Characteristics of Movement Disorders in Patients with Autoimmune GFAP Astrocytopathy
Abstract
:1. Introduction
2. Patients and Methods
2.1. Patients
2.2. Measurement of CSF GFAP-IgG
2.3. Statistical Analysis
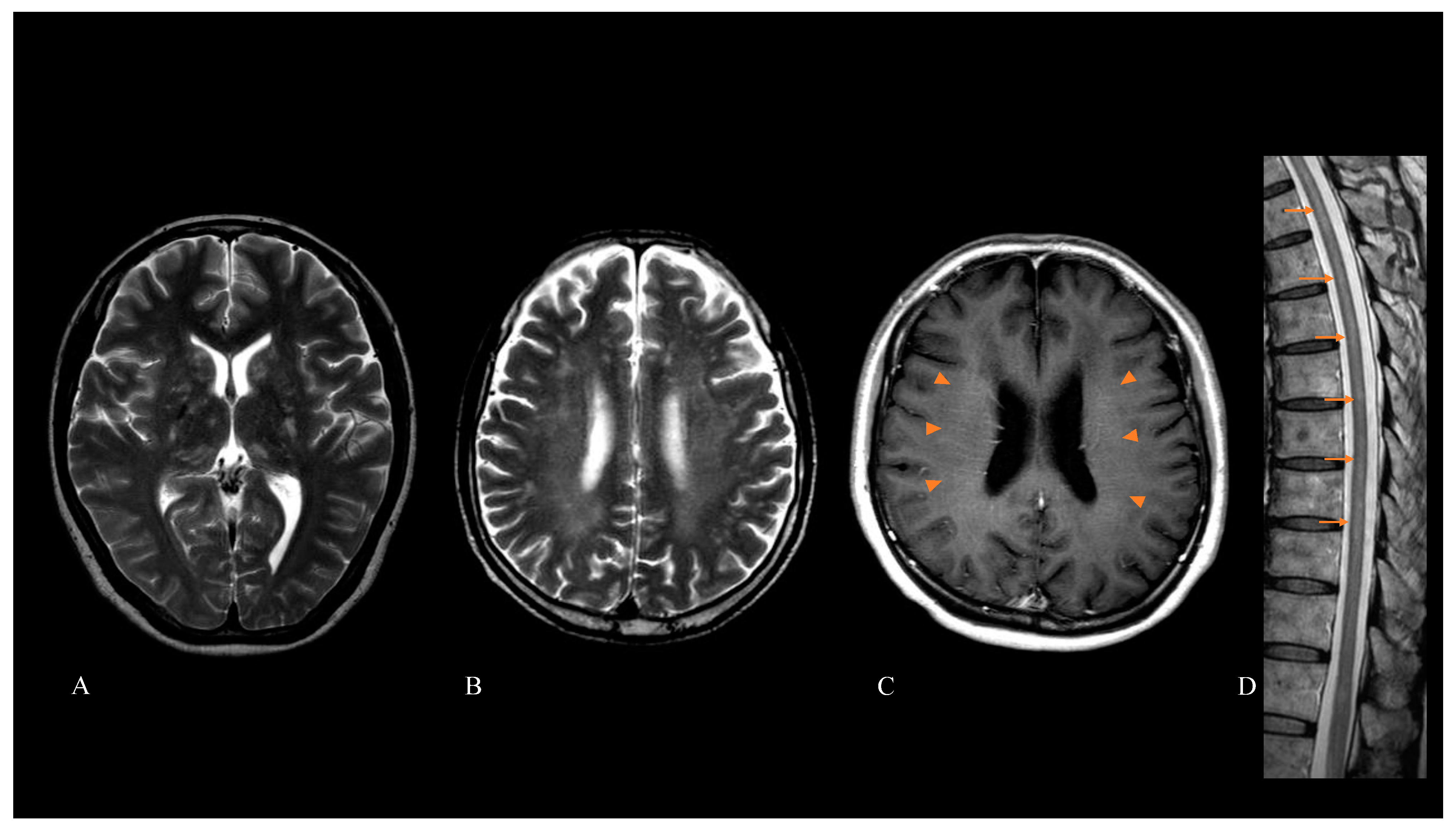
3. Results
3.1. Demographics, Clinical Features, CSF Characteristics, and Neuroimaging in Autoimmune GFAP-A Patients
3.2. Types and Frequencies of Movement Disorders in GFAP-A Patients
3.3. Characteristics of GFAP-A Patients with Movement Disorders
3.4. Literature Review of Movement Disorders in GFAP-A Patients
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fang, B.; McKeon, A.; Hinson, S.R.; Kryzer, T.J.; Pittock, S.J.; Aksamit, A.J.; Lennon, V.A. Autoimmune glial fibrillary acidic protein astrocytopathy: A novel meningoencephalomyelitis. JAMA Neurol. 2016, 73, 1297–1307. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, E.P.; Hinson, S.R.; Lennon, V.A.; Fang, B.; Aksamit, A.J.; Morris, P.P.; Basal, E.; Honorat, J.A.; Alfugham, N.B.; Linnoila, J.J.; et al. Glial fibrillary acidic protein immunoglobulin G as biomarker of autoimmune astrocytopathy: Analysis of 102 patients. Ann. Neurol. 2017, 81, 298–309. [Google Scholar] [CrossRef] [PubMed]
- Dubey, D.; Hinson, S.R.; Jolliffe, E.A.; Zekeridou, A.; Flanagan, E.P.; Pittock, S.J.; Basal, E.; Drubach, D.A.; Lachance, D.H.; Lennon, V.A.; et al. Autoimmune GFAP astrocytopathy: Prospective evaluation of 90 patients in 1 year. J. Neuroimmunol. 2018, 321, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Long, Y.; Liang, J.; Xu, H.; Huang, Q.; Yang, J.; Gao, C.; Qiu, W.; Lin, S.; Chen, X. Autoimmune glial fibrillary acidic protein astrocytopathy in Chinese patients: A retrospective study. Eur. J. Neurol. 2018, 25, 477–483. [Google Scholar] [CrossRef] [PubMed]
- Iorio, R.; Damato, V.; Evoli, A.; Gessi, M.; Gaudino, S.; Di Lazzaro, V.; Spagni, G.; Sluijs, J.A.; Hol, E.M. Clinical and immunological characteristics of the spectrum of GFAP autoimmunity: A case series of 22 patients. J. Neurol. Neurosurg. Psychiatry 2018, 89, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Dumonceau, A.G.; Ameli, R.; Rogemond, V.; Ruiz, A.; Joubert, B.; Muñiz-Castrillo, S.; Vogrig, A.; Picard, G.; Ambati, A.; Benaiteau, M.; et al. Glial fibrillary acidic protein autoimmunity: A French cohort study. Neurology 2022, 98, e653–e668. [Google Scholar] [CrossRef]
- Zarkali, A.; Cousins, O.; Athauda, D.; Moses, S.; Moran, N.; Harikrishnan, S. Glial fibrillary acidic protein antibody-positive meningoencephalomyelitis. Pract. Neurol. 2018, 18, 315–319. [Google Scholar] [CrossRef]
- Kimura, A.; Takekoshi, A.; Yoshikura, N.; Hayashi, Y.; Shimohata, T. Clinical characteristics of autoimmune GFAP astrocytopathy. J. Neuroimmunol. 2019, 332, 91–98. [Google Scholar] [CrossRef]
