Longitudinal Changes in Cognition, Behaviours, and Functional Abilities in the Three Main Variants of Primary Progressive Aphasia: A Literature Review
Abstract
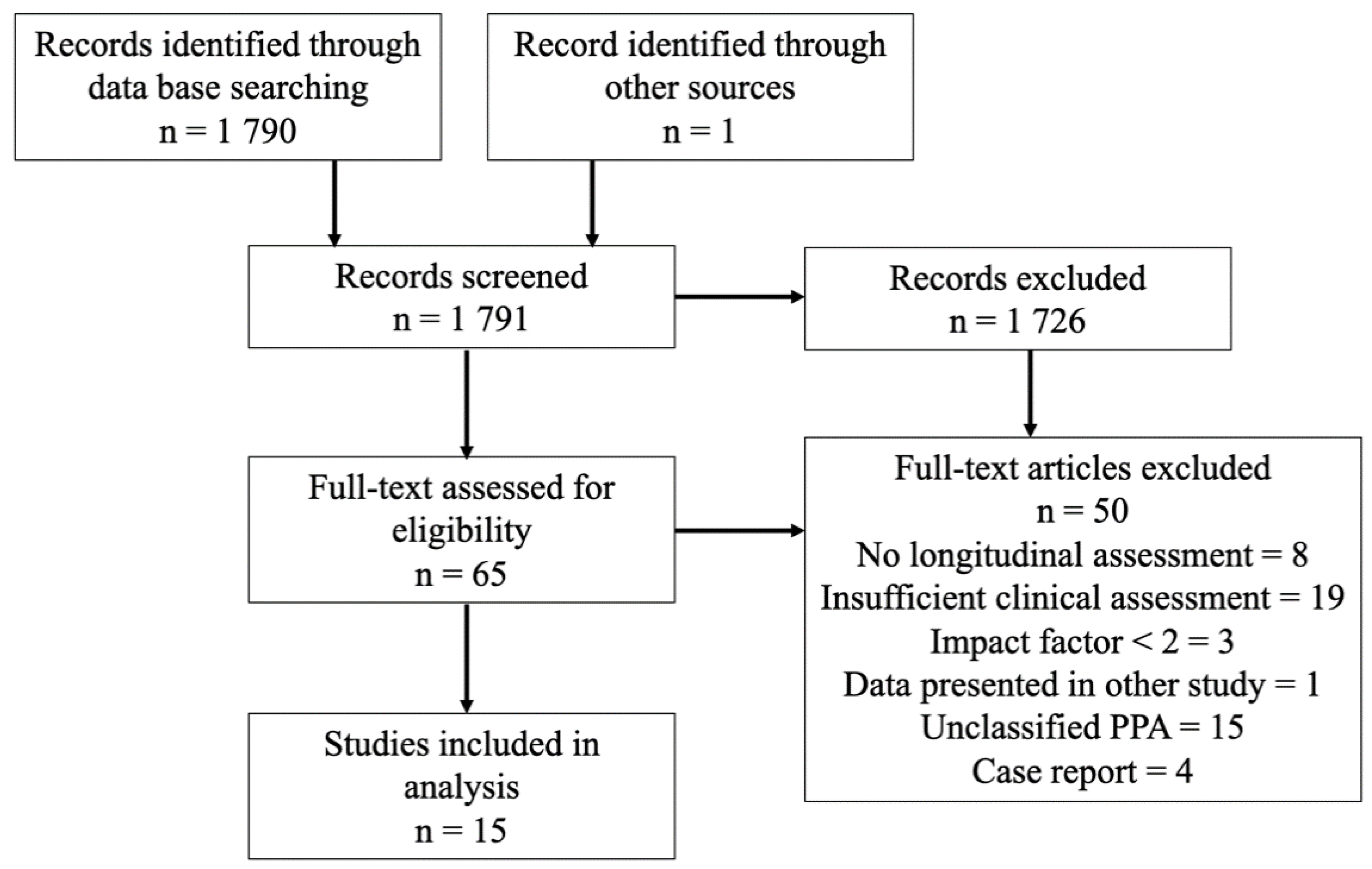
:1. Introduction
2. Materials and Methods
3. Results
Clinical Assessment
4. Discussion
4.1. Similarities between All Three Main Variants of PPA
4.2. Non-Fluent Variant of Primary Progressive Aphasia
4.3. Semantic Variant of Primary Progressive Aphasia
4.4. Logopenic Variant of Primary Progressive Aphasia
4.5. Survival Data
4.6. Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mesulam, M.-M. Primary progressive aphasia. Ann. Neurol. 2001, 49, 425–432. [Google Scholar] [CrossRef]
- Mesulam, M.-M. Slowly progressive aphasia without generalized dementia. Ann. Neurol. 1982, 11, 592–598. [Google Scholar] [CrossRef] [PubMed]
- Snowden, J.; Goulding, P.J.; David, N. Semantic dementia: A form of circumscribed cerebral atrophy. Behav. Neurol. 1989, 2, 167–182. [Google Scholar] [CrossRef]
- Neary, D.; Snowden, J.; Gustafson, L.; Passant, U.; Stuss, D.; Black, S.; Freedman, M.; Kertesz, A.; Robert, P.H.; Albert, M.; et al. Frontotemporal lobar degeneration: A consensus on clinical diagnostic criteria. Neurology 1998, 51, 1546–1554. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gorno-Tempini, M.L.; Dronkers, N.F.; Rankin, K.P.; Ogar, M.J.; Phengrasamy, L.; Rosen, H.J.; Johnson, J.K.; Weiner, M.W.; Miller, B.L. Cognition and anatomy in three variants of primary progressive aphasia. Ann. Neurol. 2004, 55, 335–346. [Google Scholar] [CrossRef] [Green Version]
- Gorno-Tempini, M.L.; Hillis, A.E.; Weintraub, S.; Kertesz, A.; Mendez, M.; Cappa, S.F.; Ogar, J.M.; Rohrer, J.D.; Black, S.; Boeve, B.F.; et al. Classification of primary progressive aphasia and its variants. Neurology 2011, 76, 1006–1014. [Google Scholar] [CrossRef] [Green Version]
- Botha, H.; Josephs, K.A. Primary Progressive Aphasias and Apraxia of Speech. Contin. Lifelong Learn. Neurol. 2019, 25, 101–127. [Google Scholar] [CrossRef]
- Bergeron, D.; Gorno-Tempini, M.L.; Rabinovici, G.D.; Santos-Santos, M.A.; Seeley, W.; Miller, B.L.; Pijnenburg, Y.; Keulen, M.A.; Groot, C.; Van Berckel, B.N.M.; et al. Prevalence of amyloid-β pathology in distinct variants of primary progressive aphasia. Ann. Neurol. 2018, 84, 729–740. [Google Scholar] [CrossRef] [Green Version]
- Botha, H.; Duffy, J.R.; Whitwell, J.L.; Strand, E.A.; Machulda, M.M.; Schwarz, C.; Reid, R.I.; Spychalla, A.J.; Senjem, M.L.; Jones, D.T.; et al. Classification and clinicoradiologic features of primary progressive aphasia (PPA) and apraxia of speech. Cortex 2015, 69, 220–236. [Google Scholar] [CrossRef] [Green Version]
