Benzodiazepines and Mood Stabilizers in Schizophrenia Patients Treated with Oral versus Long-Acting Injectable Antipsychotics—An Observational Study
Abstract
1. Introduction
Aims
2. Materials and Methods
2.1. Data Source
2.2. Study Design
2.3. Statistical Analysis
3. Results
3.1. General Findings
3.2. Mood Stabilizers
3.3. Benzodiazepines
3.4. Monotherapy versus Polytherapy
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| LAI- | long-acting injectable |
| MS- | mood stabilizer |
| BZD- | benzodiazepine |
| OAP- | oral antipsychotics |
| SGA- | second-generation antipsychotic |
| GABA- | gamma-aminobutyric acid |
| VPA- | valproic acid |
| AD- | Antidepressant |
| FGA- | first-generation antipsychotics |
| SD- | standard deviation |
| OLZ- | olanzapine |
| RIS- | risperidone |
| PAL- | paliperidone |
| ARI- | aripiprazole |
| QUE- | quetiapine |
| AMI- | amisulpride |
| ZIP- | ziprasidone |
| HAL- | haloperidol |
| FLX- | flupenthixol |
| ZUC- | zuclopenthixol |
| LEV- | levomepromazine |
| TIA- | tiapridal |
| CLZ- | clozapine |
| EPS- | extrapyramidal symptoms |
References
- Kahn, R.S.; Sommer, I.E.; Murray, R.M.; Meyer-Lindenberg, A.; Weinberger, D.R.; Cannon, T.D.; O’Donovan, M.; Correll, C.U.; Kane, J.M.; van Os, J.; et al. Schizophrenia. Nat. Rev. Dis. Primers 2015, 1, 15067. [Google Scholar] [CrossRef] [PubMed]
- Johnstone, E.C.; Geddes, J. How high is the relapse rate in schizophrenia? Acta Psychiatr. Scand. Suppl. 1994, 382, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Lader, M. What is relapse in schizophrenia? Int. Clin. Psychopharmacol. 1995, 9 (Suppl. S5), 5–9. [Google Scholar] [CrossRef] [PubMed]
- Temmingh, H.; Stein, D.J. Anxiety in Patients with Schizophrenia: Epidemiology and Management. CNS Drugs 2015, 29, 819–832. [Google Scholar] [CrossRef] [PubMed]
- Enticott, P.G.; Ogloff, J.R.; Bradshaw, J.L. Response inhibition and impulsivity in schizophrenia. Psychiatry Res. 2008, 157, 251–254. [Google Scholar] [CrossRef]
- Xi, S.J.; Shen, M.X.; Wang, Y.; Zhou, W.; Xiao, S.Y.; Tebes, J.K.; Yu, Y. Depressive symptoms, anxiety symptoms, and their co-occurrence among people living with schizophrenia in China: Prevalence and correlates. J. Clin. Psychol. 2021, 77, 2137–2146. [Google Scholar] [CrossRef]
- Correll, C.U.; Citrome, L.; Haddad, P.M.; Lauriello, J.; Olfson, M.; Calloway, S.M.; Kane, J.M. The Use of Long-Acting Injectable Antipsychotics in Schizophrenia: Evaluating the Evidence. J. Clin. Psychiatry 2016, 77 (Suppl. S3), 3–24. [Google Scholar] [CrossRef]
- Taipale, H.; Mittendorfer-Rutz, E.; Alexanderson, K.; Majak, M.; Mehtälä, J.; Hoti, F.; Jedenius, E.; Enkusson, D.; Leval, A.; Sermon, J.; et al. Antipsychotics and mortality in a nationwide cohort of 29,823 patients with schizophrenia. Schizophr. Res. 2018, 197, 274–280. [Google Scholar] [CrossRef]
