Neurosurgical Treatment of Pain
Abstract
1. Introduction
2. Mechanisms of Pain Modulation
2.1. Nociceptors and Peripheral Nerves
2.2. Spinal Ganglion-Dorsal Root
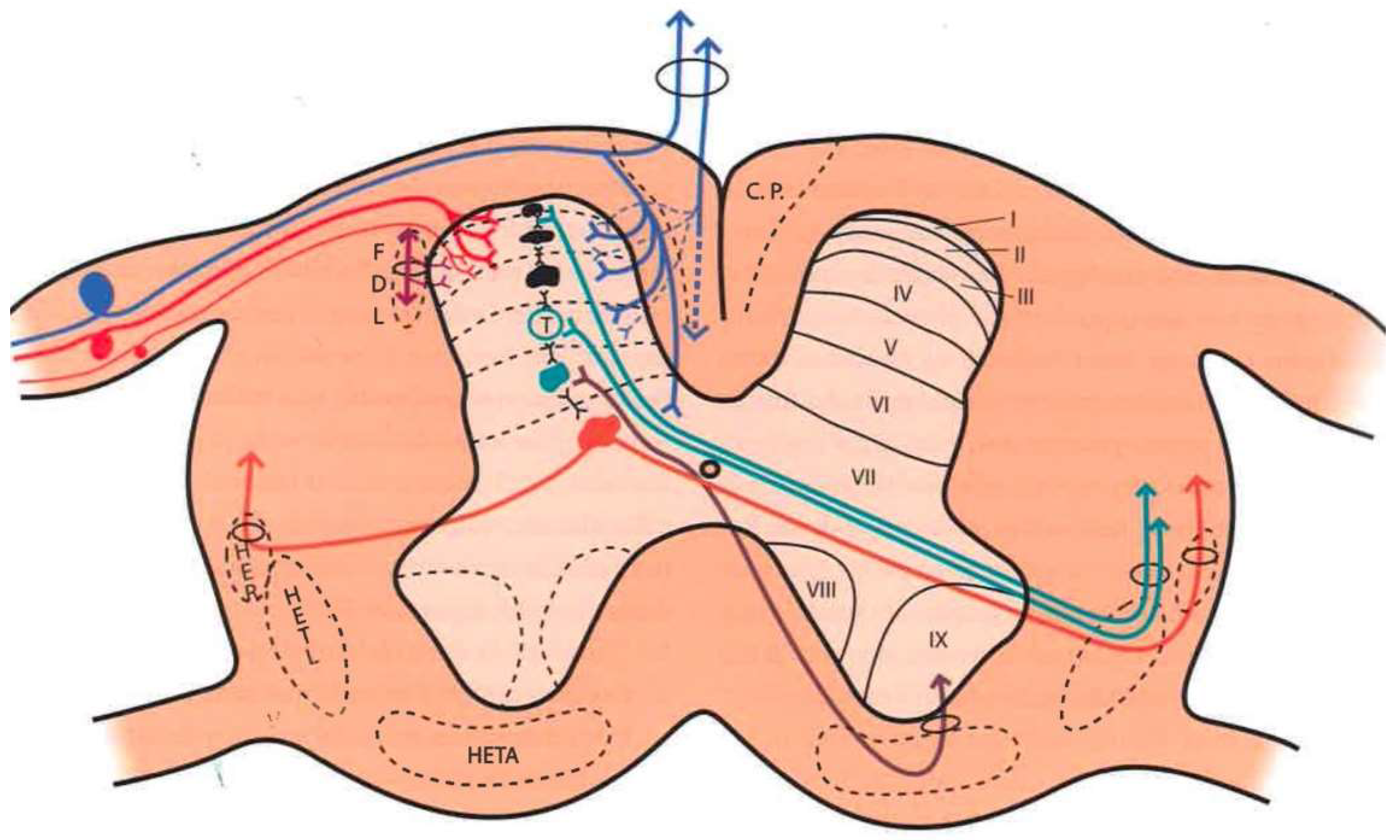
2.3. Dorsal Horn of the Spinal Cord
- Lamina I (marginal layer of Waldeyer) consists of large neurons whose afferents are mostly formed by Aδ fibers and, to a lesser extent, unmyelinated C fibers [18]. They project to the ventral posterior (VP) and intralaminar thalamic nuclei as well as to the periaqueductal gray matter (PAG) [19,20]. These are type I neurons, as described by Iggo [21], responding to painful or almost noxious stimuli.
- Laminae II and III (substantia gelatinosa of Rolando) contain small-sized neurons receiving most afferents through C fibers, and the rest through collaterals of Aβ fibers of the posterior spinal cord [18]. Their extensions form part of Lissauer’s dorsolateral fasciculus along three or more segments. Their function is still obscure, acting as interneurons and maintaining an inhibitory or excitatory influence on the cells of laminae I, IV, and V [22].
- The cells of laminae IV, V, and VI (nucleus proprius, neck, or reticular substance and base, respectively, of the dorsal horn) are Iggo type II nociceptive neurons, which respond to low-threshold peripheral mechanical stimuli, but increase their firing frequency when the stimulus becomes noxious [23]. For Mayer and Price [24], the activation of these type II neurons can lead to experiencing pain. From these laminae originates the spinothalamic tract, which projects to the sensory ventral posteromedial and posterolateral (VPM and VPL) thalamic nuclei, as well as PAG.
- Lamina VII (intermediate zone) gives rise to the spinoreticular tract, next to the spinothalamic tract, and its neurons respond to stimuli that trigger the highest-threshold receptors [25].
2.4. Pain Information Pathways
2.4.1. Neospinothalamic Tract or Lateral Spinothalamic Tract
2.4.2. Paleospinothalamic Tract
2.4.3. Archispinothalamic Tract
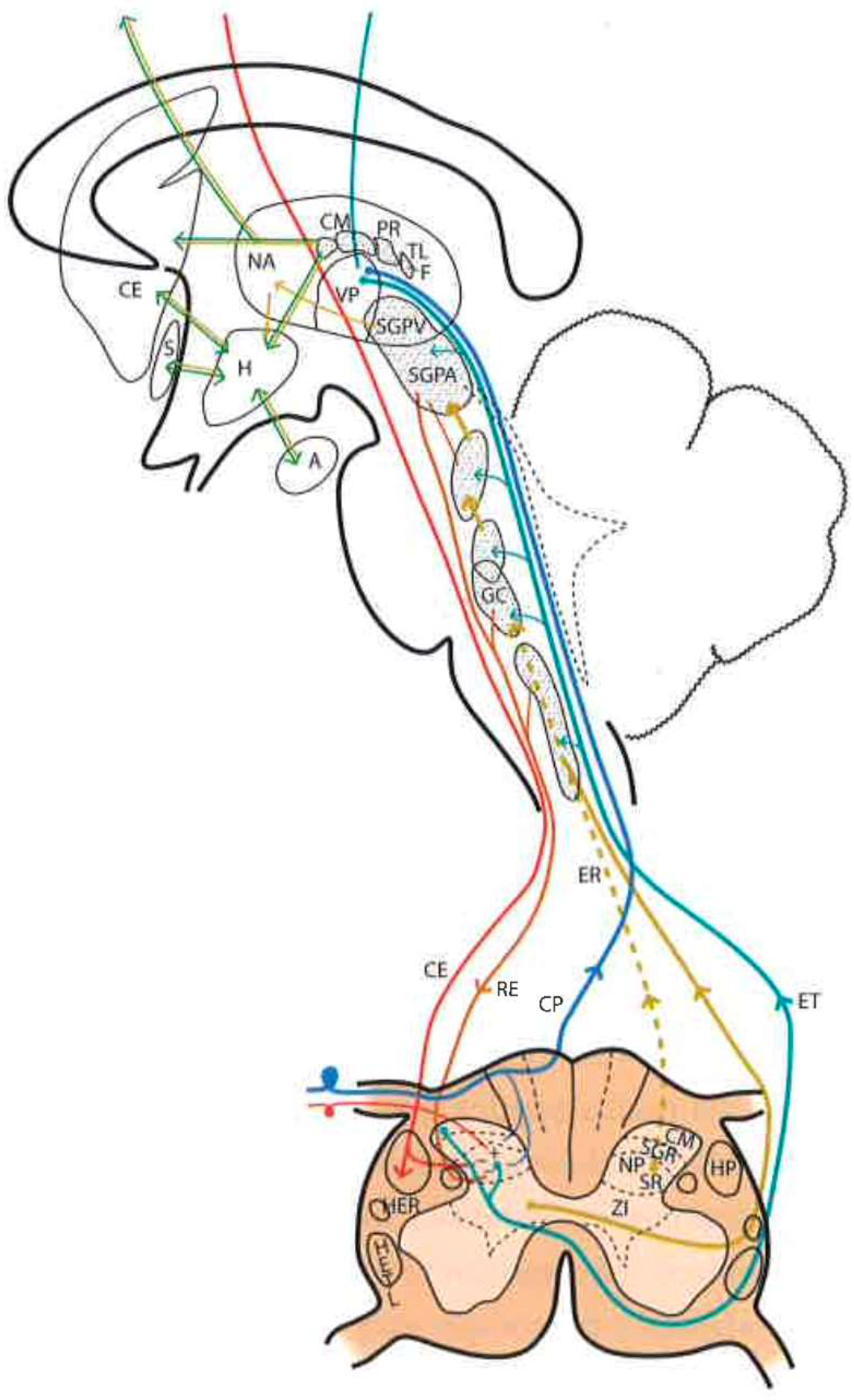
2.5. Brainstem Reticular Formation
- Gigantocellular reticular nucleus and lateral reticular region, at the level of the medulla oblongata and pons—they receive projections from the spinothalamic tract as well as from the deeper areas of spinal cord gray matter [31].
- Ventrolateral PAG and periventricular gray matter (PVG)—lateral to the third ventricle, in the region of the posterior commissure [28], are the areas whose stimulation produces the most effective analgesia with minimal side effects. This SPA [26] may last for several hours after stimulation, suggesting the activation of a multisynaptic system with a mechanism similar to opioid analgesia [32]. Both areas are, indeed, rich in opioid receptors, and their stimulation causes the release of endorphins in CSF. There is, on the other hand, cross-tolerance between SPA and opioid analgesia, just as SPA can be reversed by naloxone [33].
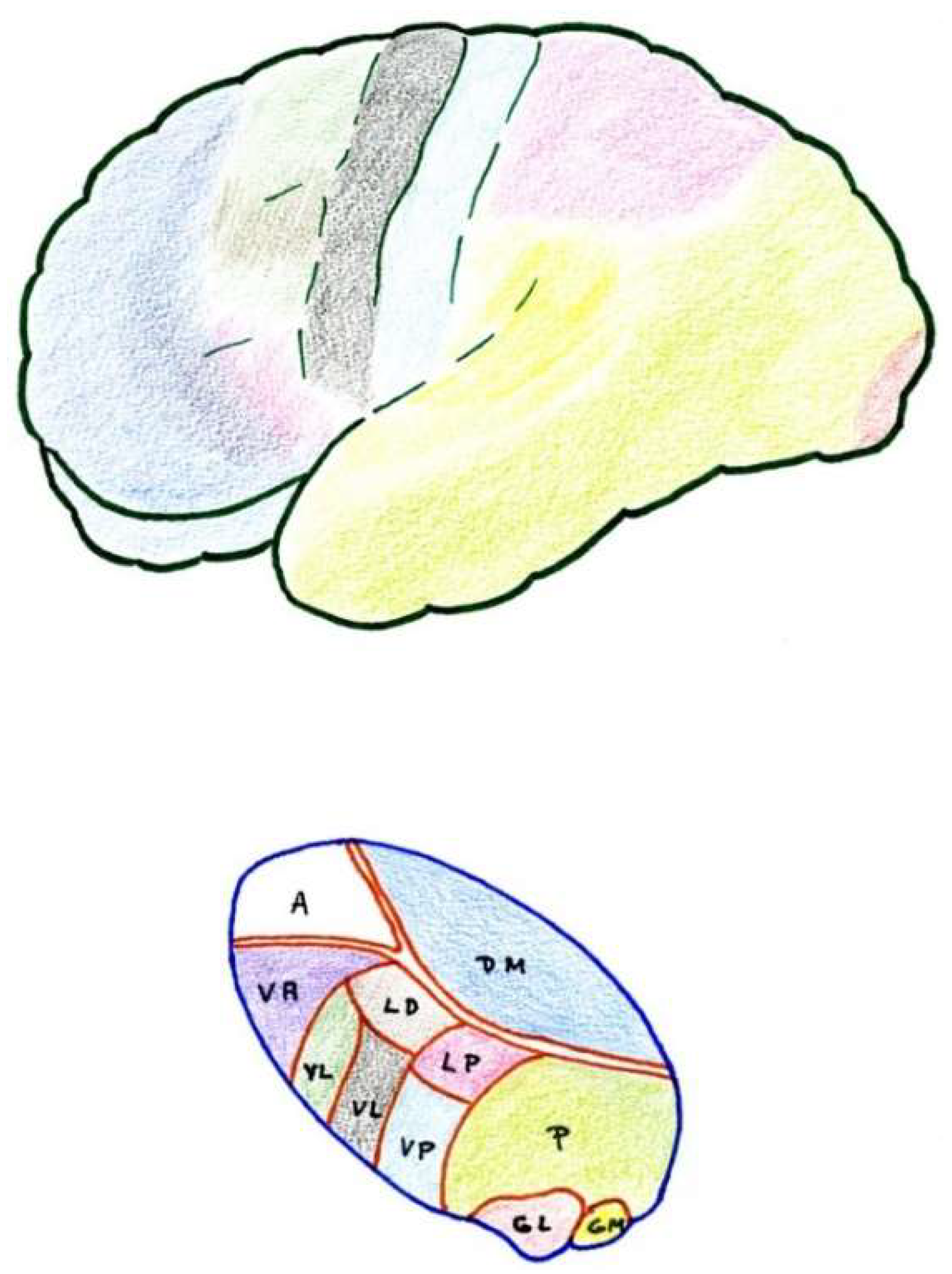
2.6. Thalamus
2.6.1. Ventrobasal Complex
2.6.2. Posterior Complex
2.6.3. Intralaminar Nuclei
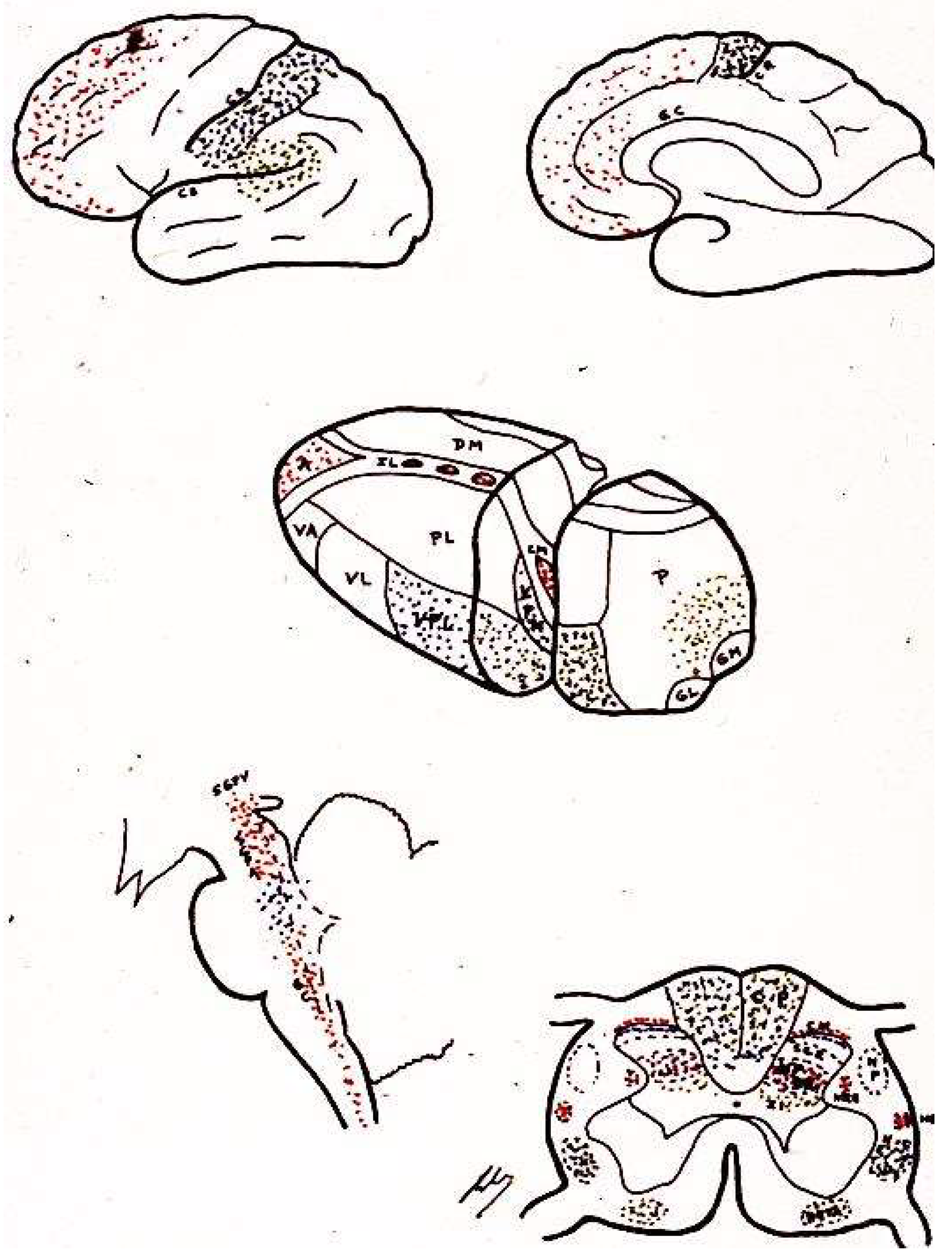
2.7. Cerebral Cortex
2.7.1. Primary
2.7.2. Secondary
2.7.3. Tertiary
2.8. Pain Modulation Pathways
2.8.1. Neurophysiological
2.8.2. Neurochemistry
2.9. Gate Control Theory
3. Pain, from the Clinical Point of View
3.1. Types of Pain
3.1.1. According to Duration: ACUTE or CHRONIC
- Acute—Up to 2 months from onset
- b.
