Potential Targets for Noninvasive Brain Stimulation on Depersonalization-Derealization Disorder
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.1.1. Participants and Study Design
2.1.2. Assessment
2.1.3. Inclusion and Exclusion Criteria
2.2. MRI Data Acquisition
2.3. FC Analysis
2.4. Sample Size Estimation
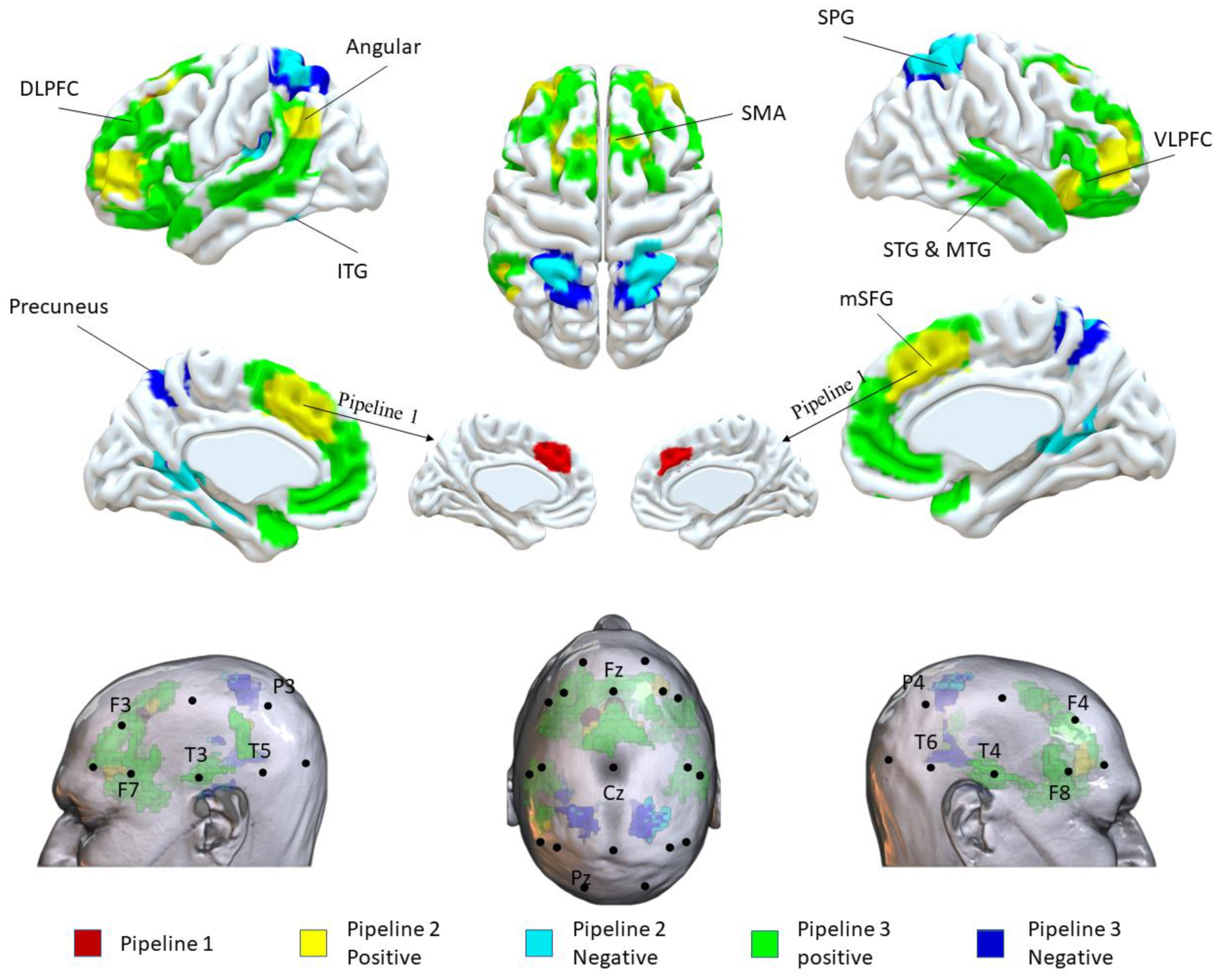
2.5. Identifying Potential Locations for NIBS
2.5.1. Pipeline 1 Identified Potential Targets from the Meta-Analysis
2.5.2. Pipeline 2 Identified Potential Targets from DPD-Network-Based FC Analysis
2.5.3. Pipeline 3 Identified Potential Targets from DPD-ROIs Based FC Analysis
2.5.4. Visualization
3. Results
3.1. Meta-Analysis Results
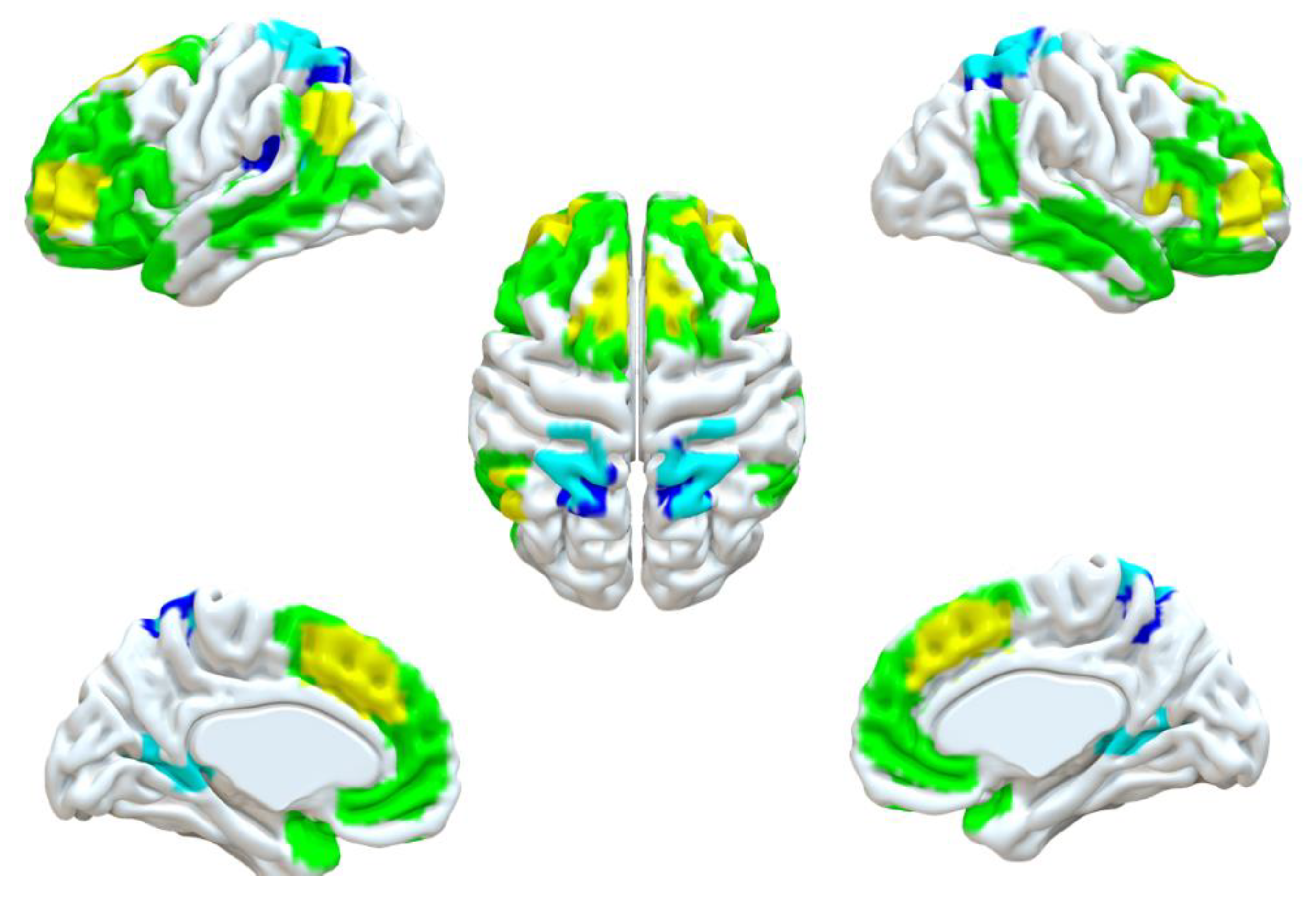
3.2. Potential Targets Identified from Three Pipelines
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-5, 5th ed.; American Psychiatric Association, Ed.; American Psychiatric Association: Washington, DC, USA, 2013; ISBN 978-0-89042-554-1. [Google Scholar]
- Hürlimann, F.; Kupferschmid, S.; Simon, A.E. Cannabis-Induced Depersonalization Disorder in Adolescence. Neuropsychobiology 2012, 65, 141–146. [Google Scholar] [CrossRef]
- Michal, M.; Adler, J.; Wiltink, J.; Reiner, I.; Tschan, R.; Wölfling, K.; Weimert, S.; Tuin, I.; Subic-Wrana, C.; Beutel, M.E.; et al. A Case Series of 223 Patients with Depersonalization-Derealization Syndrome. BMC Psychiatry 2016, 16, 203. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Simeon, D.; Guralnik, O.; Schmeidler, J.; Knutelska, M. Fluoxetine Therapy in Depersonalisation Disorder: Randomised Controlled Trial. Br. J. Psychiatry 2004, 185, 31–36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, W.E.; Kwok, C.H.T.; Hunter, E.C.M.; Richards, M.; David, A.S. Prevalence and Childhood Antecedents of Depersonalization Syndrome in a UK Birth Cohort. Soc. Psychiatry Psychiatr. Epidemiol. 2012, 47, 253–261. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aderibigbe, Y.A.; Bloch, R.M.; Walker, W.R. Prevalence of Depersonalization and Derealization Experiences in a Rural Population. Soc. Psychiatry Psychiatr. Epidemiol. 2001, 36, 63–69. [Google Scholar] [CrossRef]
- Guralnik, O.; Schmeidler, J.; Simeon, D. Feeling Unreal: Cognitive Processes in Depersonalization. Am. J. Psychiatry 2000, 157, 103–109. [Google Scholar] [CrossRef]
