Hydrophobic Ceramic Membranes for Water Desalination
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
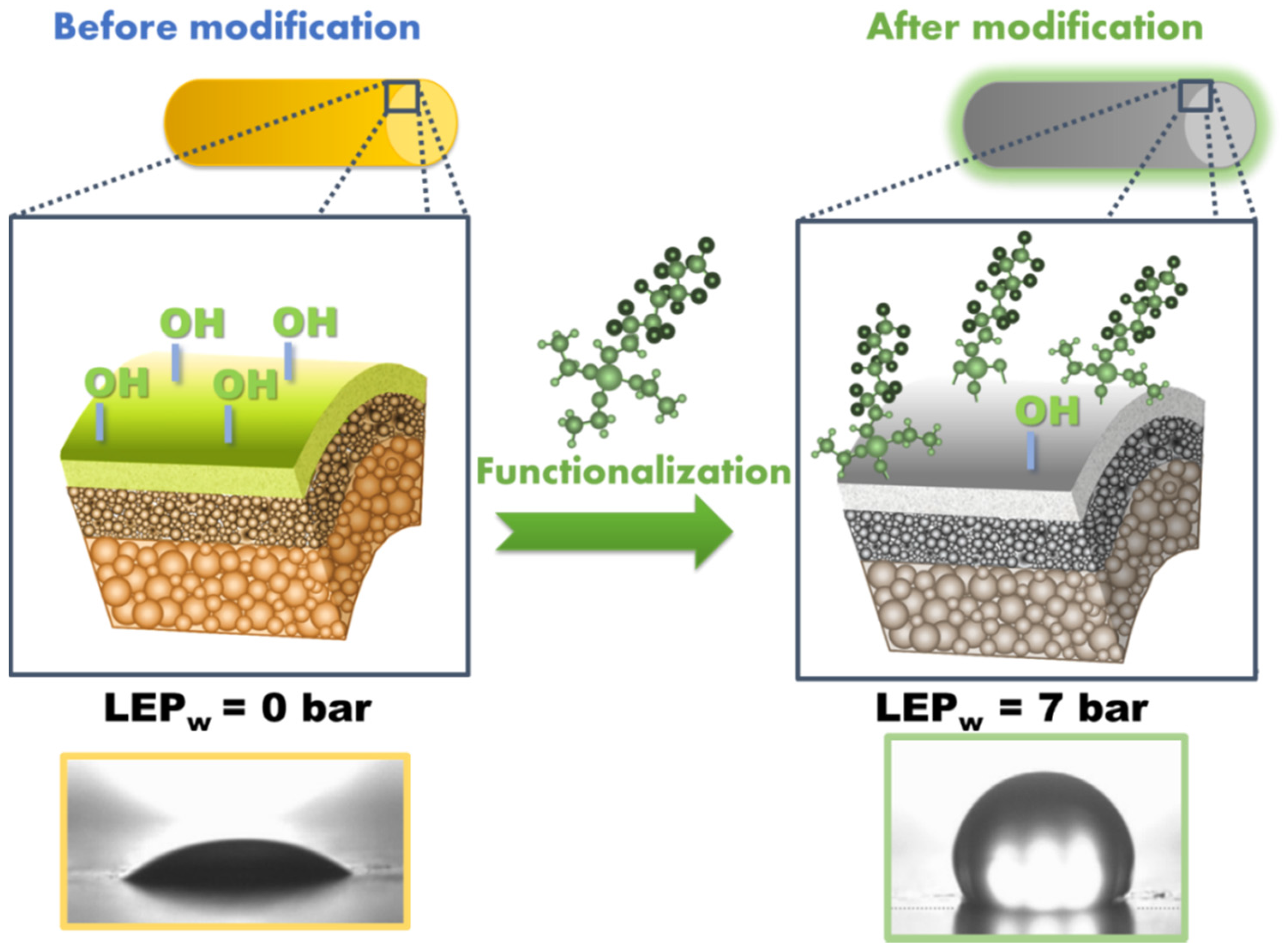
2.2. Experimental Protocol of Ceramic Membranes Modification
2.3. Modified Membrane Characterization—Analytical Methods
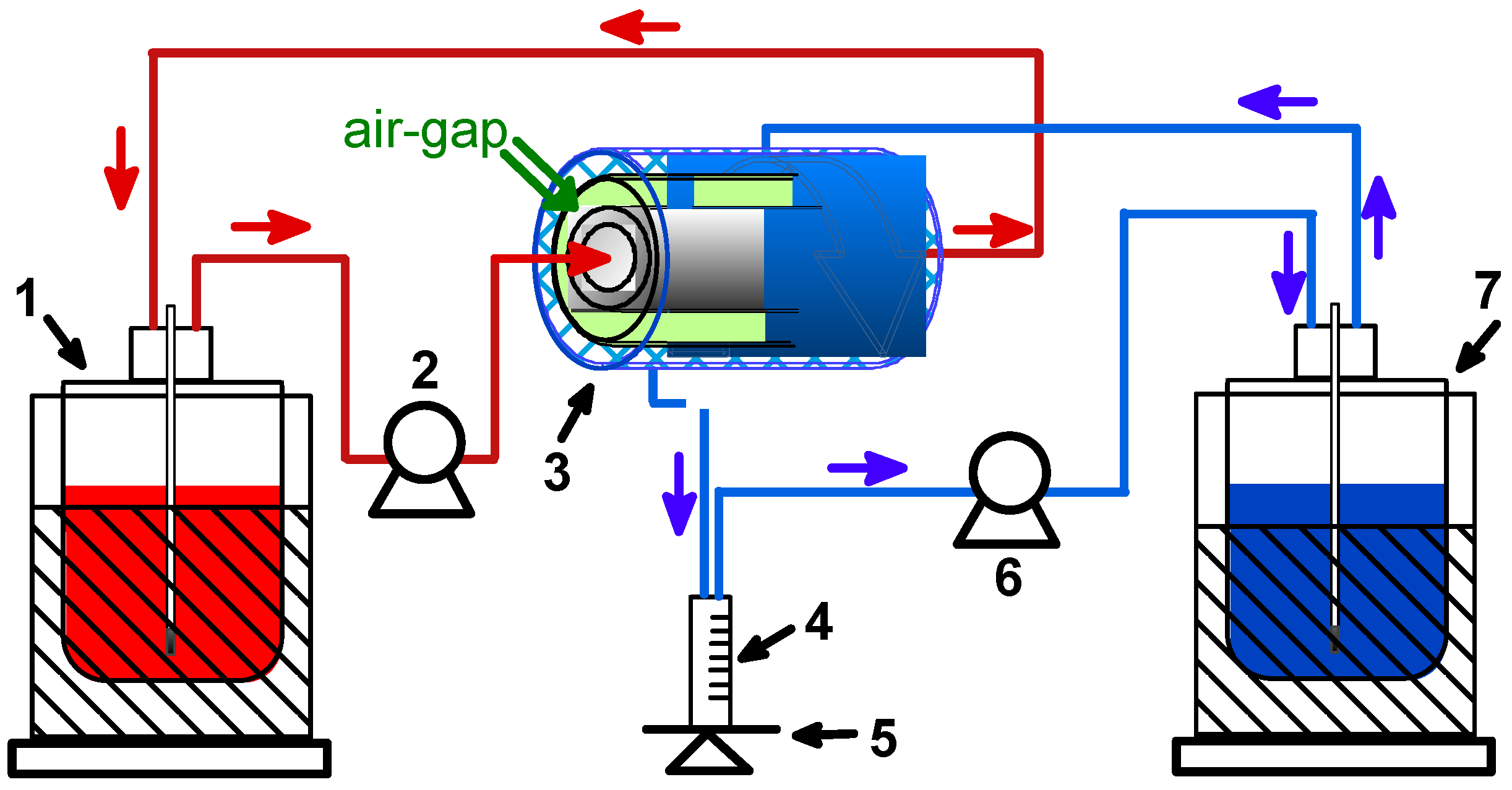
2.4. Air-Gap Membrane Distillation (AGMD)
3. Results and Discussion
3.1. Pristine Ceramic Membranes
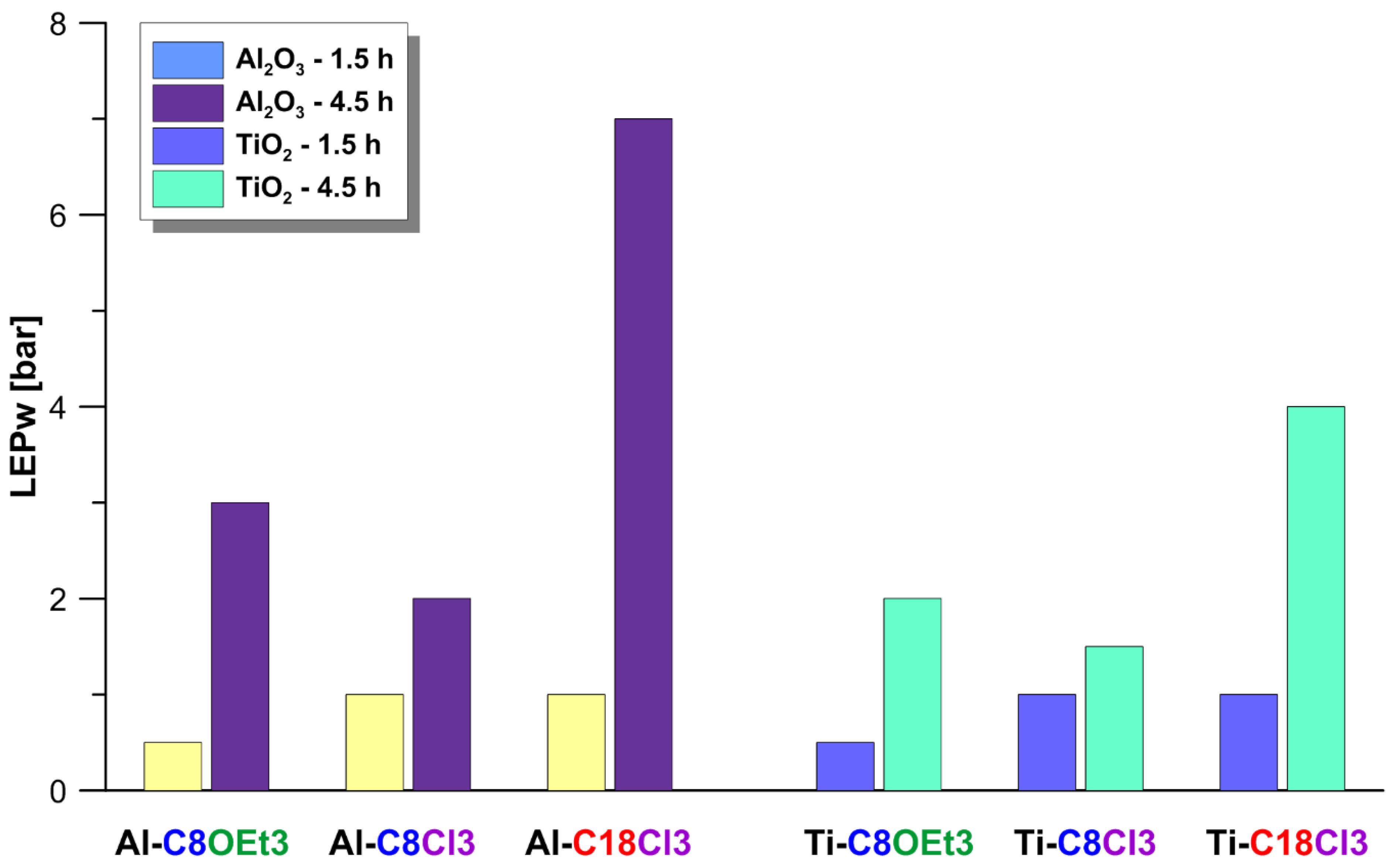
3.2. Effectiveness of Membrane Hydrophobization
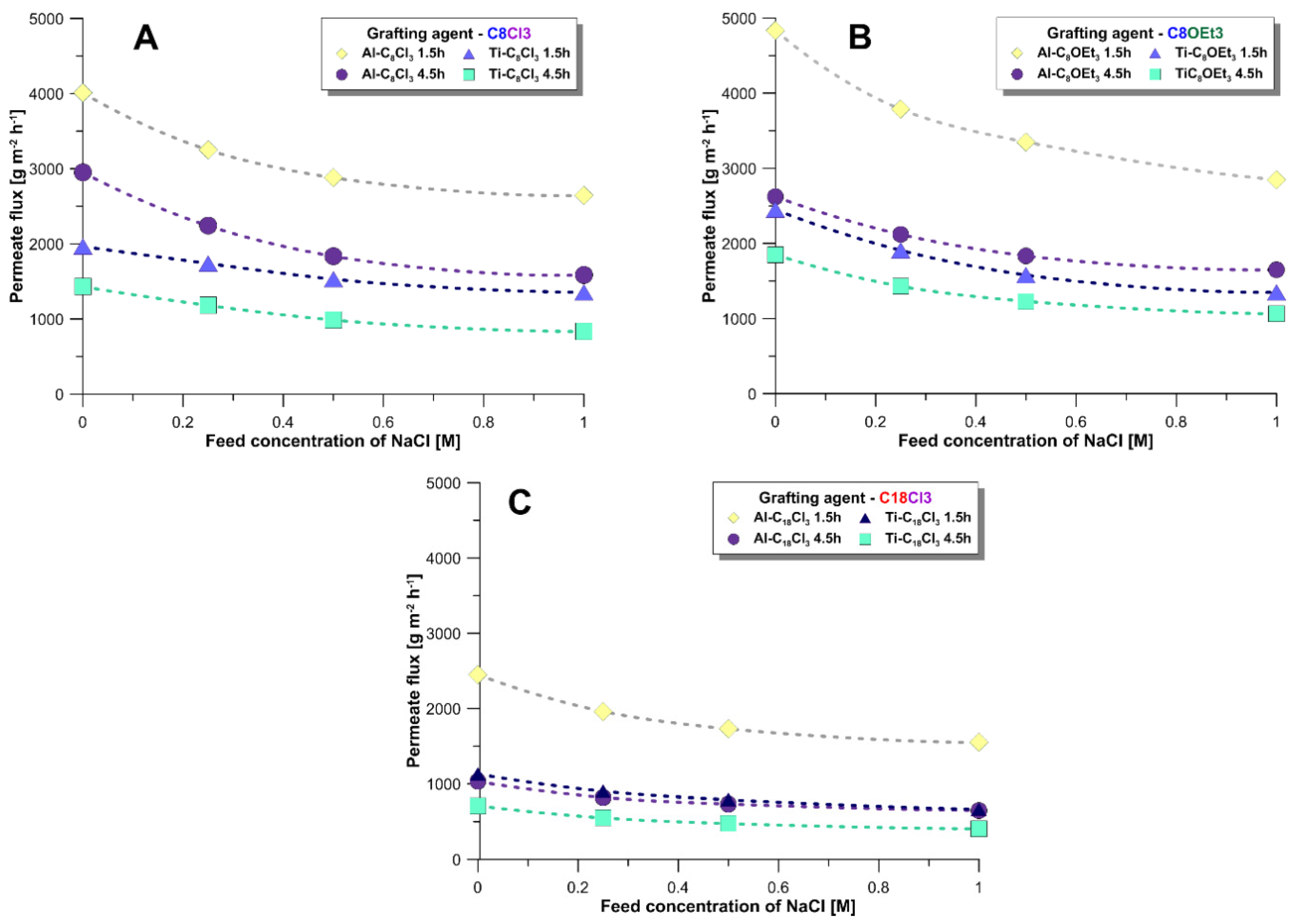
3.3. Air-Gap Membrane Distillation—Separation Efficiency of Modified Ceramic Membranes
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Woo, Y.C.; Tijing, L.D.; Shim, W.-G.; Choi, J.-S.; Kim, S.-H.; He, T.; Drioli, E.; Shon, H.K. Water desalination using graphene-enhanced electrospun nanofiber membrane via air gap membrane distillation. J. Membr. Sci. 2016, 520, 99–110. [Google Scholar] [CrossRef]
- Quist-Jensen, C.A.; Macedonio, F.; Conidi, C.; Cassano, A.; Aljlil, S.; Alharbi, O.A.; Drioli, E. Direct contact membrane distillation for the concentration of clarified orange juice. J. Food Eng. 2016, 187, 37–43. [Google Scholar] [CrossRef]
- Wang, P.; Chung, T.-S. Recent advances in membrane distillation processes: Membrane development, configuration design and application exploring. J. Membr. Sci. 2015, 474, 39–56. [Google Scholar] [CrossRef]
- Drioli, E.; Ali, A.; Simone, S.; Macedonio, F.; Al-Jlil, S.A.; Al Shabonah, F.S.; Al-Romaih, H.S.; Al-Harbi, O.; Figoli, A.; Criscuoli, A. Novel pvdf hollow fiber membranes for vacuum and direct contact membrane distillation applications. Sep. Purif. Technol. 2013, 115, 27–38. [Google Scholar] [CrossRef]
- Quist-Jensen, C.A.; Macedonio, F.; Horbez, D.; Drioli, E. Reclamation of sodium sulfate from industrial wastewater by using membrane distillation and membrane crystallization. Desalination 2017, 401, 112–119. [Google Scholar] [CrossRef]
- El-Bourawi, M.S.; Ding, Z.; Ma, R.; Khayet, M. A framework for better understanding membrane distillation separation process. J. Membr. Sci. 2006, 285, 4–29. [Google Scholar] [CrossRef]
