Central Causation of Autism/ASDs via Excessive [Ca2+]i Impacting Six Mechanisms Controlling Synaptogenesis during the Perinatal Period: The Role of Electromagnetic Fields and Chemicals and the NO/ONOO(-) Cycle, as Well as Specific Mutations
Abstract
1. Introduction
1.1. Autism/ASD Causation via Disruption of Synaptogenesis during the Perinatal Period: A Brief Review of Much of the Evidence
- Perinatal synaptogenesis is dysfunctional in human ASDs and animal models of ASD;
- Synaptic developmental dysfunction is raised by agents causing autism and lowered by agents found to be helpful in autism treatment;
- [Ca2+]i signaling is very important in autism/ASDs;
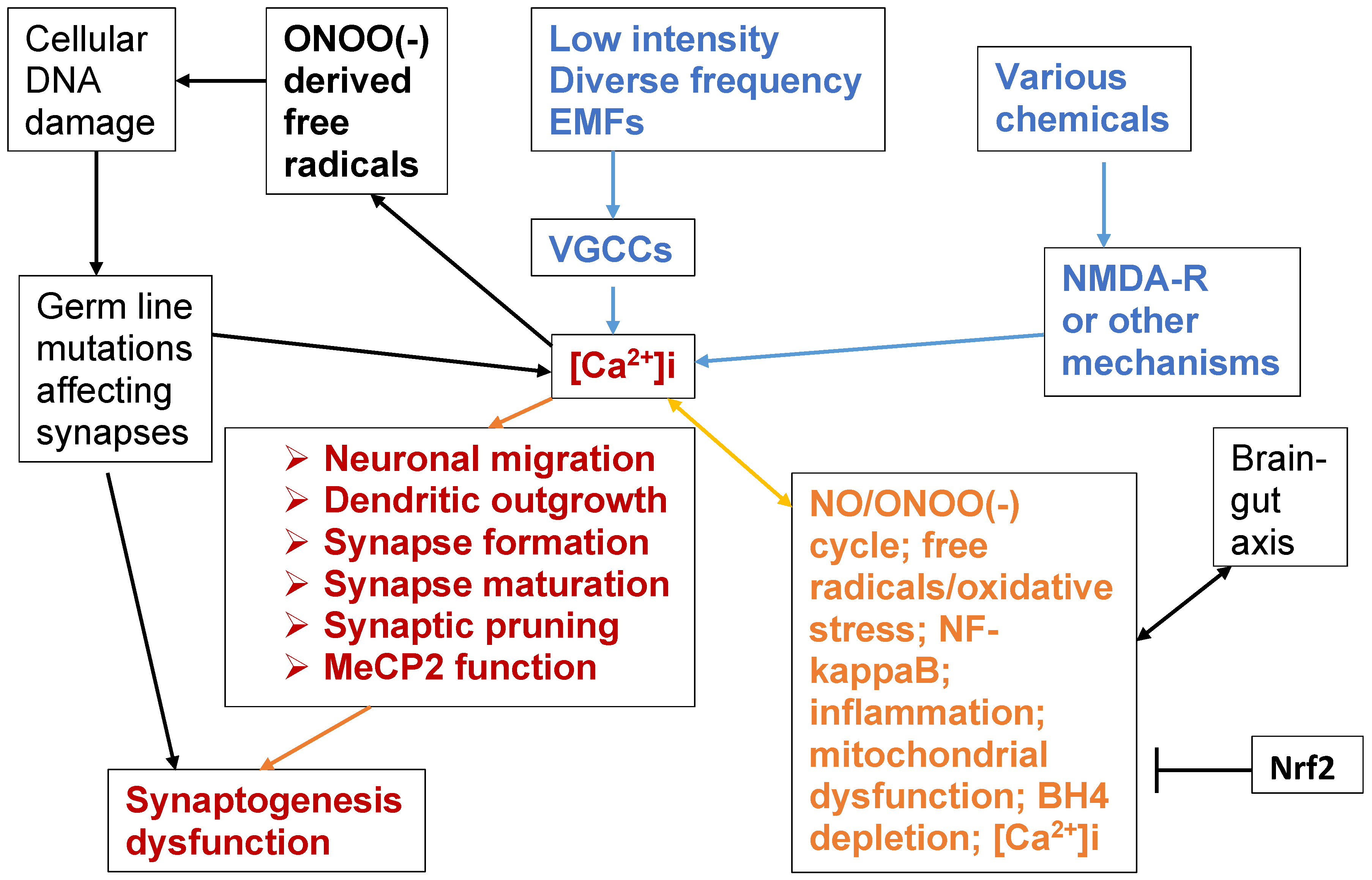
- Each of the six mechanisms that have important roles in synaptogenesis are regulated by [Ca2+]i and can therefore be impacted by either inappropriate [Ca2+]i elevation, or possibly even inappropriate [Ca2+]i depression;
- Each of those six mechanisms are impacted in ASDs;
- Each of those same six mechanisms are regulated by CamKII protein kinase, the most active calcium-regulated protein kinase in the brain;
- Synaptogenesis dysfunction produces tissue connectivity changes in the autism/ASD brain which are thought, in turn, to produce many autism symptoms. It may act, in part, to disrupt and change neural network formation, a possible central causal mechanism of autism/ASDs;
- Aberrant synaptogenesis causes macrocephaly which has much higher prevalence in individuals with autism/ASDs.
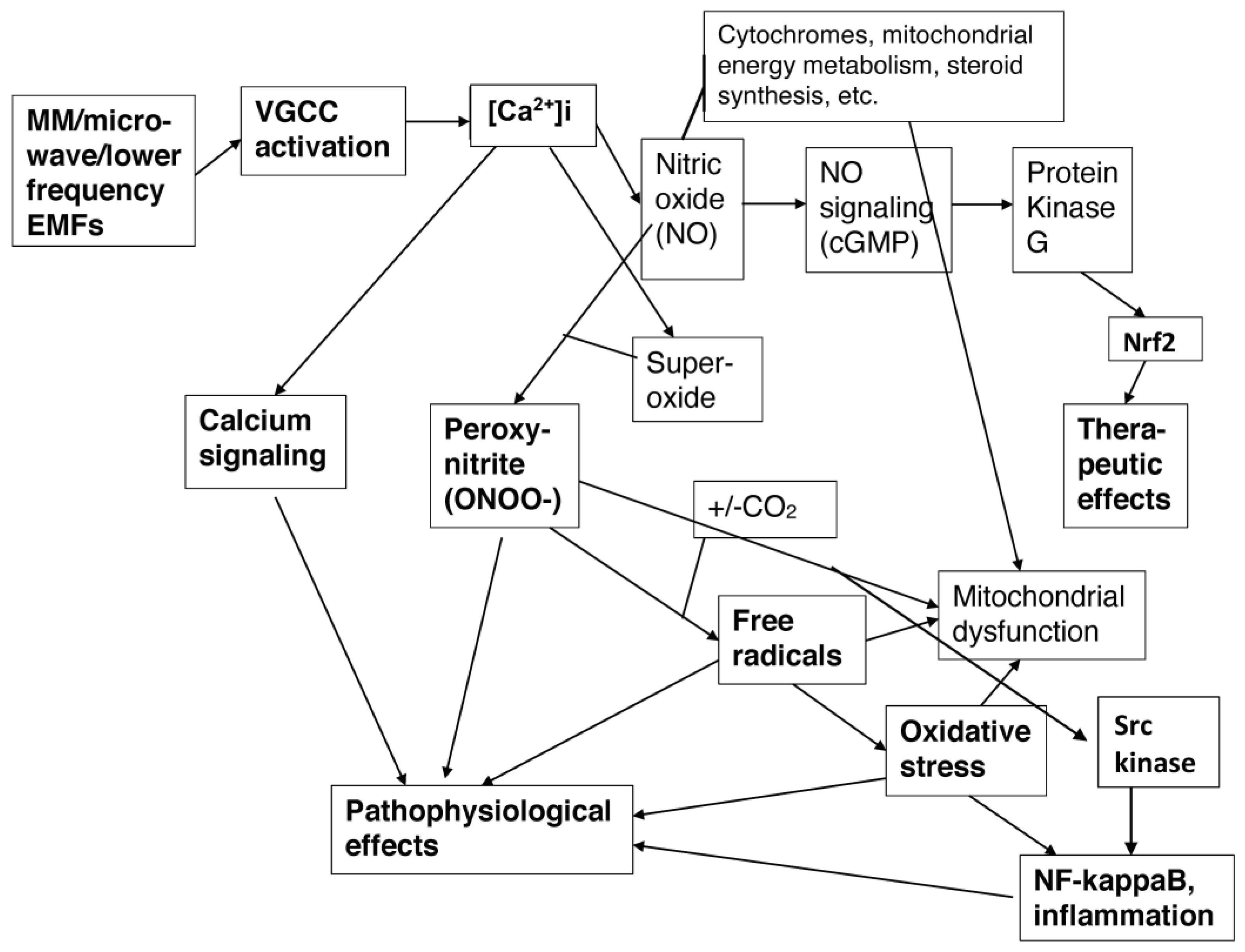
2. How Electronically-Generated EMFs Act via Two Distinct Mechanisms to Activate the VGCCs and Produce Excessive [Ca2+]i
2.1. EMFs as a Possible Major Cause of the Autism/ASD Epidemic
2.2. Genetic Evidence for VGCC Activity Roles in Autism
2.3. EMF Effects on Neurite Outgrowth from Neural Stem Cells
2.4. Epidemiology of EMFs and Autism/ASD Prevalence
2.5. EMF Causation of ASD-like Conditions in Animals
2.6. VGCC Activation Can Act Independently of [Ca2+]i
2.7. ASD Causation and Electronically-Generated EMF Exposures: A Summary of the Evidence
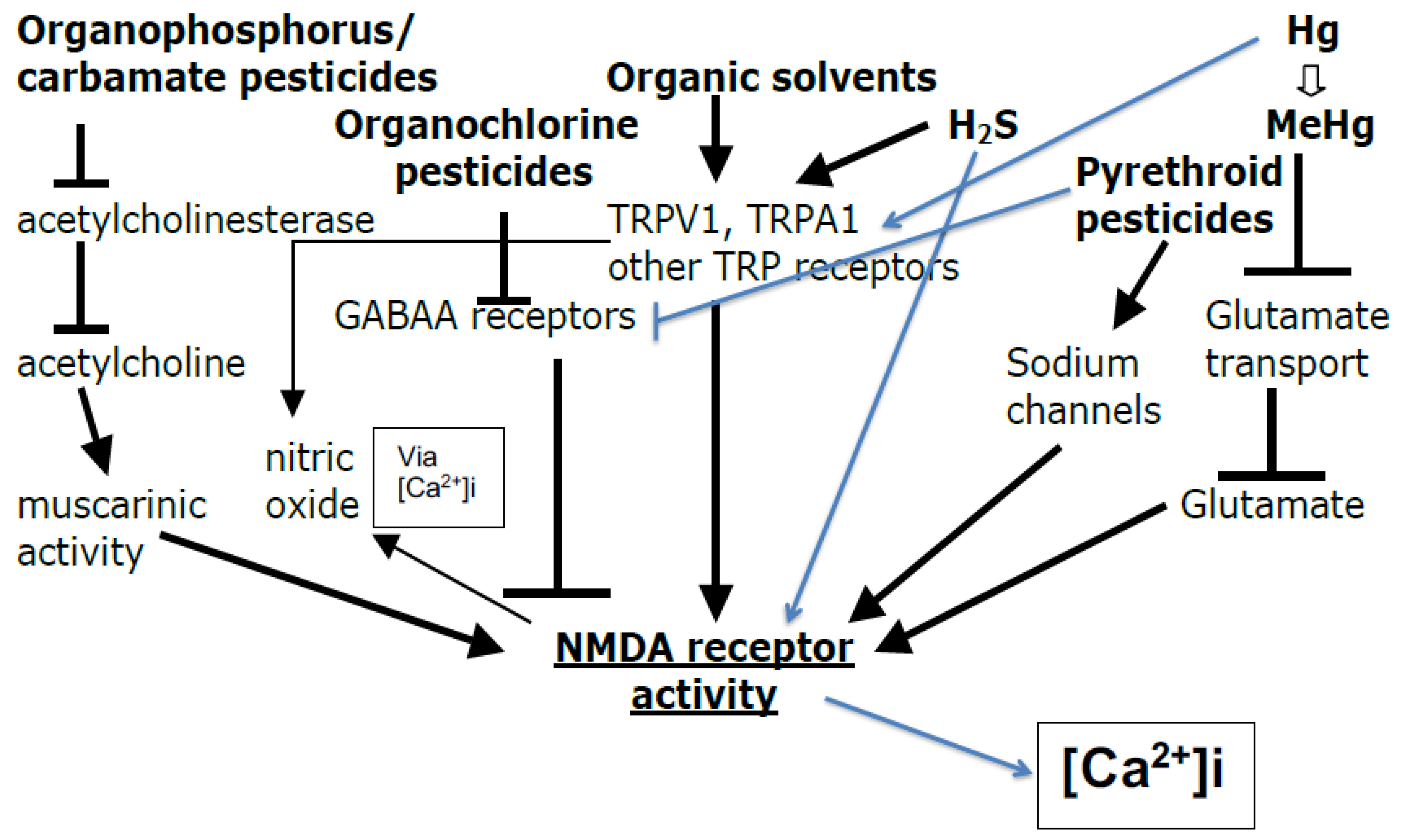
3. Organization of the Sections on the NMDA Receptors (NMDA-Rs) and Their Roles in Chemical Toxicity and Autism/ASD Causation
- The genetic evidence showing that NMDA-R activity is important in autism causation;
- The complex properties of NMDA-Rs, where an understanding of those properties is essential for understanding how toxic chemicals can act to increase NMDA-R activity and therefore cause autism/ASDs;
- Section 3 discusses whether and how various toxic chemicals can act to activate the NMDA-Rs or via other mechanisms to increase [Ca2+]i levels and therefore, potentially act to cause autism.
