Clinical Manifestations of Body Memories: The Impact of Past Bodily Experiences on Mental Health
Abstract
1. General Introduction
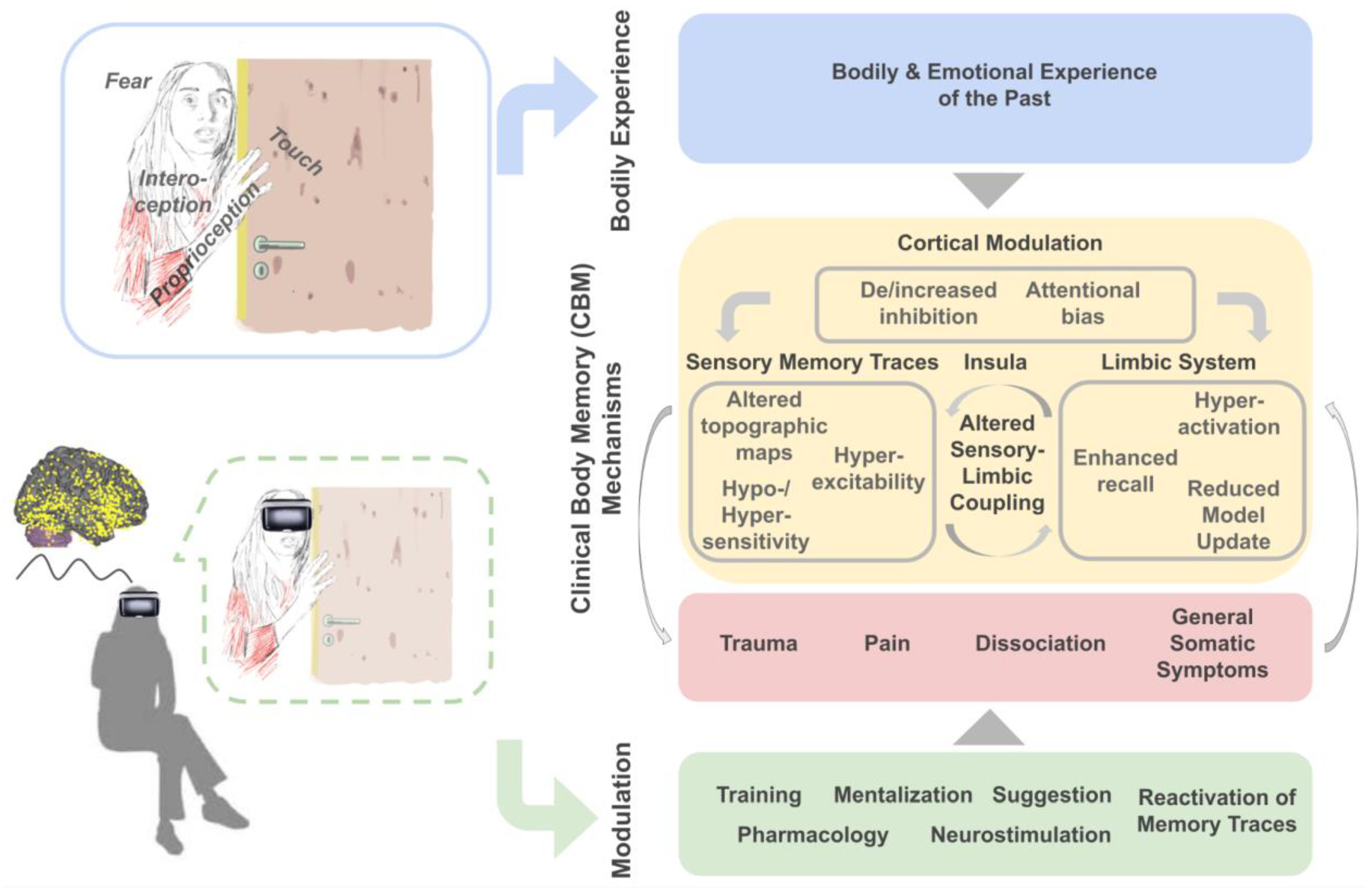
2. Clinical Body Memory (CBM) Manifestations
2.1. Trauma
2.2. Pain
2.3. Dissociation
2.4. General Somatic Symptoms
3. Clinical Body Memory (CBM) Mechanisms
3.1. Summary and Outlook
3.2. Possible Interventions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fuchs, T. The Phenomenology of Body Memory. In Body Memory, Metaphor and Movement; Koch, S.C., Fuchs, T., Summa, M., Müller, C., Eds.; John Benjamins Publishing: Amsterdam, The Netherlands, 2012; Volume 84, pp. 9–22. ISBN 978-90-272-8167-8. [Google Scholar]
- Freud, S. Die Traumdeutung; Nikol: Hamburg, Germany, 1900. [Google Scholar]
- Frankel, J.B. Ferenczi’s Trauma Theory. Am. J. Psychoanal. 1998, 58, 41–61. [Google Scholar] [CrossRef] [PubMed]
- Merleau-Ponty, M. Phenomenology of Perception; Routledge & Kegan Paul: London, UK, 1962. [Google Scholar]
- Riva, G. The Neuroscience of Body Memory: From the Self through the Space to the Others. Cortex 2018, 104, 241–260. [Google Scholar] [CrossRef] [PubMed]
- Berthier, M.; Starkstein, S.; Leiguarda, R. Asymbolia for Pain: A Sensory-Limbic Disconnection Syndrome. Ann. Neurol. 1988, 24, 41–49. [Google Scholar] [CrossRef]
- Geschwind, N. Disconnexion Syndromes in Animals and Man. Brain 1965, 88, 237–294. [Google Scholar] [CrossRef] [PubMed]
- Friedman, D.P.; Murray, E.A.; O’Neill, J.B.; Mishkin, M. Cortical Connections of the Somatosensory Fields of the Lateral Sulcus of Macaques: Evidence for a Corticolimbic Pathway for Touch. J. Comp. Neurol. 1986, 252, 323–347. [Google Scholar] [CrossRef] [PubMed]
- Buchanan, T.W.; Tranel, D.; Kirschbaum, C. Hippocampal Damage Abolishes the Cortisol Response to Psychosocial Stress in Humans. Horm. Behav. 2009, 56, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Bach, D.R.; Guitart-Masip, M.; Packard, P.A.; Miró, J.; Falip, M.; Fuentemilla, L.; Dolan, R.J. Human Hippocampus Arbitrates Approach-Avoidance Conflict. Curr. Biol. 2014, 24, 1435. [Google Scholar] [CrossRef] [PubMed]
- Clark, I.A.; Maguire, E.A. Remembering Preservation in Hippocampal Amnesia. Annu. Rev. Psychol. 2016, 67, 51–82. [Google Scholar] [CrossRef]
- Bauer, J. Das Gedächtnis des Körpers: Wie Beziehungen und Lebensstile Unsere Gene Steuern, 2nd ed.; Eichborn AG: Frankfurt am Main, Germany, 2013; ISBN 978-3-492-30185-5. [Google Scholar]
- Burgress, A.W.; Holmstrom, L.L. Rape Trauma Syndrome. Am. J. Psychiatry 1974, 131, 981–986. [Google Scholar] [CrossRef]
- Gaensbauer, T.J. Embodied Simulation, Mirror Neurons, and the Reenactment of Trauma in Early Childhood. Neuropsychoanalysis 2011, 13, 91–107. [Google Scholar] [CrossRef]
- Terr, L.C. What Becomes of Infantile Traumatic Memories?: An Adult “Wild Child” Is Asked to Remember. Psychoanal. Study Child 2013, 67, 197–214. [Google Scholar] [CrossRef] [PubMed]
- Lucas, M.V.; Anderson, L.C.; Bolling, D.Z.; Pelphrey, K.A.; Kaiser, M.D. Dissociating the Neural Correlates of Experiencing and Imagining Affective Touch. Cereb. Cortex 2015, 25, 2623–2630. [Google Scholar] [CrossRef] [PubMed]
- Morrison, I.; Björnsdotter, M.; Olausson, H. Vicarious Responses to Social Touch in Posterior Insular Cortex Are Tuned to Pleasant Caressing Speeds. J. Neurosci. 2011, 31, 9554–9562. [Google Scholar] [CrossRef] [PubMed]
- Bion, W.R. Learning from Experience; Heinemann Medical Books: London, UK, 1962; ISBN 978-0-429-91563-5. [Google Scholar]
- Leuzinger-Bohleber, M. Den Körper in der Seele Entdecken: Embodiment und Die Annäherung an das Nicht-Repräsentierte. Psyche 2014, 68, 922–950. [Google Scholar]
- Fonagy, P.; Gergely, G.; Jurist, E.L.; Target, M. Affect Regulation, Mentalization, and the Development of the Self, 1st ed.; Routledge: London, UK, 2002; ISBN 978-0-429-47164-3. [Google Scholar]
- Friston, K. The Free-Energy Principle: A Unified Brain Theory? Nat. Rev. Neurosci. 2010, 11, 127–138. [Google Scholar] [CrossRef]
