Caffeine Consuming Children and Adolescents Show Altered Sleep Behavior and Deep Sleep
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants and Scheduling
2.2. Actigraphy, Sleep Diary and Questionnaires
2.3. Attention Task
2.4. Sleep Assessment
2.5. Statistics
3. Results
3.1. Demographic Characteristics
| Caffeine Consumers (n = 16) | Controls (n = 16) | |
|---|---|---|
| Age (years); [f, m] | 14.5 ± 0.4 (11.9–16.8); [15.1 ± 0.6, 14.0 ± 0.4] | 14.4 ± 0.4 (11.8–16.4); [15.0 ± 0.5, 13.8 ± 0.5] |
| Caffeine consumption (mg/kg/d) | 2.5 ± 0.4 * | 0.1 ± 0.0 * |
| Chronotype | 4.3 ± 0.2 | 3.8 ± 0.2 |
| Socio Economic Status, SES | 5.0 ± 0.5(5) | 3.8 ± 0.3(7) |
| Sex (f, m) | 8 f, 8 m | 8 f, 8 m |
| Tanner puberty scale; [f, m] | 7.7 ± 0.7(3); [8.4 ± 1.3, 7.9 ± 0.9] | 7.5 ± 0.6(2); [7.7 ± 1.1, 7.4 ± 0.7] |
| Weight (kg) | 52.3 ± 2.6 | 51.2 ± 3.7(6) |
3.2. Sleep Diary, Architecture, Subjective Records and Attention Task
| Data | Point in time | Sleep Variables | Caffeine Consumers (n = 16) | Controls (n = 16) | Estimated n to Reveal Group Difference |
|---|---|---|---|---|---|
| Diary | Week prior | Rise time (hrs:min) | 07:29 ± 7.6 | 07:20 ± 7.5 ° | 170 |
| Bed time (Lights out) (hrs:min) | 23:14 ± 11.4 * | 22:17 ± 15.4 ° | 13 | ||
| Time in bed entire week (hrs:min) | 08:10 ± 0:13 * | 09:03 ± 0:16 ° | 18 | ||
| Time in bed week days (hrs:min) | 08:07 ± 0:19 | 08:28 ± 0:10 | 120 | ||
| Time in bed weekend days (hrs:min) | 09:04 ± 0:16 | 09:42 ± 0:27 | 81 | ||
| Difference time in bed weekend-days and time in bed weekdays (hrs:min) | 01:03 ± 0:19 | 01:18 ± 0:21 | 342 | ||
| Last caffeine consumption (hrs:min) | 16:37 ± 00:46 | n.a. | n.a. | ||
| Sleep architecture and continuity (EEG) | Night of assessment | Time in bed (hrs:min) | 07:48 ± 15.4 | 08:10 ± 16.7 | 157 |
| Total sleep time (hrs:min) | 06:59 ± 0:15 | 07:39 ± 0:18 | 44 | ||
| Sleep onset latency (min) | 20.3 ± 4.1 | 15.8 ± 1.8 | 108 | ||