- Subacute—From 2 to 6 months from onset
- c.
- Chronic—From 6 months to 8 years
- d.
- Hyperchronic—3 to 12 years
3.1.2. According to the Mechanisms Involved: NOCICEPTIVE and NEUROPATHIC
3.1.3. According to Life Expectancy: BENIGN and MALIGNANT PAIN
3.2. Pain from Ongoing Nociception
3.2.1. Ascending Pathways
Sensory-Discriminative Component of Pain
- Ia.
- Spinothalamic
- Pain-heat
- Somatotopic distribution
- Ib.
- Posterior spinal cords
- Touch, vibration, discrimination
- Proprioception
- Somatotopic distribution
- II.
- Thalamus
- VPM, VPL, and Po (Posterior Complex)
- III.
- Primary and secondary sensory cortex
Affective-Emotional Component (Unpleasant)
- Ia.
- Spinoreticular tract
- Medullary reticular substance
- Laminae V-VIII
- Does not localize pain
- Acts on autonomic mechanisms
- Ib.
- Spinomesencephalic tract
- PAG
- Somatotopic distribution
- Ic.
- Ventral spinothalamic tract
- VPL and Po (Posterior Complex)
- Id.
- Multisynaptic ascending system
- II.
- Hypothalamus
- III.
- Medial thalamus
- IV.
- Limbic system
3.2.2. Descending Pathways
- Ia.
- Posterior spinal cords
- Cerebral cortex
- Diencephalon
- Ib.
- Spinoreticular tract
- Originates from the PAG
3.2.3. Examples of Chronic Pain Syndromes from Ongoing Nociception
- Low back pain (failed back syndrome)
- Ischemic pain (angina pectoris)
- Coccygodynia
- Myofascial syndromes (fibromyalgia)
- Postoperative (thoracic surgery)
- Cancer pain
- …
3.3. Neuropathic Pain
- Postherpetic neuralgia
- Brachial plexus avulsion
- Phantom limb pain
- Reflex sympathetic dystrophy
- Causalgia
- Spinal cord injury
- Thalamic syndrome
- Cancer pain
- …
3.4. Other Pain Syndromes
- Headaches and migraines
- Essential trigeminal neuralgia
- Secondary trigeminal neuralgia
- Atypical facial neuralgia
- Glossopharyngeal neuralgia
- Arnold’s suboccipital neuralgia
- Other neuralgias
- Orofacial pain
- …
4. Neurosurgical Procedures
4.1. Introduction
4.2. History of Pain Neurosurgery