- Friston, K.; Kiebel, S. Predictive Coding under the Free-Energy Principle. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 1211–1221. [Google Scholar] [CrossRef]
- Seth, A.K. Interoceptive Inference, Emotion, and the Embodied Self. Trends Cogn. Sci. 2013, 17, 565–573. [Google Scholar] [CrossRef]
- Friston, K.; FitzGerald, T.; Rigoli, F.; Schwartenbeck, P.; Doherty, J.O.; Pezzulo, G. Active Inference and Learning. Neurosci. Biobehav. Rev. 2016, 68, 862–879. [Google Scholar] [CrossRef]
- Pezzulo, G.; Rigoli, F.; Friston, K. Active Inference, Homeostatic Regulation and Adaptive Behavioural Control. Prog. Neurobiol. 2015, 134, 17–35. [Google Scholar] [CrossRef]
- Fotopoulou, A.; Tsakiris, M. Mentalizing Homeostasis: The Social Origins of Interoceptive Inference. Neuropsychoanalysis 2017, 19, 3–28. [Google Scholar] [CrossRef]
- Holmes, J.; Nolte, T. “Surprise” and the Bayesian Brain: Implications for Psychotherapy Theory and Practice. Front. Psychol. 2019, 10, 592. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, S.; Dodgson, G.; Meares, K. Predictive Processing and the Varieties of Psychological Trauma. Front. Psychol. 2017, 8, 1840. [Google Scholar] [CrossRef] [PubMed]
- Barron, H.C.; Auksztulewicz, R.; Friston, K. Prediction and Memory: A Predictive Coding Account. Prog. Neurobiol. 2020, 192, 101821. [Google Scholar] [CrossRef] [PubMed]
- Logue, M.W.; Van Rooij, S.J.H.; Dennis, E.L.; Davis, S.L.; Hayes, J.P.; Stevens, J.S.; Densmore, M.; Haswell, C.C.; Ipser, J.; Koch, S.B.J.; et al. Smaller Hippocampal Volume in Posttraumatic Stress Disorder: A Multisite ENIGMA-PGC Study: Subcortical Volumetry Results From Posttraumatic Stress Disorder Consortia. Biol. Psychiatry 2017, 83, 244–253. [Google Scholar] [CrossRef] [PubMed]
- O’Doherty, D.C.M.; Chitty, K.M.; Saddiqui, S.; Bennett, M.R.; Lagopoulos, J. A Systematic Review and Meta-Analysis of Magnetic Resonance Imaging Measurement of Structural Volumes in Posttraumatic Stress Disorder. Psychiatry Res. 2015, 232, 1–33. [Google Scholar] [CrossRef]
- Riem, M.M.E.; Alink, L.R.A.; Out, D.; Van Ijzendoorn, M.H.; Bakermans-Kranenburg, M.J. Beating the Brain about Abuse: Empirical and Meta-Analytic Studies of the Association between Maltreatment and Hippocampal Volume across Childhood and Adolescence. Dev. Psychopathol. 2015, 27, 507–520. [Google Scholar] [CrossRef]
- Schmaal, L.; Veltman, D.J.; Van Erp, T.G.M.; Sämann, P.G.; Frodl, T.; Jahanshad, N.; Loehrer, E.; Tiemeier, H.; Hofman, A.; Niessen, W.J.; et al. Subcortical Brain Alterations in Major Depressive Disorder: Findings from the ENIGMA Major Depressive Disorder Working Group. Mol. Psychiatry 2015, 21, 806–812. [Google Scholar] [CrossRef]
- Hayes, J.P.; Hayes, S.; Miller, D.R.; Lafleche, G.; Logue, M.W.; Verfaellie, M. Automated Measurement of Hippocampal Subfields in PTSD: Evidence for Smaller Dentate Gyrus Volume. J. Psychiatr. Res. 2017, 95, 247–252. [Google Scholar] [CrossRef]
- Teicher, M.H.; Anderson, C.M.; Polcari, A. Childhood Maltreatment Is Associated with Reduced Volume in the Hippocampal Subfields CA3, Dentate Gyrus, and Subiculum. Proc. Natl. Acad. Sci. USA 2012, 109, E563–E572. [Google Scholar] [CrossRef]
- Stevens, J.S.; Reddy, R.; Kim, Y.J.; Van Rooij, S.J.H.; Ely, T.D.; Hamann, S.; Ressler, K.J.; Jovanovic, T. Episodic Memory after Trauma Exposure: Medial Temporal Lobe Function Is Positively Related to Re-Experiencing and Inversely Related to Negative Affect Symptoms. Neuroimage Clin. 2017, 17, 650–658. [Google Scholar] [CrossRef]
- Bartolomei, F.; Lagarde, S.; Médina Villalon, S.; McGonigal, A.; Benar, C.G. The “Proust Phenomenon”: Odor-Evoked Autobiographical Memories Triggered by Direct Amygdala Stimulation in Human. Cortex 2017, 90, 173–175. [Google Scholar] [CrossRef] [PubMed]
- Brewin, C.R.; Gregory, J.D.; Lipton, M.; Burgess, N. Intrusive Images in Psychological Disorders: Characteristics, Neural Mechanisms, and Treatment Implications. Psychol. Rev. 2010, 117, 210–232. [Google Scholar] [CrossRef] [PubMed]