- Alkhudhiri, A.; Darwish, N.; Hilal, N. Membrane distillation: A comprehensive review. Desalination 2012, 287, 2–18. [Google Scholar] [CrossRef]
- Shannon, M.A.; Bohn, P.W.; Elimelech, M.; Georgiadis, J.G.; Marinas, B.J.; Mayes, A.M. Science and technology for water purification in the coming decades. Nature 2008, 452, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Drioli, E.; Wu, Y. Membrane distillation: An experimental study. Desalination 1985, 53, 339–346. [Google Scholar] [CrossRef]
- Drioli, E.; Wu, Y.; Calabro, V. Membrane distillataion in the treatment of aqueous solutions. J. Membr. Sci. 1987, 33, 277–284. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, M.; Sun, D.; Li, B.; Li, P. Evaluation of commercial PTFE membranes for desalination of brine water through vacuum membrane distillation. Chem. Eng. J. 2016, 110, 52–63. [Google Scholar] [CrossRef]
- Tong, D.; Wang, X.; Ali, M.; Lan, C.Q.; Wang, Y.; Drioli, E.; Wang, Z.; Cui, Z. Preparation of Hyflon AD60/PVDF composite hollow fiber membranes for vacuum membrane distillation. Sep. Purif. Technol. 2016, 157, 1–8. [Google Scholar] [CrossRef]
- Rácz, G.; Kerker, S.; Schmitz, O.; Schnabel, B.; Kovács, Z.; Vatai, G.; Ebrahimi, M.; Czermak, P. Experimental determination of liquid entry pressure (LEP) in vacuum membrane distillation for oily wastewaters. Membr. Water Treat. 2015, 6, 237–249. [Google Scholar] [CrossRef]
- Kujawa, J.; Guillen-Burrieza, E.; Arafat, H.A.; Kurzawa, M.; Wolan, A.; Kujawski, W. Raw juice concentration by osmotic membrane distillation process with hydrophobic polymeric membranes. Food Bioprocess Technol. 2015, 8, 2146–2158. [Google Scholar] [CrossRef]
- Guo, F.; Servi, A.; Liu, A.; Gleason, K.K.; Rutledge, G.C. Desalination by membrane distillation using electrospun polyamide fiber membranes with surface fluorination by chemical vapor deposition. ACS Appl. Mater. Interfaces 2015, 7, 8225–8232. [Google Scholar] [CrossRef] [PubMed]
