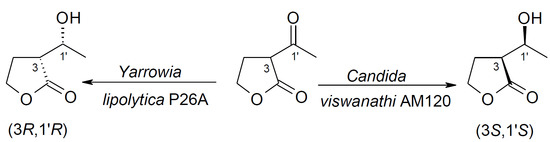
Yeast-Mediated Stereoselective Reduction of α-Acetylbutyrolactone
Abstract
1. Introduction
2. Materials and Methods
2.1. Analysis
2.2. Synthesis of Standards
2.3. Microorganisms
2.4. Screening Procedure
2.5. Preparation of Resting Cell Suspension
2.6. Biotransformation in the Presence of Organic Solvents
2.7. Biotransformation in the Presence of Deep Eutectic Solvents
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bräutigam, S.; Dennewald, D.; Schürmann, M.; Lutje-Spelberg, J.; Pitner, W.-R.; Weuster-Botz, D. Whole-cell biocatalysis: Evaluation of new hydrophobic ionic liquids for efficient asymmetric reduction of prochiral ketones. Enz. Microb. Technol. 2009, 45, 310–316. [Google Scholar] [CrossRef]
- Ribeiro, J.B.; Andrade de Sousa, L.M.; da Volta Soares, M.; da Conceição, M.; Ramos, K.V.; Aquino Neto, F.R.; Mansour Fraga, C.A.; Ferreira Leite, S.G.; Cordeiro, Y.; Antunes, O.A.C. Microbial reduction of α-acetyl-γ-butyrolactone. Tetrahedron Asymmetry 2006, 17, 984–988. [Google Scholar] [CrossRef]
- Venkataraman, S.; Roy, R.K.; Chadha, A. Asymmetric reduction of alkyl-3-oxobutanoates by Candida parapsilosis ATCC 7330: Insights into solvent and substrate optimisation of the biocatalytic reaction. Appl. Biochem. Biotechnol. 2013, 171, 756–770. [Google Scholar] [CrossRef] [PubMed]
- Contente, M.L.; Molinari, F.; Zambelli, P.; De Vitis, V.; Gandolfi, R.; Pinto, A.; Romano, D. Biotransformation of aromatic ketones and ketoesters with the non-conventional yeast Pichia glucozyma. Tetrahedron Lett. 2014, 55, 7051–7053. [Google Scholar] [CrossRef]
- Dennewald, D.; Pitner, W.-R.; Weuster-Botz, D. Recycling of the ionic liquid phase in process integrated biphasic whole-cell biocatalysis. Proc. Biochem. 2011, 46, 1132–1137. [Google Scholar] [CrossRef]
- Abbott, A.P.; Capper, G.; Davies, D.L.; Rasheed, R.K.; Tambyrajah, V. Novel solvent properties of choline chloride/urea mixtures. Chem. Commun. 2003, 70–71. [Google Scholar] [CrossRef]
- Abbott, A.P.; Capper, G.; Gray, S. Design of improved deep eutectic solvents using hole theory. ChemPhysChem 2006, 7, 803–806. [Google Scholar] [CrossRef] [PubMed]
- Abo-Hamad, A.; Hayyan, M.; AlSaadi, M.A.; Hashim, M.A. Potential application of deep eutectic solvents in nanotechnology. Chem. Eng. J. 2015, 273, 551–567. [Google Scholar] [CrossRef]
