Psychometric Properties of NASA-TLX and Index of Cognitive Activity as Measures of Cognitive Workload in Older Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Procedure
2.2.1. Demographic and Clinical Information
2.2.2. n-Back Test
2.2.3. National Aeronautics and Space Administration Task Load Index
2.2.4. Index of Cognitive Activity
2.2.5. P3 Event-Related Potential
2.3. Data Analysis
3. Results
3.1. Participant Characteristics
3.2. Test–Retest Reliability of NASA-TLX
3.3. Test–Retest Reliability of ICA
3.4. Convergent Validity of NASA-TLX
3.5. Convergent Validity of ICA
3.6. Correlation between ICA and NASA-TLX
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cowan, N. The magical mystery four: How is working memory capacity limited, and why? Curr. Dir. Psychol. Sci. 2010, 19, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Kahneman, D. Attention and Effort; Prentice Hall: Englewood Cliffs, NJ, USA, 1973; Volume 1063. [Google Scholar]
- Bruya, B.; Tang, Y.-Y. Is attention really effort? Revisiting Daniel Kahneman’s influential 1973 book Attention and effort. Front. Psychol. 2018, 9, 1133. [Google Scholar] [CrossRef] [PubMed]
- Ranchet, M.; Morgan, J.C.; Akinwuntan, A.E.; Devos, H. Cognitive workload across the spectrum of cognitive impairments: A systematic review of physiological measures. Neurosci. Biobehav. Rev. 2017, 80, 516–537. [Google Scholar] [CrossRef] [PubMed]
- Hart, S.G.; Staveland, L.E. Development of NASA-TLX (Task Load Index): Results of empirical and theoretical research. In Advances in Psychology; Hancock, P.A., Meshkati, N., Eds.; Elsevier BV: Amsterdam, The Netherlands, 1988; pp. 139–183. [Google Scholar]
- Dias, R.D.; Ngo-Howard, M.C.; Boskovski, M.T.; Zenati, M.A.; Yule, S.J. Systematic review of measurement tools to assess surgeons’ intraoperative cognitive workload. Br. J. Surg. 2018, 105, 491–501. [Google Scholar] [CrossRef] [PubMed]
- Hart, S.G. Nasa-task load index (NASA-TLX); 20 years later. Proc. Hum. Fact. Ergon. Soc. Annu. Meet. 2006, 50, 904–908. [Google Scholar] [CrossRef]
- Tubbs-Cooley, H.L.; Mara, C.A.; Carle, A.C.; Gurses, A.P. The NASA Task Load Index as a measure of overall workload among neonatal, paediatric and adult intensive care nurses. Intensiv. Crit. Care Nurs. 2018, 46, 64–69. [Google Scholar] [CrossRef]
- Devos, H.; Burns, J.M.; Liao, K.; Ahmadnezhad, P.; Mahnken, J.D.; Brooks, W.M.; Gustafson, K. Reliability of P3 event-related potential during working memory across the spectrum of cognitive aging. Front. Aging Neurosci. 2020, 12, 566391. [Google Scholar] [CrossRef]
