Beyond Reading Modulation: Temporo-Parietal tDCS Alters Visuo-Spatial Attention and Motion Perception in Dyslexia
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
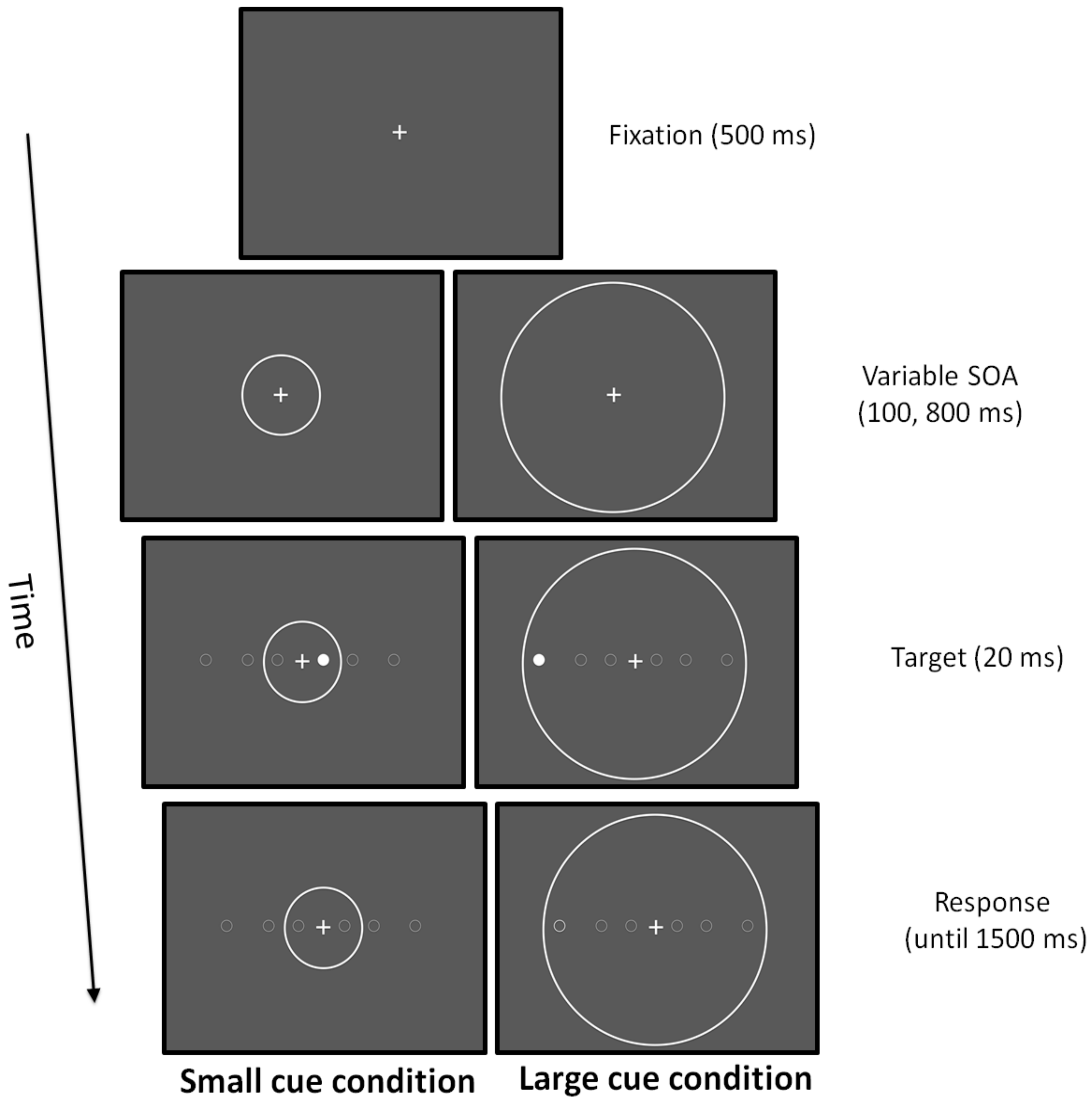
2.2. Experimental Design
2.3. Stimulation Conditions
2.4. Reading and Neuropsychological Tasks
2.4.1. Words, Pseudowords, and Text Reading
2.4.2. Lexical Decision
2.4.3. Phoneme Blending
2.4.4. Working Memory
2.4.5. Rapid Automatized Naming
2.4.6. Coherent Dot Motion
2.4.7. Attentional Zooming
2.5. Statistical Analyses
3. Results
3.1. Words, Pseudowords, and Text Reading
3.2. Lexical Decision
3.3. Phoneme Blending, Working Memory, and Rapid Automatized Naming
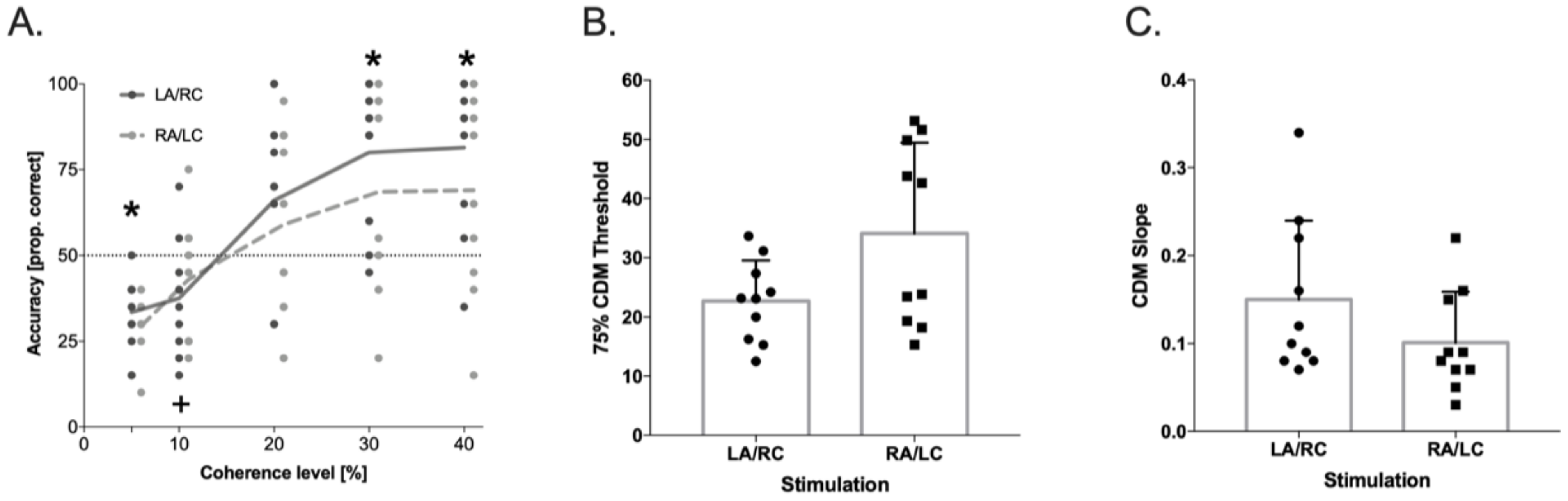
3.4. Coherent Dot Motion
3.5. Attentional Zooming
3.6. Correlations between Reading and Neuropsychological Changes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Gabrieli, J.D. Dyslexia: A new synergy between education and cognitive neuroscience. Science 2009, 325, 280–283. [Google Scholar] [CrossRef]
- Peterson, R.L.; Pennington, B.F. Developmental dyslexia. Annu. Rev. Clin. Psychol. 2015, 11, 283–307. [Google Scholar] [CrossRef] [PubMed]
- Richlan, F. The Functional Neuroanatomy of Letter-Speech Sound Integration and Its Relation to Brain Abnormalities in Developmental Dyslexia. Front. Hum. Neurosci. 2019, 13, 21. [Google Scholar] [CrossRef]
- Richlan, F.; Kronbichler, M.; Wimmer, H. Meta-analyzing brain dysfunctions in dyslexic children and adults. NeuroImage 2011, 56, 1735–1742. [Google Scholar] [CrossRef] [PubMed]
- Richlan, F. Developmental dyslexia: Dysfunction of a left hemisphere reading network. Front. Hum. Neurosci. 2012, 6, 120. [Google Scholar] [CrossRef]
- Hoeft, F.; Hernandez, A.; McMillon, G.; Taylor-Hill, H.; Martindale, J.L.; Meyler, A.; Keller, T.A.; Siok, W.T.; Deutsch, G.K.; Just, M.A.; et al. Neural basis of dyslexia: A comparison between dyslexic and nondyslexic children equated for reading ability. J. Neurosci. 2006, 26, 10700–10708. [Google Scholar] [CrossRef] [PubMed]
- Corbetta, M.; Patel, G.; Shulman, G.L. The reorienting system of the human brain: From environment to theory of mind. Neuron 2008, 58, 306–324. [Google Scholar] [CrossRef]
- Bertoni, S.; Franceschini, S.; Puccio, G.; Mancarella, M.; Gori, S.; Facoetti, A. Action Video Games Enhance Attentional Control and Phonological Decoding in Children with Developmental Dyslexia. Brain Sci. 2021, 11, 171. [Google Scholar] [CrossRef]
- Menghini, D.; Finzi, A.; Benassi, M.; Bolzani, R.; Facoetti, A.; Giovagnoli, S.; Ruffino, M.; Vicari, S. Different underlying neurocognitive deficits in developmental dyslexia: A comparative study. Neuropsychologia 2010, 48, 863–872. [Google Scholar] [CrossRef] [PubMed]
- Franceschini, S.; Gori, S.; Ruffino, M.; Pedrolli, K.; Facoetti, A. A causal link between visual spatial attention and reading acquisition. Curr. Biol. 2012, 22, 814–819. [Google Scholar] [CrossRef] [PubMed]