- Perry, D.C.; Datta, S.; Miller, Z.; Rankin, K.P.; Gorno-Tempini, M.L.; Kramer, J.H.; Rosen, H.J.; Seeley, W.W.; Miller, B.L. Factors that predict diagnostic stability in neurodegenerative dementia. J. Neurol. 2019, 266, 1998–2009. [Google Scholar] [CrossRef]
- Volkmer, A.; Rogalski, E.; Henry, M.; Taylor-Rubin, C.; Ruggero, L.; Khayum, R.; Kindell, J.; Gorno-Tempini, M.L.; Warren, J.D.; Rohrer, J.D. Speech and language therapy approaches to managing primary progressive aphasia. Pract. Neurol. 2020, 20, 154–161. [Google Scholar] [CrossRef]
- Fried-Oken, M.; Mooney, A.; Peters, B. Supporting communication for patients with neurodegenerative disease. Neurorehabilitation 2015, 37, 69–87. [Google Scholar] [CrossRef]
- Mesulam, M.-M.; Lalehzari, N.; Rahmani, F.; Ohm, D.; Shahidehpour, R.; Kim, G.; Gefen, T.; Weintraub, S.; Bigio, E.; Geula, C. Cortical cholinergic denervation in primary progressive aphasia with Alzheimer pathology. Neurology 2019, 92, e1580–e1588. [Google Scholar] [CrossRef] [PubMed]
- Davies, K.; Howe, T. Experiences of Living with Primary Progressive Aphasia: A Scoping Review of Qualitative Studies. Am. J. Alzheimer’s Dis. Other Dement. 2020, 35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pozzebon, M.; Douglas, J.; Ames, D. Facing the Challenges of Primary Progressive Aphasia: The Spousal Perspective. J. Speech, Lang. Hear. Res. 2018, 61, 2292–2312. [Google Scholar] [CrossRef]
- Rutherford, S. Our journey with primary progressive aphasia. Aphasiology 2014, 28, 900–908. [Google Scholar] [CrossRef]
- Beales, A.; Bates, K.; Cartwright, J.; Whitworth, A. Lost for words: Perspectives and experiences of people with primary progressive aphasia and Alzheimer’s disease and their families of participation in a lexical retrieval intervention. Int. J. Speech-Lang. Pathol. 2019, 21, 483–492. [Google Scholar] [CrossRef]
- Lavoie, M.; Bier, N.; Laforce, R., Jr.; Macoir, J. Improvement in functional vocabulary and generalization to conversation following a self-administered treatment using a smart tablet in primary progressive aphasia. Neuropsychol. Rehabil. 2020, 30, 1224–1254. [Google Scholar] [CrossRef]
- Volkmer, A.; Spector, A.; Meitanis, V.; Warren, J.; Beeke, S. Effects of functional communication interventions for people with primary progressive aphasia and their caregivers: A systematic review. Aging Ment. Health 2020, 24, 1381–1393. [Google Scholar] [CrossRef]
- Volkmer, A.; Spector, A.; Swinburn, K.; Warren, J.D.; Beeke, S. Using the Medical Research Council Framework for the development of a communication partner training intervention for people with primary progressive aphasia (PPA): Better Conversations with PPA. BMC Geriatr. 2021. [Google Scholar] [CrossRef]