- Bopp, K.L.; Verhaeghen, P. Aging and n-back performance: A meta-analysis. J. Gerontol. Ser. B 2018, 75, 229–240. [Google Scholar] [CrossRef]
- Schmiedek, F.; Li, S.-C.; Lindenberger, U. Interference and facilitation in spatial working memory: Age-associated differences in lure effects in the n-back paradigm. Psychol. Aging 2009, 24, 203–210. [Google Scholar] [CrossRef]
- Miller, K.; Price, C.; Okun, M.; Montijo, H.; Bowers, D. Is the N-Back task a valid neuropsychological measure for assessing working memory? Arch. Clin. Neuropsychol. 2009, 24, 711–717. [Google Scholar] [CrossRef]
- Ahmadlou, M.; Adeli, A.; Bajo, R.; Adeli, H. Complexity of functional connectivity networks in mild cognitive impairment subjects during a working memory task. Clin. Neurophysiol. 2014, 125, 694–702. [Google Scholar] [CrossRef] [PubMed]
- Galluzzi, S.; Nicosia, F.; Geroldi, C.; Alicandri, A.; Bonetti, M.; Romanelli, G.; Zulli, R.; Frisoni, G.B. Cardiac autonomic dysfunction is associated with white matter lesions in patients with mild cognitive impairment. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2009, 64, 1312–1315. [Google Scholar] [CrossRef] [PubMed]
- Granholm, E.; Panizzon, M.S.; Elman, J.A.; Jak, A.J.; Hauger, R.L.; Bondi, M.W.; Lyons, M.J.; Franz, C.E.; Kremen, W.S. Pupillary responses as a biomarker of early risk for Alzheimer’s Disease. J. Alzheimer’s Dis. 2017, 56, 1419–1428. [Google Scholar] [CrossRef] [PubMed]
- Braak, H.; Thal, D.R.; Ghebremedhin, E.; Del Tredici, K. Stages of the pathologic process in Alzheimer Disease: Age categories from 1 to 100 years. J. Neuropathol. Exp. Neurol. 2011, 70, 960–969. [Google Scholar] [CrossRef]
- Wilson, R.S.; Nag, S.; Boyle, P.A.; Hizel, L.P.; Yu, L.; Buchman, A.S.; Schneider, J.A.; Bennett, D.A. Neural reserve, neuronal density in the locus ceruleus, and cognitive decline. Neurology 2013, 80, 1202–1208. [Google Scholar] [CrossRef]
- Chandler, D.; Jensen, P.; McCall, J.G.; E Pickering, A.; Schwarz, L.A.; Totah, N.K. Redefining noradrenergic neuromodulation of behavior: Impacts of a modular locus coeruleus architecture. J. Neurosci. 2019, 39, 8239–8249. [Google Scholar] [CrossRef]
- Samuels, E.R.; Szabadi, E. Functional neuroanatomy of the noradrenergic locus coeruleus: Its roles in the regulation of arousal and autonomic function part I: Principles of functional organisation. Curr. Neuropharmacol. 2008, 6, 235–253. [Google Scholar] [CrossRef]