4.3. Ablative Techniques
4.3.1. Peripheral Neurectomy
4.3.2. Rhizotomy and Ganglionectomy
4.3.3. Sympathectomy
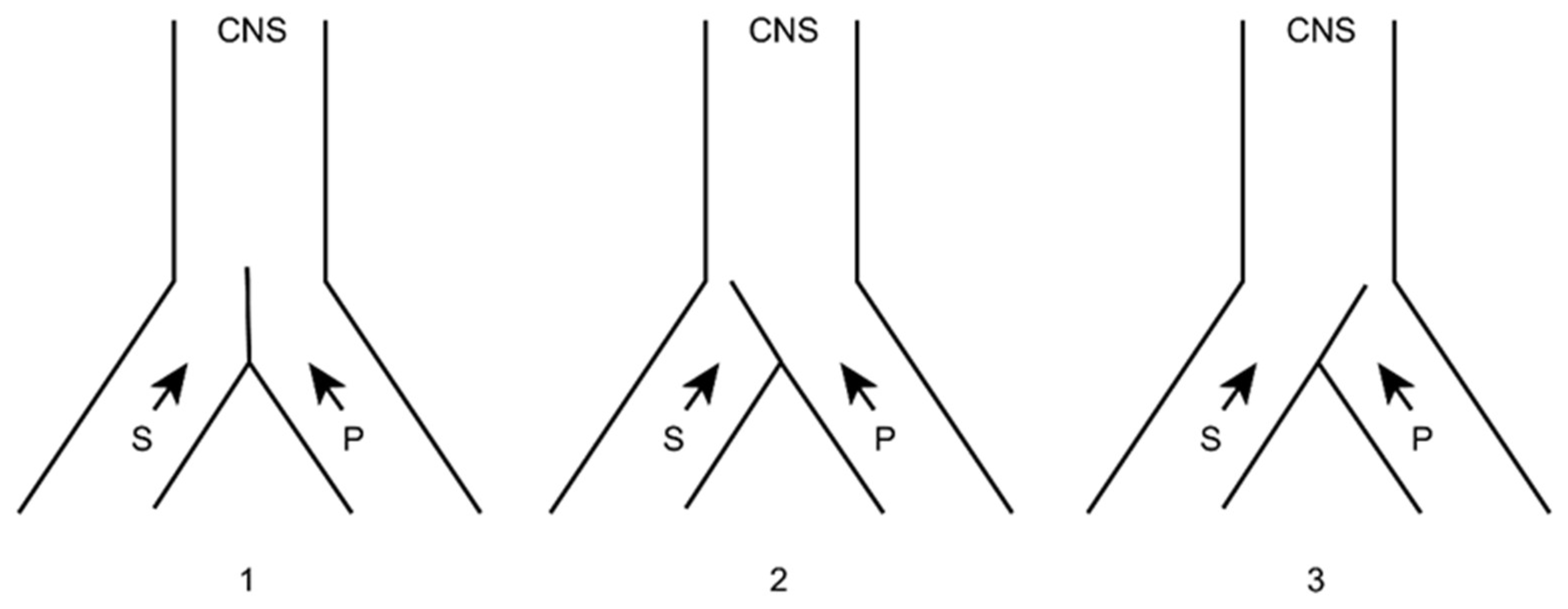
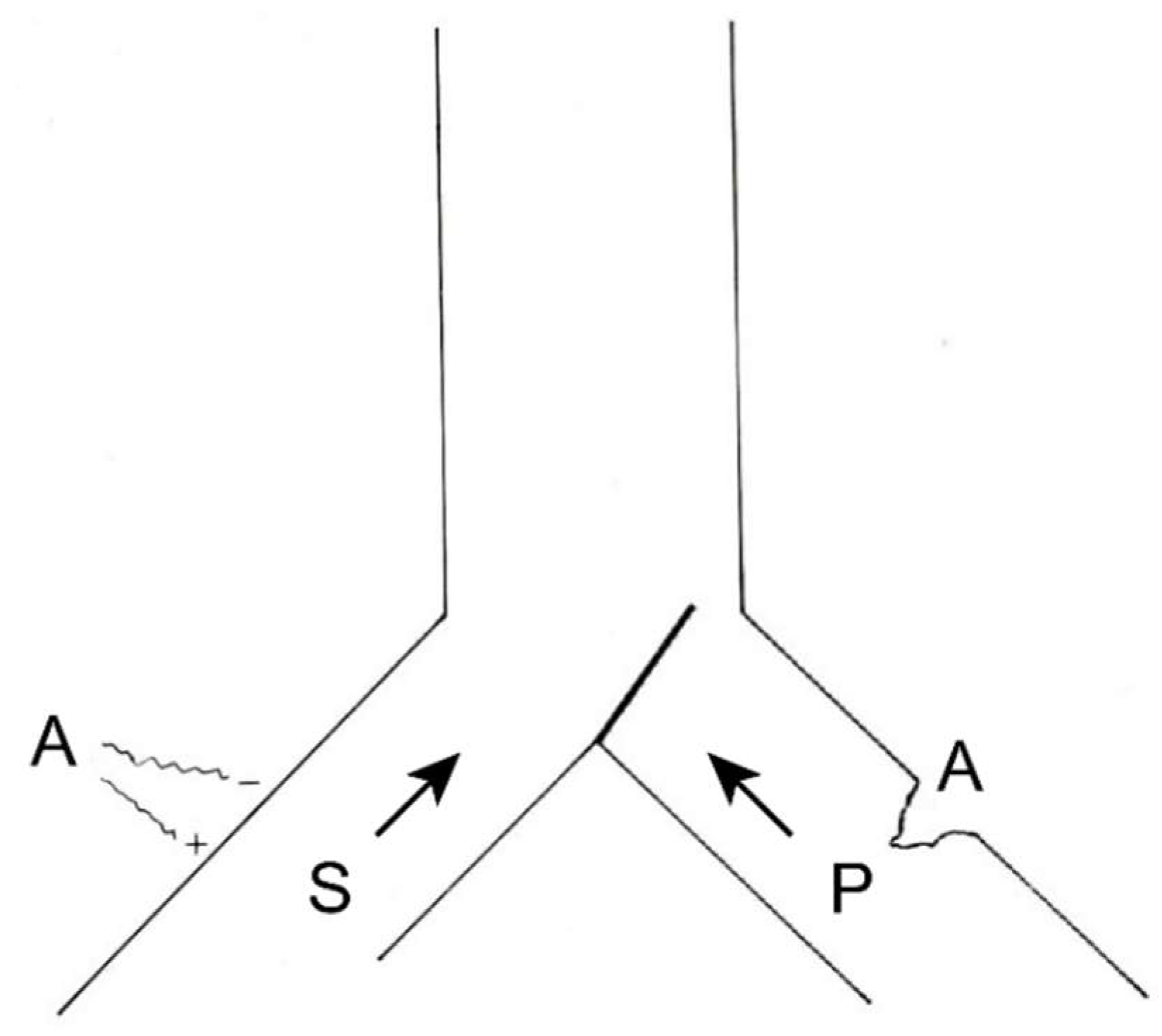
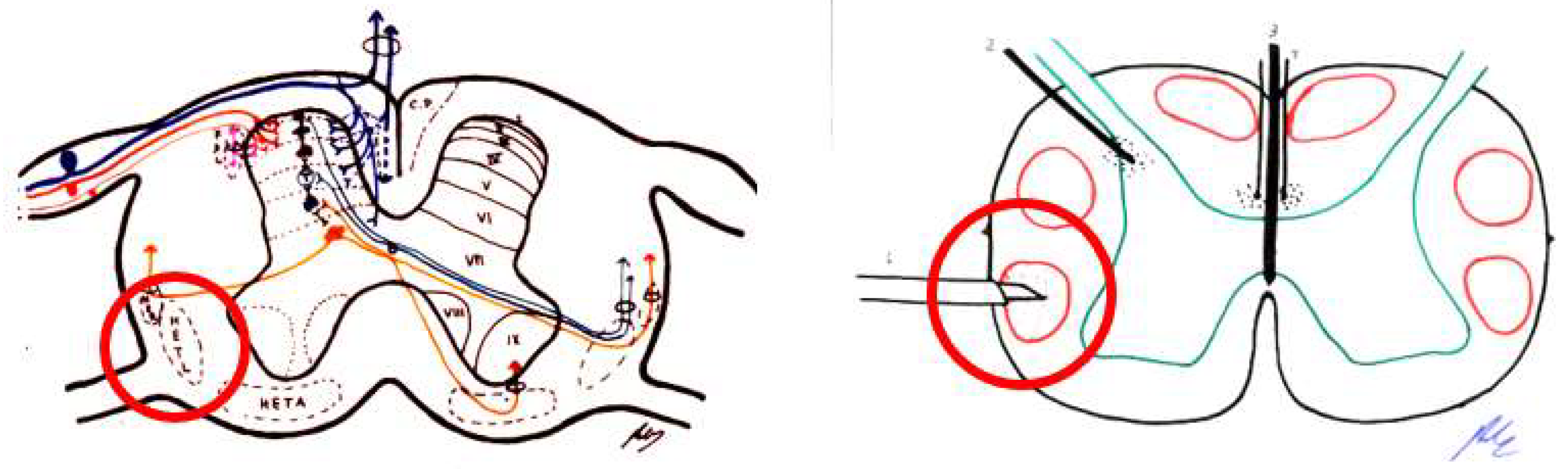
4.3.4. Dorsal Root Entry Zone (DREZ) Lesion
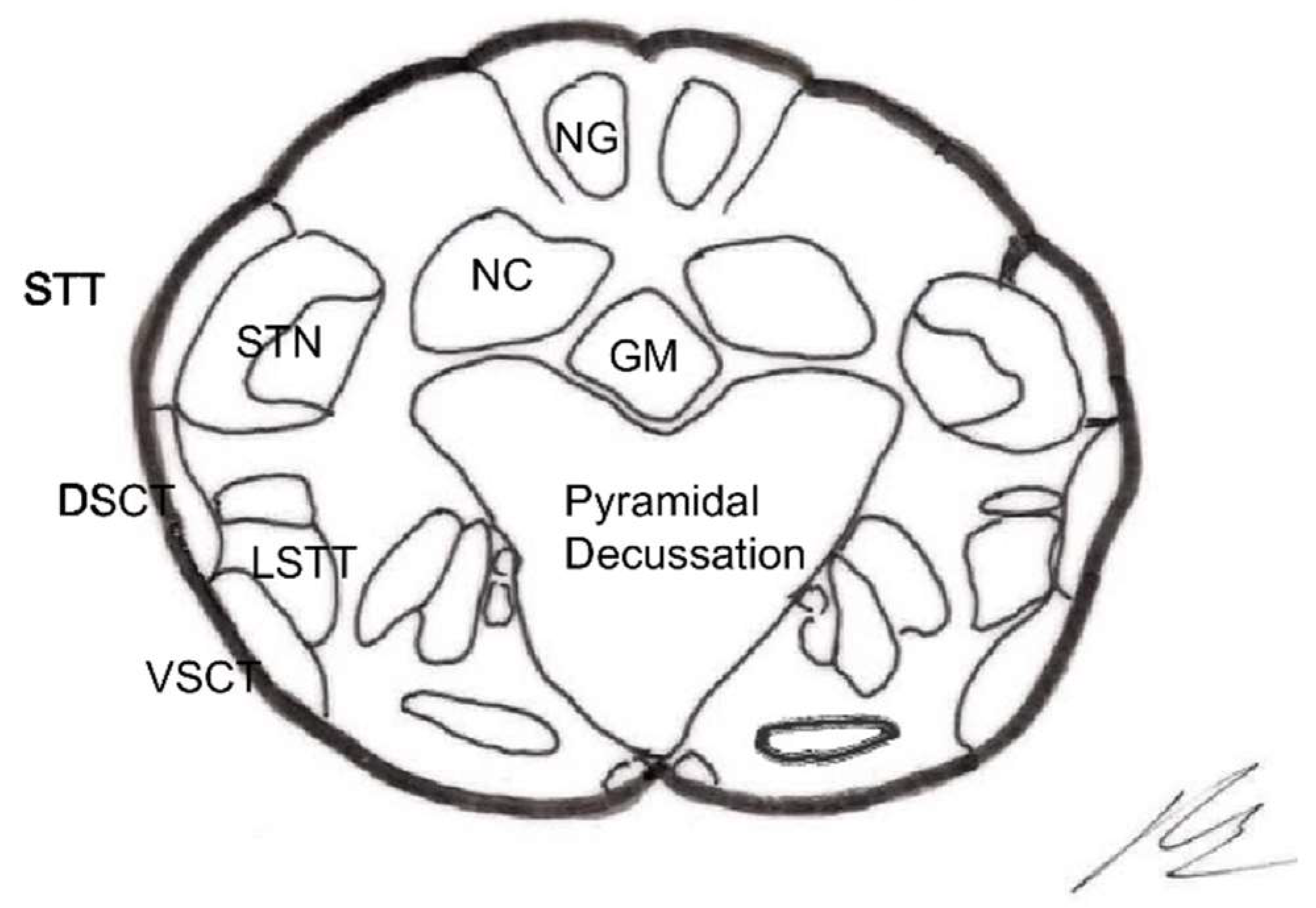
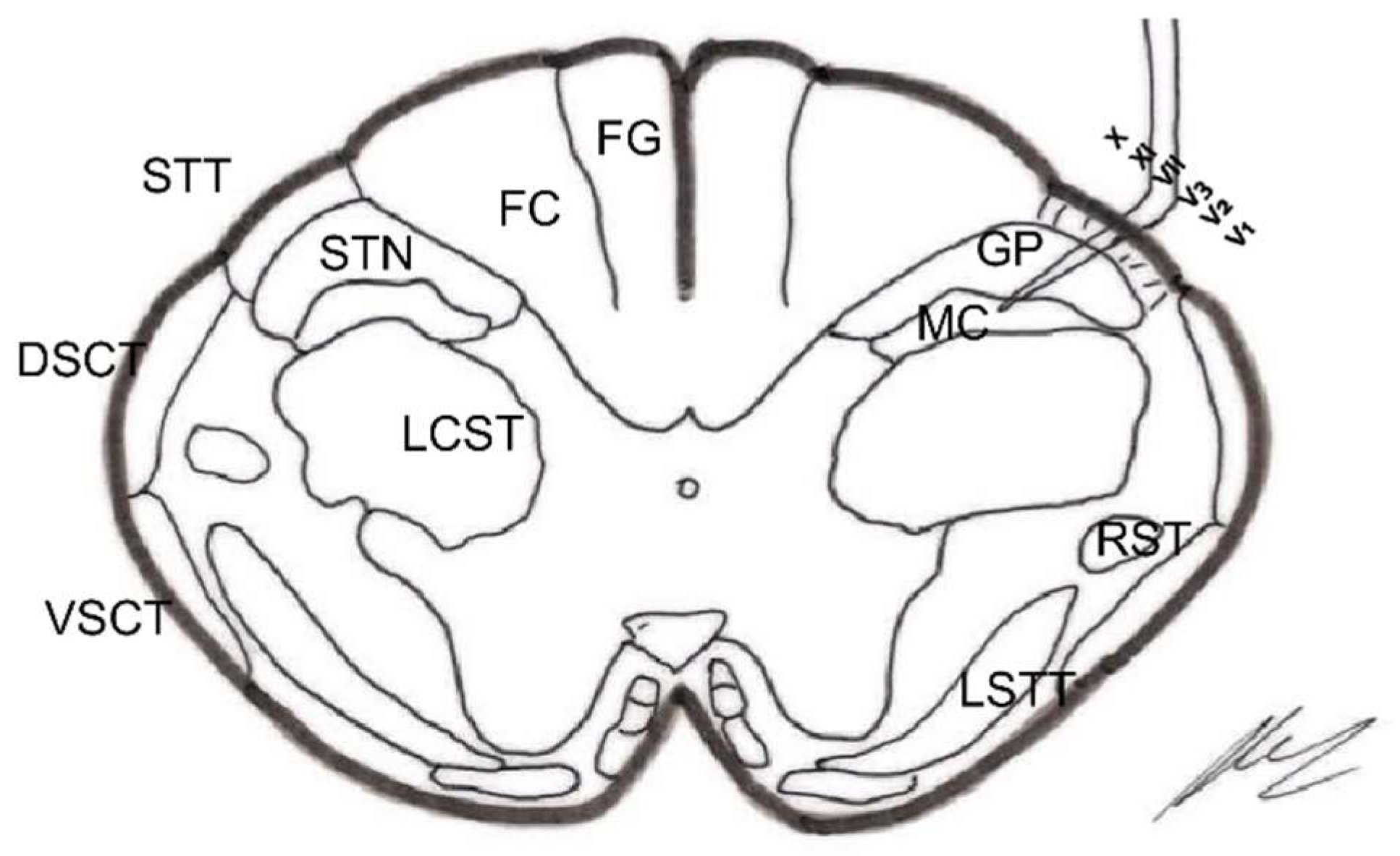
4.3.5. Cordotomy
4.3.6. Myelotomy
Commissural Myelotomy
Extralemniscal Myelotomy
4.3.7. Procedures at the Level of the Brainstem
At the Level of the Trigeminal Complex
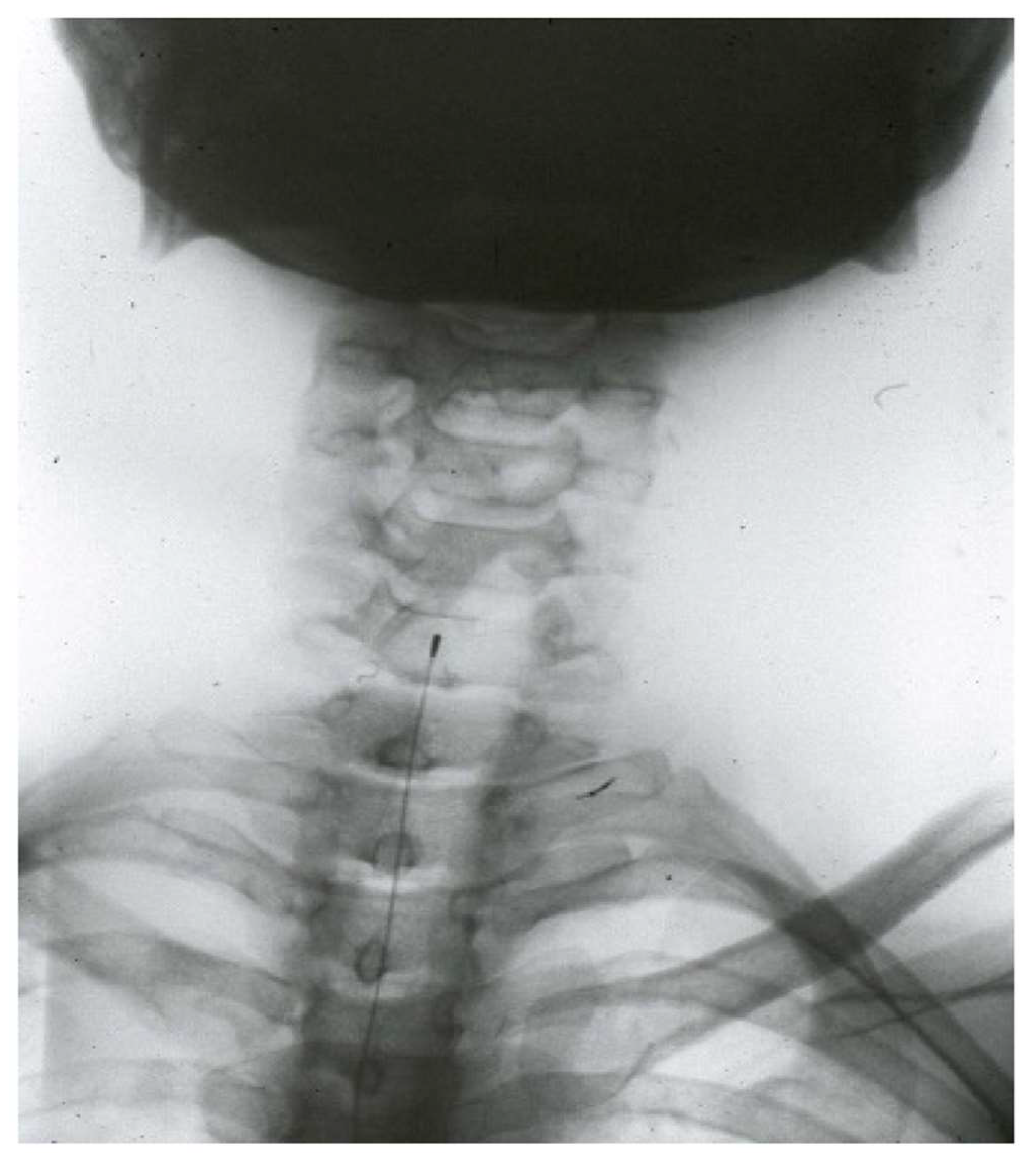
- Trigeminal tractotomy—The trigeminal tract also carries nociceptive afferents from cranial nerves VII, IX, and X. Sjöqvist [119] proposed in 1938 that this tract be severed, just before entering the caudate nucleus. The procedure involves a transverse section, about 8 mm below the obex (Figure 10). It generates ipsilateral hypoalgesia in the face, mouth, pinna, back of the tongue, and pharynx. Hitchcock [115,116], in 1970, described a percutaneous technique.
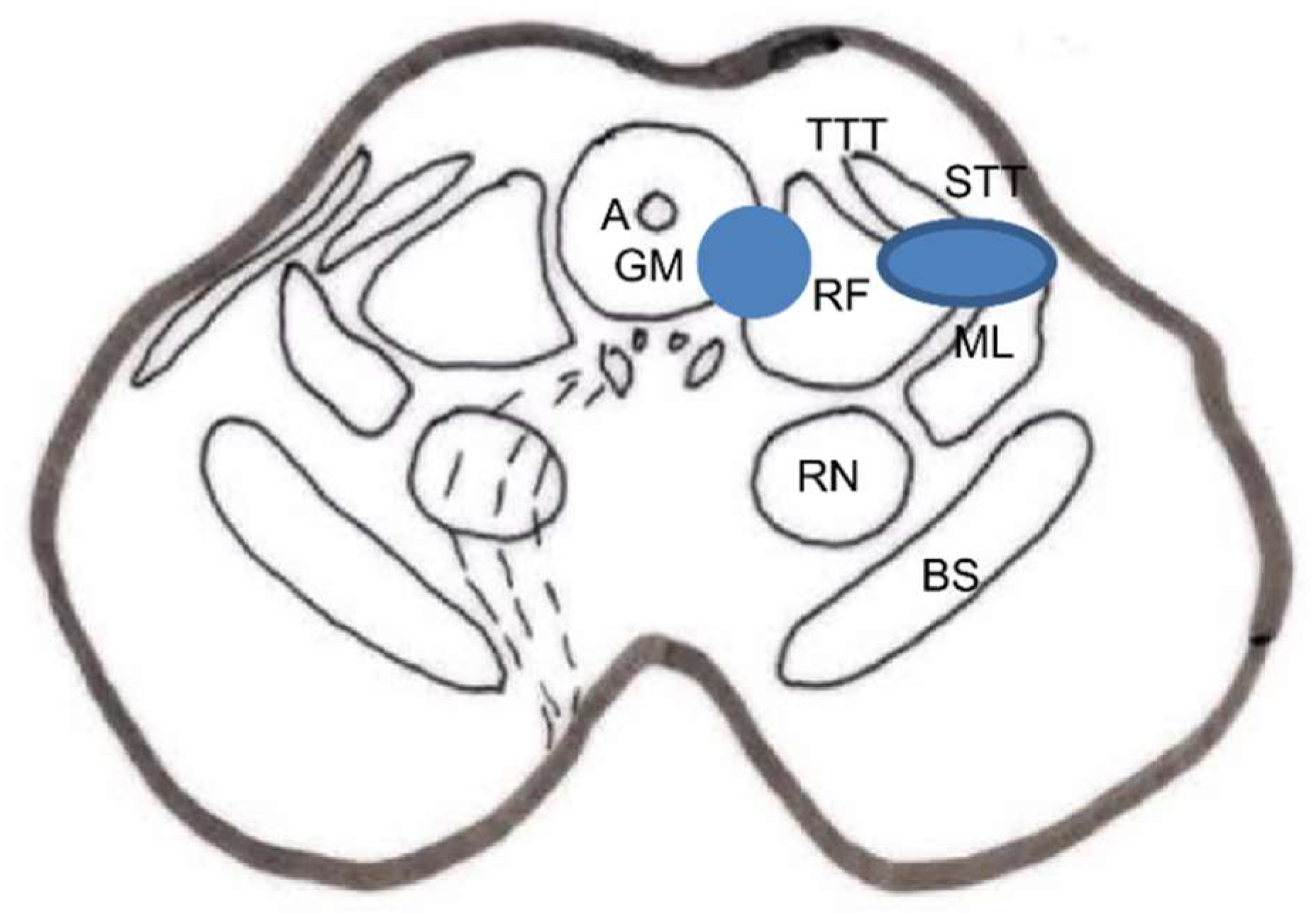
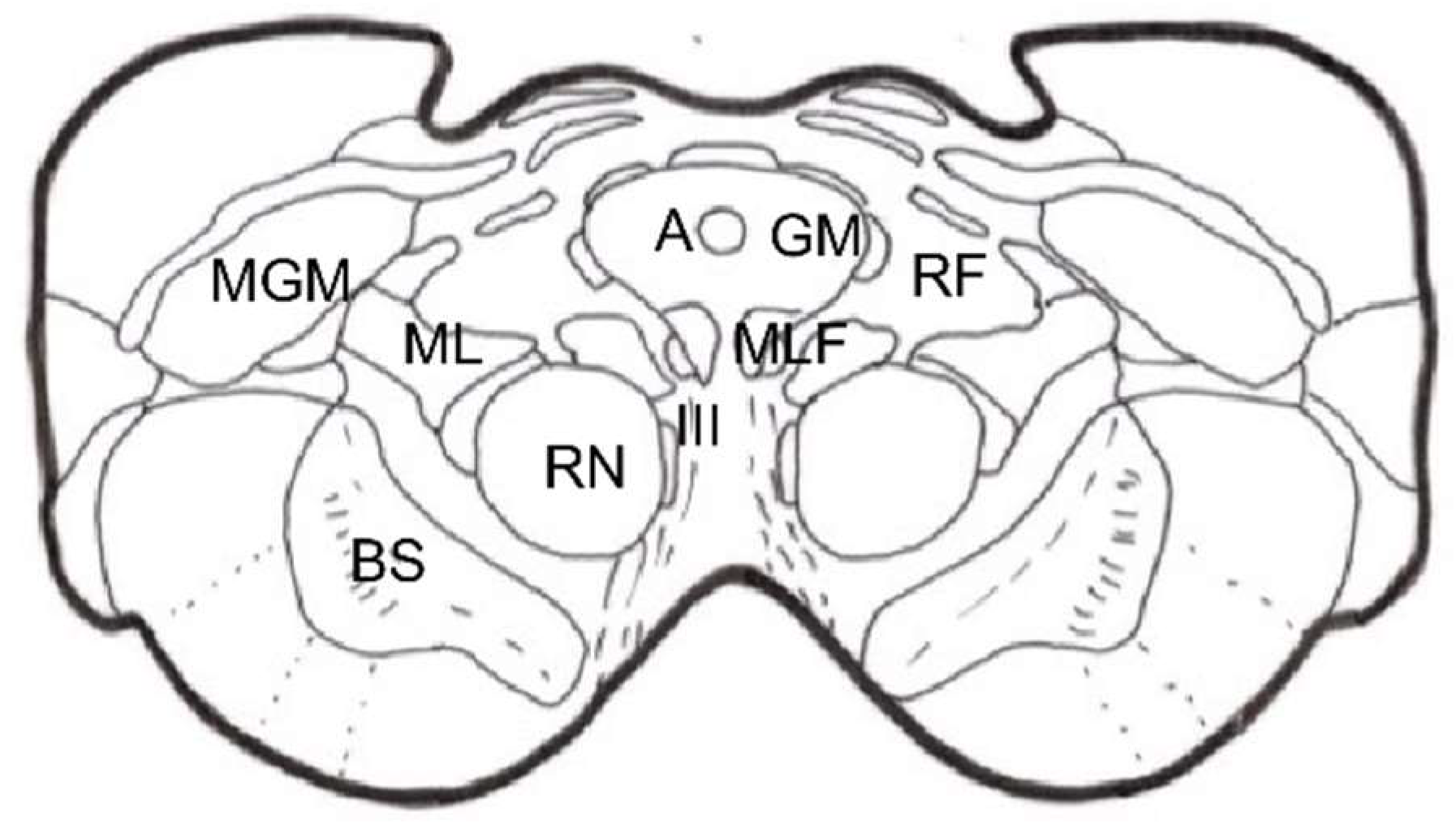
Stereotactic Mesencephalotomy
4.3.8. Cortical-Subcortical Lesions
Medial Thalamotomy
Cingulotomy
Hypophysectomy
4.4. Analgesic Neurostimulation Techniques
4.4.1. Historical Introduction
4.4.2. Transcutaneous Electrical Nerve Stimulation
Mechanism of action of TENS
- Production of a peripheral blockade of the painful stimulus, using an antidromic mechanism [168];
- “Gate control theory” by Melzack and Wall [10];
- Theory of the “central control trigger” by Melzack and Casey, which implies nociceptive information ascending through the posterior spinal cords and triggering a central inhibitory mechanism that, downstream, acts at the level of pain stimulus entry into the dorsal horn;
- Reducing the sympathetic tone [169]; and
- An action mechanism similar to acupuncture, as described by Fox and Melzack [167].