- Guralnik, O.; Giesbrecht, T.; Knutelska, M.; Sirroff, B.; Simeon, D. Cognitive Functioning in Depersonalization Disorder. J. Nerv. Ment. Dis. 2007, 195, 983–988. [Google Scholar] [CrossRef]
- Michal, M.; Beutel, M.E.; Grobe, T.G. How Often Is the Depersonalization-Derealization Disorder (ICD-10: F48. 1) Diagnosed in the Outpatient Health-Care Service? Z. Fur Psychosom. Med. Und Psychother. 2010, 56, 74–83. [Google Scholar] [CrossRef]
- Schlax, J.; Wiltink, J.; Beutel, M.E.; Münzel, T.; Pfeiffer, N.; Wild, P.; Blettner, M.; Ghaemi Kerahrodi, J.; Michal, M. Symptoms of Depersonalization/Derealization Are Independent Risk Factors for the Development or Persistence of Psychological Distress in the General Population: Results from the Gutenberg Health Study. J. Affect. Disord. 2020, 273, 41–47. [Google Scholar] [CrossRef]
- Orrù, G.; Bertelloni, D.; Cesari, V.; Conversano, C.; Gemignani, A. Targeting Temporal Parietal Junction for Assessing and Treating Disembodiment Phenomena: A Systematic Review of TMS Effect on Depersonalization and Derealization Disorders (DPD) and Body Illusions. AIMS Neurosci. 2021, 8, 181–194. [Google Scholar] [CrossRef]
- Hunter, E.C.M.; Baker, D.; Phillips, M.L.; Sierra, M.; David, A.S. Cognitive-Behaviour Therapy for Depersonalisation Disorder: An Open Study. Behav. Res. Ther. 2005, 43, 1121–1130. [Google Scholar] [CrossRef] [PubMed]
- Karris, B.C.; Capobianco, M.; Wei, X.; Ross, L. Treatment of Depersonalization Disorder with Repetitive Transcranial Magnetic Stimulation. J. Psychiatr. Pract. 2017, 23, 141–144. [Google Scholar] [CrossRef] [PubMed]
- Milev, R.V.; Giacobbe, P.; Kennedy, S.H.; Blumberger, D.M.; Daskalakis, Z.J.; Downar, J.; Modirrousta, M.; Patry, S.; Vila-Rodriguez, F.; Lam, R.W.; et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) 2016 Clinical Guidelines for the Management of Adults with Major Depressive Disorder. Can. J. Psychiatry 2016, 61, 561–575. [Google Scholar] [CrossRef] [PubMed]
- Osoegawa, C.; Gomes, J.S.; Grigolon, R.B.; Brietzke, E.; Gadelha, A.; Lacerda, A.L.T.; Dias, Á.M.; Cordeiro, Q.; Laranjeira, R.; de Jesus, D.; et al. Non-Invasive Brain Stimulation for Negative Symptoms in Schizophrenia: An Updated Systematic Review and Meta-Analysis. Schizophr. Res. 2018, 197, 34–44. [Google Scholar] [CrossRef] [PubMed]
- Rapinesi, C.; Kotzalidis, G.D.; Ferracuti, S.; Sani, G.; Girardi, P.; Del Casale, A. Brain Stimulation in Obsessive-Compulsive Disorder (OCD): A Systematic Review. Curr. Neuropharmacol 2019, 17, 787–807. [Google Scholar] [CrossRef]