3.1. Mutations Changing the Activity of the NMDA-Rs Have Been Shown to Cause Autism
3.2. Complex Properties of NMDA-Rs
3.3. Diverse Chemicals Act Primarily but Not Solely via Increased NMDA-R Activity
3.4. Agents That Lower VGCC Activity or NMDA-R Activity Are Reported to Produce Protective Effects in Autism Rat Models
4. GABAA Switching during the Early Post-Natal Period—Another Issue That Makes ASD Causation More Complex
5. Mutations Having Major Roles in Causing Forms of Autism: Focus on the Mechanism of Autism Causation
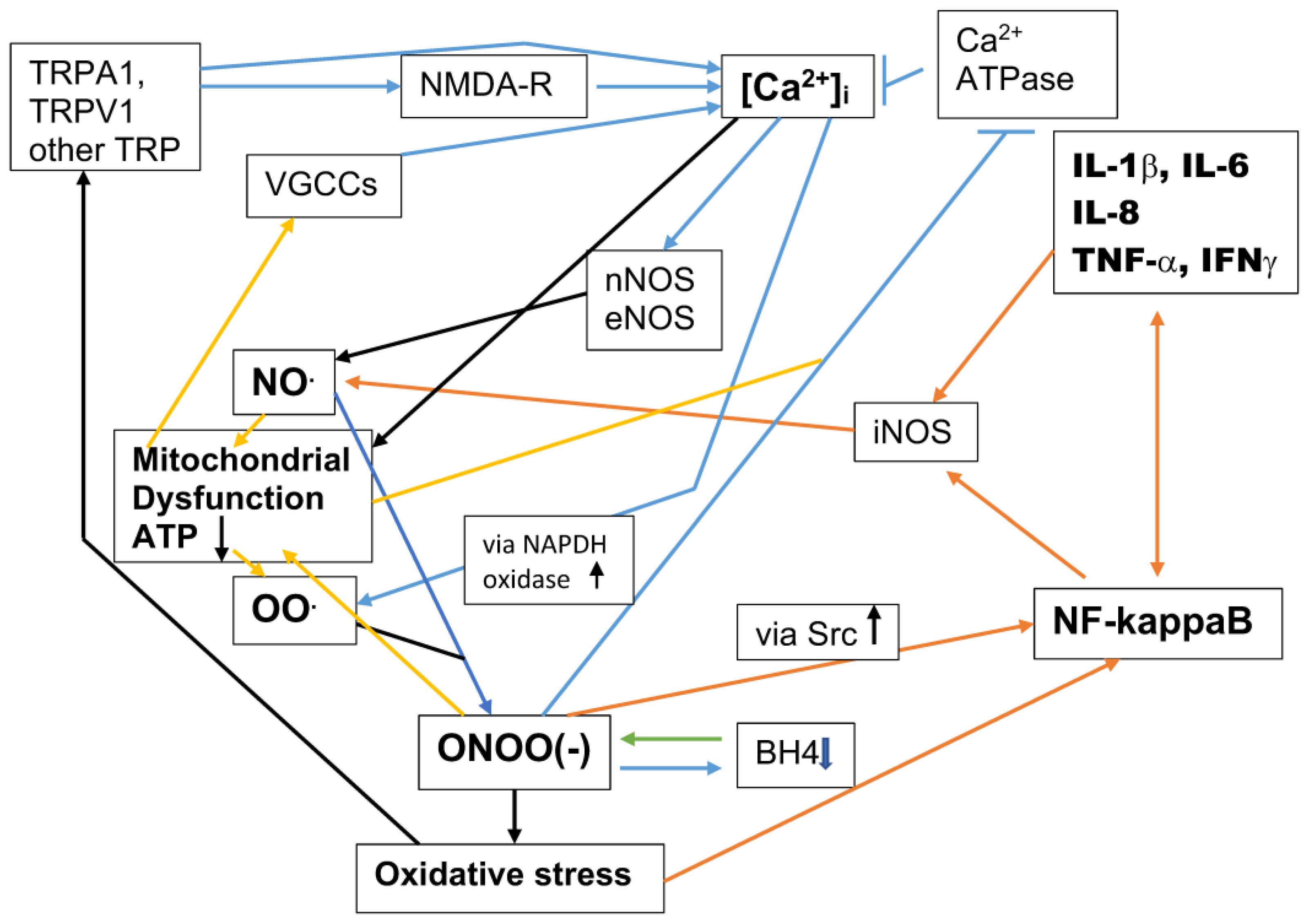
6. The NO/ONOO(-) Cycle and the Chronic Nature of Autism/ASDs
| Peroxynitrite (ONOO-) | Peroxynitrite was shown to be elevated in autism based on a test using 3-nitrotyrosine as a specific marker for peroxynitrite (Sajdel-Sulkowska et al. [213]; De Felice et al. [214]; Nadeem et al. [215,216]; Algahtani et al. [217]; Carey et al. [218]). Peroxynitrite elevation by the NO/ONOO(-) cycle, as discussed here and in thousands of other papers, produces oxidative stress. Peroxynitrite is scavenged through its reaction oxidizing 5-methyltetrahydrofolate (5-MTHF) [219,220], thus depleting cells of a very important methyl donor, while lowering peroxynitrite. These properties of peroxynitrite mean that both the oxidative stress and the low methylation status of autism patients reported by James et al. [221] may be a consequence of peroxynitrite elevation. |
| Oxidative stress | See above. There are dozens of papers showing oxidative stress in people with autism/ASDs, as well as in animal models. The following examples each provide strong evidence for an oxidative stress role in ASD causation: Carey et al. [218]; De Felice et al. [214]; Golomb [212]; James et al. [221]; McGinnis [94]; Nadeem et al. [215,216]; Saidel-Sulkowska et al. [213], and Bjorklund et al. [222]. |
| NF-kappaB | NF-kappaB has been among the less studied of the measurable elements of the cycle in autism. NF-kappaB has been found to be elevated in cases of autism in Young et al. [223]; Nadeen et al. [216]; Liao and Li [52], and Theoharides et al. [224]. Young et al. [223] may be of special interest, in that the paper was completely focused on NF-kappaB measurements. They used an immunoassay for NF-kappa that can be scored under a microscope, studying post-mortem orbitofrontal cortex brain tissues of patients with ASDs and matched normal controls. Young et al. [223] found very highly statistically significant elevation of NF-kappaB in ASD vs. controls in each of the three cell types studied, neurons, astrocytes, and microglia. They also found that there were similar elevations in both the nuclear localized and extranuclear NF-kappaB in ASD tissues. Because the NO/ONOO(-) cycle predicts cycle elevation of the synthesis of NF-kappaB, these findings are in good agreement with prediction. Alomar et al. [225] showed that NF-kappaB elevation has a causal role in a mouse model of autism. |
| Inflammatory cytokines | Eftekharian et al. [226] found that each of the five inflammatory cytokines predicted to be directly raised by NF-kappaB elevation were raised in autism patients, as well as several other cytokines. Theoharides et al. [224], Kutuk et al. [227], and Ferencova et al. [228] found, similarly, in their large studies, that the same five inflammatory cytokines directly raised by NF-kappaB were elevated in patients with ASDs, as were multiple additional cytokines. Mehta et al. [229] showed that four of those five cytokines (all except IFNγ) were elevated in a rat model of ASD. |
| Mitochondrial dysfunction | Mitochondrial dysfunction has been demonstrated in individuals with autism/ASDs. Napolioni et al. [230] ascribed much of the mitochondrial dysfunction seen in those with ASDs to the dysfunction of the calcium-regulated aspartate/glutamate carrier. Others reviewing mitochondrial dysfunction in autism include Golomb [212], Rossignol and Frye [231], and Palmieri and Persico [232]. When Palmieri and Persico [232] asked whether mitochondrial dysfunction is either the cause or effect of ASDs, they concluded it is both, consistent with the NO/ONOO(-) cycle predictions that all cycle elements must be both cause and effect. |
| [Ca2+]i | Autism/ASDs caused by excessive [Ca2+]i has been documented throughout this paper. |
| Tetrahydrobiopterin (BH4) depletion | Tani et al. [233] and Frye [234,235] have each demonstrated a deficiency of tetrahydrobiopterin (BH4) in autism patients. A series of studies have each shown that BH4 supplementation is useful in autism treatment (Naruse et al. [236]; Takesada et al. [237]; Frye [234]). The NO/ONOO(-) cycle produces, as part of the cycle, peroxynitrite-mediated oxidation of BH4 to dihydrobiopterin (BH2), thus lowering BH4 levels. Consequently, BH4 supplementation will not only provide the BH4 cofactor for several enzymes, including the nitric oxide synthases, lowering the NO/ONOO(-) cycle in that way, but will also lower peroxynitrite by chemically reacting with it, lowering the cycle in a second way. |
| iNOS | iNOS has been found to be elevated in patients with ASDs (Nadeem et al. [215,216]). Mehta et al. [226] showed that iNOS was elevated in a rat model of ASDs. |
| Nitric oxide (NO) | Several reviews documented elevated nitric oxide in patients with ASDs, including Nadeem et al. [215,216] and Tripathy et al. [238]). Mehta et al. [229] showed that nitric oxide was elevated in a rat model of ASDs. |
| RhoA | In the paper showing that the NO/ONOO(-) cycle is central to the etiology of heart failure, it was shown that RhoA functioned as possibly tissue-limited cycle element [209]. RhoA is implicated as a causal element of autism/ASDs, including in stabilization of synapses (Richter et al. [239]; Luo et al. [240]. Hayashi et al. [241]), providing further evidence consistent with a NO/ONOO(-) cycle role in autism/ASDs. |
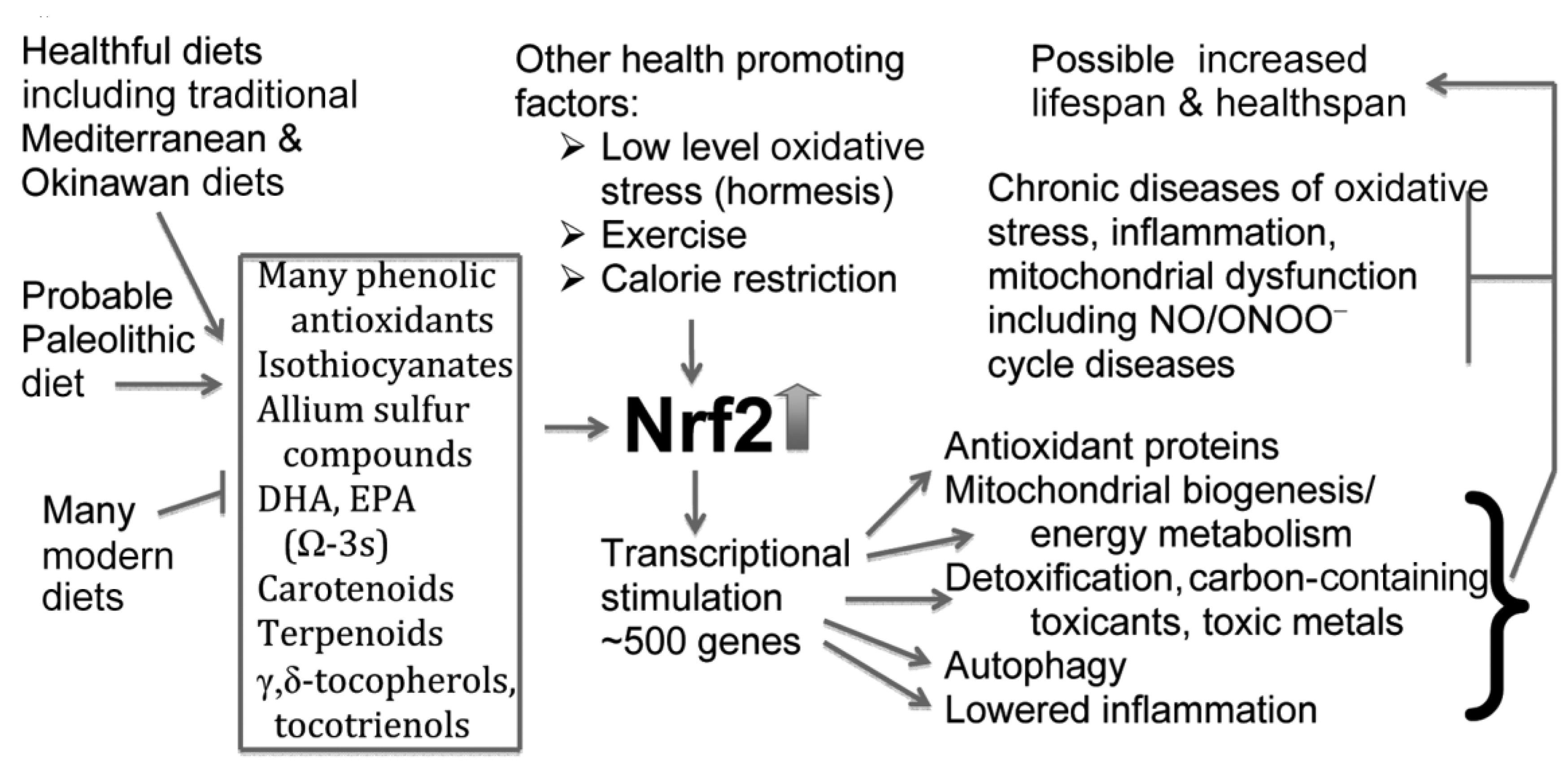
6.1. The NO/ONOO(-) Cycle and the Chronic Nature of Autism/ASDs: Therapeutic Approaches by Raising Nrf2 and AMPK
7. Discussion and Conclusions
- The special role of perinatal events is not new;
- The essential role of elevated [Ca2+]i and consequent calcium signaling is not new;
- The special roles of synaptic changes and synaptogenesis is not new;
- Even the roles of depressed levels of [Ca2+]i is not new;
- The importance of EMF exposures is not new, although the detailing of diverse types of supportive evidence is new;
- The importance of chemical exposure and even the action of diverse chemicals via increased [Ca2+]i is not new;
- The NO/ONOO(-) cycle as an apparent cause of chronic disease is not new, although documentation of its role in autism/ASDs is, to my knowledge, new;
- The role of raising Nrf2 in the prevention or treatment of apparent NO/ONOO(-) cycle diseases is not new, nor is it possible role in treatment or prevention of ASDs;
- Mutational causation of autism/ASDs via mutations causing changes in [Ca2+]i or in synaptogenesis is not new.
