Perinatal MAO Inhibition Produces Long-Lasting Impairment of Serotonin Function in Offspring
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Behavioral Assessment
2.3. Brain Preparation
2.4. Autoradiography
2.5. Image Analysis
2.6. Data Analysis
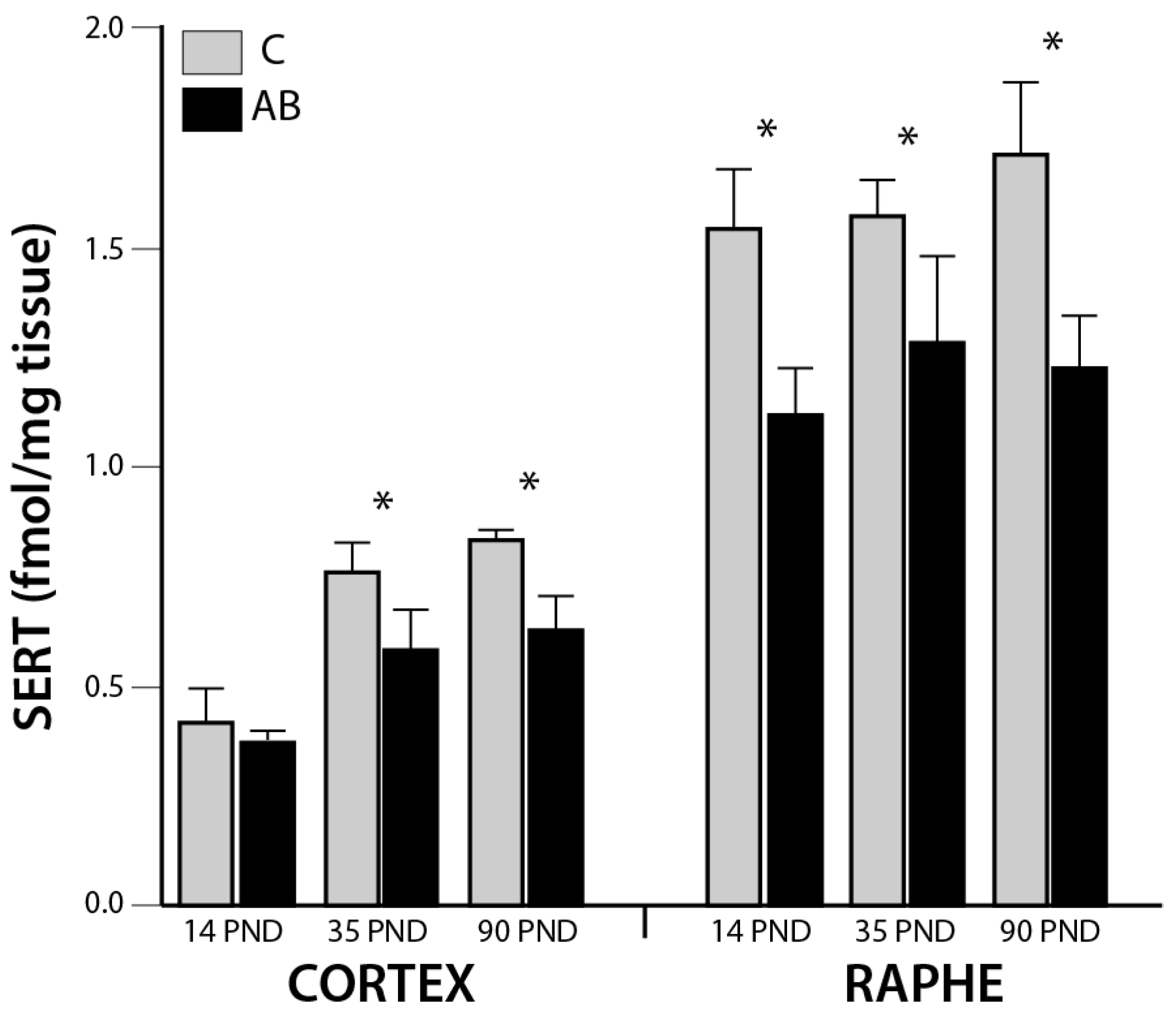
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Brown, G.L.; Goodwin, F.K.; Bunney, W.E., Jr. Human aggression and suicide: Their relationship to neuropsychiatric diagnoses and serotonin metabolism. Adv. Biochem. Psychopharmacol. 1982, 34, 287–307. [Google Scholar] [CrossRef] [PubMed]
- Brown, G.L.; Linnoila, M.I. CSF serotonin metabolite (5-HIAA) studies in depression, impulsivity, and violence. J. Clin. Psychiatry 1990, 51, 31–41. [Google Scholar] [PubMed]
- Kruesi, M.J.; Rapoport, J.L.; Hamburger, S.; Hibbs, E.; Potter, W.Z.; Lenane, M.; Brown, G.L. Cerebrospinal fluid monoamine metabolites, aggression, and impulsivity in disruptive behavior disorders of children and adolescents. Arch. Gen. Psychiatry 1990, 47, 419–426. [Google Scholar] [CrossRef] [PubMed]
- Soderstrom, H.; Blennow, K.; Manhem, A.; Forsman, A. CSF studies in violent offenders. I. 5-HIAA as a negative and hva as a positive predictor of psychopathy. J. Neural Transm. (Vienna) 2001, 108, 869–878. [Google Scholar] [CrossRef] [PubMed]
- Popova, N.K. From gene to aggressive behavior: The role of brain serotonin. Neurosci. Behav. Physiol. 2008, 38, 471–475. [Google Scholar] [CrossRef] [PubMed]
- Yu, Q.; Teixeira, C.M.; Mahadevia, D.; Huang, Y.; Balsam, D.; Mann, J.J.; Gingrich, J.A.; Ansorge, M.S. Dopamine and serotonin signaling during two sensitive developmental periods differentially impact adult aggressive and affective behaviors in mice. Mol. Psychiatry 2014, 19, 688–698. [Google Scholar] [CrossRef] [PubMed]
- Kolla, N.J.; Matthews, B.; Wilson, A.A.; Houle, S.; Bagby, R.M.; Links, P.; Simpson, A.I.; Hussain, A.; Meyer, J.H. Lower monoamine oxidase-a total distribution volume in impulsive and violent male offenders with antisocial personality disorder and high psychopathic traits: An [(11)c] harmine positron emission tomography study. Neuropsychopharmacology 2015, 40, 2596–2603. [Google Scholar] [CrossRef] [PubMed]
- Chester, D.S.; DeWall, C.N.; Derefinko, K.J.; Estus, S.; Peters, J.R.; Lynam, D.R.; Jiang, Y. Monoamine oxidase A (MAOA) genotype predicts greater aggression through impulsive reactivity to negative affect. Behav. Brain Res. 2015, 283, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Brunner, H.G.; Nelen, M.; Breakefield, X.O.; Ropers, H.H.; van Oost, B.A. Abnormal behavior associated with a point mutation in the structural gene for monoamine oxidase A. Science 1993, 262, 578–580. [Google Scholar] [CrossRef] [PubMed]
- Brunner, H.G.; Nelen, M.R.; van Zandvoort, P.; Abeling, N.G.; van Gennip, A.H.; Wolters, E.C.; Kuiper, M.A.; Ropers, H.H.; van Oost, B.A. X-linked borderline mental retardation with prominent behavioral disturbance: Phenotype, genetic localization, and evidence for disturbed monoamine metabolism. Am. J. Hum. Genet. 1993, 52, 1032–1039. [Google Scholar] [PubMed]
