Problems in Classifying Mild Cognitive Impairment (MCI): One or Multiple Syndromes?
Abstract
1. Introduction
2. Importance of Neuropsychological Assessment in Determining MCI Subtypes
3. Problems Related to the Conceptualization and Operationalization of Maintaining the Functionality of Activities of Daily Living
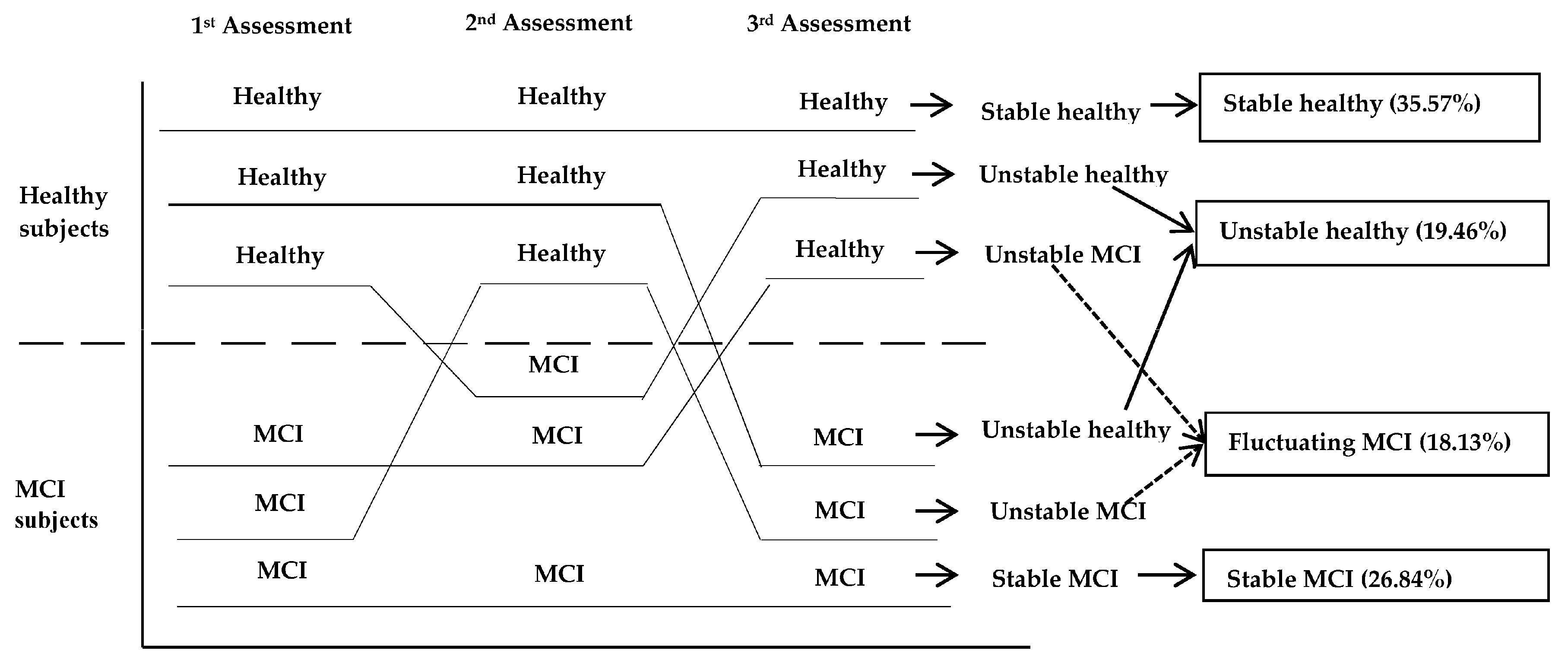
4. Analysis of Some of the Methods to Diagnose MCI and Its Possible Sub-Types
5. Social Factors Associated to Mild Cognitive Impairment
6. Conclusions
Author Contributions
Conflicts of Interest
References
- Petersen, R.C.; Smith, G.E.; Waring, S.C.; Ivnik, R.J.; Kokmen, E.; Tangelos, E.G. Aging, memory, and mild cognitive impairment. Int. Psychogeriatr. 1997, 9, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Petersen, R.C.; Smith, G.E.; Waring, S.C.; Ivnik, R.J.; Tangalos, E.G.; Kokmen, E. Mild cognitive impairment: Clinical characterization and outcome. Arch. Neurol. 1999, 56, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Winblad, B.; Palmer, K.; Kivipelto, M.; Jelic, V.; Fratiglioni, L.; Wahlund, L.; Nordberg, A.; Bäckman, L.; Albert, M.; Almkvist, O. Mild cognitive impairment–beyond controversies, towards a consensus: Report of the International Working Group on Mild Cognitive Impairment. J. Intern. Med. 2004, 256, 240–246. [Google Scholar] [CrossRef] [PubMed]
- Panza, F.; D’introno, A.; Colacicco, A.M.; Capurso, C.; Del Parigi, A.; Caselli, R.J.; Pilotto, A.; Argentieri, G.; Scapicchio, P.L.; Scafato, E. Current epidemiology of mild cognitive impairment and other predementia syndromes. Am. J. Geriatr. Psychiatry 2005, 13, 633–644. [Google Scholar] [CrossRef] [PubMed]
- Petersen, R.C.; Doody, R.; Kurz, A.; Mohs, R.C.; Morris, J.C.; Rabins, P.V.; Ritchie, K.; Rossor, M.; Thal, L.; Winblad, B. Current concepts in mild cognitive impairment. Arch. Neurol. 2001, 58, 1985–1992. [Google Scholar] [CrossRef] [PubMed]
- Petersen, R.C. Mild cognitive impairment as a diagnostic entity. J. Intern. Med. 2004, 256, 183–194. [Google Scholar] [CrossRef] [PubMed]
- Petersen, R.C.; Negash, S. Mild cognitive impairment: An overview. CNS Spectr. 2008, 13, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Bangen, K.J.; Jak, A.J.; Schiehser, D.M.; Delano-Wood, L.; Tuminello, E.; Han, S.D.; Delis, D.C.; Bondi, M.W. Complex activities of daily living vary by mild cognitive impairment subtype. J. Int. Neuropsychol. Soc. 2010, 16, 630–639. [Google Scholar] [CrossRef] [PubMed]
- Busse, A.; Hensel, A.; Guhne, U.; Angermeyer, M.C.; Riedel-Heller, S.G. Mild cognitive impairment: Long-term course of four clinical subtypes. Neurology 2006, 67, 2176–2185. [Google Scholar] [CrossRef] [PubMed]
- Jak, A.J.; Preis, S.R.; Beiser, A.S.; Seshadri, S.; Wolf, P.A.; Bondi, M.W.; Au, R. Neuropsychological Criteria for Mild Cognitive Impairment and Dementia Risk in the Framingham Heart Study. J. Int. Neuropsychol. Soc. 2016, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Lopez, O.L.; Becker, J.T.; Chang, Y.F.; Sweet, R.A.; DeKosky, S.T.; Gach, M.H.; Carmichael, O.T.; McDade, E.; Kuller, L.H. Incidence of mild cognitive impairment in the Pittsburgh Cardiovascular Health Study-Cognition Study. Neurology 2012, 79, 1599–1606. [Google Scholar] [CrossRef] [PubMed]
- Petersen, R.C.; Caracciolo, B.; Brayne, C.; Gauthier, S.; Jelic, V.; Fratiglioni, L. Mild cognitive impairment: A concept in evolution. J. Intern. Med. 2014, 275, 214–228. [Google Scholar] [CrossRef] [PubMed]
- Delano-Wood, L.; Bondi, M.W.; Sacco, J.; Abeles, N.; Jak, A.J.; Libon, D.J.; Bozoki, A. Heterogeneity in mild cognitive impairment: Differences in neuropsychological profile and associated white matter lesion pathology. J. Int. Neuropsychol. Soc. 2009, 15, 906–914. [Google Scholar] [CrossRef] [PubMed]
- Edmonds, E.C.; Delano-Wood, L.; Clark, L.R.; Jak, A.J.; Nation, D.A.; McDonald, C.R.; Libon, D.J.; Au, R.; Galasko, D.; Salmon, D.P. Susceptibility of the conventional criteria for mild cognitive impairment to false-positive diagnostic errors. Alzheimer’s Dement. 2015, 11, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Eppig, J.; Wambach, D.; Nieves, C.; Price, C.C.; Lamar, M.; Delano-Wood, L.; Giovannetti, T.; Bettcher, B.M.; Penney, D.L.; Swenson, R. Dysexecutive functioning in mild cognitive impairment: Derailment in temporal gradients. J. Int. Neuropsychol. Soc. 2012, 18, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Libon, D.J.; Xie, S.X.; Eppig, J.; Wicas, G.; Lamar, M.; Lippa, C.; Bettcher, B.M.; Price, C.C.; Giovannetti, T.; Swenson, R. The heterogeneity of mild cognitive impairment: A neuropsychological analysis. J. Int. Neuropsychol. Soc. 2010, 16, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, P.B.; Mielke, M.M.; Appleby, B.; Oh, E.; Leoutsakos, J.; Lyketsos, C.G. Neuropsychiatric symptoms in MCI subtypes: The importance of executive dysfunction. Int. J. Geriatr. Psychiatry 2011, 26, 364–372. [Google Scholar] [CrossRef] [PubMed]
