Parkinson’s Disease Motor Symptom Progression Slowed with Multisensory Dance Learning over 3-Years: A Preliminary Longitudinal Investigation
Abstract
1. Introduction
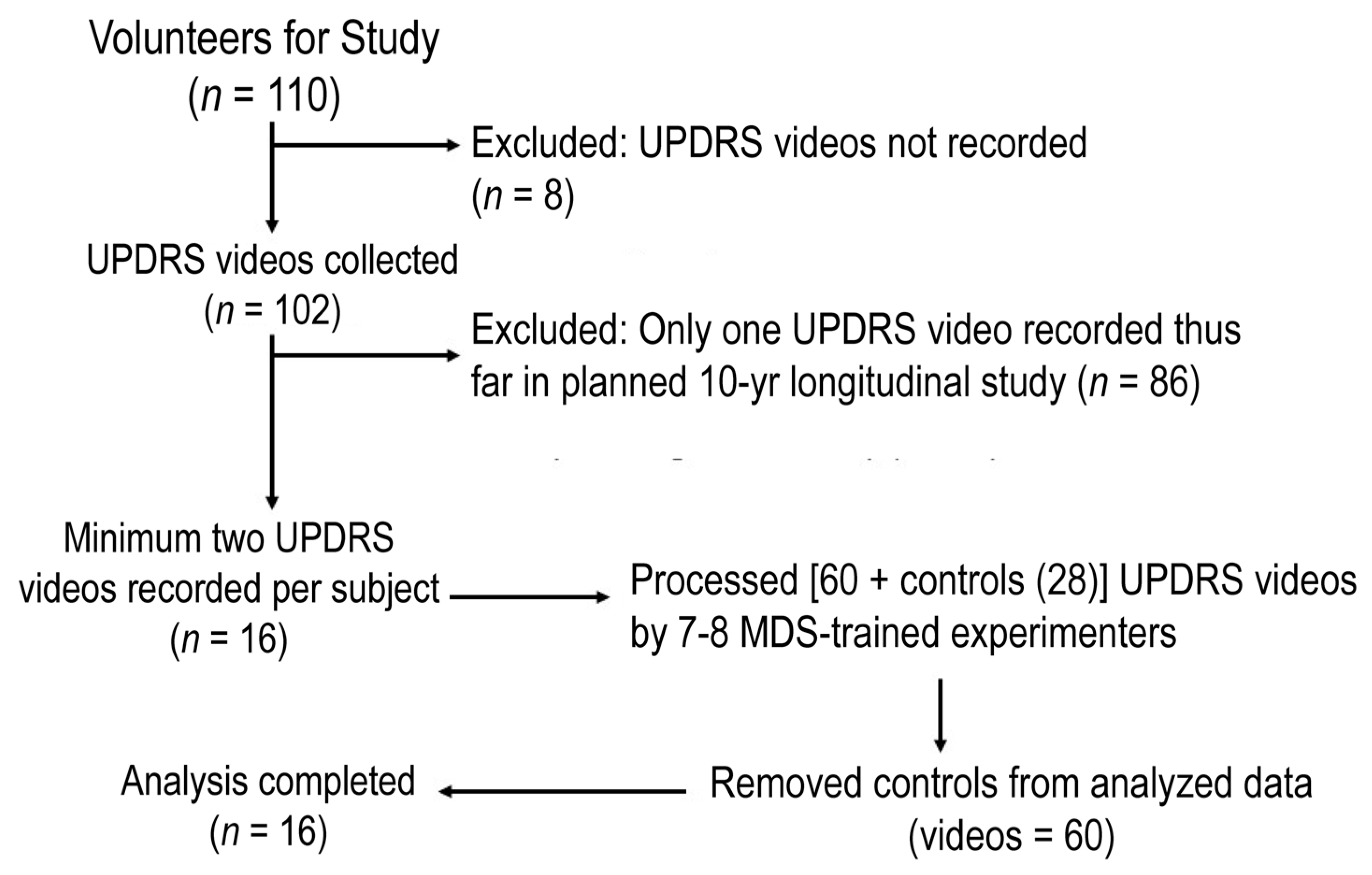
2. Materials and Methods
2.1. Participants
2.2. Ethical Compliance Statement
2.3. Measures
2.4. Procedure
2.5. Analysis
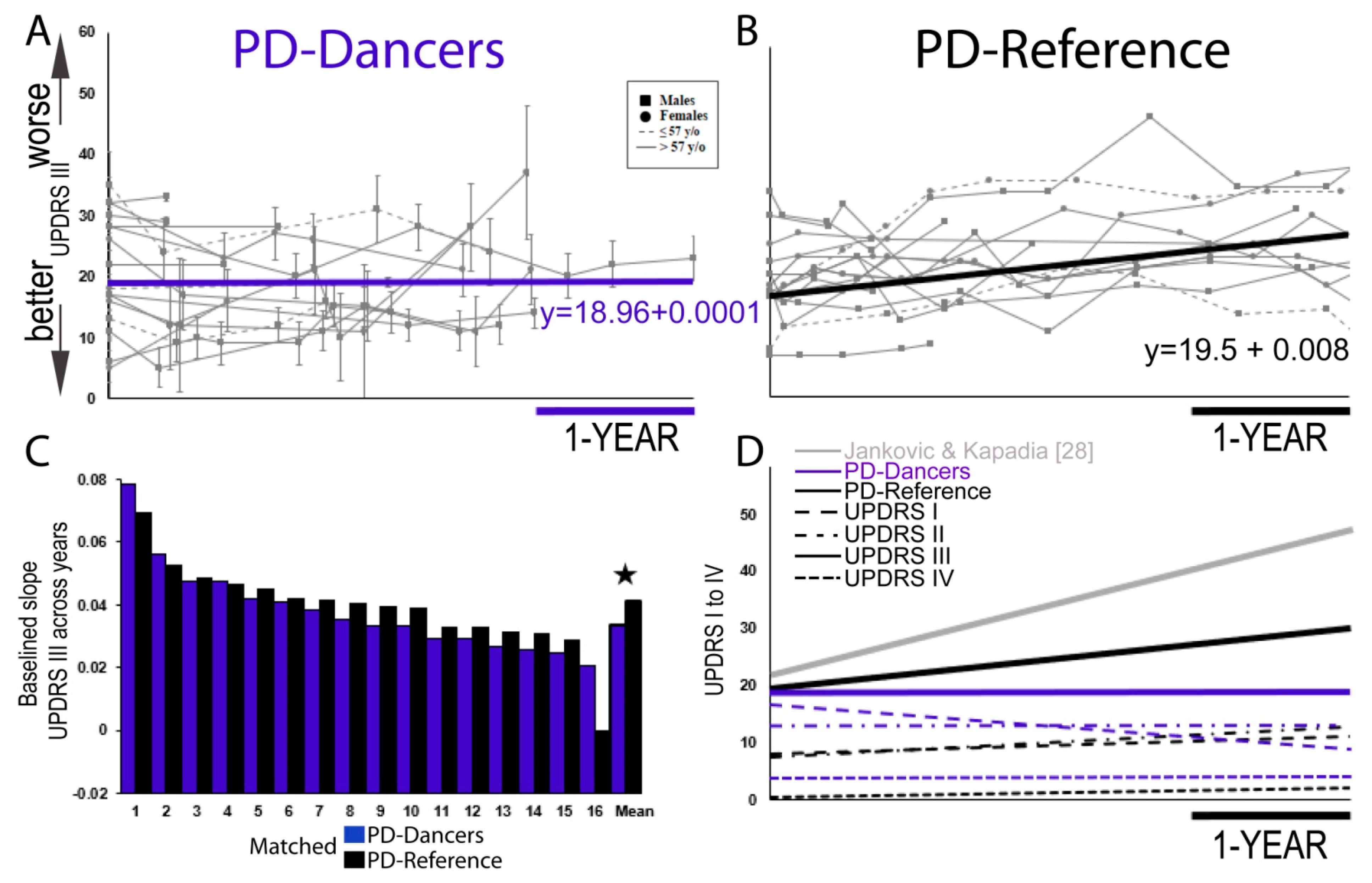
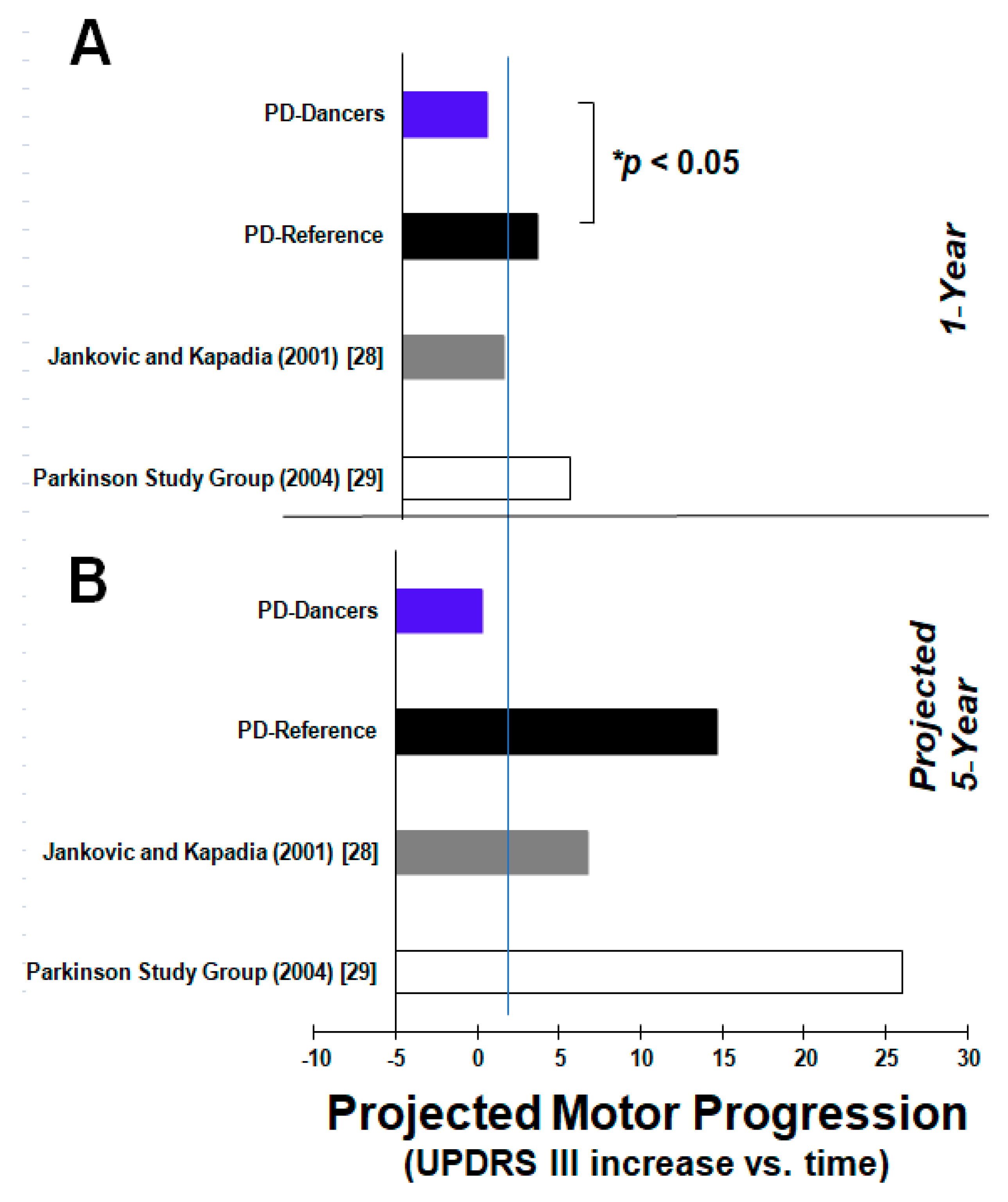
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lozano, A.; Lang, A. First of Two Parts. N. Engl. J. Med. 1998, 339, 1044–1053. [Google Scholar]
- Tan, M.M.; Lawton, M.A.; Jabbari, E.; Reynolds, R.H.; Iwaki, H.; Blauwendraat, C.; Kanavou, S.; Pollard, M.I.; Hubbard, L.; Malek, N.; et al. Genome-wide association studies of cognitive and motor progression in Parkinson’s disease. Mov. Disord. 2021, 36, 424–433. [Google Scholar] [CrossRef] [PubMed]
- Chaudhuri, K.R.; Martinez-Martin, P.; Brown, R.G.; Sethi, K.; Stocchi, F.; Odin, P.; Ondo, W.; Abe, K.; MacPhee, G.; MacMahon, D.; et al. The metric properties of a novel non-motor symptoms scale for Parkinson’s disease: Results from an international pilot study. Mov. Disord. 2007, 22, 1901–1911. [Google Scholar] [CrossRef]
- Martinez-Martin, P.; Rodriguez-Blazquez, C.; Kurtis, M.M.; Chaudhuri, K.R.; NMSS Validation Group. The impact of non-motor symptoms on health-related quality of life of patients with Parkinson’s disease. Mov. Disord. 2011, 26, 399–406. [Google Scholar] [CrossRef]
- Westheimer, O. Why dance for Parkinson’s disease. Top. Geriatr. Rehabil. 2008, 24, 127–140. [Google Scholar] [CrossRef]
- Heiberger, L.; Maurer, C.; Amtage, F.; Mendez-Balbuena, I.; Schulte-Mönting, J.; Hepp-Reymond, M.C.; Kristeva, R. Impact of a weekly dance class on the functional mobility and on the quality of life of individuals with Parkinson’s disease. Front. Aging Neurosci. 2011, 3, 1–15. [Google Scholar] [CrossRef]
