Investigation of Sex Differences in the Microglial Response to Binge Ethanol and Exercise
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Alcohol Administration Paradigm
2.3. Voluntary Exercise
2.4. Tissue Processing
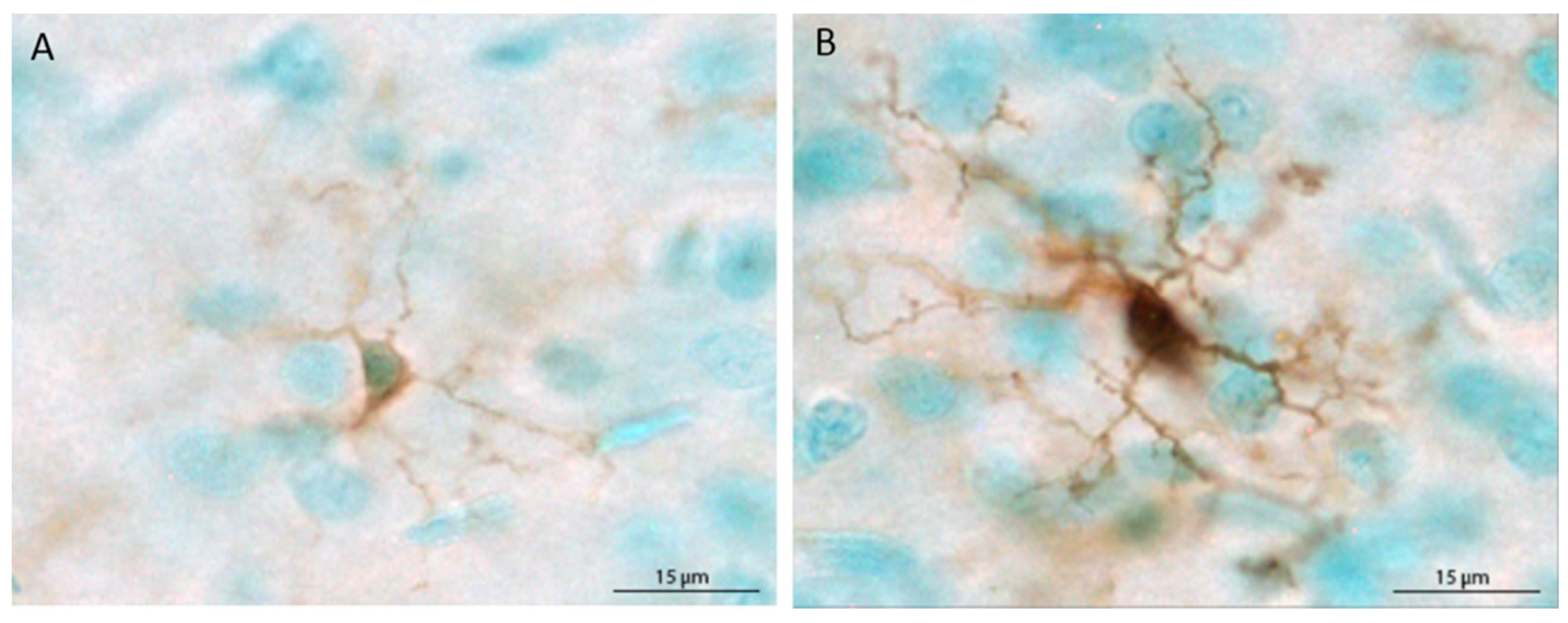
2.5. Quantification of Microglia
2.6. Statistical Analyses
3. Results
3.1. Intoxication and Exercise Data
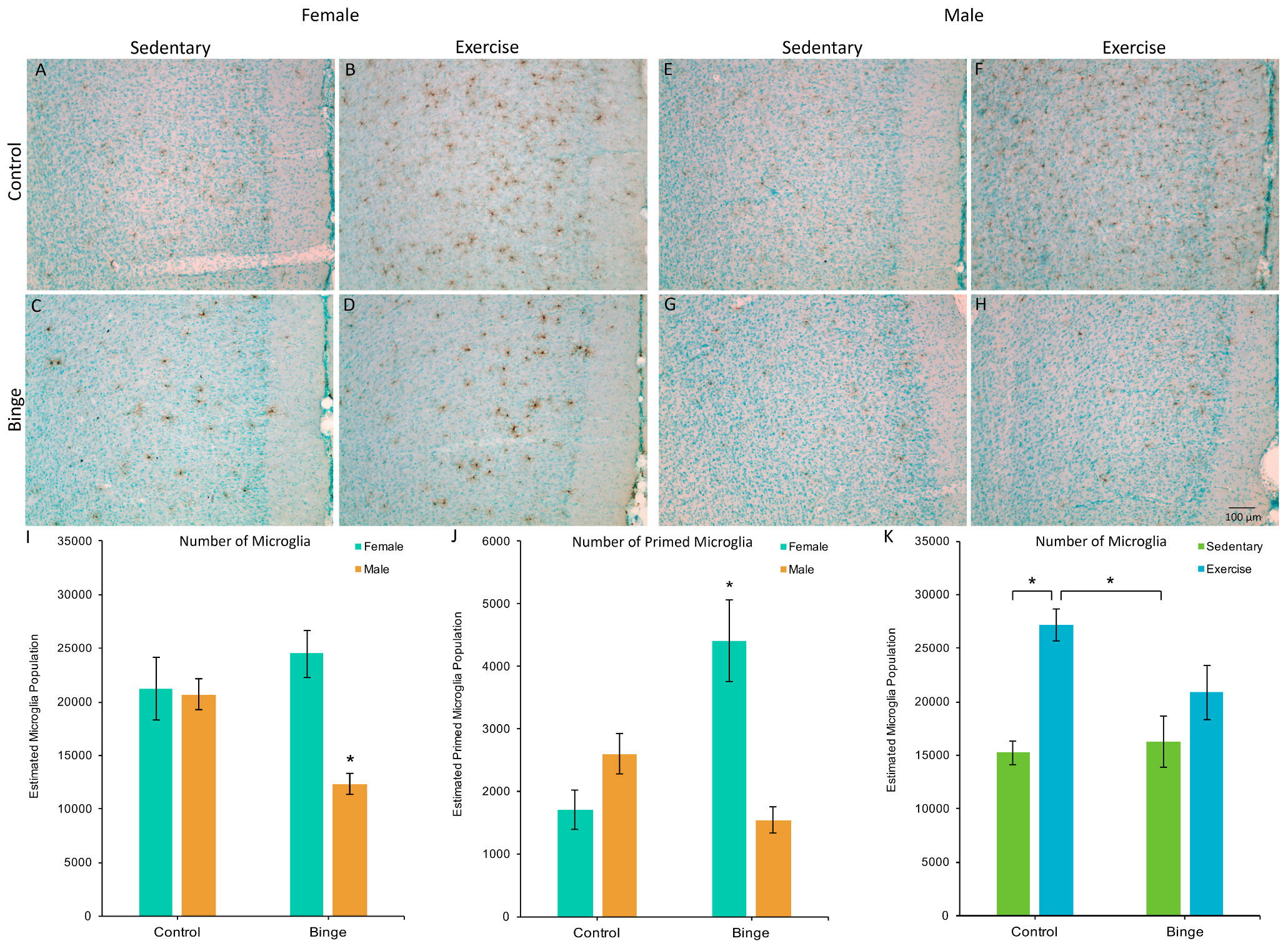
3.2. Microglia Number and Morphology in the Medial Prefrontal Cortex
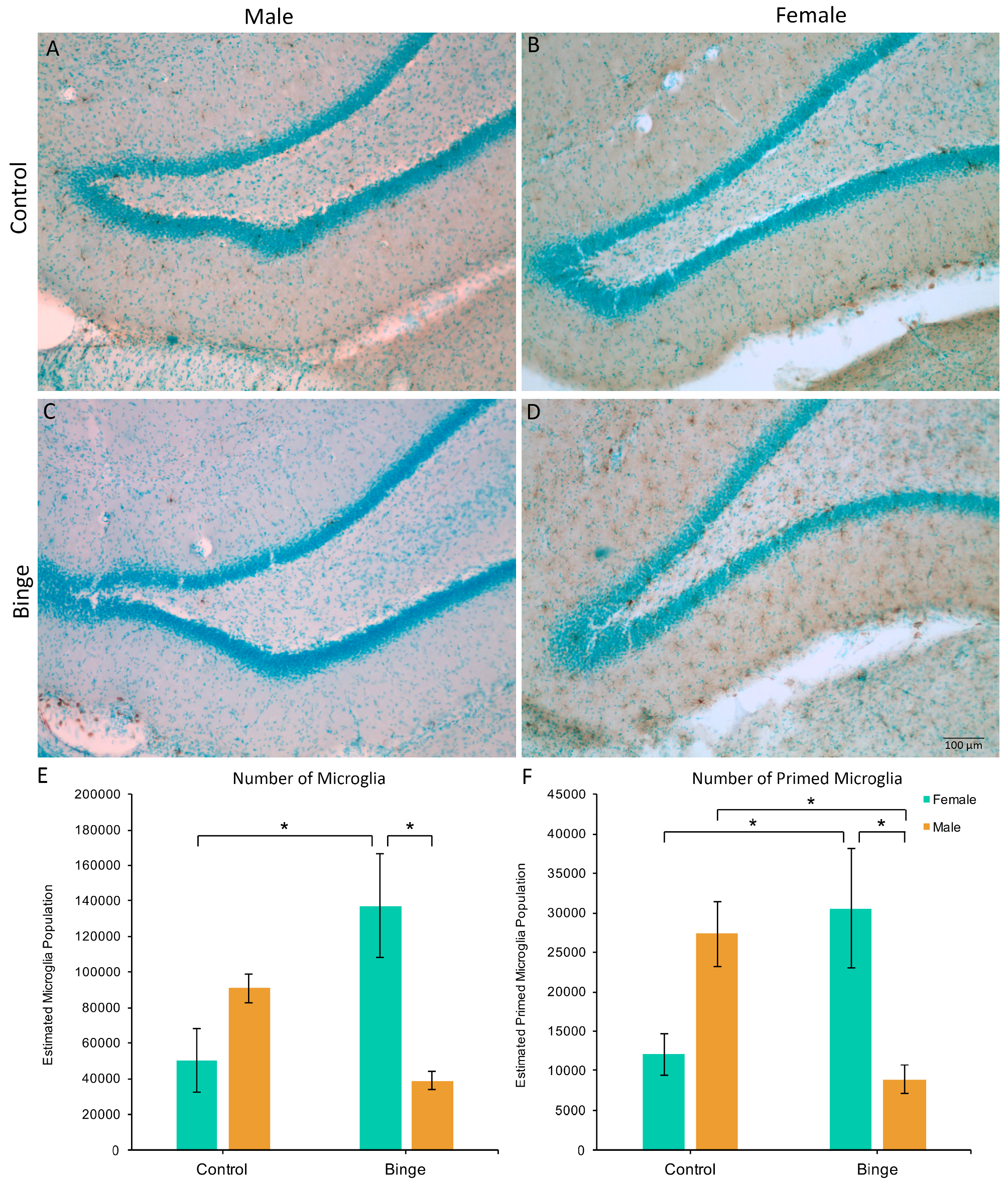
3.3. Microglia Number and Morphology in the Hippocampus
3.4. MHC II Expression in the Medial Prefrontal Cortex and Hippocampus
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- York, J.L.; Welte, J.W. Gender comparisons of alcohol consumption in alcoholic and nonalcoholic populations. J. Stud. Alcohol 1994, 55, 743–750. [Google Scholar] [CrossRef] [PubMed]
- Kessler, R.C.; McGonagle, K.A.; Zhao, S.; Nelson, C.B.; Huges, M.; Eshleman, S.; Wittchen, H.U.; Kendler, K.S. Lifetime and 12-month prevalence of DSM-III-R psychiatric disorders in the United States. Results from the National Comorbidity Survey. Arch. Gen. Psychiatry 1994, 51, 8–19. [Google Scholar] [CrossRef] [PubMed]
- Smith, W.B.; Weisner, C. Women and alcohol problems: A critical analysis of the literature and unanswered questions. Alcohol. Clin. Exp. Res. 2000, 24, 1320–1321. [Google Scholar] [CrossRef] [PubMed]
- Walter, H.; Dvorak, A.; Gutierrez, K.; Zitterl, W.; Lesch, O.M. Gender differences: Does alcohol affect females more than males? Neuropsychopharmacol. Hung. 2005, 7, 78–82. [Google Scholar] [PubMed]
- Ashley, M.J.; Olin, J.S.; le Riche, W.H.; Kornaczewski, A.; Schmidt, W.; Rankin, J.G. Morbidity in alcoholics. Evidence for accelerated development of physical disease in women. Arch. Intern. Med. 1977, 137, 883–887. [Google Scholar] [CrossRef] [PubMed]
- Hommer, D.W. Male and female sensitivity to alcohol-induced brain damage. Alcohol Res. Health 2003, 27, 181–185. [Google Scholar] [PubMed]
- Prendergast, M.A. Do women possess a unique susceptibility to the neurotoxic effects of alcohol? J. Am. Med. Womens Assoc. 2004, 59, 225–227. [Google Scholar]
- Sharrett-Field, L.; Butler, T.R.; Reynolds, A.R.; Berry, J.N.; Prendergast, M.A. Sex differences in neuroadaptation to alcohol and withdrawal neurotoxicity. Pflugers Arch. 2013, 465, 643–654. [Google Scholar] [CrossRef] [PubMed]
