Dose–Response of Creatine Supplementation on Cognitive Function in Healthy Young Adults
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Design
2.2. Participants
2.3. Pre-Post-Testing Measures
2.4. Supplementation and Randomization
2.5. Fnirs
2.6. Diet
2.7. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Martínez, L.D.; Bezard, M.; Brunotto, M.; Dodelson de Kremer, R. Creatine metabolism: Detection of creatine and guanidinoacetate in saliva of healthy subjects. Acta Odontológica Latinoam. 2016, 29, 49–53. [Google Scholar]
- Roschel, H.; Gualano, B.; Ostojic, S.M.; Rawson, E.S. Creatine supplementation and brain health. Nutrients 2021, 13, 586. [Google Scholar] [CrossRef] [PubMed]
- Guimarães-Ferreira, L. Role of the phosphocreatine system on energetic homeostasis in skeletal and cardiac muscles. Einstein 2014, 12, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Kreider, R.B.; Kalman, D.S.; Antonio, J.; Ziegenfuss, T.N.; Wildman, R.; Collins, R.; Candow, D.G.; Kleiner, S.M.; Almada, A.L.; Lopez, H.L. International Society of Sports Nutrition position stand: Safety and efficacy of creatine supplementation in exercise, sport, and medicine. J. Int. Soc. Sports Nutr. 2017, 14, 18. [Google Scholar] [CrossRef] [PubMed]
- Candow, D.G.; Forbes, S.C.; Ostojic, S.M.; Prokopidis, K.; Stock, M.S.; Harmon, K.K.; Faulkner, P. “Heads Up” for Creatine Supplementation and its Potential Applications for Brain Health and Function. Sports Med. 2023. [Google Scholar] [CrossRef] [PubMed]
- Gualano, B.; Artioli, G.G.; Poortmans, J.R.; Lancha Junior, A.H. Exploring the therapeutic role of creatine supplementation. Amino Acids 2010, 38, 31–44. [Google Scholar] [CrossRef] [PubMed]
- Rango, M.; Castelli, A.; Scarlato, G. Energetics of 3.5 s neural activation in humans: A 31P MR spectroscopy study. Magn. Reson. Med. 1997, 38, 878–883. [Google Scholar] [CrossRef]
- Sestili, P.; Martinelli, C.; Colombo, E.; Barbieri, E.; Potenza, L.; Sartini, S.; Fimognari, C. Creatine as an antioxidant. Amino Acids 2011, 40, 1385–1396. [Google Scholar] [CrossRef]
- Turner, C.E.; Byblow, W.D.; Gant, N. Creatine supplementation enhances corticomotor excitability and cognitive performance during oxygen deprivation. J. Neurosci. 2015, 35, 1773–1780. [Google Scholar] [CrossRef]
- McMorris, T.; Harris, R.C.; Swain, J.; Corbett, J.; Collard, K.; Dyson, R.J.; Dye, L.; Hodgson, C.; Draper, N. Effect of creatine supplementation and sleep deprivation, with mild exercise, on cognitive and psychomotor performance, mood state, and plasma concentrations of catecholamines and cortisol. Psychopharmacology 2006, 185, 93–103. [Google Scholar] [CrossRef]
- Machado, M.; Pereira, R. The Potential and Challenges of Creatine Supplementation for Cognition/Memory in Older Adults. Eur. J. Geriatr. Gerontol. 2023, 5, 1–5. [Google Scholar] [CrossRef]
