Cognitive Considerations in Major Depression: Evaluating the Effects of Pharmacotherapy and ECT on Mood and Executive Control Deficits
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
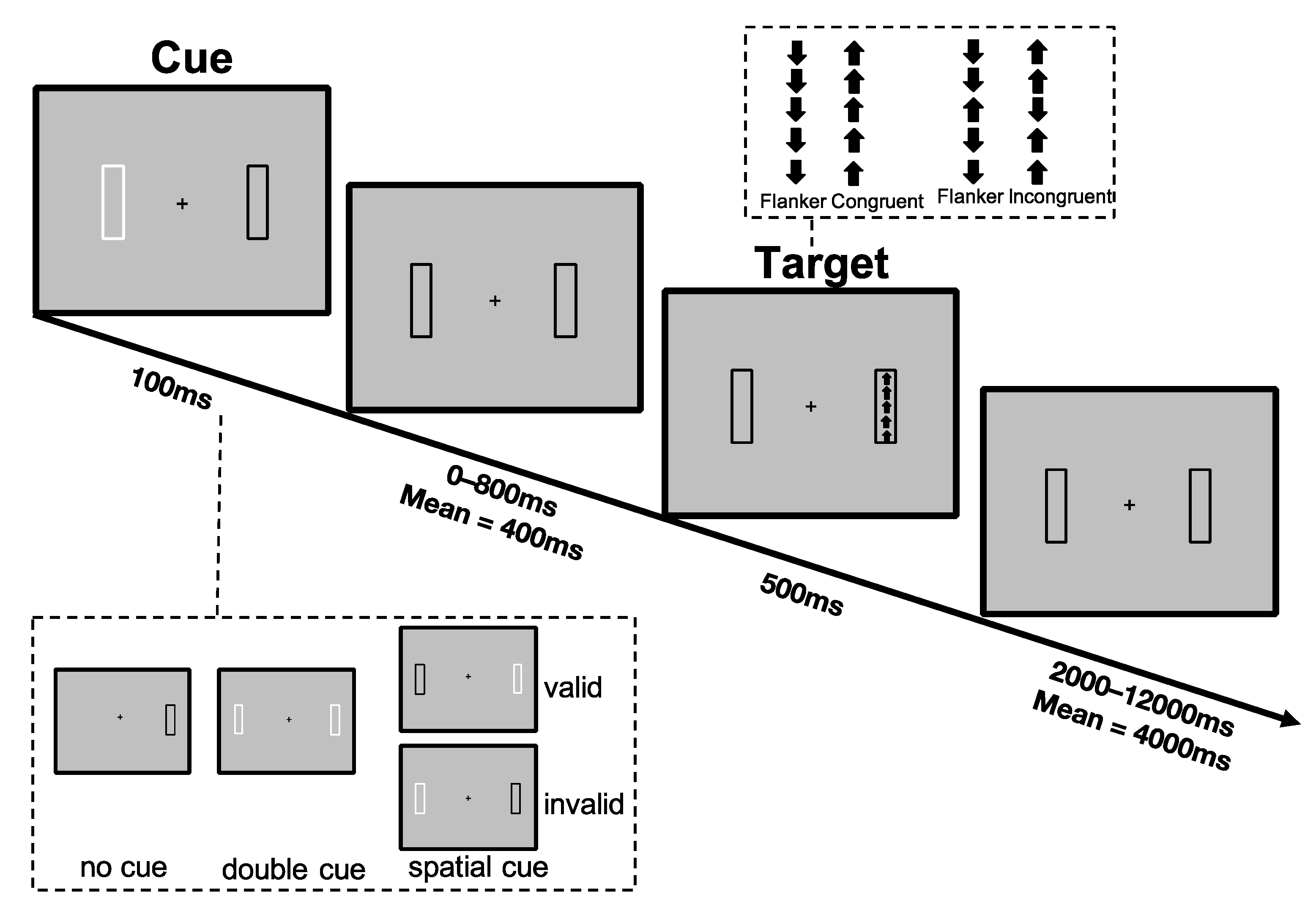
2.2. Lateralized Attention Network Test-Revised (LANT-R)
2.3. ECT Protocol
2.4. Data Analysis
3. Results
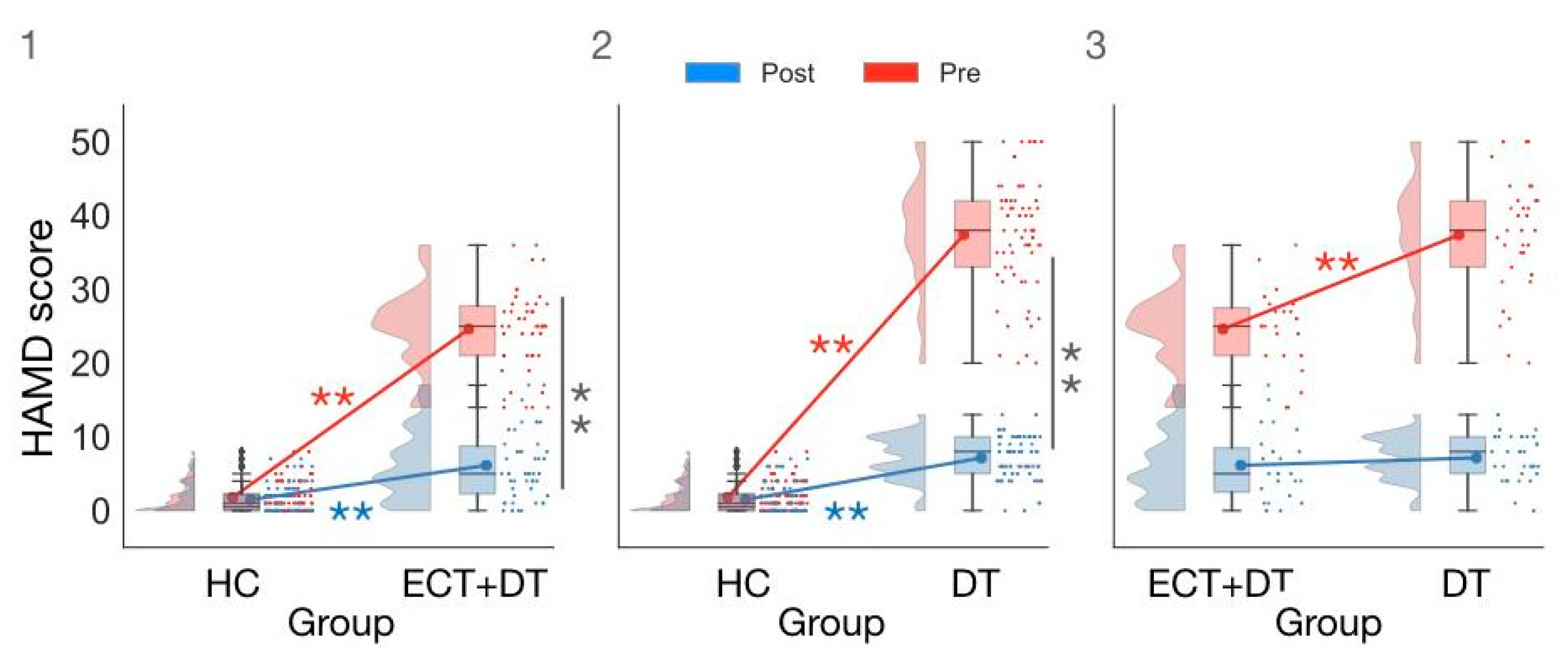
