Effective Connectivity Analysis of Brain Activated Regions during Distracted Driving
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
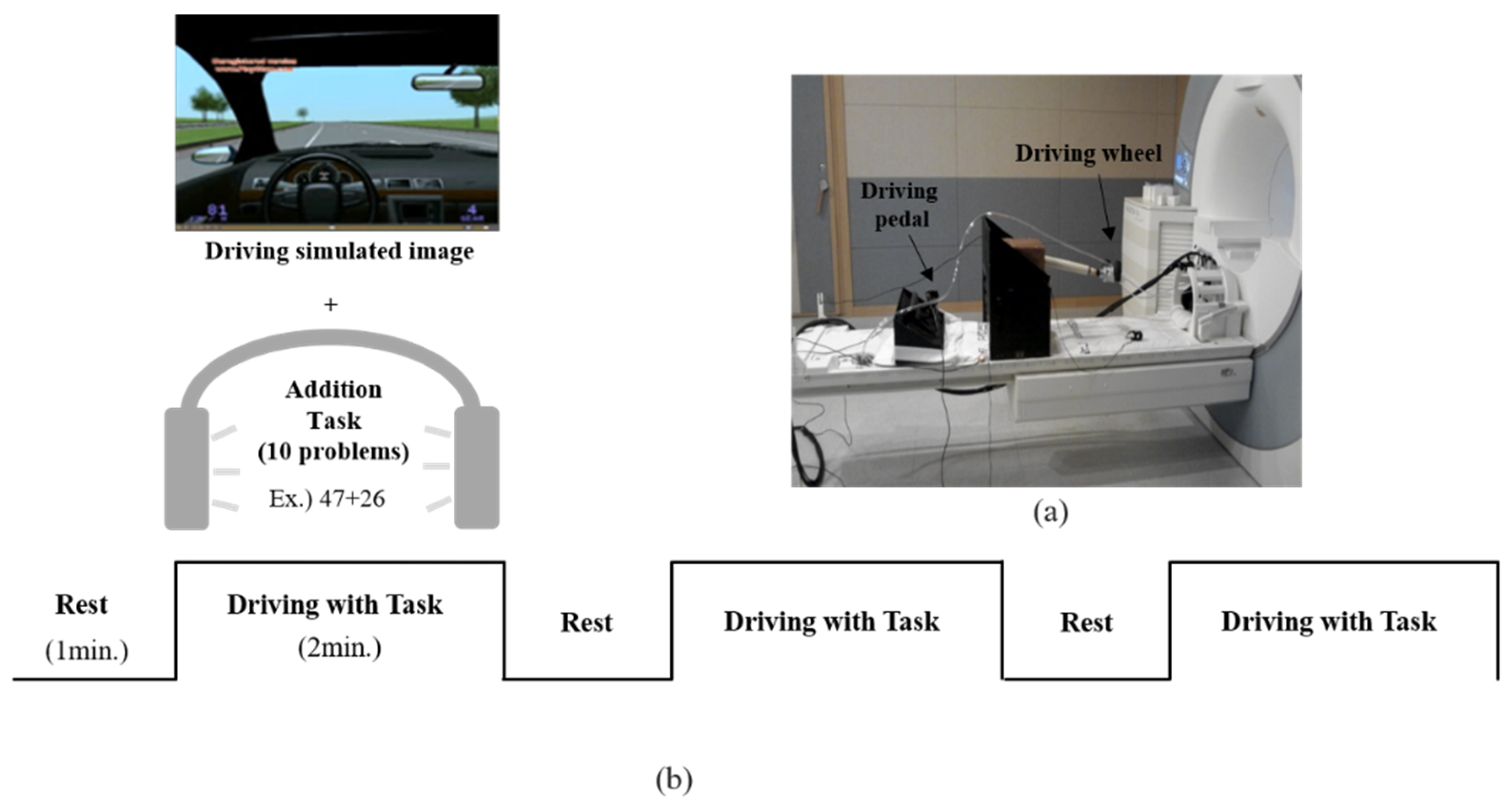
2.2. MR-Compatible Driving Simulator
2.3. Experimental Design
2.4. Image Acquisition
2.5. Image Analysis
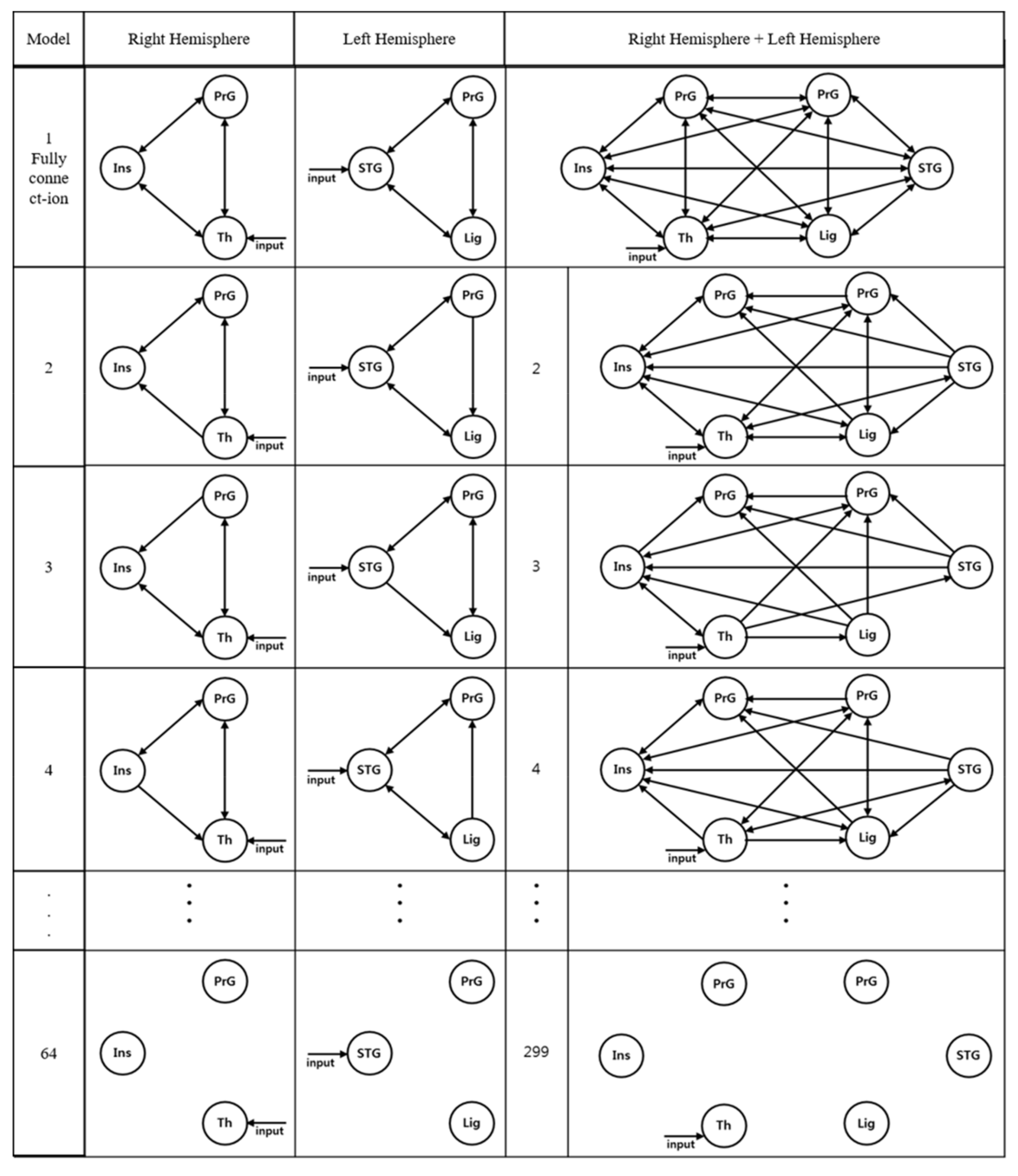
2.6. Connectivity Analysis
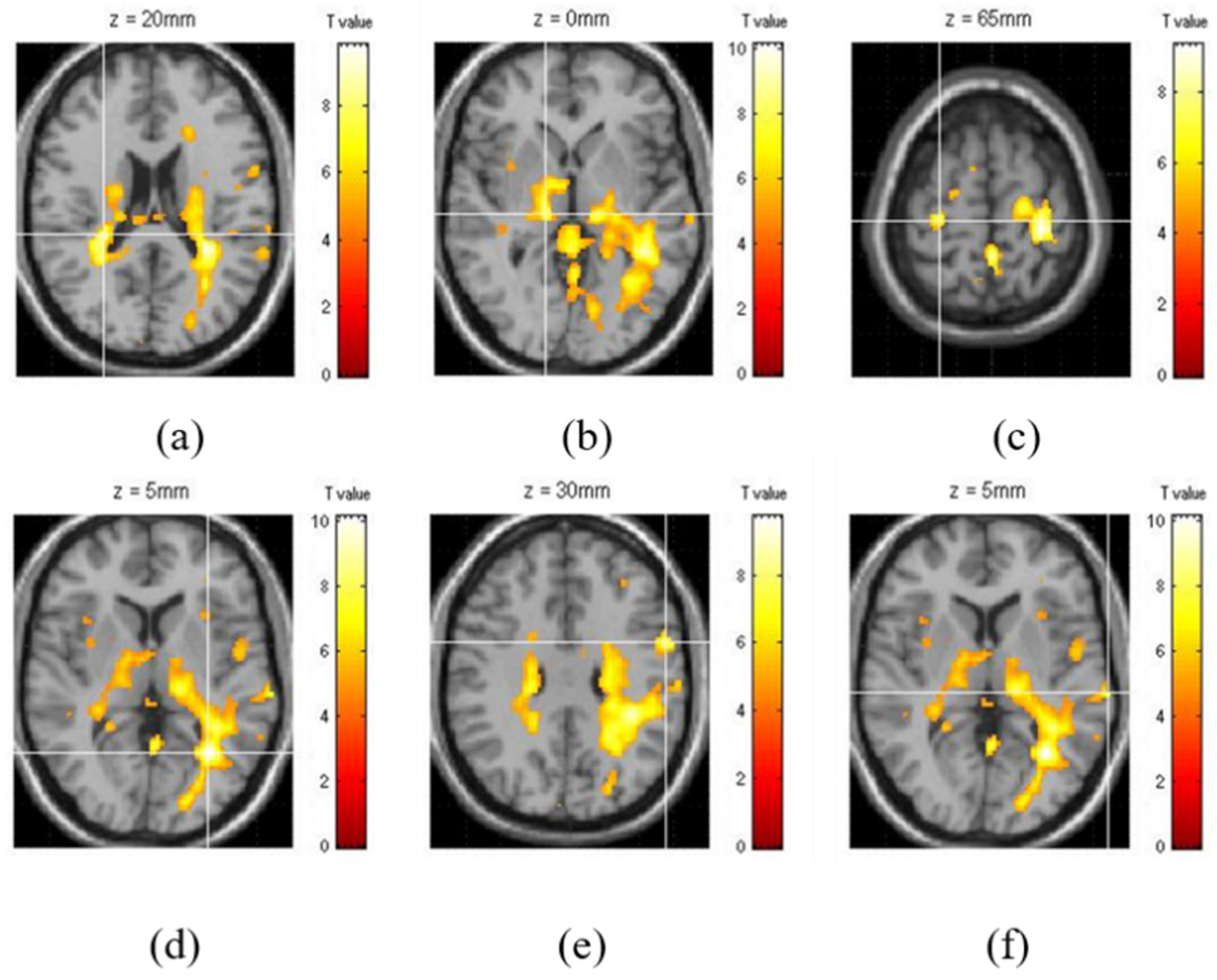
3. Results
4. Discussion
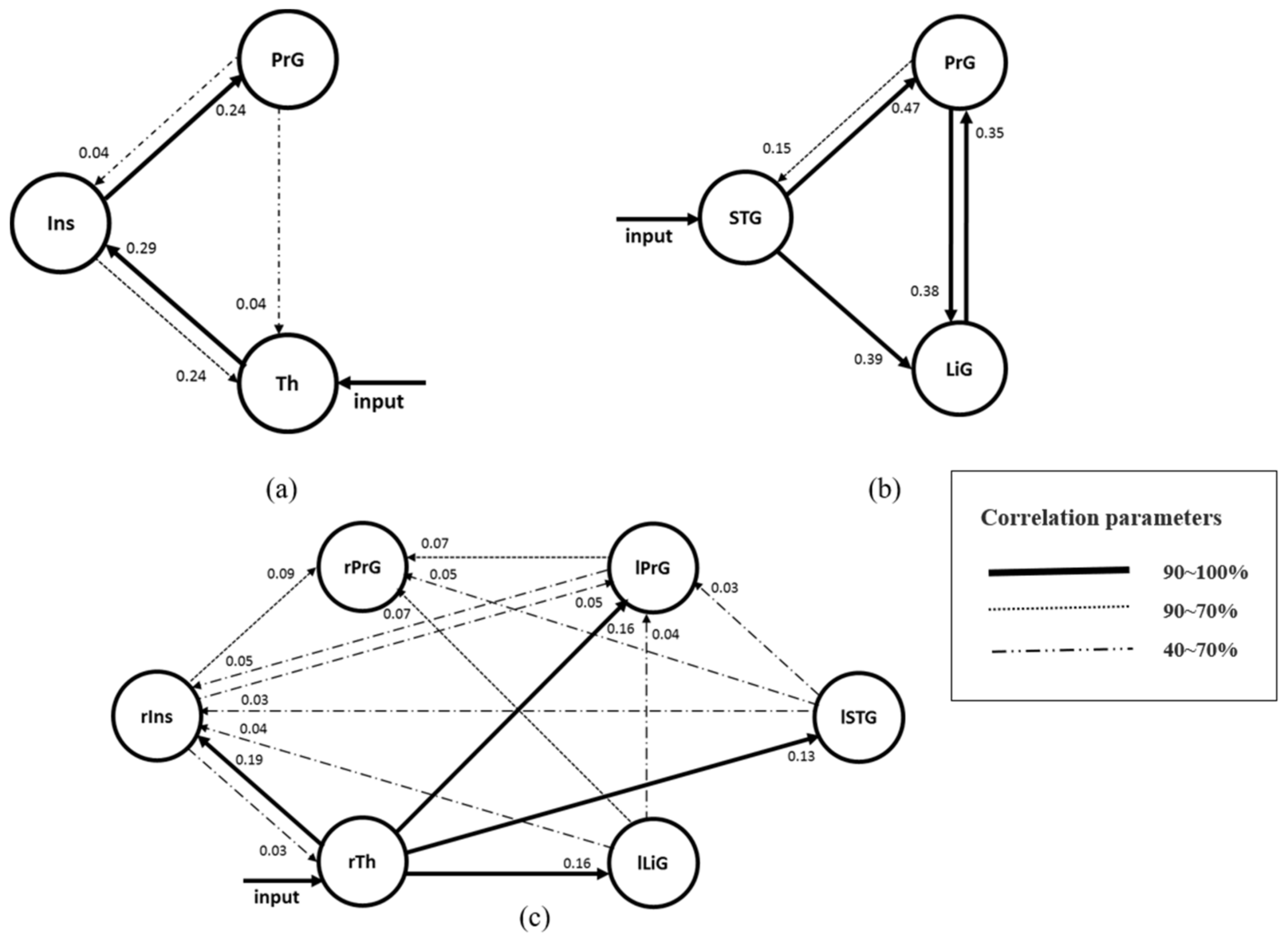
4.1. Effective Connectivity between Regions Activated in the Right Hemisphere
4.2. Effective Connectivity between Regions Activated in the Left Hemisphere
4.3. Effective Connectivity between Regions Activated in Both Hemispheres
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hsieh, L.; Young, R.A.; Bowyer, S.M.; Maran, J.E.; Genik, R.J., II. Conversation effects on neural mechanisms underlying reaction time to visual events while viewing a driving scene: fRI analysis and asynchrony model. Brain Res. 2009, 1251, 162–175. [Google Scholar] [CrossRef]
- Newman, S.D.; Keller, T.A.; Just, M.A. Volitional control of attention and brain activation in dual task performance. Hum. Brain Mapp. 2007, 28, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Mader, M.; Bresges, A.; Topal, R.; Busse, A.; Forsting, M.; Gizewski, E. Simulated car driving in fMRI-cerebral activation patterns driving an unfamiliar and a familiar route. Neurosci. Lett. 2009, 464, 222–227. [Google Scholar] [CrossRef]