- Criscuoli, A.; Carnevale, M.C. Desalination by vacuum membrane distillation: The role of cleaning on the permeate conductivity. Desalination 2015, 365, 213–219. [Google Scholar] [CrossRef]
- Gryta, M. The application of membrane distillation for broth separation in membrane bioreactors. J. Membr. Sci. Res. 2016, 2, 193–200. [Google Scholar]
- Dong, G.; Kim, J.F.; Kim, J.H.; Drioli, E.; Lee, Y.M. Open-source predictive simulators for scale-up of direct contact membrane distillation modules for seawater desalination. Desalination 2017, 402, 72–87. [Google Scholar] [CrossRef]
- Gryta, M.; Tomaszewska, M.; Karakulski, K. Wastewater treatment by membrane distillation. Desalination 2006, 198, 67–73. [Google Scholar] [CrossRef]
- Eykens, L.; De Sitter, K.; Dotremont, C.; Pinoy, L.; Van der Bruggen, B. How to optimize the membrane properties for membrane distillation: A review. Ind. Eng. Chem. Res. 2016, 55, 9333–9343. [Google Scholar] [CrossRef]
- Peydayesh, M.; Kazemi, P.; Bandegi, A.; Mohammadi, T.; Bakhtiari, O. Treatment of bentazon herbicide solutions by vacuum membrane distillation. J. Water Process Eng. 2015, 8, e17–e22. [Google Scholar] [CrossRef]
- Li, X.; Yu, X.; Cheng, C.; Deng, L.; Wang, M.; Wang, X. Electrospun superhydrophobic organic/inorganic composite nanofibrous membranes for membrane distillation. ACS Appl. Mater. Interfaces 2015, 7, 21919–21930. [Google Scholar] [CrossRef] [PubMed]
- Guillen-Burrieza, E.; Servi, A.; Lalia, B.S.; Arafat, H.A. Membrane structure and surface morphology impact on the wetting of md membranes. J. Membr. Sci. 2015, 483, 94–103. [Google Scholar] [CrossRef]
- Xu, W.-T.; Zhao, Z.-P.; Liu, M.; Chen, K.-C. Morphological and hydrophobic modifications of PVDF flat membrane with silane coupling agent grafting via plasma flow for VMD of ethanol–water mixture. J. Membr. Sci. 2015, 491, 110–120. [Google Scholar] [CrossRef]
- Chen, K.; Xiao, C.; Huang, Q.; Liu, H.; Liu, H.; Wu, Y.; Liu, Z. Study on vacuum membrane distillation (VMD) using fep hollow fiber membrane. Desalination 2015, 375, 24–32. [Google Scholar] [CrossRef]