3.1. Demographic and Clinical Differences across Groups
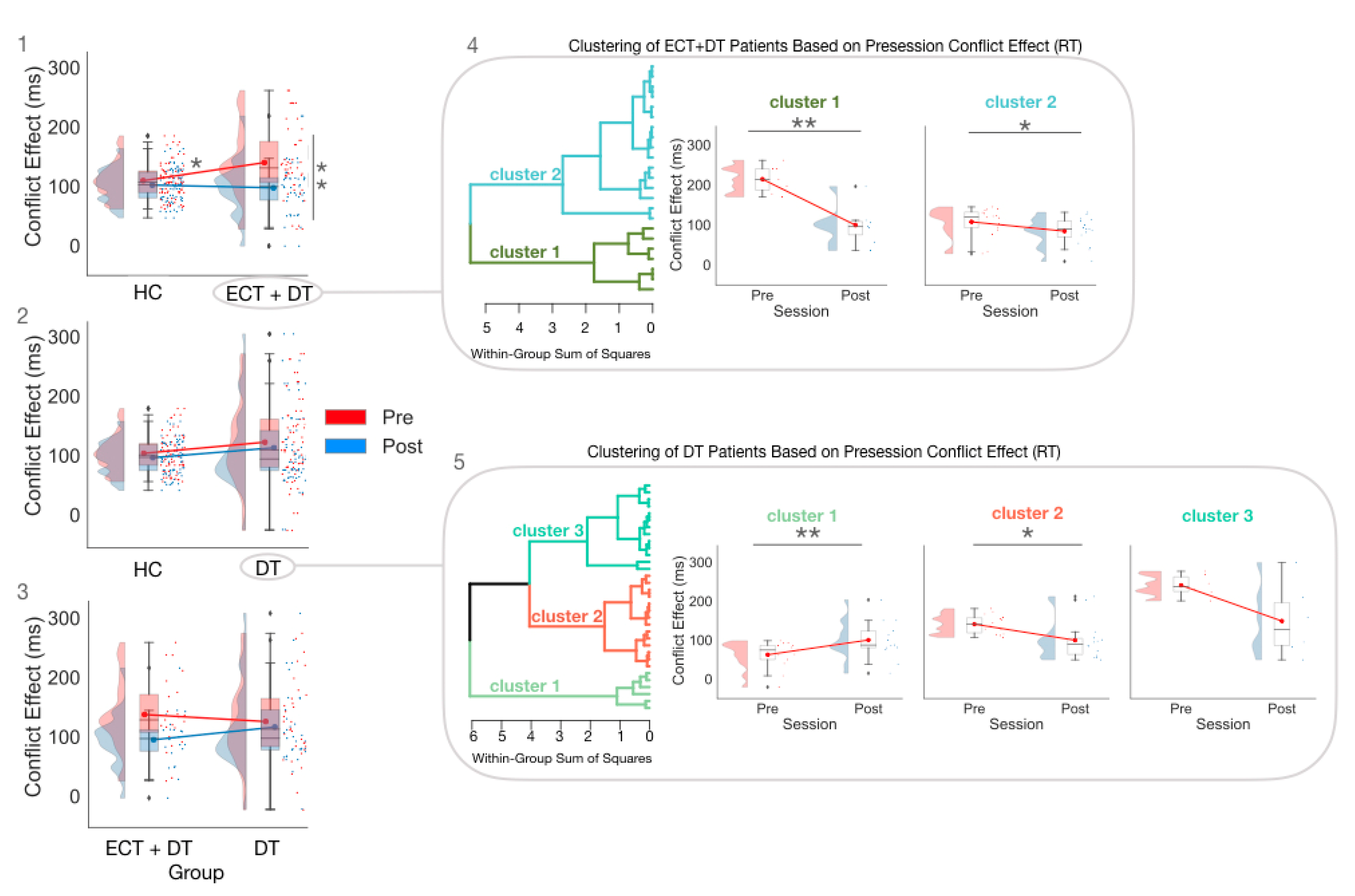
3.2. Treatment Effects on Executive Control Measures Estimated on RT
3.3. Exploratory Cluster Analysis Conducted Separately for the ECT and DT Groups on RT CE
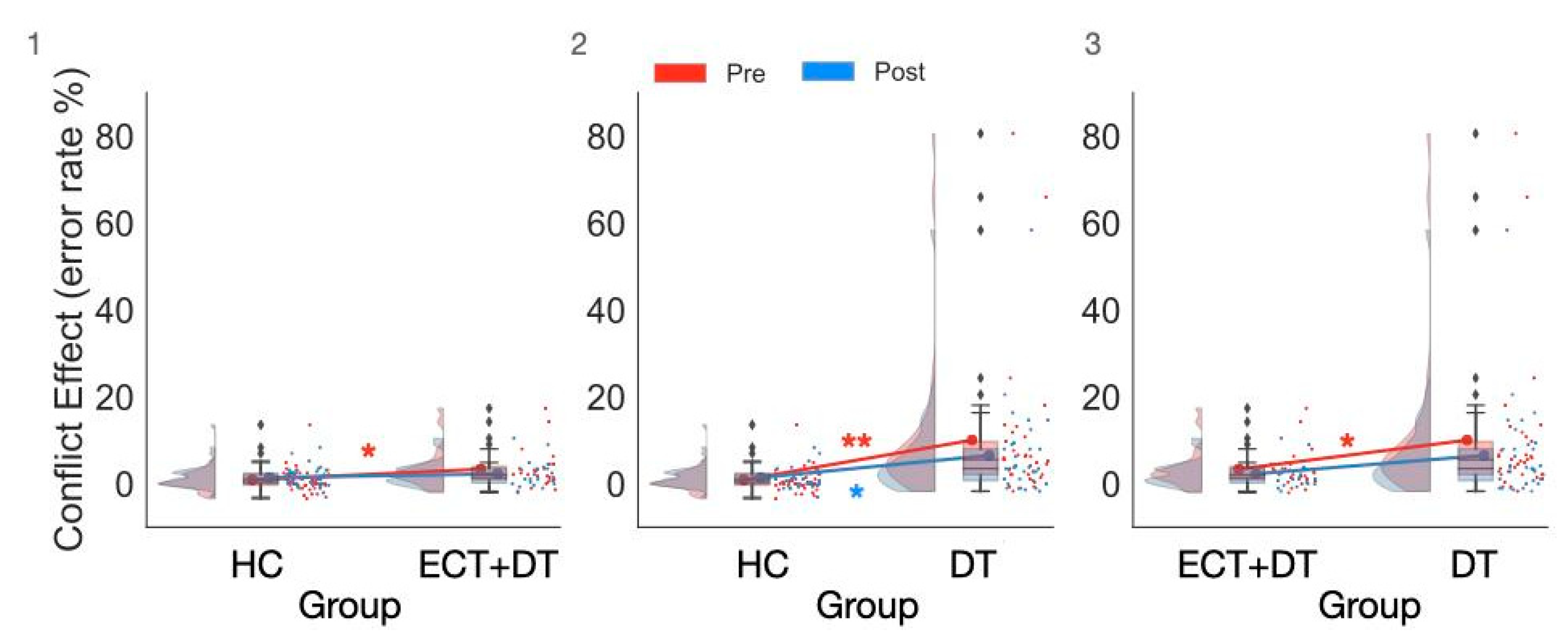
3.4. Treatment Effects on Executive Control Measures Estimated on ER
3.5. Exploratory Correlation Analyses between Executive Control Function, Clinical Symptoms, and Illness Duration
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- National Institute of Mental Health (NIMH). Major Depression. 2019. Available online: https://www.nimh.nih.gov/health/topics/depression (accessed on 22 January 2022).