- Uchiyama, Y.; Ebe, K.; Kozato, A.; Okada, T.; Sadato, N. The neural substrates of driving at a safe distance: A functional MRI study. Neurosci. Lett. 2003, 352, 199–202. [Google Scholar] [CrossRef] [PubMed]
- Uchiyama, Y.; Toyoda, H.; Sakai, H.; Shin, D.; Ebe, K.; Sadato, N. Suppression of brain activity related to a car-following task with an auditory task: An fMRI study. Transp. Res. Part F Traffic Psychol. Behav. 2012, 15, 25–37. [Google Scholar] [CrossRef]
- Michon, J.A. Explanatory pitfalls and rule-based driver models. Accid. Anal. Prev. 1989, 21, 341–353. [Google Scholar] [CrossRef]
- Graydon, F.X.; Young, R.; Benton, M.D.; Genik, R.J., II; Posse, S.; Hsieh, L.; Green, C. Visual event detection during simulated driving: Identifying the neural correlates with functional neuroimaging. Transp. Res. Part F Traffic Psychol. Behav. 2004, 7, 271–286. [Google Scholar] [CrossRef]
- Just, M.A.; Keller, T.A.; Cynkar, J. A decrease in brain activation associated with driving when listening to someone speak. Brain Res. 2008, 1205, 70–80. [Google Scholar] [CrossRef]
- Young, R.A.; Hsieh, L.; Graydon, F.X.; Genik, R.J., II; Benton, M.D.; Green, C.C.; Bowyer, S.M.; Moran, J.E.; Tepley, N. Mind-on-the-Drive: Real-time functional neuroimaging of cognitive brain mechanisms underlying driver performance and distraction. SAE 2005 World Congr. Exhib. 2005, 114, 454–472. [Google Scholar]
- Wang, L.; Liu, Q.; Shen, H.; Li, H.; Hu, D. Large-scale functional brain network changes in taxi drivers: Evidence from resting-state fMRI. Hum. Brain Mapp. 2015, 36, 862–871. [Google Scholar] [CrossRef]