- Wang, C.C.; Billett, E.; Borchert, A.; Kuhn, H.; Ufer, C. Monoamine oxidases in development. Cell. Mol. Life Sci. 2013, 70, 599–630. [Google Scholar] [CrossRef] [PubMed]
- Finberg, J.P.; Rabey, J.M. Inhibitors of MAO-A and MAO-B in psychiatry and neurology. Front. Pharmacol. 2016, 7, 340. [Google Scholar] [CrossRef] [PubMed]
- Palmer, E.E.; Leffler, M.; Rogers, C.; Shaw, M.; Carroll, R.; Earl, J.; Cheung, N.W.; Champion, B.; Hu, H.; Haas, S.A.; et al. New insights into brunner syndrome and potential for targeted therapy. Clin. Genet. 2016, 89, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Cases, O.; Seif, I.; Grimsby, J.; Gaspar, P.; Chen, K.; Pournin, S.; Muller, U.; Aguet, M.; Babinet, C.; Shih, J.C.; et al. Aggressive behavior and altered amounts of brain serotonin and norepinephrine in mice lacking maoa. Science 1995, 268, 1763–1766. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Holschneider, D.P.; Wu, W.; Rebrin, I.; Shih, J.C. A spontaneous point mutation produces monoamine oxidase a/b knock-out mice with greatly elevated monoamines and anxiety-like behavior. J. Biol. Chem. 2004, 279, 39645–39652. [Google Scholar] [CrossRef] [PubMed]
- Bortolato, M.; Godar, S.C.; Alzghoul, L.; Zhang, J.; Darling, R.D.; Simpson, K.L.; Bini, V.; Chen, K.; Wellman, C.L.; Lin, R.C.; et al. Monoamine oxidase A and A/B knockout mice display autistic-like features. Int. J. Neuropsychopharmacol. 2013, 16, 869–888. [Google Scholar] [CrossRef] [PubMed]
- Owesson, C.A.; Hopwood, S.E.; Callado, L.F.; Seif, I.; McLaughlin, D.P.; Stamford, J.A. Altered presynaptic function in monoaminergic neurons of monoamine oxidase-a knockout mice. Eur. J. Neurosci. 2002, 15, 1516–1522. [Google Scholar] [CrossRef] [PubMed]
- Singh, C.; Bortolato, M.; Bali, N.; Godar, S.C.; Scott, A.L.; Chen, K.; Thompson, R.F.; Shih, J.C. Cognitive abnormalities and hippocampal alterations in monoamine oxidase a and b knockout mice. Proc. Natl. Acad. Sci. USA 2013, 110, 12816–12821. [Google Scholar] [CrossRef] [PubMed]
- Vishnivetskaya, G.B.; Skrinskaya, J.A.; Seif, I.; Popova, N.K. Effect of mao a deficiency on different kinds of aggression and social investigation in mice. Aggress. Behav. 2007, 33, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Godar, S.C.; Bortolato, M.; Frau, R.; Dousti, M.; Chen, K.; Shih, J.C. Maladaptive defensive behaviours in monoamine oxidase A-deficient mice. Int. J. Neuropsychopharmacol. 2011, 14, 1195–1207. [Google Scholar] [CrossRef] [PubMed]
- Holschneider, D.P.; Scremin, O.U.; Huynh, L.; Chen, K.; Seif, I.; Shih, J.C. Regional cerebral cortical activation in monoamine oxidase A-deficient mice: Differential effects of chronic versus acute elevations in serotonin and norepinephrine. Neuroscience 2000, 101, 869–877. [Google Scholar] [CrossRef]
- Chen, K.; Kardys, A.; Chen, Y.; Flink, S.; Tabakoff, B.; Shih, J.C. Altered gene expression in early postnatal monoamine oxidase A knockout mice. Brain Res. 2017, 1669, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Grimsby, J.; Toth, M.; Chen, K.; Kumazawa, T.; Klaidman, L.; Adams, J.D.; Karoum, F.; Gal, J.; Shih, J.C. Increased stress response and beta-phenylethylamine in MAOB-deficient mice. Nat. Genet. 1997, 17, 206–210. [Google Scholar] [CrossRef] [PubMed]
