Resting-State Functional Connectivity in Mathematical Expertise
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Resting-State fMRI Acquisition
2.3. Resting-State fMRI Data Analysis
3. Results
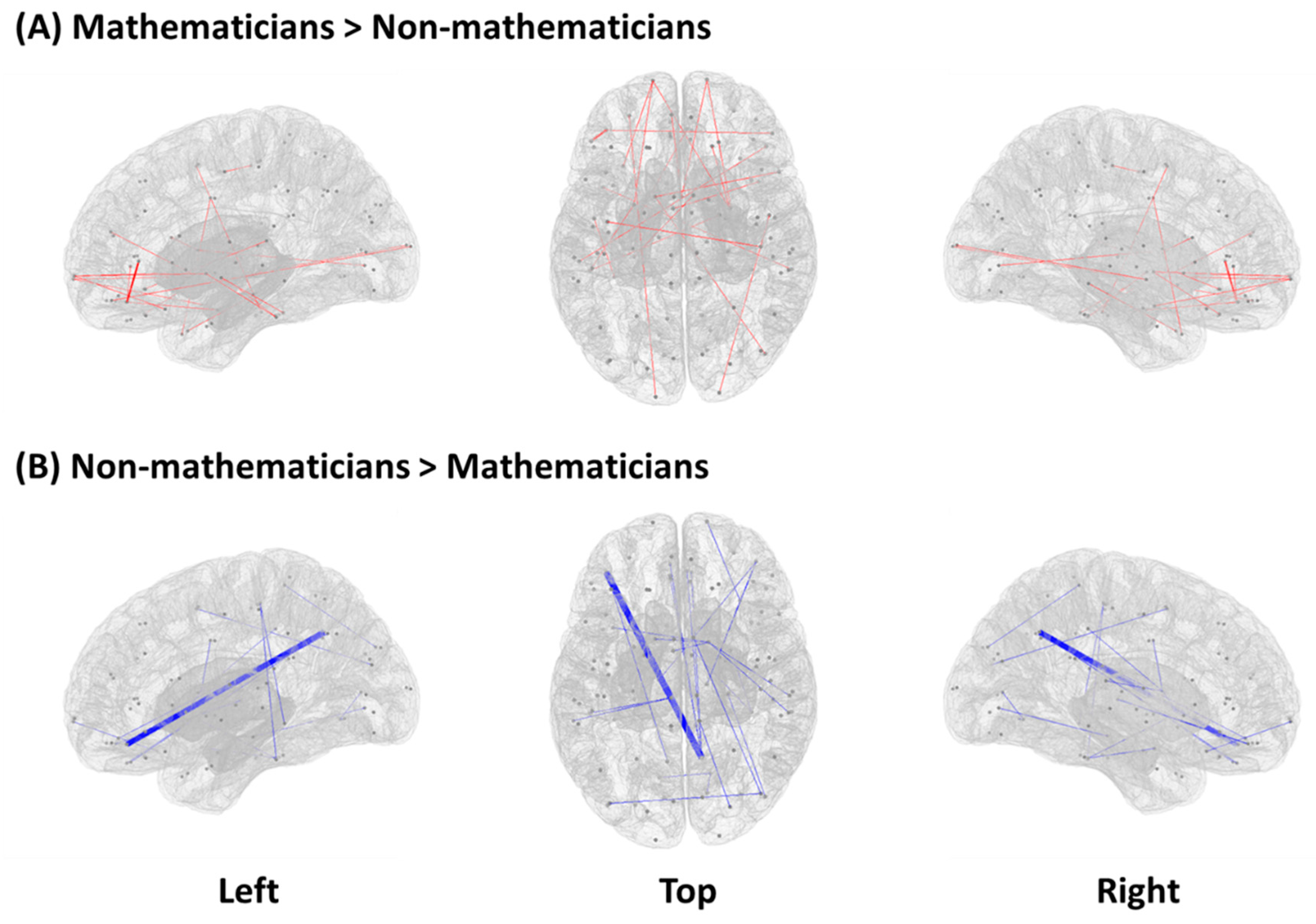
3.1. Functional Connectivity between Mathematicians and Non-Mathematicians
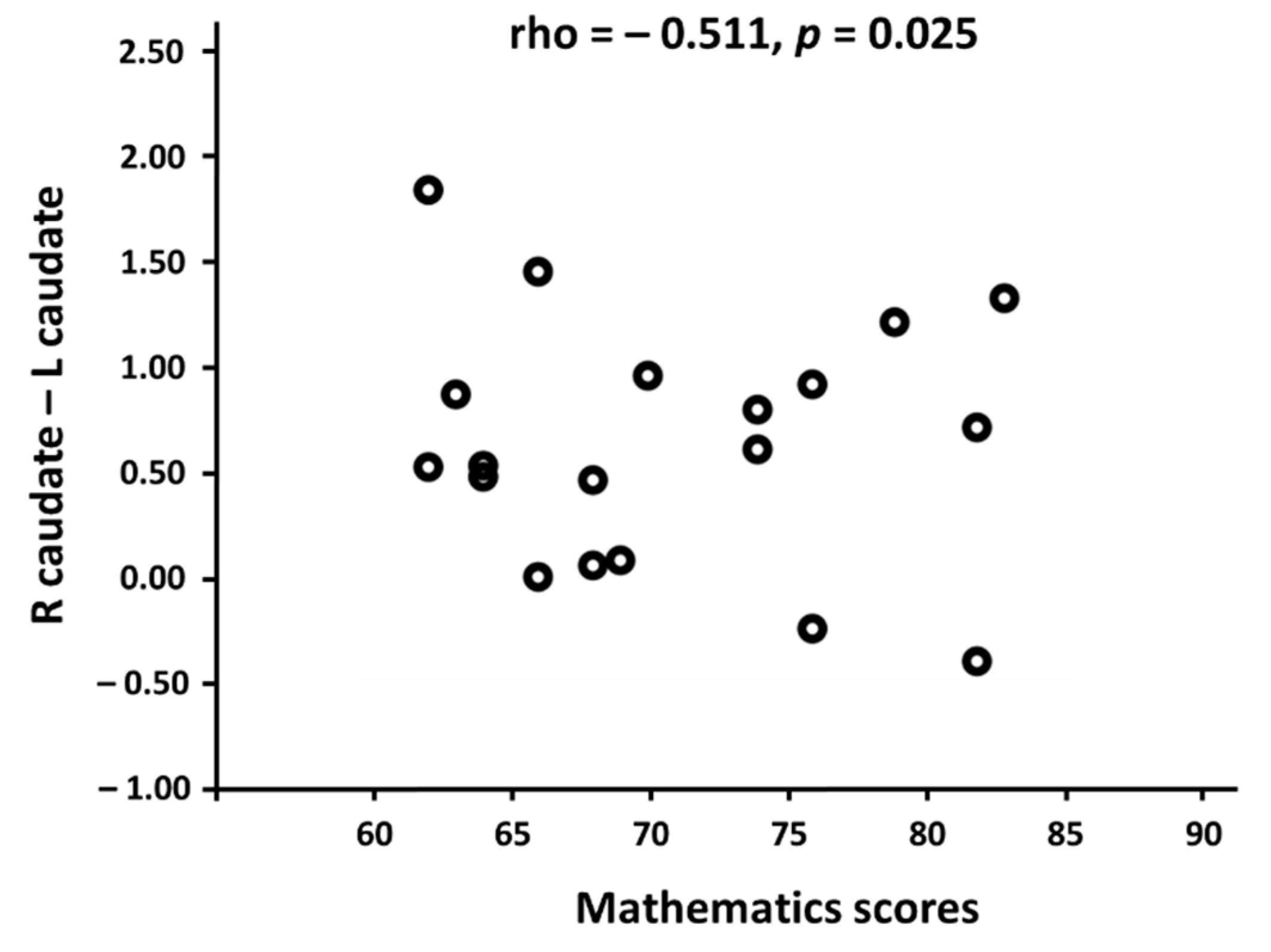
3.2. Linking Functional Connectivity to Mathematics Scores
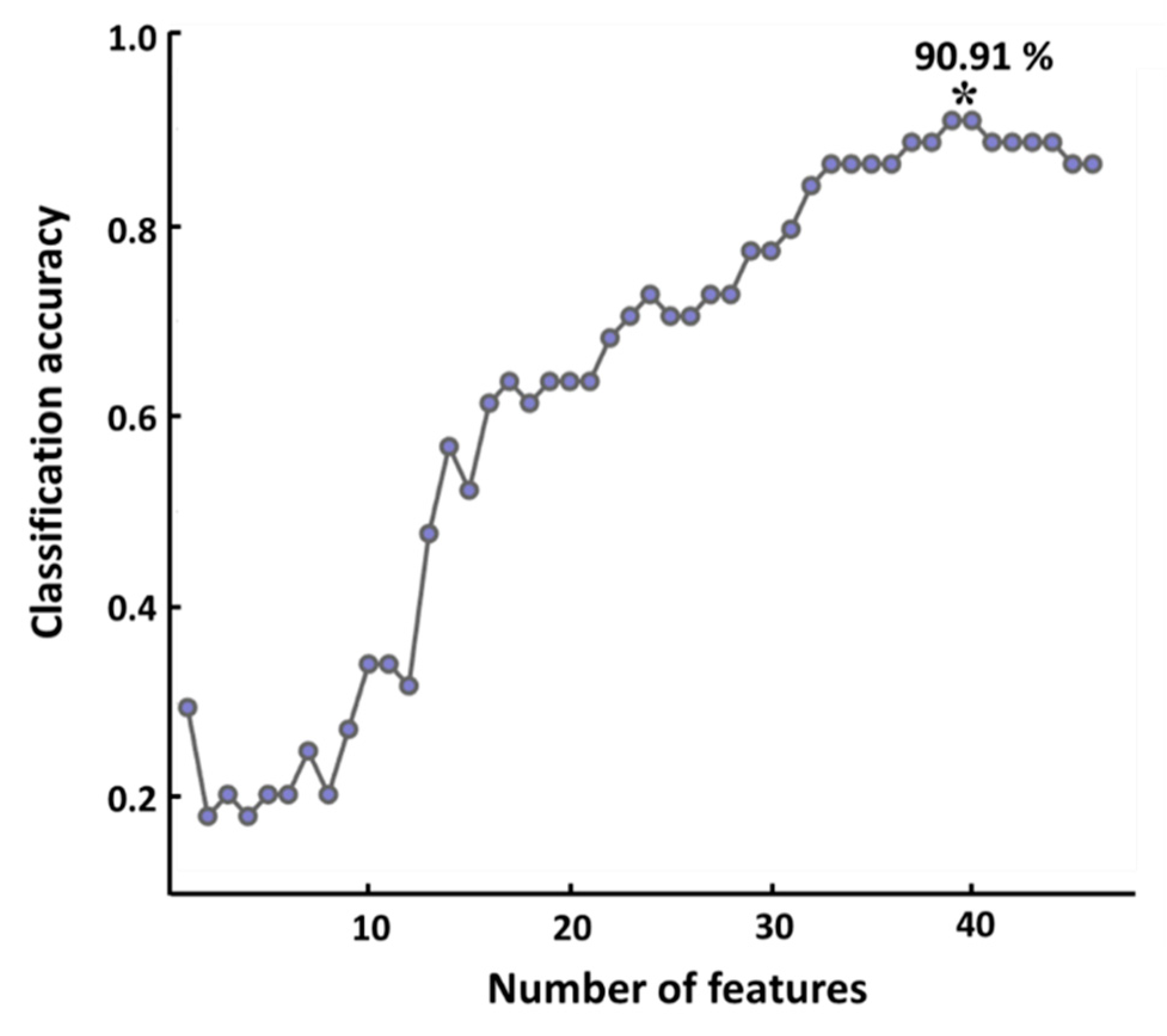
3.3. Classification Performance
4. Discussion
4.1. Resting-State Functional Connectivity for Detecting Group-Specific Features
4.2. Functional Connectivity in Mathematicians
4.3. Mathematicians’ Preconfigured Functional Connectivity for Their Expertise
4.4. Neural Efficiency Correlated with the Functional Connectivity in the Caudate Nucleus
4.5. Classification Accuracy
4.6. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Jeon, H.-A.; Friederici, A.D. What Does “Being an Expert” Mean to the Brain? Functional Specificity and Connectivity in Expertise. Cereb. Cortex 2017, 27, 5603–5615. [Google Scholar] [CrossRef]
- Jeon, H.-A.; Kuhl, U.; Friederici, A.D. Mathematical expertise modulates the architecture of dorsal and cortico-thalamic white matter tracts. Sci. Rep. 2019, 9, 6825. [Google Scholar] [CrossRef]
- Menon, V. Developmental cognitive neuroscience of arithmetic: Implications for learning and education. ZDM 2010, 42, 515–525. [Google Scholar] [CrossRef]
- Duncan, G.J.; Dowsett, C.J.; Claessens, A.; Magnuson, K.; Huston, A.C.; Klebanov, P.; Pagani, L.S.; Feinstein, L.; Engel, M.; Brooks-Gunn, J. School readiness and later achievement. Dev. Psychol. 2007, 43, 1428. [Google Scholar] [CrossRef] [PubMed]
- Biswal, B.; Yetkin, F.Z.; Haughton, V.M.; Hyde, J.S. Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magn. Reason. Med. 1995, 34, 537–541. [Google Scholar] [CrossRef]
- Fair, D.A.; Schlaggar, B.L.; Cohen, A.L.; Miezin, F.M.; Dosenbach, N.U.; Wenger, K.K.; Fox, M.D.; Snyder, A.Z.; Raichle, M.E.; Petersen, S.E. A method for using blocked and event-related fMRI data to study “resting state” functional connectivity. Neuroimage 2007, 35, 396–405. [Google Scholar] [CrossRef]
- Fox, M.D.; Raichle, M.E. Spontaneous fluctuations in brain activity observed with functional magnetic resonance imaging. Nat. Rev. Neurosci. 2007, 8, 700–711. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Gohel, S.; Vachha, B. Current methods and new directions in resting state fMRI. Clin. Imaging 2020, 65, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Damaraju, E.; Phillips, J.; Lowe, J.R.; Ohls, R.; Calhoun, V.D.; Caprihan, A. Resting-state functional connectivity differences in premature children. Front. Syst. Neurosci. 2010, 4. [Google Scholar] [CrossRef] [PubMed]
- Smitha, K.; Akhil Raja, K.; Arun, K.; Rajesh, P.; Thomas, B.; Kapilamoorthy, T.; Kesavadas, C. Resting state fMRI: A review on methods in resting state connectivity analysis and resting state networks. Neuroradiol. J. 2017, 30, 305–317. [Google Scholar] [CrossRef]
