Motion, Relation, and Passion in Brain Physiological and Cognitive Aging
Abstract
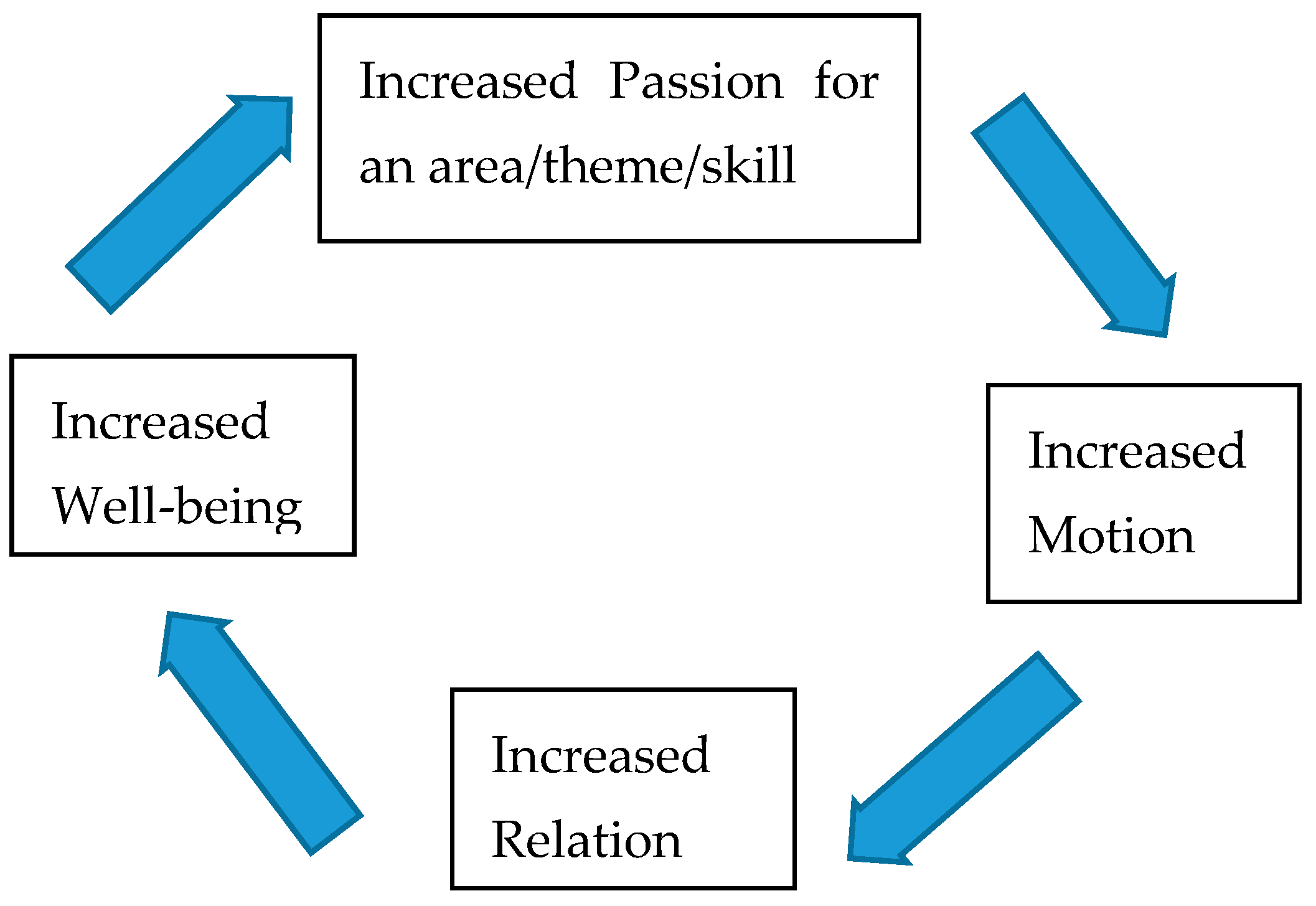
:1. Introduction
2. Motion
3. Relation
4. Passion
5. Limitations and Future Directions
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Giorgio, A.; Santelli, L.; Tomassini, V.; Bosnell, R.; Smith, S.; De Stefano, N.; Johansen-Berg, H. Age-related changes in grey and white matter structure throughout adulthood. Neuroimage 2010, 51, 943–951. [Google Scholar] [CrossRef]
- Cabeza, R.; Albert, M.; Belleville, S.; Craik, F.I.M.; Duarte, A.; Grady, C.L.; Lindenberger, U.; Nyberg, L.; Park, D.C.; Reuter-Lorenz, P.A.; et al. Maintenance, reserve and compensation: The cognitive neuroscience of healthy ageing. Nat. Rev. Neurosci. 2018, 19, 701–710. [Google Scholar] [CrossRef]
- Principles of Neural Science; Kandel, E.R.; Schwartz, J.H.; Jessell, T.M.; Siegelbaum, S.; Hudspeth, A.J.; Mack, S. (Eds.) McGraw-hill: New York, NY, USA, 2000; Volume 4, pp. 1227–1246. [Google Scholar]
- Shagrir, O. Why we view the brain as a computer. Synthese 2006, 153, 393–416. [Google Scholar] [CrossRef]
- Jiang, G.; Yin, X.; Li, C.; Li, L.; Zhao, L.; Evans, A.C.; Jiang, T.; Wu, J.; Wang, J. The Plasticity of Brain Gray Matter and White Matter following Lower Limb Amputation. Neural Plast. 2015, 2015, 823185. [Google Scholar] [CrossRef]
- Pascual-Leone, A.; Amedi, A.; Fregni, F.; Merabet, L.B. The plastic human brain cortex. Annu. Rev. Neurosci 2005, 28, 377–401. [Google Scholar] [CrossRef] [PubMed]
- Vaynman, S.; Gomez-Pinilla, F. Revenge of the “sit”: How lifestyle impacts neuronal and cognitive health through molecular systems that interface energy metabolism with neuronal plasticity. J. Neurosci. Res. 2006, 84, 699–715. [Google Scholar] [CrossRef] [PubMed]
- Falck, R.S.; Davis, J.C.; Liu-Ambrose, T. What is the association between sedentary behaviour and cognitive function? A systematic review. Br. J. Sports Med. 2017, 51, 800–811. [Google Scholar] [CrossRef] [PubMed]
- Phillips, C. Lifestyle modulators of neuroplasticity: How physical activity, mental engagement, and diet promote cognitive health during aging. Neural Plast. 2017, 2017, 3589271. [Google Scholar] [CrossRef]
- Schmithorst, V.J.; Wilke, M.; Dardzinski, B.J.; Holland, S.K. Cognitive functions correlate with white matter architecture in a normal pediatric population: A diffusion tensor MRI study. Hum. Brainmapping 2005, 26, 139–147. [Google Scholar] [CrossRef]
- Bartzokis, G. Age-related myelin breakdown: A developmental model of cognitive decline and Alzheimer’s disease. Neurobiol. Aging 2004, 25, 5–18. [Google Scholar] [CrossRef]
- Sowell, E.R.; Peterson, B.S.; Thompson, P.M.; Welcome, S.E.; Henkenius, A.L.; Toga, A.W. Mapping cortical change across the human life span. Nat. Neurosci. 2003, 6, 309. [Google Scholar] [CrossRef] [PubMed]
