Is Hypnotic Induction Necessary to Experience Hypnosis and Responsible for Changes in Brain Activity?
Abstract
1. Introduction
2. Participants and Study Design
2.1. Participants
2.2. Experimental Procedure
2.3. Signal Acquisition and Analysis
2.4. Variables
2.5. Statistical Analysis
3. Results
3.1. EEG Spectral Analysis
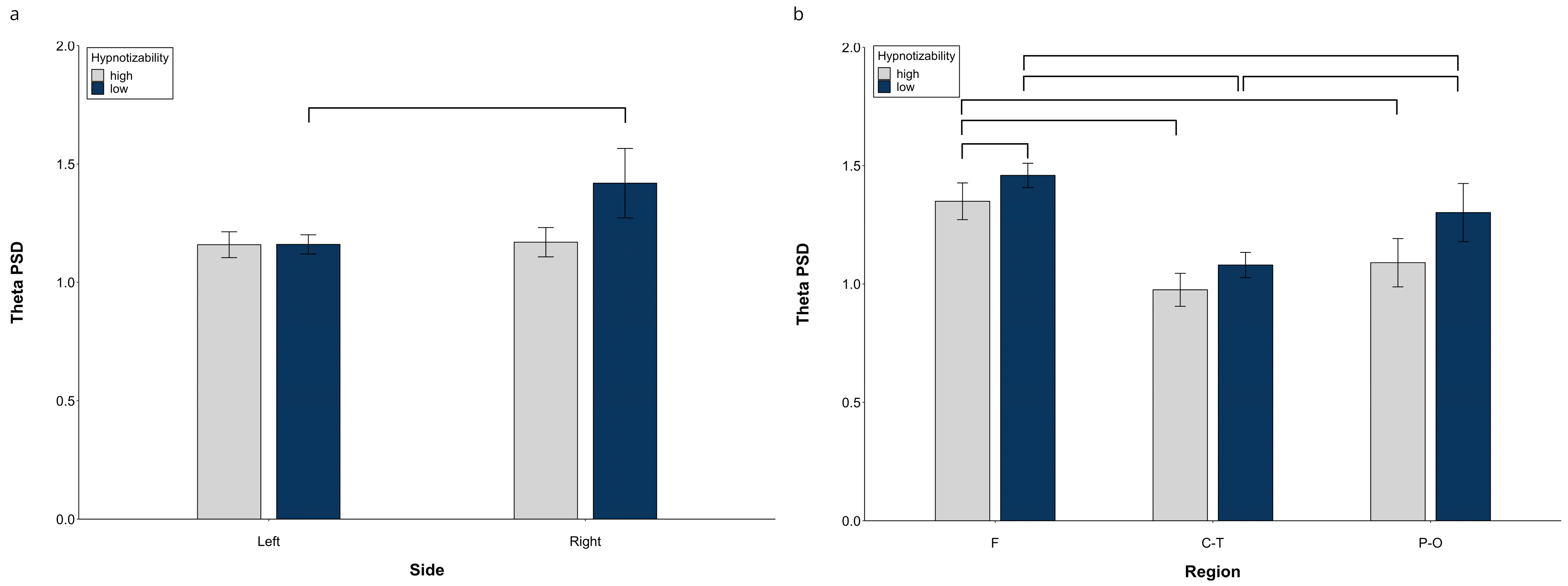
3.1.1. Theta PSD
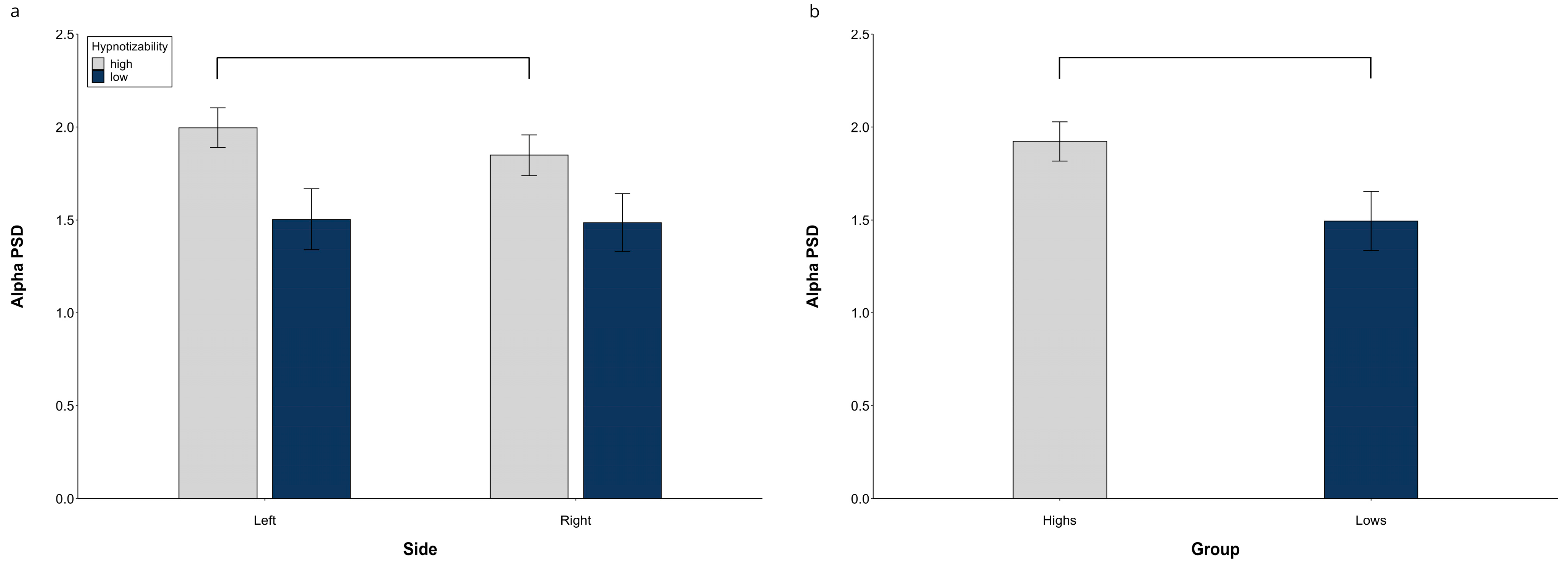
3.1.2. Alpha PSD
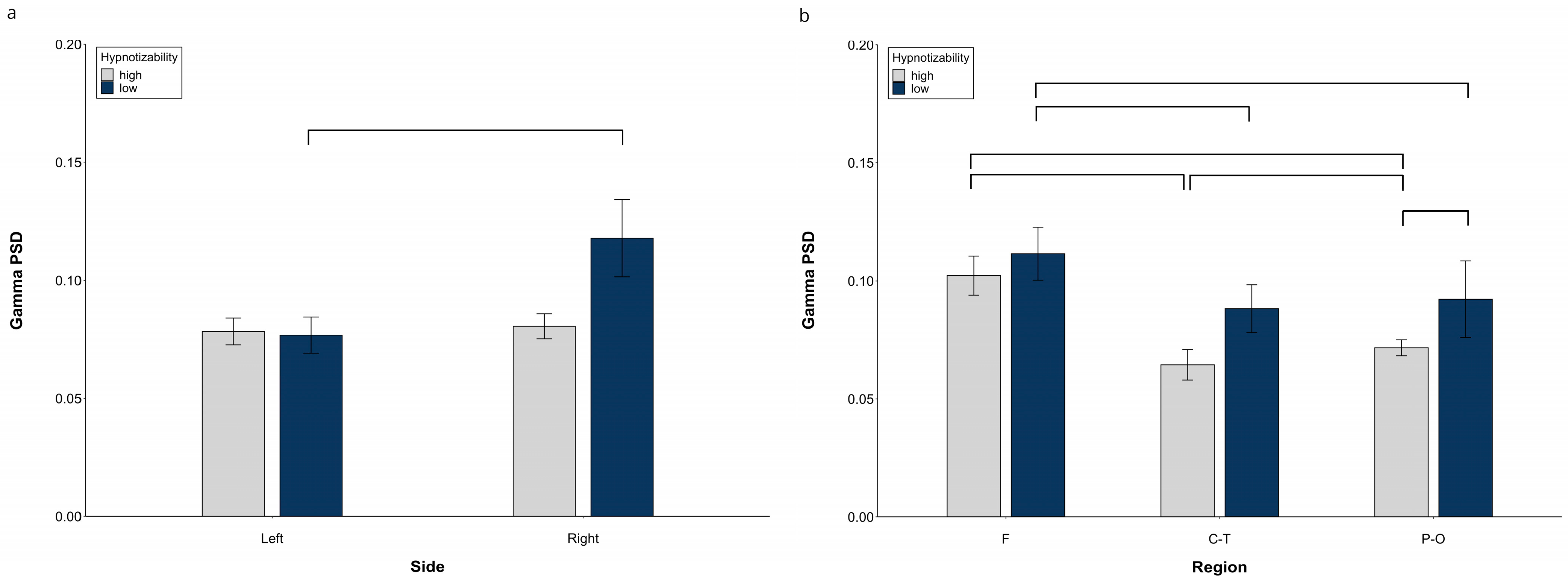
3.1.3. Gamma PSD
3.2. EEG Recurrence Quantification Analysis
4. Discussion
5. Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Elkins, G.R.; Barabasz, A.F.; Council, J.R.; Spiegel, D. Advancing Research and Practice: The Revised APA Division 30 Definition of Hypnosis. Am. J. Clin. Hypn. 2015, 57, 378–385. [Google Scholar] [CrossRef] [PubMed]
- Pekala, R.J.; Kumar, V.K.; Maurer, R.; Elliott-Carter, N.; Moon, E.; Mullen, K. Suggestibility, Expectancy, Trance State Effects, and Hypnotic Depth: I. Implications for Understanding Hypnotism. Am. J. Clin. Hypn. 2010, 52, 275–290. [Google Scholar] [CrossRef] [PubMed]
- Pekala, R.J.; Maurer, R.; Kumar, V.K.; Elliott-Carter, N.; Mullen, K. Trance State Effects and Imagery Vividness Before and During a Hypnotic Assessment: A Preliminary Study. Int. J. Clin. Exp. Hypn. 2010, 58, 383–416. [Google Scholar] [CrossRef] [PubMed]
- Iber, C.; Ancoli-Israel, S.; Chesson, A.; Quan, S. The AASM Manual for Scoring of Sleep and Associated Events: Rules, Terminology, and Technical Specifications; American Academy of Sleep Medicine: Westchester, IL, USA, 2007. [Google Scholar]
- Cardeña, E.; Jönsson, P.; Terhune, D.B.; Marcusson-Clavertz, D. The Neurophenomenology of Neutral Hypnosis. Cortex 2013, 49, 375–385. [Google Scholar] [CrossRef]
- Halligan, P.W.; Oakley, D.A. Hypnosis and Cognitive Neuroscience: Bridging the Gap. Cortex 2013, 49, 359–364. [Google Scholar] [CrossRef]
- Jiang, H.; White, M.P.; Greicius, M.D.; Waelde, L.C.; Spiegel, D. Brain Activity and Functional Connectivity Associated with Hypnosis. Cereb. Cortex 2017, 27, 4083–4093. [Google Scholar] [CrossRef]