- Shen, H.; Li, Z.; Qina, J.; Liu, Q.; Wang, L.; Zeng, L.L.; Li, H.; Hu, D. Changes in functional connectivity dynamics associated with vigilance network in taxi drivers. NeuroImage 2016, 124, 367–378. [Google Scholar] [CrossRef]
- Choi, M.H.; Kim, H.S.; Chung, S.C. Evaluation of Effective Connectivity Between Brain Areas Activated During Simulated Driving Using Dynamic Causal Modeling. Front. Behav. Neurosci. 2020, 23, 158. [Google Scholar] [CrossRef] [PubMed]
- Corbetta, M.; Kincade, J.M.; Shulman, G.L. Neural systems for visual orienting and their relationships to spatial working memory. J. Cogn. Neurosci. 2002, 14, 508–523. [Google Scholar] [CrossRef]
- Schall, J.D. On the role of frontal eye field in guiding attention and saccades. Vision Res. 2004, 44, 1453–1467. [Google Scholar] [CrossRef] [PubMed]
- Mustari, M.J.; Ono, S.; Das, V.E. Signal processing and distribution in cortical-brainstem pathways for smooth pursuit eye movements. Ann. N. Y. Acad. Sci. 2009, 1164, 147–151. [Google Scholar] [CrossRef] [PubMed]
- Bush, G.; Luu, P.; Posner, M.I. Cognitive and emotional influences in anterior cingulate cortex. Trends Cognit. Sci. 2000, 4, 215–222. [Google Scholar] [CrossRef]
- Gharib, S.; Zare-Sadeghi, A.; Zakerian, S.A.; Haidari, M.R. The neural basis of hazard perception differences between novice and experienced drivers—An fMRI study. Excli. J. 2020, 19, 547–566. [Google Scholar] [PubMed]