- Cordes, D.; Haughton, V.; Arfanakis, K.; Wendt, G.; Turski, P.A.; Moritz, C.; Quigley, M.A.; Meyerand, M. Mapping functionally related regions of brain with functional connectivity MRI (fcMRI). Am. J. Neuroradiol. 2000, 21, 1636–1644. [Google Scholar] [PubMed]
- Lowe, M.J.; Mock, B.J.; Sorenson, J.A. Functional connectivity in single and multislice echoplanar imaging using resting-state fluctuations. Neuroimage 1998, 7, 119–132. [Google Scholar] [CrossRef]
- Fox, M.; Snyder, A.; Vincent, J.; Corbetta, M.; Van Essen, D.; Raichle, M. From The Cover: The human brain is intrinsically organized into dynamic, anticorrelated functional networks. Proc. Natl. Acad. Sci. USA 2005, 102, 9673–9678. [Google Scholar] [CrossRef]
- Greicius, M.D.; Krasnow, B.; Reiss, A.L.; Menon, V. Functional connectivity in the resting brain: A network analysis of the default mode hypothesis. Proc. Natl. Acad. Sci. USA 2003, 100, 253–258. [Google Scholar] [CrossRef]
- Fransson, P. Spontaneous low-frequency BOLD signal fluctuations: An fMRI investigation of the resting-state default mode of brain function hypothesis. Hum. Brain. Mapp. 2005, 26, 15–29. [Google Scholar] [CrossRef] [PubMed]
- Laufs, H.; Krakow, K.; Sterzer, P.; Eger, E.; Beyerle, A.; Salek-Haddadi, A.; Kleinschmidt, A. Electroencephalographic signatures of attentional and cognitive default modes in spontaneous brain activity fluctuations at rest. Proc. Natl. Acad. Sci. USA 2003, 100, 11053–11058. [Google Scholar] [CrossRef] [PubMed]
- Rombouts, S.A.; Stam, C.J.; Kuijer, J.P.; Scheltens, P.; Barkhof, F. Identifying confounds to increase specificity during a “no task condition”. Evidence for hippocampal connectivity using fMRI. Neuroimage 2003, 20, 1236–1245. [Google Scholar] [CrossRef]
- Vincent, J.; Snyder, A.; Fox, M.; Shannon, B.; Andrews-Hanna, J.; Raichle, M.; Buckner, R. Coherent spontaneous activity identifies a hippocampal-parietal memory network. J. Neurophysiol. 2007, 96, 3517–3531. [Google Scholar] [CrossRef]
- Hampson, M.; Peterson, B.S.; Skudlarski, P.; Gatenby, J.C.; Gore, J.C. Detection of functional connectivity using temporal correlations in MR images. Hum. Brain. Mapp. 2002, 15, 247–262. [Google Scholar] [CrossRef]
- Fox, M.D.; Corbetta, M.; Snyder, A.Z.; Vincent, J.L.; Raichle, M.E. Spontaneous neuronal activity distinguishes human dorsal and ventral attention systems. Proc. Natl. Acad. Sci. USA 2006, 103, 10046–10051. [Google Scholar] [CrossRef]
- Sridharan, D.; Levitin, D.J.; Menon, V. A critical role for the right fronto-insular cortex in switching between central-executive and default-mode networks. Proc. Natl. Acad. Sci. USA 2008, 105, 12569–12574. [Google Scholar] [CrossRef]
- Christoff, K.; Gordon, A.M.; Smallwood, J.; Smith, R.; Schooler, J.W. Experience sampling during fMRI reveals default network and executive system contributions to mind wandering. Proc. Natl. Acad. Sci. USA 2009, 106, 8719–8724. [Google Scholar] [CrossRef] [PubMed]
- Taylor, K.S.; Seminowicz, D.A.; Davis, K.D. Two systems of resting state connectivity between the insula and cingulate cortex. Hum. Brain. Mapp. 2009, 30, 2731–2745. [Google Scholar] [CrossRef]
