Heart Rate Variability and Pain: A Systematic Review
Abstract
1. Introduction
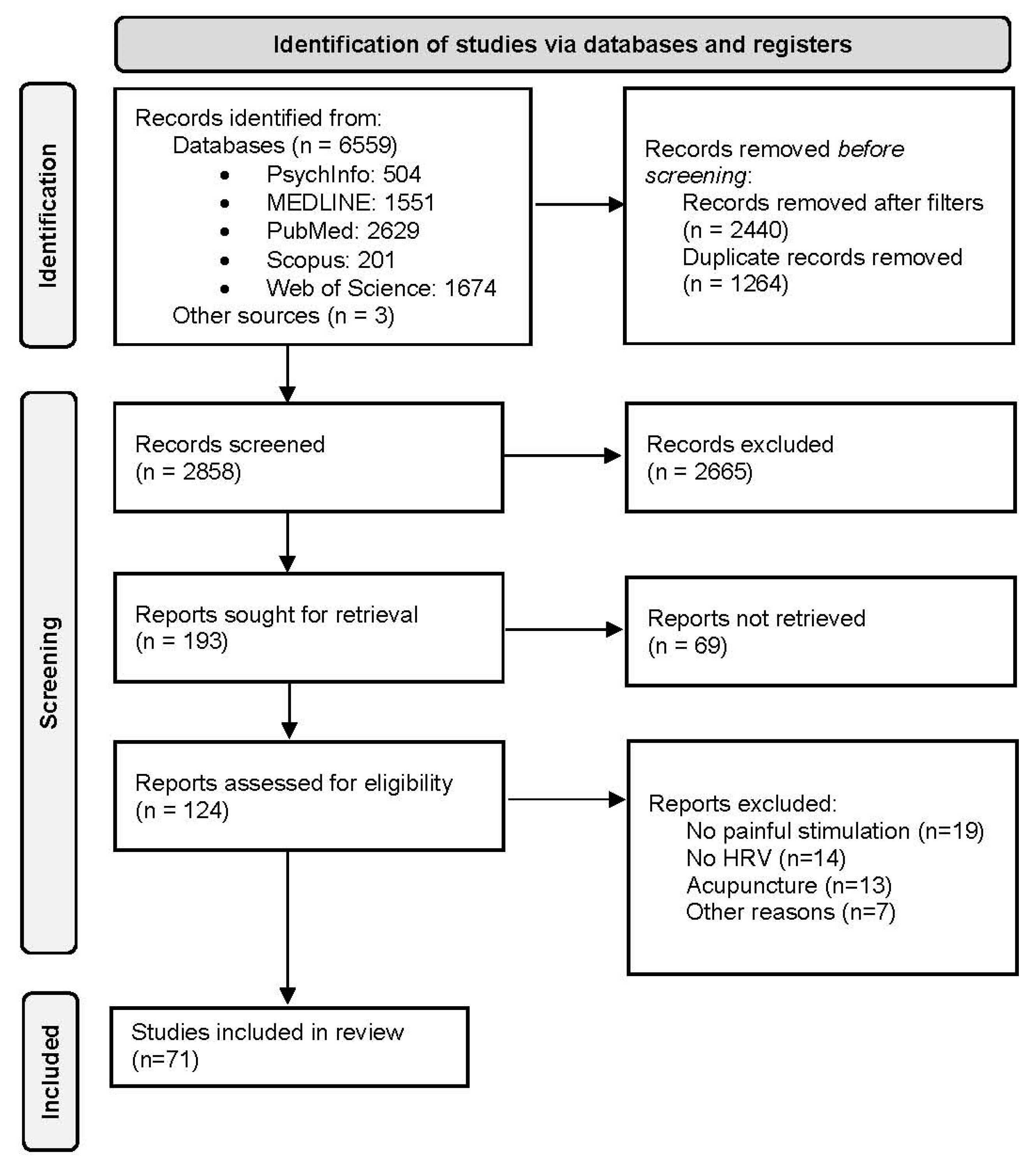
2. Method
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Raja, S.N.; Carr, D.B.; Cohen, M.; Finnerup, N.B.; Flor, H.; Gibson, S.; Keefe, F.J.; Mogil, J.S.; Ringkamp, M.; Vader, K. The revised international association for the study of pain definition of pain: Concepts, challenges, and compromises. Pain 2020, 161, 1976–1982. [Google Scholar] [CrossRef] [PubMed]
- IASP, I. Subcommittee on taxonomy. Pain terms. A list with definitions and notes on usage. Pain 1979, 6, 249–252. [Google Scholar]
- Lumley, M.A.; Cohen, J.L.; Borszcz, G.S.; Cano, A.; Radcliffe, A.M.; Porter, L.S.; Schubiner, H.; Keefe, F.J. Pain and emotion: A biopsychosocial review of recent research. J. Clin. Psychol. 2011, 67, 942–968. [Google Scholar] [CrossRef] [PubMed]
- Kumar, K.H.; Elavarasi, P. Definition of pain and classification of pain disorders. J. Adv. Clin. Res. Insights 2016, 3, 87–90. [Google Scholar] [CrossRef]
- Ellison, D.L. Physiology of pain. Crit. Care Nurs. Clin. 2017, 29, 397–406. [Google Scholar] [CrossRef] [PubMed]
- Hudspith, M.J.; Siddall, P.J.; Munglani, R. Physiology of pain. In Fundamentals of Anaesthesia; Cambridge University Press: Cambridge, UK, 2006; pp. 267–285. [Google Scholar]
- Chu, Y.; Zhao, X.; Han, J.; Su, Y. Physiological signal-based method for measurement of pain intensity. Front. Neurosci. 2017, 11, 279. [Google Scholar] [CrossRef] [PubMed]
- Paine, P.; Worthen, S.F.; Gregory, L.J.; Thompson, D.G.; Aziz, Q. Personality differences affect brainstem auto-nomic responses to visceral pain. Neurogastroenterol. Motil. 2009, 21, 1155-e98. [Google Scholar] [CrossRef]
- Cruccu, G.; Anand, P.; Attal, N.; Garcia-Larrea, L.; Haanpää, M.; Jørum, E.; Serra, J.; Jensen, T.S. EFNS guidelines on neuropathic pain assessment. Eur. J. Neurol. 2004, 11, 153–162. [Google Scholar] [CrossRef]
- Huskisson, E.C. Measurement of pain. Lancet 1974, 304, 1127–1131. [Google Scholar] [CrossRef]
- Gracely, R.H.; McGrath, P.; Dubner, R. Ratio scales of sensory and affective verbal pain descriptors. Pain 1978, 5, 5–18. [Google Scholar] [CrossRef]
- Bourne, S.; Machado, A.G.; Nagel, S.J. Basic anatomy and physiology of pain pathways. Neurosurg. Clin. N. Am. 2014, 25, 629–638. [Google Scholar] [CrossRef] [PubMed]
- Craig, A. A new view of pain as a homeostatic emotion. Trends Neurosci. 2003, 26, 303–307. [Google Scholar] [CrossRef]
- Koenig, J.; Jarczok, M.N.; Ellis, R.; Hillecke, T.; Thayer, J. Heart rate variability and experimentally induced pain in healthy adults: A systematic review. Eur. J. Pain 2013, 18, 301–314. [Google Scholar] [CrossRef] [PubMed]
- Thayer, J.F.; Lane, R.D. A model of neurovisceral integration in emotion regulation and dysregulation. J. Affect. Disord. 2000, 61, 201–216. [Google Scholar] [CrossRef]
- Thayer, J.F.; Lane, R.D. The role of vagal function in the risk for cardiovascular disease and mortality. Biol. Psychol. 2007, 74, 224–242. [Google Scholar] [CrossRef]
- Benarroch, E.E. Pain-autonomic interactions. Neurol. Sci. 2006, 27, s130–s133. [Google Scholar] [CrossRef]
- Thayer, J.F.; Åhs, F.; Fredrikson, M.; Sollers, J.J.; Wager, T.D. A meta-analysis of heart rate variability and neu-roimaging studies: Implications for heart rate variability as a marker of stress and health. Neurosci. Biobehav. Rev. 2012, 36, 747–756. [Google Scholar] [CrossRef]
- Forte, G.; Morelli, M.; Grässler, B.; Casagrande, M. Decision making and Heart Rate variability: A systematic review. Appl. Cogn. Psychol. 2021, 36, 100–110. [Google Scholar] [CrossRef]
- Forte, G.; Favieri, F.; Casagrande, M. Heart rate variability and cognitive function: A systematic review. Front. Neurosci. 2019, 13, 710. [Google Scholar] [CrossRef]
- Forte, G.; Morelli, M.; Casagrande, M. Heart rate variability and decision-making: Autonomic responses in making decisions. Brain Sci. 2021, 11, 243. [Google Scholar] [CrossRef]
- Forte, G.; Favieri, F.; Oliha, E.O.; Marotta, A.; Casagrande, M. Anxiety and attentional processes: The role of resting heart rate variability. Brain Sci. 2021, 11, 480. [Google Scholar] [CrossRef] [PubMed]
- Laborde, S.; Mosley, E.; Thayer, J.F. Heart rate variability and cardiac vagal tone in psychophysiological research–recommendations for experiment planning, data analysis, and data reporting. Front. Psychol. 2017, 8, 213. [Google Scholar] [CrossRef] [PubMed]
- Tracy, L.M.; Koenig, J.; Georgiou-Karistianis, N.; Gibson, S.J.; Giummarra, M.J. Heart rate variability is associated with thermal heat pain threshold in males, but not females. Int. J. Psychophysiol. 2018, 131, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Benarroch, E.E. Pain-autonomic interactions: A selective review. Clin. Auton. Res. 2001, 11, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Bruehl, S.; Chung, O.Y. Interactions between the cardiovascular and pain regulatory systems: An updated review of mechanisms and possible alterations in chronic pain. Neurosci. Biobehav. Rev. 2004, 28, 395–414. [Google Scholar] [CrossRef]
- Nielsen, R.; Nikolajsen, L.; Krøner, K.; Mølgaard, H.; Vase, L.; Jensen, T.S.; Terkelsen, A.J. Pre-operative baroreflex sensitivity and efferent cardiac parasympathetic activity are correlated with post-operative pain. Acta Anaesthesiol. Scand. 2014, 59, 475–485. [Google Scholar] [CrossRef]
- Bruehl, S.; Chung, O.Y.; Jirjis, J.N.; Biridepalli, S. Prevalence of clinical hypertension in patients with chronic pain compared to nonpain general medical patients. Clin. J. Pain 2005, 21, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Van Den Houte, M.; Van Oudenhove, L.; Bogaerts, K.; Van Diest, I.; Van den Bergh, O. Endogenous pain modulation: Association with resting heart rate variability and negative affectivity. Pain Med. 2018, 19, 1587–1596. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. PLoS Med. 2009, 62, e1–e34. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J. Linee guida per il reporting di revisioni sistematiche e meta-analisi: Il PRISMA Statement. PLoS Med 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Acevedo, A.M.; Leger, K.A.; Jenkins, B.N.; Pressman, S.D. Keep calm or get excited? Examining the effects of differ-ent types of positive affect on responses to acute pain. J. Posit. Psychol. 2020, 1–10. [Google Scholar] [CrossRef]