- Beatty, J.; Lucero-Wagoner, B. The pupillary system. In Handbook of Psychophysiology, 2nd ed.; Cambridge University Press: New York, NY, USA, 2000; pp. 142–162. [Google Scholar]
- Kremen, W.S.; Panizzon, M.S.; Elman, J.A.; Granholm, E.L.; Andreassen, O.A.; Dale, A.M.; Gillespie, N.A.; Gustavson, D.E.; Logue, M.W.; Lyons, M.J.; et al. Pupillary dilation responses as a midlife indicator of risk for Alzheimer’s disease: Association with Alzheimer’s disease polygenic risk. Neurobiol. Aging 2019, 83, 114–121. [Google Scholar] [CrossRef]
- Alnaes, D.; Sneve, M.H.; Espeseth, T.; Endestad, T.; Van De Pavert, S.H.P.; Laeng, B. Pupil size signals mental effort deployed during multiple object tracking and predicts brain activity in the dorsal attention network and the locus coeruleus. J. Vis. 2014, 14, 1. [Google Scholar] [CrossRef]
- Mathur, A.; Gehrmann, J.; Atchison, D.A. Pupil shape as viewed along the horizontal visual field. J. Vis. 2013, 13, 3. [Google Scholar] [CrossRef]
- Marshall, S. The Index of Cognitive Activity: Measuring cognitive workload. In Proceedings of the IEEE 7th Conference on Human Factors and Power Plants, Institute of Electrical and Electronics Engineers (IEEE), Scottsdale, AZ, USA, 15–19 September 2002; pp. 75–79. [Google Scholar]
- Duchowski, A.T.; Krejtz, K.; Kreitz, I.; Biele, C.; Niedzielska, A.; Kiefer, P.; Raubal, M.; Giannopoulos, I. The index of pupillary activity. In Proceedings of the 2018 CHI Conference on Human Factors in Computing Systems, Montreal, QC, Canada, 21–26 April 2018; ACM Press: New York, NY, USA. [Google Scholar]
- Stark, L.; Campbell, F.W.; Atwood, J. Pupil unrest: An example of noise in a biological servomechanism. Nat. Cell Biol. 1958, 182, 857–858. [Google Scholar] [CrossRef] [PubMed]
- Demberg, V.; Sayeed, A. The frequency of rapid pupil dilations as a measure of linguistic processing difficulty. PLoS ONE 2016, 11, e0146194. [Google Scholar] [CrossRef] [PubMed]
- Devos, H.; Akinwuntan, A.E.; Alissa, N.; Morohunfola, B.; Lynch, S. Cognitive performance and cognitive workload in multiple sclerosis: Two different constructs of cognitive functioning? Mult. Scler. Relat. Disord. 2020, 38, 101505. [Google Scholar] [CrossRef] [PubMed]
- Kahya, M.; Moon, S.; Lyons, K.E.; Pahwa, R.; Akinwuntan, A.E.; Devos, H. Pupillary response to cognitive demand in Parkinson’s Disease: A pilot study. Front. Aging Neurosci. 2018, 10, 90. [Google Scholar] [CrossRef]
- Moon, S.; Kahya, M.; Lyons, K.E.; Pahwa, R.; Akinwuntan, A.; Devos, H. Cognitive workload during verbal abstract reasoning in Parkinson’s disease: A pilot study. Int. J. Neurosci. 2020, 1–7. [Google Scholar] [CrossRef]