Indications and Results
4.4.3. Peripheral Nerve Stimulation
4.4.4. Chronic, Analgesic Spinal Cord Stimulation (SCS)
Mechanisms of Action of SCS
- Antidromic stimulation of the posterior spinal cord and the theory proposed by Melzack and Wall [74], directly on the inhibitory neurons of the SGR or indirectly through the inhibition of T cells in lamina V.
- Conduction block of the spinothalamic tracts [193].
- Orthodromic firing of the VPL thalamic nucleus and subsequent negative feedback on reticular thalamic nuclei [49].
Indications and Results
- Chronic postoperative low back pain (“failed back syndrome” or “low back pain syndrome”), one of the most common causes of disability in middle-aged people.
- Postamputation pain and phantom limb pain, especially in the lower extremities.
- Other neuritic pain (post-traumatic, diabetes…).
- Peripheral deafferentation pain (postherpetic neuralgia, post-cordotomy pain, and brachial plexus avulsion, fundamentally). In these cases, the statistics are few and contradictory.
- Ischemic pain, which deserves mentioning. SCS in ischemic pain was first proposed by Dooley [189], and later systematized and disseminated by Meglio et al. [201]. SCS not only produced a very significant decrease in pain, but also increased blood flow in the extremities. The explanation for this increase is not yet clear, with several mechanisms proposed [202,203]: (a) antidromic activation of C fibers of the dorsal spinal roots; (b) activation of ascending pathways to higher autonomic centers; and (c) segmental inhibition of sympathetic vasoconstrictor fibers.
- f.
- Malignant pain. The success rate is very low and recent pharmacological advances raise doubts on the indication for SCS in these patients.
- g.
- Other indications. If the type of pain is not included in those for which this modality has greater chances of success, the following conditions described by Long et al. [210] must be met: (a) clear physiological cause of pain; (b) no personality disorders; (c) anxiety and depression, if present, are adequately treated; and (d) previous positive pain control test using TENS.
4.4.5. Analgesic Deep Brain Stimulation (DBS)
Thalamic Nuclei
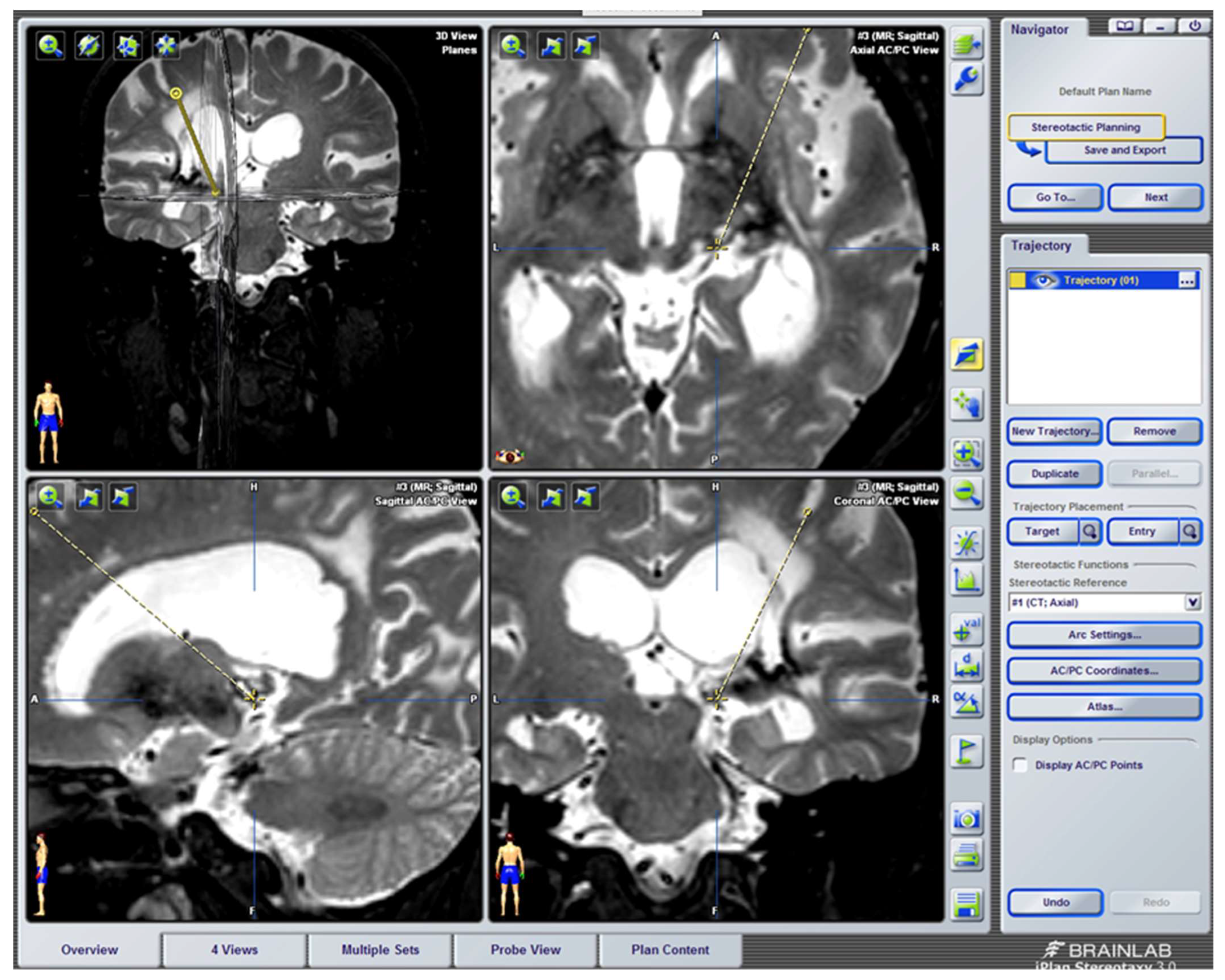
Implantation Technique
Indications and Outcomes
- Deafferentation pain with an extensive area of skin anesthesia. Positive long-term results were achieved in 50% of cases, including lesions of the brachial plexus, postherpetic neuralgia, postrhizotomy or postcordotomy pain, and thalamic syndrome.
- Other deafferentation pain. This includes peripheral nerve injuries (from wounds, crushing, surgery, radiotherapy, etc.), spinal cord or mesencephalic lesions, either vascular or in multiple sclerosis (resulting in hyperpathy-hyperesthesia syndrome), and traumatic paraplegia.
Posterior Limb of the Internal Capsule
Mesencephalic Lemniscus Medialis
Central Gray Matter
Indications and Results
Hypothalamus
Reflections and Current State of Analgesic DBS
- As in the first period, there was a rich basic pathophysiological contribution, simultaneous with published clinical results. In this second stage, there were no great advances in the basic pathophysiological studies advocating analgesic DBS, although reports on clinical advances, based on neuroimaging, have supported what has already been stated.
- 2.
- Regarding the implantation of intracranial electrodes, there have been great advances that have modified the surgical placement techniques and improved intra- and postoperative control. We refer to the use of CT and MRI fusion programs [249] and new neuronavigation software, including the different stereotactic atlases [250]. In addition, improvements in intraoperative neurophysiological exploration, through prior recording with microelectrodes, compared to classic stimulation with implantable macroelectrodes, should also be mentioned [251]. Microelectrode recording and stimulation are very useful at the level of the sensory thalamic nuclei, in which there is a somatotopic distribution; however, it is not as useful as at the PAG-PVG level [250].
- 3.
- DBS was restricted during the two preceding decades to a few centers in Europe and America. Despite this, in the last decade of the last century, more than 1000 patients were treated using this procedure [252].
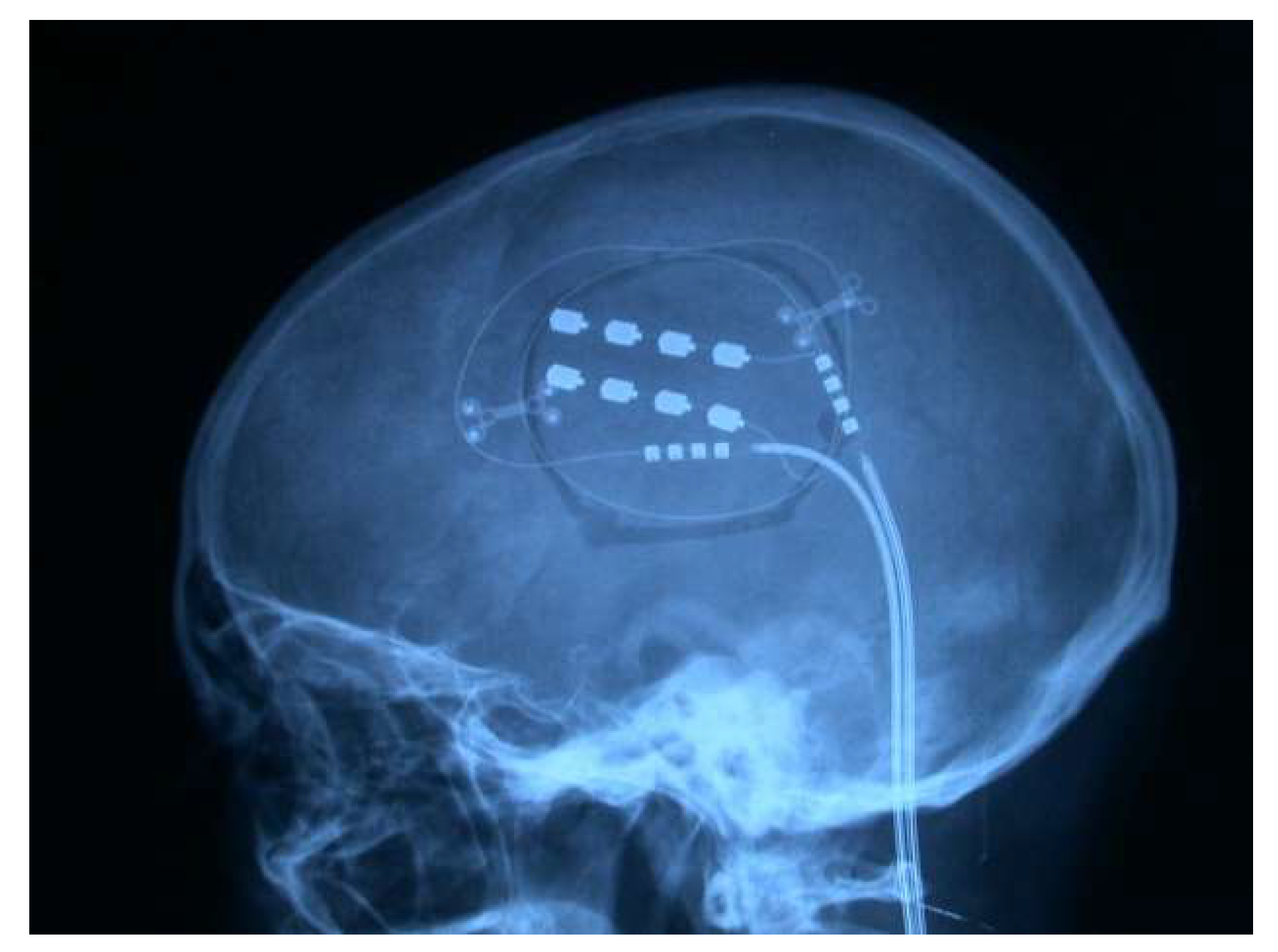
4.4.6. Analgesic Motor Cortex Stimulation (MCS)
Indications and Outcomes
- From the point of view of scientific evidence, the series have better methodological quality than the DBS series [87]. For this procedure, double-blind tests were carried out because patients do not notice anything during stimulation, allowing the treatment to be switched on and off [266,267,268], finding a placebo effect in 35% of patients [269].
- Although it is difficult to summarize all the series, the best responses (up to 70% of patients with more than a 50% reduction in pain intensity) occurred in the following order: facial neuralgia, brachial plexus avulsion, peripheral neuropathies, spinal cord injury, thalamic pain, and phantom limb pain) [266,270,271].
- This technique does not reduce pain to zero for all patients. However, like DBS, it has a very low complication rate, with greater than 60% of patients reporting excellent or good results regarding their pain (reduction of more than 50%, with improved quality of life) [273].
- The mechanism of action involves activating the inhibitory system and modulating the activity of the anterior area of the corpus callosum (pgACC, pregenual anterior cingulate cortex) and the PAG [264].