- Bortolato, M.; Godar, S.C.; Tambaro, S.; Li, F.G.; Devoto, P.; Coba, M.P.; Chen, K.; Shih, J.C. Early postnatal inhibition of serotonin synthesis results in long-term reductions of perseverative behaviors, but not aggression, in mao a-deficient mice. Neuropharmacology 2013, 75, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Whitaker-Azmitia, P.M.; Zhang, X.; Clarke, C. Effects of gestational exposure to monoamine oxidase inhibitors in rats: Preliminary behavioral and neurochemical studies. Neuropsychopharmacology 1994, 11, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Mejia, J.M.; Ervin, F.R.; Baker, G.B.; Palmour, R.M. Monoamine oxidase inhibition during brain development induces pathological aggressive behavior in mice. Biol. Psychiatry 2002, 52, 811–821. [Google Scholar] [CrossRef]
- Kaplan, H.; Sadock, B. Kaplan and Sadock’s Synopsis of Psychiatry; Williams and Williams: Baltimore, MD, USA, 1998; Volume 8. [Google Scholar]
- Brunner, H.G. Maoa deficiency and abnormal behaviour: Perspectives on an association. Ciba Found. Symp. 1996, 194, 155–164. [Google Scholar] [PubMed]
- Gingrich, J.A.; Malm, H.; Ansorge, M.S.; Brown, A.; Sourander, A.; Suri, D.; Teixeira, C.M.; Caffrey Cagliostro, M.K.; Mahadevia, D.; Weissman, M.M. New insights into how serotonin selective reuptake inhibitors shape the developing brain. Birth Defects Res. 2017, 109, 924–932. [Google Scholar] [CrossRef] [PubMed]
- Weyler, W.; Hsu, Y.P.; Breakefield, X.O. Biochemistry and genetics of monoamine oxidase. Pharmacol. Ther. 1990, 47, 391–417. [Google Scholar] [CrossRef]
- Kepser, L.J.; Homberg, J.R. The neurodevelopmental effects of serotonin: A behavioural perspective. Behav. Brain Res. 2015, 277, 3–13. [Google Scholar] [CrossRef] [PubMed]
- McLearn, G.; Wilson, J.; Meredith, M. The Use of Isogenic and Heterogenic Mouse Stocks in Behavioural Research; Linzey, G., Thiessen, D.D., Eds.; Contributions to Behaviour Genetic Analysis: The Mouse Prototype; Appleton Century Crofts, Educational Division, Meredith Corporation: New York, NY, USA, 1970; pp. 11–12. [Google Scholar]
- Coulter, C.L.; Happe, H.K.; Bergman, D.A.; Murrin, L.C. Localization and quantification of the dopamine transporter: Comparison of [3H]WIN 35,428 and [125I]RTI-55. Brain Res. 1995, 690, 217–224. [Google Scholar] [CrossRef]
- Pradhan, A.A.; Cumming, P.; Clarke, P.B. [125I]Epibatidine-labelled nicotinic receptors in the extended striatum and cerebral cortex: Lack of association with serotonergic afferents. Brain Res. 2002, 954, 227–236. [Google Scholar] [CrossRef]
- Rothman, R.B.; Silverthorn, M.L.; Glowa, J.R.; Matecka, D.; Rice, K.C.; Carroll, F.I.; Partilla, J.S.; Uhl, G.R.; Vandenbergh, D.J.; Dersch, C.M. Studies of the biogenic amine transporters. VII. Characterization of a novel cocaine binding site identified with [125I]RTI-55 in membranes prepared from human, monkey and guinea pig caudate. Synapse 1998, 28, 322–338. [Google Scholar] [CrossRef]