- Mansbach, W.E.; Mace, R.A.; Clark, K.M. Mild cognitive impairment (MCI) in long-term care patients: Subtype classification and occurrence. Aging Ment. Health 2016, 20, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Brandt, J.; Aretouli, E.; Neijstrom, E.; Samek, J.; Manning, K.; Albert, M.S.; Bandeen-Roche, K. Selectivity of executive function deficits in mild cognitive impairment. Neuropsychology 2009, 23, 607. [Google Scholar] [CrossRef] [PubMed]
- Albert, M.S.; DeKosky, S.T.; Dickson, D.; Dubois, B.; Feldman, H.H.; Fox, N.C.; Gamst, A.; Holtzman, D.M.; Jagust, W.J.; Petersen, R.C. The diagnosis of mild cognitive impairment due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer’s Dement. 2011, 7, 270–279. [Google Scholar] [CrossRef] [PubMed]
- Greenaway, M.C.; Duncan, N.L.; Hanna, S.; Smith, G.E. Predicting functional ability in mild cognitive impairment with the Dementia Rating Scale-2. Int. Psychogeriatr. 2012, 24, 987–993. [Google Scholar] [CrossRef] [PubMed]
- Saunders, N.L.; Summers, M.J. Longitudinal deficits to attention, executive, and working memory in subtypes of mild cognitive impairment. Neuropsychology 2011, 25, 237. [Google Scholar] [CrossRef] [PubMed]
- Schmitter-Edgecombe, M.; Parsey, C.M. Cognitive correlates of functional abilities in individuals with mild cognitive impairment: Comparison of questionnaire, direct observation, and performance-based measures. Clin. Neuropsychol. 2014, 28, 726–746. [Google Scholar] [CrossRef] [PubMed]
- Putcha, D.; Tremont, G. Predictors of independence in instrumental activities of daily living: Amnestic versus non-amnestic MCI. J. Clin. Exp. Neuropsychol. 2016, 38, 991–1004. [Google Scholar] [CrossRef] [PubMed]
- Sachdev, P.S.; Lipnicki, D.M.; Kochan, N.A.; Crawford, J.D.; Thalamuthu, A.; Andrews, G.; Brayne, C.; Matthews, F.E.; Stephan, B.C.; Lipton, R.B. The prevalence of mild cognitive impairment in diverse geographical and ethnocultural regions: The COSMIC collaboration. PLoS ONE 2015, 10, e0142388. [Google Scholar] [CrossRef] [PubMed]
- Apostolo, J.; Holland, C.; O’Connell, M.D.; Feeney, J.; Tabares-Seisdedos, R.; Tadros, G.; Campos, E.; Santos, N.; Robertson, D.A.; Marcucci, M. Mild cognitive decline. A position statement of the Cognitive Decline Group of the European Innovation Partnership for Active and Healthy Ageing (EIPAHA). Maturitas 2016, 83, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Dubois, B.; Feldman, H.H.; Jacova, C.; Cummings, J.L.; DeKosky, S.T.; Barberger-Gateau, P.; Delacourte, A.; Frisoni, G.; Fox, N.C.; Galasko, D. Revising the definition of Alzheimer’s disease: A new lexicon. Lancet Neurol. 2010, 9, 1118–1127. [Google Scholar] [CrossRef]
- Breitner, J. Observations on DSM-5 Mild Neurocognitive Disorder vs. its predecessor, Mild Cognitive Impairment. Acta Psychiatr. Scand. 2015, 131, 15–17. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5); American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Pírez, G.; Santabárbara, J.; Lopez-Anton, R.; Gracia-García, P.; Lobo, E.; De la Cámara, C.; Marcos, G.; Lobo, A. Different subpopulations of mild cognitive impairment are identified by using Petersen’s or DSM-5 criteria. Eur. J. Psychiatry 2017, 31, 80–86. [Google Scholar] [CrossRef]
- American Psychological Association. Supplement to Diagnostic and Statistical Manual of Mental Disorders (DSM-5-UPDATE); American Psychological Association: Washington, DC, USA, 2015. [Google Scholar]
- Espinosa, A.; Alegret, M.; Pesini, P.; Valero, S.; Lafuente, A.; Buendía, M.; San José, I.; Ibarria, M.; Tejero, M.A.; Giménez, J. Cognitive Composites Domain Scores Related to Neuroimaging Biomarkers within Probable-Amnestic Mild Cognitive Impairment-Storage Subtype. J. Alzheimer’s Dis. 2017, 57, 447–459. [Google Scholar] [CrossRef]
- Lopez, O.L.; Kuller, L.H.; Becker, J.T.; Dulberg, C.; Sweet, R.A.; Gach, H.M.; DeKosky, S.T. Incidence of dementia in mild cognitive impairment in the cardiovascular health study cognition study. Arch. Neurol. 2007, 64, 416–420. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Mardomingo, M.; García-Herranz, S.; Peraita-Adrados, H. Detección precoz del deterioro cognitivo leve y conversión a la enfermedad de Alzheimer: Un estudio longitudinal de casos. Psicogeriatra 2010, 2, 105–111. [Google Scholar]