- It does not include number 4 above, that abnormally low [Ca2+]i also can cause autism/ASDs;
- It does not include the finding in [138] that VGCCs can have roles in ASD causation independent of [Ca2+]i, although, as discussed in the text, this can be produced via direct protein–protein interactions;
- It was correctly pointed out to the author by one reviewer that glial cells [260,261,262,263], including astrocytes [264,265,266,267,268], have roles in synaptogenesis, but only the roles of neurons were included in Figure 1. Furthermore, glia, including astrocytes, have NMDA-Rs [269,270], as well as the almost universally found VGCCs, and glial calcium signaling is of great importance.
Funding
Acknowledgments
Conflicts of Interest
References
- Kanner, L. Autistic disturbances of affective contact. Nerv. Child 1943, 2, 217–260. [Google Scholar]
- Boyd, B.A.; Conroy, M.A.; Mancil, G.R.; Nakao, T. Effects of circumscribed interests on the social behaviors of children with autism spectrum disorders. J. Autism Dev. Disord. 2007, 37, 1550–1561. [Google Scholar] [CrossRef] [PubMed]
- Richler, J.; Lord, C. Association between restricted and repetitive behaviors and nonverbal IQ in children with autism spectrum disorders. Child Neuropsychol. 2006, 12, 247–267. [Google Scholar]
- Nevison, C.; Blaxill, M.; Nahorodny, W. California Autism Prevalence Trends from 1931 to 2014 and Comparison to National ASD Data from IDEA and ADDM. J Autism Dev. Disord. 2018, 48, 4103–4117. [Google Scholar] [CrossRef]
- Lathe, R. Electromagnetic Radiation and Autism. E-J. Appl. Psychol. 2009, 5, 11–30. [Google Scholar] [CrossRef]
- Newschaffer, C.J.; Falb, M.D.; Gurney, J.G. National autism prevalence trends from United States special education data. Pediatrics 2005, 115, e277–e282. [Google Scholar] [CrossRef]
- Maenner, M.J.; Shaw, K.A.; Bakian, A.V.; Bilder, D.A.; Durkin, M.S.; Esler, A.; Furnier, S.M.; Hallas, L.; Hall-Lande, J.; Hudson, A.; et al. Prevalence and characteristics of autism spectrum disorder among children aged 8 years—Autism and developmental disabilities monitoring network, 11 sites, United States, 2018. MMWR Surveill. Summ. 2021, 70, 1–16. [Google Scholar] [CrossRef]
- Bourgeron, T. A synaptic trek to autism. Curr. Opin. Neurobiol. 2009, 19, 231–234. [Google Scholar] [CrossRef]
- Das, U.N. Chapter 1—A perinatal strategy to prevent autism. In Autism 360°; Academic Press: Cambridge, MA, USA, 2020; pp. 3–32. [Google Scholar]
- Yang, E.J.; Ahn, S.; Lee, K.; Mahmood, U.; Kim, H.S. Early behavioral abnormalities and perinatal alterations of PTEN/AKT pathway in valproic acid autism model mice. PLoS ONE 2016, 11, e0157202. [Google Scholar]
- Koyama, R.; Ikegaya, Y. Microglia in pathogenesis of autism spectrum disorders. Neurosci. Res. 2015, 100, 1–5. [Google Scholar] [CrossRef]
- Fujimura, M.; Usuki, F. Pregnant rats exposed to low-level methylmercury exhibit cerebellar synaptic and neuritic remodeling during the perinatal period. Arch. Toxicol. 2020, 94, 1335–1347. [Google Scholar] [CrossRef] [PubMed]
- Masini, E.; Loi, E.; Vega-Benedetti, A.F.; Carta, M.; Doneddu, G.; Fadda, R.; Zavattari, P. An overview of the main genetic, epigenetic and environmental factors involved in autism spectrum disorder focusing on synaptic activity. Int. J. Mol. Sci. 2020, 21, 8290. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Fan, T.; Li, X.; Liu, L.; Mao, F.; Li, Y.; Miao, Z.; Zeng, C. Npas3 deficiency impairs cortical astrogenesis and induces autistic-like behaviors. Cell Rep. 2022, 40, 111289. [Google Scholar] [CrossRef] [PubMed]
- Bokobza, C.; Van Steenwinkel, J.; Mani, S.; Mezger, V. Neural inflammation in preterm babies and autism spectrum. Pediatr. Res. 2019, 85, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Pizzarelli, R.; Cherubini, E. Alterations in GABAergic signaling of autism spectrum. Neural Plast. 2011, 2011, 297153. [Google Scholar] [CrossRef]
- Zhu, Y. Perinatal Epigenetic Signature of Autism Spectrum Disorder. Ph.D. Thesis, University of California Davis, Davis, CA, USA, 2021; p. 28498790. [Google Scholar]
- Das, U.N. Autism as a disorder of deficiency of brain-derived neurotrophic factor and altered metabolism of polyunsaturated fatty acids. Nutrition 2013, 29, 1175–1185. [Google Scholar] [CrossRef]
- Schiavi, S.; Carbone, E.; Melancia, F.; Buzzelli, V.; Manduca, A.; Campolongo, P. Perinatal supplementation with omega-3 fatty acids corrects the aberrant social and cognitive traits observed in a genetic model of autism based on FMR1 deletion in rats. Nutr. Neurosci. 2022, 25, 898–911. [Google Scholar] [CrossRef] [PubMed]
- Chaste, P.; Leboyer, M. Autism risk factors: Genes, environment, and gene-environment interactions. Dialogues Nutr. Neurosci. 2012, 14, 281–292. [Google Scholar] [CrossRef]
- Ey, E.; Leblond, C.S.; Bourgeron, T. Behavioral profiles of mouse models of autism spectrum disorders. Autism Res. 2011, 4, 5–16. [Google Scholar] [CrossRef]
- O’Connor, M. The Role of Autism Gene NEXMIF in Neuronal Development, Synapse Formation, and Behaviors. Ph.D. Thesis, Boston University, Boston, MA, USA, 2021. [Google Scholar]
- Tate, K.M. Role of Antidepressants in Fetal Synapse Formation in Autism Spectrum Disorders. Master’s Thesis, East Carolina University, Greenville, NC, USA, 2018. Available online: https://thescholarship.ecu.edu/handle/10342/6985 (accessed on 22 January 2024).
- Ranger, P.; Ellenbroek, B.A. Perinatal influences of valproate on brain and behaviour: An animal model for autism. In Neurotoxin Modeling of Brain Disorders—Life-Long Outcomes in Behavioral Teratology; Kostrzewa, R.M., Archer, T., Eds.; Current Topics in Behavioral Neurosciences; Springer: Cham, Switzerland, 2015; Volume 29. [Google Scholar] [CrossRef]
- Rybakowski, F.; Chojnicka, I.; Dziechciarz, P.; Horvath, A.; Janas-Kozik, M.; Jeziorek, A.; Pisula, E.; Piwowarczyk, A.; Słopień, A.; Sykut-Cegielska, J.; et al. The role of genetic factors and pre-and perinatal influences in the etiology of autism spectrum disorders-indications for genetic referral. Psychiatr. Pol. 2016, 50, 543–554. [Google Scholar] [CrossRef]
- Fujimura, M.; Cheng, J.; Zhao, W. Perinatal exposure to low-dose methylmercury induces dysfunction of motor coordination with decreases in synaptophysin expression in the cerebellar granule cells. Brain Res. 2012, 1464, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Belmonte, M.K.; Allen, G.; Beckel-Mitchener, A. Autism and abnormal development of brain connectivity. J. Neurosci. 2004, 24, 9228–9231. [Google Scholar] [CrossRef] [PubMed]
- Rane, P.; Cochran, D.; Hodge, S.M. Connectivity in autism: A review of MRI connectivity studies. Harv. Rev. Psychiatry 2015, 23, 223–244. [Google Scholar] [CrossRef] [PubMed]
- Rolls, E.T.; Zhou, Y.; Cheng, W.; Gilson, M.; Deco, G.; Feng, J. Effective connectivity in autism. Autism Res. 2020, 13, 32–44. [Google Scholar] [CrossRef] [PubMed]
- Hull, J.V.; Dokovna, L.B.; Jacokes, C.J.; Torgerson, C.M.; Irimia, A.; Van Horn, J.D. Resting-state functional connectivity in autism spectrum disorders: A review. Front. Psychiatry 2017, 7, 205. [Google Scholar] [CrossRef] [PubMed]
- Uddin, L.Q.; Supekar, K.; Menon, V. Reconceptualizing functional brain connectivity in autism from a developmental perspective. Front. Hum. Neurosci. 2013, 7, 458. [Google Scholar] [CrossRef] [PubMed]
- Wylie, K.P.; Tregellas, J.R.; Bear, J.J.; Legget, K.T. Autism spectrum disorder symptoms are associated with connectivity between large-scale neural networks and brain regions involved in social processing. J. Autism Dev. Disord. 2020, 50, 2765–2778. [Google Scholar] [CrossRef] [PubMed]
- Dajani, D.R.; Burrows, C.A.; Nebel, M.B.; Mostofsky, S.H.; Gates, K.M.; Uddin, L.Q. Parsing heterogeneity in autism spectrum disorder and attention-deficit/hyperactivity disorder with individual connectome mapping. Brain Connect. 2019, 9, 673–691. [Google Scholar] [CrossRef]
- Leisman, G.; Melillo, R.; Melillo, T. Prefrontal functional connectivities in autism spectrum disorders: A connectopathic disorder affecting movement, interoception, and cognition. Brain Res. Bull. 2023, 198, 65–76. [Google Scholar] [CrossRef]
- Stamou, M.; Streifel, K.M.; Goines, P.E.; Lein, P.J. Neuronal connectivity as a convergent target of gene× environment interactions that confer risk for autism spectrum disorders. Neurotoxicol. Teratol. 2013, 36, 3–16. [Google Scholar] [CrossRef]
- Reiner, O.; Karzbrun, E.; Kshirsagar, A.; Kaibuchi, K. Regulation of neuronal migration, an emerging topic in autism spectrum disorders. J. Neurochem. 2016, 136, 440–456. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.H.; Wu, N.; Yuan, X.B. Toward a better understanding of neuronal migration deficits in autism spectrum disorders. Front. Cell Dev. Biol. 2019, 7, 205. [Google Scholar] [CrossRef] [PubMed]
- Wegiel, J.; Kuchna, I.; Nowicki, K.; Imaki, H.; Wegiel, J.; Marchi, A.; Ma, S.Y.; Chauhan, V.; Bobrowicz, T.W.; de Leon, M.; et al. The neuropathology of autism: Defects of neurogenesis and neuronal migration, and dysplastic changes. Acta Neuropathol. 2010, 119, 755–770. [Google Scholar] [CrossRef]
- Peñagarikano, O.; Abrahams, B.S.; Herman, E.I.; Winden, K.D.; Gdalyahu, A.; Dong, H.; Sonnenblick, L.I.; Gruver, R.; Almajano, J.; Bragin, A.; et al. Absence of CNTNAP2 leads to epilepsy, neuronal migration abnormalities, and core autism-related deficits. Cell 2011, 147, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Hussman, J.P.; Chung, R.H.; Griswold, A.J.; Jaworski, J.M.; Salyakina, D.; Ma, D.; Konidari, I.; Whitehead, P.L.; Vance, J.M.; Martin, E.R.; et al. A noise-reduction GWAS analysis implicates altered regulation of neurite outgrowth and guidance in autism. Mol. Autism 2011, 2, 1. [Google Scholar] [CrossRef]
- Martinez-Cerdeño, V. Dendrite and spine modifications in autism and related neurodevelopmental disorders in patients and animal models. Dev. Neurobiol. 2017, 77, 393–404. [Google Scholar] [CrossRef] [PubMed]
- Coley, A.A.; Gao, W.J. PSD95: A synaptic protein implicated in schizophrenia or autism. Prog. Neuropsychopharmacol. Biol. Psychiatry 2018, 82, 187–194. [Google Scholar] [CrossRef]
- Hansel, C. Deregulation of synaptic plasticity in autism. Neurosci. Lett. 2019, 688, 58–61. [Google Scholar] [CrossRef]
- Thomas, M.S.C.; Davis, R.; Karmiloff-Smith, A.; Knowland, V.C.P.; Charman, T. The over-pruning hypothesis of autism. Dev. Sci. 2016, 19, 284–305. [Google Scholar] [CrossRef]
- Picard, N.; Fagiolini, M. MeCP2: An epigenetic regulator of critical periods. Curr. Opin. Neurobiol. 2019, 59, 95–101. [Google Scholar] [CrossRef]
- Vuu, Y.M.; Roberts, C.T.; Rastegar, M. MeCP2 Is an Epigenetic Factor That Links DNA Methylation with Brain Metabolism. Int. J. Mol. Sci. 2023, 24, 4218. [Google Scholar] [CrossRef] [PubMed]
- LaSalle, J.M.; Masui, D.H. Evolving role of MeCP2 in Rett syndrome and autism. Epigenomics 2009, 1, 119–130. [Google Scholar] [CrossRef]
- Qiu, Z.; Cheng, J. The role of calcium-dependent gene expression in autism spectrum disorders. Neurosignals 2010, 18, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Krey, J.F.; Dolmetsch, R.E. Molecular mechanisms of autism: A possible role for Ca2+ signaling. Curr. Opin. Neurobiol. 2007, 17, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Lohmann, C. Calcium signaling and the development of specific neuronal connections. Prog. Brain Res. 2009, 175, 443–452. [Google Scholar] [PubMed]
- Lozac, N.B.S. Central Role of Voltage Gated Calcium Channels and Intercellular Calcium Homeostasis in Autism. 2010. Available online: https://kipdf.com/central-role-of-voltage-gated-calcium-channels-and-intercellular-calcium-homeost_5ac409f91723dd67feabd55f.html (accessed on 12 February 2024).