- Coulter, C.L.; Happe, H.K.; Murrin, L.C. Dopamine transporter development in postnatal rat striatum: An autoradiographic study with [3H]WIN 35,428. Brain Res. Dev. Brain Res. 1997, 104, 55–62. [Google Scholar] [CrossRef]
- Galineau, L.; Kodas, E.; Guilloteau, D.; Vilar, M.P.; Chalon, S. Ontogeny of the dopamine and serotonin transporters in the rat brain: An autoradiographic study. Neurosci. Lett. 2004, 363, 266–271. [Google Scholar] [CrossRef] [PubMed]
- Tarazi, F.I.; Tomasini, E.C.; Baldessarini, R.J. Postnatal development of dopamine and serotonin transporters in rat caudate-putamen and nucleus accumbens septi. Neurosci. Lett. 1998, 254, 21–24. [Google Scholar] [CrossRef]
- Zhou, F.C.; Sari, Y.; Zhang, J.K. Expression of serotonin transporter protein in developing rat brain. Brain Res. Dev. Brain Res. 2000, 119, 33–45. [Google Scholar] [CrossRef]
- Bortolato, M.; Chen, K.; Shih, J.C. Monoamine oxidase inactivation: From pathophysiology to therapeutics. Adv. Drug Deliv. Rev. 2008, 60, 1527–1533. [Google Scholar] [CrossRef] [PubMed]
- Sallinen, V.; Sundvik, M.; Reenila, I.; Peitsaro, N.; Khrustalyov, D.; Anichtchik, O.; Toleikyte, G.; Kaslin, J.; Panula, P. Hyperserotonergic phenotype after monoamine oxidase inhibition in larval zebrafish. J. Neurochem. 2009, 109, 403–415. [Google Scholar] [CrossRef] [PubMed]
- Berger-Sweeney, J.; Hohmann, C.F. Behavioral consequences of abnormal cortical development: Insights into developmental disabilities. Behav. Brain Res. 1997, 86, 121–142. [Google Scholar] [CrossRef]
- Brown, A.S.; Gyllenberg, D.; Malm, H.; McKeague, I.W.; Hinkka-Yli-Salomaki, S.; Artama, M.; Gissler, M.; Cheslack-Postava, K.; Weissman, M.M.; Gingrich, J.A.; et al. Association of selective serotonin reuptake inhibitor exposure during pregnancy with speech, scholastic, and motor disorders in offspring. JAMA Psychiatry 2016, 73, 1163–1170. [Google Scholar] [CrossRef] [PubMed]
- Malm, H.; Brown, A.S.; Gissler, M.; Gyllenberg, D.; Hinkka-Yli-Salomaki, S.; McKeague, I.W.; Weissman, M.; Wickramaratne, P.; Artama, M.; Gingrich, J.A.; et al. Gestational exposure to selective serotonin reuptake inhibitors and offspring psychiatric disorders: A national register-based study. J. Am. Acad. Child Adolesc. Psychiatry 2016, 55, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Andalib, S.; Emamhadi, M.R.; Yousefzadeh-Chabok, S.; Shakouri, S.K.; Hoilund-Carlsen, P.F.; Vafaee, M.S.; Michel, T.M. Maternal ssri exposure increases the risk of autistic offspring: A meta-analysis and systematic review. Eur. Psychiatry 2017, 45, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, Y.C.; Keskin-Arslan, E.; Acar, S. Prenatal antidepressant use and risk of autism spectrum disorders in children. JAMA Pediatr. 2016, 170, 712. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, Y.C.; Keskin-Arslan, E.; Acar, S.; Sozmen, K. Prenatal selective serotonin reuptake inhibitor use and the risk of autism spectrum disorder in children: A systematic review and meta-analysis. Reprod. Toxicol. 2016, 66, 31–43. [Google Scholar] [CrossRef] [PubMed]