5. Final Comment
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Bonica, J.J. The need of a taxonomy. Pain 1979, 6, 247–252. [Google Scholar]
- Leriche, R. The Surgery of Pain; Williams & Wilkins: Baltimore, MD, USA, 1939. [Google Scholar]
- Loonen, A.J.M.; Ivanova, S.A. Circuits regulating pleasure and happiness: The evolution of reward-seeking and misery-fleeing behavioral mechanisms in vertebrates. Front. Neurosci. 2015, 9, 394. [Google Scholar] [CrossRef] [PubMed]
- De Ridder, D.; Adhia, D.; Vanneste, S. The anatomy of pain and suffering in the brain and its clinical implications. Neurosci. Biobehav. Rev. 2021, 130, 125–146. [Google Scholar] [CrossRef] [PubMed]
- Bonica, J. Pain; Raven Press: New York, NY, USA, 1980. [Google Scholar]
- Adrian, E. The messages in sensory nerve fibres and their interpretation. Proc. R. Soc. B Biol. Sci. 1931, 109, 1–18. [Google Scholar]
- Baumann, T.K. Physiologic anatomy of nociception. In Surgical Management of Pain; Burchiel, K., Ed.; Thieme: New York, NY, USA, 2002; pp. 2–24. [Google Scholar]
- Bonica, J.J. Causalgia and other reflex sympathetic dystrophy. In The Management of Pain; Bonica, J., Ed.; Lea & Febiger: Philadelphia, PA, USA, 1990; pp. 220–243. [Google Scholar]
- Frey, M.V. Für eine Anatomisch-Physiologische Arbeitsgemeinschaft. Wilhelm. Roux Arch. Entwickl. Mech. Org. 1925, 106, 1–5. [Google Scholar] [CrossRef]
- Melzack, R.; Wall, P.D. Pain mechanisms: A new theory. Science 1965, 150, 971–979. [Google Scholar] [CrossRef]
- Kerr, F.W.L.; Wilson, P.R. Pain. Annu. Rev. Neurosci. 1978, 1, 83–102. [Google Scholar] [CrossRef]
- Löken, L.S.; Wessberg, J.; Morrison, I.; McGlone, F.; Olausson, H. Coding of pleasant touch by unmyelinated afferents in humans. Nat. Neurosci. 2009, 12, 547–548. [Google Scholar] [CrossRef]
- Kulkarni, B.; Bentley, D.E.; Elliott, R.; Youell, P.; Watson, A.; Derbyshire, S.W.G.; Frackowiak, R.S.J.; Friston, K.J.; Jones, A.K.P. Attention to pain localization and unpleasantness discriminates the functions of the medial and lateral pain systems. Eur. J. Neurosci. 2005, 21, 3133–3142. [Google Scholar] [CrossRef]
- Hökfelt, T.; Kellerth, J.O.; Nilsson, G.; Pernow, B. Substance P: Localization in the central nervous system and in some primary sensory neurons. Science 1975, 190, 889–890. [Google Scholar] [CrossRef]
- Randić, M.; Miletić, V. Effect of substance P in cat dorsal horn neurones activated by noxious stimuli. Brain Res. 1977, 128, 164–169. [Google Scholar] [CrossRef]
- Sola, R. Neuromodulación del dolor mediante estimulación eléctrica. In Neurocirugia Funcional y Estereotáctica; Sola, R., Navarrete, E.G., Eds.; Viguera: Barcelona, Spain, 2011; pp. 361–396. [Google Scholar]
- Rexed, B. The cytoarchitectonic organization of the spinal cord in the cat. J. Comp. Neurol. 1952, 96, 415–495. [Google Scholar] [CrossRef] [PubMed]
- Kumazawa, T.; Perl, E.R. Excitation of marginal and substantia gelatinosa neurons in the primate spinal cord: Indications of their place in dorsal horn functional organization. J. Comp. Neurol. 1978, 177, 417–434. [Google Scholar] [CrossRef] [PubMed]
- Kerr, F.W.L. Neuroanatomical substrates of nociception in the spinal cord. Pain 1975, 1, 325–356. [Google Scholar] [CrossRef]
- Kerr, F.W.L. The ventral spinothalamic tract and other ascending systems of the ventral funiculus of the spinal cord. J. Comp. Neurol. 1975, 159, 335–355. [Google Scholar] [CrossRef]
- Iggo, A. Is the physiology of cutaneous receptors determined by morphology? Prog. Brain Res. 1976, 43, 15–31. [Google Scholar]
- Cervero, F.; Molony, V.; Iggo, A. Extracellular and intracellular recordings from neurones in the substantia gelatinosa Rolandi. Brain Res. 1977, 136, 565–569. [Google Scholar] [CrossRef]
- Hillman, P.; Wall, P.D. Inhibitory and excitatory factors influencing the receptive fields of lamina 5 spinal cord cells. Exp. Brain Res. 1969, 9, 284–306. [Google Scholar] [CrossRef]
- Mayer, D.J.; Price, D.D. Central nervous system mechanisms of analgesia. Pain 1976, 2, 379–404. [Google Scholar] [CrossRef]
- Albe-Fessard, D. Electrophysiological methods for the identification of thalamic nuclei. Z. Neurol. 1973, 205, 15–28. [Google Scholar] [CrossRef]
- Trevino, D.L.; Coulter, J.D.; Willis, W.D. Location of cells of origin of spinothalamic tract in lumbar enlargement of the monkey. J. Neurophysiol. 1973, 36, 750–761. [Google Scholar] [CrossRef]
- Kevetter, G.A.; Haber, L.H.; Yezierski, R.P.; Chung, J.M.; Martin, R.F.; Willis, W.D. Cells of origin of the spinoreticular tract in the monkey. J. Comp. Neurol. 1982, 207, 61–74. [Google Scholar] [CrossRef]
- Reynolds, D.V. Surgery in the rat during electrical analgesia induced by focal brain stimulation. Science 1969, 164, 444–445. [Google Scholar] [CrossRef]
- Mayer, D.J.; Hayes, R.L. Stimulation-produced analgesia: Development of tolerance and cross-tolerance to morphine. Science 1975, 188, 941–943. [Google Scholar] [CrossRef]
- Richardson, D.E.; Akil, H. Pain reduction by electrical brain stimulation in man. J. Neurosurg. 1977, 47, 178–183. [Google Scholar] [CrossRef]
- Willis, W.D. The Pain System. The Neural Basis of Nociceptive Transmission in the Mammalian Nervous System; Karger: Basilea, Switzerland, 1985. [Google Scholar]
- Hosobuchi, Y.; Adams, J.E.; Linchitz, R. Pain relief by electrical stimulation of the central gray matter in humans and its reversal by naloxone. Science 1977, 197, 183–186. [Google Scholar] [CrossRef]
- Akil, H.; Mayer, D.J.; Liebeskind, J.C. Antagonism of stimulation-produced analgesia by naloxone, a narcotic antagonist. Science 1976, 191, 961–962. [Google Scholar] [CrossRef]
- Mehler, W. The anatomy of the so-called “pain tract” en man: An analysis of the course and distribution of the ascending fibres of the fasciculus anterolateralis. In Basic Research in Paraplegia; French, J., Porter, R., Eds.; Thomas: Springfield, IL, USA, 1962; pp. 26–55. [Google Scholar]
- Bowsher, D. Role of the reticular formation in responses to noxious stimulation. Pain 1976, 2, 361–378. [Google Scholar] [CrossRef]
- Tasker, R.R.; Emmers, R. The Human Somesthetic Thalamus; Raven Press: New York, NY, USA, 1975. [Google Scholar]
- Hassler, R.; Riechert, T. Clinical and anatomical findings in stereotactic pain operations on the thalamus. Arch. Psychiatr. Nervenkr. Z. Gesamte Neurol. Psychiatr. 1959, 200, 93–122. [Google Scholar] [CrossRef]
- Shigenaga, Y.; Inoki, R. Effect of morphine on single unit responses in ventrobasal complex (VB) and posterior nuclear group (PO) following tooth pulp stimulation. Brain Res. 1976, 103, 152–156. [Google Scholar] [CrossRef]
- Albe-Fessard, D.; Levante, A.; Lamour, Y. Origin of spino-thalamic tract in monkeys. Brain Res. 1974, 65, 503–509. [Google Scholar] [CrossRef]
- Penfield, W.; Jasper, H. Epilepsy and Functional Anatomy of the Human Brain; Little Brown Co.: Boston, MA, USA, 1954. [Google Scholar]
- White, J.; Sweet, W. Pain and the Neurosurgeon. A Forty-Year Experience; Charles C Thomas Publications: Springfield, IL, USA, 1969. [Google Scholar]
- Treede, R.D.; Kenshalo, D.R.; Gracely, R.H.; Jones, A.K.P. The cortical representation of pain. Pain 1999, 79, 105–111. [Google Scholar] [CrossRef]
- Beissner, F.; Meissner, K.; Bär, K.J.; Napadow, V. The autonomic brain: An activation likelihood estimation meta-analysis for central processing of autonomic function. J. Neurosci. 2013, 33, 10503–10511. [Google Scholar] [CrossRef] [PubMed]
- Tolomeo, S.; Christmas, D.; Jentzsch, I.; Johnston, B.; Sprengelmeyer, R.; Matthews, K.; Douglas Steele, J. A causal role for the anterior mid-cingulate cortex in negative affect and cognitive control. Brain 2016, 139, 1844–1854. [Google Scholar] [CrossRef] [PubMed]
- Melzack, R.; Casey, K. Sensory, motivational and central control determinants of pain. A new conceptual model. In The Skin Senses; Kenshalo, D., Ed.; Thomas: Springfield, IL, USA, 1968; pp. 423–439. [Google Scholar]
- Melzack, R. Prolonged relief of pain by brief, intense transcutaneous somatic stimulation. Pain 1975, 1, 357–373. [Google Scholar] [CrossRef]
- Shealy, C.N.; Mortimer, J.T.; Reswick, J.B. Electrical inhibition of pain by stimulation of the dorsal column: Preliminary clinical reports. Anesth. Analg. 1967, 46, 489–491. [Google Scholar] [CrossRef]
- Adams, J.E.; Hosobuchi, Y.; Fields, H.L. Stimulation of internal capsule for relief of chronic pain. J. Neurosurg. 1974, 41, 740–744. [Google Scholar] [CrossRef]
- Bowsher, D. Thalamic convergence and divergence of information generated by noxious stimulation. In Annals of Neurology; Bonica, J., Ed.; Raven Press: New York, NY, USA, 1974; pp. 223–232. [Google Scholar]
- Sheng, H.Y.; Qu, C.L.; Huo, F.Q.; Du, J.Q.; Tang, J.S. D2-like but not D1-like dopamine receptors are involved in the ventrolateral orbital cortex-induced antinociception: A GABAergic modulation mechanism. Exp. Neurol. 2009, 215, 128–134. [Google Scholar] [CrossRef]
- Kong, J.; Loggia, M.L.; Zyloney, C.; Tu, P.; LaViolette, P.; Gollub, R.L. Exploring the brain in pain: Activations, deactivations and their relation. Pain 2010, 148, 257–267. [Google Scholar] [CrossRef]
- Simons, L.E.; Moulton, E.A.; Linnman, C.; Carpino, E.; Becerra, L.; Borsook, D. The human amygdala and pain: Evidence from neuroimaging. Hum. Brain Mapp. 2014, 35, 527–538. [Google Scholar] [CrossRef]
- Henderson, L.A.; Peck, C.C.; Petersen, E.T.; Rae, C.D.; Youssef, A.M.; Reeves, J.M.; Wilcox, S.L.; Akhter, R.; Murray, G.M.; Gustin, S.M. Chronic pain: Lost inhibition? J. Neurosci. 2013, 33, 7574–7582. [Google Scholar] [CrossRef]
- Hughes, J.; Smith, T.W.; Kosterlitz, H.W.; Fothergill, L.A.; Morgan, B.A.; Morris, H.R. Identification of two related pentapeptides from the brain with potent opiate agonist activity. Nature 1975, 258, 577–580. [Google Scholar] [CrossRef]
- Bloom, F.; Segal, D.; Ling, N.; Guillemin, R. Endorphins: Profound behavioral effects in rats suggest new etiological factors in mental illness. Science 1976, 194, 630–632. [Google Scholar] [CrossRef]
- Guillemin, R. The expanding significance of hypothalamic peptides, or, is endocrinology a branch of neuroendocrinology? Recent Prog. Horm. Res. 1976, 33, 1–28. [Google Scholar]
- Akil, H.; Watson, S.J.; Barchas, J.D.; Li, C.H. β-endorphin immunoreactivity in rat and human blood: Radioimmunoassay, comparative levels and physiological alterations. Life Sci. 1979, 24, 1659–1665. [Google Scholar] [CrossRef]
- Basbaum, A.I.; Fields, H.L. Endogenous pain control mechanisms: Review and hypothesis. Ann. Neurol. 1978, 4, 451–462. [Google Scholar] [CrossRef]
- Treede, R.D.; Rief, W.; Barke, A.; Aziz, Q.; Bennett, M.I.; Benoliel, R.; Cohen, M.; Evers, S.; Finnerup, N.B.; First, M.B.; et al. Chronic pain as a symptom or a disease: The IASP Classification of Chronic Pain for the International Classification of Diseases (ICD-11). Pain 2019, 160, 19–27. [Google Scholar] [CrossRef]
- Chapman, C.R.; Vierck, C.J. The transition of acute postoperative pain to chronic pain: An integrative overview of research on mechanisms. J. Pain 2017, 18, 359.e1–359.e38. [Google Scholar] [CrossRef]
- Hendler, N. The four stages of pain. In Diagnosis and Treatment of Chronic Pain; Hendler, N., Long, D., Wise, T.N., Eds.; John Wright PSG Inc.: Boston, MA, USA, 1982; pp. 1–8. [Google Scholar]
- Vachon-Presseau, E.; Martel, M.O.; Roy, M.; Caron, E.; Albouy, G.; Marin, M.F.; Plante, I.; Sullivan, M.J.; Lupien, S.J.; Rainville, P. Acute stress contributes to individual differences in pain and pain-related brain activity in healthy and chronic pain patients. J. Neurosci. 2013, 33, 6826–6833. [Google Scholar] [CrossRef]
- Ikeda, E.; Li, T.; Kobinata, H.; Zhang, S.; Kurata, J. Anterior insular volume decrease is associated with dysfunction of the reward system in patients with chronic pain. Eur. J. Pain 2018, 22, 1170–1179. [Google Scholar] [CrossRef]
- Galambos, A.; Szabó, E.; Nagy, Z.; Édes, A.E.; Kocsel, N.; Juhász, G.; Kökönyei, G. A systematic review of structural and functional MRI studies on pain catastrophizing. J. Pain Res. 2019, 12, 1155–1178. [Google Scholar] [CrossRef] [PubMed]
- Leknes, S.; Berna, C.; Lee, M.C.; Snyder, G.D.; Biele, G.; Tracey, I. The importance of context: When relative relief renders pain pleasant. Pain. 2013, 154, 402–410. [Google Scholar] [CrossRef] [PubMed]
- Meacham, K.; Shepherd, A.; Mohapatra, D.P.; Haroutounian, S. Neuropathic Pain: Central vs. Peripheral Mechanisms. Curr. Pain Headache Rep. 2017, 21, 28. [Google Scholar] [CrossRef] [PubMed]
- Tasker, R. Deafferentation pain syndromes. In Deafferentation Pain Syndromes: Pathophysiology and Treatment; Nashold, B., Ovelmen-Levittt, J., Eds.; Raven Press: New York, NY, USA, 1991; pp. 241–258. [Google Scholar]
- Yezierski, R. Central pain. In Surgical Management of Pain; Burchiel, K., Ed.; Thieme: New York, NY, USA, 2002; pp. 42–64. [Google Scholar]
- Marchet, A. History of a singular nervous or paralytic affection, attended with anomalous morbid sensations. Med. Chir. Tr. 1811, 2, 217–235. [Google Scholar] [CrossRef]
- Behan, R. Pain: Its Origin, Conduction, Perception and Diagnostic Significance; D. Appleton and Company: New York, NY, USA; London, UK, 1914. [Google Scholar]
- Holmes, G. Pain of central origin. In Contributions to Medical and Biological Research; Osles, W., Ed.; Paul B Hoeber: New York, NY, USA, 1919; pp. 235–246. [Google Scholar]