- Liao, X.; Li, Y. Genetic associations between voltage-gated calcium channels and autism spectrum disorder: A systematic review. Mol. Brain 2020, 13, 96. [Google Scholar] [CrossRef]
- Pourtavakoli, A.; Ghafouri-Fard, S.; Eslami, S. Calcium signaling in neurodevelopment and pathophysiology of autism spectrum disorders. Mol. Biol. Rep. 2022, 49, 10811–10823. [Google Scholar] [CrossRef]
- Nguyen, R.L.; Medvedeva, Y.V.; Ayyagari, T.E.; Schmunk, G.; Gargus, J.J. Intracellular calcium dysregulation in autism spectrum disorder: An analysis of converging organelle signaling pathways. Biochim. Biophys. Acta—Mol. Cell Res. 2018, 1865, 1718–1732. [Google Scholar] [CrossRef] [PubMed]
- Baucum, J.; Shonesy, B.C.; Rose, K.L. Quantitative proteomics analysis of CaMKII phosphorylation and the CaMKII interactome in the mouse forebrain. ACS Chem. Neurosci. 2015, 6, 615–631. [Google Scholar] [CrossRef]
- Jeong, J.; Li, Y.; Roche, K.W. CaMKII phosphorylation regulates synaptic enrichment of Shank3. eNeuro 2021, 8. [Google Scholar] [CrossRef]
- Sudarov, A.; Gooden, F.; Tseng, D.; Gan, W.B.; Ross, M.E. Lis1 controls dynamics of neuronal filopodia and spines to impact synaptogenesis and social behaviour. EMBO Mol. Med. 2013, 5, 591–607. [Google Scholar] [CrossRef] [PubMed]
- Nicole, O.; Pacary, E. Camkiiβ in neuronal development and plasticity: An emerging candidate in brain diseases. Int. J. Mol. Sci. 2020, 21, 7272. [Google Scholar] [CrossRef] [PubMed]
- Uribe, E.; Wix, R. Neuronal migration, apoptosis and bipolar disorder. Rev. Psiquiatr. Salud Ment. (Engl. Ed.) 2012, 5, 127–133. [Google Scholar] [CrossRef]
- Borodinsky, L.N.; O’Leary, D.; Neale, J.H.; Vicini, S.; Coso, O.A.; Fiszman, M.L. GABA-induced neurite outgrowth of cerebellar granule cells is mediated by GABAA receptor activation, calcium influx and CaMKII and erk1/2 pathways. J. Neurochem. 2003, 84, 1411–1420. [Google Scholar] [CrossRef]
- Lohmann, C.; Wong, R.O.L. Regulation of dendritic growth and plasticity by local and global calcium dynamics. Cell Calcium 2005, 37, 403–409. [Google Scholar] [CrossRef]
- Rongo, C.; Kaplan, J.M. CaMKII regulates the density of central glutamatergic synapses in vivo. Nature 1999, 402, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Diniz, L.P.; Tortell, V.; Garcia, M.N.; Araújo, A.P.B.; Melo, H.M.; Seixas da Silva, G.S.; De Felice, F.G. Astrocyte transforming growth factor beta 1 promotes inhibitory synapse formation via CaM kinase II signaling. Glia 2014, 62, 1917–1931. [Google Scholar] [CrossRef]
- Gambrill, A.C.; Barria, A. NMDA receptor subunit composition controls synaptogenesis and synapse stabilization. Proc. Natl. Acad. Sci. USA 2011, 108, 5855–5860. [Google Scholar] [CrossRef]
- Lisman, J.; Cooper, K.; Sehgal, M.; Silva, A.J. Memory formation depends on both synapse-specific modifications of synaptic strength and cell-specific increases in excitability. Nat. Neurosci. 2018, 21, 309–314. [Google Scholar] [CrossRef]
- Wu, G.Y.; Cline, H.T. Stabilization of dendritic arbor structure in vivo by CaMKII. Science 1998, 279, 222–226. [Google Scholar] [CrossRef]
- Zhao, F.; He, J.; Tang, J.; Cui, N.; Shi, Y.; Li, Z.; Liu, S. Brain milieu induces early microglial maturation through the BAX-Notch axis. Nat. Commun. 2022, 13, 6117. [Google Scholar] [CrossRef]
- Zhou, Z.; Hong, E.J.; Cohen, S.; Zhao, W.; Ho, H.H.; Schmidt, L.; Chen, W.G.; Lin, Y.; Savner, E.; Griffith, E.C.; et al. Brain-specific phosphorylation of MeCP2 regulates activity-dependent Bdnf transcription, dendritic growth, and spine maturation. Neuron 2006, 52, 252–269. [Google Scholar] [CrossRef] [PubMed]
- Lee, L.C.; Su, M.T.; Huang, H.Y.; Cho, Y.C.; Yeh, T.K.; Chang, C.-Y. Association of CaMK2A and MeCP2 signaling pathways with cognitive ability in adolescents. Mol. Brain 2021, 14, 152. [Google Scholar] [CrossRef]
- Miles, J.H.; Hadden, L.L.; Takahashi, T.N.; Hillman, R.E. Head circumference is an independent clinical finding associated with autism. Am. J. Med. Genet. 2000, 95, 339–350. [Google Scholar] [CrossRef]
- Bolton, P.F.; Roobol, M.; Allsop, L.; Pickles, A. Association between idiopathic infantile macrocephaly and autism spectrum disorders. Lancet 2001, 358, P726–P727. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.C.; Chen, Y.; Page, D.T. Hyperconnectivity of prefrontal cortex to amygdala projections in a mouse model of macrocephaly/autism syndrome. Nat. Commun. 2016, 7, 13421. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, J.; Man, H.Y. Fundamental elements in autism: From neurogenesis and neurite growth to synaptic plasticity. Front. Cell Neurosci. 2017, 11, 359. [Google Scholar] [CrossRef]
- Giannandrea, M.; Bianchi, V.; Mignogna, M.L.; Sirri, A.; Carrabino, S.; d’Elia, E.; Vecellio, M.; Russo, S.; Cogliati, F.; Larizza, L.; et al. Mutations in the small GTPase gene RAB39B are responsible for X-linked mental retardation associated with autism, epilepsy, and macrocephaly. Am. J. Hum. Genet. 2010, 86, 185–195. [Google Scholar] [CrossRef]
- Marchese, M.; Valvo, G.; Moro, F.; Sicca, F.; Santorelli, F.M. Targeted Gene Resequencing (Astrochip) to explore the tripartite synapse in autism–epilepsy phenotype with macrocephaly. Neuromol. Med. 2016, 18, 69–80. [Google Scholar] [CrossRef]
- Fu, C.; Ngo, J.; Zhang, S.; Lu, L.; Miron, A.; Schafer, S.; Gage, F.H.; Jin, F.; Schumacher, F.R.; Wynshaw-Boris, A. Novel correlative analysis identifies multiple genomic variations impacting ASD with macrocephaly. Hum. Mol. Genet. 2023, 10, 1589–1606. [Google Scholar] [CrossRef]
- McCaffery, P.; Deutsch, C.K. Macrocephaly and the control of brain growth in autistic disorders. Prog. Neurobiol. 2005, 77, 38–56. [Google Scholar] [CrossRef]
- Baple, E.L.; Maroofian, R.; Chioza, B.A.; Izadi, M.; Cross, H.E.; Al-Turki, S.; Barwick, K.; Skrzpiec, A.; Pawlak, R.; Wagner, K.; et al. Mutations in KPTN cause macrocephaly, neurodevelopmental delay, and seizures. Am. J. Hum. Genet. 2014, 94, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S. Michael Faraday: Discovery of electromagnetic induction. Resonance 2002, 7, 35–45. [Google Scholar] [CrossRef]
- Purcell, E.M. Electricity and Magnetism, 2nd ed.; Berkeley Physics Course; McGraw-Hill: New York, NY, USA, 1985; Volume 2. [Google Scholar]
- Pall, M.L. Millimeter (MM) wave and microwave frequency radiation produce deeply penetrating effects: The biology and the physics. Rev. Environ. Health 2022, 37, 247–258. [Google Scholar] [CrossRef] [PubMed]
- Fröhlich, H. Long-range coherence and energy storage in biological systems. Int. J. Quantum Chem. 1968, 2, 641–649. [Google Scholar] [CrossRef]
- Brizhik, S. Influence of electromagnetic field on soliton-mediated charge transport in biological systems. Electromagn. Biol. Med. 2015, 34, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Pall, M.L. Electromagnetic fields act via activation of voltage-gated calcium channels to produce beneficial or adverse effects. J. Cell. Mol. Med. 2013, 17, 958–965. [Google Scholar] [CrossRef] [PubMed]
- Pilla, A.A. Nonthermal electromagnetic fields: From first messenger to therapeutic applications. Electromagn. Biol. Med. 2013, 32, 123–136. [Google Scholar] [CrossRef] [PubMed]
- Walleczek, J. Electromagnetic field effects on cells of the immune system: The role of calcium signaling. FASEB J. 1992, 6, 3177–3185. [Google Scholar] [CrossRef]
- Pall, M.L. Wi-Fi is an important threat to human health. Environ. Res. 2018, 164, 405–416. [Google Scholar] [CrossRef]
- Catterall, W. Voltage-gated sodium channels at 60: Structure, function and pathophysiology. J. Physiol. 2012, 590, 2577–2589. [Google Scholar] [CrossRef]
- Azarov, J.E.; Semenov, I.; Casciola, M.; Pakhomov, A.G. Excitation of murine cardiac myocytes by nanosecond pulsed electric field. J. Cardiovasc. Electrophysiol. 2019, 30, 392–401. [Google Scholar] [CrossRef]
- Pakhomov, A.G.; Pakhomova, O.N. The interplay of excitation and electroporation in nanosecond pulse stimulation. Bioelectrochemistry 2020, 136, 107598. [Google Scholar] [CrossRef] [PubMed]
- Pall, M.L. Low Intensity Electromagnetic Fields Act via Voltage-Gated Calcium Channel (VGCC) Activation to Cause Very Early Onset Alzheimer’s Disease: 18 Distinct Types of Evidence. Curr. Alzheimer Res. 2022, 19, 119–132. [Google Scholar] [CrossRef] [PubMed]
- El-Swefy, S.; Soliman, H.; Huessein, M. Calcium channel blockade alleviates brain injury induced by long term exposure to an electromagnetic field. J. Appl. Biomed. 2008, 6, 153–163. [Google Scholar] [CrossRef]
- Kane, R.C. A possible association between fetal/neonatal exposure to radiofrequency electromagnetic radiation and the increased frequency of autism spectrum disorders (ASD). Med. Hypotheses 2004, 62, 195–197. [Google Scholar] [CrossRef]
- McGinnis, W.R. Oxidative stress in autism. Integr. Med. 2005, 3, 42–57. [Google Scholar]
- Thornton, I.M. Out of time: A possible link between mirror neurons, autism and electromagnetic radiation. Med. Hypotheses 2006, 67, 378–382. [Google Scholar] [CrossRef] [PubMed]
- Klinghardt, D. Autism May Be Linked to Electromagnetic Radiation Levels in Mother’s Bedroom during Pregnancy. 2008. Available online: https://electromagnetichealth.org/media-stories/#Autism (accessed on 10 October 2023).
- Lathe, R. Environmental factors and limbic vulnerability in childhood. Am. J. Biochem. Biotechnol. 2008, 4, 183–197. [Google Scholar] [CrossRef][Green Version]
- Currenti, S.A. Understanding and determining the etiology of autism. Cell. Mol. Neurobiol. 2010, 30, 161–171. [Google Scholar] [CrossRef]
- Goldworthy, A. How Electromagnetically-Induced Cell Leakage May Cause Autism. 2011. Available online: http://mcs-america.org/June2011pg2345.pdf (accessed on 8 June 2023).