| Wakefulness after sleep onset (min) | 32.1 ± 6.7 | 17.6 ± 2.5 | 26 | ||
| Stage 1 (% of total sleep time) | 6.8 ± 1.3 | 5.8 ± 0.6 | 221 | ||
| Stage 2 (% of total sleep time) | 48.6 ± 1.4 | 49.1 ± 1.9 | 2689 | ||
| Slow-wave sleep (% of total sleep time) | 27.2 ± 2.0 | 24.1 ± 1.8 | 91 | ||
| REM sleep (% of total sleep time) | 17.5 ± 1.0 | 20.2 ± 1.7 | 62 | ||
| Sleep efficiency (total sleep time/time in bed) (%) | 89.6 ± 1.9 | 93.7 ± 0.6 | 24 | ||
| Subjective reports/attention task | Morning after assessment | Reaction time in attention task (ms) | 283.6 ± 8.4 °° | 287.9 ± 7.9 °° | 733 |
| Subjectively reported morning tiredness (relaxed vs. tired) (%) | 51.9 ± 3.4 | 53.7 ± 5.8 | 1637 |
3.3. EEG Power
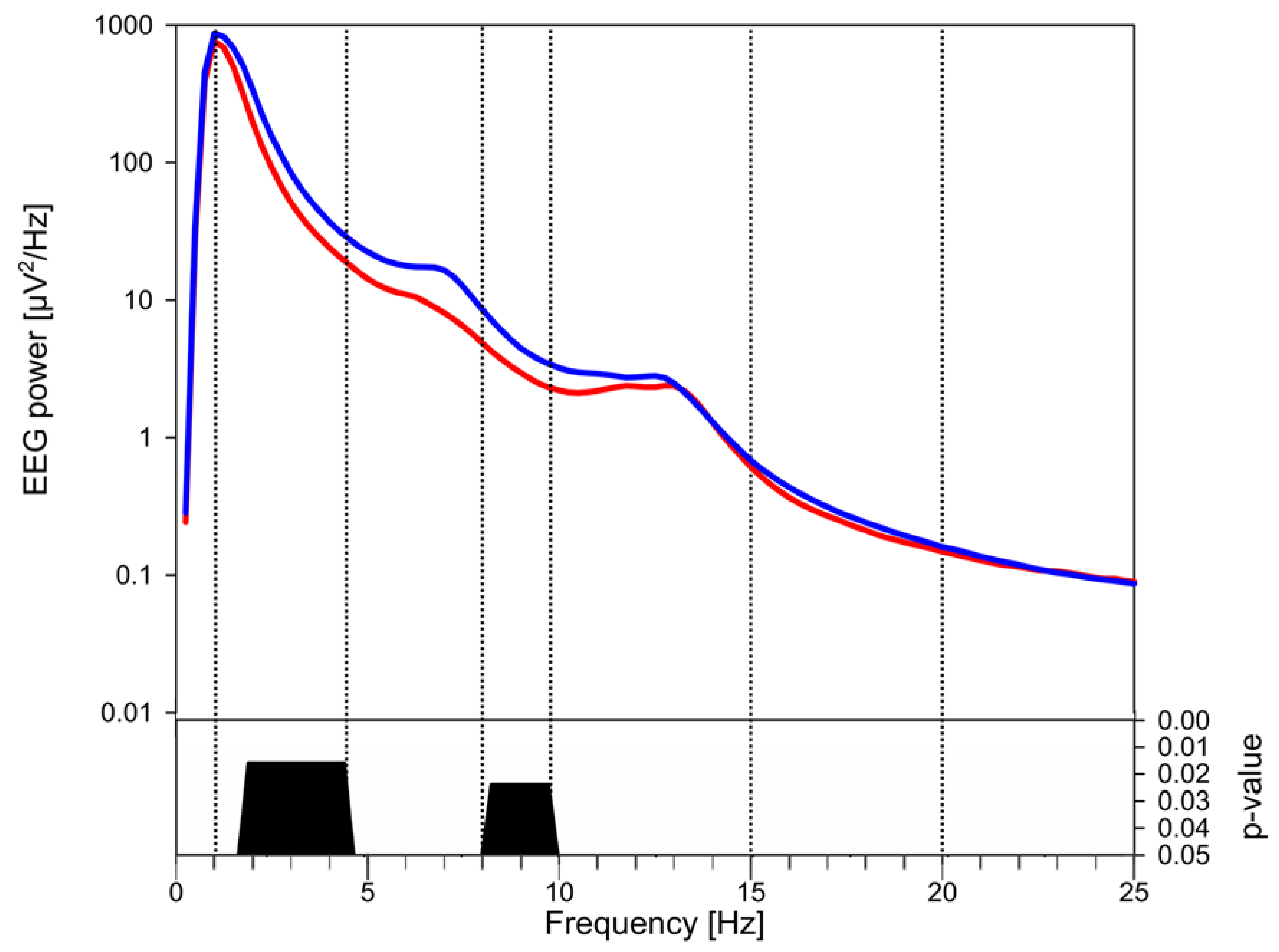
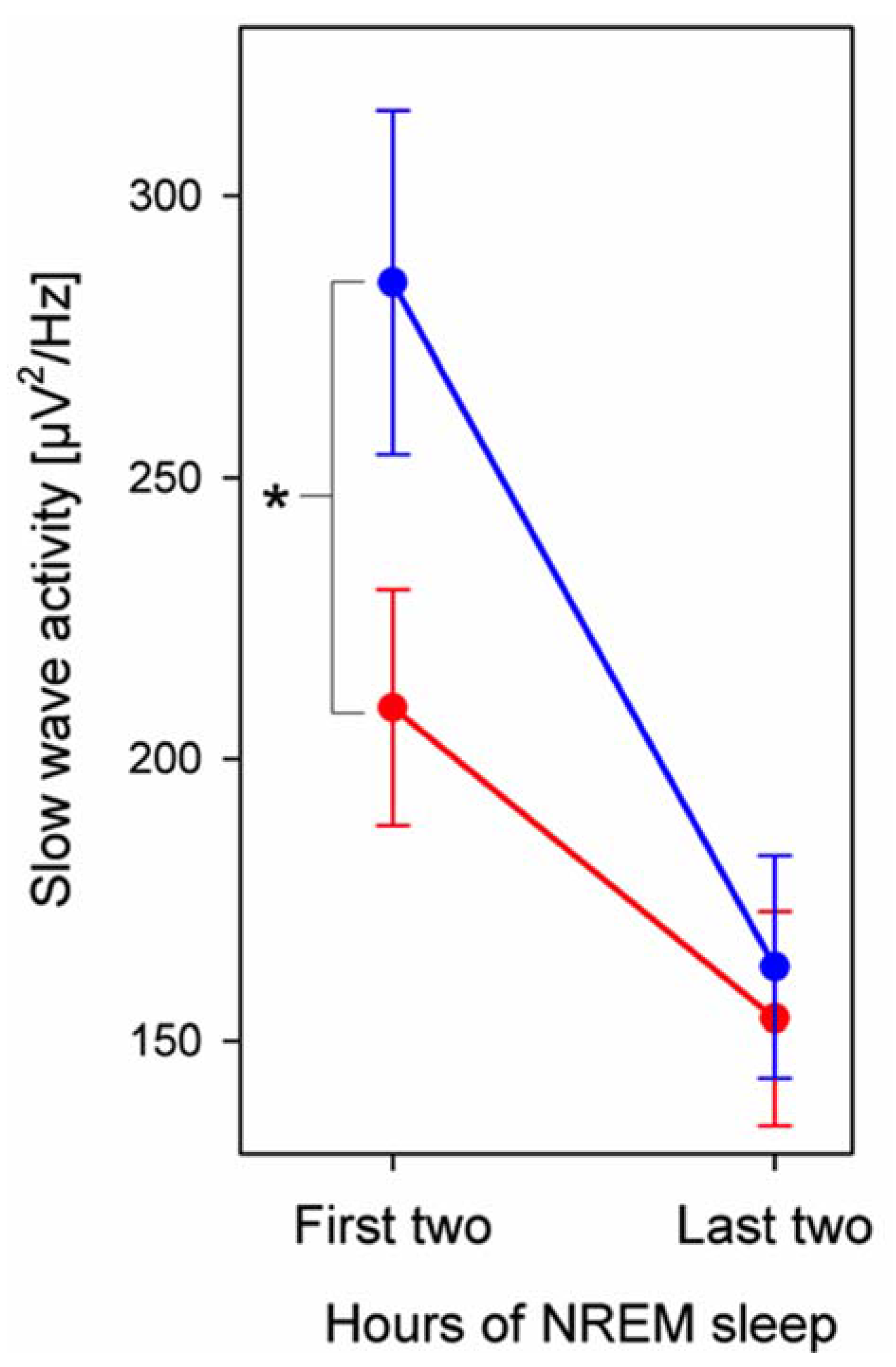
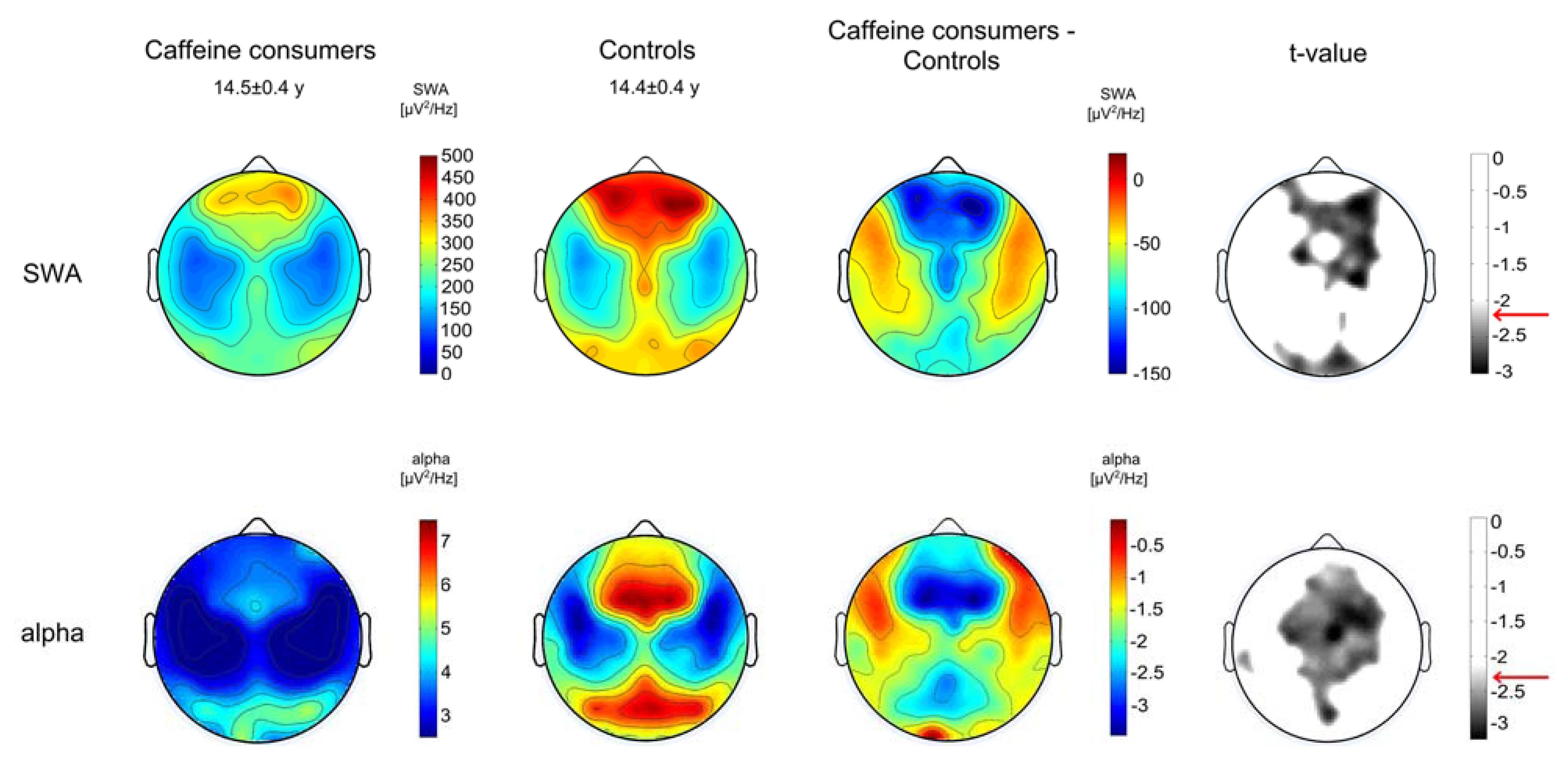
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fredholm, B.B.; Bättig, K.; Holmén, J.; Nehlig, A.; Zvartau, E.E. Actions of caffeine in the brain with special reference to factors that contribute to its widespread use. Pharmacol. Rev. 1999, 51, 83–133. [Google Scholar] [PubMed]
- Yoshimura, H. The potential of caffeine for functional modification from cortical synapses to neuron networks in the brain. Curr. Neuropharmacol. 2005, 3, 309–316. [Google Scholar] [CrossRef] [PubMed]
- McCarley, R.W. Neurobiology of REM and NREM sleep. Sleep Med. 2007, 8, 302–330. [Google Scholar] [CrossRef] [PubMed]
- Borbely, A.A. A two process model of sleep regulation. Hum. Neurobiol. 1982, 1, 195–204. [Google Scholar] [PubMed]
- Borbely, A.A.; Achermann, P. Sleep Homeostasis and Models of Sleep Regulation. In Principles and Practice of Sleep Medicine, 4th ed.; Elsevier Saunders: Philadelphia, USA, 2005; pp. 405–417. [Google Scholar]
- Vyazovskiy, V.V.; Olcese, U.; Lazimy, Y.M.; Faraguna, U.; Esser, S.K.; Williams, J.C.; Cirelli, C.; Tononi, G. Cortical firing and sleep homeostasis. Neuron 2009, 63, 865–878. [Google Scholar] [CrossRef] [PubMed]