- World Health Organization. Global Burden of Mental Disorders and the Need for a Comprehensive, Coordinated Response from Health and Social Sectors at the Counter Level; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Clark, M.; DiBenedetti, D.; Perez, V. Cognitive dysfunction and work productivity in major depressive disorder. Expert Rev. Pharm. Outcomes Res. 2016, 16, 455–463. [Google Scholar] [CrossRef]
- American Psychological Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychological Association: Washington, DC, USA, 2013. [Google Scholar]
- Ten Have, M.; Lamers, F.; Wardenaar, K.; Beekman, A.; de Jonge, P.; van Dorsselaer, S.; Tuithof, M.; Kleinjan, M.; de Graaf, R. The identification of symptom-based subtypes of depression: A nationally representative cohort study. J. Affect. Disord. 2016, 190, 395–406. [Google Scholar] [CrossRef]
- Waszczuk, M.A.; Zimmerman, M.; Ruggero, C.; Li, K.; MacNamara, A.; Weinberg, A.; Hajcak, G.; Watson, D.; Kotov, R. What do clinicians treat: Diagnoses or symptoms? The incremental validity of a symptom-based, dimensional characterization of emotional disorders in predicting medication prescription patterns. Compr. Psychiatry 2017, 79, 80–88. [Google Scholar] [CrossRef]
- Buckman, J.E.; Underwood, A.; Clarke, K.; Saunders, R.; Hollon, S.D.; Fearon, P.; Pilling, S. Risk factors for relapse and recurrence of depression in adults and how they operate: A four-phase systematic review and meta-synthesis. Clin. Psychol. Rev. 2018, 64, 13–38. [Google Scholar] [CrossRef]
- Kendler, K.S. The phenomenology of major depression and the representativeness and nature of DSM criteria. Am. J. Psychiatry 2016, 173, 771–780. [Google Scholar] [CrossRef]
- Baune, B.T.; Malhi, G.S.; Morris, G.; Outhred, T.; Hamilton, A.; Das, P.; Bassett, D.; Berk, M.; Boyce, P.; Lyndon, B.; et al. Cognition in depression: Can we THINC-it better? J. Affect. Disord. 2018, 225, 559–562. [Google Scholar] [CrossRef]
- Huang, C.L.-C. The value of patient-administered depression rating scale in detecting cognitive deficits in depressed patients. J. Clin. Med. Res. 2010, 2, 27. [Google Scholar] [CrossRef][Green Version]
- Fried, E.I.; Nesse, R.M. Depression sum-scores don’t add up: Why analyzing specific depression symptoms is essential. BMC Med. 2015, 13, 72. [Google Scholar] [CrossRef]
- Belgaied, W.; Samp, J.; Vimont, A.; Rémuzat, C.; Aballéa, S.; El Hammi, E.; Kooli, A.; Toumi, M.; Akhras, K. Routine clinical assessment of cognitive functioning in schizophrenia, major depressive disorder, and bipolar disorder. Eur. Neuropsychopharmacol. 2014, 24, 133–141. [Google Scholar] [CrossRef]
- Bakkour, N.; Samp, J.; Akhras, K.; El Hammi, E.; Soussi, I.; Zahra, F.; Duru, G.; Kooli, A.; Toumi, M. Systematic review of appropriate cognitive assessment instruments used in clinical trials of schizophrenia, major depressive disorder and bipolar disorder. Psychiatry Res. 2014, 216, 291–302. [Google Scholar] [CrossRef]
- Knight, M.J.; Lyrtzis, E.; Baune, B.T. The association of cognitive deficits with mental and physical Quality of Life in Major Depressive Disorder. Compr. Psychiatry 2020, 97, 152147. [Google Scholar] [CrossRef]
- LeMoult, J.; Gotlib, I.H. Depression: A cognitive perspective. Clin. Psychol. Rev. 2019, 69, 51–66. [Google Scholar] [CrossRef]
- De Raedt, R.; Koster, E.H. Understanding vulnerability for depression from a cognitive neuroscience perspective: A reappraisal of attentional factors and a new conceptual framework. Cogn. Affect. Behav. Neurosci. 2010, 10, 50–70. [Google Scholar] [CrossRef]
- Joormann, J.; Vanderlind, W.M. Emotion regulation in depression: The role of biased cognition and reduced cognitive control. Clin. Psychol. Sci. 2014, 2, 402–421. [Google Scholar] [CrossRef]
- Yaroslavsky, I.; Allard, E.S.; Sanchez-Lopez, A. Can’t look away: Attention control deficits predict rumination, depression symptoms and depressive affect in daily life. J. Affect. Disord. 2019, 245, 1061–1069. [Google Scholar] [CrossRef]
- Cotrena, C.; Branco, L.D.; Shansis, F.M.; Fonseca, R.P. Executive function impairments in depression and bipolar disorder: Association with functional impairment and quality of life. J. Affect. Disord. 2016, 190, 744–753. [Google Scholar] [CrossRef]