- Yan, L.; Wen, T.; Zhang, J.; Chang, L.; Wang, Y.; Liu, M.; Ding, C.; Yan, F. An Evaluation of Executive Control Function and Its Relationship with Driving Performance. Sensors 2021, 21, 1763. [Google Scholar] [CrossRef]
- Chen, P.; Chen, F.; Zhang, L.; Ma, X.; Pan, X. Examining the influence of decorated sidewaall in road tunnels using fMRI technology. Tunn. Undergr. Space Technol. 2020, 99, 103362. [Google Scholar] [CrossRef]
- Alexander, M.C.; Sherrilene, C.; Desiree, L.; Irene, A.M.; Michael, S.O.; Aparna, W.S.; Nikolaus, R.M. Driving Performance and Behaviors: A Comparison of Gender Differences in Parkinson’s Disease. Traffic Inj. Prev. 2013, 14, 340–345. [Google Scholar]
- Cordellieri, P.; Baralla, F.; Ferlazzo, F.; Sgalla, R.; Piccardi, L.; Giannini, A.M. Gender Effects in Young Road Users on Road Safety Attitudes, Behaviors and Risk Perception. Front. Psychol. 2016, 27, 1412. [Google Scholar] [CrossRef] [PubMed]
- Oldfield, R.C. The assessment and analysis of handedness: The Edinburgh inventory. Neuropsychologia 1971, 9, 97–113. [Google Scholar] [CrossRef]
- Friston, K.J.; Buchel, C. Attentional modulation of effective connectivity from V2 to V5/MT in humans. Proc. Natl. Acad. Sci. USA 2000, 97, 7591–7596. [Google Scholar] [CrossRef]
- Evarts, E.V.; Thach, W.T. Motor Mechanisms of the CNS: Cerebrocerebellar Interrelations. Annu. Rev. Physiol. 1969, 31, 451–498. [Google Scholar] [CrossRef]
- Kunimatsu, J.; Tanaka, M. Roles of the primate motor thalamus in the generation of antisaccades. J. Neurosci. 2010, 30, 5108–5117. [Google Scholar] [CrossRef]