- Myers, J.S.; Alissa, N.; Mitchell, M.; Dai, J.; He, J.; Moon, S.; O’Dea, A.; Klemp, J.; Kurylo, M.; Akinwuntan, A.; et al. Pilot feasibility study examining pupillary response during driving simulation as a measure of cognitive load in breast cancer survivors. Oncol. Nurs. Forum 2020, 47, 203–212. [Google Scholar] [CrossRef]
- Myers, J.S.; Kahya, M.; Mitchell, M.; Dai, J.; He, J.; Moon, S.; Hamilton, K.; Valla, M.; O’Dea, A.; Klemp, J.; et al. Pupillary response: Cognitive effort for breast cancer survivors. Support. Care Cancer 2019, 27, 1121–1128. [Google Scholar] [CrossRef]
- Ranchet, M.; Orlosky, J.; Morgan, J.; Qadir, S.; Akinwuntan, A.E.; Devos, H. Pupillary response to cognitive workload during saccadic tasks in Parkinson’s disease. Behav. Brain Res. 2017, 327, 162–166. [Google Scholar] [CrossRef]
- Wang, C.; Gao, J.; Li, M.; Qi, H.; Zhao, T.; Zhang, B.; Zhou, C.; Fang, S. Association of cognitive impairment and mood disorder with event-related potential P300 in patients with cerebral small vessel diseases. Neuro Endocrinol. Lett. 2019, 40, 333–341. [Google Scholar]
- Jervis, B.W.; Bigan, C.; Besleaga, M.; Jervis, M.W. New-Onset Alzheimer’s Disease and normal subjects 100% differentiated by pam. J. Alzheimer’s Dis. Other Dement. 2019, 34, 308–313. [Google Scholar] [CrossRef]
- Ghani, U.; Signal, N.; Niazi, I.K.; Taylor, D. ERP based measures of cognitive workload: A review. Neurosci. Biobehav. Rev. 2020, 118, 18–26. [Google Scholar] [CrossRef]
- Hedges, D.; Janis, R.; Mickelson, S.; Keith, C.; Bennett, D.; Brown, B.L. P300 amplitude in Alzheimer’s Disease. Clin. EEG Neurosci. 2016, 47, 48–55. [Google Scholar] [CrossRef]
- Pedroso, R.V.; Fraga, F.J.; Icassatti Corazza, D.; Almeida Andreatto, C.A.; Gomes de Melo Coelho, F.; Riani Costa, J.L.; Ferreira Santos-Galduróz, R. Latência e amplitude do P300 auditivo na doença de Alzheimer: Uma revisão sistemática. Braz. J. Otorhinolaryngol. 2012, 78, 126–132. [Google Scholar] [CrossRef]
- Lee, M.-S.; Lee, S.-H.; Moon, E.-O.; Moon, Y.-J.; Kim, S.; Kim, S.-H.; Jung, I.-K. Neuropsychological correlates of the P300 in patients with Alzheimer’s disease. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2013, 40, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Chapman, R.M.; McCrary, J.W.; Gardner, M.N.; Sandoval, T.C.; Guillily, M.D.; Reilly, L.A.; DeGrush, E. Brain ERP components predict which individuals progress to Alzheimer’s Disease and which do not. Neurobiol. Aging 2011, 32, 1742–1755. [Google Scholar] [CrossRef] [PubMed]