- Bisby, J.A.; Horner, A.J.; Hørlyck, L.D.; Burgess, N. Opposing Effects of Negative Emotion on Amygdalar and Hippocampal Memory for Items and Associations. Soc. Cogn. Affect. Neurosci. 2016, 11, 981–990. [Google Scholar] [CrossRef] [PubMed]
- Browning, M.; Fletcher, P.; Sharpe, M. Can Neuroimaging Help Us to Understand and Classify Somatoform Disorders? A Systematic and Critical Review. Psychosom. Med. 2011, 73, 173–184. [Google Scholar] [CrossRef]
- Brandl, F.; Weise, B.; Mulej Bratec, S.; Jassim, N.; Hoffmann Ayala, D.; Bertram, T.; Ploner, M.; Sorg, C. Common and Specific Large-Scale Brain Changes in Major Depressive Disorder, Anxiety Disorders, and Chronic Pain: A Transdiagnostic Multimodal Meta-Analysis of Structural and Functional MRI Studies. Neuropsychopharmacology 2022, 2022, 1071–1080. [Google Scholar] [CrossRef]
- Flor, H. Painful Memories. Can We Train Chronic Pain Patients to “forget” Their Pain? EMBO Rep. 2002, 3, 288–291. [Google Scholar] [CrossRef]
- Flor, H.; Elbert, T.; Knecht, S.; Wienbruch, C.; Pantev, C.; Birbaumer, N.; Larbig, W.; Taub, E. Phantom-Limb Pain as a Perceptual Correlate of Cortical Reorganization Following Arm Amputation. Nature 1995, 375, 482–484. [Google Scholar] [CrossRef]
- Makin, T.R.; Scholz, J.; Filippini, N.; Henderson Slater, D.; Tracey, I.; Johansen-Berg, H. Phantom Pain Is Associated with Preserved Structure and Function in the Former Hand Area. Nat. Commun. 2013, 4, 1570. [Google Scholar] [CrossRef]
- Birbaumer, N.; Lutzenberger, W.; Montoya, P.; Larbig, W.; Unertl, K.; Töpfner, S.; Grodd, W.; Taub, E.; Flor, H. Effects of Regional Anesthesia on Phantom Limb Pain Are Mirrored in Changes in Cortical Reorganization. J. Neurosci. 1997, 17, 5503–5508. [Google Scholar] [CrossRef]
- Katz, J.; Melzack, R. Pain “memories” in Phantom Limbs: Review and Clinical Observations. Pain 1990, 43, 319–336. [Google Scholar] [CrossRef]
- Flor, H.; Braun, C.; Elbert, T.; Birbaumer, N. Extensive Reorganization of Primary Somatosensory Cortex in Chronic Back Pain Patients. Neurosci. Lett. 1997, 224, 5–8. [Google Scholar] [CrossRef]
- Makin, T.R.; Flor, H. Brain (Re)Organisation Following Amputation: Implications for Phantom Limb Pain. Neuroimage 2020, 218, 116943. [Google Scholar] [CrossRef] [PubMed]
- Diers, M.; Koeppe, C.; Yilmaz, P.; Thieme, K.; Markela-Lerenc, J.; Schiltenwolf, M.; Van Ackern, K.; Flor, H. Pain Ratings and Somatosensory Evoked Responses to Repetitive Intramuscular and Intracutaneous Stimulation in Fibromyalgia Syndrome. J. Clin. Neurophysiol. 2008, 25, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Den Boer, C.; Dries, L.; Terluin, B.; Van Der Wouden, J.C.; Blankenstein, A.H.; Van Wilgen, C.P.; Lucassen, P.; Van Der Horst, H.E. Central Sensitization in Chronic Pain and Medically Unexplained Symptom Research: A Systematic Review of Definitions, Operationalizations and Measurement Instruments. J. Psychosom. Res. 2019, 117, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Loggia, M.L.; Cahalan, C.M.; Harris, R.E.; Beissner, F.; Garcia, R.G.; Kim, H.; Wasan, A.D.; Edwards, R.R.; Napadow, V. The Somatosensory Link: S1 Functional Connectivity Is Altered by Sustained Pain and Associated with Clinical/Autonomic Dysfunction in Fibromyalgia. Arthritis Rheumatol. 2015, 67, 1395–1405. [Google Scholar] [CrossRef]
- Kikkert, S.; Mezue, M.; O’Shea, J.; Henderson Slater, D.; Johansen-Berg, H.; Tracey, I.; Makin, T.R. Neural Basis of Induced Phantom Limb Pain Relief. Ann. Neurol. 2019, 85, 59–73. [Google Scholar] [CrossRef]
- Mancini, F.; Wang, A.P.; Schira, M.M.; Isherwood, Z.J.; McAuley, J.H.; Iannetti, G.D.; Sereno, M.I.; Moseley, G.L.; Rae, C.D. Fine-Grained Mapping of Cortical Somatotopies in Chronic Complex Regional Pain Syndrome. J. Neurosci. 2019, 39, 9185–9196. [Google Scholar] [CrossRef]
- Hechler, T.; Endres, D.; Thorwart, A. Why Harmless Sensations Might Hurt in Individuals with Chronic Pain: About Heightened Prediction and Perception of Pain in the Mind. Front. Psychol. 2016, 7, 1638. [Google Scholar] [CrossRef]