- Jokel, R.; Meltzer, J. Group intervention for individuals with primary progressive aphasia and their spouses: Who comes first? J. Commun. Disord. 2017, 66, 51–64. [Google Scholar] [CrossRef]
- Taylor-Rubin, C.; Azizi, L.; Croot, K.; Nickels, L. Primary Progressive Aphasia Education and Support Groups: A Clinical Evaluation. Am. J. Alzheimer’s Dis. Other Dement. 2020, 35, 1533317519895638. [Google Scholar] [CrossRef] [Green Version]
- Kansal, K.; Mareddy, M.; Sloane, K.L.; Minc, A.A.; Rabins, P.V.; Mcgready, J.B.; Onyike, C.U. Survival in Frontotemporal Dementia Phenotypes: A Meta-Analysis. Dement. Geriatr. Cogn. Disord. 2016, 41, 109–122. [Google Scholar] [CrossRef]
- Rogalski, E.; Cobia, D.; Harrison, T.M.; Wieneke, C.; Weintraub, S.; Mesulam, M.-M. Progression of language decline and cortical atrophy in subtypes of primary progressive aphasia. Neurology 2011, 76, 1804–1810. [Google Scholar] [CrossRef] [Green Version]
- Hsieh, S.; Hodges, J.; Leyton, C.E.; Mioshi, E. Longitudinal Changes in Primary Progressive Aphasias: Differences in Cognitive and Dementia Staging Measures. Dement. Geriatr. Cogn. Disord. 2012, 34, 135–141. [Google Scholar] [CrossRef]
- Leyton, C.E.; Hsieh, S.; Mioshi, E.; Hodges, J.R. Cognitive decline in logopenic aphasia: More than losing words. Neurology 2013, 80, 897–903. [Google Scholar] [CrossRef]
- Linds, A.B.; Kirstein, A.B.; Freedman, M.; Verhoeff, N.P.; Wolf, U.; Chow, T.W. Trajectories of Behavioural Disturbances Across Dementia Types. Can. J. Neurol. Sci. 2015, 42, 389–394. [Google Scholar] [CrossRef] [Green Version]
- Matias-Guiu, J.A.; Cabrera-Martín, M.N.; Moreno-Ramos, T.; García-Ramos, R.; Porta-Etessam, J.; Carreras, J.L.; Matías-Guiu, J. Clinical course of primary progressive aphasia: Clinical and FDG-PET patterns. J. Neurol. 2015, 262, 570–577. [Google Scholar] [CrossRef]
- Gómez-Tortosa, E.; Rigual, R.; Prieto-Jurczynska, C.; Mahíllo-Fernández, I.; Guerrero, R.; Pérez-Pérez, J.; Sainz, M.J. Behavioral Evolution of Progressive Semantic Aphasia in Comparison with Nonfluent Aphasia. Dement. Geriatr. Cogn. Disord. 2016, 41, 1–8. [Google Scholar] [CrossRef]
- O’Connor, C.M.C.; Clemson, L.; Flanagan, E.; Kaizik, C.; Brodaty, H.; Hodges, J.R.; Piguet, O.; Mioshi, E. The Relationship between Behavioural Changes, Cognitive Symptoms, and Functional Disability in Primary Progressive Aphasia: A Longitudinal Study. Dement. Geriatr. Cogn. Disord. 2016, 42, 215–226. [Google Scholar] [CrossRef] [Green Version]
- Van Langenhove, T.; Leyton, C.E.; Piguet, O.; Hodges, J.R. Comparing Longitudinal Behavior Changes in the Primary Progressive Aphasias. J. Alzheimer’s Dis. 2016, 53, 1033–1042. [Google Scholar] [CrossRef] [PubMed]