- Zaragoza, G.; Ruiz-Aguirre, A.; Guillén-Burrieza, E. Efficiency in the use of solar thermal energy of small membrane desalination systems for decentralized water production. Appl. Energy 2014, 130, 491–499. [Google Scholar] [CrossRef]
- Drioli, E.; Ali, A.; Macedonio, F. Membrane distillation: Recent developments and perspectives. Desalination 2015, 356, 56–84. [Google Scholar] [CrossRef]
- Michalkiewicz, B.; Ziebro, J.; Tomaszewska, M. Preliminary investigation of low pressure membrane distillation of methyl bisulphate from its solutions in fuming sulphuric acid combined with hydrolysis to methanol. J. Membr. Sci. 2006, 286, 223–227. [Google Scholar] [CrossRef]
- Orecki, A.; Tomaszewska, M.U.; Morawski, A.W. Treatment of natural waters by nanofiltration. Przem. Chem. 2006, 85, 1067–1070. [Google Scholar]
- Drioli, E. Guest editorial for the special issue: Membrane distillation and related membrane systems. Desalination 2013, 323, 1. [Google Scholar] [CrossRef]
- Ahmad, N.A.; Leo, C.P.; Ahmad, A.L. Superhydrophobic alumina membrane by steam impingement: Minimum resistance in microfiltration. Sep. Purif. Technol. 2013, 107, 187–194. [Google Scholar] [CrossRef]
- Bico, J.; Thiele, U.; Quéré, D. Wetting of textured surfaces. Colloids Surf. A Physicochem. Eng. Asp. 2002, 206, 41–46. [Google Scholar] [CrossRef]
- Onda, T.; Shibuichi, S.; Satoh, N.; Tsujii, K. Super-water-repellent fractal surfaces. Langmuir 1996, 12, 2125–2127. [Google Scholar] [CrossRef]
- Chen, W.; Fadeev, A.Y.; Hsieh, M.C.; Öner, D.; Youngblood, J.; McCarthy, T.J. Ultrahydrophobic and ultralyophobic surfaces: Some comments and examples. Langmuir 1999, 15, 3395–3399. [Google Scholar] [CrossRef]
- Wu, Y.; Sugimura, H.; Inoue, Y.; Takai, O. Thin films with nanotextures for transparent and ultra water-repellent coatings produced from trimethylmethoxysilane by microwave plasma cvd. Chem. Vap. Depos. 2002, 8, 47–50. [Google Scholar] [CrossRef]
- Tsujii, K.; Yamamoto, T.; Onda, T.; Shibuichi, S. Super oil-repellent surfaces. Angew. Chem. Int. Ed. 1997, 36, 1011–1012. [Google Scholar] [CrossRef]