- Westheimer, O.; McRae, C.; Henchcliffe, C.; Fesharaki, A.; Glazman, S.; Ene, H.; Bodis-Wollner, I. Dance for PD: A preliminary investigation of effects on motor function and quality of life among persons with Parkinson’s disease (PD). J. Neural. Transm. 2015, 122, 1263–1270. [Google Scholar] [CrossRef]
- Houston, S.; McGill, A. A mixed-methods study into ballet for people living with Parkinson’s. Arts Health 2013, 5, 103–119. [Google Scholar] [CrossRef]
- Bearss, K.A.; McDonald, K.C.; Bar, R.J.; DeSouza, J.F. Improvements in balance and gait speed after a 12 week dance intervention for Parkinson’s disease. Adv. Int. Med. 2017, 4, 10–13. [Google Scholar] [CrossRef]
- Volpe, D.; Signorini, M.; Marchetto, A.; Lynch, T.; Morris, M.E. A comparison of Irish set dancing and exercises for people with Parkinson’s disease: A phase II feasibility study. BMC Geriatr. 2013, 13, 54. [Google Scholar] [CrossRef]
- Delabary, M.D.; Monterio, E.M.; Donida, R.D.; Wolffenbuttel, M.W.; Peyré-Tartaruga, L.P.; Haas, A.H. Can Samba and Forro Brazilian rhythmic dance be more effective than walking in improving functional mobility and spatiotemporal gait parameters in patients with Parkinson’s disease? BMC Neurol. 2020, 20, 305. [Google Scholar]
- Mandelbaum, R.; Lo, A.C. Examining Dance as an Intervention in Parkinson’s Disease: A Systematic Review. Am. J. Danc. Ther. 2014, 36, 160–175. [Google Scholar] [CrossRef]
- Giménez-Llort, L.; Castillo-Mariqueo, L. PasoDoble, a Proposed Dance/Music for People with Parkinson’s Disease and Their Caregivers. Front. Neurol. 2020, 11, 1378. [Google Scholar] [CrossRef]
- Cameron, I.G.M.; Brien, D.C.; Links, K.; Robichaud, S.; Ryan, J.D.; Munoz, D.P.; Chow, T.W. Changes to Saccade Behaviors in Parkinson’s Disease Following Dancing and Observation of Dancing. Front. Neurol. 2013, 4, 22. [Google Scholar] [CrossRef][Green Version]
- Hackney, M.E.; Earhart, G.M. Effects of dance on balance and gait in severe Parkinson disease: A case study. Disabil. Rehabil. 2009, 32, 679–684. [Google Scholar] [CrossRef]