- Talarowska, M.; Zajączkowska, M.; Gałecki, P. Cognitive functions in first-episode depression and recurrent depressive disorder. Psychiatr. Danub. 2015, 27, 38–43. [Google Scholar]
- Godard, J.; Grondin, S.; Baruch, P.; Lafleur, M.F. Psychosocial and neurocognitive profiles in depressed patients with major depressive disorder and bipolar disorder. Psychiatry Res. 2011, 190, 244–252. [Google Scholar] [CrossRef]
- Bowie, C.R.; Gupta, M.; Holshausen, K.; Jokic, R.; Best, M.; Milev, R. Cognitive remediation for treatment-resistant depression: Effects on cognition and functioning and the role of online homework. J. Nerv. Ment. Dis. 2013, 201, 680–685. [Google Scholar] [CrossRef]
- Jaeger, J.; Berns, S.; Uzelac, S.; Davis-Conway, S. Neurocognitive deficits and disability in major depressive disorder. Psychiatry Res. 2006, 145, 39–48. [Google Scholar] [CrossRef]
- Petersen, S.E.; Posner, M.I. The attention system of the human brain: 20 years after. Annu. Rev. Neurosci. 2012, 35, 73–89. [Google Scholar] [CrossRef] [PubMed]
- Posner, M.I.; Petersen, S.E. The attention system of the human brain. Annu. Rev. Neurosci. 1990, 13, 25–42. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, E.E.; Heeren, A.; McNally, R.J. Unpacking rumination and executive control: A network perspective. Clin. Psychol. Sci. 2017, 5, 816–826. [Google Scholar] [CrossRef]
- Keilp, J.G.; Gorlyn, M.; Russell, M.; Oquendo, M.A.; Burke, A.K.; Harkavy-Friedman, J.; Mann, J.J. Neuropsychological function and suicidal behavior: Attention control, memory and executive dysfunction in suicide attempt. Psychol. Med. 2013, 43, 539–551. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; McCandliss, B.D.; Sommer, T.; Raz, A.; Posner, M.I. Testing the efficiency and independence of attentional networks. J. Cogn. Neurosci. 2002, 14, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Spagna, A.; Kim, T.H.; Wu, T.; Fan, J. Right hemisphere superiority for executive control of attention. Cortex 2020, 122, 263–276. [Google Scholar] [CrossRef]
- Fernandez-Duque, D.; Baird, J.A.; Posner, M.I. Executive attention and metacognitive regulation. Conscious. Cogn. 2000, 9, 288–307. [Google Scholar] [CrossRef]
- Paelecke-Habermann, Y.; Pohl, J.; Leplow, B. Attention and executive functions in remitted major depression patients. J. Affect. Disord. 2005, 89, 125–135. [Google Scholar] [CrossRef]
- RP Alves, M.; Yamamoto, T.; Arias-Carrión, O.; BF Rocha, N.; E Nardi, A.; Machado, S.; Cardoso, A. Executive function impairments in patients with depression. CNS Neurol. Disord.-Drug Targets 2014, 13, 1026–1040. [Google Scholar] [CrossRef]
- Eriksen, B.A.; Eriksen, C.W. Effects of noise letters upon the identification of a target letter in a nonsearch task. Percept. Psychophys. 1974, 16, 143–149. [Google Scholar] [CrossRef]
- Stroop, J.R. Studies of interference in serial verbal reactions. J. Exp. Psychol. 1935, 18, 643. [Google Scholar] [CrossRef]
- Simon, J.R. The effects of an irrelevant directional cue on human information processing. In Advances in Psychology; Elsevier: Amsterdam, The Netherlands, 1990; pp. 31–86. [Google Scholar]
- Epp, A.M.; Dobson, K.S.; Dozois, D.J.; Frewen, P.A. A systematic meta-analysis of the Stroop task in depression. Clin. Psychol. Rev. 2012, 32, 316–328. [Google Scholar] [CrossRef]
- Kikuchi, T.; Miller, J.M.; Schneck, N.; Oquendo, M.A.; Mann, J.J.; Parsey, R.V.; Keilp, J.G. Neural responses to incongruency in a blocked-trial Stroop fMRI task in major depressive disorder. J. Affect. Disord. 2012, 143, 241–247. [Google Scholar] [CrossRef]
- Tian, Y.; Du, J.; Spagna, A.; Mackie, M.A.; Gu, X.; Dong, Y.; Fan, J.; Wang, K. Venlafaxine treatment reduces the deficit of executive control of attention in patients with major depressive disorder. Sci. Rep. 2016, 6, 28028. [Google Scholar] [CrossRef] [PubMed]
- Gruber, S.; Rathgeber, K.; Bräunig, P.; Gauggel, S. Stability and course of neuropsychological deficits in manic and depressed bipolar patients compared to patients with major depression. J. Affect. Disord. 2007, 104, 61–71. [Google Scholar] [CrossRef] [PubMed]
- Sommerfeldt, S.L.; Cullen, K.R.; Han, G.; Fryza, B.J.; Houri, A.K.; Klimes-Dougan, B. Executive attention impairment in adolescents with major depressive disorder. J. Clin. Child Adolesc. Psychol. 2016, 45, 69–83. [Google Scholar] [CrossRef] [PubMed]