- Tadanaga, K.; Katata, N.; Minami, T. Formation process of super-water-repellent Al2O3 coating films with high transparency by the sol–gel method. J. Am. Ceram. Soc. 1997, 80, 3213–3216. [Google Scholar] [CrossRef]
- Alami Younssi, S.; Iraqi, A.; Rafiq, M.; Persin, M.; Larbot, A.; Sarrazin, J. γ alumina membranes grafting by organosilanes and its application to the separation of solvent mixtures by pervaporation. Sep. Sci. Technol. 2003, 32, 175–179. [Google Scholar] [CrossRef]
- Kujawa, J.; Cerneaux, S.; Kujawski, W. Investigation of the stability of metal oxide powders and ceramic membranes grafted by perfluoroalkylsilanes. Colloids Surf. A Physicochem. Eng. Asp. 2014, 443, 109–117. [Google Scholar] [CrossRef]
- Kujawa, J.; Kujawski, W.; Koter, S.; Jarzynka, K.; Rozicka, A.; Bajda, K.; Cerneaux, S.; Persin, M.; Larbot, A. Membrane distillation properties of TiO2 ceramic membranes modified by perfluoroalkylsilanes. Desalin. Water Treat. 2013, 51, 1352–1361. [Google Scholar] [CrossRef]
- Kujawa, J.; Kujawski, W.; Koter, S.; Rozicka, A.; Cerneaux, S.; Persin, M.; Larbot, A. Efficiency of grafting of Al2O3, TiO2 and ZrO2 powders by perfluoroalkylsilanes. Colloid Surfaces A 2013, 420, 64–73. [Google Scholar] [CrossRef]
- Kujawski, W.; Kujawa, J.; Wierzbowska, E.; Cerneaux, S.; Bryjak, M.; Kujawski, J. Influence of hydrophobization conditions and ceramic membranes pore size on their properties in vacuum membrane distillation of water–organic solvent mixtures. J. Membr. Sci. 2016, 499, 442–451. [Google Scholar] [CrossRef]
- Kujawska, A.; Kujawski, J.K.; Bryjak, M.; Cichosz, M.; Kujawski, W. Removal of volatile organic compounds from aqueous solutions applying thermally driven membrane processes. 2. Air gap membrane distillation. J. Membr. Sci. 2016, 499, 245–256. [Google Scholar] [CrossRef]
- Kujawa, J.; Kujawski, W. Functionalization of ceramic metal oxide powders and ceramic membranes by perfluoroalkylsilanes and alkylsilanes possessing different reactive groups: Physicochemical and tribological properties. ACS Appl. Mater. Interfaces 2016, 8, 7509–7521. [Google Scholar] [CrossRef] [PubMed]
