Modulation of Hippocampal Astroglial Activity by Synaptamide in Rats with Neuropathic Pain
Abstract
:1. Introduction
2. Materials and Methods
2.1. N-Docosahexaenoylethanolamine
2.2. Animals
2.3. CCI Procedure and Treatments
2.4. Behavioral Testing
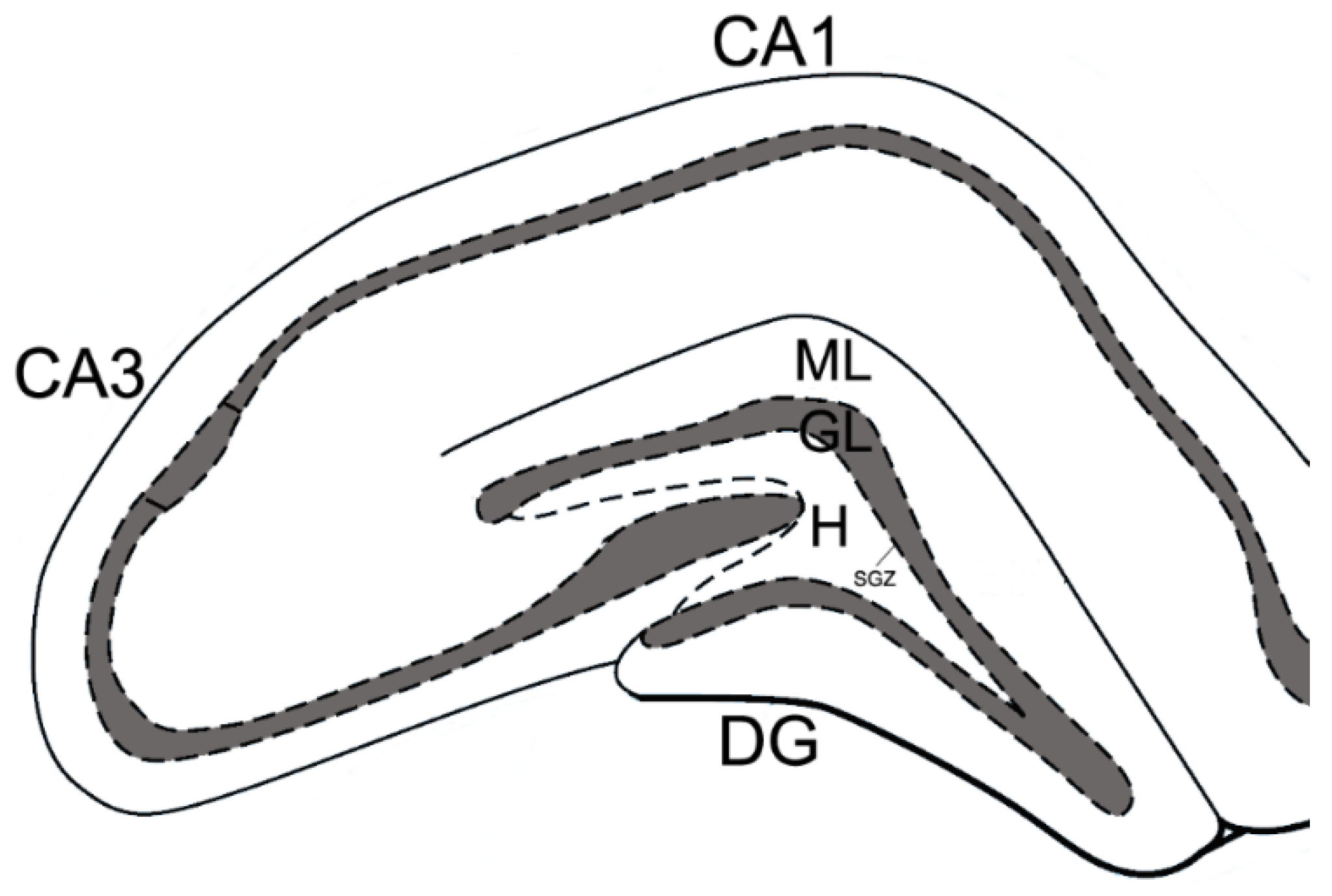
2.5. Immunohistochemistry
2.6. Cell Culture
2.7. ELISA
2.8. Calcium Imaging
2.9. Quantitation of N-Acylethanolamines
2.10. Statistical Data Processing
3. Results
3.1. Behavioral Studies
3.1.1. Working Memory
3.1.2. Locomotor Activity
3.1.3. Anxiety
3.1.4. Long-Term Memory
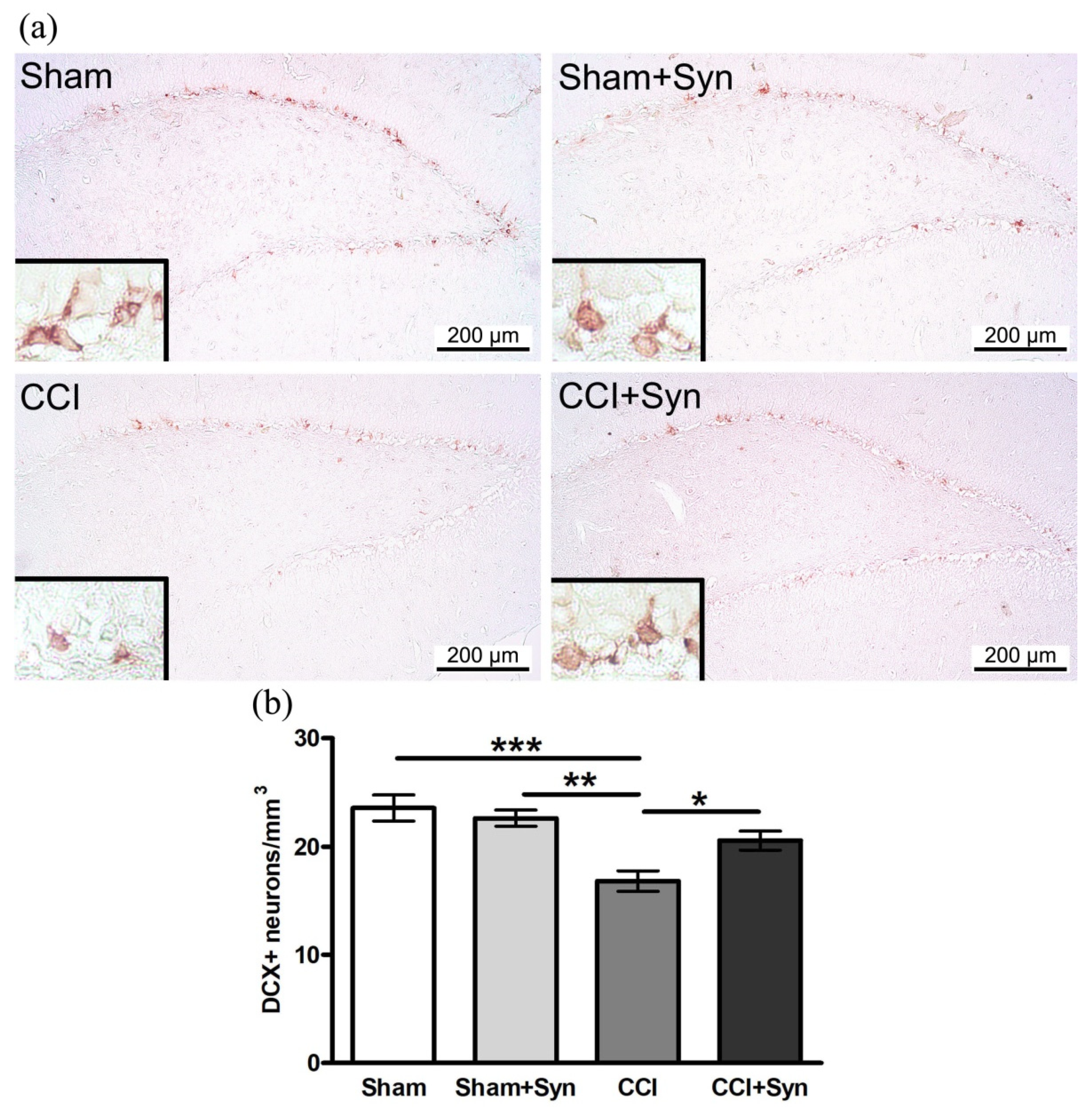
3.2. Synaptamide Prevents the Impairment of Neurogenesis after CCI
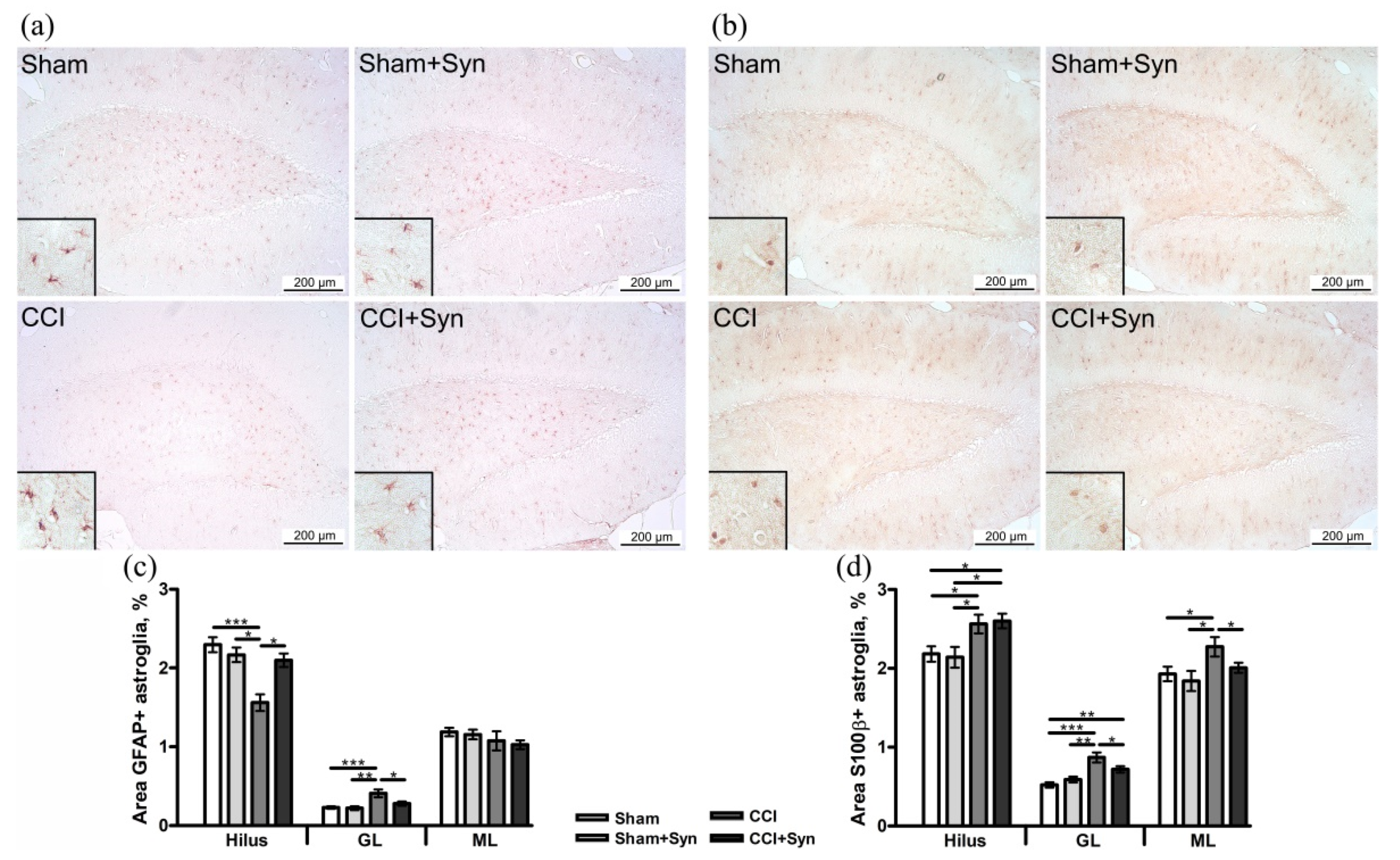
3.3. Synaptamide Stabilizes Astrogliosis after CCI
3.4. CCI Influences the Activity of NMDAR1 in the Hippocampus
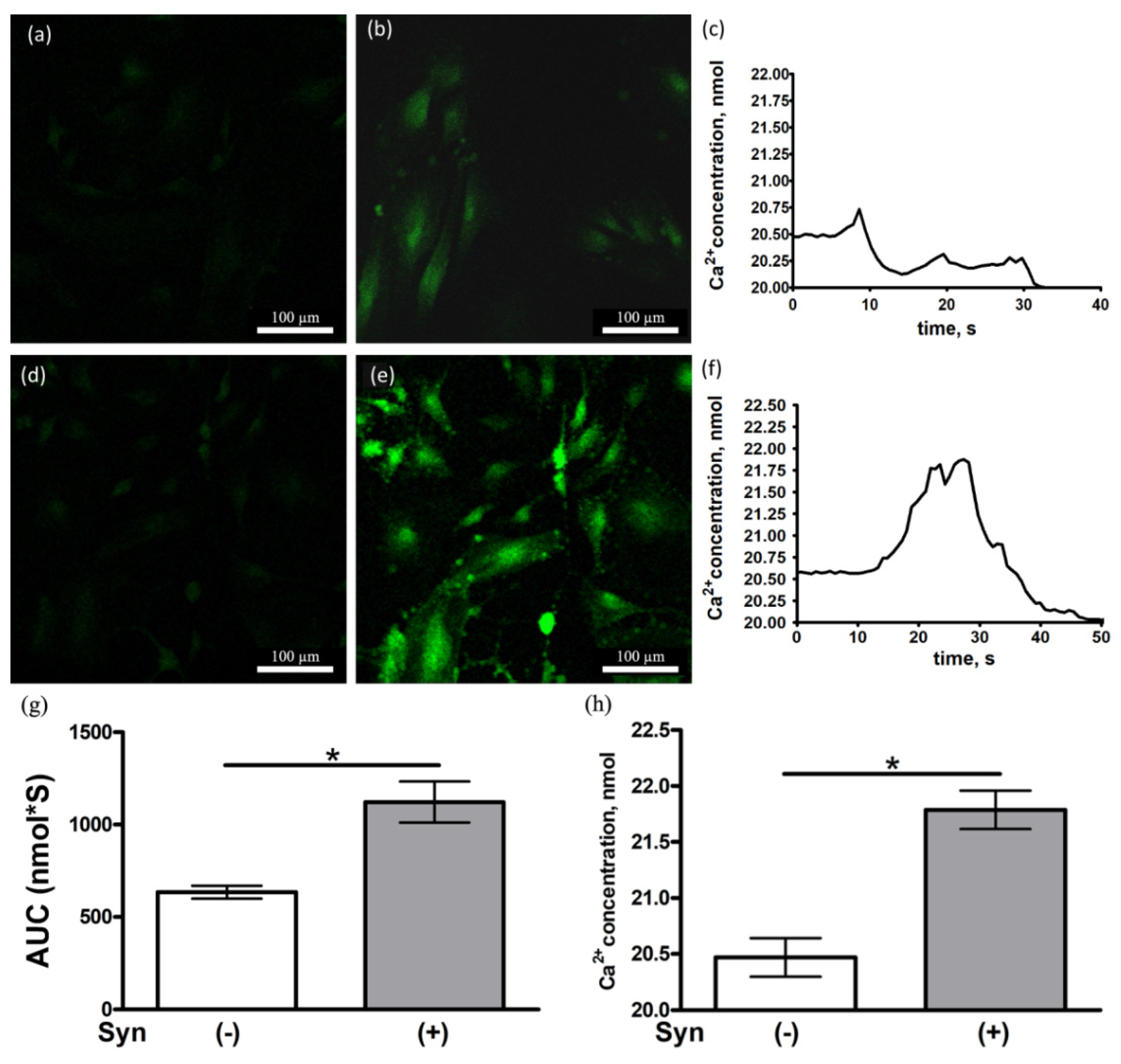
3.5. Synaptamide Increases the Ca2+ Concentration in Cultured Astrocytes
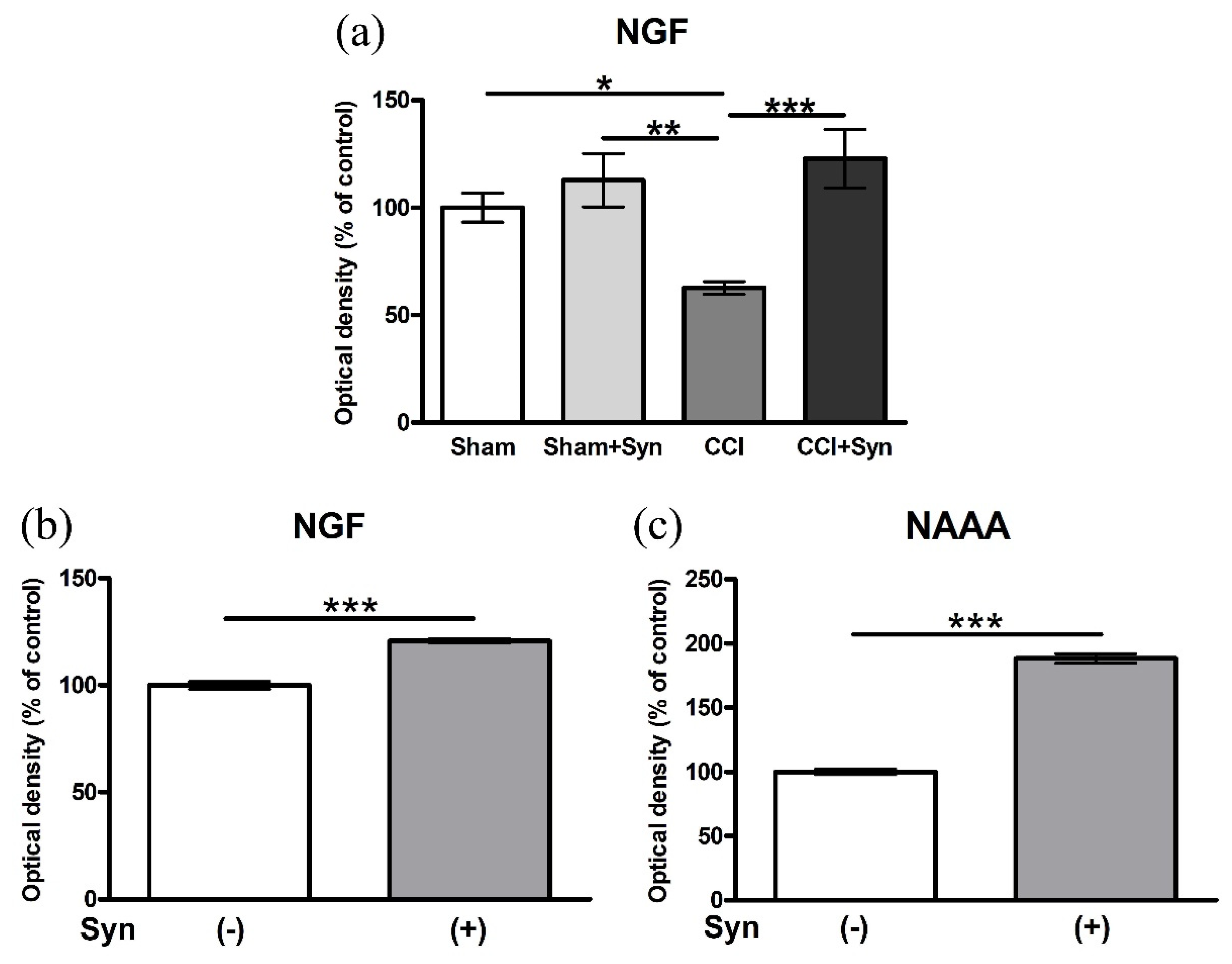
3.6. Synaptamide Increases the Activity of Nerve Growth Factor (NGF) In Vivo and In Vitro
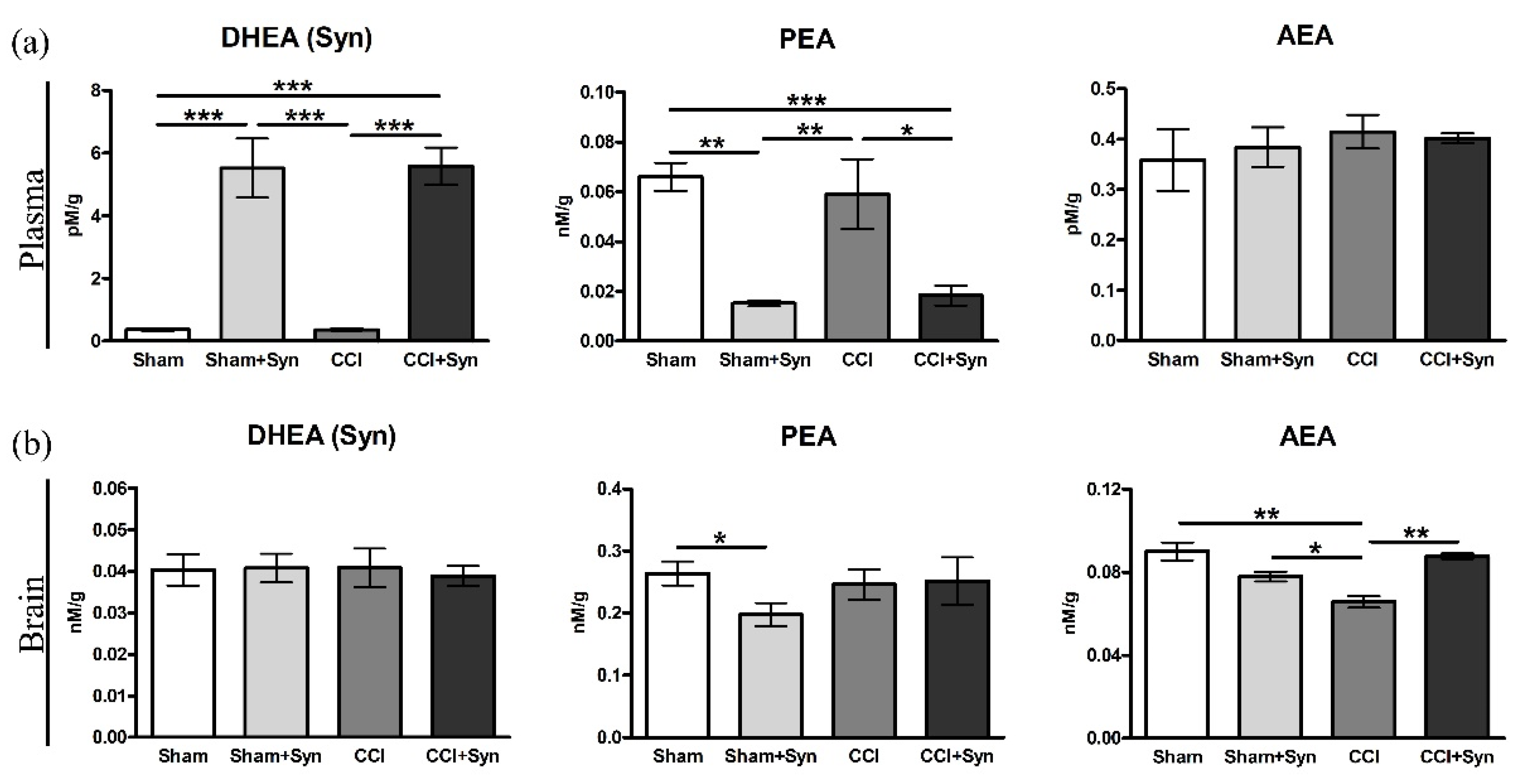
3.7. N-Acylethanolamines Composition in Plasma and Brain after CCI and Synaptamide Treatment
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Colloca, L.; Ludman, T.; Bouhassira, D.; Baron, R.; Dickenson, A.H.; Yarnitsky, D.; Freeman, R.; Truini, A.; Attal, N.; Finnerup, N.B.; et al. Neuropathic pain. Nat. Rev. Dis. Primers 2017, 3, 17002. [Google Scholar] [CrossRef] [Green Version]
- Woolf, C.J.; Mannion, R.J. Neuropathic pain: Aetiology, symptoms, mechanisms, and management. Lancet 1999, 353, 1959–1964. [Google Scholar] [CrossRef]