- Landry, M.; Lifshitz, M.; Raz, A. Brain Correlates of Hypnosis: A Systematic Review and Meta-Analytic Exploration. Neurosci. Biobehav. Rev. 2017, 81, 75–98. [Google Scholar] [CrossRef]
- De Pascalis, V.; Santarcangelo, E.L. Hypnotizability-Related Asymmetries: A Review. Symmetry 2020, 12, 1015. [Google Scholar] [CrossRef]
- Dunham, C.M.; Burger, A.J.; Hileman, B.M.; Chance, E.A.; Hutchinson, A.E. Bispectral Index Alterations and Associations With Autonomic Changes During Hypnosis in Trauma Center Researchers: Formative Evaluation Study. JMIR Form. Res. 2021, 5, e24044. [Google Scholar] [CrossRef]
- Jensen, M.P.; Adachi, T.; Hakimian, S. Brain Oscillations, Hypnosis, and Hypnotizability. Am. J. Clin. Hypn. 2015, 57, 230–253. [Google Scholar] [CrossRef]
- Rho, G.; Callara, A.L.; Petri, G.; Nardelli, M.; Scilingo, E.P.; Greco, A.; Pascalis, V.D. Linear and Nonlinear Quantitative EEG Analysis during Neutral Hypnosis Following an Opened/Closed Eye Paradigm. Symmetry 2021, 13, 1423. [Google Scholar] [CrossRef]
- Keshmiri, S.; Alimardani, M.; Shiomi, M.; Sumioka, H.; Ishiguro, H.; Hiraki, K. Higher Hypnotic Suggestibility Is Associated with the Lower EEG Signal Variability in Theta, Alpha, and Beta Frequency Bands. PLoS ONE 2020, 15, e0230853. [Google Scholar] [CrossRef]
- Wutz, A.; Loonis, R.; Roy, J.E.; Donoghue, J.A.; Miller, E.K. Different Levels of Category Abstraction by Different Dynamics in Different Prefrontal Areas. Neuron 2018, 97, 716–726.e8. [Google Scholar] [CrossRef]
- Florian, G.; Andrew, C.; Pfurtscheller, G. Do Changes in Coherence Always Reflect Changes in Functional Coupling? Electroencephalogr. Clin. Neurophysiol. 1998, 106, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Tuominen, J.; Kallio, S.; Kaasinen, V.; Railo, H. Segregated Brain State during Hypnosis. Neurosci. Conscious. 2021, 2021, niab002. [Google Scholar] [CrossRef] [PubMed]
- Baghdadi, G.; Nasrabadi, A.M. EEG Phase Synchronization during Hypnosis Induction. J. Med. Eng. Technol. 2012, 36, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Yargholi, E.; Nasrabadi, A.M. The Impacts of Hypnotic Susceptibility on Chaotic Dynamics of EEG Signals during Standard Tasks of Waterloo-Stanford Group Scale. J. Med. Eng. Technol. 2013, 37, 273–281. [Google Scholar] [CrossRef]