- Kishimoto, T.; Hagi, K.; Kurokawa, S.; Kane, J.M.; Correll, C.U. Long-acting injectable versus oral antipsychotics for the maintenance treatment of schizophrenia: A systematic review and comparative meta-analysis of randomised, cohort, and pre-post studies. Lancet Psychiatry 2021, 8, 387–404. [Google Scholar] [CrossRef]
- Kane, J.M.; McEvoy, J.P.; Correll, C.U.; Llorca, P.M. Controversies Surrounding the Use of Long-Acting Injectable Antipsychotic Medications for the Treatment of Patients with Schizophrenia. CNS Drugs 2021, 35, 1189–1205. [Google Scholar] [CrossRef]
- Ifteni, P.; Petric, P.S.; Teodorescu, A. Rating Opportunity for Long-Acting Injectable Antipsychotic Initiation Index (ROLIN). Front. Psychiatry 2021, 12, 767756. [Google Scholar] [CrossRef] [PubMed]
- Lehman, A.F.; Lieberman, J.A.; Dixon, L.B.; McGlashan, T.H.; Miller, A.L.; Perkins, D.O.; Kreyenbuhl, J.; American Psychiatric Association; Steering Committee on Practice Guidelines. Practice guideline for the treatment of patients with schizophrenia, second edition. Am. J. Psychiatry 2004, 161 (Suppl. S2), i–iv+1–56. [Google Scholar]
- Lähteenvuo, M.; Tiihonen, J. Antipsychotic Polypharmacy for the Management of Schizophrenia: Evidence and Recommendations. Drugs 2021, 81, 1273–1284. [Google Scholar] [CrossRef] [PubMed]
- Ballon, J.; Stroup, T.S. Polypharmacy for schizophrenia. Curr. Opin. Psychiatry 2013, 26, 208–213. [Google Scholar] [CrossRef]
- Baandrup, L. Polypharmacy in schizophrenia. Basic Clin. Pharmacol. Toxicol. 2020, 126, 183–192. [Google Scholar] [CrossRef]
- Włodarczyk, A.; Szarmach, J.; Cubała, W.J.; Wiglusz, M.S. Benzodiazepines in combination with antipsychotic drugs for schizophrenia: GABA-ergic targeted therapy. Psychiatr. Danub. 2017, 29 (Suppl. S3), 345–348. [Google Scholar]
- Szarmach, J.; Włodarczyk, A.; Cubała, W.J.; Wiglusz, M.S. Benzodiazepines as adjunctive therapy in treatment refractory symptoms of schizophrenia. Psychiatr. Danub. 2017, 29 (Suppl. S3), 349–352. [Google Scholar]
- Stimmel, G.L. Benzodiazepines in schizophrenia. Pharmacotherapy 1996, 16 Pt 2, 148S–151S, discussion 166S–168S. [Google Scholar]
- Van Kammen, D.P. gamma-Aminobutyric acid (Gaba) and the dopamine hypothesis of schizophrenia. Am. J. Psychiatry 1977, 134, 138–143. [Google Scholar] [CrossRef]
- Sim, F.; Sweetman, I.; Kapur, S.; Patel, M.X. Re-examining the role of benzodiazepines in the treatment of schizophrenia: A systematic review. J. Psychopharmacol. 2015, 29, 212–223. [Google Scholar] [CrossRef]
- Strømme, M.F.; Mellesdal, L.S.; Bartz-Johannesen, C.A.; Kroken, R.A.; Krogenes, M.L.; Mehlum, L.; Johnsen, E. Use of Benzodiazepines and Antipsychotic Drugs Are Inversely Associated with Acute Readmission Risk in Schizophrenia. J. Clin. Psychopharmacol. 2022, 42, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Fontanella, C.A.; Campo, J.V.; Phillips, G.S.; Hiance-Steelesmith, D.L.; Sweeney, H.A.; Tam, K.; Lehrer, D.; Klein, R.; Hurst, M. Benzodiazepine use and risk of mortality among patients with schizophrenia: A retrospective longitudinal study. J. Clin. Psychiatry 2016, 77, 661–667. [Google Scholar] [CrossRef] [PubMed]