- Kujawa, J.; Cerneaux, S.; Kujawski, W.; Bryjak, M.; Kujawski, J. How to functionalize ceramics by perfluoroalkylsilanes for membrane separation process? Properties and application of hydrophobized ceramic membranes. ACS Appl. Mater. Interfaces 2016, 8, 7564–7577. [Google Scholar] [CrossRef] [PubMed]
- Efome, J.E.; Baghbanzadeh, M.; Rana, D.; Matsuura, T.; Lan, C.Q. Effects of superhydrophobic SiO2 nanoparticles on the performance of PVDF flat sheet membranes for vacuum membrane distillation. Desalination 2015, 373, 47–57. [Google Scholar] [CrossRef]
- Hosseinabadi, S.R.; Wyns, K.; Buekenhoudt, A.; Van der Bruggen, B.; Ormerod, D. Performance of grignard functionalized ceramic nanofiltration membranes. Sep. Purif. Technol. 2015, 147, 320–328. [Google Scholar] [CrossRef]
- Rezaei Hosseinabadi, S.; Wyns, K.; Meynen, V.; Carleer, R.; Adriaensens, P.; Buekenhoudt, A.; Van der Bruggen, B. Organic solvent nanofiltration with grignard functionalised ceramic nanofiltration membranes. J. Membr. Sci. 2014, 454, 496–504. [Google Scholar] [CrossRef]
- Fane, A.G.; Schofield, R.W.; Fell, C.J.D. The efficient use of energy in membrane distillation. Desalination 1987, 64, 231–243. [Google Scholar] [CrossRef]
- Qtaishat, M.R.; Banat, F. Desalination by solar powered membrane distillation systems. Desalination 2013, 308, 186–197. [Google Scholar] [CrossRef]
- Nene, S.; Kaur, S.; Sumod, K.; Joshi, B.; Raghavarao, K.S.M.S. Membrane distillation for the concentration of raw cane-sugar syrup and membrane clarified sugarcane juice. Desalination 2002, 147, 157–160. [Google Scholar] [CrossRef]
- Sakai, K.; Koyano, T.; Muroi, T.; Tamura, M. Effects of temperature and concentration polarization on water vapour permeability for blood in membrane distillation. Chem. Eng. J. 1988, 38, B33–B39. [Google Scholar] [CrossRef]
- Sakai, K.; Muroi, T.; Ozawa, K.; Takesawa, S.; Tamura, M.; Nakane, T. Extraction of solute-free water from blood by membrane distillation. Am. Soc. Artif. Intern. Organ. Trans. 1986, 32, 397–400. [Google Scholar] [CrossRef]