- Liu, M.G.; Chen, J. Roles of the hippocampal formation in pain information processing. Neurosci. Bull. 2009, 25, 237–266. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mutso, A.A.; Radzicki, D.; Baliki, M.N.; Huang, L.; Banisadr, G.; Centeno, M.V.; Radulovic, J.; Martina, M.; Miller, R.J.; Apkarian, A.V. Abnormalities in hippocampal functioning with persistent pain. J. Neurosci. 2012, 32, 5747–5756. [Google Scholar] [CrossRef] [Green Version]
- Fiore, N.T.; Austin, P.J. Glial-cytokine-neuronal adaptations in the ventral hippocampus of rats with affective behavioral changes following peripheral nerve injury. Neuroscience 2018, 390, 119–140. [Google Scholar] [CrossRef] [PubMed]
- Tyrtyshnaia, A.A.; Egorova, E.L.; Starinets, A.A.; Ponomarenko, A.I.; Ermolenko, E.V.; Manzhulo, I.V. N-Docosahexaenoylethanolamine Attenuates Neuroinflammation and Improves Hippocampal Neurogenesis in Rats with Sciatic Nerve Chronic Constriction Injury. Mar. Drugs 2020, 18, 516. [Google Scholar] [CrossRef] [PubMed]
- Haim, B.L.; Rowitch, D.H. Functional diversity of astrocytes in neural circuit regulation. Nat. Rev. Neurosci. 2017, 18, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Grace, P.M.; Hutchinson, M.R.; Maier, S.F.; Watkins, L.R. Pathological pain and the neuroimmune interface. Nat. Rev. Immunol. 2014, 14, 217–231. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aloe, L.; Rocco, M.; Bianchi, P.; Manni, L. Nerve growth factor: From the early discoveries to the potential clinical use. J. Transl. Med. 2012, 10, 239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bannister, K.; Sachau, J.; Baron, R.; Dickenson, A.H. Neuropathic pain: Mechanism-based therapeutics. Annu. Rev. Pharmacol. Toxicol. 2020, 60, 257–274. [Google Scholar] [CrossRef]