- Ash, S.; Nevler, N.; Phillips, J.; Irwin, D.; McMillan, C.T.; Rascovsky, K.; Grossman, M. A longitudinal study of speech production in primary progressive aphasia and behavioral variant frontotemporal dementia. Brain Lang. 2019, 194, 46–57. [Google Scholar] [CrossRef]
- Ferrari, C.; Polito, C.; Vannucchi, S.; Piaceri, I.; Bagnoli, S.; Lombardi, G.; Lucidi, G.; Berti, V.; Nacmias, B.; Sorbi, S. Primary Progressive Aphasia: Natural History in an Italian Cohort. Alzheimer Dis. Assoc. Disord. 2019, 33, 42–46. [Google Scholar] [CrossRef]
- Funayama, M.; Nakagawa, Y.; Nakajima, A.; Takata, T.; Mimura, Y.; Mimura, M. Dementia trajectory for patients with logopenic variant primary progressive aphasia. Neurol. Sci. 2019, 40, 2573–2579. [Google Scholar] [CrossRef]
- Cosseddu, M.; Benussi, A.; Gazzina, S.; Alberici, A.; Dell’Era, V.; Manes, M.; Cristillo, V.; Borroni, B.; Padovani, A. Progression of behavioural disturbances in frontotemporal dementia: A longitudinal observational study. Eur. J. Neurol. 2020, 27, 265–272. [Google Scholar] [CrossRef]
- Foxe, D.; Irish, M.; Hu, A.; Carrick, J.; Hodges, J.R.; Ahmed, R.M.; Burrell, J.R.; Piguet, O. Longitudinal cognitive and functional changes in primary progressive aphasia. J. Neurol. 2021, 268, 1951–1961. [Google Scholar] [CrossRef]
- Ulugut, H.; Stek, S.; Wagemans, L.E.E.; Jutten, R.J.; Keulen, M.A.; Bouwman, F.H.; Prins, N.D.; Lemstra, A.W.; Krudop, W.; Teunissen, C.E.; et al. The natural history of primary progressive aphasia: Beyond aphasia. J. Neurol. 2021, 1–11. [Google Scholar] [CrossRef]
- Hsieh, S.; Schubert, S.; Hoon, C.; Mioshi, E.; Hodges, J.R. Validation of the Addenbrooke’s Cognitive Examination III in Frontotemporal Dementia and Alzheimer’s Disease. Dement. Geriatr. Cogn. Disord. 2013, 36, 242–250. [Google Scholar] [CrossRef]
- Mioshi, E.; Dawson, K.; Mitchell, J.; Arnold, R.; Hodges, J.R. The Addenbrooke’s Cognitive Examination Revised (ACE-R): A brief cognitive test battery for dementia screening. Int. J. Geriatr. Psychiatry 2006, 21, 1078–1085. [Google Scholar] [CrossRef]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”: A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Morris, J.C. The Clinical Dementia Rating (CDR): Current version and scoring rules. Neurology 1991, 41, 1588–1592. [Google Scholar] [CrossRef]
- Knopman, D.S.; Kramer, J.H.; Boeve, B.F.; Caselli, R.J.; Graff-Radford, N.R.; Mendez, M.F.; Miller, B.L.; Mercaldo, N. Development of methodology for conducting clinical trials in frontotemporal lobar degeneration. Brain 2008, 131, 2957–2968. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wear, H.J.; Scrymgeour-Wedderburn, C.; Mioshi, E.; Williams-Gray, C.H.; Mason, S.L.; Barker, R.A.; Hodges, J.R. The Cambridge Behavioural Inventory revised. Dement. Neuropsychol. 2008, 2, 102–107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wechsler, D. Wechsler Adult Intelligence Scale—Fourth Edition (WAIS-IV). [Database Record]; APA PsycTests: New York, NY, USA, 2008. [Google Scholar] [CrossRef]
