Repetitive Transcranial Magnetic Stimulation as an Add-On Treatment for Cognitive Impairment in Alzheimer’s Disease and Its Impact on Self-Rated Quality of Life and Caregiver’s Burden
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Design
2.3. Outcome Measures
2.3.1. Cognitive Assessment
2.3.2. Functional Ability and Quality of Life Assessment
2.3.3. Psychiatric Assessment
2.4. Statistical Analysis
3. Results
3.1. Demographic and Baseline Characteristics
3.2. Effect of 10 HF-rTMS Sessions
3.3. Correlations between Response to Treatment and Quality of Life
3.4. Side Effects
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Aisen, P.S.; Cummings, J.; Jack, C.R., Jr.; Morris, J.C.; Sperling, R.; Frölich, L.; Jones, R.W.; Dowsett, S.A.; Matthews, B.R.; Raskin, J.; et al. On the path to 2025: Understanding the Alzheimer’s disease continuum. Alzheimer’s Res. Ther. 2017, 9, 60. [Google Scholar] [CrossRef]
- Lyketsos, C.G.; Carrillo, M.C.; Ryan, J.M.; Khachaturian, A.S.; Trzepacz, P.; Amatniek, J.; Cedarbaum, J.; Brashear, R.; Miller, D.S. Neuropsychiatric symptoms in Alzheimer’s disease. Alzheimer’s Dement. 2011, 7, 532–539. [Google Scholar] [CrossRef]
- Sivertsen, H.; Bjørkløf, G.H.; Engedal, K.; Selbaek, G.; Helvik, A.-S. Depression and Quality of Life in Older Persons: A Review. Dement. Geriatr. Cogn. Disord. 2015, 40, 311–339. [Google Scholar] [CrossRef]
- Novais, F.; Starkstein, S. Phenomenology of Depression in Alzheimer’s Disease. J. Alzheimer’s Dis. 2015, 47, 845–855. [Google Scholar] [CrossRef] [PubMed]
- Rutherford, G.; Gole, R.; Moussavi, Z. rTMS as a Treatment of Alzheimer’s Disease with and without Comorbidity of Depression: A Review. Neurosci. J. 2013, 2013, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Joe, E.; Ringman, J.M. Cognitive symptoms of Alzheimer’s disease: Clinical management and prevention. BMJ 2019, 367, l6217. [Google Scholar] [CrossRef]
- Weiler, M.; Stieger, K.C.; Long, J.M.; Rapp, P.R. Transcranial Magnetic Stimulation in Alzheimer’s Disease: Are We Ready? Eneuro 2020, 7. [Google Scholar] [CrossRef]
- Demirtas-Tatlidede, A.; Vahabzadeh-Hagh, A.M.; Pascual-Leone, A. Can noninvasive brain stimulation enhance cognition in neuropsychiatric disorders? Neuropharmacology 2013, 64, 566–578. [Google Scholar] [CrossRef]
- Chang, C.-H.; Lane, H.-Y.; Lin, C.-H. Brain Stimulation in Alzheimer’s Disease. Front. Psychiatry 2018, 9, 201. [Google Scholar] [CrossRef]
- Fregni, F.; Pascual-Leone, A. Transcranial magnetic stimulation for the treatment of depression in neurologic disorders. Curr. Psychiatry Rep. 2005, 7, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Chou, Y.-H.; That, V.T.; Sundman, M. A systematic review and meta-analysis of rTMS effects on cognitive enhancement in mild cognitive impairment and Alzheimer’s disease. Neurobiol. Aging 2020, 86, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Haffen, E.; Chopard, G.; Pretalli, J.-B.; Magnin, E.; Nicolier, M.; Monnin, J.; Galmiche, J.; Rumbach, L.; Pazart, L.; Sechter, D.; et al. A case report of daily left prefrontal repetitive transcranial magnetic stimulation (rTMS) as an adjunctive treatment for Alzheimer disease. Brain Stimul. 2012, 5, 264–266. [Google Scholar] [CrossRef]