- Bennys, K.; Portet, F.; Touchon, J.; Rondouin, G. Diagnostic value of event-related evoked potentials N200 and P300 subcomponents in early diagnosis of Alzheimer’s Disease and mild cognitive impairment. J. Clin. Neurophysiol. 2007, 24, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Rossini, P.; Di Iorio, R.; Vecchio, F.; Anfossi, M.; Babiloni, C.; Bozzali, M.; Bruni, A.; Cappa, S.; Escudero, J.; Fraga, F.; et al. Early diagnosis of Alzheimer’s disease: The role of biomarkers including advanced EEG signal analysis. Report from the IFCN-sponsored panel of experts. Clin. Neurophysiol. 2020, 131, 1287–1310. [Google Scholar] [CrossRef] [PubMed]
- Boutros, N.; Torello, M.W.; Burns, E.M.; Wu, S.-S.; Nasrallah, H.A. Evoked potentials in subjects at risk for Alzheimer’s Disease. Psychiatry Res. 1995, 57, 57–63. [Google Scholar] [CrossRef]
- Nieuwenhuis, S.; De Geus, E.J.; Aston-Jones, G. The anatomical and functional relationship between the P3 and autonomic components of the orienting response. Psychophysiology 2011, 48, 162–175. [Google Scholar] [CrossRef]
- Murphy, P.R.; Robertson, I.H.; Balsters, J.H.; O’Connell, R.G. Pupillometry and P3 index the locus coeruleus-noradrenergic arousal function in humans. Psychophysiology 2011, 48, 1532–1543. [Google Scholar] [CrossRef]
- Sperling, A.R.; Aisen, P.S.; Beckett, L.A.; Bennett, D.A.; Craft, S.; Fagan, A.M.; Iwatsubo, T.; Jack, C.R., Jr.; Kaye, J.; Montine, T.J.; et al. Toward defining the preclinical stages of Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer’s Dement. 2011, 7, 280–292. [Google Scholar] [CrossRef] [PubMed]
- Vidoni, E.D.; Yeh, H.-W.; Morris, J.K.; Newell, K.L.; Alqahtani, A.; Burns, N.C.; Burns, J.M.; Billinger, S.A. Cerebral β-Amyloid angiopathy is associated with earlier dementia onset in Alzheimer’s Disease. Neurodegener. Dis. 2016, 16, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Nasreddine, Z.S.; Phillips, N.A.; Bedirian, V.; Charbonneau, S.; Whitehead, V.; Collin, I.; Cummings, J.L.; Chertkow, H. The Montreal cognitive assessment, MoCA: A brief screening tool for mild cognitive impairment. J. Am. Geriatr. Soc. 2005, 53, 695–699. [Google Scholar] [CrossRef] [PubMed]
- Gevins, A.S.; Smith, M.E.; McEvoy, L.K.; Ilan, A.B.; Chan, C.S.; Jiang, A.; Sam-Vargas, L.; Abraham, G. A cognitive and neurophysiological test of change from an individual’s baseline. Clin. Neurophysiol. 2011, 122, 114–120. [Google Scholar] [CrossRef]