- Seeley, W.W.; Menon, V.; Schatzberg, A.F.; Keller, J.; Glover, G.H.; Kenna, H.; Reiss, A.L.; Greicius, M.D. Dissociable intrinsic connectivity networks for salience processing and executive control. J. Neurosci. 2007, 27, 2349–2356. [Google Scholar] [CrossRef] [PubMed]
- Yeo, B.T.; Krienen, F.M.; Sepulcre, J.; Sabuncu, M.R.; Lashkari, D.; Hollinshead, M.; Roffman, J.L.; Smoller, J.W.; Zollei, L.; Polimeni, J.R.; et al. The organization of the human cerebral cortex estimated by intrinsic functional connectivity. J. Neurophysiol. 2011, 106, 1125–1165. [Google Scholar] [CrossRef]
- Chen, B.; Xu, T.; Zhou, C.; Wang, L.; Yang, N.; Wang, Z.; Dong, H.-M.; Yang, Z.; Zang, Y.-F.; Zuo, X.-N.; et al. Individual variability and test-retest reliability revealed by ten repeated resting-state brain scans over one month. PLoS ONE 2015, 10, e0144963. [Google Scholar] [CrossRef] [PubMed]
- Shen, H.H. Core Concept: Resting-state connectivity. Proc. Natl. Acad. Sci. USA 2015, 112, 14115–14116. [Google Scholar] [CrossRef]
- Wang, X.; Wang, R.; Li, F.; Lin, Q.; Zhao, X.; Hu, Z. Large-scale granger causal brain network based on resting-state fMRI data. Neuroscience 2020, 425, 169–180. [Google Scholar] [CrossRef]
- Sherman, L.E.; Rudie, J.D.; Pfeifer, J.H.; Masten, C.L.; McNealy, K.; Dapretto, M. Development of the default mode and central executive networks across early adolescence: A longitudinal study. Dev. Cogn. Neurosci. 2014, 10, 148–159. [Google Scholar] [CrossRef]
- Ming, S.; Yong, L.; Yuan, Z.; Kun, W.; Chunshui, Y.; Tianzi, J. Default network and intelligence difference. IEEE Trans. Auton. Ment. Dev. 2009, 1, 101–109. [Google Scholar] [CrossRef]
- Suprano, I.; Delon-Martin, C.; Kocevar, G.; Stamile, C.; Hannoun, S.; Achard, S.; Badhwar, A.; Fourneret, P.; Revol, O.; Nusbaum, F.; et al. Topological modification of brain networks organization in children with high intelligence quotient: A resting-state fMRI study. Front. Hum. Neurosci. 2019, 13, 450. [Google Scholar] [CrossRef] [PubMed]
- Reineberg, A.E.; Gustavson, D.E.; Benca, C.; Banich, M.T.; Friedman, N.P. The relationship between resting state network connectivity and individual differences in executive functions. Front. Psychol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Desmond, J.E.; Glover, G.H. Estimating sample size in functional MRI (fMRI) neuroimaging studies: Statistical power analyses. J. Neurosci. Methods 2002, 118, 115–128. [Google Scholar] [CrossRef]
- Yeung, A.W. An updated survey on statistical thresholding and sample size of fMRI studies. Front. Hum. Neurosci. 2018, 12, 16. [Google Scholar] [CrossRef]
- Yeung, A.W.; Wong, N.S.; Eickhoff, S.B. Empirical assessment of changing sample-characteristics in task-fMRI over two decades: An example from gustatory and food studies. Hum. Brain. Mapp. 2020, 41, 2460–2473. [Google Scholar] [CrossRef] [PubMed]
- Orru, G.; Pettersson-Yeo, W.; Marquand, A.F.; Sartori, G.; Mechelli, A. Using support vector machine to identify imaging biomarkers of neurological and psychiatric disease: A critical review. Neurosci. Biobehav. Rev. 2012, 36, 1140–1152. [Google Scholar] [CrossRef]