- Ge, Y.; Grossman, R.I.; Babb, J.S.; Rabin, M.L.; Mannon, L.J.; Kolson, D.L. Age-related total gray matter and white matter changes in normal adult brain. Part I: Volumetric MR imaging analysis. Am. J. Neuroradiol. 2002, 23, 1327–1333. [Google Scholar] [PubMed]
- Leversen, J.S.; Haga, M.; Sigmundsson, H. From children to adults: Motor performance across the life-span. PLoS ONE 2012, 7, e38830. [Google Scholar] [CrossRef] [PubMed]
- Webb, S.J.; Monk, C.S.; Nelson, C.A. Mechanisms of postnatal neurobiological development: Implications for human development. Dev. Neuropsychol. 2001, 19, 147–171. [Google Scholar] [CrossRef]
- Peters, A.; Morrison, J.H.; Rosene, D.L.; Hyman, B.T. Are neurons lost from the primate cerebral cortex during normal aging? Cereb. Cortex 1998, 8, 295–300. [Google Scholar] [CrossRef]
- Farkas, E.; Luiten, P.G. Cerebral microvascular pathology in aging and Alzheimer’s disease. Prog. Neurobiol. 2001, 64, 575–611. [Google Scholar] [CrossRef]
- Edelman, G.M. Neural Darwinism: The Theory of Neuronal Group Selection; Basic Books; American Psychological Association (APA): Washington, DC, USA, 1987. [Google Scholar]
- Edelman, G.M. Bright Air, Brilliant Fire: On the Matter of the Mind; Basic books; American Psychological Association (APA): Washington, DC, USA, 1992. [Google Scholar]
- Edelman, G.M. Neural Darwinism: Selection and reentrant signaling in higher brain function. Neuron 1993, 10, 115–125. [Google Scholar] [CrossRef]
- Cabeza, R. Hemispheric Asymmetry Reduction in Older Adults: The HAROLD Model. Psych Aging 2002, 17, 85–100. [Google Scholar] [CrossRef]
- Cabeza, R.; Anderson, N.D.; Locantore, J.K.; McIntosh, A.R. Aging Gracefully: Compensatory Brain Activity in High-Performing Older Adults. Neuroimage 2002, 17, 1394–1402. [Google Scholar] [CrossRef]
- Ward, N.S. Compensatory mechanisms in the aging motor system. Ageing Res. Rev. 2006, 5, 239–254. [Google Scholar] [CrossRef]
- Kleim, J.A.; Jones, T.A. Principles of experience-dependent neural plasticity: Implications for rehabilitation after brain damage. J. Speech Lang. Hear. Res. 2008, 51. [Google Scholar] [CrossRef]
- Baltes, P.B. Theoretical Propositions of Life-Span Developmental Psychology. Dev. Psych. 1987, 2, 611–626. [Google Scholar] [CrossRef]
- Burke, S.N.; Barnes, C.A. Neural plasticity in the ageing brain. Nat. Rev. Neurosc. 2006, 7, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Resnick, S.M.; Pham, D.L.; Kraut, M.A.; Zonderman, A.B.; Davatzikos, C. Longitudinal Magnetic Resonance Imaging Studies of Older Adults: A Shrinking Brain. J. Neurosci. 2003, 23, 3295–3301. [Google Scholar] [CrossRef]
- Tanaka, M.; Toldi, J.; Vécsei, L. Exploring the etiological links behind neurodegenerative diseases: Inflammatory cytokines and bioactive kynurenines. Int. J. Mol. Sci. 2020, 21, 2431. [Google Scholar] [CrossRef]