- García-Herranz, S.; Díaz-Mardomingo, M.C.; Peraita, H. Evaluación y seguimiento del envejecimiento sano y con deterioro cognitivo leve (DCL) a través del TAVEC. Anales de Psicología 2014, 30, 372–379. [Google Scholar] [CrossRef]
- Loewenstein, D.A.; Acevedo, A.; Small, B.J.; Agron, J.; Crocco, E.; Duara, R. Stability of different subtypes of mild cognitive impairment among the elderly over a 2- to 3-year follow-up period. Dement. Geriatr. Cogn. Disord. 2009, 27, 418–423. [Google Scholar] [CrossRef] [PubMed]
- Kaduszkiewicz, H.; Eisele, M.; Wiese, B.; Prokein, J.; Luppa, M.; Luck, T.; Jessen, F.; Bickel, H.; Mosch, E.; Pentzek, M.; et al. Study on Aging, Cognition, and Dementia in Primary Care Patients (AgeCoDe). Study Group Prognosis of mild cognitive impairment in general practice: Results of the German AgeCoDe study. Ann. Fam. Med. 2014, 12, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Klekociuk, S.Z.; Summers, J.J.; Vickers, J.C.; Summers, M.J. Reducing false positive diagnoses in mild cognitive impairment: The importance of comprehensive neuropsychological assessment. Eur. J. Neurol. 2014, 21, 1330–1336. [Google Scholar] [CrossRef] [PubMed]
- García-Herranz, S. Relevancia de los Test Neuropsicológicos de Evaluación de la Memoria Episódica y de la Función Ejecutiva Como Marcadores en la Detección Precoz del Deterioro Cognitivo Ligero (DCL): Seguimiento Longitudinal. Ph.D. Thesis, Universidad Nacional de Educación a Distancia (UNED), Madrid, Spain, 2013. [Google Scholar]
- Fratiglioni, L.; Winblad, B.; Von Strauss, E. Prevention of Alzheimer’s disease and dementia. Major findings from the Kungsholmen Project. Physiol. Behav. 2007, 92, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, J.; Arnold, R.; Dawson, K.; Nestor, P.J.; Hodges, J.R. Outcome in subgroups of mild cognitive impairment (MCI) is highly predictable using a simple algorithm. J. Neurol. 2009, 256, 1500–1509. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.E.; Bondi, M.W. Mild Cognitive Impairment and Dementia: Definitions, Diagnosis, and Treatment; Oxford University Press: New York, NY, USA, 2013. [Google Scholar]
- Petersen, R.C. Clinical practice. Mild cognitive impairment. N. Engl. J. Med. 2011, 364, 2227–2234. [Google Scholar] [CrossRef] [PubMed]
- Artero, S.; Ancelin, M.L.; Portet, F.; Dupuy, A.; Berr, C.; Dartigues, J.F.; Tzourio, C.; Rouaud, O.; Poncet, M.; Pasquier, F.; et al. Risk profiles for mild cognitive impairment and progression to dementia are gender specific. J. Neurol. Neurosurg. Psychiatry 2008, 79, 979–984. [Google Scholar] [CrossRef] [PubMed]
- Migliacci, M.; Scharovsky, D.; Gonorazky, S. Deterioro cognitivo leve: Características neuropsicológicas de los distintos subtipos. Revista de Neurología 2009, 48, 237–241. [Google Scholar] [PubMed]
- Pusswald, G.; Moser, D.; Gleiß, A.; Janzek-Hawlat, S.; Auff, E.; Dal-Bianco, P.; Lehrner, J. Prevalence of mild cognitive impairment subtypes in patients attending a memory outpatient clinic—Comparison of two modes of mild cognitive impairment classification. Results of the Vienna Conversion to Dementia Study. Alzheimer’s Dement. 2013, 9, 366–376. [Google Scholar] [CrossRef] [PubMed]
- Ganguli, M.; Snitz, B.E.; Saxton, J.A.; Chang, C.H.; Lee, C.; Vander Bilt, J.; Hughes, T.F.; Loewenstein, D.A.; Unverzagt, F.W.; Petersen, R.C. Outcomes of mild cognitive impairment by definition: A population study. Arch. Neurol. 2011, 68, 761–767. [Google Scholar] [CrossRef] [PubMed]
- Brodaty, H.; Aerts, L.; Crawford, J.D.; Heffernan, M.; Kochan, N.A.; Reppermund, S.; Kang, K.; Maston, K.; Draper, B.; Trollor, J.N. Operationalizing the Diagnostic Criteria for Mild Cognitive Impairment: The Salience of Objective Measures in Predicting Incident Dementia. Am. J. Geriatr. Psychiatry 2016, 25, 485–497. [Google Scholar] [CrossRef] [PubMed]
- Clark, L.R.; Delano-Wood, L.; Libon, D.J.; McDonald, C.R.; Nation, D.A.; Bangen, K.J.; Jak, A.J.; Au, R.; Salmon, D.P.; Bondi, M.W. Are empirically-derived subtypes of mild cognitive impairment consistent with conventional subtypes? J. Int. Neuropsychol. Soc. 2013, 19, 635–645. [Google Scholar] [CrossRef] [PubMed]
- Silva, D.; Guerreiro, M.; Maroco, J.; Santana, I.; Rodrigues, A.; Bravo Marques, J.; de Mendonca, A. Comparison of four verbal memory tests for the diagnosis and predictive value of mild cognitive impairment. Dement. Geriatr. Cogn. Dis. Extra 2012, 2, 120–131. [Google Scholar] [CrossRef] [PubMed]
- Valls-Pedret, C.; Molinuevo, J.L.; Rami, L. Diagnóstico precoz de la enfermedad de Alzheimer: Fase prodrómica y preclínica. Rev. Neurol. 2010, 51, 471–480. [Google Scholar] [PubMed]