- Martin, A.L.; Jolliffe, E.; Hertweck, S.P. Ovarian teratoma associated with coexisting anti-N-methyl-D-aspartate receptor and glial fibrillary acidic protein autoimmune meningoencephalitis in an adolescent girl: A case report. J. Pediatr. Adolesc. Gynecol. 2018, 31, 321–324. [Google Scholar] [CrossRef]
- Shu, Y.; Long, Y.; Chang, Y.; Li, R.; Sun, X.; Wang, Y.; Huang, Y.; Li, J.; Chen, J.; Yang, Y.; et al. Brain immunohistopathology in a patient with autoimmune glial fibrillary acidic protein astrocytopathy. Neuroimmunomodulation 2018, 25, 1–6. [Google Scholar] [CrossRef]
- Yuan, Z.; Li, H.; Huang, L.; Fu, C.; Chen, Y.; Zhi, C.; Qiu, W.; Long, Y. CD8+ T-cell predominance in autoimmune glial fibrillary acidic protein astrocytopathy. Eur. J. Neurol. 2021, 28, 2121–2125. [Google Scholar] [CrossRef]
- Yang, X.; Liang, J.; Huang, Q.; Xu, H.; Gao, C.; Long, Y.; Xiao, X. Treatment of autoimmune glial fibrillary acidic protein astrocytopathy: Follow-up in 7 cases. Neuroimmunomodulation 2017, 24, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Kunchok, A.; Zekeridou, A.; McKeon, A. Autoimmune glial fibrillary acidic protein astrocytopathy. Curr. Opin. Neurol. 2019, 32, 452–458. [Google Scholar] [CrossRef]
- Tomczak, A.; Su, E.; Tugizova, M.; Carlson, A.M.; Kipp, L.B.; Feng, H.; Han, M.H. A case of GFAP-astroglial autoimmunity presenting with reversible Parkinsonism. Mult. Scler. Relat. Disord. 2019, 39, 101900. [Google Scholar] [CrossRef] [PubMed]
- Novo, A.C.; Venegas Pérez, B. Autoimmune glial fibrillary acidic protein astrocytopathy presented as ataxia, myoclonus and bulbar syndrome: A case report and review of the literature. BMJ Neurol. Open 2021, 3, e000142. [Google Scholar] [CrossRef]
- Asan, L.; Klebe, S.; Kleinschnitz, C.; Stettner, M.; Köhrmann, M. Anti-GFAP-antibody positive postinfectious acute cerebellar ataxia and myoclonus after COVID-19: A case report. Ther. Adv. Neurol. Disord. 2021, 14, 17562864211062824. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Xu, H.; Ding, M.; Huang, Q.; Chen, B.; Yang, H.; Liu, T.; Long, Y.; Gao, C. Overlapping autoimmune syndromes in patients with glial fibrillary acidic protein antibodies. Front. Neurol. 2018, 9, 251. [Google Scholar] [CrossRef]
- Theroux, L.M.; Goodkin, H.P.; Heinan, K.C.; Quigg, M.; Brenton, J.N. Extreme delta brush and distinctive imaging in a pediatric patient with autoimmune GFAP astrocytopathy. Mult. Scler. Relat. Disord. 2018, 26, 121–123. [Google Scholar] [CrossRef]
- Luessi, F.; Engel, S.; Spreer, A.; Bittner, S.; Zipp, F. GFAPα IgG-associated encephalitis upon daclizumab treatment of MS. Neurol. Neuroimmunol. Neuroinflamm. 2018, 5, e481. [Google Scholar] [CrossRef] [Green Version]