- Landolt, H.P. Sleep homeostasis: A role for adenosine in humans? Biochem. Pharmacol. 2008, 75, 2070–2079. [Google Scholar] [CrossRef] [PubMed]
- Landolt, H.P.; Dijk, D.J.; Gaus, S.E.; Borbely, A.A. Caffeine reduces low-frequency delta activity in the human sleep EEG. Neuropsychopharmacology 1995, 12, 229–238. [Google Scholar] [CrossRef]
- Landolt, H.P.; Werth, E.; Borbely, A.A.; Dijk, D.J. Caffeine intake (200 mg) in the morning affects human sleep and EEG power spectra at night. Brain Res. 1995, 675, 67–74. [Google Scholar] [CrossRef]
- Karacan, I.; Thornby, J.I.; Anch, M.; Booth, G.H.; Williams, R.L.; Salis, P.J. Dose-related sleep disturbances induced by coffee and caffeine. Clin. Pharmacol. Ther. 1976, 20, 682–689. [Google Scholar] [PubMed]
- Spear, L.P. Adolescent neurodevelopment. J. Adolesc. Heal. 2013. [Google Scholar] [CrossRef] [PubMed]
- Temple, J.L. Caffeine use in children: What we know, what we have left to learn, and why we should worry. Neurosci. Biobehav. Rev. 2009, 33, 793–806. [Google Scholar] [CrossRef] [PubMed]
- Orbeta, R.L.; Overpeck, M.D.; Ramcharran, D.; Kogan, M.D.; Ledsky, R. High caffeine intake in adolescents: Associations with difficulty sleeping and feeling tired in the morning. J. Adolesc. Heal. 2006, 38, 451–453. [Google Scholar] [CrossRef] [PubMed]
- Pollak, C.P.; Bright, D. Caffeine consumption and weekly sleep patterns in US seventh-, eighth-, and ninth-graders. Pediatrics 2003, 111, 42–46. [Google Scholar] [CrossRef] [PubMed]
- Friedman, N.P.; Corley, R.P.; Hewitt, J.K.; Wright, K.P. Individual differences in childhood sleep problems predict later cognitive executive control. Sleep 2009, 32, 323–333. [Google Scholar] [PubMed]