- Citrome, L. Treatment-resistant schizophrenia: What role for mood stabilizers? Curr. Psychiatry 2004, 3, 23–40. [Google Scholar]
- Chiu, C.-T.; Wang, Z.; Hunsberger, J.G.; Chuang, D.-M. Therapeutic Potential of Mood Stabilizers Lithium and Valproic Acid: Beyond Bipolar Disorder. Pharmacol. Rev. 2013, 65, 105–142. [Google Scholar] [CrossRef] [PubMed]
- Leucht, S.; Helfer, B.; Dold, M.; Kissling, W.; McGrath, J. Lithium for schizophrenia. Cochrane Database Syst. Rev. 2015, 10, CD003834. [Google Scholar] [CrossRef] [PubMed]
- Citrome, L.; Levine, J.; Allingham, B. Changes in use of valproate and other mood stabilizers for patients with schizophrenia from 1994 to 1998. Psychiatr. Serv. 2000, 51, 634–638. [Google Scholar] [CrossRef]
- Puranen, A.; Koponen, M.; Tanskanen, A.; Tiihonen, J.; Taipale, H. Use of antidepressants and mood stabilizers in persons with first-episode schizophrenia. Eur. J. Clin. Pharmacol. 2020, 76, 711–718. [Google Scholar] [CrossRef]
- Lim, W.K.; Chew, Q.H.; He, Y.L.; Si, T.M.; Chiu, F.H.; Xiang, Y.T.; Kato, T.A.; Kanba, S.; Shinfuku, N.; Lee, M.S.; et al. Coprescription of mood stabilizers in schizophrenia, dosing, and clinical correlates: An international study. Hum. Psychopharmacol. 2020, 35, 1–7. [Google Scholar] [CrossRef]
- Berle, J.Ø.; Spigset, O. Har stemningsstabiliserende legemidler noen plass i behandlingen av schizofreni? [Are mood stabilizers beneficial in the treatment of schizophrenia?]. Tidsskr Nor Laegeforen. 2005, 125, 1809–1812. [Google Scholar]
- Casey, D.E.; Daniel, D.G.; Wassef, A.A.; Tracy, K.A.; Wozniak, P.; Sommerville, K.W. Effect of divalproex combined with olanzapine or risperidone in patients with an acute exacerbation of schizophrenia. Neuropsychopharmacology 2003, 28, 182–192. [Google Scholar] [CrossRef]
- Puranen, A.; Koponen, M.; Lähteenvuo, M.; Tanskanen, A.; Tiihonen, J.; Taipale, H. Real-world effectiveness of mood stabilizer use in schizophrenia. Acta Psychiatr. Scand. 2022; ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Tseng, P.T.; Chen, Y.W.; Chung, W.; Tu, K.Y.; Wang, H.Y.; Wu, C.K.; Lin, P.Y. Significant Effect of Valproate Augmentation Therapy in Patients with Schizophrenia: A Meta-analysis Study. Medicine 2016, 95, e2475. [Google Scholar] [CrossRef] [PubMed]
- Atkins, M.; Burgess, A.; Bottomley, C.; Riccio, M. Chlorpromazine equivalents: A consensus of opinion for both clinical and research applications. Psychiatr. Bull. 1997, 21, 224–226. [Google Scholar] [CrossRef]