- Zhang, B.; Liu, J.; Bao, T.; Wilson, G.; Park, J.; Zhao, B.; Kong, J. Locations for Noninvasive Brain Stimulation in Treating Depressive Disorders: A Combination of Meta-Analysis and Resting-State Functional Connectivity Analysis. Aust. N. Z. J. Psychiatry 2020, 54, 582–590. [Google Scholar] [CrossRef]
- Ning, Y.; Zheng, S.; Feng, S.; Zhang, B.; Jia, H. Potential Locations for Non-Invasive Brain Stimulation in Treating Schizophrenia: A Resting-State Functional Connectivity Analysis. Front. Neurol. 2021, 12, 2245. [Google Scholar] [CrossRef]
- Huang, Y.; Zhang, B.; Cao, J.; Yu, S.; Wilson, G.; Park, J.; Kong, J. Potential Locations for Noninvasive Brain Stimulation in Treating Autism Spectrum Disorders—A Functional Connectivity Study. Front. Psychiatry 2020, 11, 388. [Google Scholar] [CrossRef]
- Cao, J.; Huang, Y.; Meshberg, N.; Hodges, S.A.; Kong, J. Neuroimaging-Based Scalp Acupuncture Locations for Dementia. JCM 2020, 9, 2477. [Google Scholar] [CrossRef]
- Ross, C.A.; Duffy, C.M.M.; Ellason, J.W. Prevalence, Reliability and Validity of Dissociative Disorders in an Inpatient Setting. J. Trauma Dissociation 2002, 3, 7–17. [Google Scholar] [CrossRef]
- Sheehan, D.V.; Lecrubier, Y.; Sheehan, K.H.; Amorim, P.; Janavs, J.; Weiller, E.; Hergueta, T.; Baker, R.; Dunbar, G.C. The Mini-International Neuropsychiatric Interview (M.I.N.I.): The Development and Validation of a Structured Diagnostic Psychiatric Interview for DSM-IV and ICD-10. J. Clin. Psychiatry 1998, 59 (Suppl. 20), 22–33. [Google Scholar] [PubMed]
- Sierra, M.; Berrios, G.E. The Cambridge Depersonalization Scale: A New Instrument for the Measurement of Depersonalization. Psychiatry Res. 2000, 93, 153–164. [Google Scholar] [CrossRef]
- Wulf, L.; Palm, U.; Padberg, F. Combined Therapies–RTMS Meets CBT. Novel Approach for the Treatment of Derealization/Depersonalization Syndrome. Encephale 2019, 45, S82. [Google Scholar] [CrossRef]
- Rachid, F. Treatment of a Patient with Depersonalization Disorder with Low Frequency Repetitive Transcranial Magnetic Stimulation of the Right Temporo-Parietal Junction in a Private Practice Setting. J. Psychiatr. Pract. 2017, 23, 145–147. [Google Scholar] [CrossRef]
- Sierra, M.; Baker, D.; Medford, N.; David, A.S. Unpacking the Depersonalization Syndrome: An Exploratory Factor Analysis on the Cambridge Depersonalization Scale. Psychol. Med. 2005, 35, 1523–1532. [Google Scholar] [CrossRef]