- Adler-Neal, A.L.; Waugh, C.E.; Garland, E.L.; Shaltout, H.A.; Diz, D.I.; Zeidan, F. The role of heart rate variability in mindfulness-based pain relief. J. Pain 2020, 21, 306–323. [Google Scholar] [CrossRef] [PubMed]
- de Araujo, F.X.; Schell, M.S.; Ferreira, G.E.; Pessoa, M.D.V.; de Oliveira, L.R.; Borges, B.G.; Mecagnan, F.E.; Plentz, R.D.M.; Silva, M.F. Auto-nomic function and pressure pain threshold following thoracic mobilization in asymptomatic subjects: A randomized controlled trial. J. Bodyw. Mov. Ther. 2018, 22, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Arsenault, M.; Ladouceur, A.; Lehmann, A.; Rainville, P.; Piché, M. Pain modulation induced by respiration: Phase and frequency effects. Neuroscience 2013, 252, 501–511. [Google Scholar] [CrossRef]
- Aslaksen, P.M.; Myrbakk, I.N.; Høifødt, R.S.; Flaten, M.A. The effect of experimenter gender on autonomic and subjective responses to pain stimuli. Pain 2007, 129, 260–268. [Google Scholar] [CrossRef]
- Aslaksen, P.M.; Flaten, M.A. The roles of physiological and subjective stress in the effectiveness of a placebo on experimentally induced pain. Psychosom. Med. 2008, 70, 811–818. [Google Scholar] [CrossRef]
- Balocchi, R.; Varanini, M.; Menicucci, D.; Santarcangelo, E.L.; Migliorini, S.; Fontani, G.; Carli, G. Heart rate variability in subjects with different hypnotic susceptibility receiving nociceptive stimulation and suggestions of analge-sia. In Proceedings of the 2005 IEEE Engineering in Medicine and Biology 27th Annual Conference, Shanghai, China, 17–18 January 2006; pp. 6996–6999. [Google Scholar]
- Bendixen, K.H.; Terkelsen, A.J.; Baad-Hansen, L.; Cairns, B.E.; Svensson, P. Experimental stressors alter hyper-tonic saline-evoked masseter muscle pain and autonomic response. J. Orofac. Pain 2012, 26, 191–205. [Google Scholar]
- Bendixen, K.H.; Terkelsen, A.J.; Baad-Hansen, L.; E Cairns, B.; Svensson, P. Effect of propranolol on hypertonic saline-evoked masseter muscle pain and autonomic response in healthy women during rest and mental arithmetic task. J. Orofac. Pain 2013, 27, 243–255. [Google Scholar] [CrossRef]
- Boggero, I.A.; Segerstrom, S. Maintenance of affective wellbeing following acute pain in healthy older and younger adults. J. Behav. Med. 2019, 42, 934–946. [Google Scholar] [CrossRef]
- Boggero, I.A.; Segerstrom, S.C. Self-regulatory ability, fatigue, and the experience of pain: Mechanistic insights from pain-free undergraduates. Psychophysiology 2019, 56, e13388. [Google Scholar] [CrossRef]
- Bourassa, K.J.; Ruiz, J.M.; Sbarra, D.A. The impact of physical proximity and attachment working models on car-diovascular reactivity: Comparing mental activation and romantic partner presence. Psychophysiology 2019, 56, e13324. [Google Scholar] [CrossRef]
- Burton, A.R.; Birznieks, I.; Bolton, P.S.; Henderson, L.A.; Macefield, V.G. Effects of deep and superficial experimentally induced acute pain on muscle sympathetic nerve activity in human subjects. J. Physiol. 2009, 587, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Chalaye, P.; Goffaux, P.; Lafrenaye, S.; Marchand, S. Respiratory effects on experimental heat pain and cardiac activity. Pain Med. 2009, 10, 1334–1340. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.-H. Frequency and intensity of electrical stimulation of human sympathetic ganglia affect heart rate variability and pain threshold. Appl. Sci. 2019, 9, 4490. [Google Scholar] [CrossRef]
- Chouchou, F.; Pichot, V.; Perchet, C.; Legrain, V.; Garcia-Larrea, L.; Roche, F.; Bastuji, H. Autonomic pain responses during sleep: A study of heart rate variability. Eur. J. Pain 2011, 15, 554–560. [Google Scholar] [CrossRef] [PubMed]
- Cotton, V.A.; Low, L.A.; Villemure, C.; Bushnell, M.C. Unique Autonomic Responses to Pain in Yoga Practitioners. Psychosom. Med. 2018, 80, 791–798. [Google Scholar] [CrossRef] [PubMed]
- Courtois, I.; Gholamrezaei, A.; Jafari, H.; Lautenbacher, S.; Van Diest, I.; Van Oudenhove, L.; Vlaeyen, J.W. Respiratory hypoalgesia? The effect of slow deep breathing on electrocutaneous, thermal, and mechanical pain. J. Pain 2020, 21, 616–632. [Google Scholar] [CrossRef]
- De Pascalis, V.; Scacchia, P. The influence of reward sensitivity, heart rate dynamics and EEG-delta activity on placebo analgesia. Behav. Brain Res. 2018, 359, 320–332. [Google Scholar] [CrossRef]
- Dodo, N.; Hashimoto, R. The effect of anxiety sensitivity on psychological and biological variables during the cold pressor test. Auton. Neurosci. 2017, 205, 72–76. [Google Scholar] [CrossRef]
- Evans, D.R.; Eisenlohr-Moul, T.; Button, D.F.; Baer, R.A.; Segerstrom, S.C. Self-regulatory deficits associated with unpracticed mindfulness strategies for coping with acute pain. J. Appl. Soc. Psychol. 2013, 44, 23–30. [Google Scholar] [CrossRef]
- Fauchon, C.; Faillenot, I.; Perrin, A.M.; Borg, C.; Pichot, V.; Chouchou, F.; Garcia-Larrea, L.; Peyron, R. Does an observer’s empathy influence my pain? Effect of perceived empathetic or unempathetic support on a pain test. Eur. J. Neurosci. 2017, 46, 2629–2637. [Google Scholar] [CrossRef] [PubMed]
- Fauchon, C.; Pichot, V.; Faillenot, I.; Pommier, B.; Garcia-Larrea, L.; Peyron, R.; Chouchou, F. Contextual modulation of autonomic pain reactivity. Auton. Neurosci. 2018, 212, 28–31. [Google Scholar] [CrossRef] [PubMed]
- Fazalbhoy, A.; Birznieks, I.; Macefield, V.G. Individual differences in the cardiovascular responses to tonic muscle pain: Parallel increases or decreases in muscle sympathetic nerve activity, blood pressure and heart rate. Exp. Physiol. 2012, 97, 1084–1092. [Google Scholar] [CrossRef] [PubMed]
- Fidanza, F.; Varanini, M.; Ciaramella, A.; Carli, G.; Santarcangelo, E.L. Pain modulation as a function of hypnotiz-ability: Diffuse noxious inhibitory control induced by cold pressor test vs explicit suggestions of analgesia. Physiol. Behav. 2017, 171, 135–141. [Google Scholar] [CrossRef]
- Geisler, M.; Herbsleb, M.; Bär, K.-J.; Weiss, T. Dissociation of endogenous pain inhibition due to conditioned pain modulation and placebo in male athletes versus nonathletes. Front. Psychol. 2020, 11, 553530. [Google Scholar] [CrossRef]
- Geva, N.; Pruessner, J.; Defrin, R. Triathletes lose their advantageous pain modulation under acute psychosocial stress. Med. Sci. Sports Exerc. 2017, 49, 333–341. [Google Scholar] [CrossRef]