- Sechi, E.; Morris, P.P.; McKeon, A.; Pittock, S.J.; Hinson, S.R.; Weinshenker, B.G.; Aksamit, A.J.; Krecke, K.N.; Kaufmann, T.J.; Jolliffe, E.A.; et al. Glial fibrillary acidic protein IgG related myelitis: Characterisation and comparison with aquaporin-4-IgG myelitis. J. Neurol. Neurosurg. Psychiatry 2019, 90, 488–490. [Google Scholar] [CrossRef] [Green Version]
- Tokimura, R.; Matsuda, N.; Kobayashi, S.; Kimura, A.; Kanai, K. Abnormal evoked potentials in autoimmune glial fibrillary acidic protein astrocytopathy. eNeurologicalSci. 2020, 18, 100229. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Huang, Y.; Zeng, H.; Wang, M.; Guan, S.; Chen, G.; Guan, H. Overlapping clinical syndromes in patients with glial fibrillary acidic protein IgG. Neuroimmunomodulation 2020, 27, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Oger, V.; Bost, C.; Salah, L.; Yazbeck, E.; Maurey, H.; Bellesme, C.; Sevin, C.; Adamsbaum, C.; Chrétien, P.; Benaiteau, M.; et al. Mild encephalitis/encephalopathy with reversible splenial lesion syndrome: An unusual presentation of anti-GFAP astrocytopathy. Eur. J. Paediatr. Neurol. 2020, 26, 89–91. [Google Scholar] [CrossRef] [PubMed]
- Natori, T.; Shindo, K.; Okumura, A.; Kimura, A.; Takiyama, Y. A treatable case of autoimmune GFAP astrocytopathy presenting chronic progressive impairment. Neurol. Sci. 2020, 41, 2999–3002. [Google Scholar] [CrossRef]
- Rosales, J.J.; Toledano, C.; Riverol, M.; Arbizu, J. [18F]-FDG PET imaging in autoimmune GFAP meningoencephalomyelitis. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 947–948. [Google Scholar] [CrossRef]
- Xu, L.; Xian, W.; Li, J.; Yao, X.; Long, Y. Purkinje cell (PC) antibody positivity in a patient with autoimmune glial fibrillary acidic protein (GFAP) astrocytopathy. Int. J. Neurosci. 2020, 1–9. [Google Scholar] [CrossRef]
- Osakada, Y.; Omote, Y.; Ikegami, K.; Tadokoro, K.; Sato, K.; Hishikawa, N.; Takemoto, M.; Ohta, Y.; Yamashita, T.; Kimura, A.; et al. A case of autoimmune GFAP astrocytopathy with profound weight loss and increased uptake in the spinal cord on 18F-FDG PET. Neurol. Clin. Neurosci. 2021, 9, 95–97. [Google Scholar] [CrossRef]
- Wang, H.; Chin, J.H.; Fang, B.Y.; Chen, X.; Zhao, A.L.; Ren, H.T.; Guan, H.Z. Autoimmune glial fibrillary acidic protein astrocytopathy manifesting as subacute meningoencephalitis with descending myelitis: A case report. BMC Neurol. 2020, 20, 443. [Google Scholar] [CrossRef]
- Huang, H.; Bai, K.; Fu, Y.; Lu, S.; Ran, Y.; Dang, H.; Li, J. Glial fibrillary acidic protein astrocytopathy in pediatric patients: A retrospective study. Front. Pediatr. 2021, 8, 626564. [Google Scholar] [CrossRef]