- Han, G.; Klimes-Dougan, B.; Jepsen, S.; Ballard, K.; Nelson, M.; Houri, A.; Kumra, S.; Cullen, K. Selective neurocognitive impairments in adolescents with major depressive disorder. J. Adolesc. 2012, 35, 11–20. [Google Scholar] [CrossRef]
- Pešić, V.; Petrović, J.; Jukić, M.M. Molecular Mechanism and Clinical Relevance of Ketamine as Rapid-Acting Antidepressant. Drug Dev. Res. 2016, 77, 414–422. [Google Scholar] [CrossRef]
- Huddart, R.; Hicks, J.K.; Ramsey, L.B.; Strawn, J.R.; Smith, D.M.; Babilonia, M.B.; Altman, R.B.; Klein, T.E. PharmGKB summary: Sertraline pathway, pharmacokinetics. Pharm. Genom. 2020, 30, 26. [Google Scholar] [CrossRef]
- Shilyansky, C.; Williams, L.M.; Gyurak, A.; Harris, A.; Usherwood, T.; Etkin, A. Effect of antidepressant treatment on cognitive impairments associated with depression: A randomised longitudinal study. Lancet Psychiatry 2016, 3, 425–435. [Google Scholar] [CrossRef]
- Semkovska, M.; McLoughlin, D.M. Objective cognitive performance associated with electroconvulsive therapy for depression: A systematic review and meta-analysis. Biol. Psychiatry 2010, 68, 568–577. [Google Scholar] [CrossRef] [PubMed]
- Mohn, C.; Rund, B.R. Neurocognitive profile in major depressive disorders: Relationship to symptom level and subjective memory complaints. BMC Psychiatry 2016, 16, 108. [Google Scholar] [CrossRef] [PubMed]
- Verwijk, E.; Comijs, H.C.; Kok, R.M.; Spaans, H.P.; Tielkes, C.E.; Scherder, E.J.; Stek, M.L. Short-and long-term neurocognitive functioning after electroconvulsive therapy in depressed elderly: A prospective naturalistic study. Int. Psychogeriatr. 2014, 26, 315–324. [Google Scholar] [CrossRef] [PubMed]
- First, M.B.; Gibbon, M. The Structured Clinical Interview for DSM-IV Axis I Disorders (SCID-I) and the Structured Clinical Interview for DSM-IV Axis II Disorders (SCID-II). Personal. Assess. 2004, 2, 134–143. [Google Scholar]
- Hamilton, M. The Hamilton rating scale for depression. In Assessment of Depression; Springer: Berlin/Heidelberg, Germany, 1986; pp. 143–152. [Google Scholar]
- Spagna, A.; Martella, D.; Fuentes, L.J.; Marotta, A.; Casagrande, M. Hemispheric modulations of the attentional networks. Brain Cogn. 2016, 108, 73–80. [Google Scholar] [CrossRef]
- Bai, S.; Gálvez, V.; Dokos, S.; Martin, D.; Bikson, M.; Loo, C. Computational models of Bitemporal, Bifrontal and Right Unilateral ECT predict differential stimulation of brain regions associated with efficacy and cognitive side effects. Eur. Psychiatry 2017, 41, 21–29. [Google Scholar] [CrossRef]
- Wei, Q.; Tian, Y.; Yu, Y.; Zhang, F.; Hu, X.; Dong, Y.; Chen, Y.; Hu, P.; Wang, K. Modulation of interhemispheric functional coordination in electroconvulsive therapy for depression. Transl. Psychiatry 2014, 4, e453. [Google Scholar] [CrossRef]
- Zimmerman, M.; Posternak, M.A.; Chelminski, I. Derivation of a definition of remission on the Montgomery–Asberg depression rating scale corresponding to the definition of remission on the Hamilton rating scale for depression. J. Psychiatr. Res. 2004, 38, 577–582. [Google Scholar] [CrossRef]
- Bagby, R.M.; Ryder, A.G.; Schuller, D.R.; Marshall, M.B. The Hamilton Depression Rating Scale: Has the gold standard become a lead weight? Am. J. Psychiatry 2004, 161, 2163–2177. [Google Scholar] [CrossRef]
- Stojanovic, Z.; Andric, S.; Soldatovic, I.; Dolic, M.; Spiric, Z.; Maric, N.P. Executive function in treatment-resistant depression before and after electroconvulsive therapy. World J. Biol. Psychiatry 2017, 18, 624–632. [Google Scholar] [CrossRef]
- Kato, M.; Asami, Y.; Wajsbrot, D.B.; Wang, X.; Boucher, M.; Prieto, R.; Pappadopulos, E. Clustering patients by depression symptoms to predict venlafaxine ER antidepressant efficacy: Individual patient data analysis. J. Psychiatr. Res. 2020, 129, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Nuninga, J.O.; Claessens, T.F.; Somers, M.; Mandl, R.; Nieuwdorp, W.; Boks, M.P.; Bakker, S.; Begemann, M.J.; Heringa, S.; Sommer, I.E. Immediate and long-term effects of bilateral electroconvulsive therapy on cognitive functioning in patients with a depressive disorder. J. Affect. Disord. 2018, 238, 659–665. [Google Scholar] [CrossRef] [PubMed]