- Ngo, L.T.; Okogun, J.I.; Folk, W.R. 21st century natural product research and drug development and traditional medicines. Nat. Prod. Rep. 2013, 30, 584–592. [Google Scholar] [CrossRef]
- Kim, H.Y.; Spector, A.A. N-Docosahexaenoylethanolamine: A neurotrophic and neuroprotective metabolite of docosahexaenoic acid. Mol. Asp. Med. 2018, 64, 34–44. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.Y.; Spector, A.A. Synaptamide, endocannabinoid-like derivative of docosahexaenoic acid with cannabinoid-independent function. Leukot. Essent. Fat. Acids 2013, 88, 121–125. [Google Scholar] [CrossRef] [Green Version]
- Sonti, S.; Duclos, R.I., Jr.; Tolia, M.; Gatley, S.J. N-Docosahexaenoylethanolamine (synaptamide): Carbon-14 radiolabeling and metabolic studies. Chem. Phys. Lipids 2018, 210, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Calderon, F.; Kim, H.Y. Docosahexaenoic acid promotes neurite growth in hippocampal neurons. J. Neurochem. 2004, 90, 979–988. [Google Scholar] [CrossRef] [PubMed]
- Kharebava, G.; Rashid, M.A.; Lee, J.W.; Sarkar, S.; Kevala, K.; Kim, H.Y. N-docosahexaenoylethanolamine regulates Hedgehog signaling and promotes growth of cortical axons. Biol. Open 2015, 4, 1660–1670. [Google Scholar] [CrossRef] [Green Version]
- Ponomarenko, A.I.; Tyrtyshnaia, A.A.; Pislyagin, E.A.; Dyuizen, I.V.; Sultanov, R.M.; Manzhulo, I.V. N-docosahexaenoylethanolamine reduces neuroinflammation and cognitive impairment after mild traumatic brain injury in rats. Sci. Rep. 2021, 11, 756. [Google Scholar] [CrossRef]