- Nakamura, S.; Fujioka, T.; Kawashima, S.; Kawaguchi, T.; Mizuno, M.; Omura, M.; Okita, K.; Kimura, A.; Shimohata, T.; Matsukawa, N. Self-remitting elevation of adenosine deaminase levels in the cerebrospinal fluid with autoimmune glial fibrillary acidic protein astrocytopathy: A case report and review of the literature. Intern. Med. 2021, 60, 3031–3036. [Google Scholar] [CrossRef]
- Heide, E.S.; Chaudhari, A.; Pirverdian, A.; Lai, S.; Courtney, A. Failure of IVIG in steroid-responsive autoimmune glial fibrillary acidic protein astrocytopathy: A case report. Mult. Scler. Relat. Disord. 2021, 51, 102933. [Google Scholar] [CrossRef] [PubMed]
- Toledano-Illán, C.; Esparragosa Vázquez, I.; Zelaya Huerta, M.V.; Rosales Castillo, J.J.; Paternain Nuin, A.; Arbizu Lostao, J.; García de Eulate, M.R.; Riverol Fernández, M. Autoimmune glial fibrillary acidic protein astrocytopathy: Case report of a treatable cause of rapidly progressive dementia. J. Neurol. 2021, 268, 2256–2258. [Google Scholar] [CrossRef] [PubMed]
- Adachi, H.; Shiomi, Y.; Kimura, A.; Shimohata, T.; Yoneda, Y.; Kageyama, Y. A case of autoimmune glial fibrillary acidic protein (GFAP) astrocytopathy. Rinsho. Shinkeigaku 2021, 61, 401–404. [Google Scholar] [CrossRef] [PubMed]
- Tewkesbury, G.; Song, J.W.; Perrone, C.M. Magnetic resonance imaging of autoimmune GFAP astrocytopathy. Ann. Neurol. 2021, 90, 691–692. [Google Scholar] [CrossRef] [PubMed]
- Equiza, J.; Rodríguez-Antigüedad, J.; Campo-Caballero, D.; Iruzubieta, P.; Prada, Á.; Roncancio, A.; Fernández, E.; Ganzarain Oyarbide, M.; Arruti, M.; Urtasun, M.A.; et al. Autoimmune GFAP astrocytopathy presenting with remarkable CNS hyperexcitability and oculogyric crises. J. Neuroimmunol. 2021, 359, 577695. [Google Scholar] [CrossRef]
- Xiao, J.; Chen, X.; Shang, K.; Tang, Y.; Chen, M.; Deng, G.; Qin, C.; Tian, D.S. Clinical, neuroradiological, diagnostic and prognostic profile of autoimmune glial fibrillary acidic protein astrocytopathy: A pooled analysis of 324 cases from published data and a single-center retrospective study. J. Neuroimmunol. 2021, 360, 577718. [Google Scholar] [CrossRef]
- Yamakawa, M.; Hogan, K.O.; Leever, J.; Jassam, Y.N. Autopsy Case of Meningoencephalomyelitis Associated With Glial Fibrillary Acidic Protein Antibody. Neurol. Neuroimmunol. Neuroinflamm. 2021, 8, e1081. [Google Scholar] [CrossRef]
- Kimura, A.; Kato, S.; Takekoshi, A.; Yoshikura, N.; Yanagida, N.; Kitaguchi, H.; Akiyama, D.; Shimizu, H.; Kakita, A.; Shimohata, T. Autoimmune glial fibrillary acidic protein astrocytopathy resembling isolated central nervous system lymphomatoid granulomatosis. J. Neuroimmunol. 2021, 361, 577748. [Google Scholar] [CrossRef]