- Ghione, S.; Del Seppia, C.; Mezzasalma, L.; Emdin, M.; Luschi, P. Human head exposure to a 37 Hz electromagnetic field: Effects on blood pressure, somatosensory perception, and related parameters. Bioelectromagnetics 2004, 25, 167–175. [Google Scholar] [CrossRef]
- Hohenschurz-Schmidt, D.J.; Calcagnini, G.; DiPasquale, O.; Jackson, J.B.; Medina, S.; O’Daly, O.; O’Muircheartaigh, J.; Rubio, A.D.L.; Williams, S.C.R.; McMahon, S.B.; et al. Linking pain sensation to the autonomic nervous system: The role of the anterior cingulate and periaqueductal gray resting-state networks. Front. Neurosci. 2020, 14, 147. [Google Scholar] [CrossRef]
- Huggins, J.; Rakobowchuk, M. Utility of lacrimal caruncle infrared thermography when monitoring alterations in autonomic activity in healthy humans. Graefe’s Arch. Clin. Exp. Ophthalmol. 2018, 119, 531–538. [Google Scholar] [CrossRef]
- Iorfino, F.; Alvares, G.; Guastella, A.J.; Quintana, D.S. Cold face test-induced increases in heart rate variability are abolished by engagement in a social cognition task. Psychophysiology 2016, 30, 38–46. [Google Scholar] [CrossRef]
- Jafari, H.; Gholamrezaei, A.; Franssen, M.; Van Oudenhove, L.; Aziz, Q.; Bergh, O.V.D.; Vlaeyen, J.W.; Van Diest, I. Can slow deep breathing reduce pain? An experimental study exploring mechanisms. J. Pain 2020, 21, 1018–1030. [Google Scholar] [CrossRef] [PubMed]
- Jess, G.; Pogatzki-Zahn, E.M.; Zahn, P.K.; Meyer-Frieem, C.H. Monitoring heart rate variability to assess experimentally induced pain using the analgesia nociception index: A randomised volunteer study. Eur. J. Anaesthesiol. 2016, 33, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Greenspan, J.D.; Ohrbach, R.; Fillingim, R.; Maixner, W.; Renn, C.L.; Johantgen, M.; Zhu, S.; Dorsey, S.G. Racial/ethnic differences in experimental pain sensitivity and associated factors – Cardiovascular responsiveness and psychological status. PLoS ONE 2019, 14, e0215534. [Google Scholar] [CrossRef]
- Kobuch, S.; Fazalbhoy, A.; Brown, R.; Macefield, V.G. Inter-individual responses to experimental muscle pain: Baseline physiological parameters do not determine whether muscle sympathetic nerve activity increases or decreases during pain. Front. Neurosci. 2015, 9, 471. [Google Scholar] [CrossRef]
- Konstantinou, P.; Trigeorgi, A.; Georgiou, C.; Gloster, A.T.; Panayiotou, G.; Karekla, M. Comparing apples and oranges or different types of citrus fruits? Using wearable versus stationary devices to analyze psychophysiological data. Psychophysiology 2020, 57, e13551. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Liu, J.; Che, X. Investigating the influence and a potential mechanism of self-compassion on experimental pain: Evidence from a compassionate self-talk protocol and heart rate variability. J. Pain 2019, 21, 790–797. [Google Scholar] [CrossRef]
- Martin, S.L.; Kerr, K.L.; Bartley, E.J.; Kuhn, B.L.; Palit, S.; Terry, E.L.; DelVentura, J.L.; Rhudy, J.L. Respiration-induced hypo-algesia: Exploration of potential mechanisms. J. Pain 2012, 13, 755–763. [Google Scholar] [CrossRef]
- Matthewson, G.M.; Woo, C.-W.; Reddan, M.C.; Wager, T.D. Cognitive self-regulation influences pain-related physiology. Pain 2019, 160, 2338–2349. [Google Scholar] [CrossRef]
- Meeuse, J.J.; Löwik, M.S.; Löwik, S.A.; Aarden, E.; van Roon, A.M.; Gans, R.O.; van Wijhe, M.; Lefrandt, J.D.; Reyners, A.K. Heart rate variability parameters do not correlate with pain intensity in healthy volunteers. Pain Med. 2013, 14, 1192–1201. [Google Scholar] [CrossRef]
- Nahman-Averbuch, H.; Dayan, L.; Sprecher, E.; Hochberg, U.; Brill, S.; Yarnitsky, D.; Jacob, G. Sex differences in the relationships between parasympathetic activity and pain modulation. Physiol. Behav. 2016, 154, 40–48. [Google Scholar] [CrossRef]
- Nahman-Averbuch, H.; Dayan, L.; Sprecher, E.; Hochberg, U.; Brill, S.; Yarnitsky, D.; Jacob, G. Pain modulation and autonomic function: The effect of clonidine. Pain Med. 2016, 17, 1292–1301. [Google Scholar] [CrossRef] [PubMed]
- Nahman-Averbuch, H.; Sprecher, E.; Jacob, G.; Yarnitsky, D. The relationships between parasympathetic function and pain perception: The role of anxiety. Pain Pract. 2016, 16, 1064–1072. [Google Scholar] [CrossRef] [PubMed]
- Olsson, E.M.; von Schéele, B. Relaxing on a bed of nails: An exploratory study of the effects on the autonomic, cardiovascular, and respiratory systems, and saliva cortisol. J. Altern. Complement. Med. 2011, 17, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Paine, P.; Kishor, J.; Worthen, S.F.; Gregory, L.J.; Aziz, Q. Exploring relationships for visceral and somatic pain with autonomic control and personality. Pain 2009, 144, 236–244. [Google Scholar] [CrossRef]
- Perlaki, G.; Orsi, G.; Schwarcz, A.; Bodi, P.; Plozer, E.; Biczo, K.; Aradi, M.; Doczi, T.; Komoly, S.; Hejjel, L.; et al. Pain-related autonomic response is modulated by the medial prefrontal cortex: An ECG–fMRI study in men. J. Neurol. Sci. 2015, 349, 202–208. [Google Scholar] [CrossRef]
- Petersen, K.K.; Andersen, H.H.; Tsukamoto, M.; Tracy, L.; Koenig, J.; Arendt-Nielsen, L. The effects of propranolol on heart rate variability and quantitative, mechanistic, pain profiling: A randomized placebo-controlled crossover study. Scand. J. Pain 2018, 18, 479–489. [Google Scholar] [CrossRef]
- Picchiottino, M.; Honoré, M.; Leboeuf-Yde, C.; Gagey, O.; Cottin, F.; Hallman, D.M. The effect of a single spinal manipulation on cardiovascular autonomic activity and the relationship to pressure pain threshold: A randomized, cross-over, sham-controlled trial. Chiropr. Man. Ther. 2020, 28, 1–16. [Google Scholar] [CrossRef]
- Piovesan, A.; Mirams, L.; Poole, H.; Moore, D.; Ogden, R. The relationship between pain-induced autonomic arousal and perceived duration. Emotion 2019, 19, 1148–1161. [Google Scholar] [CrossRef]
- Pollatos, O.; Füstös, J.; Critchley, H.D. On the generalised embodiment of pain: How interoceptive sensitivity modulates cutaneous pain perception. Pain 2012, 153, 1680–1686. [Google Scholar] [CrossRef]
- Pollatos, O.; Herbert, B.M.; Füstös, J.; Weimer, K.; Enck, P.; Zipfel, S. Food deprivation sensitizes pain perception. Psychophysiology 2012, 26, 1–9. [Google Scholar] [CrossRef]
- Poulsen, C.E.; Bendixen, K.H.; Terkelsen, A.J.; May, A.; Hansen, J.; Svensson, P. Region-specific effects of trigeminal capsaicin stimulation. J. Oral Facial Pain Headache 2019, 33, 318–330. [Google Scholar] [CrossRef] [PubMed]