- Gori, S.; Molteni, M.; Facoetti, A. Visual Illusions: An Interesting Tool to Investigate Developmental Dyslexia and Autism Spectrum Disorder. Front. Hum. Neurosci. 2016, 10, 175. [Google Scholar] [CrossRef]
- Carroll, J.M.; Solity, J.; Shapiro, L.R. Predicting dyslexia using prereading skills: The role of sensorimotor and cognitive abilities. J. Child. Psychol. Psychiatry 2016, 57, 750–758. [Google Scholar] [CrossRef]
- Franceschini, S.; Trevisan, P.; Ronconi, L.; Bertoni, S.; Colmar, S.; Double, K.; Facoetti, A.; Gori, S. Action video games improve reading abilities and visual-to-auditory attentional shifting in English-speaking children with dyslexia. Sci. Rep. 2017, 7, 5863. [Google Scholar] [CrossRef] [PubMed]
- Bertoni, S.; Franceschini, S.; Ronconi, L.; Gori, S.; Facoetti, A. Is excessive visual crowding causally linked to developmental dyslexia? Neuropsychologia 2019, 130, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Snowling, M.J.; Lervåg, A.; Nash, H.M.; Hulme, C. Longitudinal relationships between speech perception, phonological skills and reading in children at high-risk of dyslexia. Dev. Sci. 2019, 22, e12723. [Google Scholar] [CrossRef]
- Bradley, L.; Bryant, P.E. Categorizing sounds and learning to read: A causal connection. Nature 1983, 301, 419–421. [Google Scholar] [CrossRef]
- Franceschini, S.; Gori, S.; Ruffino, M.; Viola, S.; Molteni, M.; Facoetti, A. Action video games make dyslexic children read better. Curr. Biol. 2013, 23, 462–466. [Google Scholar] [CrossRef]
- Dehaene, S.; Cohen, L.; Morais, J.; Kolinsky, R. Illiterate to literate: Behavioural and cerebral changes induced by reading acquisition. Nat. Rev. Neurosci. 2015, 16, 234–244. [Google Scholar] [CrossRef]
- Johnson, M.H. Interactive specialization: A domain-general framework for human functional brain development? Dev. Cogn. Neurosci. 2011, 1, 7–21. [Google Scholar] [CrossRef]
- Snowling, M. Dyslexia, 2nd ed.; Blackwell Publishing: Oxford, UK, 2000. [Google Scholar]
- Vellutino, F.R.; Fletcher, J.M.; Snowling, M.J.; Scanlon, D.M. Specific reading disability (dyslexia): What have we learned in the past four decades? J. Child. Psychol. Psychiatry 2004, 45, 2–40. [Google Scholar] [CrossRef]
- Melby-Lervåg, M.; Lyster, S.A.; Hulme, C. Phonological skills and their role in learning to read: A meta-analytic review. Psychol. Bull. 2012, 138, 322–352. [Google Scholar] [CrossRef]
- Horowitz-Kraus, T.; Hutton, J.S. From emergent literacy to reading: How learning to read changes a child’s brain. Acta Paediatr. 2015, 104, 648–656. [Google Scholar] [CrossRef]
- Kosmidis, M.H.; Zafiri, M.; Politimou, N. Literacy versus formal schooling: Influence on working memory. Arch. Clin. Neuropsychol. 2011, 26, 575–582. [Google Scholar] [CrossRef]
- Kosmidis, M.H.; Tsapkini, K.; Folia, V. Lexical processing in illiteracy: Effect of literacy or education? Cortex 2006, 42, 1021–1027. [Google Scholar] [CrossRef]