- Juneidi, I.; Hayyana, M.; Hashim, M.A. Intensification of biotransformations using deep eutectic solvents: Overview and outlook. Process Biochem. 2018, 66, 33–60. [Google Scholar] [CrossRef]
- Xu, P.; Zheng, G.-W.; Zong, M.-H.; Li, N.; Lou, W.-Y. Recent progress on deep eutectic solvents in biocatalysis. Biores. Bioproc. 2017, 4, 34. [Google Scholar] [CrossRef] [PubMed]
- AlOmar, M.K.; Hayyan, M.; Alsaadi, M.A.; Akib, S.; Hayyan, A.; Hashim, M.A. Glycerol-based deep eutectic solvents: Physical properties. J. Mol. Liq. 2016, 215, 98–103. [Google Scholar] [CrossRef]
- Bubalo, M.C.; Mazur, M.; Radosevic, K.; Radojcic Redovnikov, I. Baker’s yeast-mediated asymmetric reduction of ethyl 3-oxobutanoate in deep eutectic solvents. Process Biochem. 2015, 50, 1788–1792. [Google Scholar] [CrossRef]
- Maugeri, Z.; Dominguez de Maria, P. Whole-Cell Biocatalysis in Deep-Eutectic-Solvents/Aqueous Mixtures. ChemCatChem 2014, 6, 1535–1537. [Google Scholar] [CrossRef]
- Dai, Y.; Huan, B.; Zhang, H.-S.; He, Y.-C. Effective biotransformation of ethyl 4-chloro-3-oxobutanoate into ethyl (S)-4-chloro-3-hydroxybutanoate by recombinant E. coli CCZU-T15 whole cells in [ChCl][Gly]–water media. Appl. Biochem. Biotechnol. 2017, 181, 1347–1359. [Google Scholar] [CrossRef] [PubMed]
- Vitale, P.; Abbinante, V.M.; Perna, F.M.; Salomone, A.; Cardellicchio, C.; Capriati, V. Unveiling the hidden performance of whole cells in the asymmetric bioreduction of aryl-containing ketones in aqueous deep eutectic solvents. Adv. Synth. Catal. 2017, 359, 1049–1057. [Google Scholar] [CrossRef]
- Müller, C.R.; Lavandera, I.; Gotor-Fernández, V.; Domínguez de María, P. Performance of Recombinant-Whole-Cell-Catalyzed Reductions in Deep-Eutectic-Solvent–Aqueous-Media Mixtures. ChemCatChem 2015, 7, 2654–2659. [Google Scholar] [CrossRef]
- Teixeira, L.H.P.; de Souza, M.C.B.V.; da Conceição, K.V.; Ramos, M.; de Aquino Neto, F.R.; Barreiro, E.J.; Fraga, C.A.M. Studies on the diastereoselective reduction of 2-acetyl-2-alkyl-γ-butyrolactones with boron hydrides. Synth. Commun. 2002, 32, 505–526. [Google Scholar] [CrossRef]
- Fantin, G.; Fogagnolo, M.; Giovannini, P.P.; Medici, A.; Pedrini, P.; Gardini, F.; Lanciotti, R. Anti-Prelog microbial reduction of prochiral carbonyl compounds. Tetrahedron 1996, 52, 3547–3552. [Google Scholar] [CrossRef]