- Simeon, D.; Kozin, D.S.; Segal, K.; Lerch, B.; Dujour, R.; Giesbrecht, T. De-Constructing Depersonalization: Further Evidence for Symptom Clusters. Psychiatry Res. 2008, 157, 303–306. [Google Scholar] [CrossRef]
- Blevins, C.A.; Witte, T.K.; Weathers, F.W. Factor Structure of the Cambridge Depersonalization Scale in Trauma-Exposed College Students. J. Trauma Dissociation 2013, 14, 288–301. [Google Scholar] [CrossRef]
- Aponte-Soto, M.R.; Vélez-Pastrana, M.; Martínez-Taboas, A.; González, R.A. Psychometric Properties of the Cambridge Depersonalization Scale in Puerto Rico. J. Trauma Dissociation 2014, 15, 348–363. [Google Scholar] [CrossRef]
- Yan, C.-G.; Wang, X.-D.; Zuo, X.-N.; Zang, Y.-F. DPABI: Data Processing & Analysis for (Resting-State) Brain Imaging. Neuroinform 2016, 14, 339–351. [Google Scholar] [CrossRef]
- Ashburner, J. SPM: A history. NeuroImage 2012, 62, 791–800. [Google Scholar] [CrossRef] [Green Version]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Eickhoff, S.B.; Bzdok, D.; Laird, A.R.; Kurth, F.; Fox, P.T. Activation Likelihood Estimation Meta-Analysis Revisited. NeuroImage 2012, 59, 2349–2361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Turkeltaub, P.E.; Eickhoff, S.B.; Laird, A.R.; Fox, M.; Wiener, M.; Fox, P. Minimizing Within-Experiment and within-Group Effects in Activation Likelihood Estimation Meta-Analyses. Hum. Brain Mapp. 2012, 33, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eickhoff, S.B.; Bzdok, D.; Laird, A.R.; Roski, C.; Caspers, S.; Zilles, K.; Fox, P.T. Co-Activation Patterns Distinguish Cortical Modules, Their Connectivity and Functional Differentiation. NeuroImage 2011, 57, 938–949. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eickhoff, S.B.; Laird, A.R.; Grefkes, C.; Wang, L.E.; Zilles, K.; Fox, P.T. Coordinate-Based Activation Likelihood Estimation Meta-Analysis of Neuroimaging Data: A Random-Effects Approach Based on Empirical Estimates of Spatial Uncertainty. Hum. Brain Mapp. 2009, 30, 2907–2926. [Google Scholar] [CrossRef] [Green Version]
- Ketay, S.; Hamilton, H.K.; Haas, B.W.; Simeon, D. Face Processing in Depersonalization: An FMRI Study of the Unfamiliar Self. Psychiat Res.-Neuroim. 2014, 222, 107–110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Medford, N.; Brierley, B.; Brammer, M.; Bullmore, E.T.; David, A.S.; Phillips, M.L. Emotional Memory in Depersonalization Disorder: A Functional MRI Study. Psychiatry Res. Neuroimaging 2006, 148, 93–102. [Google Scholar] [CrossRef]