- Santarcangelo, E.L.; Carli, G.; Migliorini, S.; Fontani, G.; Varanini, M.; Balocchi, R. Heart-rate control during pain and suggestions of analgesia without deliberate induction of hypnosis. Int. J. Clin. Exp. Hypn. 2008, 56, 255–269. [Google Scholar] [CrossRef] [PubMed]
- Schneider, R. Essential oil inhaler (AromaStick®) improves heat tolerance in the Hot Immersion Test (HIT). Results from two randomized, controlled experiments. J. Therm. Biol. 2020, 87, 102478. [Google Scholar] [CrossRef] [PubMed]
- Sclocco, R.; Beissner, F.; Desbordes, G.; Polimeni, J.; Wald, L.; Kettner, N.W.; Kim, J.; Garcia, R.G.; Renvall, V.; Bianchi, A.M.; et al. Neuroimaging brainstem circuitry supporting cardiovagal response to pain: A combined heart rate variability/ultrahigh-field (7 T) functional magnetic resonance imaging study. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2016, 374, 20150189. [Google Scholar] [CrossRef]
- Sharma, P.; Mavai, M.; Bhagat, O.L.; Murugesh, M.; Sircar, S. Slow deep breathing increases pain-tolerance and modulates cardiac autonomic nervous system. Indian J. Physiol. Pharmacol. 2017, 61, 107–113. [Google Scholar]
- Streff, A.; Kuehl, L.K.; Michaux, G.; Anton, F. Differential physiological effects during tonic painful hand immersion tests using hot and ice water. Eur. J. Pain 2010, 14, 266–272. [Google Scholar] [CrossRef]
- Terkelsen, A.J.; Andersen, O.K.; Molgaard, H.; Hansen, J.; Jensen, T.S. Mental stress inhibits pain perception and heart rate variability but not a nociceptive withdrawal reflex. Acta Physiol. Scand. 2004, 180, 405–414. [Google Scholar] [CrossRef]
- Terkelsen, A.J.; Mølgaard, H.; Hansen, J.; Andersen, O.K.; Jensen, T.S. Acute pain increases heart rate: Differential mechanisms during rest and mental stress. Auton. Neurosci. 2005, 121, 101–109. [Google Scholar] [CrossRef]
- Terkelsen, A.J.; Bach, F.W.; Jensen, T.S. Experimental forearm immobilization in humans induces cold and mechanical hyperalgesia. J. Am. Soc. Anesthesiol. 2008, 109, 297–307. [Google Scholar] [CrossRef]
- Tian, S.; Luo, X.; Che, X.; Xu, G. Self-compassion demonstrating a dual relationship with pain dependent on high-frequency heart rate variability. Pain Res. Manag. 2020, 2020, 3126036. [Google Scholar] [CrossRef]
- Tousignant-Laflamme, Y.; Marchand, S. Autonomic reactivity to pain throughout the menstrual cycle in healthy women. Clin. Auton. Res. 2009, 19, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Tracy, L.M.; Jarczok, M.N.; Ellis, R.J.; Bach, C.; Hillecke, T.K.; Thayer, J.F.; Koenig, J. Heart rate variability and sensitivity to experimentally induced pain: A replication. Pain Pract. 2017, 18, 687–689. [Google Scholar] [CrossRef] [PubMed]
- Treister, R.; Kliger, M.; Zuckerman, G.; Aryeh, I.G.; Eisenberg, E. Differentiating between heat pain intensities: The combined effect of multiple autonomic parameters. Pain 2012, 153, 1807–1814. [Google Scholar] [CrossRef] [PubMed]
- Walter, S.; Gruss, S.; Limbrecht-Ecklundt, K.; Traue, H.C.; Werner, P.; Al-Hamadi, A.; Diniz, N.; Silva, G.M.; Andrade, A.O. Automatic pain quantification using autonomic parameters. Psychol. Neurosci. 2014, 7, 363–380. [Google Scholar] [CrossRef]
- Ye, J.J.; Lee, K.T.; Lin, J.S.; Chuang, C.C. Observing continuous change in heart rate variability and photoplethysmography-derived parameters during the process of pain production/relief with thermal stimuli. J. Pain Res. 2017, 10, 527. [Google Scholar] [CrossRef]
- Zunhammer, M.; Eichhammer, P.; Busch, V. Do cardiorespiratory variables predict the antinociceptive effects of deep and slow breathing? Pain Med. 2013, 14, 843–854. [Google Scholar] [CrossRef]
- Malik, M. Heart rate variability: Standards of measurement, physiological interpretation, and clinical use: Task force of the European Society of Cardiology and the North American Society for Pacing and Electrophysiology. Ann. Noninvasive Electrocardiol. 1996, 1, 151–181. [Google Scholar] [CrossRef]
- Laborde, S.; Mosley, E.; Mertgen, A. Vagal tank theory: The three rs of cardiac vagal control functioning–resting, reactivity, and recovery. Front. Neurosci. 2018, 12, 458. [Google Scholar] [CrossRef]
- Jester, D.J.; Rozek, E.K.; McKelley, R.A. Heart rate variability biofeedback: Implications for cognitive and psychiatric effects in older adults. Aging Ment. Health 2019, 23, 574–580. [Google Scholar] [CrossRef]
| Author (Year) | Country | N (F/M) | Age Mean (SD), Range | Method of Pain Induction | Pain Assessment | Main Study Focus | Derived HRV Measures | Hrv and Pain-Related Findings |
|---|---|---|---|---|---|---|---|---|
| Acevedo et al., (2020) [32] | United States | 195 (138/57) | 20.3 (2.5) | Thermal pain: cold pressor task | PI on a VRS (0–100) | The attenuating role of positive affect on physiological responses to acute pain. | RMSSD | All conditions had a significant increase in RMSSD in response to the CPT. Participants in the low arousal calm and high arousal excited conditions had a significant greater PNS activation during reactivity. |
| Adler-Neal et al., (2020) [33] | United States | 62 (31/31) | 30.53 (1.32) | Thermal pain: heat (thermal stimulator) | PI and PU on a VAS (0–10) | Relationship between the PNS and mindfulness-based pain attenuation | HF | Mindfulness-induced PU reductions were associated with higher HF compared with sham-mindfulness meditation. HF significantly increased during pain stimulation. |
| Appelhans and Luecken (2008) [13] | United States | 59 (37/22) | 19.74 (1.83) | Thermal pain: cold plate | PI and PU on a NRS (0–100). PTh | Between-person variability in pain sensitivity | LF HF | PI not predicted by LF or HF. High LF predicted lower PU scores and greater of PTh (notable and moderate). No association between HF and pain measures. |
| de Araujo et al., (2018) [34] | Brazil | 57 (39/18) | 22.66 (3.9) | Pressure pain (pressure algometer) | PPTh (palmar digital agometer) | Comparing the effects of two mobilization techniques and a placebo intervention, applied on the thoracic vertebral column on HRV and on PPT in asymptomatic subjects | RR STD HR SDNN RMSSD RR trindex HF nu LF nu LH/HF ratio | No difference between groups in HRV, no difference between groups in PPT, except for a reference point (mobilization SLUMP increases PPT of ipsilateral tibialis compared to mobilization PA). |