- Dolan, E.; Gualano, B.; Rawson, E.S. Beyond muscle: The effects of creatine supplementation on brain creatine, cognitive processing, and traumatic brain injury. Eur. J. Sport Sci. 2019, 19, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Dechent, P.; Pouwels, P.; Wilken, B.; Hanefeld, F.; Frahm, J. Increase of total creatine in human brain after oral supplementation of creatine-monohydrate. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 1999, 277, R698–R704. [Google Scholar] [CrossRef] [PubMed]
- Forbes, S.C.; Cordingley, D.M.; Cornish, S.M.; Gualano, B.; Roschel, H.; Ostojic, S.M.; Rawson, E.S.; Roy, B.D.; Prokopidis, K.; Giannos, P. Effects of creatine supplementation on brain function and health. Nutrients 2022, 14, 921. [Google Scholar] [CrossRef] [PubMed]
- Borchio, L.; Machek, S.B.; Machado, M. Supplemental creatine monohydrate loading improves cognitive function in experienced mountain bikers. J. Sports Med. Phys. Fit. 2020, 60, 1168–1170. [Google Scholar] [CrossRef] [PubMed]
- Hammett, S.T.; Wall, M.B.; Edwards, T.C.; Smith, A.T. Dietary supplementation of creatine monohydrate reduces the human fMRI BOLD signal. Neurosci. Lett. 2010, 479, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Ling, J.; Kritikos, M.; Tiplady, B. Cognitive effects of creatine ethyl ester supplementation. Behav. Pharmacol. 2009, 20, 673–679. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, A.; Kato, N.; Kato, T. Effects of creatine on mental fatigue and cerebral hemoglobin oxygenation. Neurosci. Res. 2002, 42, 279–285. [Google Scholar] [CrossRef]
- Rawson, E.S.; Lieberman, H.R.; Walsh, T.M.; Zuber, S.M.; Harhart, J.M.; Matthews, T.C. Creatine supplementation does not improve cognitive function in young adults. Physiol. Behav. 2008, 95, 130–134. [Google Scholar] [CrossRef]
- Sugihara, Y.; Matsuura, T.; Kubo, Y.; Ohgomori, T. Activation of the prefrontal cortex and improvement of cognitive performance with standing on one leg. Neuroscience 2021, 477, 50–62. [Google Scholar] [CrossRef]
- Causse, M.; Chua, Z.; Peysakhovich, V.; Del Campo, N.; Matton, N. Mental workload and neural efficiency quantified in the prefrontal cortex using fNIRS. Sci. Rep. 2017, 7, 5222. [Google Scholar] [CrossRef] [PubMed]
- Salzman, T.; Tobón Vallejo, D.; Polskaia, N.; Michaud, L.; St-Amant, G.; Lajoie, Y.; Fraser, S. Hemodynamic and behavioral changes in older adults during cognitively demanding dual tasks. Brain Behav. 2021, 11, e02021. [Google Scholar] [CrossRef] [PubMed]
- Siedler, M.R.; Rodriguez, C.; Stratton, M.T.; Harty, P.S.; Keith, D.S.; Green, J.J.; Boykin, J.R.; White, S.J.; Williams, A.D.; DeHaven, B. Assessing the reliability and cross-sectional and longitudinal validity of fifteen bioelectrical impedance analysis devices. Br. J. Nutr. 2023, 130, 827–840. [Google Scholar] [CrossRef]