- Medford, N.; Sierra, M.; Stringaris, A.; Giampietro, V.; Brammer, M.J.; David, A.S. Emotional Experience and Awareness of Self: Functional MRI Studies of Depersonalization Disorder. Front. Psychol. 2016, 7, 432. [Google Scholar] [CrossRef] [Green Version]
- Lieberman, M.D.; Straccia, M.A.; Meyer, M.L.; Du, M.; Tan, K.M. Social, Self, (Situational), and Affective Processes in Medial Prefrontal Cortex (MPFC): Causal, Multivariate, and Reverse Inference Evidence. Neurosci. Amp. Biobehav. Rev. 2019, 99, 311–328. [Google Scholar] [CrossRef]
- Amati, F.; Oh, H.; Kwan, V.S.Y.; Jordan, K.; Keenan, J.P. Overclaiming and the Medial Prefrontal Cortex: A Transcranial Magnetic Stimulation Study. Cogn. Neurosci. 2010, 1, 268–276. [Google Scholar] [CrossRef]
- De Pisapia, N.; Barchiesi, G.; Jovicich, J.; Cattaneo, L. The Role of Medial Prefrontal Cortex in Processing Emotional Self-Referential Information: A Combined TMS/FMRI Study. Brain Imaging Behav. 2019, 13, 603–614. [Google Scholar] [CrossRef] [PubMed]
- Goodkind, M.; Eickhoff, S.B.; Oathes, D.J.; Jiang, Y.; Chang, A.; Jones-Hagata, L.B.; Ortega, B.N.; Zaiko, Y.V.; Roach, E.L.; Korgaonkar, M.S.; et al. Identification of a Common Neurobiological Substrate for Mental Illness. JAMA Psychiatry 2015, 72, 305. [Google Scholar] [CrossRef] [PubMed]
- Marques, R.C.; Vieira, L.; Marques, D.; Cantilino, A. Transcranial Magnetic Stimulation of the Medial Prefrontal Cortex for Psychiatric Disorders: A Systematic Review. Braz. J. Psychiatry 2019, 41, 447–457. [Google Scholar] [CrossRef] [Green Version]
- Isserles, M.; Shalev, A.Y.; Roth, Y.; Peri, T.; Kutz, I.; Zlotnick, E.; Zangen, A. Effectiveness of Deep Transcranial Magnetic Stimulation Combined with a Brief Exposure Procedure in Post-Traumatic Stress Disorder--a Pilot Study. Brain Stimul. 2013, 6, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Ceccanti, M.; Inghilleri, M.; Attilia, M.L.; Raccah, R.; Fiore, M.; Zangen, A.; Ceccanti, M. Deep TMS on Alcoholics: Effects on Cortisolemia and Dopamine Pathway Modulation. A Pilot Study. Can. J. Physiol Pharm. 2015, 93, 283–290. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiménez-Genchi, A.M. Repetitive transcranial magnetic stimulation improves depersonalization: A case report. CNS Spectrums 2004, 9, 375–376. [Google Scholar] [CrossRef]