| Arsenault et al., (2013) [35] | Canada | 20 (9/11) | 26.9 (6.1), 21–42 | Transcutaneous electrical stimulation | PI and PTh on a NRS (0–100). Pain Catastrophizing Scale. | The effects of respiration on pain modulation | LF HF | LF power higher during the two slow breathing conditions. |
| Aslaksen et al., (2007) [36] | Norway | 64 (32/32) | 23.45 (3.24), 19–40 | Thermal pain: heat (thermal stimulator) | PI and PU on a VAS (0–100) | The modulating role of experimenter gender on autonomic pain responses | LF/HF ratio | PI ratings lower for male subject × female experimenter. PU higher in women compared with men. LF/HF ratio increased during pain compared with interstimulus intervals. |
| Aslaksen and Flaten (2008) [37] | Norway | 63 (32/31) | 24.25 (5.05), 18–40 | Thermal pain: heat (thermal stimulator) | PI and PU on VAS (1–100) | The effects of placebo administration on negative emotions and pain ratings | LF/HF ratio | Lower LF/HF ratio and PI during placebo condition. |
| Balocchi et al., (2005) [38] | Italy | 21 | 22 (1.3) | Pressure pain (pressure algometer) | PI on a scale (1–10) | The effect of hypnotic susceptibility on heart rate variability, in subjects receiving nociceptive stimulation and suggestion of analgesia | LF HF | In Highs, PI different between PAIN and AN. In Lows, HF significantly increased, and LF decreased during PAIN compared with B1. |
| Bendixen et al., (2012) [39] | Denmark | 16 (16/0) | 22.9 (2.4) | Muscle pain: injection of hypertonic saline solution; thermal pain: cold pressor test | PI and PU on a NRS (0–10), pain on palpation (POP) on a NRS (0–100) | The modulating role of CPT and PASAT on muscle pain and autonomic function | Mean RR RMSSD SDNN LF HF | Decreased RMSSD, HF, and CCV-HF during CPT. PI and PU higher in HS1 than HS2 during CPT and PASAT conditions. |
| Bendixen et al., (2013) [40] | Denmark | 16(16/0) | 23.6, 20–29 | Muscle pain: injection of hypertonic saline solution | PI and PU on a NRS (0–10). Pain on palpation (POP) on a NRS (0–100) | The effect of propranolol on hypertonic saline-evoked pain and autonomic activity during rest and during PASAT | Mean RR SDNN RMSSD LF HF LF/HF ratio | Parasympathetic parameters were increased in propranolol group compared with control group. |
| Boggero and Segerstrom (2019a) [41] | United States | 100 (62/38) | Younger adults: 19.06 (1.81), 18–28; older adults: 73.44 (4.73), 65–84 | Thermal pain: hand immersion in cold water | PI on a VRS (0–10) | Strategies employed by younger and older adults in order to maintain the affective well-being after an acute pain | Log HRV | Older adults demonstrated significantly lower HRV than younger adults. No correlations between pain and HRV were reported. |
| Boggero and Segerstrom (2019b) [42] | United States | 240 (122/118) | 19.38 (2.39), 18–39 | Pressure pain (pressure algometer); thermal: immersion of the non-dominant foot in cold water | PPTh | The relationship between self-regulatory ability and the experience of pain | Log HF | No relationship between pain and HRV was found. |
| Bourassa et al., (2019) [43] | United States | 102 (77/25) | 19.1 (1.75) | Thermal pain: cold pressor task | PI on NRS (0–10) | The mediating role of a romantic partner in cardiovascular responses during the cold pressor task | RSA | PI significantly lower in the partner present condition compared with control and mental activation conditions. No significant differences in HRV between conditions. |
| Burton et al., (2009) [44] | Australia | 26 (13/13) | 28 | Muscle and subdermal pain: injection of sterile hypertonic saline solution | PI on a VAS (0–10) | The effects of deep and superficial pain on muscle sympathetic nerve activity | LF HF LF/HF ratio | Significant increase in LF/HF ratio during both muscle and superficial pain. |
| Chalaye et al., (2009) [45] | Canada | 20 (9/11) | 25.1 (5.6) | Thermal pain: heat (thermal stimulator) | PTh; PTo. | The effects of breathing on heat pain and autonomic cardiac activity | SDNN LF power HF power | SDNN and LF power significantly increased during pain in deep breathing and HR Biofeedback conditions. No significant differences in HF power. PTh significantly higher during slow deep breathing, HR Biofeedback and distraction conditions; PTo higher in slow deep breathing and HR Biofeedback conditions. |
| Cho (2019) [46] | Korea | 45 (21/24) | 22.4 (1.49) | Electrical stimuli | PTh | The effects of electrical stimulation on the autonomic nervous system | HRV | HRV significantly different between the HF-Li and LF-Hi groups immediately after stimulation and between the HF-LI and LF-Hi groups 30 min after stimulation. |
| Chouchou et al., (2011) [47] | France | 14 (4/10) | 32.8 (7.3) | Thermal pain: heat (laser) | PTh on a Likert-type scale (0–10) | The assessment of autonomic responses to pain during sleep | Mean RR Wavelet power coefficient of LF, HF, and LF/HF ratio | RR significantly decreased after the stimuli. LF and LF/HF ratio significantly increased after the stimuli. No significant differences in HF. |
| Cotton et al., (2018) [48] | United States | 34 (26/8) | 43.18 (11.68) | Thermal pain: heat (thermal stimulator) | PI on a VAS (0–200)PU on a VAS (−100–100) | Autonomic responses to pain in yoga practitioners compared to a control group | RSA SDRR RMSSD pNN50 | Yogis had significantly slower RSA during baseline compared with controls. Controls had lower RSA during pain than during warm trials. Yogis had the same lever of RSA during both pain and warm trials. |
| Courtois et al., (2020) [49] | Belgium | Ex 1: 31(31/0); Ex 2: 28 (28/0); Ex 3: 24 (24/0) | Ex 1: 22.45 (3.10); Ex 2: 20.25 (2.50); Ex 3: 22.55 (3.16) | Ex 1: electrical pain; Ex 2: Thermal (thermal stmulator); Ex 3: Mechanical pain (pressure algometer) | PTh. PI on a NRS (0–10) | The effect of slow deep breathing (SDB) on pain sensitivity, HRV, and baroreflex sensitivity | RMSSD | RMSSD increased during SDB in all experimental conditions. No differences in pain ratings were found, nor in relationships between subjective pain and HRV. |
| De Pascalis and Scacchia (2019) [50] | Italy | 65 (65/0) | 24.5 (2.5), 18–36 | Thermal pain: cold cup test | Pain expectation and PI on a NRS (0–100) | The influence of personality traits on placebo analgesia | RR SDNN LF power HF power LF/HF ratio | Negative correlation was found between pain and time domain but not between pain and frequency domain. |
