Effects of Excitatory Repetitive Transcranial Magnetic Stimulation of the P3 Point in Chronic Stroke Patients—Case Reports
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
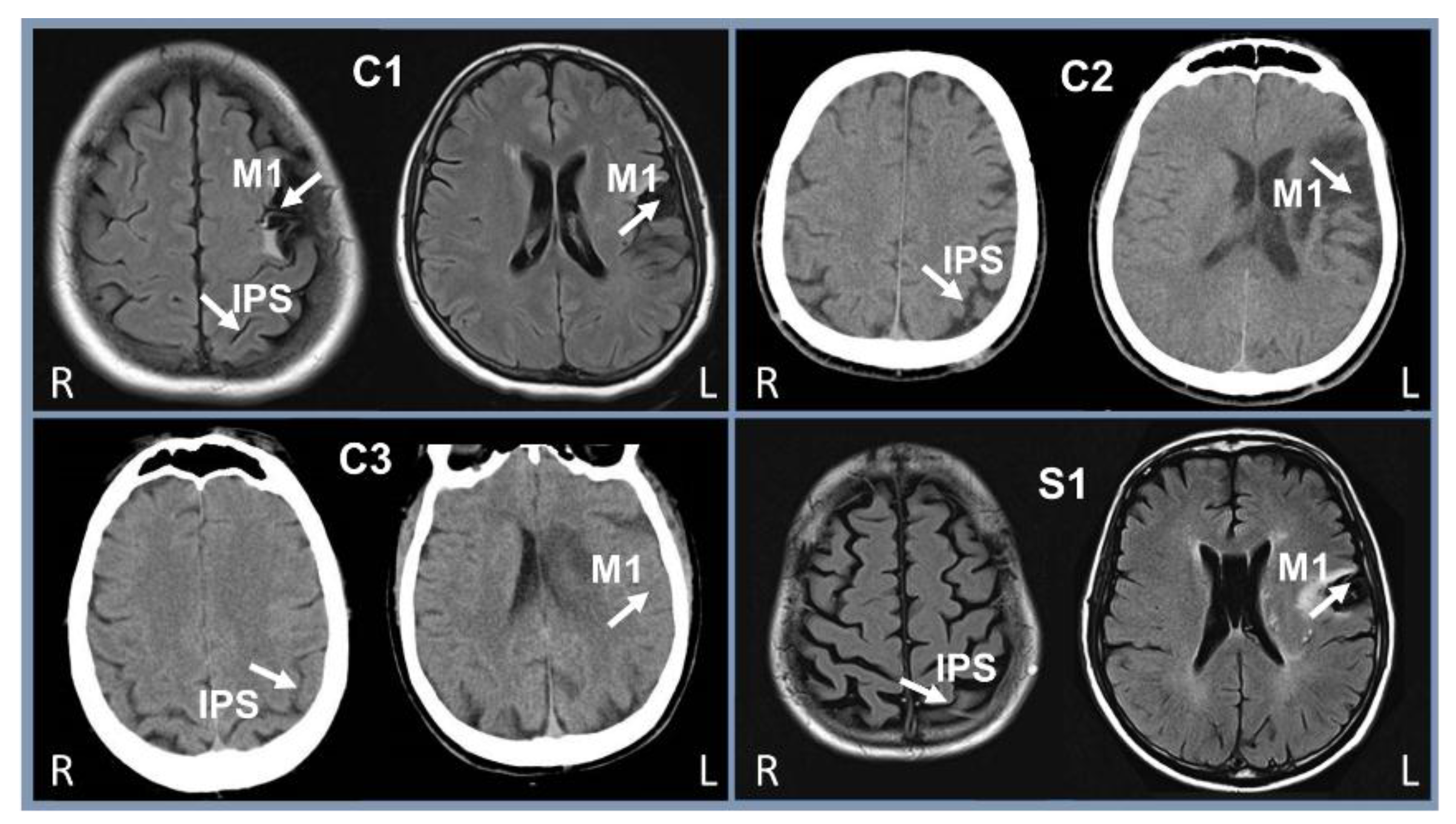
2.2. Subjects
2.3. Evaluations
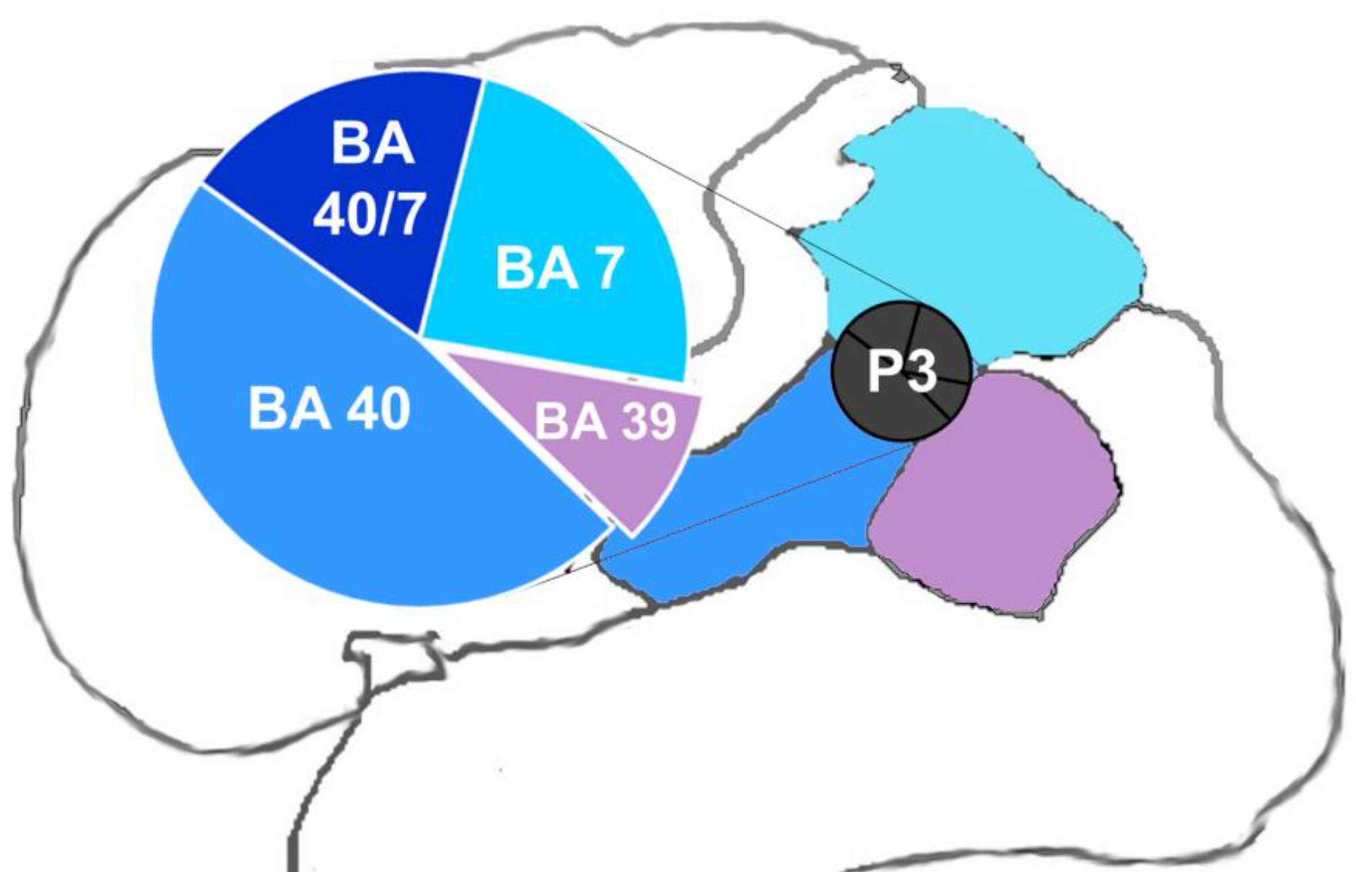
2.4. rTMS
3. Results
4. Discussion
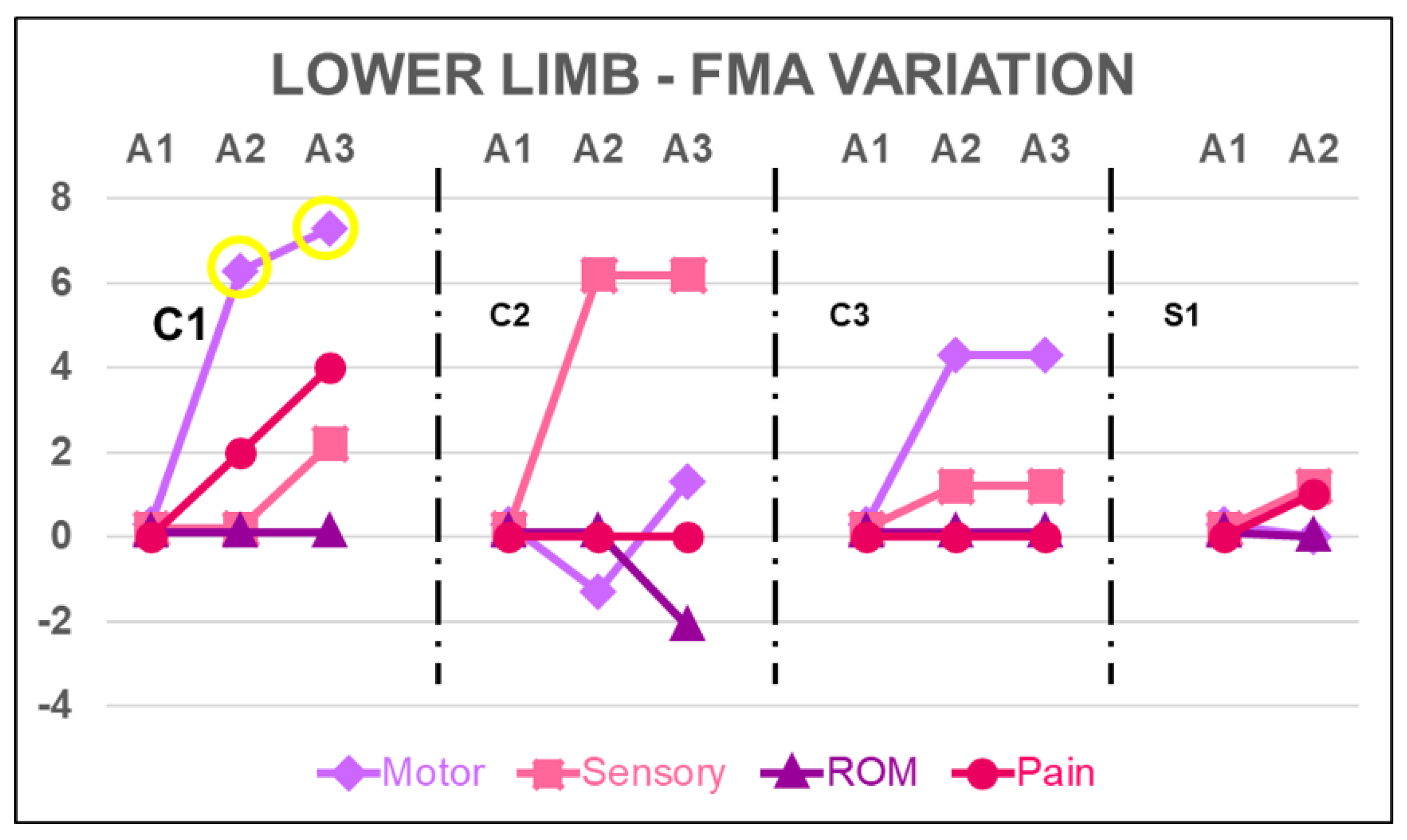
4.1. Lower Limb
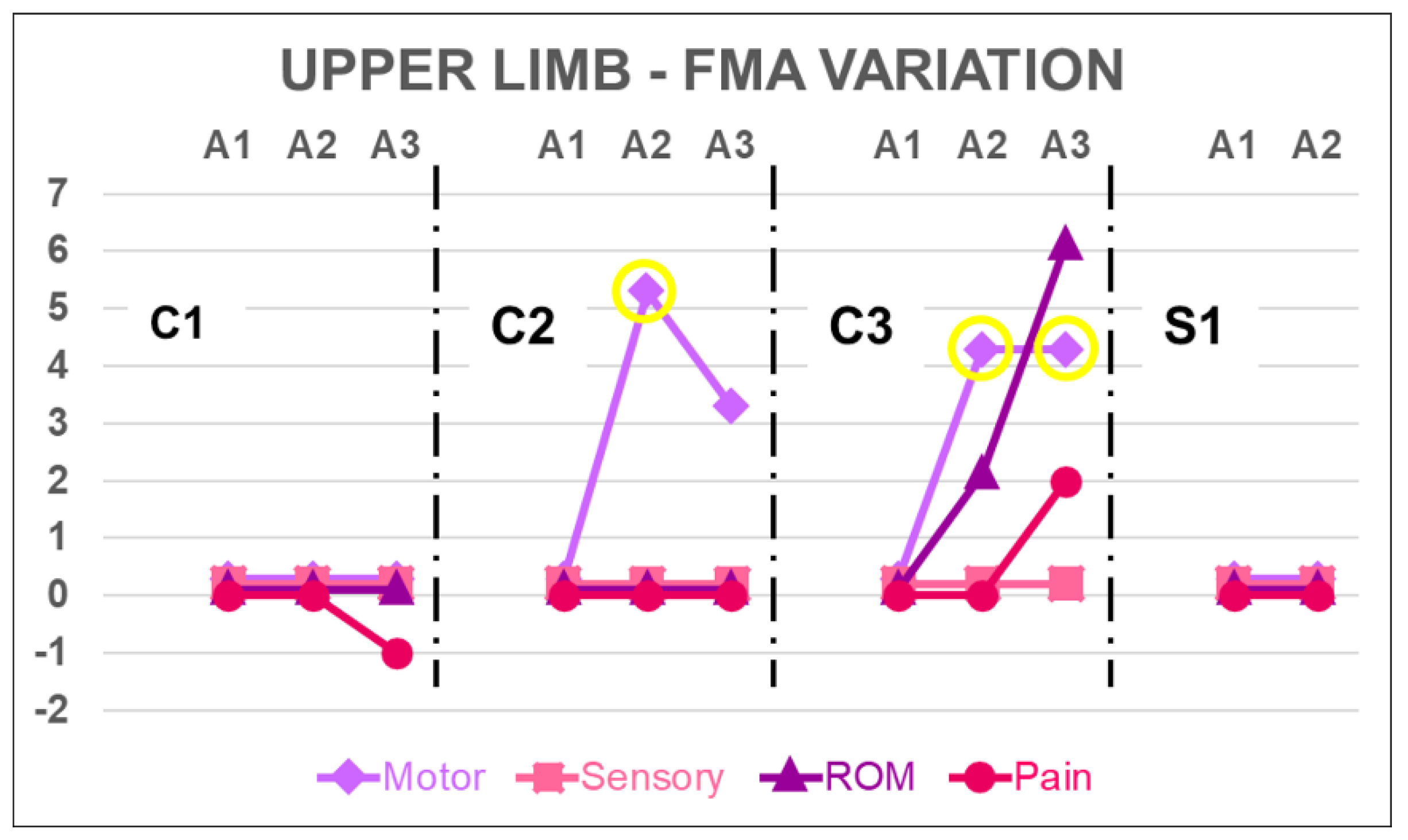
4.2. Upper Limb
4.3. Sensory Function
4.4. Limitations
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Lefaucheur, J.P.; André-Obadia, N.; Poulet, E.; Devanne, H.; Haffen, E.; Londero, A.; Cretin, B.; Leroi, A.-M.; Radtchenko, A.; Saba, G.; et al. Recommandations françaises sur l’utilisation de la stimulation magnétique transcrânienne répétitive (rTMS): Règles de sécurité et indications thérapeutiques. Neurophysiol. Clin. 2011, 41, 221–295. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, N.; Hara, T.; Yamada, N.; Niimi, M.; Kakuda, W.; Abo, M. The efficacy of high-frequency repetitive transcranial magnetic stimulation for improving apathy in chronic stroke patients. Eur. Neurol. 2017, 78, 28–32. [Google Scholar] [CrossRef] [PubMed]
- Gu, S.Y.; Chang, M.C. The effects of 10-Hz repetitive transcranial magnetic stimulation on depression in chronic stroke patients. Brain Stimul. 2017, 10, 270–274. [Google Scholar] [CrossRef] [PubMed]
- Hara, T.; Abo, M.; Kakita, K.; Mori, Y.; Yoshida, M.; Sasaki, N. The effect of selective transcranial magnetic stimulation with functional near-infrared spectroscopy and intensive speech therapy on individuals with post-stroke aphasia. Eur. Neurol. 2017, 77, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Harvey, D.Y.; Podell, J.; Turkeltaub, P.E.; Faseyitan, O.; Coslett, H.B.; Hamilton, R.H. Functional reorganization of right prefrontal cortex underlies sustained naming improvements in chronic aphasia via repetitive transcranial magnetic stimulation. Cogn. Behav. Neurol. 2017, 30, 133–144. [Google Scholar] [CrossRef] [PubMed]