- Yargholi, E.; Nasrabadi, A.M. Chaos–Chaos Transition of Left Hemisphere EEGs during Standard Tasks of Waterloo-Stanford Group Scale of Hypnotic Susceptibility. J. Med. Eng. Technol. 2015, 39, 281–285. [Google Scholar] [CrossRef]
- Benson, H.; Arns, P.A.; Hoffman, J.W. The Relaxation Response and Hypnosis. Int. J. Clin. Exp. Hypn. 1981, 29, 259–270. [Google Scholar] [CrossRef]
- Weitzenhoffer, A.M.; Hilgard, E.R. Scala Stanford di Suscettibilità Ipnotica, forme A e B [Stanford Hypnotic Susceptibility Scale, Form A]; Organizzazioni Speciali: Firenze, Italy, 1962. Original work published 1959. [Google Scholar]
- Cardeña, E.; Terhune, D.B. The Roles of Response Expectancies, Baseline Experiences, and Hypnotizability in Spontaneous Hypnotic Experiences. Int. J. Clin. Exp. Hypn. 2019, 67, 1–27. [Google Scholar] [CrossRef]
- Lynn, S.J.; Laurence, J.-R.; Kirsch, I. Hypnosis, Suggestion, and Suggestibility: An Integrative Model. Am. J. Clin. Hypn. 2015, 57, 314–329. [Google Scholar] [CrossRef] [PubMed]
- Lynn, S.J.; Green, J.P.; Polizzi, C.P.; Ellenberg, S.; Gautam, A.; Aksen, D. Hypnosis, Hypnotic Phenomena, and Hypnotic Responsiveness: Clinical and Research Foundations—A 40-Year Perspective. Int. J. Clin. Exp. Hypn. 2019, 67, 475–511. [Google Scholar] [CrossRef]
- Myga, K.A.; Kuehn, E.; Azanon, E. Autosuggestion: A Cognitive Process That Empowers Your Brain? Exp. Brain Res. 2022, 240, 381–394. [Google Scholar] [CrossRef]
- Reategui, R. The Relationship between Expectation and Hypnotic Susceptibility: A Literature Review. Am. J. Clin. Hypn. 2020, 63, 14–27. [Google Scholar] [CrossRef] [PubMed]
- Wagstaff, G.F. Hypnosis and the Relationship between Trance, Suggestion, Expectancy and Depth: Some Semantic and Conceptual Issues. Am. J. Clin. Hypn. 2010, 53, 47–59. [Google Scholar] [CrossRef]
- Derbyshire, S.W.G.; Whalley, M.G.; Oakley, D.A. Fibromyalgia Pain and Its Modulation by Hypnotic and Non-Hypnotic Suggestion: An FMRI Analysis. Eur. J. Pain 2009, 13, 542–550. [Google Scholar] [CrossRef]
- Maxwell, R.; Lynn, S.J.; Condon, L. Hypnosis, Hypnotic Suggestibility, Memory, and Involvement in Films. Conscious. Cogn. 2015, 33, 170–184. [Google Scholar] [CrossRef] [PubMed]