- Rosa, M.J.; Portugal, L.; Hahn, T.; Fallgatter, A.J.; Garrido, M.I.; Shawe-Taylor, J.; Mourao-Miranda, J. Sparse network-based models for patient classification using fMRI. Neuroimage 2015, 105, 493–506. [Google Scholar] [CrossRef]
- Saari, P.; Burunat, I.; Brattico, E.; Toiviainen, P. Decoding musical training from dynamic processing of musical features in the brain. Sci. Rep. 2018, 8, 1–12. [Google Scholar] [CrossRef]
- Ibrahimović, N.; Bulheller, S. Mathematik-Test: Grundkenntnisse für Ausbildung und Beruf; Harcourt Test Services: Frankfurt, Germany, 2005. [Google Scholar]
- Jäger, A.O.; Süß, H.-M.; Beauducel, A. Berliner Intelligenzstruktur-Test: [BIS-Test]; Hogrefe, Verlag für Psychologie: Göttingen, Germany, 1997. [Google Scholar]
- Tewes, U. Hamburg-Wechsler-Intelligenztest für Erwachsene, Revision 1991: HAWIE-R; [Testmaterial Ohne Handanweisung]; Huber: Berching, Germany, 1994. [Google Scholar]
- Power, J.D.; Barnes, K.A.; Snyder, A.Z.; Schlaggar, B.L.; Petersen, S.E. Spurious but systematic correlations in functional connectivity MRI networks arise from subject motion. Neuroimage 2012, 59, 2142–2154. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Brady, M.; Smith, S. Segmentation of brain MR images through a hidden Markov random field model and the expectation-maximization algorithm. IEEE Trans. Med. Imag. 2001, 20, 45–57. [Google Scholar] [CrossRef]
- Muschelli, J.; Nebel, M.B.; Caffo, B.S.; Barber, A.D.; Pekar, J.J.; Mostofsky, S.H. Reduction of motion-related artifacts in resting state fMRI using aCompCor. Neuroimage 2014, 96, 22–35. [Google Scholar] [CrossRef]
- Mazziotta, J.; Toga, A.; Evans, A.; Fox, P.; Lancaster, J.; Zilles, K.; Woods, R.; Paus, T.; Simpson, G.; Pike, B.; et al. A probabilistic atlas and reference system for the human brain: International Consortium for Brain Mapping (ICBM). Philos. Trans. R. Soc. Lond. B Biol. Sci. 2001, 356, 1293–1322. [Google Scholar] [CrossRef] [PubMed]
- Friston, K.J.; Holmes, A.P.; Worsley, K.J.; Poline, J.P.; Frith, C.D.; Frackowiak, R.S. Statistical parametric maps in functional imaging: A general linear approach. Hum. Brain. Mapp. 1994, 2, 189–210. [Google Scholar] [CrossRef]
- Tzourio-Mazoyer, N.; Landeau, B.; Papathanassiou, D.; Crivello, F.; Etard, O.; Delcroix, N.; Mazoyer, B.; Joliot, M. Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. NeuroImage 2002, 15, 273–289. [Google Scholar] [CrossRef]
- Koh, M.J.; Seol, J.; Kang, J.I.; Kim, B.S.; Namkoong, K.; Chang, J.W.; Kim, S.J. Altered resting-state functional connectivity in patients with obsessive-compulsive disorder: A magnetoencephalography study. Int. J. Psychophysiol. 2018, 123, 80–87. [Google Scholar] [CrossRef]
- Zar, J.H. Biostatistical Analysis; Pearson Education India: Chennai, India, 1999. [Google Scholar]
- Zhou, Y.; Liang, M.; Tian, L.; Wang, K.; Hao, Y.; Liu, H.; Liu, Z.; Jiang, T. Functional disintegration in paranoid schizophrenia using resting-state fMRI. Schizophr. Res. 2007, 97, 194–205. [Google Scholar] [CrossRef]