- Buznikov, G.A.; Lambert, H.W.; Lauder, J.M. Serotonin and serotonin-like substances as regulators of early embryogenesis and morphogenesis. Cell Tissue Res. 2001, 305, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Lauder, J.M.; Wallace, J.A.; Krebs, H.; Petrusz, P.; McCarthy, K. In vivo and in vitro development of serotonergic neurons. Brain Res. Bull. 1982, 9, 605–625. [Google Scholar] [CrossRef]
- Pendleton, R.G.; Rasheed, A.; Roychowdhury, R.; Hillman, R. A new role for catecholamines: Ontogenesis. Trends Pharmacol. Sci. 1998, 19, 248–251. [Google Scholar] [PubMed]
- Ansorge, M.S.; Morelli, E.; Gingrich, J.A. Inhibition of serotonin but not norepinephrine transport during development produces delayed, persistent perturbations of emotional behaviors in mice. J. Neurosci. 2008, 28, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Bonnin, A.; Torii, M.; Wang, L.; Rakic, P.; Levitt, P. Serotonin modulates the response of embryonic thalamocortical axons to netrin-1. Nat. Neurosci. 2007, 10, 588–597. [Google Scholar] [CrossRef] [PubMed]
- Bonnin, A.; Goeden, N.; Chen, K.; Wilson, M.L.; King, J.; Shih, J.C.; Blakely, R.D.; Deneris, E.S.; Levitt, P. A transient placental source of serotonin for the fetal forebrain. Nature 2011, 472, 347–350. [Google Scholar] [CrossRef] [PubMed]
- Whitaker-Azmitia, P.M. Serotonin and brain development: Role in human developmental diseases. Brain Res. Bull. 2001, 56, 479–485. [Google Scholar] [CrossRef]
- Eisenhofer, G.; Finberg, J.P. Different metabolism of norepinephrine and epinephrine by catechol-o-methyltransferase and monoamine oxidase in rats. J. Pharmacol. Exp. Ther. 1994, 268, 1242–1251. [Google Scholar] [PubMed]
- Eisenhofer, G.; Lenders, J.W.; Harvey-White, J.; Ernst, M.; Zametkin, A.; Murphy, D.L.; Kopin, I.J. Differential inhibition of neuronal and extraneuronal monoamine oxidase. Neuropsychopharmacology 1996, 15, 296–301. [Google Scholar] [CrossRef]
- Wayment, H.K.; Schenk, J.O.; Sorg, B.A. Characterization of extracellular dopamine clearance in the medial prefrontal cortex: Role of monoamine uptake and monoamine oxidase inhibition. J. Neurosci. 2001, 21, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Whitaker-Azmitia, P.M. Behavioral and cellular consequences of increasing serotonergic activity during brain development: A role in autism? Int. J. Dev. Neurosci. 2005, 23, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Whitaker-Azmitia, P.M.; Shemer, A.V.; Caruso, J.; Molino, L.; Azmitia, E.C. Role of high affinity serotonin receptors in neuronal growth. Ann. N. Y. Acad. Sci. 1990, 600, 315–330. [Google Scholar] [CrossRef] [PubMed]
- Ahlemeyer, B.; Beier, H.; Semkova, I.; Schaper, C.; Krieglstein, J. S-100beta protects cultured neurons against glutamate- and staurosporine-induced damage and is involved in the antiapoptotic action of the 5 HT(1A)-receptor agonist, bay x 3702. Brain Res. 2000, 858, 121–128. [Google Scholar] [CrossRef]
- Azmitia, E.C.; Dolan, K.; Whitaker-Azmitia, P.M. S-100b but not ngf, egf, insulin or calmodulin is a cns serotonergic growth factor. Brain Res. 1990, 516, 354–356. [Google Scholar] [CrossRef]