- Parris, B.A.; Dienes, Z. Hypnotic Suggestibility Predicts the Magnitude of the Imaginative Word Blindness Suggestion Effect in a Non-Hypnotic Context. Conscious. Cogn. 2013, 22, 868–874. [Google Scholar] [CrossRef]
- Faul, F.; Erdfelder, E.; Buchner, A.; Lang, A.-G. Statistical Power Analyses Using G*Power 3.1: Tests for Correlation and Regression Analyses. Behav. Res. Methods 2009, 41, 1149–1160. [Google Scholar] [CrossRef]
- Callara, A.L.; Fontanelli, L.; Belcari, I.; Rho, G.; Greco, A.; Zelič, Ž.; Sebastiani, L.; Santarcangelo, E.L. Modulation of the heartbeat evoked cortical potential by hypnotizability and hypnosis. Psychophysiology 2023, e014309. [Google Scholar] [CrossRef]
- Colloca, L.; Miller, F.G. Role of Expectations in Health. Curr. Opin. Psychiatry 2011, 24, 149–155. [Google Scholar] [CrossRef]
- Frisaldi, E.; Piedimonte, A.; Benedetti, F. Placebo and Nocebo Effects: A Complex Interplay Between Psychological Factors and Neurochemical Networks. Am. J. Clin. Hypn. 2015, 57, 267–284. [Google Scholar] [CrossRef] [PubMed]
- Delorme, A.; Makeig, S. EEGLAB: An Open Source Toolbox for Analysis of Single-Trial EEG Dynamics Including Independent Component Analysis. J. Neurosci. Methods 2004, 134, 9–21. [Google Scholar] [CrossRef] [PubMed]
- The MathWorks Inc. MATLAB, version 2020b; The MathWorks Inc.: Natick, MA, USA, 2020. [Google Scholar]
- Makeig, S.; Bell, A.J.; Jung, T.-P.; Sejnowski, T.J. Independent Component Analysis of Electroencephalographic Data. In Advances in Neural Information Processing Systems 8; Touretzky, D., Mozer, M.C., Hasselmo, M., Eds.; MIT Press: Cambridge, MA, USA, 1995; pp. 145–151. [Google Scholar]
- Onton, J.; Westerfield, M.; Townsend, J.; Makeig, S. Imaging Human EEG Dynamics Using Independent Component Analysis. Neurosci. Biobehav. Rev. 2006, 30, 808–822. [Google Scholar] [CrossRef] [PubMed]
- Wallot, S.; Roepstorff, A.; Mønster, D. Multidimensional Recurrence Quantification Analysis (MdRQA) for the Analysis of Multidimensional Time-Series: A Software Implementation in MATLAB and Its Application to Group-Level Data in Joint Action. Front. Psychol. 2016, 7, 1835. [Google Scholar] [CrossRef] [PubMed]
