Self-Reported Nonadherence to Medication Is Not Associated with Health-Related Quality of Life in Parkinson’s Disease
Abstract
1. Introduction
2. Methods
2.1. Subjects
2.2. Assessment
2.3. Statistical Analyses
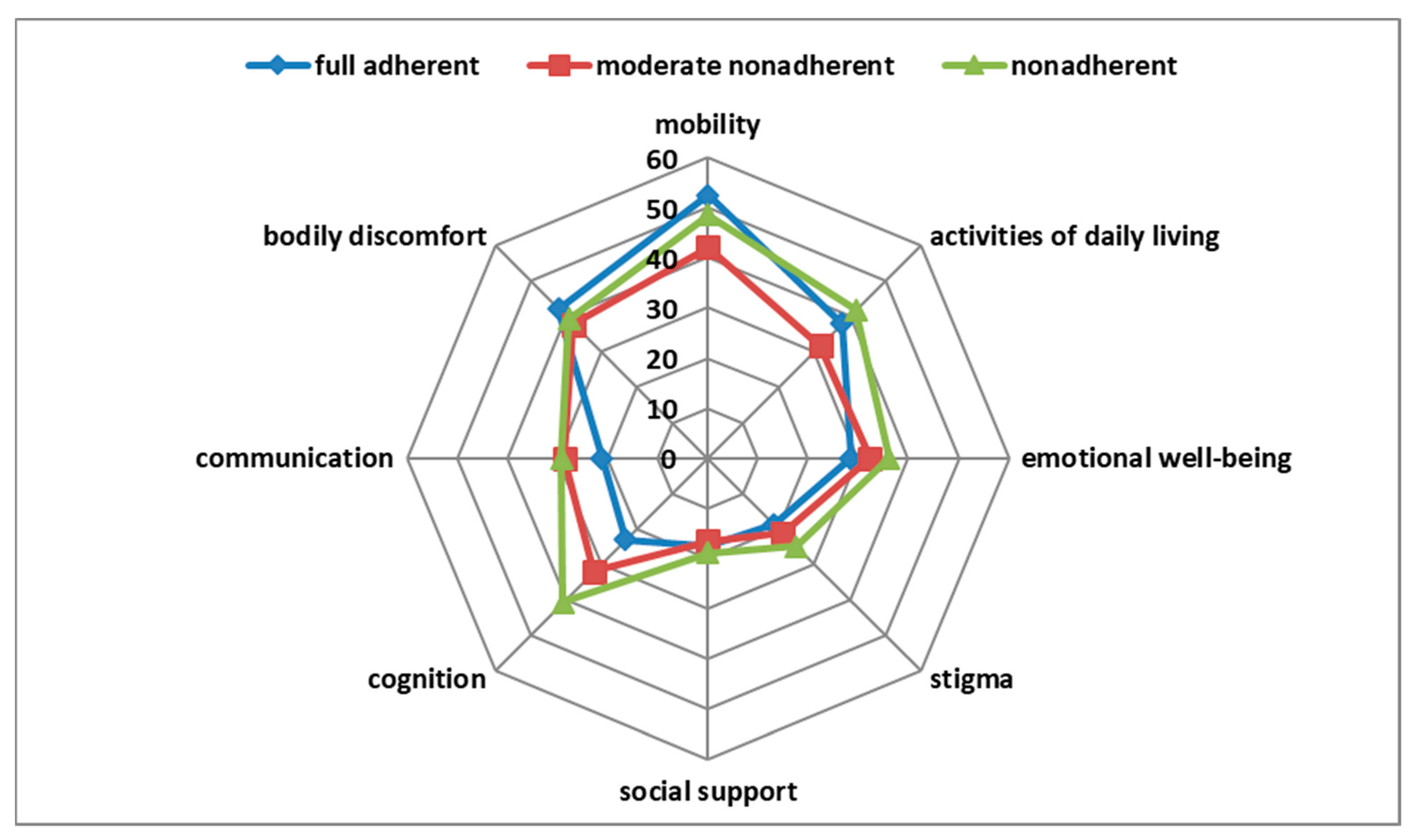
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Conn, V.S.; Ruppar, T.M.; Enriquez, M.; Cooper, P.S. Patient-Centered Outcomes of Medication Adherence Interventions: Systematic Review and Meta-Analysis. Value Health 2016, 19, 277–285. [Google Scholar] [CrossRef]
- Souza, A.C.; Borges, J.W.; Moreira, T.M. Quality of life and treatment adherence in hypertensive patients: Systematic review with meta-analysis. Rev. Saude Publica 2016, 50, 71. [Google Scholar] [CrossRef] [PubMed]
- Park, N.H.; Song, M.S.; Shin, S.Y.; Jeong, J.H.; Lee, H.Y. The effects of medication adherence and health literacy on health-related quality of life in older people with hypertension. Int. J. Older People Nurs. 2018, 13, e12196. [Google Scholar] [CrossRef] [PubMed]
- Grosset, K.A.; Bone, I.; Grosset, D.G. Suboptimal medication adherence in Parkinson’s disease. Mov. Disord. 2005, 20, 1502–1507. [Google Scholar] [CrossRef] [PubMed]
- Grosset, D.; Antonini, A.; Canesi, M.; Pezzoli, G.; Lees, A.; Shaw, K.; Cubo, E.; Martinez-Martin, P.; Rascol, O.; Negre-Pages, L.; et al. Adherence to antiparkinson medication in a multicenter European study. Mov. Disord. 2009, 24, 826–832. [Google Scholar] [CrossRef] [PubMed]
- Straka, I.; Minár, M.; Škorvánek, M.; Grofik, M.; Danterová, K.; Benetin, J.; Kurča, E.; Gažová, A.; Boleková, V.; Wyman-Chick, K.A.; et al. Adherence to Pharmacotherapy in Patients with Parkinson’s Disease Taking Three and More Daily Doses of Medication. Front. Neurol. 2019, 10. [Google Scholar] [CrossRef]
- Hanagasi, H.A.; Tufekcioglu, Z.; Emre, M. Dementia in Parkinson’s disease. J. Neurol. Sci. 2017, 374, 26–31. [Google Scholar] [CrossRef]
- Postuma, R.B.; Poewe, W.; Litvan, I.; Lewis, S.; Lang, A.E.; Halliday, G.; Goetz, C.G.; Chan, P.; Slow, E.; Seppi, K.; et al. Validation of the MDS clinical diagnostic criteria for Parkinson’s disease. Mov. Disord. 2018, 33, 1601–1608. [Google Scholar] [CrossRef] [PubMed]
- Postuma, R.B.; Berg, D.; Stern, M.; Poewe, W.; Olanow, C.W.; Oertel, W.; Obeso, J.; Marek, K.; Litvan, I.; Lang, A.E.; et al. MDS clinical diagnostic criteria for Parkinson’s disease. Mov. Disord. 2015, 30, 1591–1601. [Google Scholar] [CrossRef] [PubMed]
- Nasreddine, Z.S.; Phillips, N.A.; Bedirian, V.; Charbonneau, S.; Whitehead, V.; Collin, I.; Cummings, J.L.; Chertkow, H. The Montreal Cognitive Assessment, MoCA: A brief screening tool for mild cognitive impairment. J. Am. Geriatr. Soc. 2005, 53, 695–699. [Google Scholar] [CrossRef]
- Prell, T.; Grosskreutz, J.; Mendorf, S.; Franke, G.H.; Witte, O.W.; Kunze, A. Clusters of non-adherence to medication in neurological patients. Res. Social Adm. Pharm. 2019, 15, 1419–1424. [Google Scholar] [CrossRef] [PubMed]