- Inada, T.; Inagaki, A. Psychotropic dose equivalence in Japan. Psychiatry Clin. Neurosci. 2015, 69, 440–447. [Google Scholar] [CrossRef] [PubMed]
- Gardner, D.M.; Murphy, A.L.; O’Donnell, H.; Centorrino, F.; Baldessarini, R.J. International consensus study of antipsychotic dosing. Am. J. Psychiatry 2010, 167, 686–693. [Google Scholar] [CrossRef]
- Leucht, S.; Samara, M.; Heres, S.; Patel, M.X.; Furukawa, T.; Cipriani, A.; Geddes, J.; Davis, J.M. Dose Equivalents for Second-Generation Antipsychotic Drugs: The Classical Mean Dose Method. Schizophr. Bull. 2015, 41, 1397–1402. [Google Scholar] [CrossRef] [PubMed]
- Sajatovic, M.; Ross, R.; Legacy, S.N.; Correll, C.U.; Kane, J.M.; DiBiasi, F.; Fitzgerald, H.; Byerly, M. Identifying patients and clinical scenarios for use of long-acting injectable antipsychotics—Expert consensus survey part 1. Neuropsychiatr. Dis. Treat. 2018, 14, 1463–1474. [Google Scholar] [CrossRef]
- Offord, S.; Wong, B.; Mirski, D.; Baker, R.A.; Lin, J. Healthcare resource usage of schizophrenia patients initiating long-acting injectable antipsychotics vs oral. J. Med. Econ. 2013, 16, 231–239. [Google Scholar] [CrossRef]
- Bareis, N.; Olfson, M.; Wall, M.; Stroup, T.S. Variation in Psychotropic Medication Prescription for Adults with Schizophrenia in the United States. Psychiatr. Serv. 2022, 73, 492–500, Epub 2021 Sep 30. [Google Scholar] [CrossRef]
- Reymann, S.; Schoretsanitis, G.; Egger, S.T.; Mohonko, A.; Kirschner, M.; Vetter, S.; Homan, P.; Seifritz, E.; Burrer, A. Use of Long-Acting Injectable Antipsychotics in Inpatients with Schizophrenia Spectrum Disorder in an Academic Psychiatric Hospital in Switzerland. J. Pers. Med. 2022, 12, 441. [Google Scholar] [CrossRef]
- Stip, E.; Lachaine, J. Real-world effectiveness of long-acting antipsychotic treatments in a nationwide cohort of 3957 patients with schizophrenia, schizoaffective disorder and other diagnoses in Quebec. Ther. Adv. Psychopharmacol. 2018, 8, 287–301, Erratum in Ther. Adv. Psychopharmacol. 2018, 8, 327. [Google Scholar] [CrossRef] [PubMed]
- Pilon, D.; Joshi, K.; Tandon, N.; Lafeuille, M.H.; Kamstra, R.L.; Emond, B.; Lefebvre, P. Treatment patterns in Medicaid patients with schizophrenia initiated on a first- or second-generation long-acting injectable versus oral antipsychotic. Patient Prefer. Adherence 2017, 11, 619–629. [Google Scholar] [CrossRef] [PubMed]
- Llorca, P.M.; Bobes, J.; Fleischhacker, W.W.; Heres, S.; Moore, N.; Bent-Ennakhil, N.; Sapin, C.; Loze, J.Y.; Nylander, A.G.; Patel, M.X. Baseline results from the European non-interventional Antipsychotic Long acTing injection in schizOphrenia (ALTO) study. Eur. Psychiatry 2018, 52, 85–94. [Google Scholar] [CrossRef]
- Martin, A.; Bessonova, L.; Hughes, R.; Doane, M.J.; O’Sullivan, A.K.; Snook, K.; Cichewicz, A.; Weiden, P.J.; Harvey, P.D. Systematic Review of Real-World Treatment Patterns of Oral Antipsychotics and Associated Economic Burden in Patients with Schizophrenia in the United States. Adv. Ther. 2022, 39, 3933–3956. [Google Scholar] [CrossRef]