| Dodo and Hashimoto (2017) [51] | Japan | 74 | 21.14 (2.93) | Thermal pain: cold pressor test | Pain perception on the Wong–Baker Faces Pain Rating Scale, PI on a scale (0–5) | The relationship between anxiety sensitivity and autonomic responses during pain | CVI CSI | CVI: significantly higher during CPT in both the low-AS and the high-AS group; low group also higher in recovery compared with rest; during recovery, significantly higher in low-As group than in the high-As group. Subjective pain higher in high-As group than low-As group post-CPT. |
| Evans et al., (2014) [52] | United States | 63 (29/34) | 18.98 (1.62) | Thermal pain: cold pressor task | PTo (total time in sec) | The effects of brief mindfulness instructions on pain tolerance and HRV | Log HF power | Higher HRV at baseline positively correlated with greater PTo in the control group. |
| Fauchon et al., (2017) [53] | France | 40 (20/20) | 23.2 (8.2) | Thermal pain: heat (thermal stimulator) | PI on a VAS (0–100); PTh | The effect of perceived support on pain modulation and associated vegetative reactions | IBI | No correlation between IBI and pain. |
| Fauchon et al., (2018) [54] | France | 76 (17/59) | 27.8 (6.3) | Thermal pain: heat (thermal stimulator) | n.r. | The role of context in the autonomic responses to acute pain | LF HF LF/HF ratio | LF/HF ratio significantly increased in response to pain only during unempathetic condition. Higher LF during unempathetic condition than in neutral condition. |
| Fazalbhoy et al., (2012) [55] | Australia | 12 (1/11) | 18–48 | Muscle pain: injection of a hypertonic solution | PI on a VAS (0–10); McGill Pain Questionnaire | The cardiovascular responses to tonic pain | LF HF LF/HF ratio | Increasing MSNA group: significantly higher LF power and LF/HF ratio, lower HF power and RMSSD. |
| Fidanza et al., (2017) [56] | Italy | 51 (28/23) | 20–27 | Electrical stimulation; thermal pain: cold pressor test | PI (0–10) | The relationship between pain modulation (suggestion of analgesia VS Diffuse Noxious Inhibitory Control) and hypnotizability | LF/HF ratio | HRV was not modulated by pain experience. |
| Geisler et al., (2020) [57] | Germany | 33 (0/33) | 27.4 (5.65) | Thermal pain: heat (thermal stimulator); pressure pain (pressure algometer); thermal pain: hand immersion in cold water | PI of all stimuli on a VAS (0–100) | Differences in endogenous pain modulation in a sample of athletes and nonathletes | RMSSD SDNN | Athletes had higher RMSSD at rest compared with nonathletes. Negative association between HRV and placebo. |
| Geva et al., (2017) [58] | Israel | 25 (0/25) | 35.9 (10) | Thermal pain: heat (thermal stimulator) | PTh and Pto with the thermal stimulator; PI on a VAS (0–10) | Loss of pain modulation under acute psychosocial stress in triathletes | HRV | HRV correlated negatively with the reduction in CPM due to stress. |
| Ghione et al., (2004) [59] | Italy | 10 (0/10) | 41 (7) | Electromagnetic field exposure | PTh; PTo | The effects of an electromagnetic field on pain perception and on cardiovascular parameters | LF HF | HF progressively increased during sham exposure and remained constant during magnetic exposure. LF increased during both types of exposure. |
| Hohenschurz-Schmidt et al., (2020) [60] | United Kingdom | 21 (8/13) | 26.1 (5.2) | Thermal pain: cold (thermal stimulator) | PI and PU on a VAS (0–100) | Exploration of the neural regions underpinning the relationship between ANS and pain | Log LF Log HF LF/HF ratio | Log LF increased from baseline to cold pain. No associations between HRV and PI. During pain, a positive association was found between log LF and the functional connectivity between dACC and vmPFC. Stronger baseline PAG-vmPFC connectivity had a positive correlation with log LF and a negative correlation with PI. |
| Huggins and Rakobowchuk (2019) [61] | Canada | 16 | 18–35 | Thermal pain: cold pressor test | n.r. | The utility of lacrimal car uncle infrared thermography as a method to monitor alteration in autonomic activity | SDNN RMSSD Mean Rri | Mean RRi decreased with both CPT and MCR. |
| Iorfino et al., (2016) [62] | Canada | 25 (0/25) | 23.96 (2.19), 20–30 | Thermal pain: facial cooling | PI on a VAS | The role of the vagus in social cognition | HRV | HRV was significantly higher during FC than during NFC; HRV significantly greater during baseline than during RMET. |
| Jafari et al., (2020) [63] | Belgium | 48 (35/13) | 22.5 (3) | Thermal pain: heat (thermal stimulator) | PI on a computerized NRS (0–100); PTh | The effects of instructed breathing patterns on experimental pain | Mean IBI RMSSD | Mean IBI lower in SB, SDB-H, and SDB-L conditions compared with UB condition. RMSSD higher in both SDB conditions compared with UB and SB conditions. |
| Jess et al., (2016) [64] | Germany | 20 (0/20) | 24.2 (1.9) | Electrical pain | PI on a NRS (0–10) | The evaluation of pain using the Analgesia Nociception Index (ANI) as a measure of HRV | HRV (ANI) | HRV (ANI) scores lower after each stimulus, with a significant drop within the first 2 min after each stimulus. |
| Kim et al., (2019) [65] | United States | 3159 (1810/1349) | 26.07 (6.51) | Pressure pain (pressure algometer); mechanical cutaneous pain; thermal pain: heat (thermal stimulator) | Pressure: PTh; mechanical: PTh and PI; heat: PTh, PTo, and PI | The effects of psychological status and cardiovascular responsiveness to racial and ethnic differences in pain sensitivity | SDNN RMSSD VLF LF HF | No correlations between HRV and pain have been reported. |
| Kobuch et al., (2015) [66] | Australia | 50 (25/25) | 22.3 (1.15), 18–39 | Muscle pain: injection of hypertonic saline solution | PI on a linear potentiometer calibrated to the NRS (0–10); McGill Pain Questionnaire | The relationship between baseline physiological parameters and MSNA responses to tonic muscle pain | LF HF LF/HF ratio RMSSD | No correlations between pain and HRV have been found. |
| Kostantinou et al., (2020) [67] | Cyprus | 43 (37/6) | 21.37 (3.72) | Thermal pain: cold pressor task | PI on a VAS (0–10); PTo and PTh | Comparing psychophysiological data captured by wearable and stationary devices during experimentally induced pain | RMSSD pNN50 mean RR SDNN | Both devices registered an increase in SDNN and RMSSD and a decrease in mean RR during experimental phases. Only the wearable devices registered increased pNN50 during experimental phases. |