- Anderson, T.J.; Jenkins, I.H.; Brooks, D.J.; Hawken, M.B.; Frackowiak, R.S.; Kennard, C. Cortical control of saccades and fixation in man A PET study. Brain 1994, 117, 1073–1084. [Google Scholar] [CrossRef] [PubMed]
- Fink, G.R.; Frackowiak, R.S.; Pietrzyk, U.; Passingham, R.E. Multiple nonprimary motor areas in the human cortex. J. Neurophysiol. 1997, 77, 2164–2174. [Google Scholar] [CrossRef]
- Goulding, M. Circuits controlling vertebrate locomotion: Moving in a new direction. Nat. Rev. Neurosci. 2009, 10, 507–518. [Google Scholar] [CrossRef]
- Uddin, L.Q.; Nomi, J.S.; Hebert-Seropian, B.; Ghaziri, J.; Boucher, O. Structure and function of the human insula. J. Clin. Neurophysiol. 2017, 34, 300–306. [Google Scholar] [CrossRef]
- Cho, Y.T.; Fromm, S.; Guyer, A.E.; Detloff, A.; Pine, D.S.; Fudge, J.L.; Ernst, M. Nucleus accumbens, thalamus and insula connectivity during incentive anticipation in typical adults and adolescents. Neuroimage 2013, 1, 508–521. [Google Scholar] [CrossRef]
- Howard, M.A.; Volkov, I.O.; Mirsky, R.; Garell, P.C.; Noh, M.D.; Granner, M.; Damasio, H.; Steinschneider, M.; Reale, R.A.; Hind, J.E.; et al. Auditory cortex on the human posterior superior temporal gyrus. J. Comp. Neurol. 2000, 416, 79–92. [Google Scholar] [CrossRef]
- Choi, M.H.; Kim, H.S.; Yoon, H.J.; Lee, J.C.; Baek, J.H.; Choi, J.S.; Tack, G.R.; Min, B.C.; Lim, D.W.; Chung, S.C. Increase in brain activation due to sub-tasks during driving: fMRI study using new MR-compatible driving simulator. J. Physiol. Anthropol. 2017, 36, 11. [Google Scholar] [CrossRef]