- Petersson, K.M.; Reis, A.; Ingvar, M. Cognitive processing in literate and illiterate subjects: A review of some recent behavioral and functional neuroimaging data. Scand. J. Psychol. 2001, 42, 251–267. [Google Scholar] [CrossRef] [PubMed]
- Lazzaro, G.; Varuzza, C.; Costanzo, F.; Fucà, E.; Di Vara, S.; De Matteis, M.E.; Vicari, S.; Menghini, D. Memory Deficits in Children with Developmental Dyslexia: A Reading-Level and Chronological-Age Matched Design. Brain Sci. 2021, 11, 40. [Google Scholar] [CrossRef]
- Tallal, P. Auditory temporal perception, phonics, and reading disabilities in children. Brain Lang. 1980, 9, 182–198. [Google Scholar] [CrossRef]
- Tallal, P. Improving language and literacy is a matter of time. Nat. Rev. Neurosci. 2004, 5, 721–728. [Google Scholar] [CrossRef] [PubMed]
- Hornickel, J.; Kraus, N. Unstable representation of sound: A biological marker of dyslexia. J. Neurosci. 2013, 33, 3500–3504. [Google Scholar] [CrossRef] [PubMed]
- Hari, R.; Renvall, H. Impaired processing of rapid stimulus sequences in dyslexia. Trends Cogn. Sci. 2001, 5, 525–532. [Google Scholar] [CrossRef]
- Bosse, M.L.; Tainturier, M.J.; Valdois, S. Developmental dyslexia: The visual attention span deficit hypothesis. Cognition 2007, 104, 198–230. [Google Scholar] [CrossRef] [PubMed]
- Roach, N.W.; Hogben, J.H. Impaired filtering of behaviourally irrelevant visual information in dyslexia. Brain 2007, 130, 771–785. [Google Scholar] [CrossRef]
- Facoetti, A.; Trussardi, A.N.; Ruffino, M.; Lorusso, M.L.; Cattaneo, C.; Galli, R.; Molteni, M.; Zorzi, M. Multisensory spatial attention deficits are predictive of phonological decoding skills in developmental dyslexia. J. Cogn. Neurosci. 2010, 22, 1011–1025. [Google Scholar] [CrossRef] [PubMed]
- Lallier, M.; Tainturier, M.J.; Dering, B.; Donnadieu, S.; Valdois, S.; Thierry, G. Behavioral and ERP evidence for amodal sluggish attentional shifting in developmental dyslexia. Neuropsychologia 2010, 48, 4125–4135. [Google Scholar] [CrossRef] [PubMed]
- Zorzi, M.; Barbiero, C.; Facoetti, A.; Lonciari, I.; Carrozzi, M.; Montico, M.; Bravar, L.; George, F.; Pech-Georgel, C.; Ziegler, J.C. Extra-large letter spacing improves reading in dyslexia. Proc. Natl. Acad. Sci. USA 2012, 109, 11455–11459. [Google Scholar] [CrossRef]
- Galaburda, A.; Livingstone, M. Evidence for a magnocellular defect in developmental dyslexia. Ann. N. Y. Acad. Sci. 1993, 682, 70–82. [Google Scholar] [CrossRef] [PubMed]
- Kevan, A.; Pammer, K. Visual deficits in pre-readers at familial risk for dyslexia. Vis. Res. 2008, 48, 2835–2839. [Google Scholar] [CrossRef][Green Version]
- Kevan, A.; Pammer, K. Predicting early reading skills from pre-reading measures of dorsal stream functioning. Neuropsychologia 2009, 47, 3174–3181. [Google Scholar] [CrossRef]
- Boets, B.; Vandermosten, M.; Cornelissen, P.; Wouters, J.; Ghesquière, P. Coherent motion sensitivity and reading development in the transition from prereading to reading stage. Child. Dev. 2011, 82, 854–869. [Google Scholar] [CrossRef] [PubMed]