- Jak, A.J.; Bondi, M.W.; Delano-Wood, L.; Wierenga, C.; Corey-Bloom, J.; Salmon, D.P.; Delis, D.C. Quantification of five neuropsychological approaches to defining mild cognitive impairment. Am. J. Geriatr. Psychiatry 2009, 17, 368–375. [Google Scholar] [CrossRef] [PubMed]
- Olazarán Rodríguez, J.; Molinuevo Guix, J.L. Deterioro Cognitivo Ligero. In Guía oficial para la práctica clínica en demencias: Conceptos, criterios y recomendaciones; Molinuevo, J.L., Peña-Casanova, J., Eds.; Prous Science, SAU; Thomson Reuters: Barcelona, Spain, 2009; pp. 15–22. [Google Scholar]
- Lopez, O.L. Mild cognitive impairment. Contin. Lifelong Learn. Neurol. 2013, 19, 411. [Google Scholar] [CrossRef] [PubMed]
- Albert, M.S.; Moss, M.B.; Tanzi, R.; Jones, K. Preclinical prediction of AD using neuropsychological tests. J. Int. Neuropsychol. Soc. 2001, 7, 631–639. [Google Scholar] [CrossRef] [PubMed]
- Dubois, B.; Albert, M.L. Amnestic MCI or prodromal Alzheimer’s disease? Lancet Neurol. 2004, 3, 246–248. [Google Scholar] [CrossRef]
- Dubois, B.; Feldman, H.H.; Jacova, C.; DeKosky, S.T.; Barberger-Gateau, P.; Cummings, J.; Delacourte, A.; Galasko, D.; Gauthier, S.; Jicha, G. Research criteria for the diagnosis of Alzheimer’s disease: Revising the NINCDS–ADRDA criteria. Lancet Neurol. 2007, 6, 734–746. [Google Scholar] [CrossRef]
- Bondi, M.W.; Edmonds, E.C.; Jak, A.J.; Clark, L.R.; Delano-Wood, L.; McDonald, C.R.; Nation, D.A.; Libon, D.J.; Au, R.; Galasko, D. Neuropsychological criteria for mild cognitive impairment improves diagnostic precision, biomarker associations, and progression rates. J. Alzheimer’s Dis. 2014, 42, 275–289. [Google Scholar]
- Serra, L.; Bozzali, M.; Cercignani, M.; Perri, R.; Fadda, L.; Caltagirone, C.; Carlesimo, G.A. Recollection and familiarity in amnesic mild cognitive impairment. Neuropsychology 2010, 24, 316. [Google Scholar] [CrossRef] [PubMed]
- Molinuevo, J.L.; Gómez-Anson, B.; Monte, G.C.; Bosch, B.; Sánchez-Valle, R.; Rami, L. Neuropsychological profile of prodromal Alzheimer’s disease (Prd-AD) and their radiological correlates. Arch. Gerontol. Geriatr. 2011, 52, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Venneri, A.; Gorgoglione, G.; Toraci, C.; Nocetti, L.; Panzetti, P.; Nichelli, P. Combining neuropsychological and structural neuroimaging indicators of conversion to Alzheimer’s disease in amnestic mild cognitive impairment. Curr. Alzheimer Res. 2011, 8, 789–797. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Brennan, L.; Eppig, J.; Price, C.C.; Lamar, M.; Delano-Wood, L.; Bangen, K.J.; Edmonds, E.C.; Clark, L.; Nation, D.A. Visuoconstructional impairment in subtypes of mild cognitive impairment. Appl. Neuropsychol. Adult 2016, 23, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Salmon, D.P.; Bondi, M.W. Neuropsychological assessment of dementia. Annu. Rev. Psychol. 2009, 60, 257–282. [Google Scholar] [CrossRef] [PubMed]
- Schinka, J.A.; Loewenstein, D.A.; Raj, A.; Schoenberg, M.R.; Banko, J.L.; Potter, H.; Duara, R. Defining mild cognitive impairment: Impact of varying decision criteria on neuropsychological diagnostic frequencies and correlates. Am. J. Geriatr. Psychiatry 2010, 18, 684–691. [Google Scholar] [CrossRef] [PubMed]
- Trittschuh, E.H.; Crane, P.K.; Larson, E.B.; Cholerton, B.; McCormick, W.C.; McCurry, S.M.; Bowen, J.D.; Baker, L.D.; Craft, S. Effects of varying diagnostic criteria on prevalence of mild cognitive impairment in a community based sample. J. Alzheimer’s Dis. 2011, 25, 163–173. [Google Scholar]
- Benedet, M.J.; Alejandre, M.Á. TAVEC: Test de Aprendizaje Verbal España-Complutense: Manual; TEA Ediciones: Madrid, Spain, 1998. [Google Scholar]
- Peña-Casanova, J.; Esparcia, A.J.; Saladié, D.G.; Olmos, J.G. Normalidad, semiología y patología neuropsicológicas: Programa integrado de exploración neuropsicológica “Test Barcelona”; Masson: Barcelona, Spain, 1991. [Google Scholar]
- Reynolds, C.R. Comprehensive Trail Making Test (CTMT); Pro-Ed: Austin, TX, USA, 2002. [Google Scholar]
- Rey, A. Test de Copia y de Reproducción de Memoria de Figuras Geométricas Complejas; TEA: Madrid, Spain, 2003. [Google Scholar]
- Díaz, M.C.; Peraita, H. Detección precoz del deterioro cognitivo ligero de la tercera edad. Psicothema 2008, 20, 438–444. [Google Scholar]
- Peraita, H.; García-Herranz, S.; Díaz-Mardomingo, C. Evolution of specific cognitive subprofiles of mild cognitive impairment in a three-year longitudinal study. Curr. Aging Sci. 2011, 4, 171–182. [Google Scholar] [CrossRef] [PubMed]
- García-Herranz, S.; Díaz-Mardomingo, M.C.; Peraita, H. Neuropsychological predictors of conversion to probable Alzheimer disease in elderly with mild cognitive impairment. J. Neuropsychol. 2015, 10, 239–255. [Google Scholar] [CrossRef] [PubMed]