- Chieffo, R.; Ferrari, F.; Battista, P.; Houdayer, E.; Nuara, A.; Alemanno, F.; Abutalebi, J.; Zangen, A.; Comi, G.; Cappa, S.F.; et al. Excitatory deep transcranial magnetic stimulation with H-coil over the right homologous Broca’s region improves naming in chronic post-stroke aphasia. Neurorehabil. Neural Repair 2014, 28, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Otal, B.; Olma, M.C.; Flöel, A.; Wellwood, I. Inhibitory non-invasive brain stimulation to homologous language regions as an adjunct to speech and language therapy in post-stroke aphasia: A meta-analysis. Front. Hum. Neurosci. 2015, 28, 236. [Google Scholar] [CrossRef] [PubMed]
- Choi, G.S.; Chang, M.C. Effects of high-frequency repetitive transcranial magnetic stimulation on reducing hemiplegic shoulder pain in patients with chronic stroke: A randomized controlled trial. Int. J. Neurosci. 2018, 128, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Hosomi, K.; Kishima, H.; Oshino, S.; Hirata, M.; Tani, N.; Maruo, T.; Yorifuji, S.; Yoshimine, T.; Saitoh, Y. Cortical excitability changes after high-frequency repetitive transcranial magnetic stimulation for central poststroke pain. Pain 2013, 154, 1352–1357. [Google Scholar] [CrossRef] [PubMed]
- Lefaucheur, J.P.; Drouot, X.; Menard-Lefaucheur, I.; Zerah, F.; Bendib, B.; Cesaro, P.; Keravel, Y.; Nguyen, J.P. Neurogenic pain relief by repetitive transcranial magnetic cortical stimulation depends on the origin and the site of pain. J. Neurol. Neurosurg. Psychiatry 2004, 75, 612–616. [Google Scholar] [CrossRef] [PubMed]