- Mirian, A.; Sharma, M.; Budhram, A. Linear radial brainstem enhancement in autoimmune glial fibrillary acidic protein astrocytopathy. JAMA Neurol. 2021, 79, 82–83. [Google Scholar] [CrossRef]
- Héraud, C.; Capet, N.; Levraut, M.; Hattenberger, R.; Bourg, V.; Thomas, P.; Mondot, L.; Lebrun-Frenay, C. Glial fibrillary acidic protein (GFAP) astrocytopathy presenting as mild encephalopathy with reversible splenium lesion. Neurol. Ther. 2021. Online ahead of print. [Google Scholar] [CrossRef]
- Fang, H.; Hu, W.; Jiang, Z.; Yang, H.; Liao, H.; Yang, L.; Wu, L. Autoimmune glial fibrillary acidic protein astrocytopathy in children: A retrospective analysis of 35 cases. Front. Immunol. 2021, 12, 761354. [Google Scholar] [CrossRef] [PubMed]
- Louis, E.D.; Faust, P.L.; Vonsattel, J.P.; Honig, L.S.; Rajput, A.; Robinson, C.A.; Rajput, A.; Pahwa, R.; Lyons, K.E.; Ross, G.W.; et al. Neuropathological changes in essential tremor: 33 cases compared with 21 controls. Brain 2007, 130, 3297–3307. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Louis, E.D. Essential tremor: Evolving clinicopathological concepts in an era of intensive post-mortem enquiry. Lancet Neurol. 2010, 9, 613–622. [Google Scholar] [CrossRef]
- Welton, T.; Cardoso, F.; Carr, J.A.; Chan, L.L.; Deuschl, G.; Jankovic, J.; Tan, E.K. Essential tremor. Nat. Rev. Dis. Primers 2021, 7, 83. [Google Scholar] [CrossRef]
- Panzer, J.; Dalmau, J. Movement disorders in paraneoplastic and autoimmune disease. Curr. Opin. Neurol. 2011, 24, 346–353. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dalmau, J.; Armangué, T.; Planagumà, J.; Radosevic, M.; Mannara, F.; Leypoldt, F.; Geis, C.; Lancaster, E.; Titulaer, M.J.; Rosenfeld, M.R.; et al. An update on anti-NMDA receptor encephalitis for neurologists and psychiatrists: Mechanisms and models. Lancet Neurol. 2019, 18, 1045–1057. [Google Scholar] [CrossRef]
| Variables | Patients (%) | Median (Range) |
|---|---|---|
| Demographics | ||
| Age (N = 87) | 51 (5–83) | |
| Male | 56/87 (64) | |
| Concomitant tumor | 14/87 (16) | |
| Concomitant autoimmune disease | 2/87 (2) | |
| Period from onset to admission (days) (N = 85) | 12 (1–1054) | |
| Clinical findings | ||
| Movement disorder | 74/87 (85) | |
| Urinary dysfunction | 67/87 (77) | |
| Altered consciousness | 66/87 (76) | |
| Nuchal rigidity and/or Kernig’s sign | 56/87 (64) | |
| Fever | 54/85 (64) | |
| Hyperreflexia | 50/86 (58) | |
| Headache | 43/85 (51) | |
| Cognitive dysfunction | 42/87 (48) | |
| Psychosis | 37/87 (43) | |
| Papillary edema | 17/48 (43) | |
| Weakness | 32/87 (37) | |
| Sensory disturbance | 24/87 (28) | |
| Respiratory failure | 19/86 (22) | |
| Convulsion | 17/87 (20) | |
| Cerebrospinal fluid findings | ||