- Gori, S.; Cecchini, P.; Bigoni, A.; Molteni, M.; Facoetti, A. Magnocellular-dorsal pathway and sub-lexical route in developmental dyslexia. Front. Hum. Neurosci. 2014, 8, 460. [Google Scholar] [CrossRef]
- Lawton, T. Improving Dorsal Stream Function in Dyslexics by Training Figure/Ground Motion Discrimination Improves Attention, Reading Fluency, and Working Memory. Front. Hum. Neurosci. 2016, 10, 397. [Google Scholar] [CrossRef]
- Walsh, V. Dyslexia. Reading between the laminae. Curr. Biol. 1995, 5, 1216–1217. [Google Scholar] [CrossRef]
- Stein, J.; Walsh, V. To see but not to read; the magnocellular theory of dyslexia. Trends Neurosci. 1997, 20, 147–152. [Google Scholar] [CrossRef]
- Boden, C.; Giaschi, D. M-stream deficits and reading-related visual processes in developmental dyslexia. Psychol. Bull. 2007, 133, 346–366. [Google Scholar] [CrossRef] [PubMed]
- Laycock, R.; Crewther, S.G. Towards an understanding of the role of the ‘magnocellular advantage’ in fluent reading. Neurosci. Biobehav. Rev. 2008, 32, 1494–1506. [Google Scholar] [CrossRef] [PubMed]
- Vidyasagar, T.R.; Pammer, K. Dyslexia: A deficit in visuo-spatial attention, not in phonological processing. Trends Cogn. Sci. 2010, 14, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Gori, S.; Facoetti, A. How the visual aspects can be crucial in reading acquisition? The intriguing case of crowding and developmental dyslexia. J. Vis. 2015, 15, 15.1.8. [Google Scholar] [CrossRef] [PubMed]
- Menghini, D.; Finzi, A.; Carlesimo, G.A.; Vicari, S. Working memory impairment in children with developmental dyslexia: Is it just a phonological deficit? Dev. Neuropsychol. 2011, 36, 199–213. [Google Scholar] [CrossRef]
- Bestmann, S.; Walsh, V. Transcranial electrical stimulation. Curr. Biol. 2017, 27, R1258–R1262. [Google Scholar] [CrossRef]
- Woods, A.J.; Antal, A.; Bikson, M.; Boggio, P.S.; Brunoni, A.R.; Celnik, P.; Cohen, L.G.; Fregni, F.; Herrmann, C.S.; Kappenman, E.S.; et al. A technical guide to tDCS, and related non-invasive brain stimulation tools. Clin. Neurophysiol. 2016, 127, 1031–1048. [Google Scholar] [CrossRef]
- Kirimoto, H.; Ogata, K.; Onishi, H.; Oyama, M.; Goto, Y.; Tobimatsu, S. Transcranial direct current stimulation over the motor association cortex induces plastic changes in ipsilateral primary motor and somatosensory cortices. Clin. Neurophysiol. 2011, 122, 777–783. [Google Scholar] [CrossRef]
- Turkeltaub, P.E.; Benson, J.; Hamilton, R.H.; Datta, A.; Bikson, M.; Coslett, H.B. Left lateralizing transcranial direct current stimulation improves reading efficiency. Brain Stimul. 2012, 5, 201–207. [Google Scholar] [CrossRef]
- Younger, J.W.; Randazzo Wagner, M.; Booth, J.R. Weighing the cost and benefit of transcranial direct current stimulation on different reading subskills. Front. Neurosci. 2016, 10. [Google Scholar] [CrossRef]
- Younger, J.W.; Booth, J.R. Parietotemporal stimulation affects acquisition of novel grapheme-phoneme mappings in adult readers. Front. Hum. Neurosci. 2018, 12. [Google Scholar] [CrossRef]
