Computer versus Compensatory Calendar Training in Individuals with Mild Cognitive Impairment: Functional Impact in a Pilot Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Control Group
2.3. Intervention
2.3.1. MSS Calendar Training
2.3.2. BF Computer Training
2.4. Outcome Measures
2.4.1. Global Cognitive Status
2.4.2. Memory-Based Activities of Daily Living
2.4.3. Self-Efficacy
2.4.4. Adherence
- (1)
- brought the MSS to the appointment (1 point);
- (2)
- has at least one entry for today’s date (1 point);
- (3)
- has entries for events happening at a certain time (2 points) and any time (2 points);
- (4)
- has at least two entries for each of the two days in the journaling section (4 points).
2.5. Analysis
3. Results
3.1. Adherence
3.2. Impact of Cognition
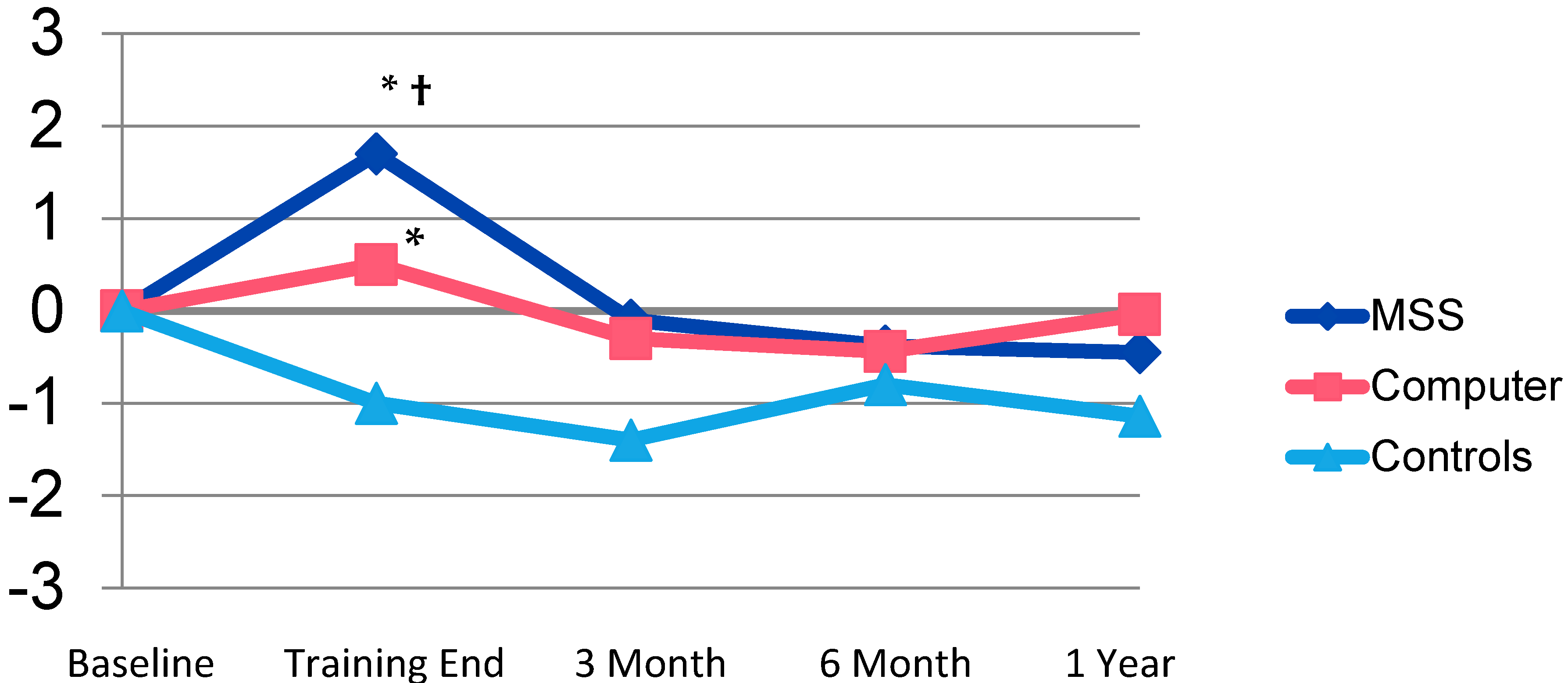
3.3. Memory-Based Activities of Daily Living
3.3.1. Interventions versus Controls
3.3.2. MSS versus BF
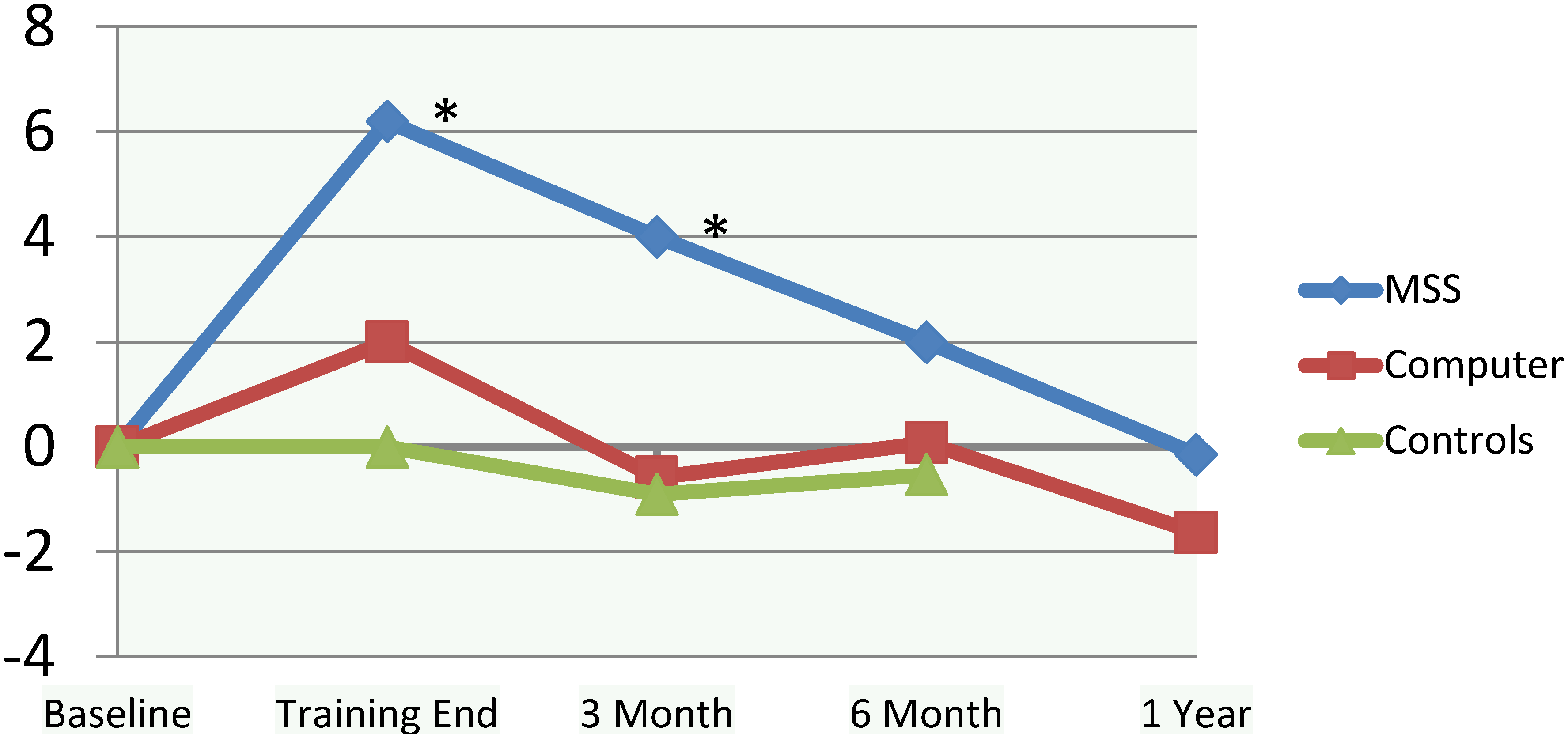
3.4. Self-Efficacy
3.4.1. Interventions versus Controls
3.4.2. MSS versus BF
4. Discussion
4.1. Findings
4.2. Challenges
4.2.1. Impact of Small Sample Sizes
4.2.2. Impact of the Heterogeneity of MCI Diagnosis
4.2.3. Impact of Providing Education
4.2.4. Impact of Subjective Report Measures
4.3. Further Clinical Implications
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- A Consensus on the Brain Training Industry from the Scientific Community (Full Statement). Available online: http://longevity.stanford.edu/a-consensus-on-the-brain-training-industry-from-the-scientific-community-2/ (accessed on 5 September 2017).