| Luo et al., (2020) [68] | China | 29 (14/15) | 19.93 (1.6), 19–27 | Thermal pain: cold (holding a bottle with iced water) | PI on a scale (0–10) | The role and mechanisms of self-compassion in pain perception | HF | Increased HFs were associated with lower PI in the self-compassion compared with control condition. HF higher in self-compassion compared with control. |
| Martin et al., (2012) [69] | United States | 30 (20/10) | 21 (5.5) | Electric stimulation | PI on a NRS (0–100); PTh | The influence of experimentally manipulated breathing on pain | Mean RR RMSSD | HRV changed during breathing manipulation, but it was not correlated with pain outcomes. |
| Matthewson et al., (2019) [70] | United States | 84 (42/42) | 27.9 (6.29) | Thermal stimuli: heat (thermal stimulator) | PI on a NRS (0–100) | The role of cognitive self-regulation in pain experience and its effects on autonomic responses | IBI | Association was found between IBI and pain. |
| Meeuse et al., (2013) [71] | Netherlands | 73 (44/29) | 30 (11) | Thermal pain: heat (thermal stimulator) | PI on a VAS (0–100) | The usefulness of HRV in quantifying pain intensity | IBI lnSDNN lnLF lnHF LF/HF ratio | lnSDNN and lnLF significantly decreased during pain compared with baseline. No significant correlation between PI and HRV parameter was found. |
| Nahman-Averbuch et al., (2016a) [72] | Israel | 40 (20/20) | 26.45 (3.85) | Thermal pain: heat (thermal stimulator) and cold (immersion of a foot in cold water); mechanical pain | PTh; PI on a NPS (0–100) | Sex differences in the relationship between pain perception and HRV | RMSSD LF HF LF/HF ratio | Women: LFnu significantly lower and Hfnu significantly higher. Men: higher RMSSD significantly negatively correlated with higher pain adaptation and with more efficient CPM response. |
| Nahman-Averbuch et al., (2016b) [73] | Israel | 40 (20/20) | 26.45 (3.85) | Thermal pain: heat (thermal stimulator) and cold (immersion of a foot in cold water); mechanical pain | M Pain: PI on a NPS (0–100); PTh CPM: PI of the TS on a COVAS; CS on an NPS | The effects of oral clonidine on pain perception | RMSSD LF nu HF nu LF/HF ratio | Higher RMSSD in clonidine group. No differences found in the other HRV parameters. |
| Nahman-Averbuch et al., (2016c) [74] | Israel | 30 (30/0) | 25.3 (4.1) | Thermal pain: heat (thermal stimulator and the immersion of a hand in hot water); mechanical pain | Thermal: PTh; PI on a NPS (0–100); mechanincal: PI on a NPS (0–100) | Effect of anxiety level on parasympathetic function and pain perception | RMSSD LF nu HF nu LF/HF ratio | Increased parasympathetic activity during recovery in both groups. In the high-anxiety group, higher RMSSD during baseline correlated with higher pain ratings during tonic pain stimulus. |
| Olsson and von Schéele (2011) [75] | Sweden | 32 (20/12) | 39.7 (8.6) | Bed of nails | PI on a NRS (0–10) | Subjective physiologic responses of lying on a bed of nails (BN) | SDNN log LF log HF log | HF higher on the BN. Higher SDNN and LF during relaxing instruction on CD while lying on the BN. |
| Paine et al., (2009a) [8] | United Kingdom | 19 (11/8) | 22–54 | Visceral pain: esophageal balloon distension | PTo; PTh | The relationship between personality and autonomic responses to visceral pain | CVC CSI | CSI increased during pain; no changes in CVC during pain. |
| Paine et al., (2009b) [76] | United Kingdom | 18 (16/2) | 35.4 (2.7) | Visceral pain: proximal and distal balloon distension; somatic pain: nail-bed stimulation | PI and PU on a VRS (0–10); PTo and PTh | The relationship between autonomic control and personality in response to visceral and somatic pain | CVT CSI | Increased CVT in the 90 s post-stimulus compared with pre-stimulus CVT. Significantly greater increase in CVT for distal balloon than for nail bed. |
| Perlaki et al., (2015) [77] | Hungary | 18 (0/18) | 22.89 (1.96) | Thermal pain: heat (thermal stimulator) | PI on a VAS (0–10); PTh | Investigating the brain structures responsible for pain-related autonomic changes | LF HF LF/HF ratio | The median COPE of left MPFC showed negative correlations with LF/HF ratio and a positive correlation with HFnu. The median COPE of right MPFC showed significant negative correlations with SDNN. |
| Petersen et al., (2018) [78] | Denmark | 25 (0/25) | 25.6, 20–37 | Pressure pain (pressure algometer); thermal pain: heat (thermal stimulator) and cold (cold pressor test) | PI on a VAS (0–10); PTh; PTo | The effect of propranolol on HRV and pain perception | Mean IBI RMSSD pNN50 | Mean IBI significantly lower and RMSSD significantly higher during CPT compared with baseline. |
| Picchiottino et al., (2020) [79] | France | 41 (22/19) | 19.9 (3.5) | Pressure pain (pressure algometer) | PTh | The effect of spinal manipulation on cardiovascular autonomic activity and the relationship to pressure pain threshold | LF HF LF/HF ratio RMSSD SDNN | Weak and moderate positive association between changes in PTh and changes in log LF. |
| Piovesan et al., (2019) [80] | United Kingdom | 40 (30/10) | 26.2 (3.91) | Electrical pain; thermal pain: heat (thermal stimulator) | PI on a NRS (0–10) | The relationship between autonomic nervous system and perceived duration of pain experience | HF nu | Only high-intensity stimuli were associated with changes in HRV. No relationship between heat pain and HRV was found. |
| Pollatos et al., (2012a) [81] | Germany (?) | 60 (30/30) | 24.4 (3.2) | Pressure pain (pressure algometer) | PTh; PTo; PI and PU on a VAS (1–9) | The role of interception sensitivity on cutaneous pain perception | LF HF LF/HF ratio | HFnu significantly decreased while LFnu and LF/HF ratio significantly increased during pain. |
| Pollatos et al., (2012b) [82] | Germany | 22 (22/0) | 24.4 (2.8), 21–31 | Pressure pain (pressure algometer) | PTh, PTo; PI and PU on a scale (1–9) | The effects of food deprivation on pain perception | HF nu LF/HF ratio | Day 1: PTo positively correlated with HF nu and inversely correlated with LF/HF ratio. Experimental group: after 24 h of food deprivation, significant positive correlation between differences in HF and PTh (hungry minus breakfast). |
| Poulsen et al., (2019) [83] | Denmark | 20 (10/10) | 25.0 (4.0) | Capsaicin application; somatosensory functions; thermal pain: heat and cold (thermal stimulator); mechanical pain (calibrated von Frey nylon filaments) | PI on a NRS (0–100) | The region-specific effects of painful stimulation | Mean RR SDNN RMSSD LF HF | Higher mean RR, increased RMSSD, SDNN, LF power, HF power, and CCV-HF power during capsaicin stimulation. |