- Onchev, G. Changes in Psychopathology and Mental Health Resilience. Front. Psychiatry 2021, 12, 676492. [Google Scholar] [CrossRef]
- Tanaka, M.; Vécsei, L. Editorial of Special Issue ‘Dissecting Neurological and Neuropsychiatric Diseases: Neurodegeneration and Neuroprotection’. Int. J. Mol. Sci. 2022, 23, 6991. [Google Scholar] [CrossRef]
- Pérez-González, M.; Badesso, S.; Lorenzo, E.; Guruceaga, E.; Pérez-Mediavilla, A.; García-Osta, A.; Cuadrado-Tejedor, M. Identifying the Main Functional Pathways Associated with Cognitive Resilience to Alzheimer’s Disease. Int. J. Mol. Sci. 2021, 22, 9120. [Google Scholar] [CrossRef]
- Hepsomali, P.; Coxon, C. Inflammation and diet: Focus on mental and cognitive health. Adv. Clin. Exp. Med. Off. Organ Wroc. Med. Univ. 2022. [Google Scholar] [CrossRef]
- Martos, D.; Tuka, B.; Tanaka, M.; Vécsei, L.; Telegdy, G. Memory enhancement with kynurenic acid and its mechanisms in neurotransmission. Biomedicines 2022, 10, 849. [Google Scholar] [CrossRef]
- Battaglia, S.; Cardellicchio, P.; Di Fazio, C.; Nazzi, C.; Fracasso, A.; Borgomaneri, S. The Influence of Vicarious Fear-Learning in “Infecting” Reactive Action Inhibition. Front. Behav. Neurosci. 2022, 16, 946263. [Google Scholar] [PubMed]
- Livingston, G.; Sommerlad, A.; Orgeta, V.; Costafreda, S.G.; Huntley, J.; Ames, D.; Ballard, C.; Banerjee, S.; Burns, A.; Cohen-Mansfield, J.; et al. Dementia prevention, intervention, and care. Lancet 2017, 390, 2673–2734. [Google Scholar] [CrossRef]
- Petersen, R.C.; Lopez, O.; Armstrong, M.J.; Getchius, T.S.; Ganguli, M.; Gloss, D.; Gronseth, G.S.; Marson, D.; Pringsheim, T.; Day, G.S.; et al. Practice guideline update summary: Mild cognitive impairment: Report of the Guideline Development, Dissemination, and Implementation. Subcommittee of the American Academy of Neurology. Neurology 2018, 90, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Petersen, R.C.; Joyner, M.J.; Jack, C.R. Cardiorespiratory Fitness and Brain Volumes. Mayo Clin. Proc. 2020, 95, 6–8. [Google Scholar] [CrossRef] [PubMed]
- Herold, F.; Hamacher, D.; Schega, L.; Müller, N.G. Thinking while moving or moving while thinking–concepts of motor-cognitive training for cognitive performance enhancement. Front. Aging Neurosci. 2018, 10, 228. [Google Scholar] [CrossRef]
- House, J.S.; Landis, K.R.; Umberson, D. Social relationships and health. Science 1988, 241, 540–545. [Google Scholar] [CrossRef]
- Gilbert, P. The Compassionate Mind; Robinson: London, UK, 2009. [Google Scholar]
- Zatorre, R.J. Predispositions and plasticity in music and speech learning: Neural correlates and implications. Science 2013, 342, 585–589. [Google Scholar] [CrossRef]
- Sigmundsson, H.; Haga, M.; Hermundsdottir, F. The passion scale: Aspects of reliability and validity of a new 8-item scale assessing passion. New Ideas Psychol. 2020, 56, 100745. [Google Scholar] [CrossRef]