- Bentwich, J.; Dobronevsky, E.; Aichenbaum, S.; Shorer, R.; Peretz, R.; Khaigrekht, M.; Marton, R.G.; Rabey, J.M. Beneficial effect of repetitive transcranial magnetic stimulation combined with cognitive training for the treatment of Alzheimer’s disease: A proof of concept study. J. Neural Transm. 2011, 118, 463–471. [Google Scholar] [CrossRef]
- McKhann, G.; Drachman, D.; Folstein, M.; Katzman, R.; Price, D.; Stadlan, E.M. Clinical diagnosis of Alzheimer’s disease: Report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 1984, 34, 939–944. [Google Scholar] [CrossRef] [PubMed]
- Monsch, A.U.; Bondi, M.W.; Salmon, D.P.; Butters, N.; Thal, L.J.; Hansen, L.A.; Wiederholt, W.C.; Cahn, D.A.; Klauber, M.R. Clinical Validity of the Mattis Dementia Rating Scale in Detecting Dementia of the Alzheimer Type. Arch. Neurol. 1995, 52, 899–904. [Google Scholar] [CrossRef]
- Goldman, W.P.; Baty, J.D.; Buckles, V.D.; Sahrmann, S.; Morris, J.C. Motor dysfunction in mildly demented AD individuals without extrapyramidal signs. Neurology 1999, 53, 956. [Google Scholar] [CrossRef]
- Reitan, R.M.; Wolfson, D. The halstead–reitan neuropsychological test battery: Research findings and clinical application. In Specific Learning Disabilities and Difficulties in Children and Adolescents: Psychological Assessment and Evaluation; Cambridge University Press: New York, NY, USA, 2001; pp. 309–346. [Google Scholar]
- Isaacs, B.; Kennie, A.T. The Set Test as an Aid to the Detection of Dementia in Old People. Br. J. Psychiatry 1973, 123, 467–470. [Google Scholar] [CrossRef]
- Barbeau, E.; Didic, M.; Tramoni, E.; Felician, O.; Joubert, S.; Sontheimer, A.; Ceccaldi, M.; Poncet, M. Evaluation of visual recognition memory in MCI patients. Neurology 2004, 62, 1317–1322. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, S.; Vanholsbeeck, G.; Chopard, G.; Pitard, A.; Tio, G.; Vandel, P.; Galmiche, J.; Rumbach, L. Comparative norms of RAPID neuropsychological battery of tests for subjects aged between 50 and 89 years. Rev. Neurol. 2010, 166, 606–614. [Google Scholar] [CrossRef]
- Katz, S.; Downs, T.D.; Cash, H.R.; Grotz, R.C. Progress in Development of the Index of ADL. Gerontologist 1970, 10, 20–30. [Google Scholar] [CrossRef]
- Lawton, M.P.; Brody, E.M. Assessment of older people: Self-maintaining and instrumental activities of daily living. Gerontologist 1969, 9, 179–186. [Google Scholar] [CrossRef]
- Logsdon, R.G.; Gibbons, L.E.; McCurry, S.M.; Teri, L. Quality of life in Alzheimer’s disease: Patient and Caregiver Reports. J. Ment. Health Aging 1999, 5, 1. Available online: https://scholar.google.com/scholar_lookup?title=Quality%20of%20Life%20in%20Alzheimer%E2%80%99s%20Disease%3A%20patient%20and%20caregiver%20reports&journal=J%20Ment%20Health%20Aging&volume=5&pages=21-32&publication_year=1999&author=Logsdon%2CR&author=Gibbons%2CLE&author=McCurry%2CSM&author=Teri%2CL (accessed on 12 October 2020).