- Park, T.; Chen, H.; Kevala, K.; Lee, J.W.; Kim, H.Y. N-Docosahexaenoylethanolamine ameliorates LPS-induced neuroinflammation via cAMP/PKA-dependent signaling. J. Neuroinflamm. 2016, 13, 284. [Google Scholar] [CrossRef] [Green Version]
- Latyshev, N.A.; Ermolenko, E.V.; Kasyanov, S.P. Concentration and purification of polyunsaturated fatty acids from squid liver processing wastes. Eur. J. Lipid Sci. Technol. 2014, 116, 1608–1613. [Google Scholar] [CrossRef]
- Bennett, G.J.; Xie, Y.K. A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain 1988, 33, 87–107. [Google Scholar] [CrossRef]
- Statler, K.D.; Alexander, H.; Vagni, V.; Holubkov, R.; Dixon, C.E.; Clark, R.S.; Jenkins, L.; Kochanek, P.M. Isoflurane exerts neuroprotective actions at or near the time of severe traumatic brain injury. Brain Res. 2006, 1076, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Hughes, R.N. The value of spontaneous alternation behavior (SAB) as a test of retention in pharmacological investigations of memory. Neurosci. Biobehav. Rev. 2004, 28, 497–505. [Google Scholar] [CrossRef]
- Ishiyama, T.; Tokuda, K.; Ishibashi, T.; Ito, A.; Toma, S.; Ohno, Y. Lurasidone (SM-13496), a novel atypical antipsychotic drug, reverses MK-801-induced impairment of learning and memory in the rat passive-avoidance test. Eur. J. Pharmacol. 2007, 572, 160–170. [Google Scholar] [CrossRef] [PubMed]
- Schildge, S.; Bohrer, C.; Beck, K.; Schachtrup, C. Isolation and culture of mouse cortical astrocytes. J. Vis. Exp. 2013, 71, 50079. [Google Scholar] [CrossRef] [Green Version]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef]