- Hommer, D.; Momenan, R.; Kaiser, E.; Rawlings, R. Evidence for a gender-related effect of alcoholism on brain volumes. Am. J. Psychiatry 2001, 158, 198–204. [Google Scholar] [CrossRef] [PubMed]
- Hommer, D.; Momenan, R.; Rawlings, R.; Ragan, P.; Williams, W.; Rio, D.; Eckardt, M. Decreased corpus callosum size among alcoholic women. Arch. Neurol. 1996, 53, 359–363. [Google Scholar] [CrossRef] [PubMed]
- Agartz, I.; Momenan, R.; Rawlings, R.R.; Kerich, M.J.; Hommer, D.W. Hippocampal volume in patients with alcohol dependence. Arch. Gen. Psychiatry 1999, 56, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Agartz, I.; Shoaf, S.; Rawlings, R.R.; Momenan, R.; Hommer, D.W. CSF monoamine metabolites and MRI brain volumes in alcohol dependence. Psychiatry Res. 2003, 122, 21–35. [Google Scholar] [CrossRef]
- Sullivan, E.V.; Rohlfing, T.; Pfefferbaum, A. Pontocerebellar volume deficits and ataxia in alcoholic men and women: No evidence for “telescoping”. Psychopharmacology 2010, 208, 279–290. [Google Scholar] [CrossRef] [PubMed]
- Demirakca, T.; Ende, G.; Kammerer, N.; Welzel-Marquez, H.; Hermann, D.; Heinz, A.; Mann, K. Effects of alcoholism and continued abstinence on brain volumes in both genders. Alcohol. Clin. Exp. Res. 2011, 35, 1678–1685. [Google Scholar] [CrossRef] [PubMed]
- Maynard, M.E.; Barton, E.A.; Robinson, C.R.; Wooden, J.I.; Leasure, J.L. Sex differences in hippocampal damage, cognitive impariment and trophic factor expression in an animal model of an Alcohol Use Disorder. Brain Struct. Funct. 2017. [Google Scholar] [CrossRef]
- Doyle, H.H.; Murphy, A.Z. Sex differences in innate immunity and its impact on opioid pharmacology. J. Neurosci. Res. 2017, 95, 487–499. [Google Scholar] [CrossRef] [PubMed]
- Acaz-Fonseca, E.; Duran, J.C.; Carrero, P.; Garcia-Segura, L.M.; Arevalo, M.A. Sex differences in glia reactivity after cortical brain injury. Glia 2015, 63, 1966–1981. [Google Scholar] [CrossRef] [PubMed]
- Ackerman, L.S. Sex hormones and the genesis of autoimmunity. Arch. Dermatol. 2006, 142, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Bouman, A.; Heineman, M.J.; Faas, M.M. Sex hormones and the immune response in humans. Hum. Reprod. Update 2005, 11, 411–423. [Google Scholar] [CrossRef] [PubMed]