- Sigmundsson, H.; Haga, M.; Hermundsdottir, F. Passion, grit and mindset in young adults: Exploring the relationship and gender differences. New Ideas Psychol. 2020, 59, 100795. [Google Scholar] [CrossRef]
- Di Liegro, C.M.; Schiera, G.; Proia, P.; Di Liegro, I. Physical activity and brain health. Genes 2019, 10, 720. [Google Scholar] [CrossRef]
- Lim, A.F.Y.; Lim, W.L.; Ch’ng, T.H. Activity-dependent synapse to nucleus signaling. Neurobiol. Learn. Mem. 2017, 138, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Bading, H. Nuclear calcium signalling in the regulation of brain function. Nat. Rev. Neurosci. 2013, 14, 593–608. [Google Scholar] [CrossRef] [PubMed]
- Parra-Damas, A.; Saura, C.A. Synapse-to-nucleus signaling in neurodegenerative and neuropsychiatric disorders. Biol. Psychiatry 2019, 86, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Di Liegro, C.M.; Schiera, G.; Di Liegro, I. Regulation of mRNA transport, localization and translation in the nervous system of mammals. Int. J. Mol. Med. 2014, 33, 747–762. [Google Scholar] [CrossRef] [PubMed]
- Marcello, E.; Di Luca, M.; Gardoni, F. Synapse-to-nucleus communication: From developmental disorders to Alzheimer’s disease. Curr. Opin. Neurobiol. 2018, 48, 160–166. [Google Scholar] [CrossRef]
- El-Sayes, J.; Harasym, D.; Turco, C.V.; Locke, M.B.; Nelson, A.J. Exercise-induced neuroplasticity: A mechanistic model and prospects for promoting plasticity. Neurosci. 2019, 25, 65–85. [Google Scholar] [CrossRef]
- Zatorre, R.J.; Fields, R.D.; Johansen-Berg, H. Plasticity in gray and white: Neuroimaging changes in brain structure during learning. Nat. Neurosci. 2012, 15, 528–536. [Google Scholar] [CrossRef]
- Wang, H.-X.; Xu, W.; Pei, J.-J. Leisure activities, cognition and dementia. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2012, 1822, 482–491. [Google Scholar] [CrossRef]
- Draganski, B.; Gaser, C.; Busch, V.; Schuierer, G.; Bogdahn, U.; May, A. Changes in grey matter induced by training. Nature 2004, 427, 311–312. [Google Scholar] [CrossRef]
- Tang, Y.Y.; Lu, Q.; Fan, M.; Yang, Y.; Posner, M.I. Mechanisms of white matter changes induced by meditation. Proc. Natl. Acad. Sci. USA 2012, 109, 10570–10574. [Google Scholar] [CrossRef]
- Hänggi, J.; Koeneke, S.; Bezzola, L.; Jäncke, L. Structural neuroplasticity in the sensorimotor network of professional female ballet dancers. Hum. Brain Mapp. 2010, 31, 1196–1206. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.; Lu, M.; Song, Z.; Wang, J. Long-term intensive training induced brain structural changes in world class gymnasts. Brain Struct. Funct. 2015, 220, 625–644. [Google Scholar] [CrossRef]
- Chaddock, L.; Voss, M.W.; Kramer, A.F. Physical activity and fitness effects on cognition and brain health in children and older adults. Kinesiol. Rev. 2012, 1, 37–45. [Google Scholar] [CrossRef]