| Cell counts (cells/μL) (N = 86) | 79 (4–472) | |
| Protein concentrations (mg/dL) (N = 86) | 146 (30–320) | |
| Oligoclonal IgG bands | 43/61 (70) | |
| Coexisting neural autoantibodies | ||
| Anti-MOG antibodies | 2/57 (4) | |
| Anti-GAD antibodies | 2/8 (25) | |
| Anti-NMDAR antibodies | 2/42 (5) | |
| Anti-AQP4 antibodies | 0/66 (0) | |
| Brain MRI findings | ||
| T2/FLAIR hyperintensity lesions | 76/86 (88) | |
| Hyperintensities in white matter | 49/84 (58) | |
| Hyperintensities in basal ganglia | 36/84 (43) | |
| Hyperintensities in cerebellum | 5/84 (6) | |
| Gadolinium enhancement lesions | 53/78 (68) | |
| LPRGE patterns | 42/78 (54) | |
| Spinal cord MRI findings | ||
| Intramedullary T2 hyperintensity lesions | 29/71 (41) | |
| Gadolinium enhancement lesions | 24/51 (47) | |
| Intramedullary enhancement lesions | 12/51 (24) | |
| Meningeal enhancement lesions | 13/51 (25) | |
| Therapies | ||
| Corticosteroid therapies | 85/87 (98) | |
| Period from onset to steroid initiation (day) (N = 83) | 24 (7–1084) | |
| Movement Disorders | Patients (%) |
|---|---|
| Ataxia | 43/87 (49) |
| Tremor | 39/87 (45) |
| Myoclonus | 32/87 (37) |
| Dyskinesia | 2/87 (2) |
| Opsoclonus | 2/87 (2) |
| Rigidity | 2/87 (2) |
| Myokymia | 1/87 (1) |
| Choreoathetosis | 1/87 (1) |
| Variables | With MDs | Without MDs | p | ||
|---|---|---|---|---|---|
| Patients (%) | Median (Range) | Patients (%) | Median (Range) | ||
| Demographics | |||||
| Age | 54 (5–83), N = 74 | 30 (17–76), N = 13 | 0.005 | ||
| Male | 50/74 (68) | 6/13 (46) | 0.208 | ||
| Concomitant tumor | 11/74 (15) | 3/12 (25) | 0.400 | ||
| Concomitant autoimmune disease | 2/74 (3) | 0/13 (0) | 1.000 | ||
| Period from onset to admission (days) | 12 (1–1054), N = 74 | 8 (1–39), N = 13 | 0.139 | ||
| Clinical features | |||||
| Urinary disturbance | 59/74 (80) | 8/13 (62) | 0.165 | ||
| Altered consciousness | 55/74 (74) | 11/13 (85) | 0.726 | ||
| Nuchal rigidity and/or Kernig’s sign | 46/74 (62) | 10/13 (77) | 0.364 | ||
| Fever | 44/72 (61) | 10/13 (77) | 0.358 | ||
| Hyperreflexia | 43/73 (59) | 7/13 (54) | 0.768 | ||
| Cognitive disfunction | 37/74 (50) | 5/13 (38) | 0.553 | ||
| Headache | 34/72 (47) | 9/13 (69) | 0.228 | ||
| Psychosis | 31/74 (42) | 6/13 (46) | 0.771 | ||
| Papillary edema | 16/35 (46) | 1/5 (20) | 0.373 | ||
| Weakness | 28/74 (38) | 4/13 (31) | 0.760 | ||
| Sensory disturbance | 22/74 (30) | 2/13 (15) | 0.501 | ||
| Respiratory failure | 14/73 (19) | 5/13 (38) | 0.150 | ||
| Convulsion | 14/74 (19) | 3/10 (30) | 0.712 | ||
| Cerebrospinal fluid | |||||
| Cell counts (cells/μL) | 79 (4–378), N = 73 | 74 (10–472), N = 13 | 0.109 | ||
| Protein concentrations (mg/dL) | 136 (54–320), N = 73 | 156 (30–241), N = 13 | 0.376 | ||