- Heth, I.; Lavidor, M. Improved reading measures in adults with dyslexia following transcranial direct current stimulation treatment. Neuropsychologia 2015, 70, 107–113. [Google Scholar] [CrossRef]
- Costanzo, F.; Varuzza, C.; Rossi, S.; Sdoia, S.; Varvara, P.; Oliveri, M.; Koch, G.; Vicari, S.; Menghini, D. Reading changes in children and adolescents with dyslexia after transcranial direct current stimulation. Neuroreport 2016, 27, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Costanzo, F.; Varuzza, C.; Rossi, S.; Sdoia, S.; Varvara, P.; Oliveri, M.; Giacomo, K.; Vicari, S.; Menghini, D. Evidence for reading improvement following tDCS treatment in children and adolescents with dyslexia. Restor. Neurol. Neurosci. 2016, 34, 215–226. [Google Scholar] [CrossRef]
- Costanzo, F.; Rossi, S.; Varuzza, C.; Varvara, P.; Vicari, S.; Menghini, D. Long-lasting improvement following tDCS treatment combined with a training for reading in children and adolescents with dyslexia. Neuropsychologia 2019, 130, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Rios, D.M.; Rios, C.M.; Bandeira, I.D.; Queiros Campbell, F.; de Carvalho Vaz, D.; Lucena, R. Impact of transcranial direct current stimulation on reading skills of children and adolescents with dyslexia. Child. Neurol. Open. 2018, 5, 2329048X1879825. [Google Scholar] [CrossRef] [PubMed]
- Lazzaro, G.; Costanzo, F.; Varuzza, C.; Rossi, S.; De Matteis, M.E.; Vicari, S.; Menghini, D. Individual Differences Modulate the Effects of tDCS on Reading in Children and Adolescents with Dyslexia. Sci. Stud. Read. 2020. [Google Scholar] [CrossRef]
- Raven, J.C. Coloured Progressive Matrices; Giunti OS Organizzazioni Speciali: Firenze, Italy, 2008. [Google Scholar]
- Raven, J.C. Standard Progressive Matrices; Giunti OS Organizzazioni Speciali: Firenze, Italy, 2008. [Google Scholar]
- Oldfield, R.C. The assessment and analysis of handedness: The Edinburgh inventory. Neuropsychologia 1971, 9, 97–113. [Google Scholar] [CrossRef]
- Sartori, G.; Job, R.; Tressoldi, P.E. DDE-2. Batteria per la Valutazione della Dislessia e della Disortografia Evolutiva; Giunti OS Organizzazioni Speciali: Firenze, Italy, 2007. [Google Scholar]
- Stella, G.; Tintoni, C. Indagine e rilevazione sulle abilità di lettura nelle scuole secondarie di secondo grado. Dislessia 2007, 4, 271–285. [Google Scholar]
- Cornoldi, C.; Pra Baldi, A.; Friso, G. MT avanzate 2—Prove MT Avanzate di Lettura e Matematica 2° per il Biennio della Scuola Secondaria di Secondo Grado; Giunti OS Organizzazioni Speciali: Firenze, Italy, 2010. [Google Scholar]
- Cornoldi, C.; Colpo, G. Nuove Prove MT per la Scuola Secondaria di I Grado; Giunti OS Organizzazioni Speciali: Firenze, Italy, 2012. [Google Scholar]
- Conners, C.K. Conners’ Rating Scales–Revised; Nobile, M., Alberti, B., Zuddas, A., Eds.; Giunti OS Organizzazioni Speciali: Firenze, Italy, 2007. [Google Scholar]