- Kramer, A.F.; Colcombe, S. Fitness effects on the cognitive function of older adults: A meta-analytic study—Revisited. Perspect. Psychol. Sci. 2018, 13, 213–217. [Google Scholar] [CrossRef]
- Colcombe, S.J.; Erickson, K.I.; Scalf, P.E.; Kim, J.S.; Prakash, R.; McAuley, E.; Elavsky, S.; Marquez, D.X.; Hu, L.; Kramer, A.F. Aerobic exercise training increases brain volume in aging humans. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2006, 61, 1166–1170. [Google Scholar] [CrossRef]
- Chaddock-Heyman, L.; Erickson, K.I.; Kienzler, C.; Drollette, E.S.; Raine, L.B.; Kao, S.C.; Bensken, J.; Weisshappel, R.; Castelli, D.M.; Hillman, C.H.; et al. Physical activity increases white matter microstructure in children. Front. Neurosci. 2018, 12, 950. [Google Scholar] [CrossRef] [PubMed]
- Colcombe, S.J.; Kramer, A.F.; Erickson, K.I.; Scalf, P.; McAuley, E.; Cohen, N.J.; Webb, A.; Jerome, G.J.; Marquez, D.X.; Elavsky, S. Cardiovascular fitness, cortical plasticity, and aging. Proc. Natl. Acad. Sci. USA 2004, 101, 3316–3321. [Google Scholar] [CrossRef] [PubMed]
- Knaepen, K.; Goekint, M.; Heyman, E.M.; Meeusen, R. Neuroplasticity—Exercise-induced response of peripheral brain-derived neurotrophic factor. Sports Med. 2010, 40, 765–801. [Google Scholar] [CrossRef] [PubMed]
- de Assis, G.G.; Almondes, K.M.D. Exercise-dependent BDNF as a modulatory factor for the executive processing of individuals in course of cognitive decline. A systematic review. Front. Psychol. 2017, 8, 584. [Google Scholar] [CrossRef]
- Kelly, M.E.; Duff, H.; Kelly, S.; McHugh Power, J.E.; Brennan, S.; Lawlor, B.A.; Loughrey, D.G. The impact of social activities, social networks, social support and social relationships on the cognitive functioning of healthy older adults: A systematic review. Syst. Rev. 2017, 6, 259. [Google Scholar] [CrossRef]
- Bennett, D.A.; Schneider, J.A.; Tang, Y.; Arnold, S.E.; Wilson, R.S. The effect of social networks on the relation between Alzheimer’s disease pathology and level of cognitive function in old people: A longitudinal cohort study. Lancet Neurol. 2006, 5, 406–412. [Google Scholar] [CrossRef]
- Fratiglioni, L.; Paillard-Borg, S.; Winblad, B. An active and socially integrated lifestyle in late life might protect against dementia. Lancet Neurol. 2004, 3, 343–353. [Google Scholar] [CrossRef]
- Barnes, L.L.; Mendes de Leon, C.F.; Wilson, R.S.; Bienias, J.L.; Evans, D.A. Social resources and cognitive decline in a population of older African Americans and whites. Neurology 2004, 63, 2322–2326. [Google Scholar] [CrossRef]
- Shankar, A.; Hamer, M.; McMunn, A.; Steptoe, A. Social Isolation and Loneliness: Relationships With Cognitive Function During 4 Years of Follow-up in the English Longitudinal Study of Ageing. Psychosom. Med. 2013, 75, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Zunzunegui, M.-V.; Alvarado, B.E.; Del Ser, T.; Otero, A. Social Networks, Social Integration, and Social Engagement Determine Cognitive Decline in Community-Dwelling Spanish Older Adults. J. Gerontol. Ser. B Psychol. Sci. Soc. Sci. 2003, 58, S93–S100. [Google Scholar] [CrossRef]