- Riddoch, G. The clinical features of central pain. Lancet 1938, 231, 1093–1098. [Google Scholar] [CrossRef]
- Bonica, J. Management of intractable pain. In New Concepts in Pain and Its Clinical Management; Way, E., Ed.; FA Davis: Philadelphia, PA, USA, 1967; pp. 155–167. [Google Scholar]
- Merskey, H.; Bogduk, N. Classification of Chronic Pain; IASP Press: Seattle, WA, USA, 1994. [Google Scholar]
- Canavero, S.; Bonicalzi, V. Central pain syndrome: Elucidation of genesis and treatment. Expert Rev. Neurother. 2007, 7, 1485–1497. [Google Scholar] [CrossRef]
- Tasker, R.R.; Organ, L.; Hawrylyshyn, P. Deafferentation and causalgia. In Pain; Bonica, J., Ed.; Raven Press: New York, NY, USA, 1980; pp. 305–329. [Google Scholar]
- Tasker, R.; Dostrovsky, J. Deafferentation and central pain. In Textbook of Pain; Wall, P., Melzack, R., Eds.; Churchill-Livingstone: Edinburgh, UK, 1989; pp. 154–180. [Google Scholar]
- Tasker, R.R. Microelectrode findings in the thalamus in chronic pain and other conditions. Stereotact. Funct. Neurosurg. 2001, 77, 166–168. [Google Scholar] [CrossRef]
- Bonica, J. Semantic, epidemiology and educational issues of central pain. In Pain and Central Nervous System Disease: The Central Pain Syndromes; Casey, K., Ed.; Raven Press: New York, NY, USA, 1991; pp. 13–29. [Google Scholar]
- Steel, T.; Burchiel, K. Ablative neurosurgical techniques in the treatment of chronic pain: Overview. In Surgical Management of Pain; Burchiel, K., Ed.; Thieme: New York, NY, USA, 2002; pp. 633–646. [Google Scholar]
- Hammond, D. Do opioids relieve central pain? In Pain and the Central Nervous System Disease: The Central Pain Syndromes; Casey, K., Ed.; Raven: New York, NY, USA, 1991; pp. 233–241. [Google Scholar]
- Horsley, V.; Taylor, J.; Colman, W.S. Remarks on the various surgical procedures devised for the relief or cure of trigeminal neuralgia (tic douloureux). Brit. Med. J. 1891, 2, 1139–1143. [Google Scholar] [CrossRef]
- Dandy, W.E. The Brain; Hoeber Medical Division: New York, NY, USA, 1969. [Google Scholar]
- Young, J.N.; Wilkins, R.H. Partial sensory trigeminal rhizotomy at the pons for trigeminal neuralgia. J. Neurosurg. 1993, 79, 680–687. [Google Scholar] [CrossRef]
- Spiegel, E.A.; Wycis, H.T.; Marks, M.; Lee, A.J. Stereotaxic apparatus for operations on the human brain. Science 1947, 106, 349–350. [Google Scholar] [CrossRef]
- Pedrosa-Sánchez, M.; de Sola, R.G. La moderna psicocirugía: Un nuevo enfoque de la neurocirugía en la enfermedad psiquiátrica. Rev. Neurol. 2003, 36, 887–897. [Google Scholar] [CrossRef] [PubMed]
- Hernando-Requejo, V.; Gómez, J.P.; Camba, E.G.; de Sola, R.G. Psicocirugía y tratamiento neuroquirúrgico del dolor: Revisión sistemática de la experiencia alcanzada en estos tipos de tratamiento. Rev. Neurol. 2004, 38, 949–956. [Google Scholar] [CrossRef] [PubMed]
- Meyerson, B.A.; Mindus, P. The role of anterior internal capsulotomy in psychiatric surgary. In Modern Stereotactic Neurosuergery; Lunsford, L.D., Ed.; Martinus Nijhoff Publishers: Boston, MA, USA, 1988. [Google Scholar]
- Scoville, W.B. Selective cortical undercutting as a means of modifying and studying frontal lobe function in man: Preliminary report of forty-three operative cases. J. Neurosurg. 1949, 6, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Whitty, C.W.; Duffield, J.E.; Tov, P.M.; Cairns, H. Anterior cingulectomy in the treatment of mental disease. Lancet 1952, 1, 475–481. [Google Scholar] [CrossRef]
- Knight, G. Stereotactic tractotomy in the surgical treatment of mental illness. J. Neurol. Neurosurg. Psychiatry 1965, 28, 304–310. [Google Scholar] [CrossRef]
- Kelly, D.; Richardson, A.; Mitchell-Heggs, N. Stereotactic limbic leucotomy: Neurophysiological aspects and operative technique. Br. J. Psychiatry 1975, 123, 133–140. [Google Scholar] [CrossRef]
- Abbe, R. Intradural section of the spinal nerves for neuralgia. Boston Med. Surg. J. 1896, 135, 329–335. [Google Scholar] [CrossRef]
- Leriche, R. La Cirugia del Dolor; Morata Ed.: Madrid, Spain, 1942. [Google Scholar]
- Spiller, W.; Martin, E. The treatment of persistent pain of organic origin in the lower part of the body by division of the anterolateral column of the spinal cord. JAMA 1912, 58, 489–490. [Google Scholar] [CrossRef]
- Armour, D. Lettsonian lecture on the surgery of the spinal cord and its membranes. Lancet 1927, 1, 691–697. [Google Scholar] [CrossRef]
- Spiegel, E.A.; Wycis, H. Mesencephalotomy in the treatment of “intractable” facial pain. AMA Arch. Neurol. Psychiatry 1953, 69, 1–13. [Google Scholar] [CrossRef]
- Penn, R.D.; Paice, J.A. Chronic intrathecal morphine for intractable pain. J. Neurosurg. 1987, 67, 182–186. [Google Scholar] [CrossRef]
- Loeser, J. Intraduction: Ablative neurosurgical operations. In The Management of Pain; Bonica, J., Ed.; Lea and Febiger: Philadelphia, PA, USA, 1990; pp. 2040–2043. [Google Scholar]
- Scoville, W.B. Extradural spinal sensory rhizotomy. J. Neurosurg. 1966, 25, 94–95. [Google Scholar] [CrossRef]
- Uematsu, S. Percutaneous electrothermocoagulation of spinal nerve trunk, ganglion and rootlets. In Operative Neurosurgical Techniques: Indications, Methods and Resuls; Smidek, H., Sweet, W., Eds.; Grune & Stratton: New York, NY, USA, 1988; pp. 1207–1221. [Google Scholar]
- Taha, J. Dorsal root ganglionectomy and dorsal rhizotomy. In Surgical Management of Pain; Burchiel, K., Ed.; Thieme: New York, NY, USA, 2002; pp. 677–687. [Google Scholar]
- Straube, S.; Derry, S.; Moore, R.A.; McQuay, H.J. Cervico-thoracic or lumbar sympathectomy for neuropathic pain and complex regional pain syndrome. Cochrane Database Syst. Rev. 2010, CD002918. [Google Scholar] [CrossRef]
- Sindou, M. Etude de la Jonction Radiculo-Médullaire Postérieure: La Radicellotomie Postérieure Sélective Dans la Chirurgie de la Douleur. Ph.D. Thesis, University Claude-Bernard, Lyon, France, 1972. [Google Scholar]
- Sindou, M. Microsurgical DREZotomy. In Operative Neurosurgical Techniques; Smideck, H., Sweet, W.H., Eds.; WP Saunders: Philadelphia, PA, USA, 1995; pp. 1613–1622. [Google Scholar]
- Sindou, M. Dorsal root entry zone lesions. In Surgical Management of Pain; Burchiel, K., Ed.; Thieme: New York, NY, USA, 2002; pp. 701–713. [Google Scholar]
- Nashold, B.S., Jr.; Urban, B.; Zorub, D.S. Phantom relief by focal destruction of substantia gelatinosa of Rolando. In Advances in Pain Research and Therapy; Bonica, J.J., Albe-Fessard, D., Eds.; Raven Press: New York, NY, USA, 1976; Volume 1, pp. 959–963. [Google Scholar]
- Nashold, B.S.; Mullen, J.B.; Avery, R. Peripheral nerve stimulation for pain relief using a multicontact electrode system. J. Neurosurg. 1979, 51, 872–873. [Google Scholar] [CrossRef]
- Sindou, M.; Turano, G.; Pantieri, R.; Mertens, P.; Mauguière, F. Intraoperative monitoring of spinal cord SEPs during microsurgical DREZotomy (MDT) for pain, spasticity and hyperactive bladder. Stereotact. Funct. Neurosurg. 1994, 62, 164–170. [Google Scholar] [CrossRef]
- Tasker, R. Percutaneous cordotomy. In Operative Neurosurgical Techniques; Schmidek, H., Sweet, W., Eds.; WB Saunders: Philadelphia, PA, USA, 1995; pp. 1595–1612. [Google Scholar]
- Javed, S.; Viswanathan, A.; Abdi, S. Cordotomy for intractable cancer pain: A narrative review. Pain Physician 2020, 23, 283–292. [Google Scholar]
- Bentley, J.N.; Viswanathan, A.; Rosenberg, W.S.; Patil, P.G. Treatment of medically refractory cancer pain with a combination of intrathecal neuromodulation and neurosurgical ablation: Case series and literature review. Pain Med. 2014, 15, 1488–1495. [Google Scholar] [CrossRef]
- Šourek, K. Commissural myelotomy. J. Neurosurg. 1969, 31, 524–527. [Google Scholar] [CrossRef]
- Adams, J.; Lippert, R.; Hosobuchi, Y. Commissural mielotomy. In Operative Neurosurgical Techniques: Indications, Methods an Results; Schmidek, S., Ed.; Grune & Stratton: New York, NY, USA, 1982; pp. 1155–1161. [Google Scholar]
- Hitchcock, E. Stereotactic cervical myelotomy. J. Neurol. Neurosurg. Psychiatry 1970, 33, 224–230. [Google Scholar] [CrossRef][Green Version]
- Hitchcock, E. Stereotactic trigeminal tractotomy. Ann. Clin. Res. 1970, 2, 131–135. [Google Scholar]
- Schvarcz, J. Stereotactic high cervical extralemniscal myelotomy for pelvic cancer pain. Acta Neurochir. Suppl. 1984, 33, 431–435. [Google Scholar]
- Nauta, H.; Westlund, K.; Willis, W. Midline myelotomy. In Surgical Management of Pain; Burchiel, K., Ed.; Thieme: New York, NY, USA, 2002; pp. 714–731. [Google Scholar]
- Sjöqvist, O. Studies on Pain Conduction in the Trigeminal Nerve: A Contribution to the Surgical Treatment of Facial Pain; Mercators Tryckeri: Helsingfors, Finland, 1938; pp. 1–139. [Google Scholar]
- Kanpolat, Y. Percutaneous stereotactic pain procedures. In Surgical Management of Pain; Burquiel, K., Ed.; Thieme: New York, NY, USA, 2002; pp. 745–762. [Google Scholar]
- Nashold, B.S.; El-Naggar, A.; Abdulhak, M.; Ovelmen-Levitt, J.; Cosman, E. Trigeminal nucleus caudalis dorsal root entry zone: A new surgical approach. Stereotact. Funct. Neurosurg. 1992, 59, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Haines, D.E. Neuroanatomy: An Atlas of Structures, Sections, and Systems; Williams & Wilkins Lippincott: Philadelphia, PA, USA, 2004. [Google Scholar]
- El-Naggar, A.; Nashold, B. Nucleus caudalis DREZ lesions for relief of intractable pain. In Neurosurgery; Rengachary, W., Ed.; Mc Graw Hill Book Co.: New York, NY, USA, 1995; pp. 4047–4054. [Google Scholar]
- Gorecki, J.; Rubin, L. Caudalis dorsal root entry zone, nucleotomy and tractotomy. In Surgical Management of Pain; Burchiel, K., Ed.; Thieme: New York, NY, USA, 2002; pp. 763–785. [Google Scholar]
- Bernard, E.J.; Nashold, B.S.; Caputi, F.; Moossy, J.J. Nucleus caudalis DREZ lesions for facial pain. Br. J. Neurosurg. 1987, 1, 81–91. [Google Scholar] [CrossRef]
- Young, R. Comentario al capítulo de Gorecki JP y Rubin LL. Caudalis dorsal root entry zone, nucleotomy and tractotomy. In Surgical Management of Pain; Burchiel, K., Ed.; Thieme: New York, NY, USA, 2002; p. 785. [Google Scholar]
- Walker, A.E. Relief of pain by mesencephalic tractotomy. Arch. Neurol. Psychiatry 1942, 48, 865–883. [Google Scholar] [CrossRef]
- Nashold, B. Brainstem stereotaxic procedures. In Stereotaxy of the Human Brain; Schaltenbrand, G., Walker, A., Eds.; Thieme: New York, NY, USA, 1982; pp. 475–483. [Google Scholar]
- Gildenberg, P. Mesencephalotomy. In Surgical Management of Pain; Burchiel, K., Ed.; Thieme: New York, NY, USA, 2002; pp. 786–794. [Google Scholar]
- Spiegel, E.A.; Kletzkin, M.; Szekely, E.G. Pain reactions upon stimulation of the tectum mesencephali. J. Neuropathol. Exp. Neurol. 1954, 13, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Amano, K. Destructive central lesions for persistent pain. In Textbook of Stereotactic and Functional Neurosurgery; Gildenberg, P., Tasker, R., Eds.; McGraw-Hill: New York, NY, USA, 1998; pp. 1425–1429. [Google Scholar]
- Kim, D.R.; Lee, S.W.; Son, B.C. Stereotactic mesencephalotomy for cancer—Related facial pain. J. Korean Neurosurg. Soc. 2014, 56, 71–74. [Google Scholar] [CrossRef]
- Gildenberg, P.L. Management of chronic pain. Appl. Neurophysiol. 1984, 47, 157–170. [Google Scholar] [CrossRef]
- Jeanmonod, D.; Maguin, M.M. Medial thalamotomy. In Surgical Management of Pain; Burchiel, K., Ed.; Thieme: New York, NY, USA, 2002; pp. 802–804. [Google Scholar]
- Young, R.F.; Jacques, D.S.; Rand, R.W.; Copcutt, B.C.; Vermeulen, S.S.; Posewitz, A.E. Technique of stereotactic medial thalamotomy with the Leksell Gamma Knife for treatment of chronic pain. Neurol. Res. 1995, 17, 59–65. [Google Scholar] [CrossRef]
- Tasker, R. Stereotactic medial thalamotomy. Is it an effective procedure. In Surgical Management of Pain; Burchiel, K., Ed.; Thieme: New York, NY, USA, 2002; pp. 805–809. [Google Scholar]
- Le Beau, J. Anterior cingulectomy in man. J. Neurosurg. 1954, 11, 268–276. [Google Scholar] [CrossRef]
- Abdelaiz, O.; Cosgrove, G. Stereotactic cingulotomy for the treatment of chronic pain. In Surgical Management of Pain; Burchiel, K., Ed.; Thieme: New York, NY, USA, 2002; pp. 812–820. [Google Scholar]
- Foltz, E.L.; White, L.E. Pain “relief” by frontal cingulumotomy. J. Neurosurg. 1962, 19, 89–100. [Google Scholar] [CrossRef]
- Sweet, W. Central mechanisms of chronic pain (neuralgias and other neurogenic pain). In Pain; Bonica, J., Ed.; Raven Press: New York, NY, USA, 1980; pp. 287–303. [Google Scholar]
- Ballantine, H.; Cosgrove, G.; Giriunas, I. Surgical treatment of intractable psychiatric illness and chronic pain by stereotactic cingulotomy. In Operative Neurosurgical Techniques: Indications, Methods and Results; Schmidek, H., Sweet, W., Eds.; Saunders: Philadelphia, PA, USA, 1995; pp. 1423–1430. [Google Scholar]
- Viswanathan, A.; Harsh, V.; Pereira, E.A.C.; Aziz, T.Z. Cingulotomy for medically refractory cancer pain. Neurosurg. Focus 2013, 35, E1. [Google Scholar] [CrossRef] [PubMed]
- Patel, N.V.; Agarwal, N.; Mammis, A.; Danish, S.F. Frameless stereotactic magnetic resonance imaging-guided laser interstitial thermal therapy to perform bilateral anterior cingulotomy for intractable pain: Feasibility, technical aspects, and initial experience in 3 patients. Oper. Neurosurg. 2015, 11, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Boccard, S.G.J.; Prangnell, S.J.; Pycroft, L.; Cheeran, B.; Moir, L.; Pereira, E.A.C.; Fitzgerald, J.J.; Green, A.L.; Aziz, T.Z. Long-term results of deep brain stimulation of the anterior cingulate cortex for neuropathic pain. World Neurosurg. 2017, 106, 625–637. [Google Scholar] [CrossRef] [PubMed]