- Abd Hamid, A.I.; Yusoff, A.N.; Mukari, S.Z.S.; Mohamad, M. Brain Activation during Addition and Subtraction Tasks In-Noise and In-Quiet. Malays J. Med. Sci. 2011, 18, 3–15. [Google Scholar]
- Banker, L.; Tadi, P. Neuroanatomy, Precentral Gyrus; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Zhang, H.; Liu, J.; Zhang, Q. Neural representations for the generation of inventive conceptions inspired by adaptive feature optimization of biological species. Cortex 2014, 50, 162–173. [Google Scholar] [CrossRef]
- Malouin, F.; Richards, C.L.; Jackson, P.L.; Dumas, F.; Doyon, J. Brain activations during motor imagery of locomotor-related tasks: A PET study. Hum. Brain Mapp. 2003, 19, 47–62. [Google Scholar] [CrossRef] [PubMed]
- Macaluso, E.; Driver, J. Spatial attention and crossmodal interactions between vision and touch. Neuropsychologia 2001, 39, 1304–1316. [Google Scholar] [CrossRef]
- Astafiev, S.V.; Stanley, C.M.; Shulman, G.L.; Corbetta, M. Extrastriate body area in human occipital cortex responds to the performance of motor actions. Nat. Neurosci. 2004, 7, 542–548. [Google Scholar] [CrossRef]
- Bremner, J.D.; Vythilingam, M.; Vermetten, E.; Vaccarino, V.; Charney, D.S. Deficits in hippocampal and anterior cingulate functioning during verbal declarative memory encoding in midlife major depression. Am. J. Psychiat. 2004, 161, 637–645. [Google Scholar] [CrossRef] [PubMed]