- Makin, T.R. Phantom Limb Pain: Thinking Outside the (Mirror) Box. Brain 2021, 144, 1929–1932. [Google Scholar] [CrossRef]
- Nijenhuis, E.R.S. Somatoform Dissociation: Major Symptoms of Dissociative Disorders. J. Trauma Dissociation 2000, 1, 7–32. [Google Scholar] [CrossRef]
- Saxe, G.N.; Chinman, G.; Berkowitz, R.; Hall, K.; Lieberg, G.; Schwartz, J.; Van Der Kolk, B.A. Somatization in Patients with Dissociative Disorders. Am. J. Psychiatry 1994, 151, 1329–1334. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nijenhuis, E.R.; Spinhoven, P.; Van Dyck, R.; Van der Hart, O.; Vanderlinden, J. The Development and Psychometric Characteristics of the Somatoform Dissociation Questionnaire (SDQ-20). J. Nerv. Ment. Dis. 1996, 184, 688–694. [Google Scholar] [CrossRef] [PubMed]
- Brewin, C.R. Memory Processes in Post-Traumatic Stress Disorder. Int. Rev. Psychiatry 2001, 13, 159–163. [Google Scholar] [CrossRef]
- Van Der Kolk, B.A. The Body Keeps the Score: Memory and the Evolving Psychobiology of Posttraumatic Stress. Harv. Rev. Psychiatry 1994, 1, 253–265. [Google Scholar] [CrossRef] [PubMed]
- Van Der Hart, O.; Bolt, H.; Van Der Kolk, B.A. Memory Fragmentation in Dissociative Identity Disorder. J. Trauma Dissociation 2005, 6, 55–70. [Google Scholar] [CrossRef]
- Van Der Kolk, B.A.; Van Der Hart, O. Pierre Janet and the Breakdown of Adaptation in Psychological Trauma. Am. J. Psychiatry 1989, 146, 1530–1540. [Google Scholar] [CrossRef] [PubMed]
- Haven, T.J. “That Part of the Body Is Just Gone”: Understanding and Responding to Dissociation and Physical Health. J. Trauma Dissociation 2009, 10, 204–218. [Google Scholar] [CrossRef]
- Ramasubbu, R. Conversion Sensory Symptoms Associated with Parietal Lobe Infarct: Case Report, Diagnostic Issues and Brain Mechanisms. J. Psychiatry Neurosci. 2002, 27, 118–122. [Google Scholar]
- Kanaan, R.A.A.; Craig, T.K.J.; Wessely, S.C.; David, A.S. Imaging Repressed Memories in Motor Conversion Disorder. Psychosom. Med. 2007, 69, 202–205. [Google Scholar] [CrossRef]
- Koreki, A.; Garfkinel, S.N.; Mula, M.; Agrawal, N.; Cope, S.; Eilon, T.; Gould Van Praag, C.; Critchley, H.D.; Edwards, M.; Yogarajah, M. Trait and State Interoceptive Abnormalities Are Associated with Dissociation and Seizure Frequency in Patients with Functional Seizures. Epilepsia 2020, 61, 1156–1165. [Google Scholar] [CrossRef]
- Schäflein, E.; Sattel, H.C.; Pollatos, O.; Sack, M. Disconnected-Impaired Interoceptive Accuracy and Its Association With Self-Perception and Cardiac Vagal Tone in Patients With Dissociative Disorder. Front. Psychol. 2018, 9, 897. [Google Scholar] [CrossRef] [PubMed]
- Medford, N.; Sierra, M.; Stringaris, A.; Giampietro, V.; Brammer, M.J.; David, A.S. Emotional Experience and Awareness of Self: Functional MRI Studies of Depersonalization Disorder. Front. Psychol. 2016, 7, 432. [Google Scholar] [CrossRef] [PubMed]
- Seth, A.K.; Suzuki, K.; Critchley, H.D. An Interoceptive Predictive Coding Model of Conscious Presence. Front. Psychol. 2011, 2, 395. [Google Scholar] [CrossRef] [PubMed]
- Edwards, M.J.; Adams, R.A.; Brown, H.; Pareés, I.; Friston, K.J. A Bayesian Account of “Hysteria”. Brain 2012, 135, 3495–3512. [Google Scholar] [CrossRef] [PubMed]
- Chalavi, S.; Vissia, E.M.; Giesen, M.E.; Nijenhuis, E.R.S.; Draijer, N.; Cole, J.H.; Dazzan, P.; Pariante, C.M.; Madsen, S.K.; Rajagopalan, P.; et al. Abnormal Hippocampal Morphology in Dissociative Identity Disorder and Post-Traumatic Stress Disorder Correlates with Childhood Trauma and Dissociative Symptoms. Hum. Brain Mapp. 2015, 36, 1692–1704. [Google Scholar] [CrossRef]
- Vermetten, E.; Schmahl, C.; Lindner, S.; Loewenstein, R.J.; Bremner, J.D. Hippocampal and Amygdalar Volumes in Dissociative Identity Disorder. Am. J. Psychiatry 2006, 163, 630–636. [Google Scholar] [CrossRef]
- Blihar, D.; Delgado, E.; Buryak, M.; Gonzalez, M.; Waechter, R. A Systematic Review of the Neuroanatomy of Dissociative Identity Disorder. Eur. J. Trauma Dissociation 2020, 4, 100148. [Google Scholar] [CrossRef]