- Lefaucheur, J.P.; Drouot, X.; Nguyen, J.P. Interventional neurophysiology for pain control: Duration of pain relief following repetitive transcranial magnetic stimulation of the motor cortex. Neurophysiol. Clin. 2001, 31, 247–252. [Google Scholar] [CrossRef]
- Choi, C.M.; Kim, J.H.; Lee, J.K.; Lee, B.Y.; Kee, H.S.; Jung, K.I.; Yoon, S.R. Effects of repetitive transcranial magnetic stimulation over trunk motor spot on balance function in stroke patients. Ann. Rehabil. Med. 2016, 40, 826–834. [Google Scholar] [CrossRef] [PubMed]
- Koyama, S.; Tanabe, S.; Takeda, K.; Warashina, H.; Sakurai, H.; Kanada, Y.; Okumura, R.; Shinoda, J.; Nagata, J.; Kanno, T. The effects of high-frequency transcranial magnetic stimulation combined with transcutaneous electrical stimulation in a severe stroke patient. Clin. Pract. 2012, 2, e89. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, N.; Mizutani, S.; Kakuda, W.; Abo, M. Comparison of the effects of high- and low-frequency repetitive transcranial magnetic stimulation on upper limb hemiparesis in the early phase of stroke. J. Stroke Cerebrovasc. Dis. 2013, 22, 413–418. [Google Scholar] [CrossRef] [PubMed]
- Yozbatiran, N.; Alonso-Alonso, M.; See, J.; Demirtas-Tatlidede, A.; Luu, D.; Motiwala, R.R.; Pascual-Leone, A.; Cramer, S.C. Safety and behavioral effects of high-frequency repetitive transcranial magnetic stimulation in stroke. Stroke 2009, 40, 309–312. [Google Scholar] [CrossRef] [PubMed]
- Ayache, S.S.; Farhat, W.H.; Zouari, H.G.; Hosseini, H.; Mylius, V.; Lefaucheur, J.P. Stroke rehabilitation using noninvasive cortical stimulation: Motor deficit. Expert Rev. Neurother. 2012, 12, 949–972. [Google Scholar] [CrossRef] [PubMed]