- Simola, P.; Liukkonen, K.; Pitkäranta, A.; Pirinen, T.; Aronen, E.T. Psychosocial and somatic outcomes of sleep problems in children: A 4-year follow-up study. Child Care Health Dev. 2014, 40, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Touchette, E.; Petit, D.; Tremblay, R.E.; Montplaisir, J.Y. Risk factors and consequences of early childhood dyssomnias: New perspectives. Sleep Med. Rev. 2009, 13, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Baker, F.C.; Driver, H.S. Self-reported sleep across the menstrual cycle in young, healthy women. J. Psychosom. Res. 2004, 56, 239–243. [Google Scholar] [CrossRef]
- Barone, J.J.; Roberts, H.R. Caffeine consumption. Food Chem. Toxicol. 1996, 34, 119–129. [Google Scholar] [CrossRef]
- Carskadon, M.A.; Acebo, C. A self-administered rating scale for pubertal development. J. Adolesc. Heal. 1993, 14, 190–195. [Google Scholar] [CrossRef]
- Feinberg, I.; Higgins, L.M.; Khaw, W.Y.; Campbell, I.G. The adolescent decline of NREM delta, an indicator of brain maturation, is linked to age and sex but not to pubertal stage. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2006, 291, R1724–R1729. [Google Scholar] [CrossRef] [PubMed]
- Largo, R.H.; Pfister, D.; Molinari, L.; Kundu, S.; Lipp, A.; Duc, G. Significance of prenatal, perinatal and postnatal factors in the development of AGA preterm infants at five to seven years. Dev. Med. Child Neurol. 1989, 31, 440–456. [Google Scholar] [CrossRef] [PubMed]
- Roenneberg, T.; Kuehnle, T.; Juda, M.; Kantermann, T.; Allebrandt, K.; Gordijn, M.; Merrow, M. Epidemiology of the human circadian clock. Sleep Med. Rev. 2007, 11, 429–438. [Google Scholar] [CrossRef] [PubMed]
- Horne, J.A.; Ostberg, O. A self-assessment questionnaire to determine morningness ± eveningness in human circadian rhythms. Int. J. Chronobiol. 1976, 4, 97–110. [Google Scholar] [PubMed]
- Smith, C.; Reilly, T.; Midkiff, K. Evaluation of three circadian rhythm questionnaires with suggestions for an improved measure of morningness. J. Appl. Psychol. 1989, 74, 728–738. [Google Scholar] [CrossRef] [PubMed]