- Goodglass, H.; Kaplan, E.; Weintraub, S. Boston Naming Test; Lea & Febiger: Boston, MA, USA, 1983. [Google Scholar]
- Sapolsky, D.; Bakkour, A.; Negreira, A.; Nalipinski, P.; Weintraub, S.; Mesulam, M.-M.; Caplan, D.; Dickerson, B.C. Cortical neuroanatomic correlates of symptom severity in primary progressive aphasia. Neurology 2010, 75, 358–366. [Google Scholar] [CrossRef] [PubMed]
- Cummings, J.L. The Neuropsychiatric Inventory: Assessing psychopathology in dementia patients. Neurology 1997, 48 (Suppl. 6), 10S–16S. [Google Scholar] [CrossRef] [Green Version]
- Kertesz, A.; Davidson, W.; Fox, H. Frontal Behavioral Inventory: Diagnostic Criteria for Frontal Lobe Dementi. Can. J. Neurol. Sci. 1997, 24, 29–36. [Google Scholar] [CrossRef]
- Gélinas, I.; Gauthier, L.; McIntyre, M.; Gauthier, S. Development of a Functional Measure for Persons with Alzheimer’s Disease: The Disability Assessment for Dementia. Am. J. Occup. Ther. 1999, 53, 471–481. [Google Scholar] [CrossRef] [Green Version]
- Pfeffer, R.I.; Kurosaki, M.T.T.; Harrah, J.C.H., Jr.; Chance, J.M.; Filos, R.S. Measurement of Functional Activities in Older Adults in the Community. J. Gerontol. 1982, 37, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Staffaroni, A.M.; Ljubenkov, P.; Kornak, J.; Cobigo, Y.; Datta, S.; Marx, G.; Walters, S.; Chiang, K.; Olney, N.; Elahi, F.; et al. Longitudinal multimodal imaging and clinical endpoints for frontotemporal dementia clinical trials. Brain 2019, 142, 443–459. [Google Scholar] [CrossRef]
- Montreuil, S.; Poulin, E.; Bergeron, D.; Sellami, L.; Verret, L.; Fortin, M.-P.; Poulin, S.; Macoir, J.; Hudon, C.; Bouchard, R.W.; et al. Cognitive Profile of the Logopenic Variant of Primary Progressive Aphasia Using the Dépistage Cognitif de Québec. Dement. Geriatr. Cogn. Disord. 2020, 49, 410–417. [Google Scholar] [CrossRef]
- Macoir, J.; Lavoie, M.; Laforce, R.J.; Brambati, S.M.; Wilson, M.A. Dysexecutive Symptoms in Primary Progressive Aphasia: Beyond Diagnostic Criteria. J. Geriatr. Psychiatry Neurol. 2017, 30, 151–161. [Google Scholar] [CrossRef]
- Eikelboom, W.; Janssen, N.; Jiskoot, L.C.; van den Berg, E.; Roelofs, A.; Kessels, R.P. Episodic and working memory function in Primary Progressive Aphasia: A meta-analysis. Neurosci. Biobehav. Rev. 2018, 92, 243–254. [Google Scholar] [CrossRef] [PubMed]