- Lally, J.; Gaughran, F.; Timms, P.; Curran, S.R. Treatment-resistant schizophrenia: Current insights on the pharmacogenomics of antipsychotics. Pharm. Pers. Med. 2016, 9, 117. [Google Scholar] [CrossRef] [PubMed]
- Miron, A.A.; Ifteni, P.I.; Teodorescu, A.; Petric, P.S. Long-Acting Injectable Antipsychotics (LAIs) Prescribing Trends during the COVID-19 Pandemic in Romania. Healthcare 2022, 10, 1265. [Google Scholar] [CrossRef] [PubMed]
- Miron, A.A.; Teodorescu, A.; Ifteni, P.; Irimie, C.A.; Dima, L.; Petric, P.S. Switch from Olanzapine Long-Acting Injectable to its Oral Equivalent during COVID-19 Pandemic: A Real World Observational Study. Psychiatr. Q. 2022, 93, 627–635. [Google Scholar] [CrossRef]
- Gruppuso, P.A.; Ahmed, R.; Adashi, E.Y. Valproate Teratogenicity: A Moving Target. Obstet. Gynecol. 2022, 140, 408–411. [Google Scholar] [CrossRef]
- Paton, C.; Citrome, L.; Fernandez-Egea, E.; Rendora, O.; Barnes, T.R.E. Who is prescribed valproate and how carefully is this treatment reviewed in UK mental health services? Data from a clinical audit. Ther. Adv. Psychopharmacol. 2022, 12, 1–15. [Google Scholar] [CrossRef]
- Wang, Y.; Xia, J.; Helfer, B.; Li, C.; Leucht, S. Valproate for schizophrenia. Cochrane Database Syst Rev. 2016, 2016, CD004028. [Google Scholar] [CrossRef]
- Leucht, S.; Helfer, B.; Dold, M.; Kissling, W.; McGrath, J. Carbamazepine for schizophrenia. Cochrane Database Syst. Rev. 2014, 2014, CD001258. [Google Scholar] [CrossRef] [PubMed]
- Premkumar, T.S.; Pick, J. Lamotrigine for schizophrenia. Cochrane Database Syst. Rev. 2006, 2006, CD005962. [Google Scholar] [CrossRef] [PubMed]
- Paton, C.; Banham, S.; Whitmore, J. Benzodiazepines in schizophrenia: Is there a trend towards long-term prescribing? Psychiatr. Bull. 2000, 24, 113–115. [Google Scholar] [CrossRef]
- Dold, M.; Li, C.; Gillies, D.; Leucht, S. Benzodiazepine augmentation of antipsychotic drugs in schizophrenia: A meta-analysis and Cochrane review of randomized controlled trials. Eur. Neuropsychopharmacol. 2013, 23, 1023–1033. [Google Scholar] [CrossRef]
- Dold, M.; Li, C.; Tardy, M.; Khorsand, V.; Gillies, D.; Leucht, S. Benzodiazepines for schizophrenia. Cochrane Database Syst. Rev. 2012, 11, CD006391. [Google Scholar] [CrossRef]
- Stewart, S.A. The effects of benzodiazepines on cognition. J. Clin. Psychiatry 2005, 66 (Suppl. S2), 9–13. [Google Scholar]
- Nielsen, S. Benzodiazepines. Curr. Top. Behav. Neurosci. 2017, 34, 141–159. [Google Scholar] [CrossRef]
- Marcum, Z.A.; Gellad, W.F. Medication adherence to multidrug regimens. Clin. Geriatr. Med. 2012, 28, 287–300. [Google Scholar] [CrossRef]