- Mantovani, A.; Simeon, D.; Urban, N.; Allart, A.; Bulow, P.M.; Lisanby, S.H. Temporo-parietal junction stimulation in the treatment of depersonalization disorder. Biol. Psychiatry 2010, 67, 45S. [Google Scholar] [CrossRef]
- Christopeit, M.; Simeon, D.; Mantovani, A. Effects of repetitive transcranial magnetic stimulation (rTMS) on specific symptom clusters in depersonalization disorder (DPD). Eur. Psychiatry 2013, 28, 1. [Google Scholar] [CrossRef]
- Jay, E.L.; Sierra, M.; Van Den Eynde, F.; Rothwell, J.C.; David, A.S. Testing a Neurobiological Model of Depersonalization Disorder Using Repetitive Transcranial Magnetic Stimulation. Brain Stimul. 2014, 7, 252–259. [Google Scholar] [CrossRef] [Green Version]
- Jay, E.L.; Nestler, S.; Sierra, M.; McClelland, J.; Kekic, M.; David, A.S. Ventrolateral Prefrontal Cortex Repetitive Transcranial Magnetic Stimulation in the Treatment of Depersonalization Disorder: A Consecutive Case Series. Psychiatry Res. 2016, 240, 118–122. [Google Scholar] [CrossRef] [Green Version]
- Koenigs, M.; Barbey, A.K.; Postle, B.R.; Grafman, J. Superior Parietal Cortex Is Critical for the Manipulation of Information in Working Memory. J. Neurosci. 2009, 29, 14980–14986. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ribeiro, J.A.; Marinho, F.V.C.; Rocha, K.; Magalhães, F.; Baptista, A.F.; Velasques, B.; Ribeiro, P.; Cagy, M.; Bastos, V.H.; Gupta, D.; et al. Low-Frequency RTMS in the Superior Parietal Cortex Affects the Working Memory in Horizontal Axis during the Spatial Task Performance. Neurol. Sci. 2018, 39, 527–532. [Google Scholar] [CrossRef]
- Caspari, N.; Arsenault, J.T.; Vandenberghe, R.; Vanduffel, W. Functional Similarity of Medial Superior Parietal Areas for Shift-Selective Attention Signals in Humans and Monkeys. Cereb. Cortex 2018, 28, 2085–2099. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Wang, J.; Zhang, Y.; Zheng, D.; Zhang, J.; Rong, M.; Wu, H.; Wang, Y.; Zhou, K.; Jiang, T. The Neuroanatomical Basis for Posterior Superior Parietal Lobule Control Lateralization of Visuospatial Attention. Front. Neuroanat. 2016, 10, 32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Simeon, D.; Knutelska, M.; Putnam, F.W.; Schmeidler, J.; Smith, L.M. Attention and Memory in Depersonalization-Spectrum Dissociative Disorders: Impact of Selective-Divided Attentional Condition, Stimulus Emotionality, and Stress. J. Trauma Dissociation 2022, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Gomez, L.J.; Dannhauer, M.; Peterchev, A.V. Fast Computational Optimization of TMS Coil Placement for Individualized Electric Field Targeting. NeuroImage 2021, 228, 117696. [Google Scholar] [CrossRef] [PubMed]