- Logsdon, R.G.; Gibbons, L.E.; McCurry, S.M.; Teri, L. Assessing Quality of Life in Older Adults with Cognitive Impairment. Psychosom. Med. 2002, 64, 510–519. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, M. A rating scale for depression. J. Neurol. Neurosurg. Psychiatry 1960, 23, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Beck, A.T.; Ward, C.H.; Mendelson, M.; Mock, J.; Erbaugh, J. An Inventory for Measuring Depression. Arch. Gen. Psychiatry 1961, 4, 561–571. [Google Scholar] [CrossRef]
- Gaudry, E.; Vagg, P.; Spielberger, C.D. Validation of the State-Trait Distinction in Anxiety Research. Multivar. Behav. Res. 1975, 10, 331–341. [Google Scholar] [CrossRef]
- Assessment, Q.O.L. The World Health Organization quality of life assessment (WHOQOL): Position paper from the World Health Organization. Soc. Sci. Med. 1995, 41, 1403–1409. [Google Scholar] [CrossRef]
- Tulving, E.; Kapur, S.; Craik, F.I.; Moscovitch, M.; Houle, S. Hemispheric encoding/retrieval asymmetry in episodic memory: Positron emission tomography findings. Proc. Natl. Acad. Sci. USA 1994, 91, 2016–2020. [Google Scholar] [CrossRef]
- Braak, H.; Braak, E. Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol. 1991, 82, 239–259. [Google Scholar] [CrossRef]
- Duff, K.; Beglinger, L.J.; Van Der Heiden, S.; Moser, D.J.; Arndt, S.; Schultz, S.K.; Paulsen, J.S. Short-term practiceeffects in amnestic mild cognitive impairment: Implications for diag-nosis and treatment. Int. Psychogeriatr. 2008, 20, 986–999. [Google Scholar] [CrossRef]
- Scharfen, J.; Jansen, K.; Holling, H. Retest effects in working memory capacity tests: A meta-analysis. Psychon. Bull. Rev. 2018, 25, 2175–2199. [Google Scholar] [CrossRef]
- Calamia, M.; Markon, K.; Tranel, D. Scoring higher the second time around: Meta-analyses of practice effects in neuropsychological assessment. Clin. Neuropsychol. 2012, 26, 543–570. [Google Scholar] [CrossRef]
- Helkala, E.L.; Kivipelto, M.; Hallikainen, M.; Alhainen, K.; Heinonen, H.; Tuomilehto, J.; Soininen, H.; Nissinen, A. Usefulness of repeated presentation of Mini-Mental State Examination as a diagnostic procedure—A population-based study. Acta. Neurol. Scand. 2002, 106, 341–346. [Google Scholar] [CrossRef]
- Schrijnemaekers, A.M.; de Jager, C.A.; Hogervorst, E.; Budge, M.M. Caseswith mild cognitive impairment and Alzheimer’s disease fail to benefitfrom repeated exposure to episodic memory tests as compared withcontrols. J. Clin. Exp. Neuropsychol. 2006, 28, 438–455. [Google Scholar] [CrossRef] [PubMed]