- Salatino, A.; Berra, E.; Troni, W.; Sacco, K.; Cauda, F.; D’Agata, F.; Geminiani, G.; Duca, S.; Dimanico, U.; Ricci, R. Behavioral and neuroplastic effects of low-frequency rTMS of the unaffected hemisphere in a chronic stroke patient: A concomitant TMS and fMRI study. Neurocase 2014, 20, 615–626. [Google Scholar] [CrossRef] [PubMed]
- Speer, A.M.; Kimbrell, T.A.; Wassermann, E.M.; Repella, J.D.; Willis, M.W.; Herscovitch, P.; Post, R.M. Opposite effects of high and low frequency rTMS on regional brain activity in depressed patients. Biol. Psychiatry 2000, 48, 1133–1141. [Google Scholar] [CrossRef]
- Restuccia, D.; Ulivelli, M.; De Capua, A.; Bartalini, S.; Rossi, S. Modulation of high-frequency (600 Hz) somatosensory-evoked potentials after rTMS of the primary sensory cortex. Eur. J. Neurosci. 2007, 26, 2349–2358. [Google Scholar] [CrossRef] [PubMed]
- McCambridge, A.B.; Stinear, J.W.; Byblow, W.D. Revisiting interhemispheric imbalance in chronic stroke: A tDCS study. Clin. Neurophysiol. 2018, 129, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.H.; You, S.H.; Ko, M.H.; Park, J.W.; Lee, K.H.; Jang, S.H.; Yoo, W.K.; Hallett, M. Repetitive transcranial magnetic stimulation-induced corticomotor excitability and associated motor skill acquisition in chronic stroke. Stroke 2006, 37, 1471–1476. [Google Scholar] [CrossRef] [PubMed]
- Shipp, S. Structure and function of the cerebral cortex. Curr. Biol. 2007, 17, R443–R449. [Google Scholar] [CrossRef] [PubMed]
- Meyer, G. Forms and spatial arrangement of neurons in the primary motor cortex of man. J. Comp. Neurol. 1987, 262, 402–428. [Google Scholar] [CrossRef] [PubMed]
- Grefkes, C.; Fink, G.R. The functional organization of the intraparietal sulcus in humans and monkeys. J. Anat. 2005, 207, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Murata, A.; Wen, W.; Asama, H. The body and objects represented in the ventral stream of the parieto-premotor network. Neurosci. Res. 2016, 104, 4–15. [Google Scholar] [CrossRef] [PubMed]