- Weintraub, S.; Dikmen, S.S.; Heaton, R.K.; Tulsky, D.S.; Zelazo, P.D.; Bauer, P.J.; Carlozzi, N.E.; Slotkin, J.; Blitz, D.; Wallner-Allen, K. Cognition assessment using the NIH Toolbox. Neurology 2013, 80, S54–S64. [Google Scholar] [CrossRef] [PubMed]
- Heaton, R.K.; Akshoomoff, N.; Tulsky, D.; Mungas, D.; Weintraub, S.; Dikmen, S.; Beaumont, J.; Casaletto, K.B.; Conway, K.; Slotkin, J. Reliability and validity of composite scores from the NIH Toolbox Cognition Battery in adults. J. Int. Neuropsychol. Soc. 2014, 20, 588–598. [Google Scholar] [CrossRef] [PubMed]
- Yoon, B.-K.; Kravitz, L.; Robergs, R. VO2max, protocol duration, and the VO2 plateau. Med. Sci. Sports Exerc. 2007, 39, 1186–1192. [Google Scholar] [CrossRef] [PubMed]
- Buchfuhrer, M.J.; Hansen, J.E.; Robinson, T.E.; Sue, D.Y.; Wasserman, K.; Whipp, B. Optimizing the exercise protocol for cardiopulmonary assessment. J. Appl. Physiol. 1983, 55, 1558–1564. [Google Scholar] [CrossRef] [PubMed]
- Vergotte, G.; Perrey, S.; Muthuraman, M.; Janaqi, S.; Torre, K. Concurrent changes of brain functional connectivity and motor variability when adapting to task constraints. Front. Physiol. 2018, 9, 909. [Google Scholar] [CrossRef]
- Obrig, H.; Villringer, A. Beyond the visible—Imaging the human brain with light. J. Cereb. Blood Flow Metab. 2003, 23, 1–18. [Google Scholar] [CrossRef]
- Duncan, A.; Meek, J.H.; Clemence, M.; Elwell, C.E.; Fallon, P.; Tyszczuk, L.; Cope, M.; Delpy, D.T. Measurement of cranial optical path length as a function of age using phase resolved near infrared spectroscopy. Pediatr. Res. 1996, 39, 889–894. [Google Scholar] [CrossRef]
- Teixeira, V.; Voci, S.M.; Mendes-Netto, R.S.; da Silva, D.G. The relative validity of a food record using the smartphone application MyFitnessPal. Nutr. Diet. 2018, 75, 219–225. [Google Scholar] [CrossRef] [PubMed]
- McMorris, T.; Mielcarz, G.; Harris, R.C.; Swain, J.P.; Howard, A. Creatine supplementation and cognitive performance in elderly individuals. Aging Neuropsychol. Cogn. 2007, 14, 517–528. [Google Scholar] [CrossRef] [PubMed]
- Lyoo, I.K.; Kong, S.W.; Sung, S.M.; Hirashima, F.; Parow, A.; Hennen, J.; Cohen, B.M.; Renshaw, P.F. Multinuclear magnetic resonance spectroscopy of high-energy phosphate metabolites in human brain following oral supplementation of creatine-monohydrate. Psychiatry Res. Neuroimaging 2003, 123, 87–100. [Google Scholar] [CrossRef] [PubMed]