- Herbert, M.R.; Sage, C. Autism and EMF? Plausibility of a pathophysiological link part I. Pathophysiology 2013, 20, 191–209. [Google Scholar] [CrossRef] [PubMed]
- Herbert, M.R.; Sage, C. Autism and EMF? Plausibility of a pathophysiological link part II. Pathophysiology 2013, 20, 211–234. [Google Scholar] [CrossRef] [PubMed]
- Ahuja, Y.R.; Sharma, S.; Bahadur, B. Autism: An epigenomic side-effect of excessive exposure to electromagnetic fields. Int. J. Med. Med. Sci. 2013, 5, 171–177. [Google Scholar]
- Bellieni, C.V.; Buonocore, G. Are electromagnetic fields in incubators a risk factor for autism? Acta Paediatr. 2017, 106, 2062. [Google Scholar] [CrossRef] [PubMed]
- Pritchard, C.; Silk, A.; Hansen, L. Are rises in Electro-Magnetic Field in the human environment, interacting with multiple environmental pollutions, the tripping point for increases in neurological deaths in the Western World? Med. Hypotheses 2019, 127, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Frey, A.H. Differential biologic effects of pulsed and continuous electromagnetic fields and mechanisms of effect. Ann. N. Y. Acad. Sci. 1974, 238, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Creighton, M.O.; Larsen, L.E.; Stewart-DeHaan, P.J.; Jacobi, J.H.; Sanwal, M.; Baskerville, J.C.; Bassen, H.E.; Brown, D.O.; Trevithick, J.R. In vitro studies of microwave-induced cataract. II. Comparison of damage observed for continuous wave and pulsed microwaves. Exp. Eye Res. 1987, 45, 357–373. [Google Scholar] [CrossRef] [PubMed]
- Belyaev, I. Non-thermal biological effects of microwaves: Current knowledge, further perspective and urgent needs. Electromagn. Biol. Med. 2005, 24, 375–403. [Google Scholar] [CrossRef]
- Belyaev, I. Biophysical mechanisms for nonthermal microwave effects. In Electromagnetic Fields in Biology and Medicine; Markov, M.S., Ed.; CRC Press: New York, NY, USA, 2015; pp. 49–67. [Google Scholar]
- Van Boxem, K.; Huntoon, M.; Van Zundert, J.; Patijn, J.; van Kleef, M.; Joosten, E.A. Pulsed radiofrequency: A review of the basic science as applied to the pathophysiology of radicular pain: A call for clinical translation. Reg. Anesth. Pain Med. 2014, 39, 149–159. [Google Scholar] [CrossRef]
- Barrett, C.F.; Tsien, R.W. The Timothy syndrome mutation differentially affects voltage- and calcium-dependent inactivation of Cav1.2 L-type calcium channels. Proc. Natl. Acad. Sci. USA 2008, 105, 2157–2162. [Google Scholar] [CrossRef]
- Splawski, I.; Timothy, K.W.; Sharpe, L.M.; Decher, N.; Kumar, P.; Bloise, R.; Napolitano, C.; Schwartz, P.J.; Joseph, R.M.; Conduris, K.; et al. Ca(V)1.2 calcium channel dysfunction causes a multisystem disorder including arrhythmia and autism. Cell 2004, 119, 19–31. [Google Scholar] [CrossRef] [PubMed]
- Depil, K.; Beyl, S.; Stary-Weinzinger, A.; Hohaus, E.; Timin, E.; Hering, S. Timothy mutation disrupts the link between activation and inactivation in Cav1.2 protein. J. Biol. Chem. 2011, 286, 31557–31564. [Google Scholar] [CrossRef] [PubMed]
- Bader, P.L.; Faizi, M.; Kim, L.H.; Owen, S.F.; Tadross, M.R.; Alfa, R.W.; Bett, G.C.; Tsien, R.W.; Rasmussen, R.L.; Shamloo, M. Mouse model of Timothy syndrome recapitulates triad of autistic traits. Proc. Natl. Acad. Sci. USA 2011, 108, 15432–15437. [Google Scholar] [CrossRef]
- Breitenkamp, A.F.; Mathes, J.; Herzig, S. Voltage-gated calcium channels and autism spectrum disorders. Curr. Mol. Pharm. 2015, 8, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Ma, Q.; Liu, C.; Deng, P.; Zhu, G.; Zhang, L.; He, M.; Lu, Y.; Duan, W.; Li, M.; et al. Exposure to 1800 MHz radiofrequency radiation impairs neurite outgrowth of embryonic neural stem cells. Sci. Rep. 2014, 4, 5103. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Chen, C.; Deng, P.; Zhu, G.; Lin, M.; Zhang, L.; Xu, S.; He, M.; Lu, Y.; Duan, W.; et al. Extremely low-frequency electromagnetic fields promote in vitro neuronal differentiation and neurite outgrowth of embryonic neural stem cells via up-regulating TRPC1. PLoS ONE 2016, 11, e0150923. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Ma, Q.; Deng, P.; Lin, M.; Gao, P.; He, M.; Lu, Y.; Duan, W.; Cao, Z.; Pei, L.; et al. 1800 MHz radiofrequency electromagnetic field impairs neurite outgrowth through inhibiting EPHA5 signaling. Front. Cell Dev. Biol. 2021, 9, 657623. [Google Scholar] [CrossRef] [PubMed]
- Eghlidospour, M.; Ghanbari, A. Effects of radiofrequency exposure emitted from a GSM mobile phone on proliferation, differentiation, and apoptosis of neural stem cells. Anat. Cell Biol. 2017, 50, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Zhu, R.; Sun, Z.; Li, C.; Ramakrishna, S.; Chiu, K.; He, L. Electrical stimulation affects neural stem cell fate and function in vitro. Exp. Neurol. 2019, 319, 112963. [Google Scholar] [CrossRef]
- Li, Y.; Deng, P.; Chen, C.; Ma, Q.; Pi, H.; He, M.; Lu, Y. 1800 MHz radiofrequency electromagnetic irradiation impairs neurite outgrowth with a decrease in Rap1-GTP in primary mouse hippocampal neurons and Neuro2a cells. Front. Public Health 2021, 9, 771508. [Google Scholar] [CrossRef]
- Akaneya, Y.; Sohya, K.; Kitamura, A.; Kimura, F.; Washburn, C.; Zhou, R. Ephrin-A5 and EphA5 interaction induces synaptogenesis during early hippocampal development. PLoS ONE 2010, 5, e12486. [Google Scholar] [CrossRef] [PubMed]
- Abdul-Wajid, S.; Morales-Diaz, H.; Khairallah, S.M.; Smith, W.C. T-type calcium channel regulation of neural tube closure and EphrinA/EPHA expression. Cell Rep. 2015, 13, 829–839. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, S.; Deniz, O.G.; Önger, M.E.; Türkmen, A.P.; Yurt, K.K.; Aydin, I.; Altunkaynak, B.Z.; Davis, D. Electromagnetic field and brain development. J. Chem. Neuroanat. 2016, 75 Pt B, 52–61. [Google Scholar] [CrossRef]
- Bertrand, J.; Mars, A.; Boyle, C.; Bove, F.; Yeargin-Allsopp, M.; Decoufle, P. Prevalence of autism in the United States population: The Brick Township, New Jersey investigation. Pediatrics 2001, 108, 1155. [Google Scholar] [CrossRef] [PubMed]
- Roelfsema, M.T.; Hoekstra, R.A.; Allison, C.; Wheelright, S.; Brayne, C.; Matthews, F.E.; Baron-Cohen, S. Are autism spectrum conditions more prevalent in an information-technology region? A school-based study of three regions in the Netherlands. J. Autism Dev. Disord. 2011, 42, 734–739. [Google Scholar] [CrossRef] [PubMed]
- Zarei, S.; Mortazavi, S.M.J.; Mehdizadeh, A.R.; Jalalipour, M.; Borzou, S.; Taeb, S.; Haghani, M.; Mortazavi, S.A.R.; Shojaei-fard, M.B.; Nematollahi, S.; et al. A Challenging Issue in the Etiology of Speech Problems: The Effect of Maternal Exposure to Electromagnetic Fields on Speech Problems in the Offspring. J. Biomed. Phys. Eng. 2015, 5, 151–154. [Google Scholar] [PubMed]
- Zarei, S.; Vahab, M.; Oryadi-Zanjani, M.M.; Alighanbari, N.; Mortazavi, S.M. Mother’s Exposure to Electromagnetic Fields before and during Pregnancy is Associated with Risk of Speech Problems in Offspring. J. Biomed. Phys. Eng. 2019, 9, 61–68. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Alsaeed, I.; Al-Somali, F.; Sakhnini, L.; Aljarallah, O.S.; Hamdan, R.M.; Bubishate, S.A.; Sarfaraz, Z.K.; Kamal, A. Autism-relevant social abnormalities in mice exposed perinatally to extremely low frequency electromagnetic fields. Int. J. Dev. Neurosci. 2014, 37, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Acikgoz, B.; Ersoy, N.; Aksu, I.; Kiray, A.; Bagriyanik, H.A.; Kiraya, M. Gender differences in effects of prenatal and postnatal exposure to electromagnetic field and prenatal zinc on behaviour and synaptic proteins in rats. J. Chem. Neuroanat. 2022, 122, 102092. [Google Scholar] [CrossRef]
- Herbert, M.R. SHANK3, the synapse, and autism. N. Engl. J. Med. 2011, 365, 173–175. [Google Scholar] [CrossRef]
- Othman, H.; Ammari, M.; Rtibi, K.; Bensaid, N.; Sakly, M.; Abdelmelek, H. Postnatal development and behavior effects of in-utero exposure of rats to radiofrequency waves emitted from conventional WiFi devices. Environ. Toxicol. Pharmacol. 2017, 52, 239–247. [Google Scholar] [CrossRef]
- Othman, H.; Ammari, M.; Sakly, M.; Abdelmelek, H. Effects of prenatal exposure to WIFI signal (2.45GHz) on postnatal development and behavior in rat: Influence of maternal restraint. Behav. Brain Res. 2017, 326, 291–302. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.; Huang, H.; Yang, M.; Wu, H.; Wang, L. Enriched environment decreases cognitive impairment in elderly rats with prenatal mobile phone exposure. Front. Aging Neurosci. 2020, 12, 162. [Google Scholar] [CrossRef] [PubMed]
- Woo, C.C.; Donnelly, J.H.; Steinberg-Epstein, R. Environmental enrichment as a therapy for autism: A clinical trial replication and extension. Behav. Neurosci. 2015, 129, 412–422. [Google Scholar] [CrossRef]
- Aronoff, E.; Hillyer, R.; Leon, M. Environmental enrichment therapy for autism: Outcomes with increased access. Neural Plast. 2016, 2016, 2734915. [Google Scholar] [CrossRef]
- Aldad, T.S.; Gan, G.; Gao, X.B.; Taylor, H.S. Fetal radiofrequency radiation exposure from 800–1900 MHz-rated cellular telephones affects neurodevelopment and behavior in mice. Sci. Rep. 2012, 2, 312. [Google Scholar] [CrossRef]
- Kern, J.K.; Geier, D.A.; Sykes, L.K.; Geier, M.R.; Deth, R.C. Are ASD and ADHD a continuum? A comparison of pathophysiological similarities between the disorders. J. Atten. Disord. 2015, 19, 805–827. [Google Scholar] [CrossRef] [PubMed]
- Krey, J.F.; Paşca, S.P.; Shcheglovitov, A.; Yazawa, M.; Schwemberger, R.; Rasmussen, R.; Dolmetsch, R.F. Timothy syndrome is associated with activity-dependent dendritic retraction in rodent and human neurons. Nat. Neurosci. 2013, 16, 201–209. [Google Scholar] [CrossRef]
- Striessnig, J.; Pinggera, A.; Kaur, G.; Bock, G.; Tuluc, P. L-type Ca2+ channels in heart and brain. Wiley Interdiscip. Rev. Membr. Transp. Signal. 2014, 3, 15–38. [Google Scholar] [CrossRef]
- Kobayashi, T.; Yamada, Y.; Fukao, M.; Tsutsuura, M.; Tohse, N. Regulation of Cav1.2 current: Interaction with intracellular molecules. J. Pharm. Sci. 2007, 103, 347–353. [Google Scholar] [CrossRef]
- Lee, E.-J.; Choi, S.Y.; Kim, E. NMDA receptor dysfunction in autism spectrum disorders. Curr. Opin. Pharm. 2015, 20, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Yoo, H.J.; Cho, I.H.; Park, M.; Yang, S.Y.; Kim, S.A. Family Based Association of GRIN2A and GRIN2B with Korean autism spectrum disorders. Neurosci. Lett. 2012, 512, 89–93. [Google Scholar] [CrossRef]
- Nisar, S.; Bhat, A.A.; Masoodi, T.; Hashem, S.; Akhtar, S.; Ali, T.A.; Amjad, S.; Chawla, S.; Bagga, P.; Frenneaux, M.P.; et al. Genetics of glutamate and its receptors in autism spectrum disorder. Mol. Psychiatry 2022, 27, 2380–2392. [Google Scholar] [CrossRef] [PubMed]
- Lim, C.S.; Kim, M.J.; Choi, J.E.; Islam, M.A.; Lee, Y.K.; Xiong, Y.; Shim, K.W.; Yang, J.E.; Lee, R.U.; Lee, J.; et al. Dysfunction of NMDA receptors in neuronal models of an autism spectrum disorder patient with a DSCAM mutation and in Dscam-knockout mice. Mol. Psychiatry 2021, 26, 7539–7549. [Google Scholar] [CrossRef] [PubMed]
- Hussman, J.P. Suppressed GABAergic inhibition as a common factor in suspected etiologies of autism. J. Autism Dev. Disord. 2001, 31, 247–248. [Google Scholar] [CrossRef]
- Tarabeux, J.; Kebir, O.; Gauthier, J.; Hamdan, F.F.; Xiong, L.; Piton, A.; Spiegelman, D.; Henrion, E.; Millet, B.; Fathalli, F.; et al. Rare mutations in N-methyl-D-aspartate glutamate receptors in autism spectrum disorders and schizophrenia. Transl. Psychiatry 2011, 1, e55. [Google Scholar] [CrossRef] [PubMed]
- Deutsch, S.I.; Luyo, Z.N.M.; Burket, J.A. Targeted NMDA receptor interventions for autism: Developmentally determined expression of GluN2B and GluN2A-containing receptors and balanced allosteric modulatory approaches. Biomolecules 2022, 12, 181. [Google Scholar] [CrossRef]