- Iber, C.; Ancoli-Israel, S.; Chesson, A.L.; Quan, S.F. For the American Academy of Sleep Medicine. In The AASM manual for the scoring of sleep and associated events: Rules, terminology and technical specifications, 1st ed.; American Academy of Sleep medicine: Westchester, Illinois, 2007. [Google Scholar]
- Huber, R.; Graf, T.; Cote, K.A.; Wittmann, L.; Gallmann, E.; Matter, D.; Schuderer, J.; Kuster, N.; Borbély, A.A.; Achermann, P. Exposure to pulsed high-frequency electromagnetic field during waking affects human sleep EEG. Neuroreport 2000, 11, 3321–3325. [Google Scholar] [CrossRef] [PubMed]
- Kurth, S.; Ringli, M.; Geiger, A.; LeBourgeois, M.; Jenni, O.G.; Huber, R. Mapping of Cortical Activity in the First Two Decades of Life: A High-Density Sleep Electroencephalogram Study. J. Neurosci. 2010, 30, 13211–13219. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, I.; Kurth, S.; Ringli, M.; Mouthon, A.-L.; Buchmann, A.; Geiger, A.; Jenni, O.G.; Huber, R. Sleep Slow-Wave Activity Reveals Developmental Changes in Experience-Dependent Plasticity. J. Neurosci. 2014, 34, 12568–12575. [Google Scholar] [CrossRef] [PubMed]
- Jenni, O.G.; Achermann, P.; Carskadon, M.A. Homeostatic sleep regulation in adolescents. Sleep 2005, 28, 1446–1454. [Google Scholar] [PubMed]
- Delorme, A.; Makeig, S. EEGLAB: An open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J. Neurosci. Methods 2004, 134, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Nichols, T.E.; Holmes, A.P. Nonparametric permutation tests for functional neuroimaging: A primer with examples. Hum. Brain Mapp. 2002, 15, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Huber, R.; Määttä, S.; Esser, S.K.; Sarasso, S.; Ferrarelli, F.; Watson, A.; Ferreri, F.; Peterson, M.J.; Tononi, G. Measures of cortical plasticity after transcranial paired associative stimulation predict changes in electroencephalogram slow-wave activity during subsequent sleep. J. Neurosci. 2008, 28, 7911–7918. [Google Scholar] [CrossRef] [PubMed]