- Kujawski, W.; Krajewska, S.; Kujawski, M.; Gazagnes, L.; Larbot, A.; Persin, M. Pervaporation properties of fluoroalkylsilane (FAS) grafted ceramic membranes. Desalination 2007, 205, 75–86. [Google Scholar] [CrossRef]
- Kujawa, J.; Cerneaux, S.; Kujawski, W. Removal of hazardous volatile organic compounds from water by vacuum pervaporation with hydrophobic ceramic membranes. J. Membr. Sci. 2015, 474, 11–19. [Google Scholar] [CrossRef]
- Kujawa, J.; Cerneaux, S.; Kujawski, W. Highly hydrophobic ceramic membranes applied to the removal of volatile organic compounds in pervaporation. Chem. Eng. J. 2015, 260, 43–54. [Google Scholar] [CrossRef]
- Amirilargani, M.; Sadrzadeh, M.; Sudholter, E.J.R.; de Smet, L.C.P.M. Surface modification methods of organic solvent nanofiltration membranes. Chem. Eng. J. 2016, 289, 562–582. [Google Scholar] [CrossRef]
- Owens, D.K.; Wendt, R.C. Estimation of the surface free energy of polymers. J. Appl. Polym. Sci. 1969, 13, 1741–1747. [Google Scholar] [CrossRef]
- Thomas, R.; Guillen-Burrieza, E.; Arafat, H.A. Pore structure control of PVDF membranes using a 2-stage coagulation bath phase inversion process for application in membrane distillation (MD). J. Membr. Sci. 2014, 452, 470–480. [Google Scholar] [CrossRef]
- Walsh, R. Bond dissociation energy values in silicon-containing compounds and some of their implications. Acc. Chem. Res. 1981, 14, 246–252. [Google Scholar] [CrossRef]
- Choi, W.; Tuteja, A.; Mabry, J.M.; Cohen, R.E.; McKinley, G.H. A modified Cassie–Baxter relationship to explain contact angle hysteresis and anisotropy on non-wetting textured surfaces. J. Colloid Interfaces Sci. 2009, 339, 208–216. [Google Scholar] [CrossRef] [PubMed]
- Woldemariam, D.; Martin, A.; Santarelli, M. Exergy analysis of air-gap membrane distillation systems for water purification applications. Appl. Sci. 2017, 7, 301. [Google Scholar] [CrossRef]
- Ko, C.-C.; Chen, C.-H.; Chen, Y.-R.; Wu, Y.-H.; Lu, S.-C.; Hu, F.-C.; Li, C.-L.; Tung, K.-L. Increasing the performance of vacuum membrane distillation using micro-structured hydrophobic aluminum hollow fiber membranes. Appl. Sci. 2017, 7, 357. [Google Scholar] [CrossRef]