- Franke, G.H.; Nentzl, J.; Jagla-Franke, M. SAMS. Stendal Adherence to Medication Score. 2020. Available online: https://www.psychometrikon.de/inhalt/suchen/test.php?id=ff32ee9ea015021c3fb047e505e2bc45 (accessed on 22 February 2021).
- DiMatteo, M.R. Variations in patients’ adherence to medical recommendations: A quantitative review of 50 years of research. Med. Care 2004, 42, 200–209. [Google Scholar] [CrossRef] [PubMed]
- Karve, S.; Cleves, M.A.; Helm, M.; Hudson, T.J.; West, D.S.; Martin, B.C. Good and poor adherence: Optimal cut-point for adherence measures using administrative claims data. Curr. Med. Res. Opin. 2009, 25, 2303–2310. [Google Scholar] [CrossRef] [PubMed]
- Hamedi-Shahraki, S.; Eshraghian, M.R.; Yekaninejad, M.S.; Nikoobakht, M.; Rasekhi, A.; Chen, H.; Pakpour, A. Health-related quality of life and medication adherence in elderly patients with epilepsy. Neurol. Neurochir. Pol. 2019, 53, 123–130. [Google Scholar] [CrossRef]
- Silavanich, V.; Nathisuwan, S.; Phrommintikul, A.; Permsuwan, U. Relationship of medication adherence and quality of life among heart failure patients. Heart Lung 2019, 48, 105–110. [Google Scholar] [CrossRef]
- Khayyat, S.M.; Mohamed, M.M.A.; Khayyat, S.M.S.; Hyat Alhazmi, R.S.; Korani, M.F.; Allugmani, E.B.; Saleh, S.F.; Mansouri, D.A.; Lamfon, Q.A.; Beshiri, O.M.; et al. Association between medication adherence and quality of life of patients with diabetes and hypertension attending primary care clinics: A cross-sectional survey. Qual. Life Res. 2019, 28, 1053–1061. [Google Scholar] [CrossRef] [PubMed]
- Tesfaye, W.; McKercher, C.; Peterson, G.; Castelino, R.; Jose, M.; Zaidi, S.; Wimmer, B. Medication Adherence, Burden and Health-Related Quality of Life in Adults with Predialysis Chronic Kidney Disease: A Prospective Cohort Study. Int. J. Environ. Res. Public Health 2020, 17, 371. [Google Scholar] [CrossRef]
- Kuhlman, G.D.; Flanigan, J.L.; Sperling, S.A.; Barrett, M.J. Predictors of health-related quality of life in Parkinson’s disease. Parkinsonism Relat. Disord. 2019, 65, 86–90. [Google Scholar] [CrossRef]
- Kuopio, A.M.; Marttila, R.J.; Helenius, H.; Toivonen, M.; Rinne, U.K. The quality of life in Parkinson’s disease. Mov. Disord. 2000, 15, 216–223. [Google Scholar] [CrossRef]
- van Uem, J.M.; Marinus, J.; Canning, C.; van Lummel, R.; Dodel, R.; Liepelt-Scarfone, I.; Berg, D.; Morris, M.E.; Maetzler, W. Health-Related Quality of Life in patients with Parkinson’s disease—A systematic review based on the ICF model. Neurosci. Biobehav. Rev. 2016, 61, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Santos García, D.; de Deus Fonticoba, T.; Suárez Castro, E.; Borrué, C.; Mata, M.; Solano Vila, B.; Cots Foraster, A.; Álvarez Sauco, M.; Rodríguez Pérez, A.B.; Vela, L.; et al. Non-motor symptoms burden, mood, and gait problems are the most significant factors contributing to a poor quality of life in non-demented Parkinson’s disease patients: Results from the COPPADIS Study Cohort. Parkinsonism Relat. Disord. 