- McKee, K.E.; Hackney, M.E. The effects of adapted tango on spatial cognition and disease severity in Parkinson’s disease. J. Mot. Behav. 2013, 45, 519–529. [Google Scholar] [CrossRef]
- de Bruin, N.; Doan, J.B.; Turnbull, G.; Suchowersky, O.; Bonfield, S.; Hu, B.; Brown, L.A. Walking with music is a safe and viable tool for gait training in Parkinson’s disease: The effect of a 13-week feasibility study on single and dual task walking. Parkinson’s Dis. 2010, 1. [Google Scholar] [CrossRef]
- Hackney, M.E.; Earhart, G.M. Effects of dance on movement control in Parkinson’s disease: A comparison of Argentine tango and American ballroom. J. Rehabil. Med. 2009, 41, 475–481. [Google Scholar] [CrossRef]
- Hackney, M.E.; Earhart, G.M. Effects of dance on gait and balance in Parkinson’s disease: A comparison of partnered and nonpartnered dance movement. Neurorehabilit. Neural. Repair. 2010, 24, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Batson, G. Feasibility of an Intensive Trial of Modern Dance for Adults with Parkinson Disease. Complement. Health Prac. Rev. 2010, 15, 65–83. [Google Scholar] [CrossRef]
- Hackney, M.E.; Earhart, G.M. Health-related quality of life and alternative forms of exercise in Parkinson disease. Park Relat. Disord. 2009, 15, 644–648. [Google Scholar] [CrossRef]
- Duncan, R.P.; Earhart, G.M. Randomized controlled trial of community-based dancing to modify disease progression in Parkinson disease. Neurorehabil. Neural. Repair. 2012, 26, 132–143. [Google Scholar] [CrossRef]
- McGill, A.; Houston, S.; Lee, R.Y. Effects of a ballet-based dance intervention on gait variability and balance confidence of people with Parkinson’s. Arts Health 2019, 11, 133–146. [Google Scholar] [CrossRef]
- McGill, A.; Houston, S.; Lee, R.Y. Effects of a ballet intervention on trunk coordination and range of motion during gait in people with Parkinson’s. Cogent. Med. 2019, 6, 1–14. [Google Scholar] [CrossRef]