- Wesseling, H.; Guest, P.C.; Lee, C.M.; Wong, E.H.F.; Rahmoune, H.; Bahm, S. Integrative proteomic analysis of the NMDA NR1 knockdown mouse model reveals effects on central and peripheral pathways associated with schizophrenia and autism spectrum disorders. Mol. Autism 2014, 5, 38. [Google Scholar] [CrossRef] [PubMed]
- McDougle, C.J.; Posey, D. Genetics of childhood disorders: XLIV. autism, part 3: Psychopharmacology of autism. J. Am. Acad. Child Adolesc. Psychiatry 2002, 41, 1380–1383. [Google Scholar] [CrossRef][Green Version]
- Sceniak, M.P.; Fedder, K.N.; Wang, Q.; Droubi, S.; Babcock, K.; Patwardhan, S.; Wright-Zornes, J.; Pham, L.; Sabo, S.L. An autism-associated mutation in GluN2B prevents NMDA receptor trafficking and interferes with dendrite growth. J. Cell Sci. 2019, 132, jcs232892. [Google Scholar] [CrossRef]
- Ransom, R.W.; Deschenes, N.L. Polyamines regulate glycine interaction with the N-methyl-D-aspartate receptor. Synapse 1990, 5, 294–298. [Google Scholar] [CrossRef]
- Guo, H.; Camargo, L.M.; Yeboah, F.; Digan, M.E.; Niu, H.; Pan, Y.; Reiling, S.; Soler-Llavina, G.; Weihofen, W.A.; Wang, H.-R.; et al. A NMDA-receptor calcium influx assay sensitive to stimulation by glutamate and glycine/D-serine. Sci. Rep. 2017, 7, 11608. [Google Scholar] [CrossRef]
- Behl, S.; Mehta, S.; Pandey, M.K. Abnormal levels of metal micronutrients and autism spectrum disorder: A perspective review. Front. Mol. Neurosci. 2020, 13, 586209. [Google Scholar] [CrossRef]
- Galland, L. Magnesium, stress and neuropsychiatric disorders. Magnes. Trace Elem. 1991, 10, 287–301. [Google Scholar]
- Skalny, A.V.; Mazaletskaya, A.L.; Ajsuvakova, O.P.; Bjorklund, G.; Skalnaya, M.G.; Chernova, L.N.; Skalny, A.A.; Tinkov, A.A. Magnesium status in children with attention-deficit/hyperactivity disorder and/or autism spectrum disorder. J. Korean Acad. Child Adolesc. Psychiatry 2020, 31, 41–45. [Google Scholar] [CrossRef]
- Botturi, A.; Ciappolino, V.; Delvecchio, G.; Boscutti, A.; Viscardi, B.; Brambilla, P. The role and the effect of magnesium in mental disorders: A systematic review. Nutrients 2020, 12, 1661. [Google Scholar] [CrossRef]
- Poddar, R.; Paul, S. Homocysteine–NMDA receptor-mediated activation of extracellular signal-regulated kinase leads to neuronal cell death. J. Neurochem. 2009, 110, 1095–1106. [Google Scholar] [CrossRef]
- Kałużna-Czaplińska, J.; Żurawicz, E.; Michalska, M.; Rynkowski, J. A focus on homocysteine in autism. Acta Biochim. Pol. 2013, 60, 137–142. [Google Scholar] [CrossRef]
- Guo, B.-Q.; Li, H.-B.; Ding, S.-B. Blood homocysteine levels in children with autism spectrum disorder: An updated systematic review and meta-analysis. Psychiatr. Res. 2020, 291, 113283. [Google Scholar] [CrossRef] [PubMed]
- Ghanizadeh, A. Increased glutamate and homocysteine and decreased glutamine levels in autism: A review and strategies for future studies of amino acids in autism. Curr. Probl. Pediatr. Adolesc. Health Care Dis. Markers 2013, 35, 536521. [Google Scholar] [CrossRef] [PubMed]
- Kalkbrenner, A.E.; Schmidt, R.J.; Penlesky, A.C. Environmental chemical exposures and autism spectrum disorders: A review of the epidemiological evidence. Curr. Probl. Pediatr. Adolesc. Health Care 2014, 44, 277–318. [Google Scholar] [CrossRef]
- Rossignol, D.A.; Genuis, S.J.; Frye, R.E. Environmental toxicants and autism spectrum disorders: A systematic review. Transl. Psychiatry 2014, 4, e360. [Google Scholar] [CrossRef]
- Fujiwara, T.; Morisaki, N.; Honda, Y.; Sampei, M.; Tani, Y. Chemicals, nutrition, and autism spectrum disorder: A mini-review. Front. Neurosci. 2016, 10, 174. [Google Scholar] [CrossRef]
- Ongono, J.S.; Beranger, R.; Baghdad, A. Pesticides used in Europe and autism spectrum disorder risk: Can novel exposure hypotheses be formulated beyond organophosphates, organochlorines, pyrethroids and carbamates?—A systematic review. Environ. Res. 2020, 187, 109646. [Google Scholar] [CrossRef]
- Pall, M.L. Multiple chemical sensitivity: Toxicological questions and mechanisms. In General and Applied Toxicology, 3rd ed.; John Wiley & Sons: Chichester, UK, 2009; pp. 2303–2352. [Google Scholar]
- Zhong, X.; Li, J.; Zhuang, Z.; Shen, Q.; Jiang, K.; Hu, Y.; Wu, D.; Xu, X. Rapid effect of bisphenol A on glutamate-induced Ca2+ influx in hippocampal neurons of rats. Mol. Cell. Endocrinol. 2019, 485, 35–43. [Google Scholar] [CrossRef]
- Xu, X.; Li, T.; Luo, Q.; Hong, X.; Xie, L.; Tian, D. Bisphenol-A rapidly enhanced passive avoidance memory and phosphorylation of NMDA receptor subunits in hippocampus of young rats. Toxicol. Appl. Pharmacol. 2011, 255, 221–228. [Google Scholar] [CrossRef]
- Eilam-Stock, T.; Serrano, P.; Frankfurt, M.; Luine, V. Bisphenol-A impairs memory and reduces dendritic spine density in adult male rats. Behav. Neurosci. 2012, 126, 175–185. [Google Scholar] [CrossRef]
- Carbone, S.E.; Ponzo, O.J.; Cutrera, R.A. Effect of N-methyl-D-aspartate receptor blockade on anxiety-like behavior induced in rats by postnatal chronic exposure to the endocrine disruptor di-2 (ethyl-hexyl phthalate) in elevated plus maze test. Res. Open World EDMJ 2019, 3, 1–6. [Google Scholar]
- Lehmann, J.; Ferkany, J.W.; Schaeffer, P.; Coyle, J.T. Dissociation between the excitatory and “excitotoxic” effects of quinolinic acid analogs on the striatal cholinergic interneuron. J. Pharmacol. Exp. Ther. 1985, 232, 873–882. [Google Scholar]
- Mohammadi, H.; Ashari, S. Mechanistic insight into toxicity of phthalates, the involved receptors, and the role of Nrf2, NF-κB, and PI3K/AKT signaling pathways. Environ. Sci. Pollut. Res. 2021, 28, 35488–35527. [Google Scholar] [CrossRef]
- Zhu, M.-M.; Li, H.-L.; Shi, L.-H.; Chen, X.-P.; Luo, J.; Zhang, Z.-L. The pharmacogenomics of valproic acid. J. Hum. Genet. 2017, 62, 1009–1014. [Google Scholar] [CrossRef] [PubMed]
- Christensen, J.; Grønborg, T.K.; Sørensen, M.J.; Schendel, D.; Parner, E.T.; Pederson, L.H.; Vestergaard, M. Prenatal valproate exposure and risk of autism spectrum disorders and childhood autism. JAMA 2013, 309, 1696–1703. [Google Scholar] [CrossRef]
- Kumar, H.; Diwan, V.; Sharma, B. Protective effect of nimodipine against valproic acid induced biochemical and behavioral phenotypes of autism. Clin. Psychopharmacol. Neurosci. 2022, 20, 725–736. [Google Scholar] [CrossRef] [PubMed]
- Abbasalipour, H.; Moghaddam, A.H.; Ranjbar, M. Sumac and gallic acid-loaded nanophytosomes ameliorate hippocampal oxidative stress via regulation of Nrf2/Keap1 pathway in autistic rats. J. Biochem. Mol. Toxicol. 2022, 36, e23035. [Google Scholar] [CrossRef] [PubMed]
- Shaw, W. Evidence that increased acetaminophen use in genetically vulnerable children appears to be a major cause of the epidemics of autism, attention deficit with hyperactivity, and asthma. J. Restor. Med. 2013, 2, 14–29. [Google Scholar] [CrossRef][Green Version]
- Avella-Garcia, C.B.; Julvez, J.; Fortuny, J.; Rebordosa, C.; García-Esteban, R.; Galan, I.R.; Tardon, A.; Rodriguez-Bernal, C.L.; Iniguez, C.; Andiarena, A.; et al. Acetaminophen use in pregnancy and neurodevelopment: Attention function and autism spectrum symptoms. Int. J. Epidemiol. 2016, 45, 1987–1996. [Google Scholar] [CrossRef] [PubMed]
- Seneff, S.; Davidson, R.M.; Liu, J. Empirical data confirm autism symptoms related to aluminum and acetaminophen exposure. Entropy 2012, 14, 2227–2253. [Google Scholar] [CrossRef]
- James, L.P.; Mayeux, P.R.; Hinson, J.A. Acetaminophen-induced hepatotoxicity. Drug Metab. Disp. 2003, 31, 1499–1506. [Google Scholar] [CrossRef] [PubMed]
- Larson, A.M. Acetaminophen hepatotoxicity. Clin. Liver Dis. 2007, 11, 525–548. [Google Scholar] [CrossRef]
- Marino, R.T.; Sidlak, A.M. Hyperammonemia in acetaminophen toxicity. Clin. Toxicol. 2022, 60, 126–130. [Google Scholar] [CrossRef]
- Haley, B.E. Mercury toxicity: Genetic susceptibility and synergistic effects. Med. Veritas 2005, 2, 535–542. [Google Scholar] [CrossRef]
- Segal, J.A.; Harris, B.D.; Kustova, Y.; Basile, A.; Skolnick, P. Aminoglycoside neurotoxicity involves NMDA receptor activation. Brain Res. 1999, 815, 270–275. [Google Scholar] [CrossRef]
- Pessah, I.N.; Cherednichenko, G.; Lein, P.J. Minding the calcium store: Ryanodine receptor activation as a convergent mechanism of PCB toxicity. Pharmacol. Ther. 2010, 125, 260–285. [Google Scholar] [CrossRef]
- Brown-Leung, J.M.; Cannon, J.R. Neurotransmission targets of per- and polyfluoroalkyl substance neurotoxicity: Mechanisms and potential implications for adverse neurological outcomes. Chem. Res. Toxicol. 2022, 35, 1312–1333. [Google Scholar] [CrossRef]
- Cao, Y.; Ng, C. Absorption, distribution, and toxicity of per- and polyfluoroalkyl substances (PFAS) in the brain: A review. Environ. Sci. Process. Impacts 2021, 23, 1623–1640. [Google Scholar] [CrossRef]
- Boretti, A. Reviewing the association between aluminum adjuvants in the vaccines and autism spectrum disorder. J. Trace Elem. Med. Biol. 2021, 66, 126764. [Google Scholar] [CrossRef]
- Kaur, I.; Behl, T.; Aleya, L.; Rahman, M.H.; Kumar, A.; Arora, S.; Akter, R. Role of metallic pollutants in neurodegeneration: Effects of aluminum, lead, mercury, and arsenic in mediating brain impairment events and autism spectrum disorder. Environ. Sci. Pollut. Res. 2021, 28, 8989–9001. [Google Scholar] [CrossRef]
- Skalny, A.V.; Aschner, M.; Jiang, Y.; Gluhcheva, Y.G.; Tizabi, Y.; Lobinski, R.; Tinkov, A.A. Molecular mechanisms of aluminum neurotoxicity: Update on adverse effects and therapeutic strategies. Adv. Neurotoxicol. 2021, 5, 1–34. [Google Scholar]
- Gatta, V.; Drago, D.; Fincati, K.; Valenti, M.T.; Dalle Carbonare, L.; Sensi, S.L.; Zatta, P. Microarray analysis on human neuroblastoma cells exposed to aluminum, β(1–42)-amyloid or the β(1–42)-amyloid aluminum complex. PLoS ONE 2011, 6, e15965. [Google Scholar] [CrossRef]
- Exley, C. Human exposure to aluminium. Environ. Sci. Process. Impacts 2013, 15, 1807–1816. [Google Scholar] [CrossRef]
- Mundy, W.R.; Kodavanki, P.R.S.; Dulchinos, V.F.; Tilson, H.A. Aluminum alters transport in plasma membrane and endoplasmic reticulum from rat brain. J. Biochem. Toxicol. 1994, 9, 17–23. [Google Scholar] [CrossRef]
- De Sautu, M.; Saffioti, N.A.; Ferreira-Gomes, M.S.; Rossi, R.C.; Rossi, P.J.F.C.; Mangialavori, I.C. Aluminum inhibits the plasma membrane and sarcoplasmic reticulum Ca2+-ATPases by different mechanisms. Biochim. Biophys. Acta—Biomembr. 2018, 1860, 1880–1888. [Google Scholar] [CrossRef]
- Cavalli, V.L.L.O.; Cattani, D.; Rieg, C.E.H.; Pierozan, P.; Zanatta, L.; Parisatto, E.B.; Wlihelm Filho, D.; Silva, F.R.M.B.; Pessoa-Pureur, R.; Zamoner, A. Roundup disrupts male reproductive functions by triggering calcium-mediated cell death in rat testis and Sertoli cells. Free Rad. Biol. Med. 2012, 65, 335–346. [Google Scholar] [CrossRef]
- Cattani, D.; De Olivera, P.A.; Cavalli, V.L.L.O.; Heinz Rieg, C.E.; Domingues, J.T.; Dal-Cim, T.; Tasca, C.I.; Silva, F.R.M.B.; Zamoner, A. Mechanisms underlying the neurotoxicity induced by glyphosate-based herbicide in immature rat hippocampus: Involvement of glutamate excitotoxicity. Toxicology 2014, 320, 34–45. [Google Scholar] [CrossRef]
- Gao, H.; Chen, J.; Ding, F.; Chou, X.; Zhang, X.; Wan, Y.; Hu, J.; Wu, Q. Activation of the N-methyl-d-aspartate receptor is involved in glyphosate-induced renal proximal tubule cell apoptosis. J. Appl. Toxicol. 2019, 39, 1096–1107. [Google Scholar] [CrossRef]
- Beecham, J.E.; Seneff, S. The possible link between autism and glyphosate acting as glycine mimetic—A review of evidence from the literature with analysis. J. Mol. Genet. Med. 2015, 9, 187. [Google Scholar]