- Webber, C.L.; Zbilut, J.P. Dynamical Assessment of Physiological Systems and States Using Recurrence Plot Strategies. J. Appl. Physiol. 1994, 76, 965–973. [Google Scholar] [CrossRef] [PubMed]
- Marwan, N.; Wessel, N.; Meyerfeldt, U.; Schirdewan, A.; Kurths, J. Recurrence-Plot-Based Measures of Complexity and Their Application to Heart-Rate-Variability Data. Phys. Rev. E 2002, 66, 026702. [Google Scholar] [CrossRef] [PubMed]
- Wallot, S.; Mønster, D. Calculation of Average Mutual Information (AMI) and False-Nearest Neighbors (FNN) for the Estimation of Embedding Parameters of Multidimensional Time Series in Matlab. Front. Psychol. 2018, 9, 1679. [Google Scholar] [CrossRef]
- Buzug, T.; Pfister, G. Optimal Delay Time and Embedding Dimension for Delay-Time Coordinates by Analysis of the Global Static and Local Dynamical Behavior of Strange Attractors. Phys. Rev. A 1992, 45, 7073–7084. [Google Scholar] [CrossRef]
- Madeo, D.; Castellani, E.; Santarcangelo, E.L.; Mocenni, C. Hypnotic Assessment Based on the Recurrence Quantification Analysis of EEG Recorded in the Ordinary State of Consciousness. Brain Cogn. 2013, 83, 227–233. [Google Scholar] [CrossRef]
- Chiarucci, R.; Madeo, D.; Loffredo, M.I.; Castellani, E.; Santarcangelo, E.L.; Mocenni, C. Cross-Evidence for Hypnotic Susceptibility through Nonlinear Measures on EEGs of Non-Hypnotized Subjects. Sci. Rep. 2014, 4, 5610. [Google Scholar] [CrossRef]
- Hiltunen, S.; Karevaara, M.; Virta, M.; Makkonen, T.; Kallio, S.; Paavilainen, P. No Evidence for Theta Power as a Marker of Hypnotic State in Highly Hypnotizable Subjects. Heliyon 2021, 7, e06871. [Google Scholar] [CrossRef] [PubMed]
- Snipes, S.; Krugliakova, E.; Meier, E.; Huber, R. The Theta Paradox: 4–8 Hz EEG Oscillations Reflect Both Sleep Pressure and Cognitive Control. J. Neurosci. 2022, 42, 8569–8586. [Google Scholar] [CrossRef]
- Ishii, R.; Canuet, L.; Ishihara, T.; Aoki, Y.; Ikeda, S.; Hata, M.; Katsimichas, T.; Gunji, A.; Takahashi, H.; Nakahachi, T.; et al. Frontal Midline Theta Rhythm and Gamma Power Changes during Focused Attention on Mental Calculation: An MEG Beamformer Analysis. Front. Hum. Neurosci. 2014, 8, 406. [Google Scholar] [CrossRef]