- Aartsen, M.J.; Smits, C.H.M.; van Tilburg, T.; Knipscheer, K.C.P.M.; Deeg, D.J.H. Activity in Older Adults: Cause or Consequence of Cognitive Functioning? A Longitudinal Study on Everyday Activities and Cognitive Performance in Older Adults. J. Gerontol. Ser. B Psychol. Sci. Soc. Sci. 2002, 57, P153–P162. [Google Scholar] [CrossRef]
- Richards, M.; Hardy, R.; Wadsworth, M.E.J. Does active leisure protect cognition? Evidence from a national birth cohort. Soc. Sci. Med. 2003, 56, 785–792. [Google Scholar] [CrossRef]
- Seeman, T.E.; Lusignolo, T.M.; Albert, M.; Berkman, L. Social relationships, social support, and patterns of cognitive aging in healthy, high-functioning older adults: MacArthur Studies of Successful Aging. Health Psychol. 2001, 20, 243–255. [Google Scholar] [CrossRef]
- Bickart, K.C.; Wright, C.I.; Dautoff, R.J.; Dickerson, B.C.; Barrett, L.F. Amygdala volume and social network size in humans. Nat. Neurosci. 2011, 14, 163–164. [Google Scholar] [CrossRef]
- Kwak, S.; Joo, W.; Youm, Y.; Chey, J. Social brain volume is associated with in-degree social network size among older adults. Proc. R. Soc. B Biol. Sci. 2018, 285, 20172708. [Google Scholar] [CrossRef] [Green Version]
- Yaffe, K.; Hoang, T. Nonpharmacologic Treatment and Prevention Strategies for Dementia. Contin. Lifelong Learn. Neurol. 2013, 19, 372–381. [Google Scholar] [CrossRef]
- Dodge, H.H.; Kadowaki, T.; Hayakawa, T.; Yamakawa, M.; Sekikawa, A.; Ueshima, H. Cognitive Impairment as a Strong Predictor of Incident Disability in Specific ADL-IADL Tasks Among Community-Dwelling Elders: The Azuchi Study. Gerontologist 2005, 45, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Mortimer, J.A.; Ding, D.; Borenstein, A.R.; DeCarli, C.; Guo, Q.; Wu, Y.; Zhao, Q.; Chu, S. Changes in Brain Volume and Cognition in a Randomized Trial of Exercise and Social Interaction in a Community-Based Sample of Non-Demented Chinese Elders. J. Alzheimer’s Dis. 2012, 30, 757–766. [Google Scholar] [CrossRef] [PubMed]
- Pitkala, K.H.; Routasalo, P.; Kautiainen, H.; Sintonen, H.; Tilvis, R.S. Effects of Socially Stimulating Group Intervention on Lonely, Older People’s Cognition: A Randomized, Controlled Trial. Am. J. Geriatr. Psychiatry 2011, 19, 654–663. [Google Scholar] [CrossRef] [PubMed]
- Toepoel, V. Ageing, Leisure, and Social Connectedness: How could Leisure Help Reduce Social Isolation of Older People? Soc. Indic. Res. 2013, 113, 355–372. [Google Scholar] [CrossRef]
- Lu, F.; Huo, Y.; Li, M.; Chen, H.; Liu, F.; Wang, Y.; Long, Z.; Duan, X.; Zhang, J.; Zeng, L.; et al. Relationship between personality and gray matter volume in healthy young adults: A voxel-based morphometric study. PLoS ONE 2014, 9, e88763. [Google Scholar] [CrossRef]