- Fantin, G.; Fogagnolo, M.; Giovannini, P.; Medici, A.; Pagnotta, E.; Pedrini, P.; Trincone, A. Synthesis of homochiral syn- and anti-α-(hydroxyethyl)-γ-buttyrolactones via microbial reduction. Tetrahedron Asymmetry 1994, 5, 1631–1634. [Google Scholar] [CrossRef]
- Tala-Tapeha, S.M.; Mahmoodib, N.O.; Ghanbari Pirbastib, F.; Azimi, A. Saccharomyces cerevisiae as a biocatalyst for different carbonyl group under green condition. Org. Chem. Res. 2016, 2, 39–42. [Google Scholar] [CrossRef]
- Romagnolo, A.; Spina, F.; Risso, S.; Brenna, E.; Crotti, M.; Varese, G.C. A competitive approach for the reduction of unsaturated compounds based on fungal ene-reductases. Mycosphere 2013, 4, 363–454. [Google Scholar] [CrossRef]
- He, C.; Chang, D.; Zhang, J. Asymmetric reduction of substituted α- and β-ketoesters by Bacillus pumilus Phe-C3. Tetrahedron Asymmetry 2008, 19, 1347–1351. [Google Scholar] [CrossRef]
- Brenna, E.; Fronza, G.; Fuganti, C.; Parmeggiani, F. Investigation of the stereochemical course of ene reductase-catalysed reactions by deuterium labelling. Isotopes Environ. Health Stud. 2015, 51, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Kalaitzakis, D.; Rozzell, J.D.; Kambourakis, S.; Smonou, I. Highly Stereoselective Reductions of α-alkyl-1,3-diketones and α-alkyl-β-keto esters catalyzed by isolated NADPH-dependent ketoreductases. Org. Lett. 2005, 7, 4799–4801. [Google Scholar] [CrossRef] [PubMed]
- Napora-Wijata, K.; Strohmeier, G.A.; Sonavane, M.N.; Avi, M.; Robins, K.; Winkler, M. Enantiocomplementary Yarrowia lipolytica oxidoreductases: Alcohol dehydrogenase 2 and short chain dehydrogenase/reductase. Biomolecules 2013, 3, 449–460. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; Xu, X.; Huang, H.; Li, S. Efficient synthesis of (R)-2-chloro-1-phenylethol using a yeast carbonyl reductase with broad substrate spectrum and 2-propanol as cosubstrate. Biochem. Eng. J. 2015, 103, 277–285. [Google Scholar] [CrossRef]
- Johanson, T.; Katz, M.; Gorwa-Grauslund, M.F. Strain engineering for stereoselective bioreduction of dicarbonyl compounds by yeast reductases. FEMS Yeast Res. 2005, 5, 513–525. [Google Scholar] [CrossRef] [PubMed]
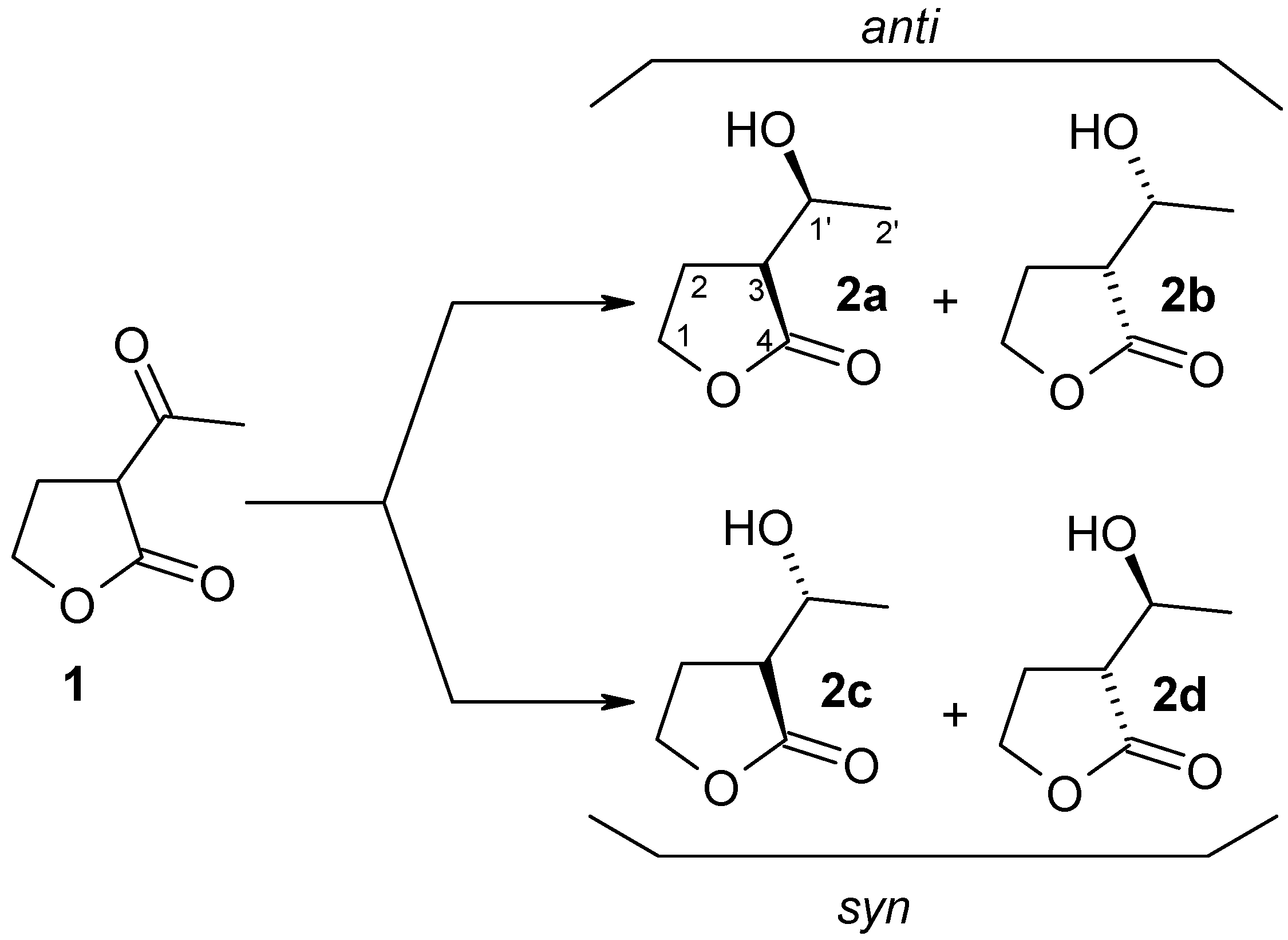
| Yeast Strain | Time [days] | Substrate | Stereoisomers of Product | ||
|---|---|---|---|---|---|
| 1 [%] | 2a + 2b (anti) [%] | 2c + 2d (syn) [%] | de [%] | ||
| Yarrowia lipolytica P26A | 1 | - | 100 | - | >99 |
| Yarrowia lipolytica AM71 | 1 | 15 | 85 | - | >99 |
| 2 | - | 100 | - | >99 | |