- Reinders, A.A.T.S.; Marquand, A.F.; Schlumpf, Y.R.; Chalavi, S.; Vissia, E.M.; Nijenhuis, E.R.S.; Dazzan, P.; Jäncke, L.; Veltman, D.J. Aiding the Diagnosis of Dissociative Identity Disorder: Pattern Recognition Study of Brain Biomarkers. Br. J. Psychiatry 2019, 215, 536–544. [Google Scholar] [CrossRef]
- Rodríguez, M.; Ceric, F.; Murgas, P.; Harland, B.; Torrealba, F.; Contreras, M. Interoceptive Insular Cortex Mediates Both Innate Fear and Contextual Threat Conditioning to Predator Odor. Front. Behav. Neurosci. 2020, 13, 283. [Google Scholar] [CrossRef]
- DeVille, D.C.; Kerr, K.L.; Avery, J.A.; Burrows, K.; Bodurka, J.; Feinstein, J.S.; Khalsa, S.S.; Paulus, M.P.; Simmons, W.K. The Neural Bases of Interoceptive Encoding and Recall in Healthy Adults and Adults With Depression. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 2018, 3, 546–554. [Google Scholar] [CrossRef]
- World Health Organization. The ICD-10 Classification of Mental and Behavioural Disorders: Clinical Descriptions and Diagnostic Guidelines; World Health Organization: Geneva, Switzerland, 1992. [Google Scholar]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th ed.; American Psychiatric Association: Washington, DC, USA, 1994. [Google Scholar]
- Van den Bergh, O.; Witthöft, M.; Petersen, S.; Brown, R.J. Symptoms and the Body: Taking the Inferential Leap. Neurosci. Biobehav. Rev. 2017, 74, 185–203. [Google Scholar] [CrossRef] [PubMed]
- Hilderink, P.H.; Collard, R.; Rosmalen, J.G.M.; Oude Voshaar, R.C. Prevalence of Somatoform Disorders and Medically Unexplained Symptoms in Old Age Populations in Comparison with Younger Age Groups: A Systematic Review. Ageing Res. Rev. 2013, 12, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Ebel, H.; Podoll, K. Komorbidität von Somatoformen Störungen Mit Anderen Psychischen Störungen; Rudolf, G., Henningsen, P., Eds.; Schattauer: Stuttgart, Germany, 1998; pp. 25–38. [Google Scholar]
- Götzmann, L.; Schnyder, U. Posttraumatic Stress Disorder (PTSD) after Heart Transplant: The Influence of Earlier Loss Experiences on Posttransplant Flashbacks. Am. J. Psychother. 2002, 56, 562–567. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bhandari, M.; Busse, J.W.; Hanson, B.P.; Leece, P.; Ayeni, O.R.; Schemitsch, E.H. Psychological Distress and Quality of Life after Orthopedic Trauma: An Observational Study. Can. J. Surg. 2008, 51, 15–22. [Google Scholar] [PubMed]
- Toyofuku, A. Psychosomatic Problems in Dentistry. Biopsychosoc. Med. 2016, 10, 14. [Google Scholar] [CrossRef]
- Chen, Y.; Wu, Y.; Mu, J.; Qiu, B.; Wang, K.; Tian, Y. Abnormal Fear Circuits Activities Correlated to Physical Symptoms in Somatic Anxiety Patients. J. Affect. Disord. 2020, 274, 54–58. [Google Scholar] [CrossRef]
- Wei, D.; Du, X.; Li, W.; Chen, Q.; Li, H.; Hao, X.; Zhang, L.; Hitchman, G.; Zhang, Q.; Qiu, J. Regional Gray Matter Volume and Anxiety-Related Traits Interact to Predict Somatic Complaints in a Non-Clinical Sample. Soc. Cogn. Affect. Neurosci. 2015, 10, 122–128. [Google Scholar] [CrossRef]
- Groen, R.N.; Van Gils, A.; Emerencia, A.C.; Bos, E.H.; Rosmalen, J.G.M. Exploring Temporal Relationships among Worrying, Anxiety, and Somatic Symptoms. J. Psychosom. Res. 2020, 146, 110293. [Google Scholar] [CrossRef]
- Liu, P.; Tu, H.; Zhang, A.; Yang, C.; Liu, Z.; Lei, L.; Wu, P.; Sun, N.; Zhang, K. Brain Functional Alterations in MDD Patients with Somatic Symptoms: A Resting-State FMRI Study. J. Affect. Disord. 2021, 295, 788–796. [Google Scholar] [CrossRef]
- Zhang, T.; Bai, T.; Xie, W.; Wei, Q.; Lv, H.; Wang, A.; Guan, J.; Tian, Y.; Wang, K. Abnormal Connectivity of Anterior-Insular Subdivisions and Relationship with Somatic Symptom in Depressive Patients. Brain Imaging Behav. 2021, 15, 1760–1768. [Google Scholar] [CrossRef]
- Katzer, A.; Oberfeld, D.; Hiller, W.; Gerlach, A.L.; Witthöft, M. Tactile Perceptual Processes and Their Relationship to Somatoform Disorders. J. Abnorm. Psychol. 2012, 121, 530–543. [Google Scholar] [CrossRef] [PubMed]