- Foster, E.R.; Golden, L.; Duncan, R.P.; Earhart, G.M. Community-based Argentine tango dance program is associated with increased activity participation among individuals with Parkinson’s disease. Arch. Phys. Med. Rehabil. 2013, 94, 240–249. [Google Scholar] [CrossRef] [PubMed]
- McRae, C.; Leventhal, D.; Westheimer, O.; Mastin, T.; Utley, J.; Russell, D. Long-term effects of Dance for PD® on self-efficacy among persons with Parkinson’s disease. Arts Health 2018, 10, 85–96. [Google Scholar] [CrossRef]
- Duncan, R.P.; Earhart, G.M. Are the effects of community-based dance on Parkinson disease severity, balance, and functional mobility reduced with time? A 2-year prospective pilot study. J. Altern. Complementary Med. 2014, 20, 757–763. [Google Scholar] [CrossRef]
- Jankovic, J.; Kapadia, A.S. Functional decline in Parkinson disease. Arch. Neurol. 2001, 58, 1611–1615. [Google Scholar] [CrossRef]
- Parkinson Study Group. Levodopa and the progression of Parkinson’s disease. N. Engl. J. Med. 2004, 351, 2498–2508. [Google Scholar] [CrossRef] [PubMed]
- Chan, P.L.; Holford, N.H. Drug treatment effects on disease progression. Annu. Rev. Pharmacol. Toxicol. 2001, 41, 625–659. [Google Scholar] [CrossRef]
- Bergen, J.L.; Toole, T.; Elliott Iii, R.G.; Wallace, B.; Robinson, K.; Maitland, C.G. Aerobic exercise intervention improves aerobic capacity and movement initiation in Parkinson’s disease patients. Neuro. Rehabil. 2002, 17, 161–168. [Google Scholar] [CrossRef]
- Sabaghi, A.; Heirani, A.; Mahmoodi, H.; Sabaghi, S. High-intensity interval training prevents cognitive-motor impairment and serum BDNF level reduction in parkinson mice model. Sport Sci. Health 2019, 3, 681–687. [Google Scholar] [CrossRef]
- O’Callaghan, A.; Harvey, M.; Houghton, D.; Gray, W.K.; Weston, K.L.; Oates, L.L.; Romano, B.; Walker, R.W. Comparing the influence of exercise intensity on brain-derived neurotrophic factor serum levels in people with Parkinson’s disease: A pilot study. Aging Clin. Exp. Res. 2020, 32, 1731–1738. [Google Scholar] [CrossRef] [PubMed]