- Wu, X.; Li, R.; Fleisher, A.S.; Reiman, E.M.; Guan, X.; Zhang, Y.; Chen, K.; Yao, L. Altered default mode network connectivity in Alzheimer’s disease—A resting functional MRI and Bayesian network study. Hum. Brain. Mapp. 2011, 32, 1868–1881. [Google Scholar] [CrossRef]
- Gentle, J.E.; Härdle, W.K.; Mori, Y. Handbook of Computational Statistics: Concepts and Methods; Springer Science & Business Media: Berlin, Germany, 2012. [Google Scholar]
- Alimardani, F.; Cho, J.; Boostani, R.; Hwang, H. Classification of bipolar disorder and schizophrenia using steady-state visual evoked potential based features. IEEE Access 2018, 6, 40379–40388. [Google Scholar] [CrossRef]
- Shim, M.; Hwang, H.-J.; Kim, D.-W.; Lee, S.-H.; Im, C.-H. Machine-learning-based diagnosis of schizophrenia using combined sensor-level and source-level EEG features. Schizophr. Res. 2016, 176, 314–319. [Google Scholar] [CrossRef]
- Raichle, M.E.; Macleod, A.M.; Snyder, A.Z.; Powers, W.J.; Gusnard, D.A.; Shulman, G.L. A default mode of brain function. Proc. Natl. Acad. Sci. USA 2001, 98, 676–682. [Google Scholar] [CrossRef]
- Raichle, M.E.; Mintun, M.A. Brain work and brain imaging. Annu. Rev. Neurosci. 2006, 29, 449–476. [Google Scholar] [CrossRef]
- Stevens, M.C. The contributions of resting state and task-based functional connectivity studies to our understanding of adolescent brain network maturation. Neurosci. Biobehav. Rev. 2016, 70, 13–32. [Google Scholar] [CrossRef]
- Jiang, L.; Qiao, K.; Li, C. Distance-based functional criticality in the human brain: Intelligence and emotional intelligence. BMC Bioinform. 2021, 22, 32. [Google Scholar] [CrossRef]
- Beaty, R.E.; Kenett, Y.N.; Christensen, A.P.; Rosenberg, M.D.; Benedek, M.; Chen, Q.; Fink, A.; Qiu, J.; Kwapil, T.R.; Kane, M.J.; et al. Robust prediction of individual creative ability from brain functional connectivity. Proc. Natl. Acad. Sci. USA 2018, 115, 1087–1092. [Google Scholar] [CrossRef] [PubMed]
- Lewis, C.M.; Baldassarre, A.; Committeri, G.; Romani, G.L.; Corbetta, M. Learning sculpts the spontaneous activity of the resting human brain. Proc. Natl. Acad. Sci. USA 2009, 106, 17558–17563. [Google Scholar] [CrossRef] [PubMed]
- Duan, X.; He, S.; Liao, W.; Liang, D.; Qiu, L.; Wei, L.; Li, Y.; Liu, C.; Gong, Q.; Chen, H. Reduced caudate volume and enhanced striatal-DMN integration in chess experts. NeuroImage 2012, 60, 1280–1286. [Google Scholar] [CrossRef]
- Adamson, M.M.; Taylor, J.L.; Heraldez, D.; Khorasani, A.; Noda, A.; Hernandez, B.; Yesavage, J.A. Higher landing accuracy in expert pilots is associated with lower activity in the caudate nucleus. PLoS ONE 2014, 9, e112607. [Google Scholar] [CrossRef] [PubMed]
- Balleine, B.W.; Liljeholm, M.; Ostlund, S.B. The integrative function of the basal ganglia in instrumental conditioning. Behav. Brain Res. 2009, 199, 43–52. [Google Scholar] [CrossRef]
- Graybiel, A.M.; Grafton, S.T. The striatum: Where skills and habits meet. Cold. Spring. Harb. Perspect. Biol. 2015, 7, a021691. [Google Scholar] [CrossRef]