- Verhagen, L.; Dijkerman, H.C.; Grol, M.J.; Toni, I. Perceptuo-motor interactions during prehension movements. J. Neurosci. 2008, 28, 4726–4735. [Google Scholar] [CrossRef] [PubMed]
- Gutteling, T.P.; Park, S.Y.; Kenemans, J.L.; Neggers, S.F.W. TMS of the anterior intraparietal area selectively modulates orientation change detection during action preparation. J. Neurophysiol. 2013, 110, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Rice, N.J.; Tunik, E.; Grafton, S.T. The anterior intraparietal sulcus mediates grasp execution, independent of requirement to update: New insights from transcranial magnetic stimulation. J. Neurosci. 2006, 26, 8176–8182. [Google Scholar] [CrossRef] [PubMed]
- Rice, N.J.; Tunik, E.; Cross, E.S.; Grafton, S.T. On-line grasp control is mediated by the contralateral hemisphere. Brain Res. 2007, 17, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Herwig, U.; Satrapi, P.; Schönfeldt-Lecuona, C. Using the International 10–20 EEG System for positioning of transcranial magnetic stimulation. Brain Topogr. 2003, 16, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Yang, J. The influence of motor expertise on the brain activity of motor task performance: A meta-analysis of functional magnetic resonance imaging studies. Cogn. Affect. Behav. Neurosci. 2015, 15, 381–394. [Google Scholar] [CrossRef] [PubMed]
- Loubinoux, I.; Carel, C.; Pariente, J.; Dechaumont, S.; Albucher, J.F.; Marque, P.; Manelfe, C.; Chollet, F. Correlation between cerebral reorganization and motor recovery after subcortical infarcts. Neuroimage 2003, 20, 2166–2180. [Google Scholar] [CrossRef] [PubMed]