- Michels, L.; Bucher, K.; Lüchinger, R.; Klaver, P.; Martin, E.; Jeanmonod, D.; Brandeis, D. Simultaneous EEG-FMRI during a Working Memory Task: Modulations in Low and High Frequency Bands. PLoS ONE 2010, 5, e10298. [Google Scholar] [CrossRef] [PubMed]
- Onton, J.; Delorme, A.; Makeig, S. Frontal Midline EEG Dynamics during Working Memory. NeuroImage 2005, 27, 341–356. [Google Scholar] [CrossRef] [PubMed]
- McGeown, W.J.; Mazzoni, G.; Vannucci, M.; Venneri, A. Structural and Functional Correlates of Hypnotic Depth and Suggestibility. Psychiatry Res. Neuroimaging 2015, 231, 151–159. [Google Scholar] [CrossRef]
- Cooper, N.R.; Burgess, A.P.; Croft, R.J.; Gruzelier, J.H. Investigating Evoked and Induced Electroencephalogram Activity in Task-Related Alpha Power Increases during an Internally Directed Attention Task. NeuroReport 2006, 17, 205–208. [Google Scholar] [CrossRef]
- Başar, E. The Theory of the Whole-Brain-Work. Int. J. Psychophysiol. 2006, 60, 133–138. [Google Scholar] [CrossRef]
- Klimesch, W.; Sauseng, P.; Hanslmayr, S. EEG Alpha Oscillations: The Inhibition–Timing Hypothesis. Brain Res. Rev. 2007, 53, 63–88. [Google Scholar] [CrossRef]
- Gruzelier, J.H. Frontal Functions, Connectivity and Neural Efficiency Underpinning Hypnosis and Hypnotic Susceptibility. Contemp. Hypnosis 2006, 23, 15–32. [Google Scholar] [CrossRef]
- Sebastiani, L.; Simoni, A.; Gemignani, A.; Ghelarducci, B.; Santarcangelo, E.L. Relaxation as a Cognitive Task. Arch. Ital. Biol. 2005, 143, 1–12. [Google Scholar] [PubMed]
- Cavallaro, F.I.; Cacace, I.; Del Testa, M.; Andre, P.; Carli, G.; De Pascalis, V.; Rocchi, R.; Santarcangelo, E.L. Hypnotizability-Related EEG Alpha and Theta Activities during Visual and Somesthetic Imageries. Neurosci. Lett. 2010, 470, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Ibáñez-Marcelo, E.; Campioni, L.; Phinyomark, A.; Petri, G.; Santarcangelo, E.L. Topology Highlights Mesoscopic Functional Equivalence between Imagery and Perception: The Case of Hypnotizability. NeuroImage 2019, 200, 437–449. [Google Scholar] [CrossRef]