- Couillard-Despres, S.; Winner, B.; Schaubeck, S.; Aigner, R.; Vroemen, M.; Weidner, N.; Bogdahn, U.; Winkler, J.; Kuhn, H.-G.; Aigner, L. Doublecortin expression levels in adult brain reflect neurogenesis. Eur. J. Neurosci. 2005, 21, 1–14. [Google Scholar] [CrossRef]
- Donegan, M.; Kernisant, M.; Cua, C.; Jasmin, L.; Ohara, P.T. Satellite glial cell proliferation in the trigeminal ganglia after chronic constriction injury of the infraorbital nerve. Glia 2013, 61, 2000–2008. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, A. NMDA receptor function during senescence: Implication on cognitive performance. Front. Neurosci. 2015, 9, 473. [Google Scholar] [CrossRef]
- Winkler, J.; Ramirez, G.A.; Kuhn, H.G.; Peterson, D.A.; Day-Lollini, P.A.; Stewart, G.R.; Tuszynski, M.H.; Gage, F.H.; Thal, L.J. Reversible Schwann cell hyperplasia and sprouting of sensory and sympathetic neurites after intraventricular administration of nerve growth factor. Ann. Neurol. 1997, 41, 82–93. [Google Scholar] [CrossRef] [PubMed]
- Shelton, D.L.; Reichardt, L.F. Studies on the expression of the beta nerve growth factor (NGF) gene in the central nervous system: Level and regional distribution of NGF mRNA suggest that NGF functions as a trophic factor for several distinct populations of neurons. Proc. Natl. Acad. Sci. USA 1986, 83, 2714–2718. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paton, K.F.; Shirazi, R.; Vyssotcki, M.; Kivell, B.M. N-docosahexaenoyl ethanolamine (synaptamide) has antinociceptive effects in male mice. Eur. J. Pain 2020, 24, 1990–1998. [Google Scholar] [CrossRef]
- Ueda, N.; Tsuboi, K.; Uyama, T. N-acylethanolamine metabolism with special reference to N-acylethanolamine-hydrolysing acid amidase (NAAA). Prog. Lipid Res. 2010, 49, 299–315. [Google Scholar] [CrossRef]