- Chase, W.G.; Simon, H.A. Perception in chess. Cogn. Psychol. 1973, 4, 55–81. [Google Scholar] [CrossRef]
- Ericsson, K.A.; Kintsch, W. Long-term working memory. Psychol. Rev. 1995, 102, 211–245. [Google Scholar] [CrossRef]
- Gobet, F.; Simon, H.A. Templates in chess memory: A mechanism for recalling several boards. Cogn. Psychol. 1996, 31, 1–40. [Google Scholar] [CrossRef]
- Maruyama, M.; Pallier, C.; Jobert, A.; Sigman, M.; Dehaene, S. The cortical representation of simple mathematical expressions. Neuroimage 2012, 61, 1444–1460. [Google Scholar] [CrossRef]
- Wang, Y.; Zuo, C.; Wang, D.; Tao, S.; Hao, L. Reduced thalamus volume and enhanced thalamus and fronto-parietal network integration in the chess experts. Cereb. Cortex. 2020, 30, 5560–5569. [Google Scholar] [CrossRef] [PubMed]
- Hwang, K.; Bertolero, M.A.; Liu, W.B.; D’Esposito, M. The human thalamus is an integrative hub for functional brain networks. J. Neurosci. 2017, 37, 5594–5607. [Google Scholar] [CrossRef]
- Browning, P.G.; Chakraborty, S.; Mitchell, A.S. Evidence for mediodorsal thalamus and prefrontal cortex interactions during cognition in macaques. Cereb. Cortex 2015, 25, 4519–4534. [Google Scholar] [CrossRef]
- Fama, R.; Sullivan, E.V. Thalamic structures and associated cognitive functions: Relations with age and aging. Neurosci. Biobehav. Rev. 2015, 54, 29–37. [Google Scholar] [CrossRef]
- Sommer, M.A.; Wurtz, R.H. Influence of the thalamus on spatial visual processing in frontal cortex. Nature 2006, 444, 374–377. [Google Scholar] [CrossRef] [PubMed]
- Pergola, G.; Danet, L.; Pitel, A.L.; Carlesimo, G.A.; Segobin, S.; Pariente, J.; Suchan, B.; Mitchell, A.S.; Barbeau, E.J. The regulatory role of the human mediodorsal thalamus. Trends Cogn. Sci. 2018, 22, 1011–1025. [Google Scholar] [CrossRef]
- Huth, A.G.; de Heer, W.A.; Griffiths, T.L.; Theunissen, F.E.; Gallant, J.L. Natural speech reveals the semantic maps that tile human cerebral cortex. Nature 2016, 532, 453–458. [Google Scholar] [CrossRef] [PubMed]
- Amalric, M.; Dehaene, S. Origins of the brain networks for advanced mathematics in expert mathematicians. Proc. Natl. Acad. Sci. USA 2016, 113, 4909–4917. [Google Scholar] [CrossRef]
- Schultz, D.H.; Cole, M.W. Higher intelligence is associated with less task-related brain network reconfiguration. J. Neurosci. 2016, 36, 8551–8561. [Google Scholar] [CrossRef]
- Cole, M.W.; Bassett, D.S.; Power, J.D.; Braver, T.S.; Petersen, S.E. Intrinsic and task-evoked network architectures of the human brain. Neuron 2014, 83, 238–251. [Google Scholar] [CrossRef]
- Neubauer, A.C.; Fink, A. Intelligence and neural efficiency: Measures of brain activation versus measures of functional connectivity in the brain. Intelligence 2009, 37, 223–229. [Google Scholar] [CrossRef]
- Grahn, J.A.; Parkinson, J.A.; Owen, A.M. The cognitive functions of the caudate nucleus. Prog. Neurobiol. 2008, 86, 141–155. [Google Scholar] [CrossRef]