- Pennell, L.M.; Galligan, C.L.; Fish, E.N. Sex affects immunity. J. Autoimmun. 2012, 38, J282–J291. [Google Scholar] [CrossRef] [PubMed]
- Andersen, K.; Launer, E.L.; Dewey, R.M.; Letenneur, M.L.; Ott, M.A.; Copeland, M.J.; Dartigues, M.; Kragh-Sorensen, M.P.; Baldereschi, M.M.; Brayne, M.C.; et al. Gender differences in the incidence of AD and vascular dementia: The EURODEM Studies. EURODEM Incidence Research Group. Neurology 1999, 53, 1992–1997. [Google Scholar] [CrossRef] [PubMed]
- Hebert, L.E.; Weuve, J.; Scherr, P.A.; Evans, D.A. Alzheimer disease in the United States (2010–2050) estimated using the 2010 census. Neurology 2013, 80, 1778–1783. [Google Scholar] [CrossRef] [PubMed]
- Koch-Henriksen, N.; Sorensen, P.S. The changing demographic pattern of multiple sclerosis epidemiology. Lancet Neurol. 2010, 9, 520–532. [Google Scholar] [CrossRef]
- Harbo, H.F.; Gold, R.; Tintoré, M. Sex and gender issues in multiple sclerosis. Ther. Adv. Neurol. Disord. 2013, 6, 237–248. [Google Scholar] [CrossRef] [PubMed]
- Buse, D.C.; Loder, E.W.; Gorman, J.A.; Stewart, W.F.; Reed, M.L.; Fanning, K.M.; Serrano, D.; Lipton, R.B. Sex differences in the prevalence, symptoms, and associated features of migraine, probable migraine and other severe headache: Results of the American Migraine Prevalence and Prevention (AMPP) Study. Headache 2013, 53, 1278–1299. [Google Scholar] [CrossRef] [PubMed]
- White, K.P.; Speechley, M.; Harth, M.; Ostbye, T. The London Fibromyalgia Epidemiology Study: The prevalence of fibromyalgia syndrome in London, Ontario. J. Rheumatol. 1999, 26, 1570–1576. [Google Scholar] [PubMed]
- Wolfe, F.; Ross, K.; Anderson, J.; Russell, I.J.; Hebert, L. The prevalence and characteristics of fibromyalgia in the general population. Arthritis Rheum. 1995, 38, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Vincent, A.; Lahr, B.D.; Wolfe, F.; Clauw, D.J.; Whipple, M.O.; Oh, T.H.; Barton, D.L.; St. Sauver, J. Prevalence of fibromyalgia: A population-based study in Olmsted County, Minnesota, utilizing the Rochester Epidemiology Project. Arthritis Care Res. 2013, 65, 786–792. [Google Scholar] [CrossRef] [PubMed]
- Plotnikoff, R.; Karunamuni, N.; Lytvyak, E.; Penfold, C.; Schopflocher, D.; Imayama, I.; Johnson, S.T.; Raine, K. Osteoarthritis prevalence and modifiable factors: A population study. BMC Public Health 2015, 15, 1195. [Google Scholar] [CrossRef] [PubMed]