- Weise, K.; Numssen, O.; Thielscher, A.; Hartwigsen, G.; Knösche, T.R. A Novel Approach to Localize Cortical TMS Effects. NeuroImage 2020, 209, 116486. [Google Scholar] [CrossRef]
- Saturnino, G.B.; Siebner, H.R.; Thielscher, A.; Madsen, K.H. Accessibility of Cortical Regions to Focal TES: Dependence on Spatial Position, Safety, and Practical Constraints. NeuroImage 2019, 203, 116183. [Google Scholar] [CrossRef]
- Thielscher, A.; Antunes, A.; Saturnino, G.B. Field Modeling for Transcranial Magnetic Stimulation: A Useful Tool to Understand the Physiological Effects of TMS? In Proceedings of the 2015 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Milan, Italy, 25–29 August 2015; Volume 2015, pp. 222–225. [Google Scholar] [CrossRef]
| ID | x | y | z | ALE | P | Z | Label | BA |
|---|---|---|---|---|---|---|---|---|
| 1 | −1 | 16 | 35 | 9.25 × 10−3 | 1.93 × 10−5 | 4.12 | CG-L | 32 |
| 2 | −1 | 25 | 41 | 7.04 × 10−3 | 1.73 × 10−4 | 3.58 | mFG-L | 8 |
| 3 | −1 | 25 | 35 | 6.82 × 10−3 | 2.39 × 10−4 | 3.49 | mFG-L | 9 |
| 4 | −1 | 15 | 25 | 6.63 × 10−3 | 3.05 × 10−4 | 3.43 | AC-L | 33 |
| 5 | −18 | 19 | 38 | 6.33 × 10−3 | 4.41 × 10−4 | 3.33 | mFG-L | 6 |
| 6 | −1 | 30 | 22 | 4.97 × 10−3 | 2.78 × 10−3 | 2.77 | AC-L | 32 |
| Untreated (n = 52) | Treated (n = 24) | |
|---|---|---|
| Gender (Male/Female) | 38/14 | 17/7 |
| Age | 23.9 ± 5.2 | 23.5 ± 6.7 |
| Education | 13.6 ± 2.6 | 13.9 ± 3.1 |
| Duration (Years) | 5.5 ± 4.8 | 4.5 ± 4.7 |
| CDS total score | 162.6 ± 45.5 | 180.9 ± 58.1 |
| Numbing | 31.5 ± 14.2 | 35.7 ± 16.4 |
| Unreality of Self | 40.0 ± 15.4 | 40.1 ± 14.0 |
| Perceptual alterations | 20.1 ± 12.7 | 20.8 ± 10.9 |
| Unreality of surroundings | 14.1 ± 5.6 | 16.0 ± 5.0 |
| Temporal disintegration | 21.0 ± 9.6 | 23.3 ± 10.7 |
| Peak Coordinates | |||||||
|---|---|---|---|---|---|---|---|
| Cluster | Cluster Size | Peak Intensity | x | y | z | Brain Regions | 10–20 System Locations |
| Pipeline 1 | |||||||
| 1 | 572 | 0.0084 | 0 | 24 | 38 | mSFG | Fz |
| Pipeline 2 Positive | |||||||
| 1 | 1098 | 30.0245 | 0 | 24 | 38 | mSFG | Fz |
| 2 | 544 | 21.024 | −32 | 48 | 0 | SFG and MFG_L | F3 |
| 3 | 360 | 20.0082 | 34 | 48 | 8 | MFG_R | F4 |