- Obbels, J.; Verwijk, E.; Vansteelandt, K.; Dols, A.; Bouckaert, F.; Schouws, S.; Vandenbulcke, M.; Emsell, L.; Stek, M.; Sienaert, P. Long-term neurocognitive functioning after electroconvulsive therapy in patients with late-life depression. Acta Psychiatr. Scand. 2018, 138, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Masand, P.S.; Gupta, S. Long-term side effects of newer-generation antidepressants: SSRIS, venlafaxine, nefazodone, bupropion, and mirtazapine. Ann. Clin. Psychiatry 2002, 14, 175–182. [Google Scholar] [CrossRef]
- Bet, P.M.; Hugtenburg, J.G.; Penninx, B.W.; Hoogendijk, W.J. Side effects of antidepressants during long-term use in a naturalistic setting. European Neuropsychopharmacology 2013, 23, 1443–1451. [Google Scholar] [CrossRef]
- Fitzgerald, P.J. Forbearance for fluoxetine: Do monoaminergic antidepressants require a number of years to reach maximum therapeutic effect in humans? Int. J. Neurosci. 2014, 124, 467–473. [Google Scholar] [CrossRef]
- Herrera-Guzmán, I.; Gudayol-Ferré, E.; Herrera-Guzmán, D.; Guàrdia-Olmos, J.; Hinojosa-Calvo, E.; Herrera-Abarca, J.E. Effects of selective serotonin reuptake and dual serotonergic–noradrenergic reuptake treatments on attention and executive functions in patients with major depressive disorder. Psychiatry Res. 2010, 177, 323–329. [Google Scholar] [CrossRef]
- Vilgis, V.; Silk, T.J.; Vance, A. Executive function and attention in children and adolescents with depressive disorders: A systematic review. Eur. Child Adolesc. Psychiatry 2015, 24, 365–384. [Google Scholar] [CrossRef]
- Snyder, H.R. Major depressive disorder is associated with broad impairments on neuropsychological measures of executive function: A meta-analysis and review. Psychol. Bull. 2013, 139, 81. [Google Scholar] [CrossRef]
- Southworth, F.; Grafton, B.; MacLeod, C.; Watkins, E. Heightened ruminative disposition is associated with impaired attentional disengagement from negative relative to positive information: Support for the “impaired disengagement” hypothesis. Cogn. Emot. 2017, 31, 422–434. [Google Scholar] [CrossRef]
- Bourne, V.J. The divided visual field paradigm: Methodological considerations. Laterality 2006, 11, 373–393. [Google Scholar] [CrossRef] [PubMed]
- Asanowicz, D.; Marzecová, A.; Jaśkowski, P.; Wolski, P. Hemispheric asymmetry in the efficiency of attentional networks. Brain Cogn. 2012, 79, 117–128. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Heilman, K.M.; van den Abell, T. Right hemisphere dominance for attention: The mechanism underlying hemispheric asymmetries of inattention (neglect). Neurology 1980, 30, 327. [Google Scholar] [CrossRef]
- Bartolomeo, P.; Malkinson, T.S. Hemispheric lateralization of attention processes in the human brain. Curr. Opin. Psychol. 2019, 29, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Chica, A.B.; de Schotten, M.T.; Toba, M.; Malhotra, P.; Lupiáñez, J.; Bartolomeo, P. Attention networks and their interactions after right-hemisphere damage. Cortex 2012, 48, 654–663. [Google Scholar] [CrossRef]
- Thiebaut De Schotten, M.; Dell’Acqua, F.; Forkel, S.; Simmons, A.; Vergani, F.; Murphy, D.G.; Catani, M. A lateralized brain network for visuo-spatial attention. Nat. Preced. 2011. [Google Scholar] [CrossRef]
- Gálvez, V.; Li, A.; Oxley, C.; Waite, S.; De Felice, N.; Hadzi-Pavlovic, D.; Kumar, D.; Page, A.C.; Hooke, G.; Loo, C.K. Health related quality of life after ECT for depression: A study exploring the role of different electrode-placements and pulse-widths. J. Affect. Disord. 2016, 206, 268–272. [Google Scholar] [CrossRef]
- Shafi, R.M.; Kung, S.; Johnson, E.K.; Lapid, M.I.; Rasmussen, K.G. Decreasing Treatment Duration of Electroconvulsive Therapy (ECT) Using Daily Right Unilateral Ultrabrief Instead of Bitemporal ECT. J. Psychiatr. Pract.® 2018, 24, 410–415. [Google Scholar] [CrossRef]
- Su, L.; Jia, Y.; Liang, S.; Shi, S.; Mellor, D.; Xu, Y. Multicenter randomized controlled trial of bifrontal, bitemporal, and right unilateral electroconvulsive therapy in major depressive disorder. Psychiatry Clin. Neurosci. 2019, 73, 636–641. [Google Scholar] [CrossRef]
- Mackin, R.S.; Areán, P.A. Impaired financial capacity in late life depression is associated with cognitive performance on measures of executive functioning and attention. J. Int. Neuropsychol. Soc. 2009, 15, 793–798. [Google Scholar] [CrossRef]
| HC | ECT | DT | ||||