- Lenz, K.M.; McCarthy, M.M. A starring role for microglia in brain sex differences. Neuroscientist 2015, 21, 306–321. [Google Scholar] [CrossRef] [PubMed]
- Nelson, L.H.; Warden, S.; Lenz, K.M. Sex differences in microglial phagocytosis in the neonatal hippocampus. Brain Behav. Immun. 2017, 64, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, J.M.; Sholar, P.W.; Bilbo, S.D. Sex differences in microglial colonization of the developing rat brain. J. Neurochem. 2012, 120, 948–963. [Google Scholar] [CrossRef] [PubMed]
- Doyle, H.H.; Eidson, L.N.; Sinkiewicz, D.M.; Murphy, A.Z. Sex differences in microglia activity within the periaqueductal gray of the rat: A potential mechanism driving the dimorphic effects of morphine. J. Neurosci. 2017, 37, 3202–3214. [Google Scholar] [CrossRef] [PubMed]
- Bollinger, J.L.; Bergeon Burns, C.M.; Wellman, C.L. Differential effects of stress on microglial cell activation in male and female medial prefrontal cortex. Brain Behav. Immun. 2016, 52, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Perry, V.H.; Holmes, C. Microglial priming in neurodegenerative disease. Nat. Rev. Neurol. 2014, 10, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Chastain, L.G.; Sarkar, D.K. Role of microglia in regulation of ethanol neurotoxic action. Int. Rev. Neurobiol. 2014, 118, 81–103. [Google Scholar] [PubMed]
- Zhao, Y.N.; Wang, F.; Fan, Y.X.; Ping, G.F.; Yang, J.Y.; Wu, C.F. Activated microglia are implicated in cognitive deficits, neuronal death, and successful recovery following intermittent ethanol exposure. Behav. Brain Res. 2013, 236, 270–282. [Google Scholar] [CrossRef] [PubMed]
- Marshall, S.A.; Geil, C.R.; Nixon, K. Prior Binge Ethanol Exposure Potentiates the Microglial Response in a Model of Alcohol-Induced Neurodegeneration. Brain Sci. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Qin, L.; Crews, F.T. Chronic ethanol increases systemic TLR3 agonist-induced neuroinflammation and neurodegeneration. J. Neuroinflamm. 2012, 9, 130. [Google Scholar] [CrossRef] [PubMed]