- Ikuma, L.H.; Nussbaum, M.A.; Babski-Reeves, K.L. Reliability of physiological and subjective responses to physical and psychosocial exposures during a simulated manufacturing task. Int. J. Ind. Ergon. 2009, 39, 813–820. [Google Scholar] [CrossRef]
- Marshall, S.P. Method and Apparatus for Eye Tracking and Monitoring Pupil Dilation to Evaluate Cognitive Activity. U.S. Patent 6090051A, 18 July 2000. [Google Scholar]
- Vogels, J.; Demberg, V.; Kray, J. The index of cognitive activity as a measure of cognitive processing load in dual task settings. Front. Psychol. 2018, 9, 2276. [Google Scholar] [CrossRef]
- Delorme, A.; Makeig, S. EEGLAB: An open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J. Neurosci. Methods 2004, 134, 9–21. [Google Scholar] [CrossRef]
- Lopez-Calderon, J.; Luck, S.J. ERPLAB: An open-source toolbox for the analysis of event-related potentials. Front. Hum. Neurosci. 2014, 8, 213. [Google Scholar] [CrossRef]
- Shrout, P.E.; Fleiss, J.L. Intraclass correlations: Uses in assessing rater reliability. Psychol. Bull. 1979, 86, 420–428. [Google Scholar] [CrossRef]
- Cicchetti, D.V. Guidelines, criteria, and rules of thumb for evaluating normed and standardized assessment instruments in psychology. Psychol. Assess. 1994, 6, 284. [Google Scholar] [CrossRef]
- Bland, J.M.; Altman, D.G. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986, 1, 307–310. [Google Scholar] [CrossRef]
- Donoghue, D.; Stokes, E.K. Physiotherapy research and older people (PROP) group how much change is true change? The minimum detectable change of the Berg Balance Scale in elderly people. J. Rehabil. Med. 2009, 41, 343–346. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.M.; Wang, Z.-M.; Wang, M.-Z.; Lan, Y.-J. The appraisal of reliability and validity of subjective workload assessment technique and NASA-task load index. Zhonghua Lao Dong Wei Sheng Zhi Ye Bing Za Zhi, 2005, 23, 178–181. (In Chinese) [Google Scholar]
- Lew, H.L.; Gray, M.; Poole, J.H. Temporal stability of auditory event-related potentials in healthy individuals and patients with traumatic brain injury. J. Clin. Neurophysiol. 2007, 24, 392–397. [Google Scholar] [CrossRef]
- Kahya, M.; Lyons, K.E.; Pahwa, R.; Akinwuntan, A.E.; He, J.; Devos, H. Reliability and validity of pupillary response during dual-task balance in Parkinson Disease. Arch. Phys. Med. Rehabil. 2020. [Google Scholar] [CrossRef]