- Katzer, A.; Oberfeld, D.; Hiller, W.; Witthöft, M. Tactile Perceptual Processes and Their Relationship to Medically Unexplained Symptoms and Health Anxiety. J. Psychosom. Res. 2011, 71, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Bogaerts, K.; Millen, A.; Li, W.; De Peuter, S.; Van Diest, I.; Vlemincx, E.; Fannes, S.; Van den Bergh, O. High Symptom Reporters Are Less Interoceptively Accurate in a Symptom-Related Context. J. Psychosom. Res. 2008, 65, 417–424. [Google Scholar] [CrossRef] [PubMed]
- Bogaerts, K.; Van Eylen, L.; Li, W.; Bresseleers, J.; Van Diest, I.; De Peuter, S.; Stans, L.; Decramer, M.; Van den Bergh, O. Distorted Symptom Perception in Patients with Medically Unexplained Symptoms. J. Abnorm. Psychol. 2010, 119, 226–234. [Google Scholar] [CrossRef] [PubMed]
- Barsky, A.J.; Wyshak, G. Hypochondriasis and Somatosensory Amplification. Br. J. Psychiatry 1990, 157, 404–409. [Google Scholar] [CrossRef] [PubMed]
- Voss, J.L.; Bridge, D.J.; Cohen, N.J.; Walker, J.A. A Closer Look at the Hippocampus and Memory. Trends Cogn. Sci. 2017, 21, 577–588. [Google Scholar] [CrossRef] [PubMed]
- Quian Quiroga, R. Neuronal Codes for Visual Perception and Memory. Neuropsychologia 2016, 83, 227–241. [Google Scholar] [CrossRef][Green Version]
- Turk-Browne, N.B. The Hippocampus as a Visual Area Organized by Space and Time: A Spatiotemporal Similarity Hypothesis. Vis. Res. 2019, 165, 123–130. [Google Scholar] [CrossRef]
- Zimmermann, K.; Eschen, A. Brain Regions Involved in Subprocesses of Small-Space Episodic Object-Location Memory: A Systematic Review of Lesion and Functional Neuroimaging Studies. Memory 2017, 25, 487–519. [Google Scholar] [CrossRef]
- Postma, A.; Kessels, R.P.C.; Van Asselen, M. How the Brain Remembers and Forgets Where Things Are: The Neurocognition of Object-Location Memory. Neurosci. Biobehav. Rev. 2008, 32, 1339–1345. [Google Scholar] [CrossRef]
- Behrendt, R.-P. Hippocampus and Consciousness. Rev. Neurosci. 2013, 24, 239–266. [Google Scholar] [CrossRef] [PubMed]
- Kwok, S.C.; Xu, X.; Duan, W.; Wang, X.; Tang, Y.; Allé, M.C.; Berna, F. Autobiographical and Episodic Memory Deficits in Schizophrenia: A Narrative Review and Proposed Agenda for Research. Clin. Psychol. Rev. 2021, 83, 101956. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Kuhn, S.K.; Jobson, L.; Haque, S. A Review of Autobiographical Memory Studies on Patients with Schizophrenia Spectrum Disorders. BMC Psychiatry 2019, 19, 361. [Google Scholar] [CrossRef] [PubMed]
- Rolls, E.T. The Cingulate Cortex and Limbic Systems for Emotion, Action, and Memory. Brain Struct. Funct. 2019, 224, 3001–3018. [Google Scholar] [CrossRef] [PubMed]
- Schacter, D.L.; Benoit, R.G.; Szpunar, K.K. Episodic Future Thinking: Mechanisms and Functions. Curr. Opin. Behav. Sci. 2017, 17, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Van Schie, K.; Geraerts, E.; Anderson, M.C. Emotional and Non-Emotional Memories Are Suppressible under Direct Suppression Instructions. Cogn. Emot. 2013, 27, 1122–1131. [Google Scholar] [CrossRef] [PubMed]
- Wolbers, T.; Hegarty, M. What Determines Our Navigational Abilities? Trends Cogn. Sci. 2010, 14, 138–146. [Google Scholar] [CrossRef]
- Burgess, N.; Maguire, E.A.; O’Keefe, J. The Human Hippocampus and Spatial and Episodic Memory. Neuron 2002, 35, 625–641. [Google Scholar] [CrossRef]
- Smith, R.; Badcock, P.; Friston, K.J. Recent Advances in the Application of Predictive Coding and Active Inference Models within Clinical Neuroscience. Psychiatry Clin. Neurosci. 2021, 75, 3–13. [Google Scholar] [CrossRef]
- Koren, T.; Yifa, R.; Amer, M.; Krot, M.; Boshnak, N.; Ben-Shaanan, T.L.; Azulay-Debby, H.; Zalayat, I.; Avishai, E.; Hajjo, H.; et al. Insular Cortex Neurons Encode and Retrieve Specific Immune Responses. Cell 2021, 184, 5902–5915.e17. [Google Scholar] [CrossRef]