- Costanzo, F.; Menghini, D.; Caltagirone, C.; Oliveri, M.; Vicari, S. High frequency rTMS over the left parietal lobule increases non-word reading accuracy. Neuropsychologia 2012, 50, 2645–2651. [Google Scholar] [CrossRef] [PubMed]
- Costanzo, F.; Menghini, D.; Caltagirone, C.; Oliveri, M.; Vicari, S. How to improve reading skills in dyslexics: The effect of high frequency rTMS. Neuropsychologia 2013, 51, 2953–2959. [Google Scholar] [CrossRef] [PubMed]
- Denckla, M.B.; Rudel, R.G. Rapid “automatized” naming (R.A.N): Dyslexia differentiated from other learning disabilities. Neuropsychologia 1976, 14, 471–479. [Google Scholar] [CrossRef]
- Britten, K.H.; Shadlen, M.N.; Newsome, W.T.; Movshon, J.A. The analysis of visual motion: A comparison of neuronal and psychophysical performance. J. Neurosci. 1992, 12, 4745–4765. [Google Scholar] [CrossRef] [PubMed]
- Pilly, P.K.; Seitz, A.R. What a difference a parameter makes: A psychophysical comparison of random dot motion algorithms. Vis. Res. 2009, 49, 1599–1612. [Google Scholar] [CrossRef]
- Ronconi, L.; Gori, S.; Ruffino, M.; Franceschini, S.; Urbani, B.; Molteni, M.; Facoetti, A. Decreased coherent motion discrimination in autism spectrum disorder: The role of attentional zoom-out deficit. PLoS ONE 2012, 7, e49019. [Google Scholar] [CrossRef] [PubMed]
- Facoetti, A.; Molteni, M. The gradient of visual attention in developmental dyslexia. Neuropsychologia 2001, 39, 352–357. [Google Scholar] [CrossRef]
- Ronconi, L.; Gori, S.; Ruffino, M.; Molteni, M.; Facoetti, A. Zoom-out attentional impairment in children with autism spectrum disorder. Cortex 2013, 49, 1025–1033. [Google Scholar] [CrossRef] [PubMed]
- Gori, S.; Yazdanbakhsh, A. The riddle of the Rotating-Tilted-Lines illusion. Perception 2008, 37, 631–635. [Google Scholar] [CrossRef] [PubMed]
- Cancer, A.; Antonietti, A. tDCS Modulatory Effect on Reading Processes: A Review of Studies on Typical Readers and Individuals with Dyslexia. Front. Behav. Neurosci. 2018, 12, 162. [Google Scholar] [CrossRef]
- Livingstone, M.S.; Rosen, G.D.; Drislane, F.W.; Galaburda, A.M. Physiological and anatomical evidence for a magnocellular defect in developmental dyslexia. Proc. Natl. Acad. Sci. USA 1991, 88, 7943–7947. [Google Scholar] [CrossRef] [PubMed]
- Eden, G.F.; VanMeter, J.W.; Rumsey, J.M.; Maisog, J.M.; Woods, R.P.; Zeffiro, T.A. Abnormal processing of visual motion in dyslexia revealed by functional brain imaging. Nature 1996, 382, 66–69. [Google Scholar] [CrossRef] [PubMed]
- Demb, J.B.; Boynton, G.M.; Best, M.; Heeger, D.J. Psychophysical evidence for a magnocellular pathway deficit in dyslexia. Vis. Res. 1998, 38, 1555–1559. [Google Scholar] [CrossRef]
- Ben-Shachar, M.; Dougherty, R.F.; Deutsch, G.K.; Wandell, B.A. Contrast responsivity in MT+ correlates with phonological awareness and reading measures in children. NeuroImage 2007, 37, 1396–1406. [Google Scholar] [CrossRef][Green Version]