- Cattani, D.; Cesconetto, P.A.; Tavares, M.K.; Parisotto, E.B.; De Olivera, P.A.; Heinz Rieg, C.E.; Leite, M.C.; Prediger, R.D.S.; Wendt, N.C.; Razzera, G.; et al. Developmental exposure to glyphosate-based herbicide and depressive-like behavior in adult offspring: Implication of glutamate excitotoxicity and oxidative stress. Toxicology 2017, 387, 67–80. [Google Scholar] [CrossRef]
- Hultberg, M. Cysteine turnover in human cell lines is influenced by glyphosate. Environ. Toxicol. Pharmacol. 2007, 24, 19–22. [Google Scholar] [CrossRef]
- Demirkaya, S.K.; Orak, C.; Şirinyıldız, F.; Ferhat, S.; Taşlı, Y.G. Effects of riluzole and gabapentin on spatial learning, locomotor activity of anxiety and social behaviours in autism-like rat model. Psychopharmacology 2019, 29, 121–122. [Google Scholar]
- Cherubini, E.; Di Cristo, G.; Avoli, M. Dysregulation of GABAergic signaling in neurodevelomental disorders: Targeting cation-chloride co-transporters to re-establish a proper E/I balance. Front. Cell. Neurosci. 2022, 15, 813441. [Google Scholar] [CrossRef]
- Hwang, H.M.; Ku, R.Y.; Hashimoto-Torii, K. Prenatal Environment That Affects Neuronal Migration. Front. Cell Dev. Biol. 2019, 7, 138. [Google Scholar] [CrossRef]
- Ang, E.S.B.C., Jr.; Gluncic, V.; Duque, A.; Rakic, P. Prenatal exposure to ultrasound waves impacts neuronal migration in mice. Proc. Natl. Acad. Sci. USA 2006, 103, 12903–12910. [Google Scholar] [CrossRef] [PubMed]
- Huguet, G.; Ey, E.; Bourgeron, T. The genetic landscapes of autism spectrum disorders. Annu. Rev. Genom. Hum. Genet. 2013, 14, 191–213. [Google Scholar] [CrossRef]
- Crawley, J.N. Translational animal models of autism and neurodevelopmental disorders. Dialogues Clin. Neurosci. 2012, 14, 293–305. [Google Scholar] [CrossRef] [PubMed]
- Persico, A.M.; Napolioni, V. Autism genetics. Behav. Brain Res. 2013, 251, 95–112. [Google Scholar] [CrossRef]
- Pall, M.L. Elevated, sustained peroxynitrite levels as the cause of chronic fatigue syndrome. Med. Hypotheses 2000, 54, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Pall, M.L. Explaining “Unexplained Illnesses”: Disease Paradigm for Chronic Fatigue Syndrome, Multiple Chemical Sensitivity, Fibromyalgia, Post-Traumatic Stress Disorder, Gulf War Syndrome and Others; Harrington Park Press: New York, NY, USA, 2007. [Google Scholar]
- Pall, M.L. The NO/ONOO-cycle as the central cause of heart failure. Int. J. Mol. Sci. 2013, 14, 22274–22330. [Google Scholar] [CrossRef]
- Brown, G.C.; Bal-Price, A. Inflammatory neurodegeneration mediated by nitric oxide, glutamate, and mitochondria. Mol. Neurobiol. 2003, 27, 325–355. [Google Scholar] [CrossRef] [PubMed]
- Brown, G.C.; Neher, J.J. Inflammatory neurodegeneration and mechanisms of microglial killing of neurons. Mol. Neurobiol. 2010, 41, 242–247. [Google Scholar] [CrossRef]
- Golomb, B. Oxidative stress and mitochondrial injury in chronic multisymptom conditions: From gulf war illness to autism spectrum disorder. Nat. Preced. 2012. [Google Scholar] [CrossRef]
- Sajdel-Sulkowska, E.M.; Lipinski, B.; Windom, H.; Audhya, T.; McGinnis, W. Oxidative stress in autism: Elevated cerebellar 3-nitrotyrosine levels. Am. J. Biochem. Biotechnol. 2008, 4, 73–84. [Google Scholar]
- De Felice, A.; Greco, A.; Calamandrei, G.; Minghetti, L. Prenatal exposure to the organophosphate insecticide chlorpyrifos enhances brain oxidative stress and prostaglandin E2 synthesis in a mouse model of idiopathic autism. J. Neuroinflammation 2016, 13, 149. [Google Scholar] [CrossRef] [PubMed]
- Nadeem, A.; Ahmad, S.F.; Al-Harbi, N.O.; Attia, S.M.; Alshammari, M.A.; Alzahrani, K.S.; Bakheet, S.A. Increased oxidative stress in the cerebellum and peripheral immune cells leads to exaggerated autism-like repetitive behavior due to deficiency of antioxidant response in BTBR T + tf/J mice. Prog. Neuropsychopharmacol. Biol. Psychiatry 2019, 89, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Nadeem, A.; Ahmad, S.F.; Al-Ayadhi, L.Y.; Attia, S.M.; Al-Harbi, N.O.; Alzahrani, K.S.; Bakheet, S.A. Differential regulation of Nrf2 is linked to elevated inflammation and nitrative stress in monocytes of children with autism. Psychoneuroendocrinology 2020, 113, 104554. [Google Scholar] [CrossRef] [PubMed]
- Algahtani, M.M.; Sheikh, F.; Ahmad, S.F.; Alkharashi, L.A.; Al-Harbi, N.O.; Alanazi, W.A.; Alhamed, A.S.; Attia, S.M.; Bakheet, S.A.; Ibrahim, K.E.; et al. Exposure to methylmercury at juvenile stage worsens autism-like symptoms in adult BTBR T+tf/J mice due to lack of nuclear factor erythroid 2-related factor 2 signaling upregulation in periphery and brain. Toxics 2023, 11, 546. [Google Scholar] [CrossRef] [PubMed]
- Carey, M.E.; Rando, J.; Melnyk, S.; James, S.J.; Snyder, N.; Salafia, C.; Croen, L.A.; Fallin, M.D.; Hertz-Picciotto, I.; Volk, H.; et al. Examining associations between prenatal biomarkers of oxidative stress and ASD-related outcomes using quantile regression. J. Autism Dev. Disord. 2023, 53, 2975–2985. [Google Scholar] [CrossRef] [PubMed]
- Rezk, B.M.; Haenen, G.R.M.M.; van der Vijgh, W.J.F.; Bast, A. Tetrahydrofolate and 5-methyltetrahydrofolate are folates with high antioxidant activity. Identification of the antioxidant pharmacophore. FEBS Lett. 2003, 555, 601–605. [Google Scholar] [CrossRef] [PubMed]
- Antoniades, C.; Shirodaria, C.; Warrick, N.; Cai, S.; de Bono, J.; Lee, J.; Leeson, P.; Neubauer, S.; Ratnatunga, C.; Pillai, R.; et al. 5-Methyltetrahydrofolate rapidly improves endothelial function and decreases superoxide production in human vessels. Effects on vascular tetrahydrobiopterin availability and endothelial nitric oxide synthase coupling. Circulation 2006, 114, 1193–1201. [Google Scholar] [CrossRef] [PubMed]
- James, S.J.; Cutler, P.; Melnyk, S.; Jernigan, S.; Janak, S.L.; Gaylor, D.W.; Neubrander, J.A. Metabolic biomarkers of increased oxidative stress and impaired methylation capacity in children with autism. Am. J. Clin. Nutr. 2004, 80, 1611–1617. [Google Scholar] [CrossRef]
- Bjørklund, G.; Tinkov, A.A.; Hosnedlová, B.; Kizek, R.; Ajsuvakova, O.P.; Chirumbolo, S.; Skalnaya, M.G.; Peana, M.; Dadar, M.; El-Ansari, A.; et al. The role of glutathione redox imbalance in autism spectrum disorder: A review. Free Radic. Biol. Med. 2020, 160, 149–162. [Google Scholar] [CrossRef]
- Young, A.M.; Campbell, E.; Lynch, S.; Suckling, J.; Powis, S.J. Aberrant NF-kappaB expression in autism spectrum condition: A mechanism for neuroinflammation. Front. Psychiatry 2011, 2, 27. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, T.C.; Tsilioni, I.; Patel, A.B.; Doyle, R. Atopic diseases and inflammation of the brain in the pathogenesis of autism spectrum disorders. Transl. Psychiatry 2016, 6, e844. [Google Scholar] [CrossRef] [PubMed]
- Alomar, H.A.; Ansari, M.A.; Nadeem, A.; Attia, S.M.; Bakheet, S.A.; Al-Mazroua, H.A.; Hussein, M.H.; Algarni, M.H.; Ahmad, S.F. A potent and selective CXCR2 antagonist improves neuroimmune dysregulation through the inhibition of NF-κB and notch inflammatory signaling in the BTBR mouse model of autism. J. Neuroimmunol. 2023, 377, 578069. [Google Scholar] [CrossRef] [PubMed]
- Eftekharian, M.M.; Ghafouri-Fard, S.; Noroozi, R.; Omrani, M.D.; Arsang-Jang, S.; Ganji, M.; Gharzi, V.; Noroozi, H.; Komaki, A.; Mazdeh, M.; et al. Cytokine profile in autistic patients. Cytokine 2018, 108, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Kutuk, M.O.; Tufan, E.; Gokcen, C.; Kilicaslan, F.; Karadag, M.; Mutluer, T.; Yektas, C.; Coban, N.; Kandemir, H.; Buber, A.; et al. Cytokine expression profiles in Autism spectrum disorder: A multi-center study from Turkey. Cytokine 2020, 133, 155152. [Google Scholar] [CrossRef] [PubMed]
- Ferencova, N.; Visnovcova, Z.; Ondrejka, I.; Hrtanek, I.; Bujnakova, I.; Kovacova, V.; Macejova, A.; Tonhajzerova, I. Peripheral inflammatory markers in autism spectrum disorder and attention deficit/hyperactivity disorder at adolescent age. Int. J. Mol. Sci. 2023, 24, 11710. [Google Scholar] [CrossRef] [PubMed]
- Mehta, R.; Bhandari, R.; Kuhad, A. Effects of catechin on a rodent model of autism spectrum disorder: Implications for the role of nitric oxide in neuroinflammatory pathway. Psychopharmacology 2021, 238, 3249–3271. [Google Scholar] [CrossRef] [PubMed]
- Napolioni, V.; Persico, A.M.; Porcelli, V.; Palmieri, L. The mitochondrial aspartate/glutamate carrier AGC1 and calcium homeostasis: Physiological links and abnormalities in autism. Mol. Neurobiol. 2011, 44, 83–92. [Google Scholar] [CrossRef]
- Rossignol, D.A.; Frye, R.E. Mitochondrial dysfunction in autism spectrum disorders: A systematic review and meta-analysis. Mol. Psychiatry 2012, 17, 290–314. [Google Scholar] [CrossRef]
- Palmieri, L.; Persico, A.M. Mitochondrial dysfunction in autism spectrum disorders: Cause or effect? Biochim. Biophys. Acta—Bioenerg. 2010, 1797, 1130–1137. [Google Scholar] [CrossRef]
- Tani, Y.; Fernell, E.; Watanabe, Y.; Kanai, T.; Langstrom, B. Decrease in 6R-5,6,7,8-tetrahydrobiopterin content in cerebrospinal fluid of autistic patients. Neurosci. Lett. 1994, 181, 169–172. [Google Scholar] [CrossRef] [PubMed]
- Frye, R.E. Central tetrahydrobiopterin concentration in neurodevelopmental disorders. Front. Neurosci. 2010, 4, 1619. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Frye, R.E. Tetrahydrobiopterin deficiency in autism spectrum disorder. N. Am. J. Med. Sci. 2014, 7, 93–96. [Google Scholar]
- Naruse, H.; Takesada, M.; Nagahata, M.; Kazamatsuri, H.Y.N.; Yamazaki, K. An open clinical study of sapropterin hydocloride (R-tetrahydrobiopterin SUN 588) in infantile autism: Clinical study using a Rating Scale for Abnormal Behaviors in Children. Rinsho Iyaku 1990, 6, 1343–1368. (In Japanese) [Google Scholar]
- Takesada, M.; Naruse, H.; Nagahata, M. An open clinical study of sapropterin hydrochloride (R-tetrahydrobiopterin, R-THBP) in infantile autism: Clinical effects and long-term follow-up. In Neurobiology of Infantile Autism: Proceedings of the International Symposium on Neurobiology of Infantile Autism, Tokyo, Japan, 10–11 November 1990; Naruse, H., Ornitz, E., Eds.; Elsevier Science Publishers: New York, NY, USA, 1990; pp. 355–358. [Google Scholar]
- Tripathi, M.K.; Kartawy, M.; Amal, H. The role of nitric oxide in brain disorders: Autism spectrum disorder and other psychiatric, neurological, and neurodegenerative disorders. Redox Biol. 2020, 34, 101567. [Google Scholar] [CrossRef] [PubMed]
- Richter, M.; Murtaza, N.; Scharrenberg, R.; White, S.H.; Johanns, O.; Walker, S.; Yuen, R.K.C.; Schwanke, B.; Bedurfig, B.; Henis, M.; et al. Altered TAOK2 activity causes autism-related neurodevelopmental and cognitive abnormalities through RhoA signaling. Mol. Psychiatry 2019, 24, 1329–1350. [Google Scholar] [CrossRef] [PubMed]
- Luo, T.; Ou, J.-N.; Cao, L.-F.; Peng, X.-Q.; Li, Y.-M.; Tian, Y.-Q. The autism-related lncRNA MSNP1AS regulates moesin protein to influence the RhoA, Rac1, and PI3K/Akt pathways and regulate the structure and survival of neurons. Autism Res. 2020, 13, 2073–2082. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, T.; Yoshida, T.; Ra, M.; Taguchi, R.; Mishina, M. IL1RAPL1 associated with mental retardation and autism regulates the formation and stabilization of glutamatergic synapses of cortical neurons through RhoA signaling. PLoS ONE 2013, 8, e66254. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.E.; Tran, K.; Smith, C.C.; McDonald, M.; Shejwalkar, P.; Hara, K. The role of the Nrf2/ARE antioxidant system in preventing cardiovascular diseases. Diseases 2016, 4, 34. [Google Scholar] [CrossRef]
- Forman, H.J.; Zhang, H. Targeting oxidative stress in disease: Promise and limitations of antioxidant therapy. Nat. Rev. Drug Discov. 2021, 20, 689–709. [Google Scholar] [CrossRef]