- Merege-Filho, C.A.A.; Otaduy, M.C.G.; de Sá-Pinto, A.L.; de Oliveira, M.O.; de Souza Gonçalves, L.; Hayashi, A.P.T.; Roschel, H.; Pereira, R.M.R.; Silva, C.A.; Brucki, S.M.D. Does brain creatine content rely on exogenous creatine in healthy youth? A proof-of-principle study. Appl. Physiol. Nutr. Metab. 2017, 42, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Alves, C.R.R.; Merege Filho, C.A.A.; Benatti, F.B.; Brucki, S.; Pereira, R.M.R.; de Sá Pinto, A.L.; Lima, F.R.; Roschel, H.; Gualano, B. Creatine supplementation associated or not with strength training upon emotional and cognitive measures in older women: A randomized double-blind study. PLoS ONE 2013, 8, e76301. [Google Scholar] [CrossRef] [PubMed]
- Prokopidis, K.; Giannos, P.; Triantafyllidis, K.K.; Kechagias, K.S.; Forbes, S.C.; Candow, D.G. Author’s reply: Letter to the Editor: Double counting due to inadequate statistics leads to false-positive findings in “Effects of creatine supplementation on memory in healthy individuals: A systematic review and meta-analysis of randomized controlled trials”. Nutr. Rev. 2023. [Google Scholar] [CrossRef]
- Rae, C.; Digney, A.L.; McEwan, S.R.; Bates, T.C. Oral creatine monohydrate supplementation improves brain performance: A double–blind, placebo–controlled, cross–over trial. Proc. R. Soc. Lond. B 2003, 270, 2147–2150. [Google Scholar] [CrossRef] [PubMed]
- Hultman, E.; Soderlund, K.; Timmons, J.; Cederblad, G.; Greenhaff, P. Muscle creatine loading in men. J. Appl. Physiol. 1996, 81, 232–237. [Google Scholar] [CrossRef]
- Benton, D.; Donohoe, R. The influence of creatine supplementation on the cognitive functioning of vegetarians and omnivores. Br. J. Nutr. 2011, 105, 1100–1105. [Google Scholar] [CrossRef]
- Yang, D.; Hong, K.-S.; Yoo, S.-H.; Kim, C.-S. Evaluation of neural degeneration biomarkers in the prefrontal cortex for early identification of patients with mild cognitive impairment: An fNIRS study. Front. Hum. Neurosci. 2019, 13, 317. [Google Scholar] [CrossRef]
- Herold, F.; Behrendt, T.; Törpel, A.; Hamacher, D.; Müller, N.G.; Schega, L. Cortical hemodynamics as a function of handgrip strength and cognitive performance: A cross-sectional fNIRS study in younger adults. BMC Neurosci. 2021, 22, 10. [Google Scholar] [CrossRef] [PubMed]
- Baik, J.S.; Ko, M.-H.; Ko, S.-H.; Min, J.H.; Choi, J.K.; Baek, J.Y.; Kim, T.H.; Jung, N.-Y.; Jo, Y.; Lee, T.Y. Assessment of functional near-infrared spectroscopy by comparing prefrontal cortex activity: A cognitive impairment screening tool. Alzheimer Dis. Assoc. Disord. 2022, 36, 266–268. [Google Scholar] [CrossRef] [PubMed]
- Mandrick, K.; Peysakhovich, V.; Rémy, F.; Lepron, E.; Causse, M. Neural and psychophysiological correlates of human performance under stress and high mental workload. Biol. Psychol. 2016, 121, 62–73. [Google Scholar] [CrossRef] [PubMed]
| Group (M/F) | CR10 (3/7) | CR20 (4/6) | PLA (4/6) | p-Value |
|---|---|---|---|---|
| Age (yrs) | 21.8 (4.1) | 20.4 (0.7) | 20.8 (1.4) | 0.42 |