- Gori, S.; Seitz, A.R.; Ronconi, L.; Franceschini, S.; Facoetti, A. Multiple Causal Links between Magnocellular-Dorsal Pathway Deficit and Developmental Dyslexia. Cereb. Cortex 2016, 26, 4356–4369. [Google Scholar] [CrossRef]
- Ronconi, L.; Facoetti, A.; Bulf, H.; Franchin, L.; Bettoni, R.; Valenza, E. Paternal autistic traits are predictive of infants visual attention. J. Autism Dev. Disord. 2014, 44, 1556–1564. [Google Scholar] [CrossRef] [PubMed]
- Franceschini, S.; Bertoni, S.; Gianesini, T.; Gori, S.; Facoetti, A. A different vision of dyslexia: Local precedence on global perception. Sci. Rep. 2017, 7, 17462. [Google Scholar] [CrossRef]
- de Schotten, M.T.; Dell’Acqua, F.; Forkel, S.J.; Simmons, A.; Vergani, F.; Murphy, D.G.; Catani, M.A. Lateralized brain network for visuospatial attention. Nat. Neurosci. 2011, 14, 1245–1246. [Google Scholar] [CrossRef]
- Fink, G.R.; Halligan, P.W.; Marshall, J.C.; Frith, C.D.; Frackowiak, R.S.; Dolan, R.J. Where in the brain does visual attention select the forest and the trees? Nature 1996, 382, 626–628. [Google Scholar] [CrossRef]
- Sergent, J. The cerebral balance of power: Confrontation or cooperation? J. Exp. Psychol. Hum. Percept. Perform. 1982, 8, 253–272. [Google Scholar] [CrossRef] [PubMed]
- Hoeft, F.; McCandliss, B.D.; Black, J.M.; Gantman, A.; Zakerani, N.; Hulme, C.; Lyytinen, H.; Whitfield-Gabrieli, S.; Glover, G.H.; Reiss, A.L.; et al. Neural systems predicting long-term outcome in dyslexia. Proc. Natl. Acad. Sci. USA 2011, 108, 361–366. [Google Scholar] [CrossRef] [PubMed]
- Maisog, J.M.; Einbinder, E.R.; Flowers, D.L.; Turkeltaub, P.E.; Eden, G.F. A meta-analysis of functional neuroimaging studies of dyslexia. Ann. N. Y. Acad. Sci. 2008, 1145, 237–259. [Google Scholar] [CrossRef] [PubMed]
- Vandermosten, M.; Hoeft, F.; Norton, E.S. Integrating MRI brain imaging studies of pre-reading children with current theories of developmental dyslexia: A review and quantitative meta-analysis. Curr. Opin. Behav. Sci. 2016, 10, 155–161. [Google Scholar] [CrossRef]
- Sireteanu, R.; Goertz, R.; Bachert, I.; Wandert, T. Children with developmental dyslexia show a left visual “minineglect”. Vis. Res. 2005, 45, 3075–3082. [Google Scholar] [CrossRef] [PubMed]
- Hancock, R.; Pugh, K.R.; Hoeft, F. Neural Noise Hypothesis of Developmental Dyslexia. Trends Cogn. Sci. 2017, 21, 434–448. [Google Scholar] [CrossRef] [PubMed]
- Pugh, K.R.; Frost, S.J.; Rothman, D.L.; Hoeft, F.; Del Tufo, S.N.; Mason, G.F.; Molfese, P.J.; Mencl, W.E.; Grigorenko, E.L.; Landi, N.; et al. Glutamate and choline levels predict individual differences in reading ability in emergent readers. J. Neurosci. 2014, 34, 4082–4089. [Google Scholar] [CrossRef]
- Vigneau, M.; Beaucousin, V.; Hervé, P.Y.; Jobard, G.; Petit, L.; Crivello, F.; Mellet, E.; Zago, L.; Mazoyer, B.; Tzourio-Mazoyer, N. What is right-hemisphere contribution to phonological, lexico-semantic, and sentence processing? Insights from a meta-analysis. NeuroImage 2011, 54, 577–593. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Marshall, J.C.; Weidner, R.; Fink, G.R. Zooming in and zooming out of the attentional focus: An FMRI study. Cereb. Cortex 2009, 19, 805–819. [Google Scholar] [CrossRef] [PubMed]