- Perrault, M.; Lebeau, J.; Klotz, B.; Sicard, J.; Clavel, B. L’hypophysectomie totale dans le traitment du cancer du sein: Premier cas francais: Avenir de la method. Therapie 1952, 7, 290–300. [Google Scholar]
- Luft, R.; Olivecrona, H. Experiences with hypophysectomy in man. J. Neurosurg. 1953, 10, 301–316. [Google Scholar] [CrossRef]
- Talairach, J.; Tournoux, P. Appareil de stéréotaxie hypophysaire pour voie d’abord nasale. Neurochirurgie 1955, 1, 127–131. [Google Scholar]
- West, C.; Avellanosa, A.; Brtemer, A.; Yamada, K. Hypophysectomy for relief of pain of disseminated carcinoma of the prostate. In Advances in Pain Research and Therapy; Bonica, J., Ventafrida, V., Eds.; Raven Press: New York, NY, USA, 1979; pp. 393–400. [Google Scholar]
- Polin, R.; Laws, E.; Shaffrey, M. Hypophysectomy for intractable pain from metastatis carcinoma: A historical perspective. In Surgical Management of Pain; Burchiel, K., Ed.; Thieme: New York, NY, USA, 2002; pp. 821–826. [Google Scholar]
- Sloan, P.A.; Hodes, J.; John, W. Radiosurgical pituitary ablation for cancer pain. J. Palliat. Care 1996, 12, 51–53. [Google Scholar] [CrossRef]
- Hassenbuch, S. Hypophysectomy for intractable pain from metastatis carcinoma: A historical perspective. Commentary. In Surgical Management of Pain; Burchiel, K., Ed.; Thieme: New York, NY, USA, 2002; pp. 826–827. [Google Scholar]
- Kellaway, P. The part placed by electrical fish in the early history of bioelectricity and electrotherapy. Bull. Hist. Med. 1946, 20, 112–137. [Google Scholar]
- Parent, A. Duchenne De Boulogne: A pioneer in neurology and medical photography. Can. J. Neurol. Sci. 2005, 32, 369–377. [Google Scholar] [CrossRef]
- Stillings, D. A survey of the history of electrical stimulation for pain to 1900. Med. Instrum. 1975, 9, 235–259. [Google Scholar]
- Gazdag, G.; Ungvari, G.S. Electroconvulsive therapy: 80 years old and still going strong. World. J. Psychiatr. 2019, 9, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, E.A. Electrical stimulation of sensory nerves with skin electrodes for research, diagnosis, communication and behavioral conditioning: A survey. Med. Biol. Eng. 1968, 6, 637–651. [Google Scholar] [CrossRef]
- Siegfried, J. Introduction historique. In La Neurostimulation Électrique Thérapeutique; Sedan, R., Lazorthes, Y., Eds.; Masson: Paris, France, 1977; pp. 5–10. [Google Scholar]
- Hess, W. Beiträge zur Physiologie des Hirnstammes; Georg Thieme: Leipzig, Germany, 1932. [Google Scholar]
- Wall, P.D.; Sweet, W.H. Temporary abolition of pain in man. Science 1967, 155, 108–109. [Google Scholar] [CrossRef] [PubMed]
- Mazars, G.; Roge, R.; Mazards, Y. Rèsultats de la stimulation du faisceau spino-thalamic et leur incidence sur la physiopathologie de la douleur. Rev. Neurol. 1960, 103, 136–138. [Google Scholar]
- Goldstein, A. Opioid peptides (endorphins) in pituitary and brain. Science 1976, 193, 1081–1086. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.E.; Hosobuchi, Y.; Linchitz, R. The present status of implantable intracranial stimulators for pain. Neurosurgery 1977, 24, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Cook, A.; Weinstein, S. Chronic dorsal column stimulation in multiple sclerosis. N. Y. State J. Med. 1973, 73, 2868–2872. [Google Scholar] [PubMed]
- Cooper, I.S. Effect of chronic stimulation of anterior cerebellum on neurological disease. Lancet 1973, 1, 206. [Google Scholar] [CrossRef]
- Sluka, K.; Smith, H.; Walsh, D. Transcutaneous electric nerve stimulation (TENS): A review. In Neuromodulation; Krames, E., Peckham, P., Rezai, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 335–344. [Google Scholar]
- Melzack, R.; Stillwell, D.M.; Fox, E.J. Trigger points and acupuncture points for pain: Correlations and implications. Pain 1977, 3, 3–23. [Google Scholar] [CrossRef]
- Fox, E.J.; Melzack, R. Transcutaneous electrical stimulation and acupuncture: Comparison of treatment for low-back pain. Pain 1976, 2, 141–148. [Google Scholar] [CrossRef]
- Campbell, J.N.; Taub, A. Local analgesia from percutaneous electrical stimulation. Arch. Neurol. 1973, 28, 347–350. [Google Scholar] [CrossRef] [PubMed]
- Owens, S.; Atkinson, E.R.; David, E.L. Thermographic evidence of reduced sympathetic tone with transcutaneous nerve stimulation. Anesthesiology 1979, 50, 62–64. [Google Scholar] [CrossRef] [PubMed]
- Ebershold, J.; Laws, R.; Stonnington, H.; Stillwell, C. Transcutaneous electrical stimulation for treatment of chronic pain. A preliminary report. Surg. Neurol. 1975, 4, 96–99. [Google Scholar]
- Long, D.M. Cutaneous afferent stimulation for relief of chronic pain. Neurosurgery 1974, 21, 257–268. [Google Scholar] [CrossRef]
- Mannheimer, J. Nonmedicinal and noninvasive pain control techniques in the management of rheumatic disease and related musculoskeletal disorders. J. Rheumatol. Suppl. 1987, 14, 26–32. [Google Scholar]
- Shealy, N.C. Transcutaneous electrical stimulation for control of pain. Neurosurgery 1974, 21, 269–277. [Google Scholar] [CrossRef]
- Gibson, W.; Wand, B.M.; Meads, C.; Catley, M.J.; O’Connell, N.E. Transcutaneous electrical nerve stimulation (TENS) for chronic pain—an overview of cochrane reviews. Cochrane Database Syst. Rev. 2019, 2, CD011890. [Google Scholar]
- Campbell, J.N.; Long, D.M. Peripheral nerve stimulation in the treatment of intractable pain. J. Neurosurg. 1976, 45, 692–699. [Google Scholar] [CrossRef]
- Sweet, W.H. Control of pain by direct electrical stimulation of peripheral nerves. Neurosurgery 1976, 23, 103–111. [Google Scholar] [CrossRef]
- Nielson, K.; Watts, C.; Kempclark, W. Peripheral nerve injury from implantation of chronic stimulating electrodes for pain control. Surg. Neurol. 1976, 5, 51–53. [Google Scholar]
- Nashold, B.S.; Ostdahl, R.H. Dorsal root entry zone lesions for pain relief. J. Neurosurg. 1979, 51, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Stanton-Hicks, M. Peripheral nerve stimulation for pain peripheral neuralgia and complex regional pain syndrome. In Neuromodulation; Krames, E., Peckham, P., Rezai, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 397–407. [Google Scholar]
- Meyerson, B.A.; Håkansson, S. Alleviation of atypical trigeminal pain by stimulation of the Gasserian ganglion via an implanted electrode. Acta Neurochir. Suppl. 1980, 30, 303–309. [Google Scholar] [PubMed]
- Taub, E.; Munz, M.; Tasker, R.R. Chronic electrical stimulation of the gasserian ganglion for the relief of pain in a series of 34 patients. J. Neurosurg. 1997, 86, 197–202. [Google Scholar] [CrossRef]
- Holsheimer, J. Electrical stimulation of the trigeminal tract in chronic, intractable facial neuralgia. Arch. Physiol. Biochem. 2001, 109, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Mekhail, N.A.; Estemalik, E.; Azer, G.; Davis, K.; Tepper, S.J. Safety and efficacy of occipital nerves stimulation for the treatment of chronic migraines: Randomized, double-blind, controlled single-center experience. Pain Pract. 2017, 17, 669–677. [Google Scholar] [CrossRef] [PubMed]
- Weiner, R.L. Occipital neurostimulation (ONS) for treatment of intractable headache disorders. Pain Med. 2006, 7, S137–S139. [Google Scholar] [CrossRef]
- Weiner, R.L.; Alo, K.M. Occipital neurostimulation for treatment of intractable headache síndromes. In Neuromodulation; Krames, E., Peckham, P., Rezai, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 409–416. [Google Scholar]
- Popeney, C.; Alo, K.M. Peripheral neurostimulation for the treatment of chronic, disabling transformed migraine. Headache 2003, 43, 369–375. [Google Scholar] [CrossRef]
- Nashold, B.S.; Friedman, H. Dorsal column stimulation for control of pain. Preliminary report on 30 patients. J. Neurosurg. 1972, 36, 590–597. [Google Scholar] [CrossRef]
- Sweet, W.H.; Wepsic, J.G. Stimulation of the posterior columns of the spinal cord for pain control: Indications, technique, and results. Clin. Neurosurg. 1974, 21, 278–310. [Google Scholar] [CrossRef]
- Dooley, D.M. Spinal cord stimulation. AORN J. 1976, 23, 1209–1212. [Google Scholar] [CrossRef]
- Hosobuchi, Y.; Adams, J.E.; Weinstein, P.R. Preliminary percutaneous dorsal column stimulation prior to permanent implantation. Technical note. J. Neurosurg. 1972, 37, 242–245. [Google Scholar] [CrossRef] [PubMed]
- Ray, C.D. Spinal epidural electrical stimulation for pain control. Practical details and results. Appl. Neurophysiol. 1981, 44, 194–206. [Google Scholar] [PubMed]
- Sedan, R.; Lazorthes, Y. La Neurostimulation Électrique Thérapeutique; Masson: Paris, France, 1977. [Google Scholar]
- Larson, S.J.; Sances, A.; Riegel, D.H.; Meyer, G.A.; Dallmann, D.E.; Swiontek, T. Neurophysiological effects of dorsal column stimulation in man and monkey. J. Neurosurg. 1974, 41, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Mayer, D.J.; Price, D.D.; Becker, D.P. Neurophysiological characterization of the anterolateral spinal cord neurons contributing to pain perception in man. Pain 1975, 1, 51–58. [Google Scholar] [CrossRef]
- Hosobuchi, Y.; Lamb, S.; Bascom, D. Tryptophan loading may reverse tolerance to opiate analgesics in humans: A preliminary report. Pain 1980, 9, 161–169. [Google Scholar] [CrossRef]
- Nielson, K.D.; Adams, J.E.; Hosobuchi, Y. Experience with dorsal column Stimulation for relief of chronic intractable pain 1968– 1973. Surg. Neurol. 1975, 4, 148–152. [Google Scholar] [PubMed]
- Cameron, T. Safety and efficacy of spinal cord stimulation for the treatment of chronic pain: A 20-year literature review. J. Neurosurg. 2004, 100 (Suppl. S3), 254–267. [Google Scholar] [CrossRef]
- Barolat, G. Spinal cord stimulation for painful neuropathies. In Neuromodulation; Krames, E., Peckham, P., Rezai, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 377–384. [Google Scholar]
- Prager, J.P. Neurostimulation in the treatment of complex regional pain syndrome. In Neuromodulation; Krames, E., Peckham, P., Rezai, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 385–395. [Google Scholar]
- Cruccu, G.; Aziz, T.Z.; Garcia-Larrea, L.; Hansson, P.; Jensen, T.S.; Lefaucheur, J.P.; Simpson, B.A.; Taylor, R.S. EFNS guidelines on neurostimulation therapy for neuropathic pain. Eur. J. Neurol. 2007, 14, 952–970. [Google Scholar] [CrossRef]
- Meglio, M.; Cioni, B.; Dal Lago, A.; De Santis, M.; Pola, P.; Serricchio, M. Pain control and improvement of peripheral blood flow following epidural spinal cord stimulation. J. Neurosurg. 1981, 54, 821–823. [Google Scholar] [CrossRef] [PubMed]
- Broseta, J.; Barberá, J.; de Vera, J.A.; Barcia-Salorio, J.L.; Garcia-March, G.; González-Darder, J.; Rovaina, F.; Joanes, V. Spinal cord stimulation in peripheral arterial disease. A cooperative study. J. Neurosurg. 1986, 64, 71–80. [Google Scholar] [CrossRef]
- Linderoth, B.; Foreman, R.D.; Meyerson, B.A. Mechanisms of spinal cord stimulation in neuropathic and ischemic pain syndromes. In Neuromodulation; Krames, E., Peckham, P., Rezai, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 345–354. [Google Scholar]
- Cook, A.W.; Tahmouresie, A.; Oygar, A.; Kleriga, E.; Pacheco, S.; Baggenstos, P.; Nidzgorski, F. Vascular disease of extremities. Electric stimulation of spinal cord and posterior roots. N. Y. State J. Med. 1977, 76, 366–368. [Google Scholar]
- Murphy, D.F.; Giles, K.E. Dorsal column stimulation for pain relief from intractable angina pectoris. Pain 1987, 28, 365–368. [Google Scholar] [CrossRef]
- Melero, A.; Martinez, R.; de Teresa, E.; de Sola, R.G.; Reig, E. Estimulación medular epidural en la angina refractaria al tratamiento convencional. In Estimulación Eléctrica biológica; Barbera, J., Ed.; Servicio de Publicaciones, Universidad de Cadiz: Cadiz, Spain, 1989; pp. 159–169. [Google Scholar]
- Stanik-Hutt, J.A. Management options for angina refractory to maximal medical and surgical interventions. AACN Adv. Crit. Care 2005, 16, 320–323. [Google Scholar] [CrossRef] [PubMed]
- Eckert, S.; Horstkotte, D. Management of angina pectoris: The role of spinal cord stimulation. Am. J. Cardiovasc. Drugs 2009, 9, 16–28. [Google Scholar] [CrossRef]
- DeJongste, M.J.L.; Foreman, R.D. Spinal cord stimulation for refractory angina. In Neuromodulation; Krames, E., Peckham, P., Rezai, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 831–843. [Google Scholar]
- Long, D.M.; Erickson, D.; Campbell, J.; North, R. Electrical stimulation of the spinal cord and peripheral nerves for pain control. A 10-year experience. Appl. Neurophysiol. 1981, 44, 207–217. [Google Scholar]
- Heath, R. Studies in Schizophrenia: A Multidisciplinary Approach to Mind-Brain Relationship; Harvard University Press: Cambridge, MA, USA, 1954. [Google Scholar]
- Pool, J.; Clark, W.; Hudson, P.; Lombardo, M. Hypothalamic-Hypophyseal Interrelationships; Charles C Thomas: Springfield, IL, USA, 1956. [Google Scholar]
- Ervin, F.R.; Brown, C.E.; Mark, V.H. Striatal influence on facial pain. Confin. Neurol. 1966, 27, 75–90. [Google Scholar] [CrossRef]
- Gol, A. Relief of pain by electrical stimulation of the septal area. J. Neurol. Sci. 1967, 5, 115–120. [Google Scholar] [CrossRef]