- Huber, R.; Ghilardi, M.F.; Massimini, M.; Tononi, G. Local sleep and learning. Nature 2004, 430, 78–81. [Google Scholar] [CrossRef] [PubMed]
- Werner, H.; Molinari, L.; Guyer, C.; Jenni, O.G. Agreement rates between actigraphy, diary, and questionnaire for children’s sleep patterns. Arch. Pediatr. Adolesc. Med. 2008, 162, 350–358. [Google Scholar] [CrossRef] [PubMed]
- Harrison, Y.; Horne, J.A. Sleep loss impairs short and novel language tasks having a prefrontal focus. J. Sleep Res. 1998, 7, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Van Dongen, H.P.A.; Maislin, G.; Mullington, J.M.; Dinges, D.F. The cumulative cost of additional wakefulness: Dose-response effects on neurobehavioral functions and sleep physiology from chronic sleep restriction and total sleep deprivation. Sleep 2003, 26, 117–126. [Google Scholar] [PubMed]
- Couyoumdjian, A.; Sdoia, S.; Tempesta, D.; Curcio, G.; Rastellini, E.; de Gennaro, L.; Ferrara, M. The effects of sleep and sleep deprivation on task-switching performance. J. Sleep Res. 2010, 19, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.A.; McEnany, G.; Weekes, D. Gender differences in sleep patterns for early adolescents. J. Adolesc. Health 1999, 24, 16–20. [Google Scholar] [CrossRef]
- Martinez, D.; Breitenbach, T.C.; Lenz, M.C.S. Light sleep and sleep time misperception—Relationship to alpha-delta sleep. Clin. Neurophysiol. 2010, 121, 704–711. [Google Scholar] [CrossRef] [PubMed]
- Latta, F.; Leproult, R.; Tasali, E.; Hofmann, E.; L’Hermite-Balériaux, M.; Copinschi, G.; van Cauter, E. Sex differences in nocturnal growth hormone and prolactin secretion in healthy older adults: Relationships with sleep EEG variables. Sleep 2005, 28, 1519–1524. [Google Scholar] [PubMed]