- Doody, R.S.; Pavlik, V.; Massman, P.; Rountree, S.; Darby, E.; Chan, W. Predicting progression of Alzheimer’s disease. Alzheimer’s Res. Ther. 2010, 2, 2–9. [Google Scholar] [CrossRef] [Green Version]
- Henry, M.L.; Grasso, S.M. Assessment of Individuals with Primary Progressive Aphasia. Semin. Speech Lang. 2018, 39, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Le Rhun, E.; Richard, F.; Pasquier, F. Natural history of primary progressive aphasia. Neurology 2005, 65, 887–891. [Google Scholar] [CrossRef] [PubMed]
- Lai, C. The merits and problems of Neuropsychiatric Inventory as an assessment tool in people with dementia and other neurological disorders. Clin. Interv. Aging 2014, 9, 1051–1061. [Google Scholar] [CrossRef] [Green Version]
- Mioshi, E.; Hodges, J. Rate of Change of Functional Abilities in Frontotemporal Dementia. Dement. Geriatr. Cogn. Disord. 2009, 28, 419–426. [Google Scholar] [CrossRef]
- Stern, Y. Cognitive reserve in ageing and Alzheimer’s disease. Lancet Neurol. 2012, 11, 1006–1012. [Google Scholar] [CrossRef] [Green Version]
- Stern, Y.; Barulli, D. Cognitive reserve. Handb. Clin. Neurol. 2019, 167, 181–190. [Google Scholar] [CrossRef]
- Brambati, S.M.; Amici, S.; Racine, C.A.; Neuhaus, J.; Miller, Z.; Ogar, J.; Dronkers, N.; Miller, B.L.; Rosen, H.; Gorno-Tempini, M.L. Longitudinal gray matter contraction in three variants of primary progressive aphasia: A tenser-based morphometry study. NeuroImage Clin. 2015, 8, 345–355. [Google Scholar] [CrossRef] [Green Version]
- Price, C.J. The anatomy of language: A review of 100 fMRI studies published in 2009. Ann. N. Y. Acad. Sci. 2010, 1191, 62–88. [Google Scholar] [CrossRef]
- Tetzloff, K.A.; Duffy, J.R.; Clark, H.M.; Strand, E.A.; Machulda, M.M.; Schwarz, C.G.; Senjem, M.L.; Reid, R.I.; Spychalla, A.J.; Tosakulwong, N.; et al. Longitudinal structural and molecular neuroimaging in agrammatic primary progressive aphasia. Brain 2017, 141, 302–317. [Google Scholar] [CrossRef] [PubMed]
- Rosen, H.J.; Allison, S.C.; Ogar, J.M.; Amici, S.; Rose, K.; Dronkers, N.; Miller, B.L.; Gorno-Tempini, M.L. Behavioral features in semantic dementia vs. other forms of progressive aphasias. Neurology 2006, 67, 1752–1756. [Google Scholar] [CrossRef] [PubMed]
- Singh, T.D.; Duffy, J.R.; Strand, E.A.; Machulda, M.M.; Whitwell, J.L.; Josephs, K.A. Neuropsychiatric Symptoms in Primary Progressive Aphasia and Apraxia of Speech. Dement. Geriatr. Cogn. Disord. 2015, 39, 228–238. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ulugut Erkoyun, H.; Groot, C.; Heilbron, R.; Nelissen, A.; Van Rossum, J.; Jutten, R.; Koene, T.; Van Der Flier, W.M.; Wattjes, M.P.; Scheltens, P.; et al. A clinical-radiological framework of the right temporal variant of frontotemporal dementia. Brain 2020, 143, 2831–2843. [Google Scholar] [CrossRef] [PubMed]