| Mass (kg) | 70.0 (12.0) | 76.2 (10.5) | 75.5 (13.0) | 0.46 |
| Height (m) | 1.72 (0.06) | 1.71 (0.10) | 1.72 (0.06) | 0.90 |
| Body Mass Index (kg/m2) | 23.4 (3.0) | 26.1 (2.9) | 25.5 (3.1) | 0.14 |
| BF (%) | 24.2 (6.8) | 26.1 (11.3) | 21.4 (4.9) | 0.45 |
| VO2max (mL·kg−1·min−1) | 44.4 (11.1) | 46.5 (8.7) | 47.8 (5.6) | 0.19 |
| Godin questionnaire (physical activity) | 46 (30) | 57 (24) | 44 (22) | 0.51 |
| Total calories (kcal/day) | 2088 (516) | 2143 (652) | 2084 (620) | 0.97 |
| Carbohydrate (g/day) | 252 (62) | 236 (82) | 231 (69) | 0.67 |
| Fat (g/day) | 76 (24) | 81 (34) | 77 (24) | 0.92 |
| Protein (g/day) | 98 (34) | 148 (122) | 111 (54) | 0.37 |
| Relative protein (g/kg) | 1.4 (0.4) | 1.9 (1.4) | 1.4 (0.5) | 0.42 |
| Cognitive test 1 | 62 (15) | 58 (12) | 59 (12) | 0.84 |
| Cognitive test 2 | 51 (12) | 55 (14) | 53 (9) | 0.83 |
| Cognitive test 3 | 49 (15) | 48 (6) | 48 (6) | 0.92 |
| CR10 | CR20 | PLA | Interaction p-Value | |
|---|---|---|---|---|
| Cognitive test 1 | 3.6 (−3.4, 10.6) | 19 (−0.19, 38.2) | 18.7 (3.9, 33.5) | 0.17 |
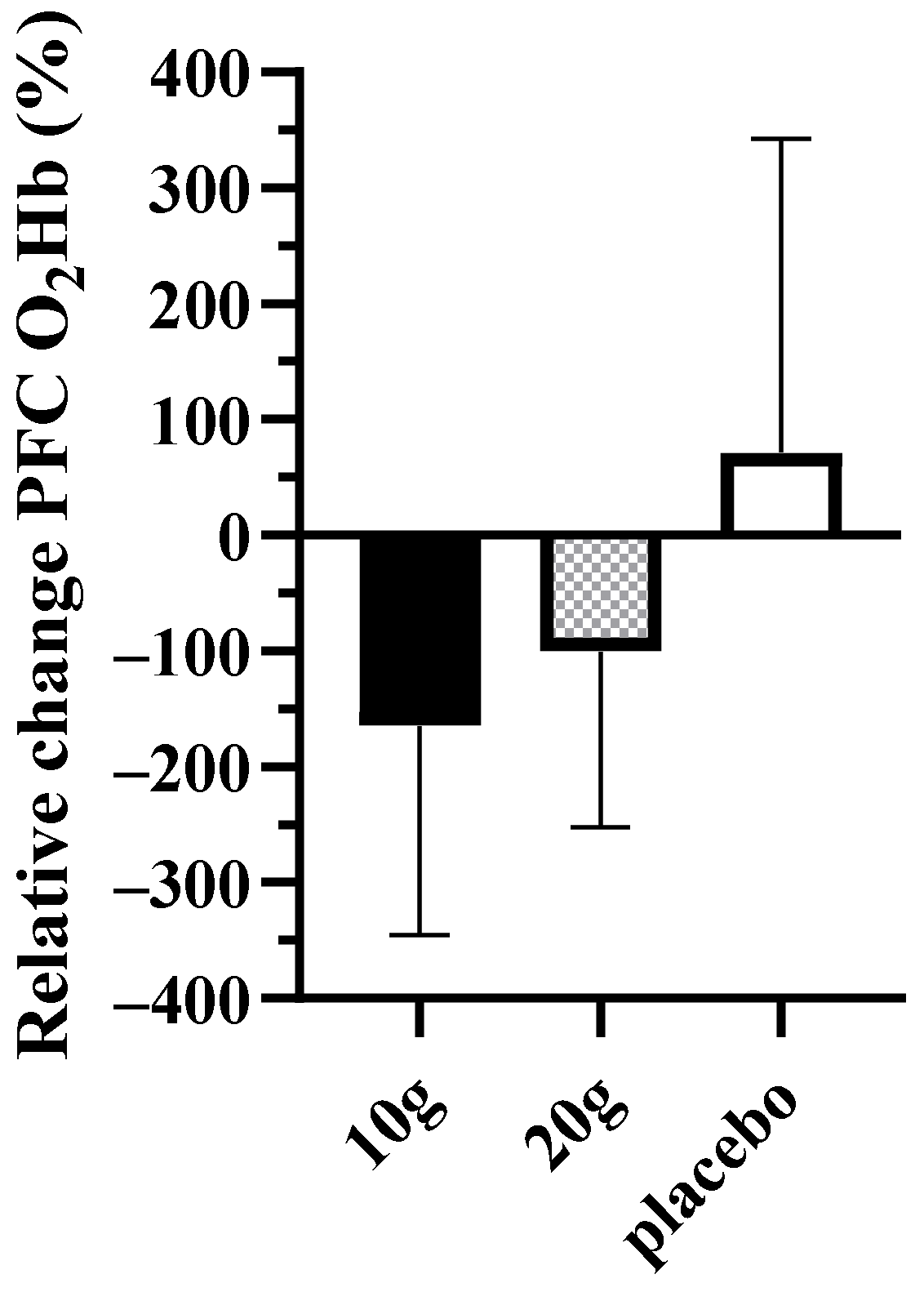
| PFC O2Hb | −164.6 (−303.5, −25.7) | −100.3 (−217.1, 16.4) | 71.7 (−154.5, 297.8) | 0.07 |
| Cognitive test 2 | 11.7 (−4.6, 27.9) | 8.8 (−7.6, 25.3) | 10 (−0.5, 20.5) | 0.95 |
| PFC O2Hb | −15 (−157.1, 127.2) | −25.9 (−92.8, 40.9) | 81.6 (−162.1, 325.3) | 0.50 |
| Cognitive test 3 | 8.2 (−4.2, 20.7) | 11.5 (3.8, 19.2) | 4.9 (−4.8, 14.7) | 0.59 |
| PFC O2Hb | 7.5 (−98, 113) | −41.1 (−84, 1.9) | −13.8 (−175.3, 147.8) | 0.76 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moriarty, T.; Bourbeau, K.; Dorman, K.; Runyon, L.; Glaser, N.; Brandt, J.; Hoodjer, M.; Forbes, S.C.; Candow, D.G. Dose–Response of Creatine Supplementation on Cognitive Function in Healthy Young Adults. Brain Sci. 2023, 13, 1276. https://doi.org/10.3390/brainsci13091276
Moriarty T, Bourbeau K, Dorman K, Runyon L, Glaser N, Brandt J, Hoodjer M, Forbes SC, Candow DG. Dose–Response of Creatine Supplementation on Cognitive Function in Healthy Young Adults. Brain Sciences. 2023; 13(9):1276. https://doi.org/10.3390/brainsci13091276
Chicago/Turabian StyleMoriarty, Terence, Kelsey Bourbeau, Katie Dorman, Lance Runyon, Noah Glaser, Jenna Brandt, Mallory Hoodjer, Scott C. Forbes, and Darren G. Candow. 2023. "Dose–Response of Creatine Supplementation on Cognitive Function in Healthy Young Adults" Brain Sciences 13, no. 9: 1276. https://doi.org/10.3390/brainsci13091276
APA StyleMoriarty, T., Bourbeau, K., Dorman, K., Runyon, L., Glaser, N., Brandt, J., Hoodjer, M., Forbes, S. C., & Candow, D. G. (2023). Dose–Response of Creatine Supplementation on Cognitive Function in Healthy Young Adults. Brain Sciences, 13(9), 1276. https://doi.org/10.3390/brainsci13091276