- Barton, E.A.; Lu, Y.; Megjhani, M.; Maynard, M.E.; Kulkarni, P.M.; Roysam, B.; Leasure, J.L. Binge alcohol alters exercise-driven neuroplasticity. Neuroscience 2017, 343, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Kubota, M.; Nakazaki, S.; Hirai, S.; Saeki, N.; Yamaura, A.; Kusaka, T. Alcohol consumption and frontal lobe shrinkage: Study of 1432 non-alcoholic subjects. J. Neurol. Neurosurg. Psychiatry 2001, 71, 104–106. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Tian, F.; Zhang, H.; Zeng, J.; Chen, T.; Wang, S.; Jia, Z.; Gong, Q. Cortical and subcortical gray matter shrinkage in alcohol-use disorders: A voxel-based meta-analysis. Neurosci. Biobehav. Rev. 2016, 66, 92–103. [Google Scholar] [CrossRef] [PubMed]
- Mandyam, C.D.; Wee, S.; Eisch, A.J.; Richardson, H.K.; Koob, G.F. Methamphetamine self-administration and voluntary exercise have opposing effects on medial prefrontal cortex gliogenesis. J. Neurosci. 2007, 27, 11442–11450. [Google Scholar] [CrossRef] [PubMed]
- Brockett, A.T.; LaMarca, E.A.; Gould, E. Physical exercise enhances cognitive flexibility as well as astrocytic and synaptic markers in the medial prefrontal cortex. PLoS ONE 2015, 10, e0124859. [Google Scholar] [CrossRef] [PubMed]
- Aloisi, F. Immune function of microglia. Glia 2001, 36, 165–179. [Google Scholar] [CrossRef] [PubMed]
- Lull, M.E.; Block, M.L. Microglial activation and chronic neurodegeneration. Neurotherapeutics 2010, 7, 354–365. [Google Scholar] [CrossRef] [PubMed]
- Henry, C.J.; Huang, Y.; Wynne, A.M.; Godbout, J.P. Peripheral lipopolysaccharide (LPS) challenge promotes microglial hyperactivity in aged mice that is associated with exaggerated induction of both pro-inflammatory IL-1beta and anti-inflammatory IL-10 cytokines. Brain Behav. Immun. 2009, 23, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Norden, D.M.; Muccigrosso, M.M.; Godbout, J.P. Microglial priming and enhanced reactivity to secondary insult in aging, and traumatic CNS injury, and neurodegenerative disease. Neuropharmacology 2015, 96 Pt A, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Fenn, A.M.; Gensel, J.C.; Huang, Y.; Popovich, P.G.; Lifshitz, J.; Godbout, J.P. Immune activation promotes depression 1 month after diffuse brain injury: A role for primed microglia. Biol. Psychiatry 2014, 76, 575–584. [Google Scholar] [CrossRef] [PubMed]
- Loane, D.J.; Kumar, A.; Stoica, B.A.; Cabatbat, R.; Faden, A.I. Progressive neurodegeneration after experimental brain trauma: Association with chronic microglial activation. J. Neuropathol. Exp. Neurol. 2014, 73, 14–29. [Google Scholar] [CrossRef] [PubMed]