- Sebastian, R.; Thompson, C.B.; Wang, N.-Y.; Wright, A.; Meyer, A.; Friedman, R.B.; Hillis, A.E.; Tippett, D.C. Patterns of decline in naming and semantic knowledge in primary progressive aphasia. Aphasiology 2017, 32, 1010–1030. [Google Scholar] [CrossRef] [PubMed]
- Rohrer, J.; Rossor, M.; Warren, J.D. Alzheimer’s pathology in primary progressive aphasia. Neurobiol. Aging 2012, 33, 744–752. [Google Scholar] [CrossRef] [Green Version]
- Tastevin, M.; Lavoie, M.; de la Sablonnière, J.; Carrier-Auclair, J.; Laforce, R., Jr. Survival in the Three Common Variants of Primary Progressive Aphasia: A Retrospective Study in a Tertiary Memory Clinic. Brain Sci. 2021, 11, 1113. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Prisma Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. BMJ 2009, 339, b2535. [Google Scholar] [CrossRef] [Green Version]
- Morhardt, D.J.; O’Hara, M.C.; Zachrich, K.; Wieneke, C.; Rogalski, E.J. Development of a Psycho-Educational Support Program for Individuals with Primary Progressive Aphasia and their Care-Partners. Dementia 2019, 18, 1310–1327. [Google Scholar] [CrossRef]
- Henry, M.L.; Hubbard, H.I.; Grasso, S.M.; Dial, H.R.; Beeson, P.M.; Miller, B.L.; Gorno-Tempini, M.L. Treatment for Word Retrieval in Semantic and Logopenic Variants of Primary Progressive Aphasia: Immediate and Long-Term Outcomes. J. Speech Lang. Hear. Res. 2019, 62, 2723–2749. [Google Scholar] [CrossRef] [PubMed]
- Pagnoni, I.; Gobbi, E.; Premi, E.; Borroni, B.; Binetti, G.; Cotelli, M.; Manenti, R. Language training for oral and written naming impairment in primary progressive aphasia: A review. Transl. Neurodegener. 2021, 10, 24. [Google Scholar] [CrossRef] [PubMed]
- Dial, H.R.; Hinshelwood, H.A.; Grasso, S.M.; Hubbard, H.I.; Gorno-Tempini, M.-L.; Henry, M.L. Investigating the utility of teletherapy in individuals with primary progressive aphasia. Clin. Interv. Aging 2019, 14, 453–471. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Authors (Year) | Participants | Study Design | Follow-Up | Clinical Assessment | Main Results |
|---|---|---|---|---|---|
| Rogalski et al. (2011) [24] | lvPPA: n = 6 nfvPPA: n = 3 svPPA: n = 4 HC: n = 27 | Prospective group study | 1 FU at 2 years |
|
|
| Hsieh et al. (2012) [25] | lvPPA: n = 9 nfvPPA: n = 12 svPPA: n = 17 AD: n = 17 | Retrospective group study | Two assessments at least 12 months apart |
|
|
| Leyton et al. (2013) [26] | lvPPA: n = 13 svPPA: n = 11 HC: n = 17 | Prospective group study | Yearly Mean duration of 3 years |
|
|
| Linds et al. (2015) [27] | nfvPPA + svPPA: n = 13 bvFTD: n = 30 AD: n = 118 | Retrospective group study | Every year |
|
|