- Liu, J.P.; Lauder, J.M. S-100 beta and insulin-like growth factor-ii differentially regulate growth of developing serotonin and dopamine neurons in vitro. J. Neurosci. Res. 1992, 33, 248–256. [Google Scholar] [CrossRef] [PubMed]
- Chubakov, A.R.; Gromova, E.A.; Konovalov, G.V.; Chumasov, E.I.; Sarkisova, E.F. Effect of serotonin on the development of a rat cerebral cortex tissue culture. Neurosci. Behav. Physiol. 1986, 16, 490–497. [Google Scholar] [CrossRef] [PubMed]
- Whitaker-Azmitia, P.M.; Lauder, J.M.; Shemmer, A.; Azmitia, E.C. Postnatal changes in serotonin receptors following prenatal alterations in serotonin levels: Further evidence for functional fetal serotonin receptors. Brain Res. 1987, 430, 285–289. [Google Scholar] [CrossRef]
- Lauder, J.M.; Liu, J.; Grayson, D.R. In utero exposure to serotonergic drugs alters neonatal expression of 5-HT(1A) receptor transcripts: A quantitative RT-PCR study. Int. J. Dev. Neurosci. 2000, 18, 171–176. [Google Scholar] [CrossRef]
- Whitaker-Azmitia, P.M.; Azmitia, E.C. Autoregulation of fetal serotonergic neuronal development: Role of high affinity serotonin receptors. Neurosci. Lett. 1986, 67, 307–312. [Google Scholar] [CrossRef]
- Shemer, A.V.; Azmitia, E.C.; Whitaker-Azmitia, P.M. Dose-related effects of prenatal 5-methoxytryptamine (5-mt) on development of serotonin terminal density and behavior. Brain Res. Dev. Brain Res. 1991, 59, 59–63. [Google Scholar] [CrossRef]
- Sikich, L.; Hickok, J.M.; Todd, R.D. 5-HT1A receptors control neurite branching during development. Brain Res. Dev. Brain Res. 1990, 56, 269–274. [Google Scholar] [CrossRef]
- Azmitia, E.C. Modern views on an ancient chemical: Serotonin effects on cell proliferation, maturation, and apoptosis. Brain Res. Bull. 2001, 56, 413–424. [Google Scholar] [CrossRef]
- Andrade, S.E.; Raebel, M.A.; Brown, J.; Lane, K.; Livingston, J.; Boudreau, D.; Rolnick, S.J.; Roblin, D.; Smith, D.H.; Willy, M.E.; et al. Use of antidepressant medications during pregnancy: A multisite study. Am. J. Obstet. Gynecol. 2008, 198, 194.e1–194.e5. [Google Scholar] [CrossRef] [PubMed]
- Bakker, M.K.; Kolling, P.; van den Berg, P.B.; de Walle, H.E.; de Jong van den Berg, L.T. Increase in use of selective serotonin reuptake inhibitors in pregnancy during the last decade, a population-based cohort study from the netherlands. Br. J. Clin. Pharmacol. 2008, 65, 600–606. [Google Scholar] [CrossRef] [PubMed]
- Hviid, A.; Melbye, M.; Pasternak, B. Use of selective serotonin reuptake inhibitors during pregnancy and risk of autism. N. Engl. J. Med. 2013, 369, 2406–2415. [Google Scholar] [CrossRef] [PubMed]
- Tran, H.; Robb, A.S. Ssri use during pregnancy. Semin. Perinatol. 2015, 39, 545–547. [Google Scholar] [CrossRef] [PubMed]
- Urato, A.C. Are the ssri antidepressants safe in pregnancy? Understanding the debate. Int. J. Risk Saf. Med. 2015, 27, 93–99. [Google Scholar] [PubMed]
- Handal, M.; Skurtveit, S.; Furu, K.; Hernandez-Diaz, S.; Skovlund, E.; Nystad, W.; Selmer, R. Motor development in children prenatally exposed to selective serotonin reuptake inhibitors: A large population-based pregnancy cohort study. BJOG 2016, 123, 1908–1917. [Google Scholar] [CrossRef] [PubMed]