- Schlaug, G.; Jäncke, L.; Huang, Y.; Staiger, J.F.; Steinmetz, H. Increased corpus callosum size in musicians. Neuropsychologia 1995, 33, 1047–1055. [Google Scholar] [CrossRef]
- Du, Y.; Fu, Z.; Calhoun, V.D. Classification and prediction of brain disorders using functional connectivity: Promising but challenging. Front. Neurosci. 2018, 12, 525. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Yu, F.; Duong, T. Multiparametric MRI characterization and prediction in autism spectrum disorder using graph theory and machine learning. PLoS ONE 2014, 9, e90405. [Google Scholar] [CrossRef]
- Wee, C.Y.; Yap, P.T.; Zhang, D.; Denny, K.; Browndyke, J.N.; Potter, G.G.; Welsh-Bohmer, K.A.; Wang, L.; Shen, D. Identification of MCI individuals using structural and functional connectivity networks. Neuroimage 2012, 59, 2045–2056. [Google Scholar] [CrossRef]
- McDonough, I.M.; Nashiro, K. Network complexity as a measure of information processing across resting-state networks: Evidence from the Human Connectome Project. Front. Hum. Neurosci. 2014, 8, 409. [Google Scholar] [CrossRef]
- Shim, M.; Kim, D.-W.; Lee, S.-H.; Im, C.-H. Disruptions in small-world cortical functional connectivity network during an auditory oddball paradigm task in patients with schizophrenia. Schizophr. Res. 2014, 156, 197–203. [Google Scholar] [CrossRef]
- Mohanty, R.; Sethares, W.A.; Nair, V.A.; Prabhakaran, V. Rethinking measures of functional connectivity via feature extraction. Sci. Rep. 2020, 10, 1–17. [Google Scholar] [CrossRef] [PubMed]

| Mathematicians | Non-Mathematicians | Statistics | |
|---|---|---|---|
| Age | 33.42 (5.62) | 27.23 (8.21) | p = 0.081 |
| Gender, M/F | 16/5 | 14/9 | p = 0.276 |
| Handedness, LQ | 92.45 (3.65) | 90.28 (8.25) | p = 0.269 |
| Years of education | 19.5 (2.7) | 16.21 (6.28) | p = 0.079 |
| Mathematics test | 70.95 (7.13) | 40.71 (7.69) | p < 0.001 |
| Intelligence test | 115.91 (12.35) | 124.27 (15.23) | p = 0.072 |
| WM (forward) | 8.9 (3.12) | 9.12 (4.2) | p = 0.319 |
| WM (backward) | 7.3 (1.9) | 7.62 (1.59) | p = 0.273 |
| 1st ROI | 2nd ROI | p-Value |
|---|---|---|
| Mathematicians > Non-mathematicians | ||
| Left lateral orbital gyrus (LOrG) | Left triangular part of the inferior frontal gyrus (TrIFG) | 0.0003 |
| Right ventral diencephalon (VDc) | Right frontal pole (FP) | 0.0011 |
| Left ventral diencephalon (VDc) | Left superior frontal gyrus (SFG) | 0.0019 |
| Left caudate nucleus (CN) | Right opercular part of the inferior frontal gyrus (OpIFG) | 0.0021 |
| Left parahippocampal gyrus (PhG) | Right supplementary motor cortex (SMC) | 0.0031 |
| Left caudate nucleus (CN) | Right inferior occipital gyrus (IOG) | 0.0039 |
| Right pallidum (Pd) | Left inferior temporal gyrus (ITG) | 0.0039 |
| Left middle cingulate gyrus (MCG) | Right temporal pole (TP) | 0.0039 |
| Left putamen (Pu) | Left frontal pole (FP) | 0.0041 |
| Right orbital part of the inferior frontal gyrus (OrIFG) | Left planum temporale (PT) | 0.0041 |
| Left putamen | Left inferior temporal gyrus | 0.0051 |
| Left ventral diencephalon | Right frontal pole | 0.0051 |
| Right postcentral gyrus | Left precentral gyrus | 0.0059 |