- Zehnder, A.E.; Blasi, S.; Berres, M.; Spiegel, R.; Monsch, A.U. Lack of practice effects on neuropsychological tests as early cognitive markers of Alzheimer disease? Am. J. Alzheimers Dis. Other Demen. 2007, 22, 416–426. [Google Scholar] [CrossRef] [PubMed]
- Cooper, D.B.; Epker, M.; Lacritz, L.; Weine, M.; Rosenberg, R.N.; Honig, L.; Cullum, C.M. Effects of practice on category fluency in Alzheimer’s disease. Clin. Neuropsychol. 2001, 15, 125–128. [Google Scholar] [CrossRef]
- Cooper, D.B.; Lacritz, L.H.; Weiner, M.F.; Rosenberg, R.N.; Cullum, C.M. Category fluency in mild cognitive impairment: Reduced effect of practice in test-retest conditions. Alzheimer Dis. Assoc. Disord. 2004, 18, 120–122. [Google Scholar] [CrossRef]
- Huntley, J.D.; Howard, R.J. Working memory in early Alzheimer’s disease: A neuropsychological review. Int. J. Geriatr. Psychiatry 2010, 25, 121–132. [Google Scholar] [CrossRef]
- Kaufman, L.D.; Pratt, J.; Levine, B.; Black, S.E. Executive deficits detected in mild Alzheimer’s disease using the antisaccade task. Brain Behav. 2011, 2, 15–21. [Google Scholar] [CrossRef]
- Boggio, P.S.; Khoury, L.P.; Martins, D.C.S.; Martins, O.E.M.S.; De Macedo, E.C.; Fregni, F. Temporal cortex direct current stimulation enhances performance on a visual recognition memory task in Alzheimer disease. J. Neurol. Neurosurg. Psychiatry 2008, 80, 444–447. [Google Scholar] [CrossRef] [PubMed]
- MacPherson, S.E.; Bozzali, M.; Cipolotti, L.; Dolan, R.J.; Rees, J.H.; Shallice, T. Effect of frontal lobe lesions on the recollection and familiarity components of recognition memory. Neuropsychol. 2008, 46, 3124–3132. [Google Scholar] [CrossRef] [PubMed]
- Brunoni, A.R.; Sampaio-Junior, B.; Moffa, A.H.; Aparício, L.V.; Gordon, P.; Klein, I.; Rios, R.M.; Razza, L.B.; Loo, C.; Padberg, F.; et al. Noninvasive brain stimulation in psychiatric disorders: A primer. Rev. Bras. Psiquiatr. 2019, 41, 70–81. [Google Scholar] [CrossRef] [PubMed]
- Lefaucheur, J.-P.; Aleman, A.; Baeken, C.; Benninger, D.H.; Brunelin, J.; Di Lazzaro, V.; Filipović, S.R.; Grefkes, C.; Hasan, A.; Hummel, F.C.; et al. Evidence-based guidelines on the therapeutic use of repetitive transcranial magnetic stimulation (rTMS): An update (2014–2018). Clin. Neurophysiol. 2020, 131, 474–528. [Google Scholar] [CrossRef] [PubMed]
| Subjects (n) | Mean | ±SD | Min | Max | |
|---|---|---|---|---|---|
| Age | 12 | 70.2 | 6.5 | 62 | 84 |
