning autism, males and females, scientists and mathematicians. J. Autism Dev. Disord. 2001, 31, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Achenbach, T.M.; Rescorla, L.A. Child Behavioural Checklist (CBCL) for Ages 1.5–5. In STOP, THAT and One Hundred Other Sleep Scales; Springer: New York, NY, USA, 2000; Traduzione italiana a cura di Frigerio A. [Google Scholar]
- Cantiani, C.; Riva, V.; Piazza, C.; Bettoni, R.; Molteni, M.; Choudhury, N.; Marino, C.; Benasich, A.A. Auditory discrimination predicts linguistic outcome in Italian infants with and without familial risk for language learning impairment. Dev. Cogn. Neurosci. 2016, 20, 23–34. [Google Scholar] [CrossRef]
- Delorme, A.; Makeig, S. EEGLAB: an open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J. Neurosci. Methods 2004, 134, 9–21. [Google Scholar] [CrossRef]
- Welch, P.D. The Use of Fast Fourier Transform for the Estimation of Power Spectra: A Method Based on Time Averaging Over Short, Modified Periodograms. IEEE Trans. Audio Electroacoust. 1967, 15, 70–73. [Google Scholar] [CrossRef]
- Marshall, P.J.; Bar-Haim, Y.; Fox, N.A. Development of the EEG from 5 months to 4 years of age. Clin. Neurophysiol. 2002, 113, 1199–1208. [Google Scholar] [CrossRef]
- Jensen, F.V.; Nielsen, T.D.; Shenoy, P.P. Sequential influence diagrams: A unified asymmetry framework. Int. J. Approx. Reason. 2006. [Google Scholar] [CrossRef][Green Version]
- Brooker, R.J.; Canen, M.J.; Davidson, R.J.; Hill Goldsmith, H. Short- and long-term stability of alpha asymmetry in infants: Baseline and affective measures. Psychophysiology 2017, 54, 1100–1109. [Google Scholar] [CrossRef]
- Ruta, L.; Mazzone, D.; Mazzone, L.; Wheelwright, S.; Baron-Cohen, S. The Autism-Spectrum Quotient—Italian Version: A Cross-Cultural Confirmation of the Broader Autism Phenotype. J. Autism Dev. Disord. 2012, 42, 625–633. [Google Scholar] [CrossRef]
- Ruzich, E.; Allison, C.; Smith, P.; Watson, P.; Auyeung, B.; Ring, H.; Baron-Cohen, S. Measuring autistic traits in the general population: A systematic review of the Autism-Spectrum Quotient (AQ) in a nonclinical population sample of 6900 typical adult males and females. Mol. Autism 2015, 6, 2. [Google Scholar] [CrossRef] [PubMed]
- Ivanova, M.Y.; Achenbach, T.M.; Rescorla, L.A.; Harder, V.S.; Ang, R.P.; Bilenberg, N.; Bjarnadottir, G.; Capron, C.; De Pauw, S.S.W.; Dias, P.; et al. Preschool psychopathology reported by parents in 23 societies: Testing the seven-syndrome model of the child behavior checklist for ages 1.5–5. J. Am. Acad. Child Adolesc. Psychiatry 2010, 49, 1215–1224. [Google Scholar] [CrossRef] [PubMed]
- Fritz, M.S.; Mackinnon, D.P. Required sample size to detect the mediated effect. Psychol. Sci. 2007, 18, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Muthén, L.K.; Muthén, B.O. Mplus User’s Guide, 7th ed.; Muthén & Muthén: Los Angeles, CA, USA, 2014. [Google Scholar]
- Iacobucci, D.; Saldanha, N.; Deng, X. A Meditation on Mediation: Evidence That Structural Equations Models Perform Better Than Regressions. J. Consum. Psychol. 2007, 17, 139–153. [Google Scholar] [CrossRef]
- Pedhazur, E.J. Multiple Regression in Behavioral Research: Explanation and Prediction, 3rd ed.; Wadsworth Thomson Learning: Belmont, CA, USA, 1982. [Google Scholar]
- Mueller, R.; Hancock, G.R. Best Practices in Structural Equation Modeling. In Best Practices in Quantitative Methods; SAGE Publications, Inc.: Thousand Oaks, CA, USA, 2008. [Google Scholar] [CrossRef]
- Mackinnon, D.P.; Lockwood, C.M.; Williams, J. Confidence Limits for the Indirect Effect: Distribution of the Product and Resampling Methods. Multivar. Behav. Res. 2004, 39, 99. [Google Scholar] [CrossRef]
- Kline, R. Exploratory and Confirmatory Factor Analysis; Routledge: Abingdon-on-Thames, UK, 2013. [Google Scholar]
- Browne, M.W.; Cudeck, R. Alternative ways of assessing model fit. Sage Focus Ed. 1993, 154, 136. [Google Scholar] [CrossRef]
- Hu, L.; Bentler, P.M. Cutoff criteria for fit indexes in covariance structure analysis: Conventional criteria versus new alternatives. Struct. Equ. Model. Multidiscip. J. 1999, 6, 1–55. [Google Scholar] [CrossRef]
- Whitley, B.E.; Kite, M.E.; Adams, H.L. Principles of Research in Behavioral Science; Psychology Press: Hove, UK, 2013; ISBN 0415879280. [Google Scholar]