- Tang, R.; Lin, Y.M.; Liu, H.X.; Wang, E.S. Neuroprotective effect of docosahexaenoic acid in rat traumatic brain injury model via regulation of TLR4/NF-Kappa B signaling pathway. Int. J. Biochem. Cell Biol. 2018, 99, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Meijerink, J.; Balvers, M.; Witkamp, R. N-acyl amines of docosahexaenoic acid and other n–3 polyunsatured fatty acids–from fishy endocannabinoids to potential leads. Br. J. Pharmacol. 2013, 169, 772–783. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bisogno, T.; Berrendero, F.; Ambrosino, G.; Cebeira, M.; Ramos, J.A.; Fernandez-Ruiz, J.J.; Di Marzo, V. Brain regional distribution of endocannabinoids: Implications for their biosynthesis and biological function. Biochem. Biophys. Res. Commun. 1999, 256, 377–380. [Google Scholar] [CrossRef] [PubMed]
- Guidi, M.; Rani, A.; Karic, S.; Severance, B.; Kumar, A.; Foster, T.C. Contribution of N-methyl-D-aspartate receptors to attention and episodic spatial memory during senescence. Neurobiol. Learn. Mem. 2015, 125, 36–46. [Google Scholar] [CrossRef] [Green Version]
- Kumar, A.; Foster, T.C. Linking redox regulation of NMDAR synaptic function to cognitive decline during aging. J. Neurosci. 2013, 33, 15710–15715. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nishikawa, M.; Kimura, S.; Akaike, N. Facilitatory effect of docosahexaenoic acid on N-methyl-D-aspartate response in pyramidal neurones of rat cerebral cortex. J. Physiol. 1994, 475, 83–93. [Google Scholar] [CrossRef]
- Schell, M.J. The N-Methyl D-Aspartate Receptor Glycine Site and D-Serine Metabolism: An evolutionary perspective. Philos. Trans. Biol. Sci. 2004, 359, 943–964. [Google Scholar] [CrossRef] [Green Version]