| MMSE | 12 | 19.1 | 5.5 | 11 | 27 |
| MDRS | 12 | 113.8 | 19.1 | 78 | 136 |
| MDRS Attention | 12 | 34.2 | 1.9 | 32 | 36 |
| MDRS I/P | 12 | 26.2 | 10.5 | 8 | 37 |
| MDRS Memory | 12 | 14.2 | 4.0 | 8 | 23 |
| MDRS Conceptualization | 12 | 33.7 | 5.2 | 23 | 39 |
| COT | 12 | 73.7 | 32.9 | 42 | 130 |
| TMT-A | 11 | 73.0 | 39.0 | 24 | 150 |
| TMT B-A | 8 | 193.7 | 187.2 | 42 | 638 |
| IST | 12 | 21.4 | 10.8 | 3 | 37 |
| DMS-48 | 12 | 81.2 | 11.9 | 64 | 98 |
| DO-30 | 12 | 25.1 | 6.7 | 14 | 30 |
| QoL-AD | 9 | 33.2 | 4.0 | 30 | 43 |
| ADL | 10 | 5.7 | 0.5 | 4.5 | 6.0 |
| IADL | 10 | 5.3 | 2.4 | 2.0 | 8.0 |
| BDI | 12 | 6.8 | 4.5 | 0 | 15 |
| HDRS-21 | 12 | 7.5 | 4.9 | 0 | 17 |
| STAI-YA | 12 | 36.3 | 11.7 | 21 | 59 |
| STAI-YB | 12 | 39.7 | 11.0 | 26 | 56 |
| D0 | QoL-AD | ADL | IADL |
|---|---|---|---|
| MMSE | 0.651 | 0.260 | 0.680 * |
| MDRS | 0.582 | 0.517 | 0.735 * |
| MDRS Attention | 0.812 * | 0.072 | 0.396 |
| MDRS I/P | 0.241 | 0.462 | 0.557 |
| MDRS Memory | 0.686 * | 0.293 | 0.855 * |
| MDRS Conceptualization | 0.308 | 0.590 | 0.604 |
| COT | −0.381 | −0.676 * | −0.830 * |
| TMT-A | −0.096 | −0.598 | −0.809 |
| TMT B-A | −0.429 | −−− † | −0.334 |
| IST | 0.051 | 0.719 * | 0.784 * |
| DMS-48 | −0.216 | 0.203 | 0.351 |
| DO-30 | 0.106 | 0.593 | 0.639 * |
| BDI | −0.359 | −0.080 | −0.434 |
| HDRS-21 | 0.140 | 0.011 | −0.034 |
| STAI-YA | −0.165 | 0.451 | −0.071 |
| STAI-YB | −0.532 | 0.360 | −0.043 |
| Measure (Corrected Significance Level) | Median [Q1;Q3] | p-Value | |
|---|---|---|---|
| D0 | M1 | ||
| Global cognition (p < 0.02) | |||
| MMSE | 20 [14;23.5] | 20.5 [16;24] | 0.30 |
| MDRS | 119.5 [100.5;127.5] | 123 [118;127] | 0.10 |
| Attention/Processing speed (p < 0.01) | |||
| COT | 59 [52;96.5] | 58 [46;87] | 0.92 |
| TMT-A | 67 [38;102] | 48 [35;70] | 0.12 |
| MDRS Attention | 35 [32;36] | 36 [35;36] | 0.25 |
| Executive function (p < 0.01) | |||
| TMT B-A | 134.5 [99.5;201] | 165.5 [91;440] | 0.38 |
| IST | 23.5 [13.5;28.5] | 21 [16;30] | 0.79 |
| MDRS I/P | 31 [19;33] | 30 [29;32] | 0.52 |
| Episodic memory (p < 0.02) | |||
| DMS-48 | 81 [70;93.5] | 87.5 [76;97] | 0.04 |
| MDRS Memory | 14.5 [11.5;16] | 14 [11;17] | 0.65 |
| Language/Semantic functioning (p < 0.02) | |||
| DO-30 | 29 [18.5;30] | 28.5 [19;30] | 0.78 |
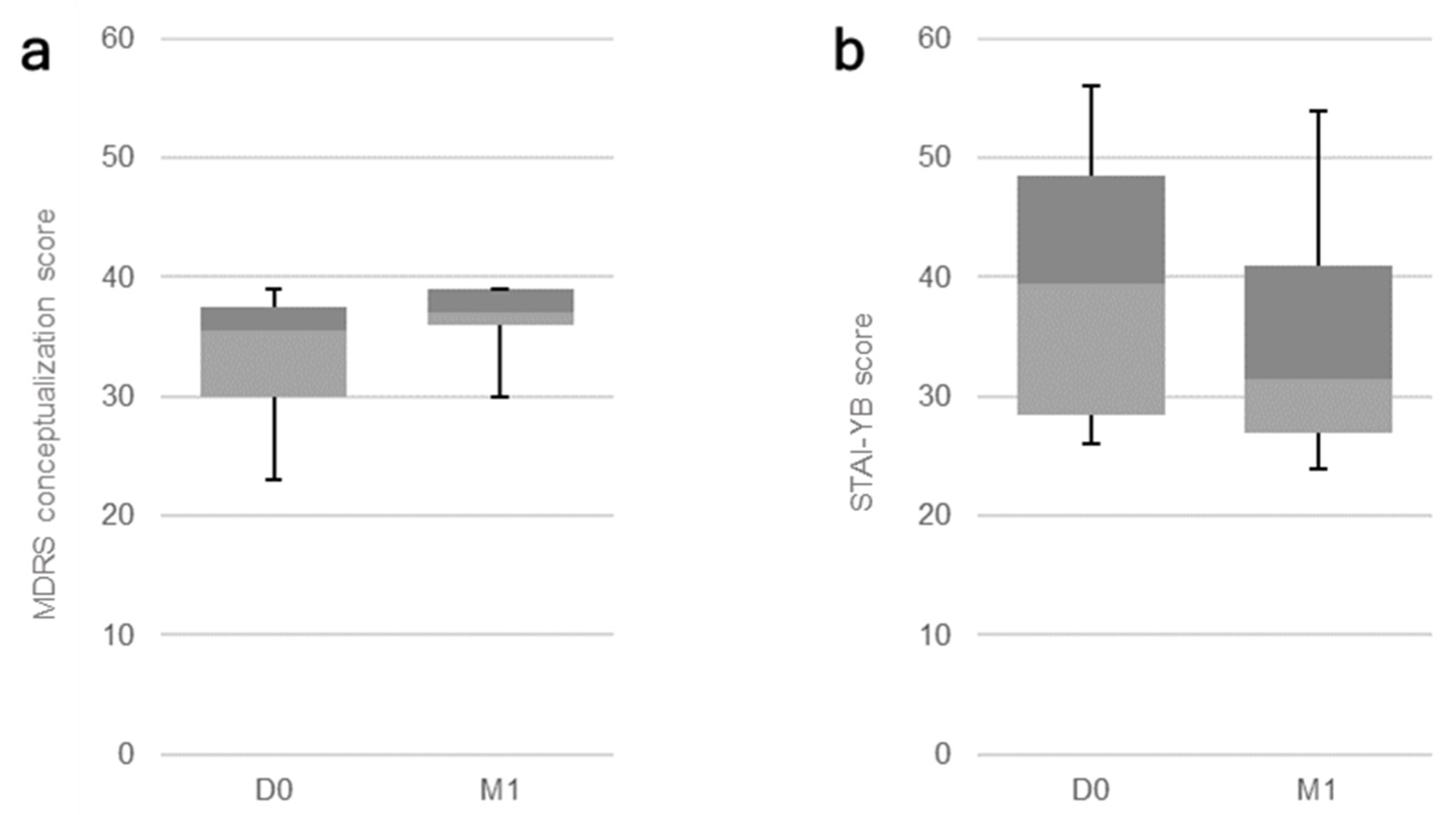
| MDRS Conceptualization | 35.5 [30;37.5] | 37 [36;39] | 0.01 * |
| Functional ability (p < 0.02) | |||
| ADL | 6 [5.5;6] | 6 [5.7;6] | 1 |
| IADL | 5 [3;8] | 6 [3.5;8] | 1 |