- Robinson, E.B.; Koenen, K.C.; McCormick, M.C.; Munir, K.; Hallett, V.; Happé, F.; Plomin, R.; Ronald, A. Evidence That Autistic Traits Show the Same Etiology in the General Population and at the Quantitative Extremes (5%, 2.5%, and 1%). Arch. Gen. Psychiatry 2011, 68, 1113. [Google Scholar] [CrossRef]
- Simon, D.M.; Wallace, M.T. Dysfunction of sensory oscillations in Autism Spectrum Disorder. Neurosci. Biobehav. Rev. 2016, 68, 848–861. [Google Scholar] [CrossRef]
- Coan, J.A.; Allen, J.J.B. Frontal EEG asymmetry as a moderator and mediator of emotion. Biol. Psychol. 2004, 67, 7–49. [Google Scholar] [CrossRef]
- Flashner, B.M.; Russo, M.E.; Boileau, J.E.; Leong, D.W.; Gallicano, G.I. Epigenetic Factors and Autism Spectrum Disorders. NeuroMol. Med. 2013, 15, 339–350. [Google Scholar] [CrossRef] [PubMed]
- Gerdts, J.; Bernier, R. The Broader Autism Phenotype and Its Implications on the Etiology and Treatment of Autism Spectrum Disorders. Autism Res. Treat. 2011, 2011, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Fombonne, E. Epidemiology of Pervasive Developmental Disorders. Pediatr. Res. 2009, 65, 591–598. [Google Scholar] [CrossRef]
- Volf, N.V.; Belousova, L.V.; Knyazev, G.G.; Kulikov, A.V. Gender differences in association between serotonin transporter gene polymorphism and resting-state EEG activity. Neuroscience 2015, 284, 513–521. [Google Scholar] [CrossRef] [PubMed]
- Frenkel, T.I.; Koss, K.J.; Donzella, B.; Frenn, K.A.; Lamm, C.; Fox, N.A.; Gunnar, M.R. ADHD Symptoms in Post-Institutionalized Children Are Partially Mediated by Altered Frontal EEG Asymmetry. J. Abnorm. Child Psychol. 2017, 45, 857–869. [Google Scholar] [CrossRef] [PubMed]
- Möricke, E.; Buitelaar, J.K.; Rommelse, N.N.J. Do We Need Multiple Informants When Assessing Autistic Traits? The Degree of Report Bias on Offspring, Self, and Spouse Ratings. J. Autism Dev. Disord. 2016, 46, 164–175. [Google Scholar] [CrossRef] [PubMed]
- Cantiani, C.; Piazza, C.; Mornati, G.; Molteni, M.; Riva, V. Oscillatory gamma activity mediates the pathway from socioeconomic status to language acquisition in infancy. Infant Behav. Dev. 2019, 57. [Google Scholar] [CrossRef] [PubMed]
| Total Sample (n = 103) | Males (n = 50) | Females (n = 53) | |
|---|---|---|---|
| Mean (±SD) | Mean (±SD) | Mean (±SD) | |
| Birthweight (grams) | 3252.20 (±468.08) | 3325.33 (±481.35) | 3179.07 (±453.03) |
| Gestational age (weeks) | 39.08 (±1.47) | 39.67 (±1.27) | 38.49 (±1.66) |
| Maternal educational level a | 58.04 (±17.05) | 56.50 (±19.39) | 59.52 (±14.49) |
| Paternal educational level a | 49.31 (±17.68) | 50.41 (±17.67) | 48.27 (±17.79) |
| Socioeconomic status b | 61.47 (±15.51) | 61.10 (±15.63) | 61.83 (±15.53) |
| Bayley cognitive subscale at 6 months c | 12.07 (±1.81) | 11.82 (±1.96) | 12.30 (±1.65) |
| Paternal AQ (raw scores) | 17.81 (±6.22) | 19.20 (±6.49) | 16.44 (±5.68) |
| Maternal AQ (raw scores) | 14.49 (±5.77) | 15.28 (±5.95) | 13.73 (±5.53) |
| Paternal AQ d | −0.26 (±1.37) | −0.53 (±1.40) | 0.003 (±1.22) |
| Maternal AQ d | 0.03 (±1.09) | −0.03 (±1.16) | 0.10 (±1.02) |
| CBCL 1½-5 Pervasive Developmental Problems e | 53.42 (±5.94) | 52.84 (±4.88) | 53.98 (±6.81) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Riva, V.; Marino, C.; Piazza, C.; Riboldi, E.M.; Mornati, G.; Molteni, M.; Cantiani, C. Paternal—but Not Maternal—Autistic Traits Predict Frontal EEG Alpha Asymmetry in Infants with Later Symptoms of Autism. Brain Sci. 2019, 9, 342. https://doi.org/10.3390/brainsci9120342
Riva V, Marino C, Piazza C, Riboldi EM, Mornati G, Molteni M, Cantiani C. Paternal—but Not Maternal—Autistic Traits Predict Frontal EEG Alpha Asymmetry in Infants with Later Symptoms of Autism. Brain Sciences. 2019; 9(12):342. https://doi.org/10.3390/brainsci9120342
Chicago/Turabian StyleRiva, Valentina, Cecilia Marino, Caterina Piazza, Elena M Riboldi, Giulia Mornati, Massimo Molteni, and Chiara Cantiani. 2019. "Paternal—but Not Maternal—Autistic Traits Predict Frontal EEG Alpha Asymmetry in Infants with Later Symptoms of Autism" Brain Sciences 9, no. 12: 342. https://doi.org/10.3390/brainsci9120342
APA StyleRiva, V., Marino, C., Piazza, C., Riboldi, E. M., Mornati, G., Molteni, M., & Cantiani, C. (2019). Paternal—but Not Maternal—Autistic Traits Predict Frontal EEG Alpha Asymmetry in Infants with Later Symptoms of Autism. Brain Sciences, 9(12), 342. https://doi.org/10.3390/brainsci9120342