- Bhalla, U.S.; Iyengar, R. Emergent properties of networks of biological signaling pathways. Science 1999, 283, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Huston, J.P.; Haas, H.L.; Boix, F.; Pfister, M.; Decking, U.; Schrader, J.; Schwarting, R.K. Extracellular adenosine levels in neostriatum and hippocampus during rest and activity periods of rats. Neuroscience 1996, 73, 99–107. [Google Scholar] [CrossRef]
- Tononi, G.; Cirelli, C. Sleep and the Price of Plasticity: From Synaptic and Cellular Homeostasis to Memory Consolidation and Integration. Neuron 2014, 81, 12–34. [Google Scholar] [CrossRef] [PubMed]
- Diekelmann, S.; Born, J. The memory function of sleep. Nat. Rev. Neurosci. 2010, 11, 114–126. [Google Scholar] [CrossRef] [PubMed]
- Paolicelli, R.C.; Bolasco, G.; Pagani, F.; Maggi, L.; Scianni, M.; Panzanelli, P.; Giustetto, M.; Ferreira, T.A.; Guiducci, E.; Dumas, L.; et al. Synaptic pruning by microglia is necessary for normal brain development. Science 2011, 333, 1456–1458. [Google Scholar] [CrossRef] [PubMed]
- Steger, R.; Kamal, A.; Lutchman, S.; Intrabartolo, L.; Sohail, R.; Brumberg, J.C. Chronic caffeine ingestion causes microglia activation, but not proliferation in the healthy brain. Brain Res. Bull. 2014, 106, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Rex, C.S.; Chen, L.Y.; Sharma, A.; Liu, J.; Babayan, A.H.; Gall, C.M.; Lynch, G. Different Rho GTPase-dependent signaling pathways initiate sequential steps in the consolidation of long-term potentiation. J. Cell Biol. 2009, 186, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Perkinson-Gloor, N.; Lemola, S.; Grob, A. Sleep duration, positive attitude toward life, and academic achievement: The role of daytime tiredness, behavioral persistence, and school start times. J. Adolesc. 2013, 36, 311–318. [Google Scholar] [CrossRef] [PubMed]
- Achermann, P.; Borbely, A.A. Sleep homeostasis and models of sleep regulation. In Principles and Practice of Sleep Medicine; Kryger, M., Roth, T., Dement, W., Eds.; 2011; pp. 431–444. [Google Scholar]