- Williams, S.M.; Diaz, C.M.; Macnab, L.T.; Sullivan, R.K.; Pow, D.V. Immunocytochemical analysis of d-serine distribution in the mammalian brain reveals novel anatomical compartmentalizations in glia and neurons. Glia 2006, 53, 401–411. [Google Scholar] [CrossRef]
- Skowrońska, K.; Obara-Michlewska, M.; Zielińska, M.; Albrecht, J. NMDA Receptors in Astrocytes: In Search for Roles in Neurotransmission and Astrocytic Homeostasis. Int. J. Mol. Sci. 2019, 20, 309. [Google Scholar] [CrossRef] [Green Version]
- Palygin, O.; Lalo, U.; Verkhratsky, A.; Pankratov, Y. Ionotropic NMDA and P2X1/5 receptors mediate synaptically induced Ca2+ signalling in cortical astrocytes. Cell Calcium 2010, 48, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Haim, B.L.; Carrillo-de Sauvage, M.A.; Ceyzériat, K.; Escartin, C. Elusive roles for reactive astrocytes in neurodegenerative diseases. Front. Cell. Neurosci. 2015, 9, 278. [Google Scholar]
- Raponi, E.; Agenes, F.; Delphin, C.; Assard, N.; Baudier, J.; Legraverend, C.; Deloulme, J.C. S100β expression defines a state in which GFAP-expressing cells lose their neural stem cell potential and acquire a more mature developmental stage. Glia 2007, 55, 165–177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Colangelo, A.M.; Cirillo, G.; Lavitrano, M.L.; Alberghina, L.; Papa, M. Targeting reactive astrogliosis by novel biotechnological strategies. Biotechnol. Adv. 2012, 30, 261–271. [Google Scholar] [CrossRef]