| Santarcangelo et al., (2008) [84] | Italy | 19 (19/0) | 21 | Pressure pain (pressure algometer) | PI on a scale (0–10) | Differences due to hypnotizability in the pain-related modulation of HRV during suggestion of analgesia | Mean RR HF LF LF/HF SDNN RMSSD CSI | Mean RR shorter during pain and AN than during baseline. SDNN shorter during pain than during baseline. |
| Schneider (2020) [85] | Germany | 40 (20/20) | 35.1, 24–55 | Thermal pain: heat (hot immersion test) | PTo, PI, and PU on an NRS (0–10) | The effects of essential oil inhaler on pain perception | RMSSD SDNN | RMSSD: significantly higher during pain than during baseline; higher in the verum condition. SDNN: larger in the verum condition than in the placebo condition. |
| Sclocco et al., (2016) [86] | United States | 11 (3/8) | 33 (4) | Pressure pain (pressure cuff) | PI on a scale (0–100) | Investigating specific brainstem nuclei involved in autonomic responses to pain | LF HF LF/HF ratio | HF power decreased during pain compared with rest. |
| Sharma et al., (2017) [87] | India | 30 (15/15) | 18–25 | Cold pain | PTh, PTo | The modulating role of slow deep breathing on pain perception and cardiac autonomic activity | Mean RR SDNN RMSSD pNN50 LF power HF power LF/HF ratio | PTo, SDNN, RMSSD, LF power, and LF/HF ratio significantly higher during SDB condition compared with spontaneous breathing. HF power significantly lower during SDB condition. |
| Streff et al., (2010) [88] | Luxemburg | 35 (18/17) | 24, 19–57 | Heat pain; cold pressor trial | PTh; PI on a NRS (0–100); PU on a VAS (0–10) | The physiological effects of two different tonic thermal stimuli | LF/HF ratio | LF/HF ratio relative to baseline higher on CPT compared with HIT. |
| Terkelsen et al., (2004) [89] | Denmark | 26 (0/26) | 24, 21–31 | Electrical stimulation (sural nerve stimulation) | PTh; PI and PU on an NRS (0–10) | The effects of mental stress on pain perception, HRV, and nociceptive withdrawal reflex | Mean RR SDNN LF HF | Pain + PASAT decreased mean RR, SDNN, LF power, CCV-LF, HF power, and CCV-HF compared with pain at baseline. Pain + attention decreased HF power. |
| Terkelsen et al., (2005) [90] | Denmark | 26 (0/26) | 24, 21–31 | Electrical stimulation (sural nerve stimulation) | PTh; PI on an NRS (0–10) | The effects of stress on the HRV responses to acute pain | Mean RR SDNN LF HF | Pain at rest: mean RR significantly decreased, LF power and CCV-LF increased. Attention to pain: mean RR decreased and CCV-LF increased. Pain + PASAT: mean RR decreased. |
| Terkelsen et al., (2008) [91] | Denmark | 45 (22/23) | 23, 18–27 | Cold pain; heat pain; pressure pain | PTh | The effects of the forearm immobilization on pain perception | Mean RR SDNN LF HF | PASAT reduced mean RR, SDNN HF power, and LF power. |
| Tian et al., (2020) [92] | China | 57 (30/27) | 20.28 (2.38), 19–33 | Cold pain | PI on a scale (0–10) | The impact of the heart rate variability on the relationship between self-compassion and pain | HF | Self-compassion was associated with increased pain when HF was lower; self-compassion was associated with lower pain when HF was higher. |
| Tousignant-Laflamme and Marchand (2009) [93] | Canada | 32 (32/0) | 34.3 (7.5) | Cold pressor test | PI on a NRS (0–100) | Autonomic reactivity to pain throughout the menstrual cycle | LF HF LF/HF ratio NN50 | No significant differences in HRV were found between rest and CPT. |
| Tracy et al., (2018a) [24] | Australia | 51 (26/25) | 21.9, 18–36 | Heat pain | PTh | Sex differences in the association between resting HRV and pain sensitivity | lnRMSSD lnLF lnHF | Higher resting LF was associated with higher PTh. In men, significant positive relationship between PTh and resting LF and HF found. |
| Tracy et al., (2018b) [94] | Germany | 35 (29/6) | 22.80 (2.45) | Cold pain | PTh; PTo; PI on a VAS (0–10) | The association between HRV and pain sensitivity | LF HF | LF and HF predicted PI. |
| Treister et al., (2012) [95] | Israel | 55 (21/34) | 25.9 (4.1), 20–37 | Heat pain | PTh; PI on an NPS (0–100) | Comparing different intensities of pain employing different autonomic parameters | HF | HF showed a negative peak (a decrease compared with pre-stimulus) followed by a gradual increase. |
| Van Den Houte et al., (2018) [29] | Belgium | 63 (48/15) | 21.49 (3.80), 18–41 | Heat pain | PTh; PI on an NRS (0–100) | The association of HRV and negative affectivity in the endogenous pain modulation | RMSSD | Baseline RMSSD significantly related to the difference in PI between the constant and offset condition. Higher RMSSD and larger offset analgesia. |
| Walter et al., (2014) [96] | Germany | 90 (45/45) | 18–65 | Heat pain | PTh; PTo | The quantification of pain experience using autonomic parameters | IBI | An association between pain and IBI was found. |
| Ye et al., (2017) [97] | Taiwan | 40 (19/21) | 22.5, 20–27 | Heat pain | PI on an NRS | Changes in physiological parameters during the process of pain production and relief | Mean RR LF HF | LF significantly changed between segments D and E. |
| Zunhammer et al., (2013) [98] | Germany | 20 (10/10) | 24.4, 20.7–28.6 | Heat and cold pain | PTh; PI and PU on a VAS (0–100) | The relationship between breathing and pain perception | SDRR | All breathing exercises with the exception of paced resting frequency significantly increased SDRR compared with baseline. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Forte, G.; Troisi, G.; Pazzaglia, M.; Pascalis, V.D.; Casagrande, M. Heart Rate Variability and Pain: A Systematic Review. Brain Sci. 2022, 12, 153. https://doi.org/10.3390/brainsci12020153
Forte G, Troisi G, Pazzaglia M, Pascalis VD, Casagrande M. Heart Rate Variability and Pain: A Systematic Review. Brain Sciences. 2022; 12(2):153. https://doi.org/10.3390/brainsci12020153
Chicago/Turabian StyleForte, Giuseppe, Giovanna Troisi, Mariella Pazzaglia, Vilfredo De Pascalis, and Maria Casagrande. 2022. "Heart Rate Variability and Pain: A Systematic Review" Brain Sciences 12, no. 2: 153. https://doi.org/10.3390/brainsci12020153
APA StyleForte, G., Troisi, G., Pazzaglia, M., Pascalis, V. D., & Casagrande, M. (2022). Heart Rate Variability and Pain: A Systematic Review. Brain Sciences, 12(2), 153. https://doi.org/10.3390/brainsci12020153