- Leucht, S.; Crippa, A.; Siafis, S.; Patel, M.X.; Orsini, N.; Davis, J.M. Dose-Response Meta-Analysis of Antipsychotic Drugs for Acute Schizophrenia. Am. J. Psychiatry 2020, 177, 342–353, Erratum in Am. J. Psychiatry 2020, 177, 272. [Google Scholar] [CrossRef]
- Sparshatt, A.; Jones, S.; Taylor, D. Quetiapine: Dose-response relationship in schizophrenia. CNS Drugs 2008, 22, 49–68, discussion 69–72. [Google Scholar] [CrossRef]
- Armitage, R.; Cole, D.; Suppes, T.; Ozcan, M.E. Effects of clozapine on sleep in bipolar and schizoaffective disorders. Prog. Neuropsychopharmacol. Biol. Psychiatry 2004, 28, 1065–1070. [Google Scholar] [CrossRef]
- Ali, T.; Sisay, M.; Tariku, M.; Mekuria, A.N.; Desalew, A. Antipsychotic-induced extrapyramidal side effects: A systematic review and meta-analysis of observational studies. PLoS ONE 2021, 16, e0257129. [Google Scholar] [CrossRef] [PubMed]
- Novick, D.; Haro, J.M.; Bertsch, J.; Haddad, P.M. Incidence of extrapyramidal symptoms and tardive dyskinesia in schizophrenia: Thirty-six-month results from the European schizophrenia outpatient health outcomes study. J. Clin. Psychopharmacol. 2010, 30, 531–540. [Google Scholar] [CrossRef] [PubMed]
- Park, S.C.; Choi, M.Y.; Choi, J.; Park, E.; Tchoe, H.J.; Suh, J.K.; Kim, Y.H.; Won, S.H.; Chung, Y.C.; Bae, K.Y.; et al. Comparative Efficacy and Safety of Long-acting Injectable and Oral Second-generation Antipsychotics for the Treatment of Schizophrenia: A Systematic Review and Meta-analysis. Clin. Psychopharmacol. Neurosci. 2018, 16, 361–375. [Google Scholar] [CrossRef] [PubMed]
- Miller, D.D. Atypical antipsychotics: Sleep, sedation, and efficacy. Prim. Care Companion J. Clin. Psychiatry 2004, 6 (Suppl. S2), 3–7. [Google Scholar]
- Wong, J.; Delva, N. Clozapine-induced seizures: Recognition and treatment. Can. J. Psychiatry 2007, 52, 457–463. [Google Scholar] [CrossRef]
- Lin, S.K. Antipsychotic Polypharmacy: A Dirty Little Secret or a Fashion? Int. J. Neuropsychopharmacol. 2020, 23, 125–131. [Google Scholar] [CrossRef]
- Tiihonen, J.; Taipale, H.; Mehtälä, J.; Vattulainen, P.; Correll, C.U.; Tanskanen, A. Association of Antipsychotic Polypharmacy vs Monotherapy with Psychiatric Rehospitalization among Adults with Schizophrenia. JAMA Psychiatry 2019, 76, 499–507. [Google Scholar] [CrossRef]
- Zhang, L.; He, S.; He, L.; Yu, W.; He, S.; Li, Y.; Yu, Y.; Zheng, Q.; Huang, J.; Shen, Y.; et al. Long-Term Antipsychotic Effectiveness and Comparison of the Efficacy of Monotherapy and Polypharmacy in Schizophrenia: A 3-Years Follow-Up “Real World” Study in China. Front. Pharmacol. 2022, 13, 860713. [Google Scholar] [CrossRef]
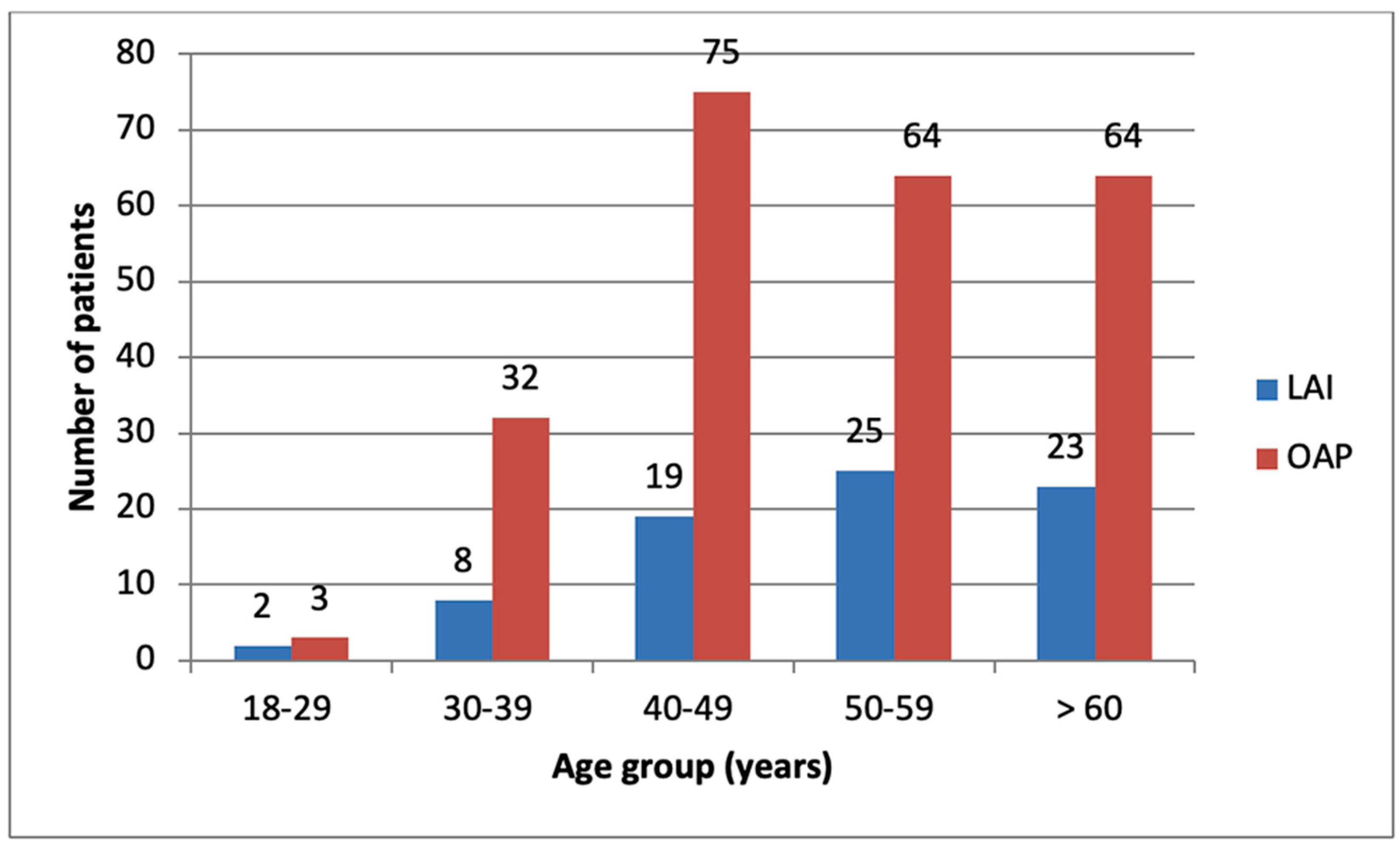
| Parameters | LAIs | OAPs | p-Value | |
|---|---|---|---|---|
| Number of patients (N, %) | 77 (24.44%) | 238 (75.56%) | p < 0.0001 | |
| Male gender (N, %) | 34 (44.15%) | 96 (40.34%) | p = 0.55 | |
| Mean age (±SD) | 52.92 (±12.24 SD) | 51.32 (±11.10 SD) | p = 0.28 | |