2019, 66, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Mendorf, S.; Witte, O.W.; Grosskreutz, J.; Zipprich, H.M.; Prell, T. What Predicts Different Kinds of Nonadherent Behavior in Elderly People with Parkinson’s Disease? Front. Med. 2020, 7. [Google Scholar] [CrossRef] [PubMed]
- Grosset, K.A.; Bone, I.; Reid, J.L.; Grosset, D. Measuring therapy adherence in Parkinson’s disease: A comparison of methods. J. Neurol. Neurosurg. Psychiatry 2006, 77, 249–251. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | n | % | |
|---|---|---|---|
| Sex | Female | 64 | 39.0 |
| Male | 100 | 61.0 | |
| Marital status | Married | 121 | 75.6 |
| Widowed or divorced | 35 | 21.9 | |
| Single | 4 | 2.5 | |
| Educational level | Low | 36 | 22.2 |
| Middle | 57 | 35.2 | |
| High | 69 | 42.6 | |
| Mean | SD | ||
| Number of medications | 7.27 | 4.29 | |
| Age (years) | 71.04 | 8.97 | |
| Disease duration (years) | 9.41 | 5.90 | |
| Hoehn and Yahr stage (median, interquartile range) | 3.0 | 0.0 | |
| BDI | 11.39 | 6.64 | |
| MoCA | 22.46 | 4.21 | |
| SAMS | 6.76 | 5.80 | |
| NMS total score | 10.39 | 5.03 | |
| MDS-UPDRS III | 25.87 | 14.09 | |
| PDQ-39 mobility | 44.64 | 27.67 | |
| PDQ-39 activities of daily living | 34.71 | 25.40 | |
| PDQ-39 emotional well-being | 32.84 | 21.67 | |
| PDQ-39 stigma | 21.77 | 20.42 | |
| PDQ-39 social support | 17.28 | 19.28 | |
| PDQ-39 cognition | 33.14 | 19.79 | |
| PDQ-39 communication | 27.88 | 20.44 | |
| PDQ-39 bodily discomfort | 38.60 | 23.62 | |
| PDQ-39 summary index | 31.36 | 16.12 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zipprich, H.M.; Mendorf, S.; Lehmann, T.; Prell, T. Self-Reported Nonadherence to Medication Is Not Associated with Health-Related Quality of Life in Parkinson’s Disease. Brain Sci. 2021, 11, 273. https://doi.org/10.3390/brainsci11020273
Zipprich HM, Mendorf S, Lehmann T, Prell T. Self-Reported Nonadherence to Medication Is Not Associated with Health-Related Quality of Life in Parkinson’s Disease. Brain Sciences. 2021; 11(2):273. https://doi.org/10.3390/brainsci11020273
Chicago/Turabian StyleZipprich, Hannah M., Sarah Mendorf, Thomas Lehmann, and Tino Prell. 2021. "Self-Reported Nonadherence to Medication Is Not Associated with Health-Related Quality of Life in Parkinson’s Disease" Brain Sciences 11, no. 2: 273. https://doi.org/10.3390/brainsci11020273
APA StyleZipprich, H. M., Mendorf, S., Lehmann, T., & Prell, T. (2021). Self-Reported Nonadherence to Medication Is Not Associated with Health-Related Quality of Life in Parkinson’s Disease. Brain Sciences, 11(2), 273. https://doi.org/10.3390/brainsci11020273