- Palin, K.; Cunningham, C.; Forse, P.; Perry, V.H.; Platt, N. Systemic inflammation switches the inflammatory cytokine profile in CNS Wallerian degeneration. Neurobiol. Dis. 2008, 30, 19–29. [Google Scholar] [CrossRef] [PubMed]
- VanGuilder, H.D.; Bixler, G.V.; Brucklacher, R.M.; Farley, J.A.; Yan, H.; Warrington, J.P.; Sonntag, W.E.; Freeman, W.M. Concurrent hippocampal induction of MHC II pathway components and glial activation with advanced aging is not correlated with cognitive impairment. J. Neuroinflamm. 2011, 8, 138. [Google Scholar] [CrossRef] [PubMed]
- Majchrowicz, E. Induction of physical dependence upon ethanol and the associated behavioral changes in rats. Psychopharmacologia 1975, 43, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Paxinos, G.; Watson, C. The Rat Brain in Stereotaxic Coordinates; Academic Press: San Diego, CA, USA, 1988. [Google Scholar]
- Wolf, S.A.; Boddeke, H.W.G.M.; Kettenmann, H. Microglia in Physiology and Disease. Annu. Rev. Physiol. 2017, 79, 619–643. [Google Scholar] [CrossRef] [PubMed]
- Alfonso-Loeches, S.; Pascual, M.; Guerri, C. Gender differences in alcohol-induced neurotoxicity and brain damage. Toxicology 2013, 311, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, C.J.; Hashimoto, J.G.; Roberts, M.L.; Bloom, S.H.; Andrew, M.R.; Wiren, K.M. Astrocyte dysfunction induced by alcohol in females but not males. Brain Pathol. 2016, 26, 433–451. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, C.J.; Hashimoto, J.G.; Roberts, M.L.; Bloom, S.H.; Beard, D.K.; Wiren, K.M. Females uniquely vulnerable to alcohol-induced neurotoxicity show altered glucocorticoid signaling. Brain Res. 2015, 1601, 102–116. [Google Scholar] [CrossRef] [PubMed]
- Nixon, K.; Kimm, D.H.; Potts, E.N.; He, J.; Crews, F.T. Distinct cell proliferation events during abstinence after alcohol dependence: Microglia proliferation precedes neurogenesis. Neurobiol. Dis. 2008, 31, 218–229. [Google Scholar] [CrossRef] [PubMed]
- Aguzzi, A.; Barres, B.A.; Bennett, M.L. Microglia: Scapegoat, saboteur, or something else? Science 2013, 339, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Streit, W.J. Microglia as neuroprotective, immunocompetent cells of the CNS. Glia 2002, 40, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Marshall, S.A.; McClain, J.A.; Kelso, M.L.; Hopkins, D.M.; Pauly, J.R.; Nixon, K. Microglial activation is not equivalent to neuroinflammation in alcohol-induced neurodegeneration: The importance of microglia phenotype. Neurobiol. Dis. 2013, 54, 239–251. [Google Scholar] [CrossRef] [PubMed]