- Nagai, M.; Kishi, K.; Kato, S. Insular Cortex and Neuropsychiatric Disorders: A Review of Recent Literature. Eur. Psychiatry 2007, 22, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Namkung, H.; Kim, S.-H.; Sawa, A. The Insula: An Underestimated Brain Area in Clinical Neuroscience, Psychiatry, and Neurology. Trends Neurosci. 2017, 40, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Bierbrauer, A.; Fellner, M.-C.; Heinen, R.; Wolf, O.T.; Axmacher, N. The Memory Trace of a Stressful Episode. Curr. Biol. 2021, 31, 5204–5213.e8. [Google Scholar] [CrossRef] [PubMed]
- Behrens, T.E.J.; Muller, T.H.; Whittington, J.C.R.; Mark, S.; Baram, A.B.; Stachenfeld, K.L.; Kurth-Nelson, Z. What Is a Cognitive Map? Organizing Knowledge for Flexible Behavior. Neuron 2018, 100, 490–509. [Google Scholar] [CrossRef]
- Bellmund, J.L.S.; Gärdenfors, P.; Moser, E.I.; Doeller, C.F. Navigating Cognition: Spatial Codes for Human Thinking. Science 2018, 362, eaat6766. [Google Scholar] [CrossRef]
- Schiller, D.; Eichenbaum, H.; Buffalo, E.A.; Davachi, L.; Foster, D.J.; Leutgeb, S.; Ranganath, C. Memory and Space: Towards an Understanding of the Cognitive Map. J. Neurosci. 2015, 35, 13904–13911. [Google Scholar] [CrossRef]
- Tavares, R.M.; Mendelsohn, A.; Grossman, Y.; Williams, C.H.; Shapiro, M.; Trope, Y.; Schiller, D. A Map for Social Navigation in the Human Brain. Neuron 2015, 87, 231–243. [Google Scholar] [CrossRef]
- Oltmer, J.; Wolbers, T.; Kuehn, E. Impaired Remapping of Social Relationships in Older Adults. Sci. Rep. 2021, 11, 21910. [Google Scholar] [CrossRef]
- Mishkin, M. Analogous Neural Models for Tactual and Visual Learning. Neuropsychologia 1979, 17, 139–151. [Google Scholar] [CrossRef]
- Flor, H.; Denke, C.; Schaefer, M.; Grüsser, S. Effect of Sensory Discrimination Training on Cortical Reorganisation and Phantom Limb Pain. Lancet 2001, 357, 1763–1764. [Google Scholar] [CrossRef]
- Sumner, J.A.; Griffith, J.W.; Mineka, S. Overgeneral Autobiographical Memory as a Predictor of the Course of Depression: A Meta-Analysis. Behav. Res. Ther. 2010, 48, 614–625. [Google Scholar] [CrossRef] [PubMed]
- Erten, M.N.; Brown, A.D. Memory Specificity Training for Depression and Posttraumatic Stress Disorder: A Promising Therapeutic Intervention. Front. Psychol. 2018, 9, 419. [Google Scholar] [CrossRef] [PubMed]
- Shelton, D.J.; Kirwan, C.B. A Possible Negative Influence of Depression on the Ability to Overcome Memory Interference. Behav. Brain Res. 2013, 256, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Koelen, J.A.; Houtveen, J.H.; Abbass, A.; Luyten, P.; Eurelings-Bontekoe, E.H.M.; Van Broeckhuysen-Kloth, S.A.M.; Bühring, M.E.F.; Geenen, R. Effectiveness of Psychotherapy for Severe Somatoform Disorder: Meta-Analysis. Br. J. Psychiatry 2014, 204, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Luyten, P.; Van Houdenhove, B.; Lemma, A.; Target, M.; Fonagy, P. A Mentalization-Based Approach to the Understanding and Treatment of Functional Somatic Disorders. Psychoanal. Psychother. 2012, 26, 121–140. [Google Scholar] [CrossRef]
- Ballespí, S.; Vives, J.; Alonso, N.; Sharp, C.; Ramírez, M.S.; Fonagy, P.; Barrantes-Vidal, N. To Know or Not to Know? Mentalization as Protection from Somatic Complaints. PLoS ONE 2019, 14, e0215308. [Google Scholar] [CrossRef]
- Myga, K.A.; Kuehn, E.; Azanon, E. Autosuggestion: A Cognitive Process That Empowers Your Brain? Exp. Brain Res. 2021, 240, 381–394. [Google Scholar] [CrossRef]
- Elsey, J.W.B.; Van Ast, V.A.; Kindt, M. Human Memory Reconsolidation: A Guiding Framework and Critical Review of the Evidence. Psychol. Bull. 2018, 144, 797–848. [Google Scholar] [CrossRef]
- Speer, M.E.; Ibrahim, S.; Schiller, D.; Delgado, M.R. Finding Positive Meaning in Memories of Negative Events Adaptively Updates Memory. Nat. Commun. 2021, 12, 6601. [Google Scholar] [CrossRef]
- Van Marle, H. PTSD as a Memory Disorder. Eur. J. Psychotraumatol. 2015, 6, 27633. [Google Scholar] [CrossRef]
- Van Dongen, E.V.; Takashima, A.; Barth, M.; Zapp, J.; Schad, L.R.; Paller, K.A.; Fernández, G. Memory Stabilization with Targeted Reactivation during Human Slow-Wave Sleep. Proc. Natl. Acad. Sci. USA 2012, 109, 10575–10580. [Google Scholar] [CrossRef] [PubMed]