- Raine, A.; Lencz, T.; Bihrle, S.; LaCasse, L.; Colletti, P. Reduced prefrontal gray matter volume and reduced autonomic activity in antisocial personality disorder. Arch. Gen. Psychiatry 2000, 57, 119–127. [Google Scholar] [CrossRef]
- Haga, M.; Pedersen, A.V.; Sigmundsson, H. Interrelationship among selected measures of motor skills. Child Care Health Dev. 2008, 34, 245–248. [Google Scholar] [CrossRef]
- Neely, A.S.; Bäkman, L. Long-Term Maintenance of Gains From Memory Training in Older Adults: Two 3½Year Follow-up Studies. J. Gerontol. 1993, 48, P233–P237. [Google Scholar] [CrossRef]
- Doistua, J.; Lazcano, I.; Madariaga, A. Serious Leisure and Passion in University Programs for Seniors. Int. J. Environ. Res. Public Health 2022, 19, 3573. [Google Scholar] [CrossRef]
- Jachimowicz, J.M.; Wihler, A.; Bailey, E.R.; Galinsky, A.D. Why grit requires perseverance and passion to positively predict performance. Proc. Natl. Acad. Sci. USA 2018, 115, 9980–9985. [Google Scholar] [CrossRef] [PubMed]
- Sigmundsson, H.; Trana, L.; Polman, R.; Haga, M. What is trained develops! theoretical perspective on skill learning. Sports 2017, 5, 38. [Google Scholar] [CrossRef] [PubMed]
- Verner-Filion, J.; Vallerand, R.J.; Amiot, C.E.; Mocanu, I. The two roads from passion to sport performance and psychological well-being: The mediating role of need satisfaction, deliberate practice, and achievement goals. Psychol. Sport Exerc. 2017, 30, 19–29. [Google Scholar] [CrossRef]
- Curran, T.; Hill, A.P.; Appleton, P.R.; Vallerand, R.J.; Standage, M. The psychology of passion: A meta-analytical review of a decade of research on intrapersonal outcomes. Motiv. Emot. 2015, 39, 631–655. [Google Scholar] [CrossRef]
- Ehling, R.; Amprosi, M.; Kremmel, B.; Bsteh, G.; Eberharter, K.; Zehentner, M.; Steiger, R.; Tuovinen, N.; Gizewski, E.R.; Benke, T.; et al. Second language learning induces grey matter volume increase in people with multiple sclerosis. PLoS ONE 2019, 14, e0226525. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Vallerand, R.J.; Padilla, A.M. On the Role of Passion in Second Language Learning and Flourishing. J. Happiness Stud. 2021, 22, 2761–2779. [Google Scholar] [CrossRef]
- Vallerand, R.J.; Salvy, S.J.; Mageau, G.A.; Elliot, A.J.; Denis, P.L.; Grouzet, F.M.; Blanchard, C. On the role of passion in performance. J. Personal. 2007, 75, 505–534. [Google Scholar] [CrossRef]
- Sigmundsson, H.; Haga, M.; Elnes, M.; Dybendal, B.H.; Hermundsdottir, F. Motivational Factors Are Varying across Age Groups and Gender. Int. J. Environ. Res. Public Health 2022, 19, 5207. [Google Scholar] [CrossRef] [PubMed]
- Sigmundsson, H. Passion, grit and mindset in the ages 14 to 77: Exploring relationship and gender differences. New Ideas Psychol. 2021, 60, 100815. [Google Scholar] [CrossRef]