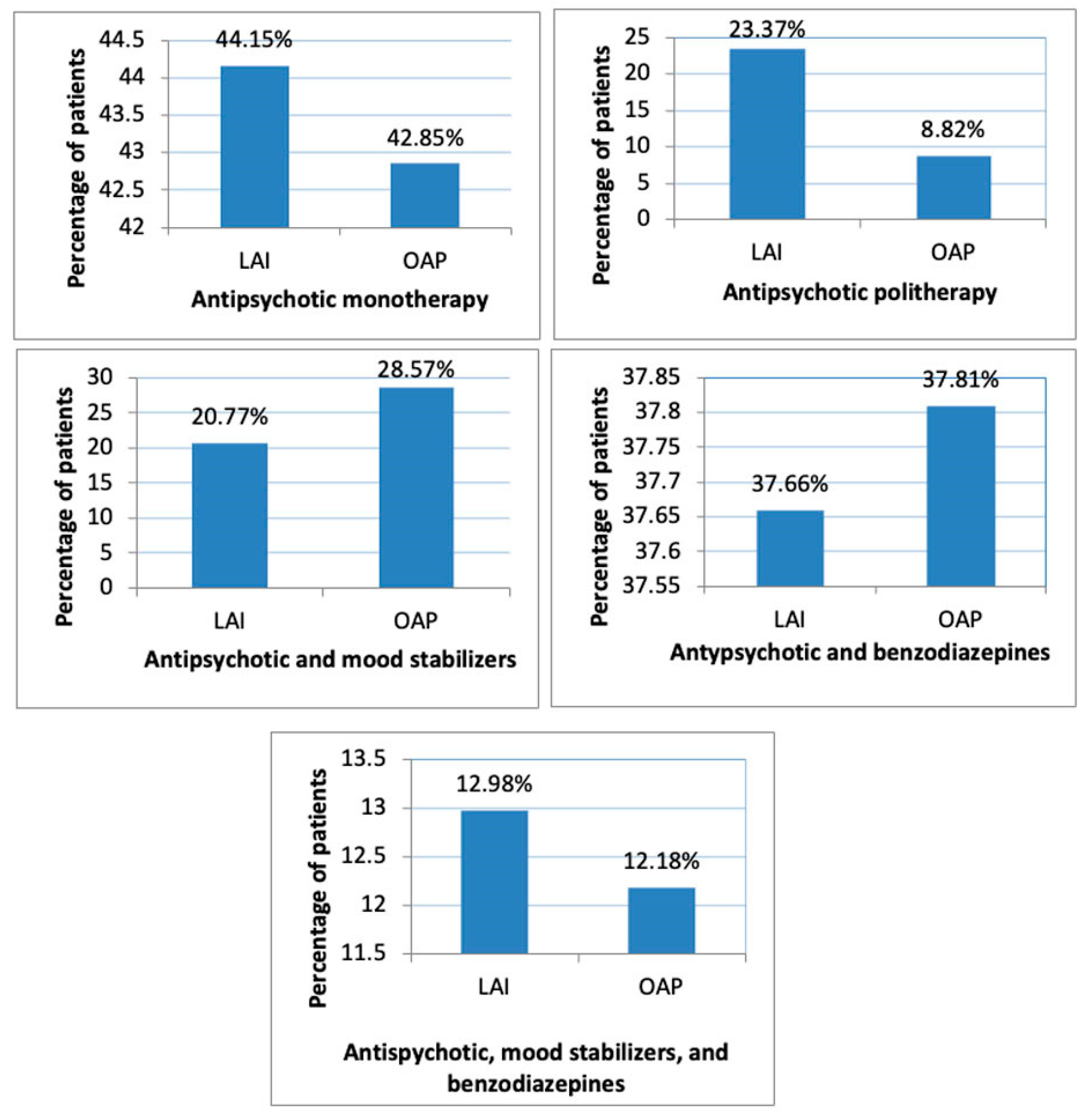
| Patients receiving BZD (N, %) | Total | 29 (37.66%) | 90 (37.81%) | p = 0.98 |
| Male | 11 (37.93%) | 34 (37.77%) | p = 0.98 | |
| Female | 18 (62.07%) | 56 (62.23%) | p = 0.93 | |
| Patients receiving MS (N, %) | Total | 16 (20.77%) | 68 (28.57%) | p = 0.17 |
| Male | 8 (50%) | 32 (47.05%) | p = 0.83 | |
| Female | 8 (50%) | 36 (52.95%) | p = 0.83 | |
| Patients receiving both MS and BZD (N, %) | Total | 10 (12.98%) | 29 (12.18%) | p = 0.85 |
| Male | 5 (50%) | 15 (51.72%) | p = 0.92 | |
| Female | 5 (50%) | 14 (48.28%) | p = 0.92 | |
| Antipsychotic type | OLZ (N, %) | 5 (6.49%) | 70 (29.41%) | p ˂ 0.0001 |
| RIS (N, %) | 16 (20.77%) | 31 (13.02%) | p = 0.09 | |
| PAL (N, %) | 9 (11.68%) | 17 (7.14%) | p = 0.20 | |
| ARI (N, %) | 9 (11.68%) | 24 (10.08%) | p = 0.69 | |
| QUE (N, %) | - | 24 (10.08%) | - | |
| AMI (N, %) | - | 20 (8.40%) | - | |
| ZIP (N, %) | - | 1 (0.42%) | - | |
| HAL (N, %) | 0 | 22 (9.24%) | - | |
| FLX (N, %) | 36 (46.75%) | - | - | |
| ZUC (N, %) | 2 (2.59%) | - | - | |
| LEV (N, %) | - | 3 (1.26%) | - | |
| TIA (N, %) | - | 1 (0.42%) | - | |
| CLZ (N, %) | - | 66 (27.73%) | - | |
| Antipsychotic (Type, Formulation) | Number of Cases | Mean Dose (mg) | Oral Dose Equivalent (mg) | Chlorpro-Mazine Equivalent (mg) | p-Value | ||
|---|---|---|---|---|---|---|---|
| olanzapine | LAI | 5 | 480 (±164.31) | 16 (±5.47) | 320 | p = 0.66 | |
| OAP | 70 | 15 (±5) | 15 (±5) | 300 | |||
| risperidone | LAI | 16 | 76.56 (±24.94) | 3.06 (±0.99) | 306 | p = 0.21 | |
| OAP | 31 | 3.58 (±1.50) | 3.58 (±1.50) | 358 | |||
| aripiprazole | LAI | 9 | 400 | 20 | 266.66 | p = 0.22 | |
| OAP | 24 | 16.875 (±7.49) | 16.875 (±7.49) | 225 | |||
| paliperidone | LAI | 9 | 180.55 (±152.97) | 9.66 (±1.32) | 483 | p = 0.005 | |
| OAP | 15 | 7.4 (±1.91) | 7.4 (±1.91) | 370 | |||
| quetiapine | LAI | - | - | - | - | NA | |
| OAP | 24 | 491.66 (±224.89) | - | 655 | |||
| amisulpride | LAI | - | - | - | - | NA | |
| OAP | 20 | 500 (±247.08) | - | 290 | |||
| ziprasidone | LAI | - | - | - | - | NA | |
| OAP | 1 | 120 | - | 200 | |||
| haloperidol | LAI | - | - | - | - | NA | |
| OAP | 22 | 3.52 (±1.61) | - | 176 | |||
| flupenthixol | LAI | 36 | 35 (±8.78) | - | 100 | NA | |
| OAP | - | - | - | - | |||
| zuclopenthixol | LAI | 2 | 200 | - | 100 | NA | |
| OAP | - | - | - | - | |||
| levomepromazine | LAI | - | - | - | - | NA | |
| OAP | 3 | 54.16 (±7.21) | - | 54.16 | |||
| tiapridal | LAI | - | - | - | - | NA | |
| OAP | 1 | 100 | - | 100 | |||
| clozapine | LAI | - | - | - | - | NA | |
| OAP | Total | 66 | 272.72 (±126.51) | - | 272.72 | ||
| clozapine monotherapy | 53 | 297.64 (±128.56) | 297.64 | ||||
| Diazepam | Alprazolam | Lorazepam | Clonazepam | Bromazepam | Nitrazepam | Cinolazepam | |
|---|---|---|---|---|---|---|---|