- Kujawa, J.; Kujawski, W. Driving force and activation energy in air-gap membrane distillation process. Chem. Pap. 2015, 69, 1438–1444. [Google Scholar] [CrossRef]
| Parameter | Pristine | Ti-C8OEt3 | Ti-C8Cl3 | Ti-C18Cl3 | |||
|---|---|---|---|---|---|---|---|
| 1.5 h | 4.5 h | 1.5h | 4.5 h | 1.5 h | 4.5 h | ||
| Contact angle (CA), deg | 40 ± 2 | 111 ± 2 | 113 ± 2 | 120 ± 2 | 128 ± 2 | 126 ± 2 | 132 ± 2 |
| SFE, mN·m−1 | 140 ± 5 | 62.5 ± 2.7 | 53.6 ± 2.3 | 43.7 ± 1.9 | 32.7 ± 1.4 | 38.2 ± 1.7 | 31.1 ± 1.4 |
| Polar part of SFE (γp), mN·m−1 | 44.8 ± 1.4 | 13.3 ± 0.6 | 11.4 ± 0.5 | 9.3 ± 0.4 | 8.6 ± 0.4 | 5.7 ± 0.3 | 5.5 ± 0.2 |
| Dispersive part of SFE (γd), mN·m−1 | 95.2 ± 3.1 | 49.2 ± 2.2 | 42.2 ± 1.8 | 34.4 ± 1.5 | 24.1 ± 1.1 | 32.5 ± 1.4 | 25.6 ± 1.1 |
| RMS, nm | 60.5 ± 5.1 | 50.9 ± 2.0 | 45.2 ± 1.8 | 39.6 ± 1.6 | 33.2 ± 1.3 | 27.8 ± 1.1 | 22.9 ± 0.9 |
| Membrane | 1.5 h Modification | 4.5 h Modification |
|---|---|---|
| Ti-C8Cl3 | 98.0–98.3 | 99.5–99.6 |
| Ti-C8OEt3 | 98.3–98.5 | 99.6–99.7 |
| Ti-C18Cl3 | 98.0–98.4 | 99.2–99.6 |
| Al-C8Cl3 | 98.1–98.3 | 99.0–99.5 |
| Al-C8OEt3 | 98.0–98.3 | 99.0–99.3 |
| Al-C18Cl3 | 98.0–98.2 | 99.0–99.3 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kujawa, J.; Cerneaux, S.; Kujawski, W.; Knozowska, K. Hydrophobic Ceramic Membranes for Water Desalination. Appl. Sci. 2017, 7, 402. https://doi.org/10.3390/app7040402
Kujawa J, Cerneaux S, Kujawski W, Knozowska K. Hydrophobic Ceramic Membranes for Water Desalination. Applied Sciences. 2017; 7(4):402. https://doi.org/10.3390/app7040402
Chicago/Turabian StyleKujawa, Joanna, Sophie Cerneaux, Wojciech Kujawski, and Katarzyna Knozowska. 2017. "Hydrophobic Ceramic Membranes for Water Desalination" Applied Sciences 7, no. 4: 402. https://doi.org/10.3390/app7040402
APA StyleKujawa, J., Cerneaux, S., Kujawski, W., & Knozowska, K. (2017). Hydrophobic Ceramic Membranes for Water Desalination. Applied Sciences, 7(4), 402. https://doi.org/10.3390/app7040402