- McNeely, M.E.; Duncan, R.P.; Earhart, G.M. A comparison of dance interventions in people with Parkinson disease and older adults. Maturitas 2015, 81, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Israili, Z.H.; Israili S, J. Tango Dance: Therapeutic Benefits: A Narrative Review; IJASSH: Ghaziabad, India, 2018. [Google Scholar]
- Shanahan, J.; Morris, M.E.; Bhriain, O.N.; Volpe, D.; Lynch, T.; Clifford, A.M. Dancing for Parkinson disease: A randomized trial of Irish set dancing compared with usual care. Arch. Phys. Med. Rehabil. 2017, 98, 1744–1751. [Google Scholar] [CrossRef] [PubMed]
- Rocha, P.A.; Slade, S.C.; McClelland, J.; Morris, M.E. Dance is more than therapy: Qualitative analysis on therapeutic dancing classes for Parkinson’s. Complementary Ther. Med. 2017, 34, 1–9. [Google Scholar] [CrossRef]
- Allen, N.E.; Sherrington, C.; Paul, S.S.; Canning, C.G. Balance and falls in Parkinson’s disease: A meta-analysis of the effect of exercise and motor training. Mov. Disord. 2011, 26, 1605–1615. [Google Scholar] [CrossRef] [PubMed]
- Shulman, L.M.; Katzel, L.I.; Ivey, F.M.; Sorkin, J.D.; Favors, K.; Anderson, K.E.; Macko, R.F. Randomized clinical trial of 3 types of physical exercise for patients with Parkinson disease. JAMA Neurol. 2013, 70, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Gibala, M.J.; Little, J.P.; MacDonald, M.J.; Hawley, J.A. Physiological adaptations to low-volume, high-intensity interval training in health and disease. J. Physiol. 2012, 590, 1077–1084. [Google Scholar] [CrossRef] [PubMed]
- Pereira, A.P.S.; Marinho, V.; Gupta, D.; Magalhães, F.; Ayres, C.; Teixeira, S. Music therapy and dance as gait rehabilitation in patients with Parkinson disease: A review of evidence. J. Geriatr. Psychiatry Neurol. 2019, 32, 49–56. [Google Scholar] [CrossRef]