- Coyle, H.; Traynor, V.; Solowij, N. Computerized and virtual reality cognitive training for individuals at high risk of cognitive decline: systematic review of the literature. Am. J. Geriatr. Psychiatry 2015, 23, 335–359. [Google Scholar] [CrossRef] [PubMed]
- Rozzini, L.; Costardi, D.; Chilovi, B.V.; Franzoni, S.; Trabucchi, M.; Padovani, A. Efficacy of cognitive rehabilitation in patients with mild cognitive impairment treated with cholinesterase inhibitors. Int. J. Geriatr. Psychiatry 2007, 22, 356–360. [Google Scholar] [CrossRef] [PubMed]
- Talassi, E.; Guerreschi, M.; Feriani, M.; Fedi, V.; Bianchetti, A.; Trabucchi, M. Effectiveness of a cognitive rehabilitation program in mild dementia (MD) and mild cognitive impairment (MCI): A case control study. Arch. Gerontol. Geriatr. 2007, 44, 391–399. [Google Scholar] [CrossRef] [PubMed]
- Chandler, M.J.; Parks, A.C.; Marsiske, M.; Rotblatt, L.J.; Smith, G.E. Everyday impact of cognitive interventions in mild cognitive impairment: A systematic review and meta-analysis. Neuropsychol. Rev. 2016, 26, 225–251. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.E.; Housen, P.; Yaffe, K.; Ruff, R.; Kennison, R.F.; Mahncke, H.W.; Zelinski, E.M. A cognitive training program based on principles of brain plasticity: Results from the Improvement in Memory with Plasticity-based Adaptive Cognitive Training (IMPACT) study. J. Am. Geriatr. Soc. 2009, 57, 594–603. [Google Scholar] [CrossRef] [PubMed]
- Barnes, D.E.; Yaffe, K.; Belfor, N.; Jagust, W.J.; DeCarli, C.; Reed, B.R.; Kramer, J.H. Computer-based cognitive training for mild cognitive impairment: Results from a pilot randomized, controlled trial. Alzheimer Dis. Assoc. Disord. 2009, 23, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Greenaway, M.C.; Hanna, S.M.; Lepore, S.W.; Smith, G.E. A behavioral rehabilitation intervention for amnestic mild cognitive impairment. Am. J. Alzheimers Dis. Other Dement. 2008, 23, 451–461. [Google Scholar] [CrossRef] [PubMed]
- Petersen, R.C. Mild cognitive impairment as a diagnostic entity. J. Intern. Med. 2004, 256, 183–194. [Google Scholar] [CrossRef] [PubMed]
- Petersen, R.C.; Morris, J.C. Mild cognitive impairment as a clinical entity and treatment target. Arch. Neurol. 2005, 62, 1160–1163, discussion 1167. [Google Scholar] [CrossRef] [PubMed]
- Jurica, P.J.; Leitten, C.L. Dementia Rating Scale-2: Professional Manual, 1st ed.; PAR, Inc.: Lutz, FL, USA, 2001. [Google Scholar]
- Pfeffer, R.I.; Kurosaki, T.T.; Harrah, C.H., Jr.; Chance, J.M.; Filos, S. Measurement of functional activities in older adults in the community. J. Gerontol. 1982, 37, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Radloff, L.S. The CES-D scale: A self-report depression scale for research in the general population. Appl. Psychol. Meas. 1977, 1, 385–401. [Google Scholar] [CrossRef]
- Locke, D.E.C.; Chandler Greenaway, M.; Duncan, N.L.; Fields, J.A.; Cuc, A.V.; Hoffman Snyder, C.; Hanna, S.; Lunde, A.; Smith, G.E. A patient-centered analysis of enrollment and retention in a randomized behavioral trial of two cognitive rehabilitation interventions for Mild Cognitive Impairment. J. Prev. Alzheimer’s Dis. 2014, 1, 143–150. [Google Scholar]
- Hepburn, K.W.; Lewis, M.; Sherman, C.W.; Tornatore, J. The savvy caregiver program: Developing and testing a transportable dementia family caregiver training program. Gerontologist 2003, 43, 908–915. [Google Scholar] [CrossRef] [PubMed]
- Zarit, S.H.; Femia, E.E.; Watson, J.; Rice-Oeschger, L.; Kakos, B. Memory Club: A group intervention for people with early-stage dementia and their care partners. Gerontologist 2004, 44, 262–269. [Google Scholar] [CrossRef] [PubMed]
- Logson, R.G.; McCurry, S.M.; Teri, L. Time-limited support groups for individuals with early stage dementia and their care partners: Preliminary outcomes from a controlled trial. Clin. Gerontol. 2006, 30, 5–19. [Google Scholar] [CrossRef]