- Kahya, M.; Liao, K.; Gustafson, K.; Akinwuntan, A.; Devos, H. Validation of pupillary response against EEG during dual-tasking postural control. Arch. Phys. Med. Rehabil. 2019, 100, e142. [Google Scholar] [CrossRef]
- Kim, M.; Barrett, A.M.; Heilman, K.M. Lateral asymmetries of pupillary responses. Cortex 1998, 34, 753–762. [Google Scholar] [CrossRef]
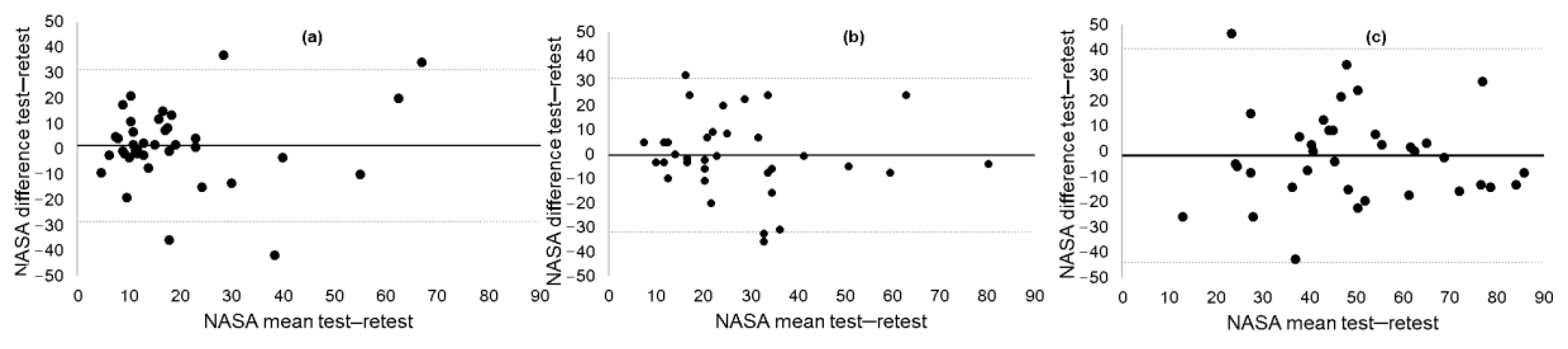
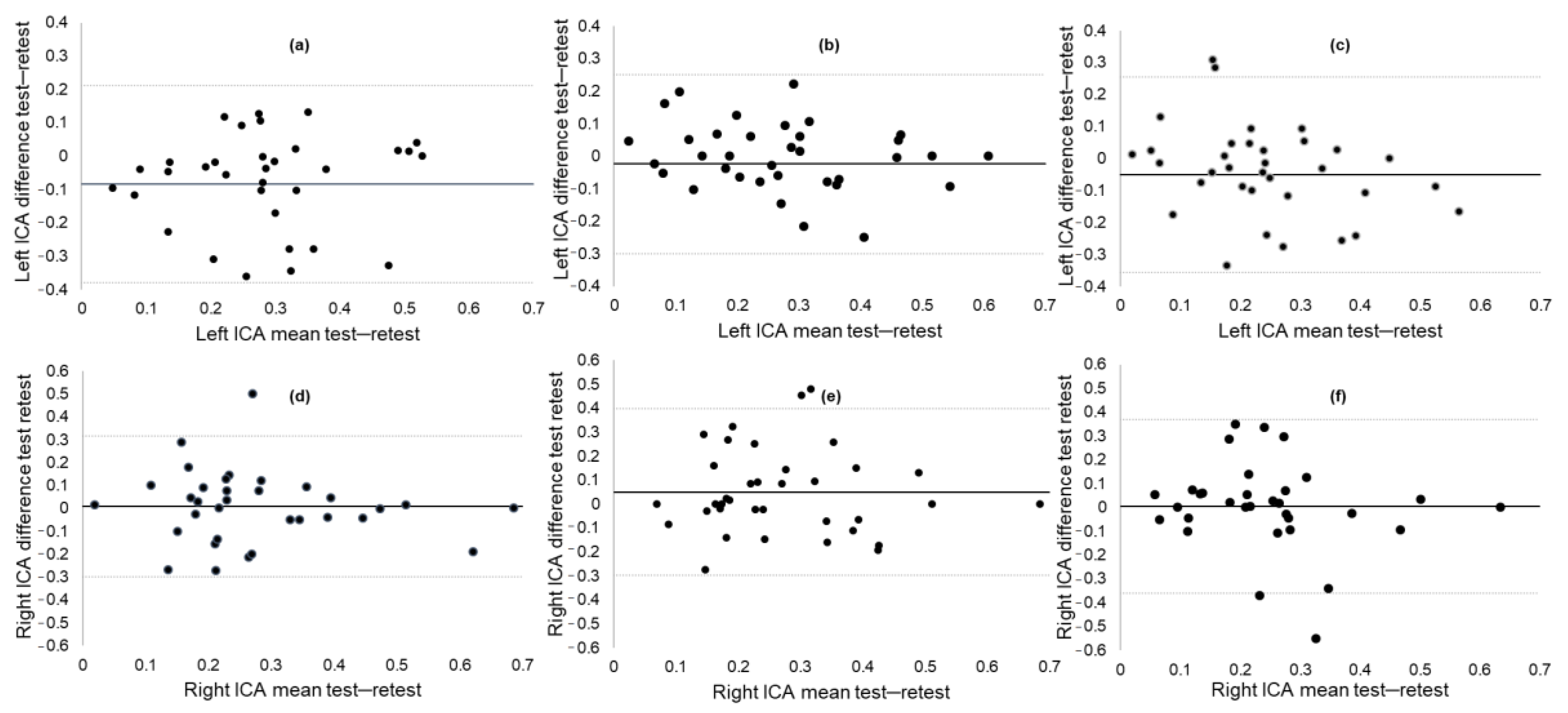
| Variable | Baseline | Follow-up | Pearson r | ICC, (95% CI) | MDC90 |
|---|---|---|---|---|---|
| 0-back, NASA-TLX | 19.51 (15.95) | 21.98 (17.89) | 0.68 a | 0.81 (0.61–0.90) a | 15.82 |
| 0-back, mean ICA L | 0.33 (0.14) | 0.24 (0.15) | 0.46 b | 0.63 (0.26–0.83) b | 0.20 |
| 0-back, mean ICA R | 0.27 (0.17) | 0.28 (0.16) | 0.55 b | 0.70 (0.40–0.85) a | 0.22 |
| 1-back, NASA-TLX | 28.24 (17.80) | 27.22 (16.96) | 0.60 a | 0.78 (0.57–0.89) a | 19.37 |
| 1-back, mean ICA L | 0.29 (0.17) | 0.27 (0.14) | 0.58 a | 0.73 (0.47–0.86) a | 0.20 |
| 1-back, mean ICA R | 0.25 (0.16) | 0.30 (0.15) | 0.39 c | 0.56 (0.12–0.78) b | 0.25 |
| 2-back, NASA-TLX | 50.92 (19.41) | 50.61 (19.13) | 0.55 b | 0.71 (0.42–0.85) a | 24.33 |
| 2-back, mean ICA L | 0.25 (0.14) | 0.23 (0.16) | 0.50 b | 0.64 (0.29–0.82) a | 0.24 |
| 2-back, mean ICA R | 0.24 (0.18) | 0.25 (0.15) | 0.45 b | 0.62 (0.20–0.82) b | 0.25 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Devos, H.; Gustafson, K.; Ahmadnezhad, P.; Liao, K.; Mahnken, J.D.; Brooks, W.M.; Burns, J.M. Psychometric Properties of NASA-TLX and Index of Cognitive Activity as Measures of Cognitive Workload in Older Adults. Brain Sci. 2020, 10, 994. https://doi.org/10.3390/brainsci10120994
Devos H, Gustafson K, Ahmadnezhad P, Liao K, Mahnken JD, Brooks WM, Burns JM. Psychometric Properties of NASA-TLX and Index of Cognitive Activity as Measures of Cognitive Workload in Older Adults. Brain Sciences. 2020; 10(12):994. https://doi.org/10.3390/brainsci10120994
Chicago/Turabian StyleDevos, Hannes, Kathleen Gustafson, Pedram Ahmadnezhad, Ke Liao, Jonathan D. Mahnken, William M. Brooks, and Jeffrey M. Burns. 2020. "Psychometric Properties of NASA-TLX and Index of Cognitive Activity as Measures of Cognitive Workload in Older Adults" Brain Sciences 10, no. 12: 994. https://doi.org/10.3390/brainsci10120994
APA StyleDevos, H., Gustafson, K., Ahmadnezhad, P., Liao, K., Mahnken, J. D., Brooks, W. M., & Burns, J. M. (2020). Psychometric Properties of NASA-TLX and Index of Cognitive Activity as Measures of Cognitive Workload in Older Adults. Brain Sciences, 10(12), 994. https://doi.org/10.3390/brainsci10120994