- Kitada, R.; Johnsrude, I.S.; Kochiyama, T.; Lederman, S.J. Brain networks involved in haptic and visual identification of facial expressions of emotion: An fMRI study. NeuroImage 2010, 49, 1677–1689. [Google Scholar] [CrossRef] [PubMed]
- Mechelli, A.; Humphreys, G.W.; Mayall, K.; Olson, A.; Price, C.J. Differential effects of word length and visual contrast in the fusiform and lingual gyri during reading. Proc. Biol. Sci. 2000, 267, 1909–1913. [Google Scholar] [CrossRef] [PubMed]
- Kozlovskiy, S.A.; Pyasik, M.M.; Korotkova, A.V.; Vartanov, A.V.; Glozman, J.M.; Kiselnikov, A.A. Activation of left lingual gyrus related to working memory for schematic faces. Int. J. Psychophysiol. 2014, 94, 241. [Google Scholar] [CrossRef]
- Borkowski, K.; Krzyzak, A.T. The generalized Stejskal-Tanner equation for non-uniform magnetic field gradients. J. Magn. Reson. 2018, 296, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Krzyzak, A.T.; Olejniczak, Z. Improving the accuracy of PGSE DTI experiments using the spatial distribution of b matrix. Magn. Reson. Imag. 2015, 33, 286–295. [Google Scholar] [CrossRef] [PubMed]
| From | |||||
| Ins | Th | PrG | |||
| Right Hemisphere | To | Ins | 0.29 (100%) | 0.04 (57%) | |
| Th | 0.24 (83%) | 0.04 (57%) | |||
| PrG | 0.24 (96%) | 0 | |||
| From | |||||
| LiG | PrG | STG | |||
| Left Hemisphere | To | LiG | 0.38 (96%) | 0.39 (98%) | |
| PrG | 0.35 (94%) | 0.47 (99%) | |||
| STG | 0 | 0.15 (74%) | |||
| From | |||||||
|---|---|---|---|---|---|---|---|