- Tononi, G.; Cirelli, C. Sleep function and synaptic homeostasis. Sleep Med. Rev. 2006, 10, 49–62. [Google Scholar] [CrossRef] [PubMed]
- Calamaro, C.J.; Mason, T.B.A.; Ratcliffe, S.J. Adolescents living the 24/7 lifestyle: Effects of caffeine and technology on sleep duration and daytime functioning. Pediatrics 2009, 123, e1005–e1010. [Google Scholar] [CrossRef] [PubMed]
- Rétey, J.V.; Adam, M.; Khatami, R.; Luhmann, U.F.O.; Jung, H.H.; Berger, W.; Landolt, H.-P.P. A genetic variation in the adenosine A2A receptor gene (ADORA2A) contributes to individual sensitivity to caffeine effects on sleep. Clin. Pharmacol. Ther. 2007, 81, 692–698. [Google Scholar] [CrossRef] [PubMed]
- Perlis, M.L.; Merica, H.; Smith, M.T.; Giles, D.E. Beta EEG activity and insomnia. Sleep Med. Rev. 2001, 5, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Olini, N.; Kurth, S.; Huber, R. The Effects of Caffeine on Sleep and Maturational Markers in the Rat. PLoS One 2013, 8, e72539. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aepli, A.; Kurth, S.; Tesler, N.; Jenni, O.G.; Huber, R. Caffeine Consuming Children and Adolescents Show Altered Sleep Behavior and Deep Sleep. Brain Sci. 2015, 5, 441-455. https://doi.org/10.3390/brainsci5040441
Aepli A, Kurth S, Tesler N, Jenni OG, Huber R. Caffeine Consuming Children and Adolescents Show Altered Sleep Behavior and Deep Sleep. Brain Sciences. 2015; 5(4):441-455. https://doi.org/10.3390/brainsci5040441
Chicago/Turabian StyleAepli, Andrina, Salome Kurth, Noemi Tesler, Oskar G. Jenni, and Reto Huber. 2015. "Caffeine Consuming Children and Adolescents Show Altered Sleep Behavior and Deep Sleep" Brain Sciences 5, no. 4: 441-455. https://doi.org/10.3390/brainsci5040441
APA StyleAepli, A., Kurth, S., Tesler, N., Jenni, O. G., & Huber, R. (2015). Caffeine Consuming Children and Adolescents Show Altered Sleep Behavior and Deep Sleep. Brain Sciences, 5(4), 441-455. https://doi.org/10.3390/brainsci5040441