- Masuda, F.; Nakajima, S.; Miyazaki, T.; Kazunari Yoshida, K.; Tsugawa, S.; Wada, M.; Ogyu, K.; Croarkin, P.E.; Blumberger, D.M.; Daskalakis, Z.J.; et al. Motor cortex excitability and inhibitory imbalance in autism spectrum disorder assessed with transcranial magnetic stimulation: A systematic review. Transl. Psychiatry 2019, 9, 110. [Google Scholar] [CrossRef] [PubMed]


| LA/RC | RA/LC | ||
|---|---|---|---|
| Reading Measures | M (SD) | M (SD) | |
| TEXT | % Errors 1 | 4.78 (4.16) | 6.89 (5.47) ** |
| Times 2 | 41.90 (23.27) | 40.72 (24.66) | |
| High Frequency words | % Errors 1 | 6.75 (8.74) | 9.00 (15.64) |
| Times 3 | 18.40 (10.99) | 20.00 (16.11) | |
| Low Frequency words | % Errors 1 | 12.25 (12.10) | 15.75 (18.52) |
| Times 3 | 28.40 (16.04) | 32.80 (26.86) | |
| Pseudo- words | % Errors 1 | 20.75 (26.90) | 20.00 (21.21) |
| Times 3 | 36.30 (15.95) | 35.66 (16.91) | |
| Lexical Decision | Reaction Times 3 | 1.34 (0.40) | 1.50 (0.42) * |
| LA/RC | RA/LC | ||
|---|---|---|---|
| Neuropsychological Tasks | M (SD) | M (SD) | |
| Phoneme Blending | Accuracy 1 | 62.80 (16.46) | 65.00 (15.58) |
| Times 2 | 22.33 (8.55) | 23.91 (10.46) | |
| N-back | Verbal 3 | 2.56 (0.60) | 2.73 (0.45) |
| Visuo-Spatial 3 | 2.78 (0.73) | 2.93 (0.70) | |
| RAN | Letters 2 | 3.75 (1.14) | 5.03 (1.34) |
| Colors 2 | 4.82 (1.11) | 3.84 (0.76) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lazzaro, G.; Bertoni, S.; Menghini, D.; Costanzo, F.; Franceschini, S.; Varuzza, C.; Ronconi, L.; Battisti, A.; Gori, S.; Facoetti, A.; et al. Beyond Reading Modulation: Temporo-Parietal tDCS Alters Visuo-Spatial Attention and Motion Perception in Dyslexia. Brain Sci. 2021, 11, 263. https://doi.org/10.3390/brainsci11020263
Lazzaro G, Bertoni S, Menghini D, Costanzo F, Franceschini S, Varuzza C, Ronconi L, Battisti A, Gori S, Facoetti A, et al. Beyond Reading Modulation: Temporo-Parietal tDCS Alters Visuo-Spatial Attention and Motion Perception in Dyslexia. Brain Sciences. 2021; 11(2):263. https://doi.org/10.3390/brainsci11020263
Chicago/Turabian StyleLazzaro, Giulia, Sara Bertoni, Deny Menghini, Floriana Costanzo, Sandro Franceschini, Cristiana Varuzza, Luca Ronconi, Andrea Battisti, Simone Gori, Andrea Facoetti, and et al. 2021. "Beyond Reading Modulation: Temporo-Parietal tDCS Alters Visuo-Spatial Attention and Motion Perception in Dyslexia" Brain Sciences 11, no. 2: 263. https://doi.org/10.3390/brainsci11020263
APA StyleLazzaro, G., Bertoni, S., Menghini, D., Costanzo, F., Franceschini, S., Varuzza, C., Ronconi, L., Battisti, A., Gori, S., Facoetti, A., & Vicari, S. (2021). Beyond Reading Modulation: Temporo-Parietal tDCS Alters Visuo-Spatial Attention and Motion Perception in Dyslexia. Brain Sciences, 11(2), 263. https://doi.org/10.3390/brainsci11020263