| Yarrowia lipolytica AM72 | 1 | 19 | 80 | - | >99 |
| 2 | 10 | 90 | - | >99 | |
| 3 | - | 100 | - | >99 | |
| Candida viswanathi AM120 | 1 | 7 | 89 | 3.9 | 92 |
| 2 | - | 95 | 4.8 | 90 | |
| Hansenula anomala C2 | 1 | 80 | 20 | - | >99 |
| 2 | 68 | 30 | 2 | 89 | |
| 3 | 60 | 38 | 2 | 89 | |
| 4 | 46 | 51 | 3 | 90 | |
| Saccharomyces cerevisiae K1 | 1 | 100 | - | - | - |
| 2 | 100 | - | - | - | |
| 3 | 100 | - | - | - | |
| 4 | 100 | - | - | - | |
| Saccharomyces pombe C1 | 1 | 100 | - | - | - |
| 2 | 100 | - | - | - | |
| 3 | 100 | - | - | - | |
| 4 | 100 | - | - | - | |
| Strain | Time [days] | 1 [%] | anti | ee [%] | syn | ee [%] | ||
|---|---|---|---|---|---|---|---|---|
| 2a (3S,1’S) [%] | 2b (3R,1’R)[%] | 2c (3S,1’R) [%] | 2d (3R,1’S) [%] | |||||
| Yarrowia lipolytica P26A | 1 | 0 | 0 | 100 | >99 | 0 | 0 | - |
| Yarrowia lipolytica AM71 | 1 | 10 | 0 | 90 | >99 | 0 | 0 | - |
| 2 | 0 | 0 | 100 | >99 | 0 | 0 | - | |
| Candida viswanathi AM120 | 1 | 3 | 81 | 12 | 73 | 0 | 4.0 | >99 |
| 2 | 0 | 83 | 13 | 73 | 0 | 4.3 | >99 | |
| Strain | Time [days] | 1 [%] | anti | ee [%] | syn | ee [%] | ||
|---|---|---|---|---|---|---|---|---|
| 2a (3S,1’S) [%] | 2b (3R,1’R) [%] | 2c (3S,1’R) [%] | 2d (3R,1’S) [%] | |||||
| Yarrowia lipolytica P26A | 1 | 2 | 1.0 | 97 | 98 | 0 | 0 | - |
| 2 | 0 | 1.3 | 99 | 97 | 0 | 0 | - | |
| Yarrowia lipolytica AM71 | 1 | 2 | 0.5 | 97 | 99 | 0 | 0 | - |
| 2 | 2 | 1 | 97 | 98 | 0 | 0 | - | |
| 3 | 0 | 2 | 98 | 96 | 0 | 0 | - | |
| Candida viswanathi AM120 | 1 | 26 | 45 | 15 | 51 | 0 | 14 | >99 |
| 2 | 7 | 60 | 18 | 55 | 0 | 15 | >99 | |
| 3 | 0 | 65 | 19 | 56 | 0 | 16 | >99 | |
| Strain | Time [days] | 1 [%] | anti | ee [%] | syn | ee [%] | ||
|---|---|---|---|---|---|---|---|---|
| 2a (3S,1’S) [%] | 2b (3R,1’R) [%] | 2c (3S,1’R) [%] | 2d (3R,1’S) [%] | |||||
| Yarrowia lipolytica P26A | 1 | 0 | 1 | 99 | 99 | 0 | 0.3 | >99 |
| Yarrowia lipolytica AM71 | 1 | 0 | 1 | 99 | 99 | 0 | 0.6 | >99 |
| Candida viswanathi AM120 | 1 | 1 | 65 | 10 | 73 | 0 | 23.9 | >99 |
| 2 | 0 | 67 | 11 | 72 | 0 | 22.4 | >99 | |
| Solvent | % of Solvent [%] | 1 [%] | anti | ee [%] | syn | ee [%] | ||
|---|---|---|---|---|---|---|---|---|
| 2a (3S,1’S) [%] | 2b (3R,1’R) [%] | 2c (3S,1’R) [%] | 2d (3R,1’S) [%] | |||||
| Candida viswanathi AM120 | ||||||||
| Ethanol | 5 | 51 | 38 | 7 | 69 | 0 | 4 | >99 |
| 10 | 100 | 0 | 0 | - | 0 | 0 | - | |