| rIns | rTh | rPrG | lLiG | lPrG | lSTG | ||
| To | rIns | 0.19 (100%) | 0 | 0.04 (62%) | 0.05 (64%) | 0.03 (60%) | |
| rTh | 0.03 (58%) | 0 | 0 | 0 | 0 | ||
| rPrG | 0.09 (76%) | 0 | 0.07 (70%) | 0.07 (72%) | 0.05 (58%) | ||
| lLiG | 0 | 0.16 (100%) | 0 | 0 | 0 | ||
| lPrG | 0.05 (64%) | 0.16 (99%) | 0 | 0.04 (60%) | 0.03 (65%) | ||
| lSTG | 0 | 0.13 (99%) | 0 | 0 | 0 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, M.-H.; Jung, J.-J.; Lee, J.-H.; Kim, Y.-J.; Kim, K.-B.; Kim, H.-S.; Yi, J.-H.; Chung, S.-C. Effective Connectivity Analysis of Brain Activated Regions during Distracted Driving. Brain Sci. 2021, 11, 690. https://doi.org/10.3390/brainsci11060690
Choi M-H, Jung J-J, Lee J-H, Kim Y-J, Kim K-B, Kim H-S, Yi J-H, Chung S-C. Effective Connectivity Analysis of Brain Activated Regions during Distracted Driving. Brain Sciences. 2021; 11(6):690. https://doi.org/10.3390/brainsci11060690
Chicago/Turabian StyleChoi, Mi-Hyun, Jin-Ju Jung, Je-Hyeop Lee, Ye-Jin Kim, Kyu-Beom Kim, Hyung-Sik Kim, Jeong-Han Yi, and Soon-Cheol Chung. 2021. "Effective Connectivity Analysis of Brain Activated Regions during Distracted Driving" Brain Sciences 11, no. 6: 690. https://doi.org/10.3390/brainsci11060690
APA StyleChoi, M.-H., Jung, J.-J., Lee, J.-H., Kim, Y.-J., Kim, K.-B., Kim, H.-S., Yi, J.-H., & Chung, S.-C. (2021). Effective Connectivity Analysis of Brain Activated Regions during Distracted Driving. Brain Sciences, 11(6), 690. https://doi.org/10.3390/brainsci11060690