- Hannerfors, A.K.; Hellgren, C.; Schijven, D.; Iliadis, S.I.; Comasco, E.; Skalkidou, A.; Olivier, J.D.; Sundstrom-Poromaa, I. Treatment with serotonin reuptake inhibitors during pregnancy is associated with elevated corticotropin-releasing hormone levels. Psychoneuroendocrinology 2015, 58, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Man, K.K.; Tong, H.H.; Wong, L.Y.; Chan, E.W.; Simonoff, E.; Wong, I.C. Exposure to selective serotonin reuptake inhibitors during pregnancy and risk of autism spectrum disorder in children: A systematic review and meta-analysis of observational studies. Neurosci. Biobehav. Rev. 2015, 49, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Gentile, S. Prenatal antidepressant exposure and the risk of autism spectrum disorders in children. Are we looking at the fall of gods? J. Affect. Disord. 2015, 182, 132–137. [Google Scholar] [CrossRef] [PubMed]
- El Marroun, H.; White, T.J.; van der Knaap, N.J.; Homberg, J.R.; Fernandez, G.; Schoemaker, N.K.; Jaddoe, V.W.; Hofman, A.; Verhulst, F.C.; Hudziak, J.J.; et al. Prenatal exposure to selective serotonin reuptake inhibitors and social responsiveness symptoms of autism: Population-based study of young children. Br. J. Psychiatry 2014, 205, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Harrington, R.A.; Lee, L.C.; Crum, R.M.; Zimmerman, A.W.; Hertz-Picciotto, I. Prenatal ssri use and offspring with autism spectrum disorder or developmental delay. Pediatrics 2014, 133, e1241–e1248. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Guan, X.W.; Ren, J.Q.; He, W. Developmental toxicity of cocaine exposure in mid-pregnancy mice. Acta Pharmacol. Sin. 2002, 23, 1029–1034. [Google Scholar] [PubMed]
- Smith, A.D.; Weiss, F. Ethanol exposure differentially alters central monoamine neurotransmission in alcohol-preferring versus -nonpreferring rats. J. Pharmacol. Exp. Ther. 1999, 288, 1223–1228. [Google Scholar] [PubMed]
- Fowler, J.S.; Volkow, N.D.; Wang, G.J.; Pappas, N.; Logan, J.; MacGregor, R.; Alexoff, D.; Shea, C.; Schlyer, D.; Wolf, A.P.; et al. Inhibition of monoamine oxidase b in the brains of smokers. Nature 1996, 379, 733–736. [Google Scholar] [CrossRef] [PubMed]
- Fowler, J.S.; Volkow, N.D.; Wang, G.J.; Pappas, N.; Logan, J.; Shea, C.; Alexoff, D.; MacGregor, R.R.; Schlyer, D.J.; Zezulkova, I.; et al. Brain monoamine oxidase a inhibition in cigarette smokers. Proc. Natl. Acad. Sci. USA 1996, 93, 14065–14069. [Google Scholar] [CrossRef] [PubMed]
| Group | Locomotor Activity | Stereotypy Count | Margin Time | Center Time |
|---|---|---|---|---|
| A | 1626 ± 135 cm | 20.0 ± 2.1 | 157.3 ± 4.1 s | 21.7 ± 4.1 s |
| B | 1732 ± 173 cm | 22.2 ± 2.4 | 150.2 ± 3.7 s | 28.8 ± 3.7 s |
| AB | 1571 ± 156 cm | 18.3 ± 2.3 | 153.2 ± 4.1 s | 25.8 ± 4.1 s |
| C | 1970 ± 198 cm | 15.5 ± 2.2 | 151.3 ± 3.2 s | 27.7 ± 3.2 s |
| Serotonin Transporter, fmol/mg Tissue | |||||||||
| PND | Striatum | Hippocampus | Cortex | Raphe | Sub nigra | ||||
| 14 | 0.39 ± 0.07 | 0.42 ± 0.19 | 0.42 ± 0.14 | 1.54 ± 0.31 | 1.47 ± 0.23 | ||||
| 35 | 1.03 ± 0.16 | 0.89 ± 0.15 | 0.73 ± 0.12 | 1.57 ± 0.18 | 2.31 ± 0.35 | ||||