- Cragnolini, A.B.; Huang, Y.; Gokina, P.; Friedman, W.J. Nerve growth factor attenuates proliferation of astrocytes via the p75 neurotrophin receptor. Glia 2009, 57, 1386–1392. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blochl, A.; Blochl, R. A cell-biological model of p75NTR signaling. J. Neurochem. 2007, 102, 289–305. [Google Scholar] [CrossRef]
- Knipper, M.; Beck, A.; Rylett, J.; Breer, H. Neurotrophin induced cAMP and IP3 responses in PC12 cells. Different pathways. FEBS Lett. 1993, 324, 147–152. [Google Scholar] [CrossRef] [Green Version]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manzhulo, I.; Manzhulo, O.; Tyrtyshnaia, A.; Ponomarenko, A.; Konovalova, S.; Ermolenko, E.; Milkina, E.; Starinets, A. Modulation of Hippocampal Astroglial Activity by Synaptamide in Rats with Neuropathic Pain. Brain Sci. 2021, 11, 1561. https://doi.org/10.3390/brainsci11121561
Manzhulo I, Manzhulo O, Tyrtyshnaia A, Ponomarenko A, Konovalova S, Ermolenko E, Milkina E, Starinets A. Modulation of Hippocampal Astroglial Activity by Synaptamide in Rats with Neuropathic Pain. Brain Sciences. 2021; 11(12):1561. https://doi.org/10.3390/brainsci11121561
Chicago/Turabian StyleManzhulo, Igor, Olga Manzhulo, Anna Tyrtyshnaia, Arina Ponomarenko, Sophia Konovalova, Ekaterina Ermolenko, Elena Milkina, and Anna Starinets. 2021. "Modulation of Hippocampal Astroglial Activity by Synaptamide in Rats with Neuropathic Pain" Brain Sciences 11, no. 12: 1561. https://doi.org/10.3390/brainsci11121561
APA StyleManzhulo, I., Manzhulo, O., Tyrtyshnaia, A., Ponomarenko, A., Konovalova, S., Ermolenko, E., Milkina, E., & Starinets, A. (2021). Modulation of Hippocampal Astroglial Activity by Synaptamide in Rats with Neuropathic Pain. Brain Sciences, 11(12), 1561. https://doi.org/10.3390/brainsci11121561