- Ruggirello, S.; Campioni, L.; Piermanni, S.; Sebastiani, L.; Santarcangelo, E.L. Does Hypnotic Assessment Predict the Functional Equivalence between Motor Imagery and Action? Brain Cogn. 2019, 136, 103598. [Google Scholar] [CrossRef]
- Jensen, M.P.; Jamieson, G.A.; Lutz, A.; Mazzoni, G.; McGeown, W.J.; Santarcangelo, E.L.; Demertzi, A.; De Pascalis, V.; Bányai, É.I.; Rominger, C.; et al. New Directions in Hypnosis Research: Strategies for Advancing the Cognitive and Clinical Neuroscience of Hypnosis. Neurosci. Conscious. 2017, 2017, nix004. [Google Scholar] [CrossRef]
- Zaccaro, A.; Piarulli, A.; Melosini, L.; Menicucci, D.; Gemignani, A. Neural Correlates of Non-Ordinary States of Consciousness in Pranayama Practitioners: The Role of Slow Nasal Breathing. Front. Syst. Neurosci. 2022, 16, 803904. [Google Scholar] [CrossRef]
- Zeig, J.; Tanev, K.S. Advancing Hypnotic Inductions: An Ericksonian Perspective. Eur. J. Psychother. Couns. 2022, 24, 457–472. [Google Scholar] [CrossRef]
- Varga, K. Possible Mechanisms of Hypnosis from an Interactional Perspective. Brain Sci. 2021, 11, 903. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Callara, A.L.; Zelič, Ž.; Fontanelli, L.; Greco, A.; Santarcangelo, E.L.; Sebastiani, L. Is Hypnotic Induction Necessary to Experience Hypnosis and Responsible for Changes in Brain Activity? Brain Sci. 2023, 13, 875. https://doi.org/10.3390/brainsci13060875
Callara AL, Zelič Ž, Fontanelli L, Greco A, Santarcangelo EL, Sebastiani L. Is Hypnotic Induction Necessary to Experience Hypnosis and Responsible for Changes in Brain Activity? Brain Sciences. 2023; 13(6):875. https://doi.org/10.3390/brainsci13060875
Chicago/Turabian StyleCallara, Alejandro Luis, Žan Zelič, Lorenzo Fontanelli, Alberto Greco, Enrica Laura Santarcangelo, and Laura Sebastiani. 2023. "Is Hypnotic Induction Necessary to Experience Hypnosis and Responsible for Changes in Brain Activity?" Brain Sciences 13, no. 6: 875. https://doi.org/10.3390/brainsci13060875
APA StyleCallara, A. L., Zelič, Ž., Fontanelli, L., Greco, A., Santarcangelo, E. L., & Sebastiani, L. (2023). Is Hypnotic Induction Necessary to Experience Hypnosis and Responsible for Changes in Brain Activity? Brain Sciences, 13(6), 875. https://doi.org/10.3390/brainsci13060875