| Matias-Guiu et al. (2015) [28] | lvPPA: n = 17 nfvPPA: n = 12 svPPA: n = 4 Unclassified: n = 2 HC: n = 16 | Prospective group study | Every 4 to 6 months Mean length unknown |
|
|
| Gómez-Tortosa et al. (2016) [29] | nfvPPA: n = 39 svPPA: n = 41 | Retrospective group study | Biannual Mean length = 5 years |
|
|
| O’Connor et al. (2016) [30] | nfvPPA: n = 11 svPPA: n = 18 | Prospective group study | Baseline and one FU at mean 1.4 years |
|
|
| Van Langenhove et al. (2016) [31] | lvPPA: n = 21 nfvPPA: n = 22 svPPA: n = 30 bvFTD: n = 33 AD: n = 31 | Prospective group study | 1 FU at a mean of 12 months |
|
|
| Ash et al. (2019) [32] | lvPPA: n = 14 nfvPPA: n = 9 svPPA: n = 11 bvFTD: n = 14 HC: n = 36 | Prospective group study | 1 FU at a mean of 26 months |
|
|
| Ferrari et al. (2019) [33] | lvPPA: n = 23 nfvPPA: n = 26 svPPA: n = 19 | Retrospective group study | M = 2.06 years Frequency unknown |
|
|
| Funayama et al. (2019) [34] | lvPPA: n = 10 | Prospective group study | Every year Duration 6 to 10 years post onset |
|
|
| Cosseddu et al. (2020) [35] | nfvPPA: n = 77 svPPA: n = 40 bvFTD: n = 286 | Retrospective and prospective group study | Every year Mean length = 3.1 years |
|
|
| Foxe et al. (2021) [36] | lvPPA: n = 41 nfvPPA: n = 44 svPPA: n = 62 HC: n = 60 | Prospective group study | FU every year |
|
|
| Ulugut et al. (2021) [37] | lvPPA: n = 18 nfvPPA: n = 22 svPPA: n = 24 | Retrospective group study | FU length 1 to 6 years |
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de la Sablonnière, J.; Tastevin, M.; Lavoie, M.; Laforce, R., Jr. Longitudinal Changes in Cognition, Behaviours, and Functional Abilities in the Three Main Variants of Primary Progressive Aphasia: A Literature Review. Brain Sci. 2021, 11, 1209. https://doi.org/10.3390/brainsci11091209
de la Sablonnière J, Tastevin M, Lavoie M, Laforce R Jr. Longitudinal Changes in Cognition, Behaviours, and Functional Abilities in the Three Main Variants of Primary Progressive Aphasia: A Literature Review. Brain Sciences. 2021; 11(9):1209. https://doi.org/10.3390/brainsci11091209
Chicago/Turabian Stylede la Sablonnière, Justine, Maud Tastevin, Monica Lavoie, and Robert Laforce, Jr. 2021. "Longitudinal Changes in Cognition, Behaviours, and Functional Abilities in the Three Main Variants of Primary Progressive Aphasia: A Literature Review" Brain Sciences 11, no. 9: 1209. https://doi.org/10.3390/brainsci11091209
APA Stylede la Sablonnière, J., Tastevin, M., Lavoie, M., & Laforce, R., Jr. (2021). Longitudinal Changes in Cognition, Behaviours, and Functional Abilities in the Three Main Variants of Primary Progressive Aphasia: A Literature Review. Brain Sciences, 11(9), 1209. https://doi.org/10.3390/brainsci11091209