- Samuel, M.; Ceballos-Baumann, A.O.; Blin, J.; Uema, T.; Boecker, H.; Passingham, R.E.; Brooks, D.J. Evidence for lateral premotor and parietal overactivity in Parkinson’s disease during sequential and bimanual movements. A PET study. Brain 1997, 120, 963–976. [Google Scholar] [CrossRef] [PubMed]
- Catalan, M.J.; Honda, M.; Weeks, R.A.; Cohen, L.G.; Hallett, M. The functional neuroanatomy of simple and complex sequential finger movements: A PET study. Brain 1998, 121, 253–264. [Google Scholar] [CrossRef] [PubMed]
- Fink, G.; Marshall, J.C.; Halligan, P.W.; Frith, C.D.; Driver, J.; Frackowiak, R.S.; Dolan, R.J. The neural consequences of conflict between intention and the senses. Brain 1999, 122, 497–512. [Google Scholar] [CrossRef] [PubMed]
- Meehan, S.K.; Staines, W.R. The effect of task-relevance on primary somatosensory cortex during continuous sensory-guided movement in the presence of bimodal competition. Brain Res. 2007, 23, 148–158. [Google Scholar] [CrossRef] [PubMed]
- Le, A.; Vesia, M.; Yan, X.; Crawford, J.D.; Niemeier, M. Parietal area BA7 integrates motor programs for reaching, grasping, and bimanual coordination. J. Neurophysiol. 2017, 117, 624–636. [Google Scholar] [CrossRef] [PubMed]
- Binkofski, F.; Buccino, G.; Stephan, K.M.; Rizzolatti, G.; Seitz, R.J.; Freund, H.J. A parieto-premotor network for object manipulation: Evidence from neuroimaging. Exp. Brain Res. 1999, 128, 210–213. [Google Scholar] [CrossRef] [PubMed]
- Krayenbühl, H.A.; Yaşargil, M.G.; Huber, P. Cerebral arteries. In Cerebral Angiography, 2nd ed.; Huber, P., Ed.; Thieme: New York, NY, USA, 1982; pp. 105–123. ISBN 978-0-86577-067-6. [Google Scholar]
- Radiopaedia.org. Available online: https://radiopaedia.org/articles/middle-cerebral-artery (accessed on 15 February 2018).