- Díaz, M.C.; Chacón, J.; Martínez, R.; Peraita, H. Stability of the cognitive dimensions of a battery of neuropsychological tests. Psicothema 2012, 24, 587–593. [Google Scholar]
- Fischer, P.; Jungwirth, S.; Zehetmayer, S.; Weissgram, S.; Hoenigschnabl, S.; Gelpi, E.; Krampla, W.; Tragl, K.H. Conversion from subtypes of mild cognitive impairment to Alzheimer dementia. Neurology 2007, 68, 288–291. [Google Scholar] [CrossRef] [PubMed]
- Lopez, O.L.; Becker, J.T.; Sweet, R.A. Non-cognitive symptoms in mild cognitive impairment subjects. Neurocase 2005, 11, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Jekel, K.; Damian, M.; Wattmo, C.; Hausner, L.; Bullock, R.; Connelly, P.J.; Dubois, B.; Eriksdotter, M.; Ewers, M.; Graessel, E. Mild cognitive impairment and deficits in instrumental activities of daily living: A systematic review. Alzheimer’s Res. Ther. 2015, 7, 17. [Google Scholar] [CrossRef] [PubMed]
- Njegovan, V.; Man-Son-Hing, M.; Mitchell, S.L.; Molnar, F.J. The hierarchy of functional loss associated with cognitive decline in older persons. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2001, 56, 638–643. [Google Scholar] [CrossRef]
- Peres, K.; Chrysostome, V.; Fabrigoule, C.; Orgogozo, J.M.; Dartigues, J.F.; Barberger-Gateau, P. Restriction in complex activities of daily living in MCI Impact on outcome. Neurology 2006, 67, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Triebel, K.L.; Martin, R.; Griffith, H.R.; Marceaux, J.; Okonkwo, O.C.; Harrell, L.; Clark, D.; Brockington, J.; Bartolucci, A.; Marson, D.C. Declining financial capacity in mild cognitive impairment A 1-year longitudinal study. Neurology 2009, 73, 928–934. [Google Scholar] [CrossRef] [PubMed]
- Fang, M.L.; Coatta, K.; Badger, M.; Wu, S.; Easton, M.; Nygård, L.; Astell, A.; Sixsmith, A. Informing understandings of mild cognitive impairment for older adults: Implications from a scoping review. J. Appl. Gerontol. 2015, 36, 808–839. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.J.; Heaton, R.K. Sensitivity and specificity of WAIS–III/WMS–III demographically corrected factor scores in neuropsychological assessment. J. Int. Neuropsychol. Soc. 2001, 7, 867–874. [Google Scholar] [PubMed]
- Callahan, C.M.; Hall, K.S.; Hui, S.L.; Musick, B.S.; Unverzagt, F.W.; Hendrie, H.C. Relationship of age, education, and occupation with dementia among a community-based sample of African Americans. Arch. Neurol. 1996, 53, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Marcopulos, B.A.; McLain, C.A. Are our norms “normal”? A 4-year follow-up study of a biracial sample of rural elders with low education. Clin. Neuropsychol. 2003, 17, 19–33. [Google Scholar] [CrossRef] [PubMed]
- Ivnik, R.J.; Malec, J.F.; Smith, G.E.; Tangalos, E.G.; Petersen, R.C.; Kokmen, E.; Kurland, L.T. Mayo’s Older Americans Normative Studies: WAIS-R norms for ages 56 to 97. Clin. Neuropsychol. 1992, 6, 1–30. [Google Scholar] [CrossRef]
- Ivnik, R.J.; Smith, G.E.; Lucas, J.A.; Tangalos, E.G.; Kokmen, E.; Petersen, R.C. Free and cued selective reminding test: MOANS norms. J. Clin. Exp. Neuropsychol. 1997, 19, 676–691. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.E.; Ivnik, R.J. Normative Neuropsychology. Mild Cognitive Impairment; Oxford: New York, NY, USA, 2003; pp. 63–88. [Google Scholar]
- Steinberg, B.A.; Bieliauskas, L.A.; Smith, G.E.; Ivnik, R.J.; Malec, J.F. Mayo’s Older Americans normative studies: Age-and IQ-adjusted norms for the auditory verbal learning test and the visual spatial learning test. Clin. Neuropsychol. 2005, 19, 464–523. [Google Scholar] [CrossRef] [PubMed]
- Lucas, J.A.; Ivnik, R.J.; Willis, F.B.; Ferman, T.J.; Smith, G.E.; Parfitt, F.C.; Petersen, R.C.; Graff-Radford, N.R. Mayo’s Older African Americans Normative Studies: Normative data for commonly used clinical neuropsychological measures. Clin. Neuropsychol. 2005, 19, 162–183. [Google Scholar] [CrossRef] [PubMed]
- Pauker, J.D. Constructing overlapping cell tables to maximize the clinical usefulness of normative test data: Rationale and an example from neuropsychology. J. Clin. Psychol. 1988, 44, 930–933. [Google Scholar] [CrossRef]
- Peña-Casanova, J.; Blesa, R.; Aguilar, M.; Gramunt-Fombuena, N.; Gomez-Anson, B.; Oliva, R.; Molinuevo, J.L.; Robles, A.; Barquero, M.S.; Antunez, C.; et al. NEURONORMA Study Team Spanish Multicenter Normative Studies (NEURONORMA Project): Methods and sample characteristics. Arch. Clin. Neuropsychol. 2009, 24, 307–319. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, L.; Rousseeuw, P.J. Finding Groups in Data: An Introduction to Cluster Analysis; John Wiley & Sons: New York, NY, USA, 2005. [Google Scholar]