- Beissner, F.; Preibisch, C.; Schweizer-Arau, A.; Popovici, R.M.; Meissner, K. Psychotherapy With Somatosensory Stimulation for Endometriosis-Associated Pain: The Role of the Anterior Hippocampus. Biological. Psychiatry 2017, 84, 734–742. [Google Scholar] [CrossRef] [PubMed]
- Pitman, R.K.; Sanders, K.M.; Zusman, R.M.; Healy, A.R.; Cheema, F.; Lasko, N.B.; Cahill, L.; Orr, S.P. Pilot Study of Secondary Prevention of Posttraumatic Stress Disorder with Propranolol. Biol. Psychiatry 2002, 51, 189–192. [Google Scholar] [CrossRef]
- Brunet, A.; Orr, S.P.; Tremblay, J.; Robertson, K.; Nader, K.; Pitman, R.K. Effect of Post-Retrieval Propranolol on Psychophysiologic Responding during Subsequent Script-Driven Traumatic Imagery in Post-Traumatic Stress Disorder. J. Psychiatr. Res. 2008, 4, 503–506. [Google Scholar] [CrossRef] [PubMed]
- Hoge, E.A.; Worthington, J.J.; Nagurney, J.T.; Chang, Y.; Kay, E.B.; Feterowski, C.M.; Katzman, A.R.; Goetz, J.M.; Rosasco, M.L.; Lasko, N.B.; et al. Effect of Acute Posttrauma Propranolol on PTSD Outcome and Physiological Responses During Script-Driven Imagery. CNS Neurosci. Ther. 2012, 18, 21–27. [Google Scholar] [CrossRef]
- Lefaucheur, J.-P.; Aleman, A.; Baeken, C.; Benninger, D.H.; Brunelin, J.; Di Lazzaro, V.; Filipović, S.R.; Grefkes, C.; Hasan, A.; Hummel, F.C.; et al. Evidence-Based Guidelines on the Therapeutic Use of Repetitive Transcranial Magnetic Stimulation (RTMS): An Update (2014–2018). Clin. Neurophysiol. 2020, 131, 474–528. [Google Scholar] [CrossRef]
- Woldemichael, B.T.; Bohacek, J.; Gapp, K.; Mansuy, I.M. Epigenetics of Memory and Plasticity. Prog. Mol. Biol. Transl. Sci. 2014, 122, 305–340. [Google Scholar] [CrossRef]
- Dudley, K.J.; Li, X.; Kobor, M.S.; Kippin, T.E.; Bredy, T.W. Epigenetic Mechanisms Mediating Vulnerability and Resilience to Psychiatric Disorders. Neurosci. Biobehav. Rev. 2011, 35, 1544–1551. [Google Scholar] [CrossRef]
- Kim, G.S.; Smith, A.K.; Nievergelt, C.M.; Uddin, M. Neuroepigenetics of Post-Traumatic Stress Disorder. Prog. Mol. Biol. Transl. Sci. 2018, 158, 227–253. [Google Scholar] [CrossRef]
- Pizzimenti, C.L.; Lattal, K.M. Epigenetics and Memory: Causes, Consequences and Treatments for Post-Traumatic Stress Disorder and Addiction. Genes. Brain Behav. 2015, 14, 73–84. [Google Scholar] [CrossRef]
- Jiménez, J.P.; Botto, A.; Herrera, L.; Leighton, C.; Rossi, J.L.; Quevedo, Y.; Silva, J.R.; Martínez, F.; Assar, R.; Salazar, L.A.; et al. Psychotherapy and Genetic Neuroscience: An Emerging Dialog. Front. Genet. 2018, 9, 257. [Google Scholar] [CrossRef] [PubMed]
- Riva, G.; Wiederhold, B.K.; Mantovani, F. Neuroscience of Virtual Reality: From Virtual Exposure to Embodied Medicine. Cyberpsychol. Behav. Soc. Netw. 2019, 22, 82–96. [Google Scholar] [CrossRef] [PubMed]
- Diersch, N.; Wolbers, T. The Potential of Virtual Reality for Spatial Navigation Research across the Adult Lifespan. J. Exp. Biol. 2019, 222, jeb187252. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, S.M.; Klotzsche, F.; Mariola, A.; Nikulin, V.; Villringer, A.; Gaebler, M. Decoding Subjective Emotional Arousal from EEG during an Immersive Virtual Reality Experience. Elife 2021, 10, e64812. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gentsch, A.; Kuehn, E. Clinical Manifestations of Body Memories: The Impact of Past Bodily Experiences on Mental Health. Brain Sci. 2022, 12, 594. https://doi.org/10.3390/brainsci12050594
Gentsch A, Kuehn E. Clinical Manifestations of Body Memories: The Impact of Past Bodily Experiences on Mental Health. Brain Sciences. 2022; 12(5):594. https://doi.org/10.3390/brainsci12050594
Chicago/Turabian StyleGentsch, Antje, and Esther Kuehn. 2022. "Clinical Manifestations of Body Memories: The Impact of Past Bodily Experiences on Mental Health" Brain Sciences 12, no. 5: 594. https://doi.org/10.3390/brainsci12050594
APA StyleGentsch, A., & Kuehn, E. (2022). Clinical Manifestations of Body Memories: The Impact of Past Bodily Experiences on Mental Health. Brain Sciences, 12(5), 594. https://doi.org/10.3390/brainsci12050594