| Quality of life (p < 0.05) | |||
| QoL-AD | 32 [31;34] | 32 [30;34] | 0.28 |
| Measure (Corrected Significance Level) | Median [Q1;Q3] | p-Value | |
|---|---|---|---|
| D0 | M1 | ||
| Depression (p < 0.02) | |||
| BDI | 7 [4;9.5] | 3.5 [0.5;6.5] | 0.03 |
| HDRS-21 | 6.5 [4.5;11] | 5.5 [2.5;8] | 0.05 |
| Anxiety (p < 0.02) | |||
| STAI-YA | 37 [26;43.5] | 33.5 [27;39.5] | 0.31 |
| STAI-YB | 39.5 [28.5;48.5] | 31.5 [27;41] | 0.01 * |
| M1-D0 | QoL-AD |
|---|---|
| MMSE | 0.413 |
| MDRS | 0.706 |
| MDRS Attention | 0.633 |
| MDRS I/P | 0.545 |
| MDRS Memory | 0.695 |
| MDRS Conceptualization | −0.222 |
| COT | −0.691 |
| TMT-A | −0.109 |
| TMT B-A | 0.200 |
| IST | 0.701 |
| DMS-48 | 0.390 |
| DO-30 | 0.241 |
| BDI | −0.768 * |
| HDRS-21 | −0.412 |
| STAI-YA | −0.337 |
| STAI-YB | −0.736 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teti Mayer, J.; Masse, C.; Chopard, G.; Nicolier, M.; Bereau, M.; Magnin, E.; Monnin, J.; Tio, G.; Haffen, E.; Vandel, P.; et al. Repetitive Transcranial Magnetic Stimulation as an Add-On Treatment for Cognitive Impairment in Alzheimer’s Disease and Its Impact on Self-Rated Quality of Life and Caregiver’s Burden. Brain Sci. 2021, 11, 740. https://doi.org/10.3390/brainsci11060740
Teti Mayer J, Masse C, Chopard G, Nicolier M, Bereau M, Magnin E, Monnin J, Tio G, Haffen E, Vandel P, et al. Repetitive Transcranial Magnetic Stimulation as an Add-On Treatment for Cognitive Impairment in Alzheimer’s Disease and Its Impact on Self-Rated Quality of Life and Caregiver’s Burden. Brain Sciences. 2021; 11(6):740. https://doi.org/10.3390/brainsci11060740
Chicago/Turabian StyleTeti Mayer, Juliana, Caroline Masse, Gilles Chopard, Magali Nicolier, Matthieu Bereau, Eloi Magnin, Julie Monnin, Gregory Tio, Emmanuel Haffen, Pierre Vandel, and et al. 2021. "Repetitive Transcranial Magnetic Stimulation as an Add-On Treatment for Cognitive Impairment in Alzheimer’s Disease and Its Impact on Self-Rated Quality of Life and Caregiver’s Burden" Brain Sciences 11, no. 6: 740. https://doi.org/10.3390/brainsci11060740
APA StyleTeti Mayer, J., Masse, C., Chopard, G., Nicolier, M., Bereau, M., Magnin, E., Monnin, J., Tio, G., Haffen, E., Vandel, P., & Bennabi, D. (2021). Repetitive Transcranial Magnetic Stimulation as an Add-On Treatment for Cognitive Impairment in Alzheimer’s Disease and Its Impact on Self-Rated Quality of Life and Caregiver’s Burden. Brain Sciences, 11(6), 740. https://doi.org/10.3390/brainsci11060740