- Campos-Magdaleno, M.; Facal, D.; Juncos-Rabadán, O.; Braña, T.; Pereiro, A.X. Cluster subtypes of the Spanish version of the California Verbal Learning Test in a sample of adults with subjective memory complaints. J. Clin. Exp. Neuropsychol. 2014, 36, 317–333. [Google Scholar] [CrossRef] [PubMed]
- Vermunt, J.K.; Magidson, J. Technical Guide for Latent GOLD 4.0: Basic and Advanced; Statistical Innovations Inc.: Belmont, MA, USA, 2005. [Google Scholar]
- Peraita, H.; Chacón, J.; Díaz-Mardomingo, C.; Martínez-Arias, R. Profiles of mild cognitive impairment (MCI) in the elderly. Span. J. Psychol. 2015, 18, E90. [Google Scholar] [CrossRef] [PubMed]
- Facal, D.; Guàrdia-Olmos, J.; Juncos-Rabadán, O. Diagnostic transitions in mild cognitive impairment by use of simple Markov models. Int. J. Geriatr. Psychiatry 2015, 30, 669–676. [Google Scholar] [CrossRef] [PubMed]
- Baumeister, R.F.; Leary, M.R. The need to belong: Desire for interpersonal attachments as a fundamental human motivation. Psychol. Bull. 1995, 117, 497. [Google Scholar] [CrossRef] [PubMed]
- Hawkley, L.C.; Cacioppo, J.T. Loneliness matters: A theoretical and empirical review of consequences and mechanisms. Ann. Behav. Med. 2010, 40, 218–227. [Google Scholar] [CrossRef] [PubMed]
- Sampson, E.L.; Bulpitt, C.J.; Fletcher, A.E. Survival of community-dwelling older people: The effect of cognitive impairment and social engagement. J. Am. Geriatr. Soc. 2009, 57, 985–991. [Google Scholar] [CrossRef] [PubMed]
- Bassuk, S.S.; Glass, T.A.; Berkman, L.F. Social disengagement and incident cognitive decline in community-dwelling elderly persons. Ann. Intern. Med. 1999, 131, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, N.; Molony, S.; Fennie, K.; Shellman, J.; McCorkle, R. Predictors of Social Isolation in Community Living Older Persons. Ph.D. Thesis, Yale University, New Haven, CT, USA, Unpublished work. 2010. [Google Scholar]
- Fratiglioni, L.; Paillard-Borg, S.; Winblad, B. An active and socially integrated lifestyle in late life might protect against dementia. Lancet Neurol. 2004, 3, 343–353. [Google Scholar] [CrossRef]
- Cacioppo, J.T.; Cacioppo, S.; Capitanio, J.P.; Cole, S.W. The neuroendocrinology of social isolation. Annu. Rev. Psychol. 2015, 66, 733–767. [Google Scholar] [CrossRef] [PubMed]
- Pillemer, S.C.; Holtzer, R. The differential relationships of dimensions of perceived social support with cognitive function among older adults. Aging Ment. Health 2016, 20, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Hultsch, D.F.; Hertzog, C.; Small, B.J.; Dixon, R.A. Use it or lose it: Engaged lifestyle as a buffer of cognitive decline in aging? Psychol. Aging 1999, 14, 245. [Google Scholar] [CrossRef] [PubMed]
- Aartsen, M.J.; Smits, C.H.; van Tilburg, T.; Knipscheer, K.C.; Deeg, D.J. Activity in older adults: Cause or consequence of cognitive functioning? A longitudinal study on everyday activities and cognitive performance in older adults. J. Gerontol. B Psychol. Sci. Soc. Sci. 2002, 57, 153–162. [Google Scholar] [CrossRef]
- Seeman, T.E.; Lusignolo, T.M.; Albert, M.; Berkman, L. Social relationships, social support, and patterns of cognitive aging in healthy, high-functioning older adults: MacArthur studies of successful aging. Health Psychol. 2001, 20, 243. [Google Scholar] [CrossRef] [PubMed]
- Fratiglioni, L.; Hui-Xin, W.; Ericsson, K.; Maytan, M.; Winblad, B. Influence of social network on occurrence of dementia: A community-based longitudinal study. Lancet 2000, 355, 1315. [Google Scholar] [CrossRef]
- James, B.D.; Wilson, R.S.; Barnes, L.L.; Bennett, D.A. Late-life social activity and cognitive decline in old age. J. Int. Neuropsychol. Soc. 2011, 17, 998–1005. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R.S.; Boyle, P.A.; James, B.D.; Leurgans, S.E.; Buchman, A.S.; Bennett, D.A. Negative social interactions and risk of mild cognitive impairment in old age. Neuropsychology 2015, 29, 561. [Google Scholar] [CrossRef] [PubMed]
- Zunzunegui, M.V.; Alvarado, B.E.; Del Ser, T.; Otero, A. Social networks, social integration, and social engagement determine cognitive decline in community-dwelling Spanish older adults. J. Gerontol. B Psychol. Sci. Soc. Sci. 2003, 58, S93–S100. [Google Scholar] [CrossRef] [PubMed]
- Tilvis, R.; Pitkala, K.H.; Jolkkonen, J.; Strandberg, T. Social networks and dementia. Lancet 2000, 356. [Google Scholar] [CrossRef]
- O’luanaigh, C.; O’connell, H.; Chin, A.; Hamilton, F.; Coen, R.; Walsh, C.; Walsh, J.; Caokley, D.; Cunningham, C.; Lawlor, B. Loneliness and cognition in older people: The Dublin Healthy Ageing study. Aging Ment. Health 2012, 16, 347–352. [Google Scholar] [CrossRef] [PubMed]