- McClain, J.A.; Morris, S.A.; Deeny, M.A.; Marshall, S.A.; Hayes, D.M.; Kiser, Z.M.; Nixon, K. Adolescent binge alcohol exposure induces long-lasting partial activation of microglia. Brain Behav. Immun. 2011, 25 (Suppl. S1), S120–S128. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, V.; Moore, V.; Brunzell, D.; Lynch, W. Sex differences in the effect of wheel running on subsequent nicotine-seeking in a rat adolescent-onset self-administration model. Psychopharmacology 2014, 231, 1753–1762. [Google Scholar] [CrossRef] [PubMed]
- Eikelboom, R.; Mills, R. A microanalysis of wheel running in male and female rats. Physiol. Behav. 1988, 43, 625–630. [Google Scholar] [CrossRef]
- Giesen, E.S.; Deimel, H.; Bloch, W. Clinical exercise interventions in alcohol use disorders: A systematic review. J. Subst. Abuse Treat. 2015, 52, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Hallgren, M.; Vancampfort, D.; Giesen, E.S.; Lundin, A.; Stubbs, B. Exercise as treatment for alcohol use disorders: Systematic review and meta-analysis. Br. J. Sports Med. 2017, 51, 1058–1064. [Google Scholar] [CrossRef] [PubMed]
- Stoutenberg, M.; Rethorst, C.D.; Lawson, O.; Read, J.P. Exercise training—A beneficial intervention in the treatment of alcohol use disorders? Drug Alcohol. Depend. 2016, 160, 2–11. [Google Scholar] [CrossRef] [PubMed]
- Robinson, D.L.; Brunner, L.J.; Gonzales, R.A. Effect of gender and estrous cycle on the pharmacokinetics of ethanol in the rat brain. Alcohol. Clin. Exp. Res. 2002, 26, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Priddy, B.M.; Carmack, S.A.; Thomas, L.C.; Vendruscolo, J.C.M.; Koob, G.F.; Vendruscolo, L.F. Sex, strain, and estrous cycle influences on alcohol drinking in rats. Pharmacol. Biochem. Behav. 2017, 152, 61–67. [Google Scholar] [CrossRef] [PubMed]

| Sex | Group | Timeline | ||
|---|---|---|---|---|
| 4 Days | 6 Days | 11 Days | ||
| Females | CON | Control Diet | Rest | Sedentary |
| CEX | Exercise | |||
| BIN | Binge Diet | Rest | Sedentary | |
| BEX | Exercise | |||