| Oligoclonal IgG bands | 39/56 (70) | 4/5 (80) | 1.000 | ||
| Coexisting neural autoantibodies | |||||
| Anti-MOG antibodies | 2/50 (4) | 0/7 (0) | 1.000 | ||
| Anti-GAD antibodies | 1/7 (14) | 1/1 (100) | 0.250 | ||
| Anti-NMDAR antibodies | 2/38 (5) | 0/4 (0) | 1.000 | ||
| Brain MRI | |||||
| T2/FLAIR hyperintensity lesions | 64/73 (88) | 12/13 (92) | 1.000 | ||
| Hyperintensities in white matter | 43/71 (61) | 6/13 (46) | 0.371 | ||
| Hyperintensities in basal ganglia | 27/71 (38) | 9/13 (70) | 0.065 | ||
| Hyperintensities in cerebellum | 4/71 (6) | 1/13 (8) | 0.578 | ||
| Gadolinium enhancement lesions | 47/67 (70) | 6/11 (55) | 0.316 | ||
| LPRGE | 36/67 (54) | 6/11 (55) | 1.000 | ||
| Spinal cord MRI | |||||
| Intramedullary T2 hyperintensity lesions | 25/60 (42) | 4/11 (36) | 1.000 | ||
| Gadolinium enhancement lesions | 21/44 (48) | 3/7 (45) | 1.000 | ||
| Intramedullary enhancement lesions | 11/44 (25) | 1/7 (14) | 1.000 | ||
| Meningeal enhancement lesions | 11/44 (25) | 2/7 (29) | 1.000 | ||
| Therapies | |||||
| Corticosteroid therapies | 72/74 (97) | 13/13 (100) | 1.000 | ||
| Period from onset to steroid initiation (days) | 25 (7–1084), N = 70 | 14 (8–60), N = 13 | 0.136 | ||
| Movement Disorders | Number of Reports | References |
|---|---|---|
| Ataxia | 23 | [1,2,3,4,5,8,9,13,15,16,17,19,22,23,24,25,26,27,32,36,38,40,41] |
| Tremor | 22 | [1,2,6,8,13,14,20,21,24,26,27,28,29,30,31,33,34,35,36,37,38,39] |
| Myoclonus | 12 | [6,8,13,14,15,16,25,32,33,35,36,37] |
| Dyskinesia | 6 | [4,5,6,17,23,41] |
| Hyperekplexia | 3 | [6,32,35] |
| Dystonia | 2 | [6,23] |
| Parkinsonism | 2 | [6,14] |
| Chorea | 1 | [13] |
| Axial stiffness | 1 | [35] |
| Grimacing | 1 | [18] |
| Oral movements | 1 | [18] |
| Catatonia | 1 | [18] |
| Oculogyric crises | 1 | [35] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kimura, A.; Takekoshi, A.; Shimohata, T. Characteristics of Movement Disorders in Patients with Autoimmune GFAP Astrocytopathy. Brain Sci. 2022, 12, 462. https://doi.org/10.3390/brainsci12040462
Kimura A, Takekoshi A, Shimohata T. Characteristics of Movement Disorders in Patients with Autoimmune GFAP Astrocytopathy. Brain Sciences. 2022; 12(4):462. https://doi.org/10.3390/brainsci12040462
Chicago/Turabian StyleKimura, Akio, Akira Takekoshi, and Takayoshi Shimohata. 2022. "Characteristics of Movement Disorders in Patients with Autoimmune GFAP Astrocytopathy" Brain Sciences 12, no. 4: 462. https://doi.org/10.3390/brainsci12040462
APA StyleKimura, A., Takekoshi, A., & Shimohata, T. (2022). Characteristics of Movement Disorders in Patients with Autoimmune GFAP Astrocytopathy. Brain Sciences, 12(4), 462. https://doi.org/10.3390/brainsci12040462