| 20 | 100 | 0 | 0 | - | 0 | 0 | - | |
| Glycerol | 5 | 0 | 78 | 15 | 67 | 0 | 7 | >99 |
| 10 | 0.5 | 76 | 16 | 64 | 0 | 7 | >99 | |
| 20 | 1.6 | 65 | 20 | 53 | 0 | 13 | >99 | |
| Hexane | 5 | 5 | 76 | 14 | 69 | 0 | 6 | >99 |
| 10 | 17 | 66 | 10 | 73 | 0 | 7 | >99 | |
| 20 | 17 | 68 | 11 | 73 | 0 | 5 | >99 | |
| Isopropanol | 5 | 3 | 81 | 12 | 73 | 0 | 4.0 | >99 |
| 10 | 1 | 81 | 14 | 71 | 0 | 4 | >99 | |
| 20 | 3 | 78 | 17 | 65 | 0 | 2 | >99 | |
| Yarrowia lipolytica P26a | ||||||||
| Ethanol | 5 | 8 | 0 | 92 | >99 | 0 | 0 | - |
| 10 | 96 | 0 | 4 | >99 | 0 | 0 | - | |
| 20 | 100 | 0 | 0 | - | 0 | 0 | - | |
| Glycerol | 5 | 0 | 0 | 100 | >99 | 0 | 0 | - |
| 10 | 0 | 0 | 100 | >99 | 0 | 0 | - | |
| 20 | 1 | 0 | 93 | >99 | 6 | 0 | >99 | |
| Hexane | 5 | 0 | 0 | 100 | >99 | 0 | 0 | - |
| 10 | 0 | 0 | 100 | >99 | 0 | 0 | - | |
| 20 | 0 | 0 | 94 | >99 | 5 | 1 | 53 | |
| Isopropanol | 5 | 3 | 0 | 95 | >99.9 | 0 | 2 | >99 |
| 10 | 2 | 0 | 97 | >99.9 | 0 | 1 | >99 | |
| 20 | 8 | 0 | 91 | >99.9 | 0 | 1 | >99 | |
| DES [%] | Time [days] | 1 [%] | anti | ee [%] | syn | ee [%] | ||
|---|---|---|---|---|---|---|---|---|
| 2a (3S,1’S) [%] | 2b (3R,1’R) [%] | 2c (3S,1’R) [%] | 2d (3R,1’S) [%] | |||||
| 10 | 1 | 1 | 81 | 11 | 76 | 0 | 7 | >99 |
| 2 | 0 | 81 | 11 | 76 | 0 | 8 | >99 | |
| 25 | 1 | 34 | 49 | 15 | 64 | 0 | 2 | >99 |
| 2 | 0 | 80 | 16 | 66 | 0 | 4 | >99 | |
| 50 | 1 | 100 | 0 | 0 | - | 0 | 0 | - |
| 2 | 72 | 17 | 11 | 20 | 0 | 2 | >99 | |
| 3 | 50 | 27 | 20 | 16 | 0 | 4 | >99 | |
| 7 | 37 | 34 | 25 | 14 | 0 | 4 | >99 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mączka, W.; Wińska, K.; Grabarczyk, M.; Żarowska, B. Yeast-Mediated Stereoselective Reduction of α-Acetylbutyrolactone. Appl. Sci. 2018, 8, 1334. https://doi.org/10.3390/app8081334
Mączka W, Wińska K, Grabarczyk M, Żarowska B. Yeast-Mediated Stereoselective Reduction of α-Acetylbutyrolactone. Applied Sciences. 2018; 8(8):1334. https://doi.org/10.3390/app8081334
Chicago/Turabian StyleMączka, Wanda, Katarzyna Wińska, Małgorzata Grabarczyk, and Barbara Żarowska. 2018. "Yeast-Mediated Stereoselective Reduction of α-Acetylbutyrolactone" Applied Sciences 8, no. 8: 1334. https://doi.org/10.3390/app8081334
APA StyleMączka, W., Wińska, K., Grabarczyk, M., & Żarowska, B. (2018). Yeast-Mediated Stereoselective Reduction of α-Acetylbutyrolactone. Applied Sciences, 8(8), 1334. https://doi.org/10.3390/app8081334