| Left fusiform gyrus | Left planum temporale | 0.0061 |
| Left anterior orbital gyrus | Left occipital pole | 0.0061 |
| Right putamen | Left frontal pole | 0.0063 |
| Left lateral orbital gyrus | Right triangular part of the inferior frontal gyrus | 0.0065 |
| Left frontal pole | Left posterior orbital gyrus | 0.0065 |
| Right occipital pole | Right planum temporale | 0.0069 |
| Right putamen | Right medial orbital gyrus | 0.0073 |
| Left amygdala | Right medial frontal cortex | 0.0079 |
| Right middle temporal gyrus | Left temporal pole | 0.0081 |
| Non-Mathematicians > Mathematicians | ||
| Left lateral orbital gyrus (LOrG) | Right precuneus (Pcun) | 0.000 |
| Right thalamus (Th) | Right lateral orbital gyrus (LOrG) | 0.0007 |
| Left thalamus (Th) | Right lateral orbital gyrus (LOrG) | 0.0011 |
| Right middle occipital gyrus (MOG) | Right supplementary motor cortex (SMC) | 0.0017 |
| Right middle occipital gyrus (MOG) | Left middle occipital gyrus (MOG) | 0.0021 |
| Left gyrus rectus (GRe) | Right precuneus (Pcun) | 0.0027 |
| Right postcentral gyrus (PcG) | Right superior occipital gyrus (SOG) | 0.0031 |
| Right medial frontal cortex (MFC) | Right precuneus (Pcun) | 0.0033 |
| Right caudate nucleus (CN) | Right planum temporale (PT) | 0.0037 |
| Left medial frontal cortex (MFC) | Right precuneus (Pcun) | 0.0041 |
| Right ventral diencephalon | Right fusiform gyrus | 0.0050 |
| Right caudate nucleus | Left middle cingulate gyrus | 0.0053 |
| Left lateral orbital gyrus | Left precuneus | 0.0061 |
| Right caudate nucleus | Left anterior insula | 0.0067 |
| Right lingual gyrus | Left lingual gyrus | 0.0067 |
| Left precentral gyrus | Left middle temporal gyrus | 0.0069 |
| Left inferior temporal gyrus | Left precentral gyrus | 0.0069 |
| Left ventral diencephalon | Right fusiform gyrus | 0.0075 |
| Right gyrus rectus | Right precuneus | 0.0081 |
| Right hippocampus | Right inferior occipital gyrus | 0.0089 |
| Right caudate nucleus | Right supramarginal gyrus | 0.0091 |
| Right caudate nucleus | Left caudate nucleus | 0.0093 |
| Right calcarine cortex | Right lingual gyrus | 0.0099 |
| Right frontal pole | Right posterior orbital gyrus | 0.0099 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shim, M.; Hwang, H.-J.; Kuhl, U.; Jeon, H.-A. Resting-State Functional Connectivity in Mathematical Expertise. Brain Sci. 2021, 11, 430. https://doi.org/10.3390/brainsci11040430
Shim M, Hwang H-J, Kuhl U, Jeon H-A. Resting-State Functional Connectivity in Mathematical Expertise. Brain Sciences. 2021; 11(4):430. https://doi.org/10.3390/brainsci11040430
Chicago/Turabian StyleShim, Miseon, Han-Jeong Hwang, Ulrike Kuhl, and Hyeon-Ae Jeon. 2021. "Resting-State Functional Connectivity in Mathematical Expertise" Brain Sciences 11, no. 4: 430. https://doi.org/10.3390/brainsci11040430
APA StyleShim, M., Hwang, H.-J., Kuhl, U., & Jeon, H.-A. (2021). Resting-State Functional Connectivity in Mathematical Expertise. Brain Sciences, 11(4), 430. https://doi.org/10.3390/brainsci11040430