- Lauzé, M.; Daneault, J.F.; Duval, C. The effects of physical activity in Parkinson’s disease: A review. J. Parkinson’s Dis. 2016, 6, 685–698. [Google Scholar] [CrossRef]
- Cheon, S.M.; Chae, B.K.; Sung, H.R.; Lee, G.C.; Kim, J.W. The efficacy of exercise programs for Parkinson’s disease: Tai Chi versus combined exercise. J. Clin. Neurol. 2013, 9, 237–243. [Google Scholar] [PubMed]
- Yang, Y.; Li, X.Y.; Gong, L.; Zhu, Y.L.; Hao, Y.L. Tai Chi for improvement of motor function, balance and gait in Parkinson’s disease: A systematic review and meta-analysis. PLoS ONE 2014, 9, e102942. [Google Scholar] [CrossRef]
- Lim, L.I.I.K.; Van Wegen, E.E.H.; De Goede, C.J.T.; Jones, D.; Rochester, L.; Hetherington, V.; Nieuwboer, A.; Willems, A.M.; Kwakkel, G. Measuring gait and gait-related activities in Parkinson’s patients own home environment: A reliability, responsiveness and feasibility study. Parkinsonism Relat. Disord. 2005, 11, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Asmundson, G.J.; Fetzner, M.G.; DeBoer, L.B.; Powers, M.B.; Otto, M.W.; Smits, J.A. Let’s get physical: A contemporary review of the anxiolytic effects of exercise for anxiety and its disorders. Depress. Anxiety 2013, 30, 362–373. [Google Scholar] [CrossRef] [PubMed]
- Fontanesi, C.; DeSouza, J.F. Beauty that Moves: Dance for Parkinson’s Effects on Affect, Self-Efficacy, Gait Symmetry, and Dual Task Performance. Front. Psychol. 2021, 11, 3896. [Google Scholar] [CrossRef] [PubMed]
| PD-Dancers | PD-Reference | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Subject | Age | Age Onset | H and Y | Total Hours in DfPD Exercises | Subject | Age | Age Onset | H and Y | PASE Activity (h) | |
| Q. 4b (Ballroom Dance) | Q. 5b (Aerobic Dance) | |||||||||
| 10× | 70 | 67 | 2 | 107 | 3002 | 68 | 60 | 2 | 0 | 0 |
| 10× | 66 | 64 | 1 | 125 | 3018 | 61 | 55 | 2 | 0 | 0 |
| 10× | 76 | 73 | 0 | 124 | 3021 | 64 | 58 | 2 | 0 | 0 |