|---|---|---|---|---|---|---|
| Mean ± SD | Median (IQR) | Mean ± SD | Median (IQR) | Mean ± SD | Median (IQR) | |
| Age (years) | 34.55 ± 11.80 | 30.50 (3.25) | 34.39 ± 10.86 | 31.00 (18) | 34.22 ± 11.57 | 33.00 (19.00) |
| Education (years) | 13.53 ± 3.82 | 15.00 (5.00) | 11.87 ± 4.16 | 14.00 (7.00) | 12.06 ± 3.31 | 11.00 (7.00) |
| HAMD Pre | 1.80 ± 1.96 | 1.00 (2.25) | 24.61 ± 5.56 | 25.00 (6.50) | 37.39 ± 7.80 | 38.00 (9.00) |
| HAMD Post | 1.48 ± 1.96 | 0.50 (2.25) | 6.13 ± 4.79 | 5.00 (6.00) | 7.15 ± 3.00 | 8.00 (5.00) |
| Illness Duration (months) | N/A | N/A | 55.91 ± 93.32 | 12.00 (45.00) | 62.09 ± 68.08 | 36.00 (102.00) |
| Relapse | N/A | N/A | First episode n = 12 | Relapse n = 11 | First episode n = 12 | Relapse n = 21 |
| Gender | Male n = 8 | Female n = 32 | Male n = 4 | Female n = 19 | Male n = 8 | Female n = 25 |
| F | p | ηp2 | BFincl | |
|---|---|---|---|---|
| Group | 0.98 | 0.38 | 0.02 | 0.21 |
| Session | 20.52 | <0.001 | 0.18 | 64,823.21 |
| Hemisphere | 6.40 | 0.01 | 0.06 | 1.05 |
| Group × Session | 4.52 | 0.01 | 0.09 | 85.56 |
| Group × Hemisphere | 1.24 | 0.29 | 0.03 | 0.10 |
| Session × Hemisphere | 2.16 | 0.15 | 0.02 | 0.29 |
| Group × Session × Hemisphere | 0.45 | 0.64 | 0.01 | 0.12 |
| HC | ECT | DT | ||||
|---|---|---|---|---|---|---|
| Mean ± SD | Median (IQR) | Mean ± SD | Median (IQR) | Mean ± SD | Median (IQR) | |
| Pre | 109.56 ± 30.44 | 106.75 (36.38) | 140.06 ± 61.03 | 131.00 (63.75) | 128.26 ± 70.96 | 115.00 (81.00) |
| Post | 101.45 ± 23.59 | 103 (28) | 88.87 ± 39.22 | 92.5 (44.5) | 108.96 ± 60.70 | 95 (48) |
| F | p | ηp2 | BFincl | |
|---|---|---|---|---|
| Group | 7.56 | <0.001 | 0.14 | 40.91 |
| Session | 2.85 | 0.10 | 0.03 | 1.25 |
| Hemisphere | 2.85 | 0.80 | 6.90 × 10−4 | 0.11 |
| Group × Session | 2.54 | 0.08 | 0.05 | 5.18 |
| Group × Hemisphere | 2.54 | 0.91 | 0.002 | 0.06 |
| Session × Hemisphere | 4.90 | 0.03 | 0.05 | 0.55 |
| Group × Session × Hemisphere | 1.53 | 0.22 | 0.03 | 0.17 |
| HC | ECT | DT | ||||
|---|---|---|---|---|---|---|
| Mean ± SD | Median (IQR) | Mean ± SD | Median (IQR) | Mean ± SD | Median (IQR) | |
| Pre | 0.88 ± 2.81 | 0.35 (2.43) | 3.34 ± 4.60 | 2.09 (3.13) | 10.03 ± 17.32 | 5.56 (7.64) |
| Post | 1.41 ± 2.21 | 1.04 (2.43) | 2.27 ± 2.98 | 1.39 (3.30) | 6.52 ± 7.29 | 3.48 (7.29) |
| HC | ECT | DT | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Correlation | τ | p | BF10 | τ | p | BF10 | τ | p | BF10 | |
| Illness Duration | HAMD Pre | - | - | - | −0.05 | 0.75 | 0.28 Ϯ | −0.03 | 0.83 | 0.23 Ϯ |
| Illness Duration | RT Pre CE | - | - | - | 0.11 | 0.49 | 0.34 | −0.6 | 0.61 | 0.26 Ϯ |
| Illness Duration | ER Pre CE | - | - | - | −0.05 | 0.75 | 0.28 Ϯ | 0.002 | 0.99 | 0.23 Ϯ |
| HAMD Pre | RT Pre CE | - | - | - | 0.05 | 0.75 | 0.28 Ϯ | −0.07 | 0.57 | 0.27 Ϯ |
| HAMD Pre | ER Pre CE | - | - | - | 0.16 | 0.29 | 0.47 | 0.10 | 0.41 | 0.32 Ϯ |
| HAMD Diff | RT CE Diff | 0.08 | 0.50 | 0.27 Ϯ | 0.18 | 0.24 | 0.53 | −0.11 | 0.38 | 0.33 Ϯ |
| HAMD Diff | ER CE Diff | 0.04 | 0.75 | 0.22 Ϯ | −0.05 | 0.75 | 0.28 Ϯ | 0.13 | 0.29 | 0.39 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spagna, A.; Wang, J.; Rosario, I.E.; Zhang, L.; Zu, M.; Wang, K.; Tian, Y. Cognitive Considerations in Major Depression: Evaluating the Effects of Pharmacotherapy and ECT on Mood and Executive Control Deficits. Brain Sci. 2022, 12, 350. https://doi.org/10.3390/brainsci12030350
Spagna A, Wang J, Rosario IE, Zhang L, Zu M, Wang K, Tian Y. Cognitive Considerations in Major Depression: Evaluating the Effects of Pharmacotherapy and ECT on Mood and Executive Control Deficits. Brain Sciences. 2022; 12(3):350. https://doi.org/10.3390/brainsci12030350
Chicago/Turabian StyleSpagna, Alfredo, Jason Wang, Isabella Elaine Rosario, Li Zhang, Meidan Zu, Kai Wang, and Yanghua Tian. 2022. "Cognitive Considerations in Major Depression: Evaluating the Effects of Pharmacotherapy and ECT on Mood and Executive Control Deficits" Brain Sciences 12, no. 3: 350. https://doi.org/10.3390/brainsci12030350
APA StyleSpagna, A., Wang, J., Rosario, I. E., Zhang, L., Zu, M., Wang, K., & Tian, Y. (2022). Cognitive Considerations in Major Depression: Evaluating the Effects of Pharmacotherapy and ECT on Mood and Executive Control Deficits. Brain Sciences, 12(3), 350. https://doi.org/10.3390/brainsci12030350