| SGA-LAI (N, %) | 4 (10.25%) | 1 (2.56%) | 3 (7.69) | 2 (5.12%) | 1 (2.56%) | 1 (2.56%) | 1 (2.56%) |
| FGA-LAI (N, %) | 8 (21.05%) | 0 | 8 (21.05%) | 1 (2.63%) | 1 (2.63%) | 0 | 1 (2.63%) |
| OAP (N, %) | 43 (18.06%) | 4 (1.68%) | 49 (20.58%) | 16 (6.72%) | 2 (0.84%) | 0 | 1 (0.42%) |
| Antipsychotic Type | Formulation | MS (N, %) | p-Value | BZD (N, %) | p-Value | MSs and BZDs (N, %) | p-Value |
|---|---|---|---|---|---|---|---|
| olanzapine | LAI (N = 5) | 1 (20%) | p = 0.77 | 0 | p = 0.10 | 0 | p = 0.42 |
| OAP (N = 70) | 18 (25.71%) | 25 (35.71%) | 8 (11.42%) | ||||
| risperidone | LAI (N = 16) | 4 (25%) | p = 0.47 | 6 (37.50%) | p = 0.93 | 3 (18.75%) | p = 0.96 |
| OAP (N = 31) | 11 (35.48%) | 12 (38.70) | 6 (19.35%) | ||||
| aripiprazole | LAI (N = 9) | 2 (22.22%) | p = 0.69 | 2 (22.22%) | p = 0.54 | 2 (22.22%) | p = 0.49 |
| OAP (N = 24) | 7 (29.16%) | 8 (33.33%) | 3 (12.50%) | ||||
| paliperidone | LAI (N = 9) | 2 (22.22%) | p = 0.57 | 3 (33.33%) | p = 0.73 | 1 (11.11%) | p = 0.70 |
| OAP (N=15) | 5 (33.33%) | 4 (26.66%) | 1 (6.66%) |
| Antipsychotic (Type, Formulation) | Number of Patients | Patients Stabilized on Monotherapy (N, %) | p-Value | |
|---|---|---|---|---|
| olanzapine | LAI | 5 | 4 (80%) | p = 0.18 |
| OAP | 70 | 32 (45.71%) | ||
| risperidone | LAI | 16 | 8 (50%) | p = 0.24 |
| OAP | 31 | 10 (32.25%) | ||
| aripiprazole | LAI | 9 | 5 (55.55%) | p = 0.25 |
| OAP | 24 | 8 (33.33%) | ||
| paliperidone | LAI | 9 | 4 (44.44%) | p = 0.83 |
| OAP | 15 | 6 (40%) | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miron, A.A.; Petric, P.S.; Teodorescu, A.; Ifteni, P.; Chele, G.; Szalontay, A.S. Benzodiazepines and Mood Stabilizers in Schizophrenia Patients Treated with Oral versus Long-Acting Injectable Antipsychotics—An Observational Study. Brain Sci. 2023, 13, 173. https://doi.org/10.3390/brainsci13020173
Miron AA, Petric PS, Teodorescu A, Ifteni P, Chele G, Szalontay AS. Benzodiazepines and Mood Stabilizers in Schizophrenia Patients Treated with Oral versus Long-Acting Injectable Antipsychotics—An Observational Study. Brain Sciences. 2023; 13(2):173. https://doi.org/10.3390/brainsci13020173
Chicago/Turabian StyleMiron, Ana Aliana, Paula Simina Petric, Andreea Teodorescu, Petru Ifteni, Gabriela Chele, and Andreea Silvana Szalontay. 2023. "Benzodiazepines and Mood Stabilizers in Schizophrenia Patients Treated with Oral versus Long-Acting Injectable Antipsychotics—An Observational Study" Brain Sciences 13, no. 2: 173. https://doi.org/10.3390/brainsci13020173
APA StyleMiron, A. A., Petric, P. S., Teodorescu, A., Ifteni, P., Chele, G., & Szalontay, A. S. (2023). Benzodiazepines and Mood Stabilizers in Schizophrenia Patients Treated with Oral versus Long-Acting Injectable Antipsychotics—An Observational Study. Brain Sciences, 13(2), 173. https://doi.org/10.3390/brainsci13020173