- Farias, S.T.; Mungas, D.; Reed, B.R.; Cahn-Weiner, D.; Jagust, W.; Baynes, K.; DeCarli, C. The measurement of everyday cognition (ECog): Scale development and psychometric properties. Neuropsychology 2008, 22, 531–544. [Google Scholar] [CrossRef] [PubMed]
- Lorig, K. Outcome Measures for Health Education and other Health Care Interventions, 1st ed.; Sage Publications: Thousand Oaks, CA, USA, 1996. [Google Scholar]
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inf. 2009, 42, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Hampstead, B.M.; Gillis, M.M.; Stringer, A.Y. Cognitive rehabilitation of memory for mild cognitive impairment: A methodological review and model for future research. J. Int. Neuropsychol. Soc. JINS 2014, 20, 135–151. [Google Scholar] [CrossRef] [PubMed]
- Albert, M.S.; DeKosky, S.T.; Dickson, D.; Dubois, B.; Feldman, H.H.; Fox, N.C.; Gamst, A.; Holtzman, D.M.; Jagust, W.J.; Petersen, R.C.; et al. The diagnosis of mild cognitive impairment due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 2011, 7, 270–279. [Google Scholar] [CrossRef] [PubMed]
- Cicerone, K.D. Comments on defining the targets, mechanisms, and active ingredients of rehabilitation. Rehabil. Psychol. 2015, 60, 136–137. [Google Scholar] [CrossRef] [PubMed]
- Farias, S.T.; Lau, K.; Harvey, D.; Denny, K.G.; Barba, C.; Mefford, A.N. Early functional limitations in cognitively normal older adults predict diagnostic conversion to mild cognitive impairment. J. Am. Geriatr. Soc. 2017, 65, 1152–1158. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Patient Group | ||
|---|---|---|---|
| MSS (n = 30) | BF (n = 27) | Controls (n = 20) | |
| Age, Mean (SD) | 76.2 (7.0) | 77.4 (7.2) | 72.3 (7.9) |
| Education, Mean (SD) | 16.0 (2.4) | 16.2 (2.6) | 16.4 (2.7) |
| Sex, male, % | 50.0 | 73.3 | 55.0 |
| White, % | 91.1 | 90.0 | 85.0 |
| Spouse as program partner, % | 91.2 | 90.0 | 75.0 |
| AChE use, % | 54.5 | 40.0 | 70.0 |
| Mean Change (SD) | Compared to Control, Cohen’s d (Significant at) | Calendar vs Computer, Cohen’s d (Significant at) | |
|---|---|---|---|
| Activities of daily living | |||
| MSS | −1.7 (3.3) a | d = 0.75 b | d = 0.39 (n = 105) |
| BF | −0.5 (2.2) a | d = 0.54 (n = 55) | |
| Controls | 1.0 (3.3) | ||
| Self-efficacy | |||
| MSS | 6.2 (12.9) a | d = 0.52 (n = 58) | d = 0.37 (n = 118) |
| BF | 2.0 (9.3) | d = 0.22 (n = 323) | |
| Controls | 0.0 (9.2) | ||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chandler, M.J.; Locke, D.E.C.; Duncan, N.L.; Hanna, S.M.; Cuc, A.V.; Fields, J.A.; Hoffman Snyder, C.R.; Lunde, A.M.; Smith, G.E. Computer versus Compensatory Calendar Training in Individuals with Mild Cognitive Impairment: Functional Impact in a Pilot Study. Brain Sci. 2017, 7, 112. https://doi.org/10.3390/brainsci7090112
Chandler MJ, Locke DEC, Duncan NL, Hanna SM, Cuc AV, Fields JA, Hoffman Snyder CR, Lunde AM, Smith GE. Computer versus Compensatory Calendar Training in Individuals with Mild Cognitive Impairment: Functional Impact in a Pilot Study. Brain Sciences. 2017; 7(9):112. https://doi.org/10.3390/brainsci7090112
Chicago/Turabian StyleChandler, Melanie J., Dona E. C. Locke, Noah L. Duncan, Sherrie M. Hanna, Andrea V. Cuc, Julie A. Fields, Charlene R. Hoffman Snyder, Angela M. Lunde, and Glenn E. Smith. 2017. "Computer versus Compensatory Calendar Training in Individuals with Mild Cognitive Impairment: Functional Impact in a Pilot Study" Brain Sciences 7, no. 9: 112. https://doi.org/10.3390/brainsci7090112
APA StyleChandler, M. J., Locke, D. E. C., Duncan, N. L., Hanna, S. M., Cuc, A. V., Fields, J. A., Hoffman Snyder, C. R., Lunde, A. M., & Smith, G. E. (2017). Computer versus Compensatory Calendar Training in Individuals with Mild Cognitive Impairment: Functional Impact in a Pilot Study. Brain Sciences, 7(9), 112. https://doi.org/10.3390/brainsci7090112