| 10× | 70 | 66 | 1 | 116 | 3028 | 76 | 71 | 2 | 0 | 0 |
| 10× | 83 | 82 | 2 | 173 | 3051 | 72 | 64 | 2 | 0 | 0 |
| 10× | 52 | 37 | 2 | 64 | 3810 | 67 | 58 | 1 | - | - |
| 10× | 59 | 50 | 2 | 113 | 3958 | 76 | 69 | 1 | - | - |
| 10× | 73 | 70 | 2 | 105 | 3962 | 69 | 63 | 1 | - | - |
| 10× | 77 | 77 | 3 | 17 | 4076 | 72 | 66 | 2 | 0 | 0 |
| 11× | 58 | 58 | 0 | 84 | 40,690 | 72 | 65 | 2 | 0 | 0 |
| 11× | 61 | 50 | 1 | 83 | 40,693 | 72 | 66 | 1 | 0 | 0 |
| 12× | 68 | 67 | 1 | 122 | 40,740 | 69 | 65 | 1 | 0 | 0 |
| 13× | 73 | 71 | 1 | 24 | 40,916 | 77 | 65 | 2 | 0 | 0 |
| 14× | - | - | 1 | 50 | 50,175 | 62 | 57 | 1 | 0 | 0 |
| 15× | 77 | 67 | 0 | 35 | 51,971 | 66 | 62 | 2 | 0 | 0 |
| 16× | 68 | 60 | 1 | 17 | 57,090 | 74 | 72 | 2 | 0 | 0 |
| Mean | 68.7 | 63.9 | 1.3 | 85.5 | 69.8 | 63.5 | 1.6 | 0.0 | 0.0 | |
| SD | 8.4 | 11.5 | 0.9 | 45.2 | 4.9 | 5.0 | 0.5 | 0.0 | 0.0 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bearss, K.A.; DeSouza, J.F.X. Parkinson’s Disease Motor Symptom Progression Slowed with Multisensory Dance Learning over 3-Years: A Preliminary Longitudinal Investigation. Brain Sci. 2021, 11, 895. https://doi.org/10.3390/brainsci11070895
Bearss KA, DeSouza JFX. Parkinson’s Disease Motor Symptom Progression Slowed with Multisensory Dance Learning over 3-Years: A Preliminary Longitudinal Investigation. Brain Sciences. 2021; 11(7):895. https://doi.org/10.3390/brainsci11070895
Chicago/Turabian StyleBearss, Karolina A., and Joseph F. X. DeSouza. 2021. "Parkinson’s Disease Motor Symptom Progression Slowed with Multisensory Dance Learning over 3-Years: A Preliminary Longitudinal Investigation" Brain Sciences 11, no. 7: 895. https://doi.org/10.3390/brainsci11070895
APA StyleBearss, K. A., & DeSouza, J. F. X. (2021). Parkinson’s Disease Motor Symptom Progression Slowed with Multisensory Dance Learning over 3-Years: A Preliminary Longitudinal Investigation. Brain Sciences, 11(7), 895. https://doi.org/10.3390/brainsci11070895