- Boss, L.; Kang, D.; Branson, S. Loneliness and cognitive function in the older adult: A systematic review. Int. Psychogeriatr. 2015, 27, 541–553. [Google Scholar] [CrossRef] [PubMed]
- Gow, A.J.; Corley, J.; Starr, J.M.; Deary, I.J. Which social network or support factors are associated with cognitive abilities in old age? Gerontology 2013, 59, 454–463. [Google Scholar] [CrossRef] [PubMed]
- Shankar, A.; Hamer, M.; McMunn, A.; Steptoe, A. Social isolation and loneliness: Relationships with cognitive function during 4 years of follow-up in the English Longitudinal Study of Ageing. Psychosom. Med. 2013, 75, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Tilvis, R.S.; Kahonen-Vare, M.H.; Jolkkonen, J.; Valvanne, J.; Pitkala, K.H.; Strandberg, T.E. Predictors of cognitive decline and mortality of aged people over a 10-year period. J. Gerontol. A Biol. Sci. Med. Sci. 2004, 59, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Lobo, A.; López-Antón, R.; De-La-Cámara, C.; Quintanilla, M.Á.; Campayo, A.; Saz, P. Non-cognitive psychopathological symptoms associated with incident mild cognitive impairment and dementia, Alzheimer’s type. Neurotox. Res. 2008, 14, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Kotwal, A.A.; Kim, J.; Waite, L.; Dale, W. Social Function and Cognitive Status: Results from a US Nationally Representative Survey of Older Adults. J. Gen. Intern. Med. 2016, 31, 854–862. [Google Scholar] [CrossRef] [PubMed]
- Kryscio, R.J.; Schmitt, F.A.; Salazar, J.C.; Mendiondo, M.S.; Markesbery, W.R. Risk factors for transitions from normal to mild cognitive impairment and dementia. Neurology 2006, 66, 828–832. [Google Scholar] [CrossRef] [PubMed]
- Verghese, J.; Cuiling, W.; Katz, M.J.; Sanders, A.; Lipton, R.B. Leisure activities and risk of vascular cognitive impairment in older adults. J. Geriatr. Psychiatry Neurol. 2009, 22, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Verghese, J.; LeValley, A.; Derby, C.; Kuslansky, G.; Katz, M.; Hall, C.; Buschke, H.; Lipton, R.B. Leisure activities and the risk of amnestic mild cognitive impairment in the elderly. Neurology 2006, 66, 821–827. [Google Scholar] [CrossRef] [PubMed]
- Lojo-Seoane, C.; Facal, D.; Juncos-Rabadán, O. ¿Previene la actividad intelectual el deterioro cognitivo? Relaciones entre reserva cognitiva y deterioro cognitivo ligero. Revista Española de Geriatría y Gerontología 2012, 47, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Ma, C.; Zhang, J.; Liang, Y.; Chen, Y.; Chen, K.; Wang, J.; Zhang, Z.; Wang, Y. Prevalence of and Potential Risk Factors for Mild Cognitive Impairment in Community-Dwelling Residents of Beijing. J. Am. Geriatr. Soc. 2013, 61, 2111–2119. [Google Scholar] [CrossRef] [PubMed]
- Ball, K.; Berch, D.B.; Helmers, K.F.; Jobe, J.B.; Leveck, M.D.; Marsiske, M.; Morris, J.N.; Rebok, G.W.; Smith, D.M.; Tennstedt, S.L. Effects of cognitive training interventions with older adults: A randomized controlled trial. JAMA 2002, 288, 2271–2281. [Google Scholar] [CrossRef] [PubMed]
- Doi, T.; Verghese, J.; Makizako, H.; Tsutsumimoto, K.; Hotta, R.; Nakakubo, S.; Suzuki, T.; Shimada, H. Effects of Cognitive Leisure Activity on Cognition in Mild Cognitive Impairment: Results of a Randomized Controlled Trial. J. Am. Med. Dir. Assoc. 2017, 18, 686–691. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R.S.; Krueger, K.R.; Arnold, S.E.; Schneider, J.A.; Kelly, J.F.; Barnes, L.L.; Tang, Y.; Bennett, D.A. Loneliness and risk of Alzheimer disease. Arch. Gen. Psychiatry 2007, 64, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Bediou, B.; Ryff, I.; Mercier, B.; Milliery, M.; Henaff, M.; d’Amato, T.; Bonnefoy, M.; Vighetto, A.; Krolak-Salmon, P. Impaired social cognition in mild Alzheimer disease. J. Geriatr. Psychiatry Neurol. 2009, 22, 130–140. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Antón, R.; Santabarbara, J.; De-la-Cámara, C.; Gracia-García, P.; Lobo, E.; Marcos, G.; Pirez, G.; Saz, P.; Haro, J.; Rodríguez-Mañas, L. Mild cognitive impairment diagnosed with the new DSM-5 criteria: Prevalence and associations with non-cognitive psychopathology. Acta Psychiatr. Scand. 2015, 131, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Tang, E.Y.; Brayne, C.; Albanese, E.; Stephan, B.C. Mild cognitive impairment definitions: More evolution than revolution. Spec. Rep. Neurodegener. Dis. Manag. 2015, 5, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Portet, F.; Ousset, P.J.; Visser, P.J.; Frisoni, G.B.; Nobili, F.; Scheltens, P.; Vellas, B.; Touchon, J. MCI Working Group of the European Consortium on Alzheimer’s Disease (EADC) Mild cognitive impairment (MCI) in medical practice: A critical review of the concept and new diagnostic procedure. Report of the MCI Working Group of the European Consortium on Alzheimer’s Disease. J. Neurol. Neurosurg. Psychiatry 2006, 77, 714–718. [Google Scholar] [PubMed]