- Oldfield, R.C. The Assessment and Analysis of Handedness: The Edinburgh Inventory. Neuropsychologia 1971, 9, 97–113. [Google Scholar] [CrossRef]
- Rossini, P.M.; Barker, A.T.; Berardelli, A.; Caramia, M.D.; Caruso, G.; Cracco, R.Q.; Dimitrijević, M.R.; Hallett, M.; Katayama, Y.; Lücking, A.L.; et al. Non-invasive electrical and magnetic stimulation of the brain, spinal cord and roots: Basic principles and procedures for routine clinical application. Report of an IFCN committee. Electroencephalogr. Clin. Neurophysiol. 1994, 91, 79–92. [Google Scholar] [CrossRef]
- Koch, G.; Versace, V.; Bonnì, S.; Lupo, F.; Lo Gerfo, E.; Oliveri, M.; Caltagirone, C. Resonance of cortico-cortical connections of the motor system with the observation of goal directed grasping movements. Neuropsychologia 2010, 48, 3513–3520. [Google Scholar] [CrossRef] [PubMed]
- Pandian, S.; Arya, K.N.; Kumar, D. Minimal clinically important difference of the lower-extremity Fugl-Meyer assessment in chronic-stroke. Top. Stroke Rehabil. 2016, 23, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Page, S.J.; Fulk, G.D.; Boyne, P. Clinically important differences for the upper-extremity Fugl-Meyer scale in people with minimal to moderate impairment due to chronic stroke. Phys. Ther. 2012, 92, 791–798. [Google Scholar] [CrossRef] [PubMed]
- Borich, M.R.; Brodie, S.M.; Gray, W.A.; Ionta, S.; Boyd, L.A.; Columbia, B. Understanding the role of the primary somatosensory cortex: Opportunities for rehabilitation. Neuropsychologia 2016, 79, 246–255. [Google Scholar] [CrossRef] [PubMed]
- Bolognini, N.; Russo, C.; Edwards, D.J. The sensory side of post-stroke motor rehabilitation. Restor. Neurol. Neurosci. 2016, 34, 571–586. [Google Scholar] [CrossRef] [PubMed]
- Walker, E.R.; Hyngstrom, A.S.; Schmit, B.D. Sensory electrical stimulation improves foot placement during targeted stepping post-stroke. Exp. Brain Res. 2014, 232, 1137–1143. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, C.B.; Medeiros, Í.R.T.; Greters, M.G.; Frota, N.A.F.; Lucato, L.T.; Scaff, M.; Conforto, A.B. Abnormal sensory integration affects balance control in hemiparetic patients within the first year after stroke. Clinics 2011, 66, 2043–2048. [Google Scholar] [CrossRef] [PubMed]
- Hattori, N.; Shibasaki, H.; Wheaton, L.; Wu, T.; Matsuhashi, M.; Hallett, M. Discrete parieto-frontal functional connectivity related to grasping. J. Neurophysiol. 2009, 101, 1267–1282. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Davare, M.; Kraskov, A.; Rothwell, J.C.; Lemon, R.N. Interactions between areas of the cortical grasping network. Curr. Opin. Neurobiol. 2011, 21, 565–570. [Google Scholar] [CrossRef] [PubMed]
- Binkofski, F.; Dohle, C.; Posse, S.; Stephan, K.M.; Hefter, H.; Seitz, R.J.; Freund, H.J. Human anterior intraparietal area subserves prehension: A combined lesion and functional MRI activation study. Neurology 1998, 50, 1253–1259. [Google Scholar] [CrossRef] [PubMed]
- Jeannerod, M. The mechanism of self-recognition in humans. Behav. Brain Res. 2003, 142, 1–15. [Google Scholar] [CrossRef]