- Sigmundsson, H.; Guðnason, S.; Jóhannsdóttir, S. Passion, grit and mindset: Exploring gender differences. New Ideas Psychol. 2021, 63, 100878. [Google Scholar] [CrossRef]
- Sigmundsson, H.; Dybendal, B.H.; Loftesnes, J.M.; Olafsson, B.; Grassini, S. Passion a key for success: Exploring motivational factors in football players. New Ideas Psychol. 2022, 65, 100932. [Google Scholar] [CrossRef]
- Wang, S.; Dai, J.; Li, J.; Wang, X.; Chen, T.; Yang, X.; He, M.; Gong, Q. Neuroanatomical correlates of grit: Growth mindset mediates the association between gray matter structure and trait grit in late adolescence. Hum. Brain Mapp. 2018, 39, 1688–1699. [Google Scholar] [CrossRef] [PubMed]
- Battaglia, S.; Fabius, J.H.; Moravkova, K.; Fracasso, A.; Borgomaneri, S. The neurobiological correlates of gaze perception in healthy individuals and neurologic patients. Biomedicines 2022, 10, 627. [Google Scholar] [CrossRef] [PubMed]
- Chen, C. Recent advances in the study of the comorbidity of depressive and anxiety disorders. Adv. Clin. Exp. Med. 2022, 31, 355–358. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Yang, F.; Zheng, J.; Wang, X.; Huang, Q. Aberrant Structure MRI in Parkinson’s Disease and Comorbidity with Depression Based on Multinomial Tensor Regression Analysis. J. Pers. Med. 2022, 12, 89. [Google Scholar] [CrossRef]
- Tanaka, M.; Spekker, E.; Szabó, Á.; Polyák, H.; Vécsei, L. Modelling the neurodevelopmental pathogenesis in neuropsychiatric disorders. Bioactive kynurenines and their analogues as neuroprotective agents—in celebration of 80th birthday of Professor Peter Riederer. J. Neural Transm. 2022, 129, 627–642. [Google Scholar] [CrossRef]
- Munro, C.A.; McCaul, M.E.; Wong, D.F.; Oswald, L.M.; Zhou, Y.; Brasic, J.; Kuwabara, H.; Kumar, A.; Alexander, M.; Ye, W.; et al. Sex differences in striatal dopamine release in healthy adults. Biol. Psychiatry 2006, 59, 966–974. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sigmundsson, H.; Dybendal, B.H.; Grassini, S. Motion, Relation, and Passion in Brain Physiological and Cognitive Aging. Brain Sci. 2022, 12, 1122. https://doi.org/10.3390/brainsci12091122
Sigmundsson H, Dybendal BH, Grassini S. Motion, Relation, and Passion in Brain Physiological and Cognitive Aging. Brain Sciences. 2022; 12(9):1122. https://doi.org/10.3390/brainsci12091122
Chicago/Turabian StyleSigmundsson, Hermundur, Benjamin H. Dybendal, and Simone Grassini. 2022. "Motion, Relation, and Passion in Brain Physiological and Cognitive Aging" Brain Sciences 12, no. 9: 1122. https://doi.org/10.3390/brainsci12091122
APA StyleSigmundsson, H., Dybendal, B. H., & Grassini, S. (2022). Motion, Relation, and Passion in Brain Physiological and Cognitive Aging. Brain Sciences, 12(9), 1122. https://doi.org/10.3390/brainsci12091122