| 90 | 1.10 ± 0.10 | 0.95 ± 0.09 | 0.84 ± 0.09 | 1.7 ± 0.37 | 2.07 ± 0.18 | ||||
| Dopamine Transporter, fmol/mg Tissue | |||||||||
| PND | Nuc accumbens | Striatum | Loc coeruleus | Sub nigra | |||||
| 14 | 1.29 ± 0.31 | 1.35 ± 0.18 | 0.91 ± 0.13 | 0.8 ± 0.09 | |||||
| 35 | 1.30 ± 0.07 | 1.39 ± 0.06 | 0.75 ± 0.2 | 0.71 ± 0.06 | |||||
| 90 | 1.64 ± 0.16 | 1.66 ± 0.13 | 0.61 ± 0.02 | 0.83 ± 0.15 | |||||
| Serotonin Transporter (fmol/mg Tissue) | Dopamine Transporter (fmol/mg Tissue) | |||||||
|---|---|---|---|---|---|---|---|---|
| Striatum | Hippocampus | Cortex | Raphe | Sub Nigra | Nuc. Accumbens | Striatum | Sub Nigra | |
| PND 14 | ||||||||
| C | 0.39 ± 0.07 | 0.42 ± 0.19 | 0.42 ± 0.14 | 1.54 ± 0.31 | 1.47 ± 0.23 | 1.29 ± 0.31 | 1.35 ± 0.18 | 0.80 ± 0.09 |
| A | 0.44 ± 0.11 | 0.51 ± 0.10 | 0.44 ± 0.04 | 1.33 ± 0.32 | 1.22 ± 0.23 | 0.96 ± 0.30 | 1.17 ± 0.15 | 0.60 ± 0.08 |
| B | 0.40 ± 0.12 | 0.50 ± 0.09 | 0.45 ± 0.09 | 1.15 ± 0.16 | 1.23 ± 0.14 | 1.11 ± 0.30 | 1.38 ± 0.41 | 0.82 ± 0.19 |
| AB | 0.39 ± 0.18 | 0.49 ± 0.12 | 0.38 ± 0.08 | 1.11 ± 0.24 | 1.29 ± 0.34 | 0.84 ± 0.21 | 1.11 ± 0.14 | 0.60 ± 0.12 |
| PND 35 | ||||||||
| C | 1.03 ± 0.16 | 0.89 ± 0.15 | 0.73 ± 0.12 | 1.57 ± 0.18 | 2.31 ± 0.35 | 1.30 ± 0.07 | 1.39 ± 0.06 | 0.71 ± 0.06 |
| A | 1.01 ± 0.19 | 0.84 ± 0.22 | 0.73 ± 0.15 | 1.52 ± 0.26 | 2.21 ± 0.32 | 1.71 ± 0.59 | 1.70 ± 0.70 | 0.73 ± 0.31 |
| B | 0.98 ± 0.13 | 0.79 ± 0.16 | 0.70 ± 0.09 | 1.60 ± 0.21 | 2.29 ± 0.28 | 1.69 ± 0.53 | 1.71 ± 0.53 | 0.75 ± 0.28 |
| AB | 0.89 ± 0.13 | 0.89 ± 0.26 | 0.59 ± 0.21 | 1.27 ± 0.46 | 2.04 ± 0.40 | 1.71 ± 1.03 | 1.64 ± 0.74 | 0.72 ± 0.33 |
| PND 90 | ||||||||
| C | 1.10 ± 0.10 | 0.95 ± 0.09 | 0.84 ± 0.09 | 1.7 ± 0.37 | 2.07 ± 0.18 | 1.64 ± 0.16 | 1.66 ± 0.13 | 0.83 ± 0.15 |
| A | 0.97 ± 0.15 | 0.79 ± 0.09 | 0.67 ± 0.10 | 1.52 ± 0.26 | 1.96 ± 0.14 | 1.46 ± 0.29 | 1.59 ± 0.33 | 0.76 ± 0.26 |
| B | 0.99 ± 0.15 | 0.90 ± 0.09 | 0.72 ± 0.08 | 1.33 ± 0.11 | 2.03 ± 0.06 | 1.73 ± 0.19 | 1.85 ± 0.35 | 0.86 ± 0.32 |
| AB | 0.90 ± 0.09 | 0.89 ± 0.14 | 0.63 ± 0.12 | 1.23 ± 0.28 | 1.96 ± 0.14 | 1.59 ± 0.28 | 1.75 ± 0.33 | 0.77 ± 0.27 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burke, M.W.; Fillion, M.; Mejia, J.; Ervin, F.R.; Palmour, R.M. Perinatal MAO Inhibition Produces Long-Lasting Impairment of Serotonin Function in Offspring. Brain Sci. 2018, 8, 106. https://doi.org/10.3390/brainsci8060106
Burke MW, Fillion M, Mejia J, Ervin FR, Palmour RM. Perinatal MAO Inhibition Produces Long-Lasting Impairment of Serotonin Function in Offspring. Brain Sciences. 2018; 8(6):106. https://doi.org/10.3390/brainsci8060106
Chicago/Turabian StyleBurke, Mark W., Myriam Fillion, Jose Mejia, Frank R. Ervin, and Roberta M. Palmour. 2018. "Perinatal MAO Inhibition Produces Long-Lasting Impairment of Serotonin Function in Offspring" Brain Sciences 8, no. 6: 106. https://doi.org/10.3390/brainsci8060106
APA StyleBurke, M. W., Fillion, M., Mejia, J., Ervin, F. R., & Palmour, R. M. (2018). Perinatal MAO Inhibition Produces Long-Lasting Impairment of Serotonin Function in Offspring. Brain Sciences, 8(6), 106. https://doi.org/10.3390/brainsci8060106