- Borstad, A.; Schmalbrock, P.; Choi, S.; Nichols-Larsen, D.S. Neural correlates supporting sensory discrimination after left hemisphere stroke. Brain Res. 2012, 1460, 78–87. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pandian, S.; Arya, K.N. Stroke-related motor outcome measures: Do they quantify the neurophysiological aspects of upper extremity recovery? J. Bodyw. Mov. Ther. 2014, 18, 412–423. [Google Scholar] [CrossRef] [PubMed]
- Gladstone, D.J.; Danells, C.J.; Black, S.E. The Fugl-Meyer Assessment of motor recovery after stroke: A critical review of its measurement properties. Neurorehabil. Neural Repair 2002, 16, 232–240. [Google Scholar] [CrossRef] [PubMed]
- Bushnell, C.; Bettger, J.P.; Cockroft, K.M.; Mattke, S.; Nilsen, D.M.; Piquado, T.; Skidmore, E.R.; Wing, K.; Yenokyan, G. Chronic stroke outcome measures for motor function intervention trials: Expert panel recommendations. Circ. Cardiovasc. Qual. Outcomes 2015, 8, S163–S169. [Google Scholar] [CrossRef] [PubMed]
| C1 | C2 | C3 | S1 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Max | A1 | A2 | A3 | A1 | A2 | A3 | A1 | A2 | A3 | A1 | A2 | ||
| LL-FMA | motor function | 34 | 27 | 33 | 34 | 29 | 28 | 30 | 17 | 21 | 21 | 18 | 18 |
| sensory function | 12 | 10 | 10 | 12 | 6 | 12 | 12 | 9 | 10 | 10 | 10 | 11 | |
| ROM | 20 | 20 | 20 | 20 | 20 | 20 | 18 | 18 | 18 | 18 | 16 | 16 | |
| joint pain | 20 | 10 | 12 | 14 | 20 | 20 | 20 | 20 | 20 | 20 | 19 | 20 | |
| UL-FMA | motor function | 66 | 66 | 66 | 66 | 13 | 18 | 16 | 4 | 8 | 8 | 2 | 2 |
| sensory function | 12 | 12 | 12 | 12 | 12 | 12 | 12 | 6 | 6 | 6 | 6 | 6 | |
| ROM | 24 | 24 | 24 | 24 | 24 | 24 | 24 | 18 | 18 | 24 | 13 | 13 | |
| joint pain | 24 | 23 | 23 | 22 | 20 | 20 | 20 | 18 | 18 | 20 | 20 | 20 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Da Silva, R.L.; De Souza, A.M.C.; Santos, F.F.; Inoue, S.T.; Higgins, J.; Frak, V. Effects of Excitatory Repetitive Transcranial Magnetic Stimulation of the P3 Point in Chronic Stroke Patients—Case Reports. Brain Sci. 2018, 8, 78. https://doi.org/10.3390/brainsci8050078
Da Silva RL, De Souza AMC, Santos FF, Inoue ST, Higgins J, Frak V. Effects of Excitatory Repetitive Transcranial Magnetic Stimulation of the P3 Point in Chronic Stroke Patients—Case Reports. Brain Sciences. 2018; 8(5):78. https://doi.org/10.3390/brainsci8050078
Chicago/Turabian StyleDa Silva, Ronaldo Luis, Angela Maria Costa De Souza, Francielly Ferreira Santos, Sueli Toshie Inoue, Johanne Higgins, and Victor Frak. 2018. "Effects of Excitatory Repetitive Transcranial Magnetic Stimulation of the P3 Point in Chronic Stroke Patients—Case Reports" Brain Sciences 8, no. 5: 78. https://doi.org/10.3390/brainsci8050078
APA StyleDa Silva, R. L., De Souza, A. M. C., Santos, F. F., Inoue, S. T., Higgins, J., & Frak, V. (2018). Effects of Excitatory Repetitive Transcranial Magnetic Stimulation of the P3 Point in Chronic Stroke Patients—Case Reports. Brain Sciences, 8(5), 78. https://doi.org/10.3390/brainsci8050078