- Sachs-Ericsson, N.; Blazer, D.G. The new DSM-5 diagnosis of mild neurocognitive disorder and its relation to research in mild cognitive impairment. Aging Ment. Health 2015, 19, 2–12. [Google Scholar] [CrossRef] [PubMed]
- Yoo, S.W.; Han, C.E.; Shin, J.S.; Seo, S.W.; Na, D.L.; Kaiser, M.; Jeong, Y.; Seong, J. A network flow-based analysis of cognitive reserve in normal ageing and Alzheimer’s disease. Sci. Rep. 2015, 5, 10057. [Google Scholar] [CrossRef] [PubMed]
- Lojo-Seoane, C.; Facal, D.; Guàrdia-Olmos, J.; Juncos-Rabadán, O. Structural model for estimating the influence of cognitive reserve on cognitive performance in adults with subjective memory complaints. Arch. Clin. Neuropsychol. 2014, 29, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Wang, H.; Sun, Q.; Yao, H.; Keegan, A.P.; Mullan, M.; Wilson, J.; Lista, S.; Leyhe, T.; Laske, C. Increased Plasma Beta-Secretase 1 May Predict Conversion to Alzheimer’s Disease Dementia in Individuals With Mild Cognitive Impairment. Biol. Psychiatry 2017. [Google Scholar] [CrossRef] [PubMed]
| References | Cognitive Processes Evaluated | MCI Subtypes |
|---|---|---|
| Petersen [2] | Episodic memory | aMCI |
| Petersen et al. [5] | Episodic memory | aMCI |
| Language | Single non-memory domain MCI | |
| Executive function | Multiple domains, slight impairment MCI | |
| Visuospatial skills | ||
| Petersen [6] | Episodic memory | Single domain aMCI |
| Petersen & Negash [7] | Language | Multiple domain aMCI |
| Executive function | Single domain naMCI | |
| Visuospatial skills | Multiple domain naMCI | |
| Edmonds et al. [14] | Memory | aMCI |
| Language | Dysnomic MCI | |
| Attention | Dysexecutive MCI) | |
| Executive function | Cluster-Derived Normal(within normal limits on cognitive testing) | |
| Eppig et al. [15] | Episodic memory | aMCI |
| Libon et al. [16] | Language | dMCI (executive and processing speed deficits) |
| Executive functioning | mx MCI (memory and language deficits) | |
| Processing speed | ||
| Visuo-construction | ||
| Rosenberg et al. [17] | Episodic memory recall | aMCI |
| Language | exMCI | |
| Attention | Both aMCI and exMCI | |
| Executive function | Neither aMCI nor exMCI | |
| Visuospatial function | ||
| Mansbach et al. [18] | Verbal memory | aMCI |
| Executive control functions | Executive MCI | |
| Attentional capacity | Multi-domain MCI | |
| Undifferentiated MCI | ||
| Albert et al. [20] | Episodic verbal memory | aMCI |
| Saunders & Summers [22] | Language | |
| Putcha & Tremont [24] | Executive function | |
| Attention | naMCI | |
| Working memory |
| Cognitive Domains | Tests |
|---|---|
| Episodic memory and learning | Verbal Learning Test Spain Complutense (Test de Aprendizaje Verbal España Complutense—TAVEC) [66] |
| The Rey-Osterrieth complex figure [69] | |
| Working Memory | Inverse Digits, Barcelona Test [67] |
| Language | Semantic and phonemic fluency, Barcelona Test [67] |
| Attention and Executive function | Direct Digits, Test Barcelona [67] |
| Comprehensive Trail-Making Test (CTMT), A and B [68] | |
| Alternating graphs and loops, Barcelona Test [67] | |
| Constructive praxis | The Rey-Osterrieth complex figure [69] |
| Praxis constructive graphics, Barcelona Test [67] | |
| Ideomotor praxis | Mimicking the use of objects and Symbolic gestures of communication, Barcelona Test [67] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Díaz-Mardomingo, M.D.C.; García-Herranz, S.; Rodríguez-Fernández, R.; Venero, C.; Peraita, H. Problems in Classifying Mild Cognitive Impairment (MCI): One or Multiple Syndromes? Brain Sci. 2017, 7, 111. https://doi.org/10.3390/brainsci7090111
Díaz-Mardomingo MDC, García-Herranz S, Rodríguez-Fernández R, Venero C, Peraita H. Problems in Classifying Mild Cognitive Impairment (MCI): One or Multiple Syndromes? Brain Sciences. 2017; 7(9):111. https://doi.org/10.3390/brainsci7090111
Chicago/Turabian StyleDíaz-Mardomingo, María Del Carmen, Sara García-Herranz, Raquel Rodríguez-Fernández, César Venero, and Herminia Peraita. 2017. "Problems in Classifying Mild Cognitive Impairment (MCI): One or Multiple Syndromes?" Brain Sciences 7, no. 9: 111. https://doi.org/10.3390/brainsci7090111
APA StyleDíaz-Mardomingo, M. D. C., García-Herranz, S., Rodríguez-Fernández, R., Venero, C., & Peraita, H. (2017). Problems in Classifying Mild Cognitive Impairment (MCI): One or Multiple Syndromes? Brain Sciences, 7(9), 111. https://doi.org/10.3390/brainsci7090111