- Pall, M.L.; Levine, S. Nrf2, a master regulator of detoxification and also antioxidant, anti-inflammatory and other cytoprotective mechanisms, is raised by health promoting factors. Sheng Li Xue Bao 2015, 67, 1–18. [Google Scholar] [PubMed]
- Yang, J.; Fu, X.; Liao, X.; Li, Y. Nrf2 activators as dietary phytochemicals against oxidative stress, inflammation, and mitochondrial dysfunction in autism spectrum disorders: A systematic review. Front. Psychiatry 2020, 11, 561998. [Google Scholar] [CrossRef]
- Yerra, V.G.; Negi, G.; Sharma, S.S.; Kumar, A. Potential therapeutic effects of the simultaneous targeting of the Nrf2 and NF-κB pathways in diabetic neuropathy. Redox Biol. 2013, 1, 394–397. [Google Scholar] [CrossRef]
- Fontes-Dutra, M.; Santos-Terra, J.; Deckmann, I.; Schwingel, G.B.; Della-Flora Nunes, G.; Hirsch, M.M.; Bauer-Negrini, G.; Riesgo, R.S.; Bambini-Junior, V.; Hedin-Pereira, C.; et al. Resveratrol prevents cellular and behavioral sensory alterations in the animal model of autism induced by valproic acid. Front. Synaptic Neurosci. 2018, 10, 9. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, M.M.; Deckmann, I.; Fontes-Dutra, M.; Bauer-Negrini, G.; Nunes, G.D.F.; Nunes, W.; Rabelo, B.; Riesgo, R.; Margis, R.; Bambini-Junior, V.; et al. Behavioral alterations in autism model induced by valproic acid and translational analysis of circulating microRNA. Food Chem. Toxicol. 2018, 115, 336–343. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.F.; Qin, L.Y.; Lian, J.Q. Resveratrol improves autistic behaviors in rat model. J. Third Mil. Med. Univ. 2017, 39, 1360–1365. [Google Scholar]
- Subasi Turgut, F.; Karadag, M.; Taysi, S.; Hangül, Z.; Gokcen, C. NRF2, KEAP1 and GSK-3 levels in autism spectrum disorder: A case control study. Int. J. Dev. Disabil. 2023, 1–11. [Google Scholar] [CrossRef]
- Porokhovnik, L.N.; Pisarev, V.M.; Chumachenko, A.G.; Chedakova, J.M.; Ershova, E.S.; Veiko, N.N.; Gorbachevskaya, N.L.; Mamokhina, U.A.; Sorokin, A.B.; Basova, A.Y.; et al. Association of NEF2L2 Rs35652124 polymorphism with Nrf2 induction and genotoxic stress biomarkers in autism. Genes 2023, 14, 718. [Google Scholar] [CrossRef]
- Wu, H.; Luan, Y.; Wang, H.; Zhang, P.; Liu, S.; Wang, P. Selenium inhibits ferroptosis and ameliorates autistic-like behaviors of BTBR mice by regulating the Nrf2/GPx4 pathway. Brain Res. 2022, 188, 38–48. [Google Scholar] [CrossRef]
- Shah, A.; Varma, M.; Bhandari, R. Exploring sulforaphane as neurotherapeutic: Targeting Nrf2-Keap & Nf-Kb pathway crosstalk in ASD. Metab. Brain Dis. 2024, 39, 373–385. [Google Scholar] [CrossRef]
- Zhao, Y.; Fu, H.; Cheng, H.; Zheng, R.; Wang, G.; Li, S.; Li, E.; Li, L. Electroacupuncture at Zusanli ameliorates the autistic-like behaviors of rats through activating the Nrf2-mediated antioxidant responses. Gene 2022, 828, 146440. [Google Scholar] [CrossRef] [PubMed]
- Bakouie, F.; Darvishi, A.; Bahrami, S.; Gharibzadeh, S. The increase of AMP-activated protein kinase during physical activities can reduce symptoms of autistic children. Neonat. Pediatr. Med. 2015, 1, S1002. [Google Scholar]
- Alsaqati, M.; Heine, V.M.; Harwood, A.J. Pharmacological intervention to restore connectivity deficits of neuronal networks derived from ASD patient iPSC with a TSC2 mutation. Mol. Autism 2020, 11, 80. [Google Scholar] [CrossRef] [PubMed]
- Lanfranchi, M.; Meyer-Dilhet, G.; Dos Reis, R.; Garcia, A.; Blondet, C.; Javin, L.; Amar, A.; Courchet, J. The AMPK-related kinase NUAK1 controls cortical axons branching though a local modulation of mitochondrial metabolic functions. BioRxiv 2020. [Google Scholar] [CrossRef]
- Diaz-Gerevini, G.T.; Repossi, G.; Dain, A.; Tarres, M.C.; Das, A.N.; Reynard, A.R. Beneficial action of resveratrol: How and why? Nutrition 2016, 32, 174–178. [Google Scholar] [CrossRef] [PubMed]
- Gantois, I.; Khoutorsky, A.; Popic, J.; Aguilar-Valles, A.; Freemantle, E.; Cao, R.; Sharma, V.; Pooters, T.; Nagpal, A.; Skalecka, A.; et al. Metformin ameliorates core deficits in a mouse model of fragile X syndrome. Nat. Med. 2017, 23, 674–677. [Google Scholar] [CrossRef] [PubMed]
- Ullian, E.M.; Christopherson, K.S.; Barres, B.A. Role for glia in synaptogenesis. Glia 2004, 47, 209–216. [Google Scholar] [CrossRef]
- Pfrieger, F.W. Role of glial cells in the formation and maintenance of synapses. Brain Res. Rev. 2010, 63, 39–46. [Google Scholar] [CrossRef]
- Pfrieger, F.W. Roles of glial cells in synapse development. Cell. Mol. Life Sci. 2009, 66, 2037–2047. [Google Scholar] [CrossRef]
- Agrawal, M.; Welshhans, K. Local translation across neural development: A focus on radial glial cells, axons, and synaptogenesis. Front. Mol. Neurosci. 2021, 14, 717170. [Google Scholar] [CrossRef]
- Slezak, M.; Pfrieger, F.W. New roles for astrocytes: Regulation of CNS synaptogenesis. Trends Neurosci. 2003, 21, P531–P535. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, K.T.; Eroglu, C. Molecular mechanisms of astrocyte-induced synaptogenesis. Curr. Opin. Neurobiol. 2017, 45, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Farhy-Tselnicker, I.; Allen, N.J. Astrocytes, neurons, synapses: A tripartite view on cortical circuit development. Neural Dev. 2018, 13, 7. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.X.; Lane, C.J.B.; Eroglu, C. Role of astrocytes in synapse formation and maturation. Curr. Top. Dev. Biol. 2021, 142, 371–402. [Google Scholar] [PubMed]
- Bosworth, A.P.; Allen, N.J. The diverse actions of astrocytes during synaptic development. Curr. Opin. Neurobiol. 2017, 47, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Verkhratsky, A.; Kirchhoff, F. NMDA receptors in glia. Neuroscientist 2007, 13, 28. [Google Scholar] [CrossRef] [PubMed]
- Verkhratsky, A.; Chvátal, A. NMDA receptors in astrocytes. Neurochem. Res. 2020, 45, 122–133. [Google Scholar] [CrossRef] [PubMed]
- Pavlík, M.; Zbojovský, J.; Kolcunová, I. Compare fabric and brick wall as shielding material dependence on electromagnetic field. In Proceedings of the 2021 IEEE 4th International Conference and Workshop Óbuda on Electrical and Power Engineering (CANDO-EPE), Budapest, Hungary, 17–18 November 2021; pp. 139–142. [Google Scholar] [CrossRef]
- Ängskog, P.; Bäckström, M.; Samuelsson, C.; Kangashaka Vallhagen, B. Shielding Effectiveness and HPM Vulnerability of Energy-Saving Windows and Window Panes. IEEE Trans. Electromagn. Compat. 2018, 61, 870–877. [Google Scholar] [CrossRef]
- Raj, A.A.; Lee, C.P.; Sidek, M.F. Protection against EMF at transmission line and tower. In Proceedings of the 2020 IEEE International Conference on Power and Energy (PECon), Penang, Malaysia, 7–8 December 2020. [Google Scholar]
- Hori, T.; Nedachi, T.; Suzuki, H.; Harakawa, S. Characterization of the suppressive effects of extremely-low-frequency electric fields on a stress-induced increase in the plasma glucocorticoid level in mice. Bioelectromagnetics 2018, 39, 516–528. [Google Scholar] [CrossRef]
- Syldona, M. Reducing the in-vitro electromagnetic field effect of cellular phones on human DNA and the intensity of their emitted radiation. Acupunct. Electrother. Res. 2007, 32, 1–14. [Google Scholar] [CrossRef]
- Verma, S.; Dhangar, M.; Paul, S.; Chaturvedi, K.; Khan, M.A.; Srivastava, A.K. Recent Advances for Fabricating Smart Electromagnetic Interference Shielding Textile: A Comprehensive Review. Electron. Mater. Lett. 2022, 18, 331–344. [Google Scholar] [CrossRef]
- Conniott, W.; Viera, J.; Caldeirinha, R.F.S. Shielding effectiveness using carbonized wood. In Proceedings of the 2019 13th European Conference on Antennas and Propagation (EuCAP), Krakow, Poland, 31 March–5 April 2019; pp. 1–4. [Google Scholar]
- Tantengco, O.A.G.; de Castro Silva, M.; Shahin, H.; Bento, G.F.C.; Cursino, G.C.; Cayenne, S. The role of nuclear factor erythroid 2–related factor 2 (NRF2) in normal and pathological pregnancy: A systematic review. Am. J. Reprod. Immunol. 2021, 86, e13496. [Google Scholar] [CrossRef]
- Zou, Y.; Hu, M.; Bao, Q.; Chan, J.Y.; Dai, G. Nrf2 participates in regulating maternal hepatic adaptations to pregnancy. J. Cell Sci. 2013, 126, 1618–1625. [Google Scholar] [CrossRef] [PubMed]
- Hussain, T.; Tan, B.; Liu, G.; Murtaza, G.; Rahu, N.; Saleem, M.; Yin, Y. Modulatory mechanism of polyphenols and Nrf2 signaling pathway in LPS challenged pregnancy disorders. Oxid. Med. Cell Longev. 2017, 2017, 8254289. [Google Scholar] [CrossRef] [PubMed]
- Aubouy, A.; Olagnier, D.; Bertin, G.; Ezinmegnon, S.; Majorel, C.; Mimar, S.; Massougbodji, A.; Deloron, P.; Pipy, B.; Coste, A. Nrf2-driven CD36 and HO-1 gene expression in circulating monocytes correlates with favourable clinical outcome in pregnancy-associated malaria. Malar. J. 2015, 14, 358. [Google Scholar] [CrossRef]
- Tossetta, G.; Fantone, S.; Piani, F.; Crescimanno, C.; Ciavattini, A.; Giannubilo, S.R.; Marzioni, D. Modulation of NRF2/KEAP1 signaling in preeclampsia. Cells 2023, 12, 1545. [Google Scholar] [CrossRef]
- Psefteli, P.M.; Morris, J.K.; Ehler, E.; Smith, L.; Bowe, J.; Mann, G.E.; Taylor, P.D.; Chapple, S.J. Sulforaphane induced NRF2 activation in obese pregnancy attenuates developmental redox imbalance and improves early-life cardiovascular function in offspring. Redox Biol. 2023, 67, 102883. [Google Scholar] [CrossRef]
- Cheng, X.; Chapple, S.J.; Patel, B.; Puszyk, W.; Sugden, D.; Yin, X.; Mayr, M.; Siow, R.C.; Mann, G.E. Gestational diabetes mellitus impairs Nrf2-mediated adaptive antioxidant defenses and redox signaling in fetal endothelial cells in utero. Diabetes 2013, 62, 4088–4097. [Google Scholar] [CrossRef] [PubMed]
- Chapple, S.J.; Puszyk, W.M.; Mann, G.E. Keap1–Nrf2 regulated redox signaling in utero: Priming of disease susceptibility in offspring. Free Rad. Biol. Med. 2015, 88, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Kweider, N.; Wruck, C.J.; Rath, W. New insights into the pathogenesis of preeclampsia–the role of nrf2 activators and their potential therapeutic impact. Geburtshilfe Frauenheilkd. 2013, 73, 1236–1240. [Google Scholar] [CrossRef]
- Mundal, S.B.; Rakner, J.J.; Silva, G.B.; Gierman, L.M.; Austdal, M.; Basnet, P.; Elschot, M.; Bakke, S.S.; Ostrop, J.; Thomsen, L.C.; et al. Divergent regulation of decidual oxidative-stress response by NRF2 and KEAP1 in preeclampsia with and without fetal growth restriction. Int. J. Mol. Sci. 2022, 23, 1966. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pall, M.L. Central Causation of Autism/ASDs via Excessive [Ca2+]i Impacting Six Mechanisms Controlling Synaptogenesis during the Perinatal Period: The Role of Electromagnetic Fields and Chemicals and the NO/ONOO(-) Cycle, as Well as Specific Mutations. Brain Sci. 2024, 14, 454. https://doi.org/10.3390/brainsci14050454
Pall ML. Central Causation of Autism/ASDs via Excessive [Ca2+]i Impacting Six Mechanisms Controlling Synaptogenesis during the Perinatal Period: The Role of Electromagnetic Fields and Chemicals and the NO/ONOO(-) Cycle, as Well as Specific Mutations. Brain Sciences. 2024; 14(5):454. https://doi.org/10.3390/brainsci14050454
Chicago/Turabian StylePall, Martin L. 2024. "Central Causation of Autism/ASDs via Excessive [Ca2+]i Impacting Six Mechanisms Controlling Synaptogenesis during the Perinatal Period: The Role of Electromagnetic Fields and Chemicals and the NO/ONOO(-) Cycle, as Well as Specific Mutations" Brain Sciences 14, no. 5: 454. https://doi.org/10.3390/brainsci14050454
APA StylePall, M. L. (2024). Central Causation of Autism/ASDs via Excessive [Ca2+]i Impacting Six Mechanisms Controlling Synaptogenesis during the Perinatal Period: The Role of Electromagnetic Fields and Chemicals and the NO/ONOO(-) Cycle, as Well as Specific Mutations. Brain Sciences, 14(5), 454. https://doi.org/10.3390/brainsci14050454