| 4 | 73 | 16.9293 | −54 | −58 | 30 | Angular_L | P3 |
| 5 | 54 | 20.9628 | 36 | 22 | −12 | IFG_R | F8 |
| Pipeline 2 Negative | |||||||
| 1 | 316 | −9.75294 | 30 | −52 | 12 | Precuneus_R | Cz-Pz |
| 2 | 299 | −6.66981 | 36 | −48 | 62 | SPG_R | Superior to P4 |
| 3 | 142 | −8.70127 | −20 | −46 | 2 | Precuneus_L | Cz-Pz |
| 4 | 118 | −6.55238 | −38 | −40 | −16 | ITG_L | T3 |
| 5 | 58 | −4.51933 | −30 | −48 | 66 | SPG_L | Superior to P3 |
| 6 | 52 | −7.962 | −36 | −32 | 20 | STG_L | T3 |
| Pipeline 3 Positive | |||||||
| 1 | 1389 | 6 | - | - | - | IFG_L | F7 |
| 1 | 499 | 6 | - | - | - | mSFG_L | Fz |
| 1 | 371 | 6 | - | - | - | SFG_R | F4 |
| 1 | 359 | 6 | - | - | - | MFG_R | F4 |
| 2 | 1494 | 6 | - | - | - | IFG_R | F8 |
| 3 | 919 | 6 | - | - | - | STG_R | T4 |
| 4 | 640 | 6 | - | - | - | MTG_L | T3 |
| 5 | 502 | 6 | - | - | - | SMG | P3 |
| 6 | 780 | 6 | - | - | - | SFG_L | Fz |
| 6 | 697 | 6 | - | - | - | SMA_L | Fz-Cz |
| 6 | 321 | 6 | - | - | - | SMA_R | Fz-Cz |
| Pipeline 3 Negative | |||||||
| 1 | 683 | 5 | - | - | - | SPG_R | Superior to P4 |
| 2 | 505 | 5 | - | - | - | SPG_L | Superior to P3 |
| 3 | 300 | 6 | - | - | - | Precuneus_R | Cz-Pz |
| 4 | 130 | 6 | - | - | - | Precuneus_L | Cz-Pz |
| 5 | 93 | 6 | - | - | - | ITG_L | T5 |
| 6 | 52 | 6 | - | - | - | STG_L | Superior to T3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, S.; Song, N.; Wang, S.; Ning, Y.; Zhu, H.; Song, M.; Jia, Y.; Jia, H. Potential Targets for Noninvasive Brain Stimulation on Depersonalization-Derealization Disorder. Brain Sci. 2022, 12, 1112. https://doi.org/10.3390/brainsci12081112
Zheng S, Song N, Wang S, Ning Y, Zhu H, Song M, Jia Y, Jia H. Potential Targets for Noninvasive Brain Stimulation on Depersonalization-Derealization Disorder. Brain Sciences. 2022; 12(8):1112. https://doi.org/10.3390/brainsci12081112
Chicago/Turabian StyleZheng, Sisi, Nan Song, Sici Wang, Yanzhe Ning, Hong Zhu, Mingkang Song, Yuan Jia, and Hongxiao Jia. 2022. "Potential Targets for Noninvasive Brain Stimulation on Depersonalization-Derealization Disorder" Brain Sciences 12, no. 8: 1112. https://doi.org/10.3390/brainsci12081112
APA StyleZheng, S., Song, N., Wang, S., Ning, Y., Zhu, H., Song, M., Jia, Y., & Jia, H. (2022). Potential Targets for Noninvasive Brain Stimulation on Depersonalization-Derealization Disorder. Brain Sciences, 12(8), 1112. https://doi.org/10.3390/brainsci12081112