| Males | CON | Control Diet | Rest | Sedentary |
| CEX | Exercise | |||
| BIN | Binge Diet | Rest | Sedentary | |
| BEX | Exercise | |||
| Intoxication Score | Indications | Dose (g/kg) |
|---|---|---|
| 0 | Normal rat | 5 |
| 1 | Hypoactive, mild ataxia | 4 |
| 2 | Ataxic, abdomen elevated | 3 |
| 3 | Ataxic with no abdomen elevation, delayed righting reflex | 2 |
| 4 | Loss of righting reflex, retains eye-blink reflex | 1 |
| 5 | Loss of righting reflex, loss of eye-blink reflex | 0 |
| Behavior Score | Indications | Behavior Score | Indications |
|---|---|---|---|
| 0 | Normal rat | 2.6 | General tremor |
| 1.0 | Hyperactivity | 3.0 | Head tremor |
| 1.4 | Tail tremor | 3.4 | Wet dog shakes |
| 1.6 | Tail spasticity | 3.6 | Chattering teeth |
| 2.0 | Caudal tremor | 3.8 | Convulsion |
| 2.4 | Splayed limbs | 4.0 | Death |
| Intoxication Behavior | p | Dose (g/kg/day) | p | BEC (mg/dL) | p | Median Withdrawal | p | Peak Withdrawal | p | |
|---|---|---|---|---|---|---|---|---|---|---|
| Male | 2.2 ± 0.1 | 0.01 | 2.9 ± 0.1 | <0.001 | 485.6 ± 9.7 | 0.15 | 1.05 ± 0.2 | 0.27 | 2.7 ± 0.2 | 0.84 |
| Female | 1.5 ± 0.2 | 3.5 ± 0.1 | 460.07 ± 14.1 | 1.4 ± 0.2 | 2.8 ± 0.09 |
| Average Distance (km/day) | p | Total Distance (km over 7 Days) | p | Peak Speed (km/h) | p | |
|---|---|---|---|---|---|---|
| Male | 1.1 ± 0.2 | <0.001 | 11.4 ± 2.06 | <0.001 | 16.2 ± 1.4 | 0.66 |
| Female | 2.7 ± 0.2 | 29.9 ± 2.7 | 17.1 ± 1.6 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barton, E.A.; Baker, C.; Leasure, J.L. Investigation of Sex Differences in the Microglial Response to Binge Ethanol and Exercise. Brain Sci. 2017, 7, 139. https://doi.org/10.3390/brainsci7100139
Barton EA, Baker C, Leasure JL. Investigation of Sex Differences in the Microglial Response to Binge Ethanol and Exercise. Brain Sciences. 2017; 7(10):139. https://doi.org/10.3390/brainsci7100139
Chicago/Turabian StyleBarton, Emily A., Cassandra Baker, and J. Leigh Leasure. 2017. "Investigation of Sex Differences in the Microglial Response to Binge Ethanol and Exercise" Brain Sciences 7, no. 10: 139. https://doi.org/10.3390/brainsci7100139
APA StyleBarton, E. A., Baker, C., & Leasure, J. L. (2017). Investigation of Sex Differences in the Microglial Response to Binge Ethanol and Exercise. Brain Sciences, 7(10), 139. https://doi.org/10.3390/brainsci7100139