- Mazars, G.; Mérienne, L.; Ciolocca, C. Intermittent analgesic thalamic stimulation. Preliminary note. Rev. Neurol. 1973, 128, 273–279. [Google Scholar]
- Kumar, K.; Toth, C.; Nath, R.K. Deep brain stimulation for intractable pain: A 15-year experience. Neurosurgery 1997, 40, 736–747. [Google Scholar] [CrossRef]
- Mundinger, F.; Salomão, J.F. Deep brain stimulation in mesencephalic lemniscus medialis for chronic pain. Acta Neurochir. Suppl. 1980, 30, 245–258. [Google Scholar]
- Hosobuchi, Y.; Adams, J.E.; Rutkin, B. Chronic thalamic stimulation for the control of facial anesthesia dolorosa. Arch. Neurol. 1973, 29, 158–161. [Google Scholar] [CrossRef] [PubMed]
- Mazars, G.; Merienne, L.; Cioloca, C. Stimulations thalamiques intermittentes antalgiques. Note preliminaire. Rev. Neurol. 1973, 128, 273–279. [Google Scholar] [PubMed]
- Mazars, G.; Merienne, L.; Cioloca, C. Etat actuel de la chirurgie de la douleur. Neurochirurgie 1976, 22, 1–174. [Google Scholar]
- Adams, J.E. The future of stereotaxic surgery. JAMA 1966, 198, 648–652. [Google Scholar] [CrossRef]
- Adams, J.E.; Hosobuchi, Y. Technique and technical problems. Neurosurgery 1977, 1, 196–199. [Google Scholar] [CrossRef] [PubMed]
- Mazars, G.; Merienne, L.; Cioloca, C. Comparative Study of Electrical Stimulation of Posterior Thalamic Nuclei, Periaqueductal Grey and Other Midline Mesencephalic Structures in Man; Raven Press: New York, NY, USA, 1979; pp. 243–245. [Google Scholar]
- Hosobuchi, Y. Chronic brain stimulation for the treatment of intractable pain. Res. Clin. Stud. Headache 1978, 5, 122–126. [Google Scholar]
- Hosobuchi, Y. The current status of analgesic brain stimulation. Acta Neurochir. Suppl. 1980, 30, 219–227. [Google Scholar]
- Hosobuchi, Y. Combined electrical stimulation of the periaqueductal gray matter and sensory thalamus. Appl. Neurophysiol. 1983, 46, 112–115. [Google Scholar] [CrossRef]
- Hosobuchi, Y. Subcortical electrical stimulation for control of intractable pain in humans. Report of 122 cases (1970–1984). J. Neurosurg. 1986, 64, 543–553. [Google Scholar] [CrossRef]
- Mazars, G.J. Intermittent stimulation of nucleus ventralis posterolateralis for intractable pain. Pain 1975, 4, 93–94. [Google Scholar] [CrossRef]
- Hosobuchi, Y.; Adams, J.E.; Rutkin, B. Chronic thalamic and internal capsule stimulation for the control of central pain. Surg. Neurol. 1975, 4, 91–92. [Google Scholar] [CrossRef]
- Dieckmann, G.; Witzmann, A. Initial and long-term results of deep brain stimulation for chronic intractable pain. Appl. Neurophysiol. 1982, 45, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Siegfried, J.; Shulman, J. Deep brain stimulation. Pacing Clin. Electrophysiol. 1987, 10, 271–272. [Google Scholar] [CrossRef] [PubMed]
- Levy, R.M.; Lamb, S.; Adams, J.E. Treatment of chronic pain by deep brain stimulation: Long term follow-up and review of the literature. Neurosurgery 1987, 21, 885–893. [Google Scholar] [CrossRef]
- Turnbull, I.M.; Shulman, R.; Woodhurst, W.B. Thalamic stimulation for neuropathic pain. J. Neurosurg. 1980, 52, 486–493. [Google Scholar] [CrossRef]
- Hosobuhi, Y.; Adams, J.E. Brain stimulation for the suppression of the intractable pain. Neurol. Surg. 1978, 6, 313–319. [Google Scholar]
- Richardson, D.E.; Akil, H. Long term results of periventricular gray self-stimulation. Neurosurgery 1977, 1, 199–202. [Google Scholar] [CrossRef]
- Meyerson, B.A. Electrostimulation procedures: Effects, presumed rationale, and possible mechanisms. Adv. Pain Res. Ther. 1983, 5, 495–534. [Google Scholar]
- Young, R.F.; Rinaldi, P.C. Brain Stimulation; Springer: New York, NY, USA, 1997. [Google Scholar]
- Leone, M.; Franzini, A.; Bussone, G. Stereotactic stimulation of posterior hypothalamic gray matter in a patient with intractable cluster headache. N. Engl. J. Med. 2001, 345, 1428–1429. [Google Scholar] [CrossRef]
- Leone, M.; Franzini, A.; Broggi, G.; Bussone, G. Hypothalamic stimulation for intractable cluster headache: Long-term experience. Neurology 2006, 67, 150–152. [Google Scholar] [CrossRef]
- Leone, M.; Franzini, A.; Cecchini, A.P.; Broggi, G.; Bussone, G. Hypothalamic deep brain stimulation in the treatment of chronic cluster headache. Ther. Adv. Neurol. Disord. 2010, 3, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Leone, M.; Franzini, A.; Cecchini, A.P.; Bussone, G. Efficacy of hypothalamic stimulation for chronic drug-resistant cluster headache. Cephalalgia 2012, 32, 267–268. [Google Scholar] [CrossRef] [PubMed]
- Schoenen, J.; Di Clemente, L.; Vandenheede, M.; Fumal, A.; De Pasqua, V.; Mouchamps, M.; Remacle, J.M.; de Noordhout, A.M. Hypothalamic stimulation in chronic cluster headache: A pilot study of efficacy and mode of action. Brain 2005, 128, 940–947. [Google Scholar] [CrossRef] [PubMed]
- Bartsch, T.; Pinsker, M.O.; Rasche, D.; Kinfe, T.; Hertel, F.; Diener, H.C.; Tronnier, V.; Mehdorn, H.M.; Volkmann, J.; Deuschl, G.; et al. Hypothalamic deep brain stimulation for cluster headache: Experience from a new multicase series. Cephalalgia 2008, 28, 285–295. [Google Scholar] [CrossRef]
- Brittain, J.S.; Green, A.L.; Jenkinson, N.; Ray, N.J.; Holland, P.; Stein, J.F.; Aziz, T.Z.; Davies, P. Local field potentials reveal a distinctive neural signature of cluster headache in the hypothalamus. Cephalalgia 2009, 29, 1165–1173. [Google Scholar] [CrossRef]
- Sillay, K.A.; Sani, S.; Starr, P.A. Deep brain stimulation for medically intractable cluster headache. Neurobiol. Dis. 2010, 38, 361–368. [Google Scholar] [CrossRef]
- Seijo, F.; Saiz, A.; Lozano, B.; Santamarta, E.; Alvarez-Vega, M.; Seijo, E.; Fernández de León, R.; Fernández-González, F.; Pascual, J. Neuromodulation of the posterolateral hypothalamus for the treatment of chronic refractory cluster headache: Experience in five patients with a modified anatomical target. Cephalalgia 2011, 31, 1634–1641. [Google Scholar] [CrossRef]
- Levy, R.; Deer, T.R.; Henderson, J. Intracranial neurostimulation for pain control. Pain Physician 2010, 13, 157–163. [Google Scholar]
- Hamani, C.; Schwalb, J.M.; Rezai, A.R.; Dostrovsky, J.O.; Davis, K.D.; Lozano, A.M. Deep brain stimulation for chronic neuropathic pain: Long-term outcome and the incidence of insertional effect. Pain 2006, 125, 188–196. [Google Scholar] [CrossRef]
- Richardson, D. Intracranial stimulation therapies: Deep-brain stimulation. In Neurosurgical Pain Management; Follet, K., Ed.; Elsevier: Philadelphia, PA, USA, 2004; pp. 156–159. [Google Scholar]
- Hamani, C.; Fontaine, D.; Lozano, A. BBS for persistent non-cancer pain. In En: Textbook of Stereotactic and Functional Neurosurgery; Lozano, A., Gildenberg, P.L., Tasker, R., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 2227–2238. [Google Scholar]
- Rezai, A.; Lozano, A. Deep brain stimulation for chronic pain. In Surgical Management of Pain; Burchiel, K., Ed.; Thieme: New York, NY, USA, 2002; pp. 565–576. [Google Scholar]
- Pereira, E.A.C.; Moir, L.; Green, A.L.; Aziz, T.Z. Deep brain stimulation for pain. In Neuromodulation; Krames, E., Peckham, P., Rezai, A., Eds.; Elsevier: Oxford, UK, 2009; pp. 499–505. [Google Scholar]
- Owen, S.L.F.; Green, A.L.; Nandi, D.; Bittar, R.G.; Wang, S.; Aziz, T.Z. Deep brain stimulation for neuropathic pain. Neuromodulation 2006, 9, 100–106. [Google Scholar] [CrossRef]
- Awan, N.R.; Lozano, A.; Hamani, C. Deep brain stimulation: Current and future perspectives. Neurosurg. Focus. 2009, 27, E2. [Google Scholar] [CrossRef] [PubMed]
- Katayama, Y.; Yamamoto, T.; Kobayashi, K.; Oshima, H.; Fukaya, C. Deep brain and motor cortex stimulation for post-stroke movement disorders and post-stroke pain. Acta Neurochir. Suppl. 2003, 87, 121–123. [Google Scholar] [PubMed]
- Bittar, R.G.; Kar-Purkayastha, I.; Owen, S.L.; Bear, R.E.; Green, A.; Wang, S.; Aziz, T.Z. Deep brain stimulation for pain relief: A meta-analysis. J. Clin. Neurosci. 2005, 12, 515–519. [Google Scholar] [CrossRef] [PubMed]
- Tsubokawa, T.; Katayama, Y.; Yamamoto, T.; Hirayama, T.; Koyama, S. Chronic motor cortex stimulation for the treatment of central pain. Acta Neurochir. Suppl. 1991, 52, 137–139. [Google Scholar]
- Meyerson, B.A.; Lindblom, U.; Linderoth, B.; Lind, G.; Herregodts, P. Motor cortex stimulation as treatment of trigeminal neuropathic pain. Acta Neurochir. Suppl. 1993, 58, 150–153. [Google Scholar]
- Nguyen, J.; Keravel, Y.; Feve, A. Treatment of deafferentation pain by chronic stimulation of the motor cortex: Report of a series of 20 cases. Acta Neurochir. Suppl. 1997, 68, 54–60. [Google Scholar]
- Tsubokawa, T. Motor cortex stimulation for relief of central deafferentation pain. In Surgical Management of Pain; Burchiel, K., Ed.; Thieme: New York, NY, USA, 2002; pp. 555–574. [Google Scholar]
- Kjerulf, T.D.; Loeser, J.D. Neuronal hyperactivity following deafferentation of the lateral cuneate nucleus. Exp. Neurol. 1973, 39, 70–85. [Google Scholar] [CrossRef]
- Hirayama, T.; Tsubokawa, T.; Katayama, Y. Chronic change in activity of thalamic relay neurons following spinothalamic tractotomy in cat: Effect of motor cortex stimulation. Pain 1990, 5, 273. [Google Scholar] [CrossRef]
- Lenz, F.A.; Kwan, H.C.; Dostrovsky, J.O.; Tasker, R.R. Characteristics of the bursting pattern of action potentials that occurs in the thalamus of patients with central pain. Brain Res. 1989, 496, 357–360. [Google Scholar] [CrossRef]
- Volkers, R.; Giesen, E.; van der Heiden, M.; Kerperien, M.; Lange, S.; Kurt, E.; van Dongen, R.; Schutter, D.; Vissers, K.C.P.; Henssen, D. Invasive motor cortex stimulation influences intracerebral structures in patients with neuropathic pain: An activation likelihood estimation meta-analysis of imaging data. Neuromodulation 2020, 23, 436–443. [Google Scholar] [CrossRef]
- De Ridder, D.; De Mulder, G.; Verstraeten, E.; Sunaert, S.; Moller, A. Somatosensory cortex stimulation for deafferentation pain. Oper. Neuromodulation 2007, 97, 67–74. [Google Scholar]
- Lefaucheur, J.P.; Drouot, X.; Cunin, P.; Bruckert, R.; Lepetit, H.; Creange, A.; Wolkenstein, P.; Maison, P.; Keravel, Y.; Nguyen, J.P. Motor cortex stimulation for the treatment of refractory peripheral neuropathic pain. Brain 2009, 132, 1463–1471. [Google Scholar] [CrossRef] [PubMed]
- Velasco, M.; Velasco, F.; Brito, F.; Velasco, A.L.; Nguyen, J.P.; Marquez, I.; Boleaga, B.; Keravel, Y. Motor cortex stimulation in the treatment of deafferentation pain. I. Localization of the motor cortex. Stereotact. Funct. Neurosurg. 2002, 79, 146–167. [Google Scholar] [CrossRef] [PubMed]
- Velasco, F.; Argüelles, C.; Carrillo-Ruiz, J.D.; Castro, G.; Velasco, A.L.; Jiménez, F.; Velasco, M. Efficacy of motor cortex stimulation in the treatment of neuropathic pain: A randomized double-blind trial. J. Neurosurg. 2008, 108, 698–706. [Google Scholar] [CrossRef] [PubMed]
- Rasche, D.; Ruppolt, M.; Stippich, C.; Unterberg, A.; Tronnier, V.M. Motor cortex stimulation for long-term relief of chronic neuropathic pain: A 10 year experience. Pain 2006, 121, 43–52. [Google Scholar] [CrossRef]
- Nguyen, J.P.; Lefaucheur, J.P.; Raoul, S.; Roualdes, V.; Péréon, Y.; Keravel, Y. Motor cortex stimulation for the treatment of neuropathic pain. In Neuromodulation; Krames, E., Peckham, P., Rezai, A., Eds.; Elsevier: Oxford, UK, 2009; pp. 515–526. [Google Scholar]
- Fontaine, D.; Hamani, C.; Lozano, A. Efficacy and safety of motor cortex stimulation for chronic neuropathic pain: Critical review of the literature. J. Neurosurg. 2009, 110, 251–256. [Google Scholar] [CrossRef]
- Henderson, J.M.; Boongird, A.; Rosenow, J.M.; LaPresto, E.; Rezai, A.R. Recovery of pain control by intensive reprogramming after loss of benefit from motor cortex stimulation for neuropathic pain. Stereotact. Funct. Neurosurg. 2004, 82, 207–213. [Google Scholar] [CrossRef]
- Lima, M.C.; Fregni, F. Motor cortex stimulation for chronic pain: Systematic review and meta-analysis of the literature. Neurology 2008, 70, 2329–2337. [Google Scholar] [CrossRef]
- Tanei, T.; Kajita, Y.; Noda, H.; Takebayashi, S.; Nakatsubo, D.; Maesawa, S.; Wakabayashi, T. Efficacy of motor cortex stimulation for intractable central neuropathic pain: Comparison of stimulation parameters between post-stroke pain and other central pain. Neurol. Med. Chir. 2011, 51, 8–14. [Google Scholar] [CrossRef]
- Medrano-Escalada, Y.; Plaza-Manzano, G.; Fernández de las Peñas, C.; Valera-Calero, J. Structural, functional and neurochemical cortical brain changes associated with chronic low back pain. Tomography 2022, 8, 2153–2163. [Google Scholar] [CrossRef]
- Machado, A.G.; Mogilner, A.Y.; Rezai, A.R. Motor cortex stimulation for persistent non-cancer pain. In Textbook of Stereotactic and Functional Neurosurgery; Lozano, A., Gildenberg, P., Tasker, R., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 2239–2249. [Google Scholar]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sola, R.G.; Pulido, P. Neurosurgical Treatment of Pain. Brain Sci. 2022, 12, 1584. https://doi.org/10.3390/brainsci12111584
Sola RG, Pulido P. Neurosurgical Treatment of Pain. Brain Sciences. 2022; 12(11):1584. https://doi.org/10.3390/brainsci12111584
Chicago/Turabian StyleSola, Rafael G., and Paloma Pulido. 2022. "Neurosurgical Treatment of Pain" Brain Sciences 12, no. 11: 1584. https://doi.org/10.3390/brainsci12111584
APA StyleSola, R. G., & Pulido, P. (2022). Neurosurgical Treatment of Pain. Brain Sciences, 12(11), 1584. https://doi.org/10.3390/brainsci12111584




