The Chemical Behaviors of Nitrogen Dioxide Absorption in Sulfite Solution
Abstract
:1. Introduction
2. Experimental Section
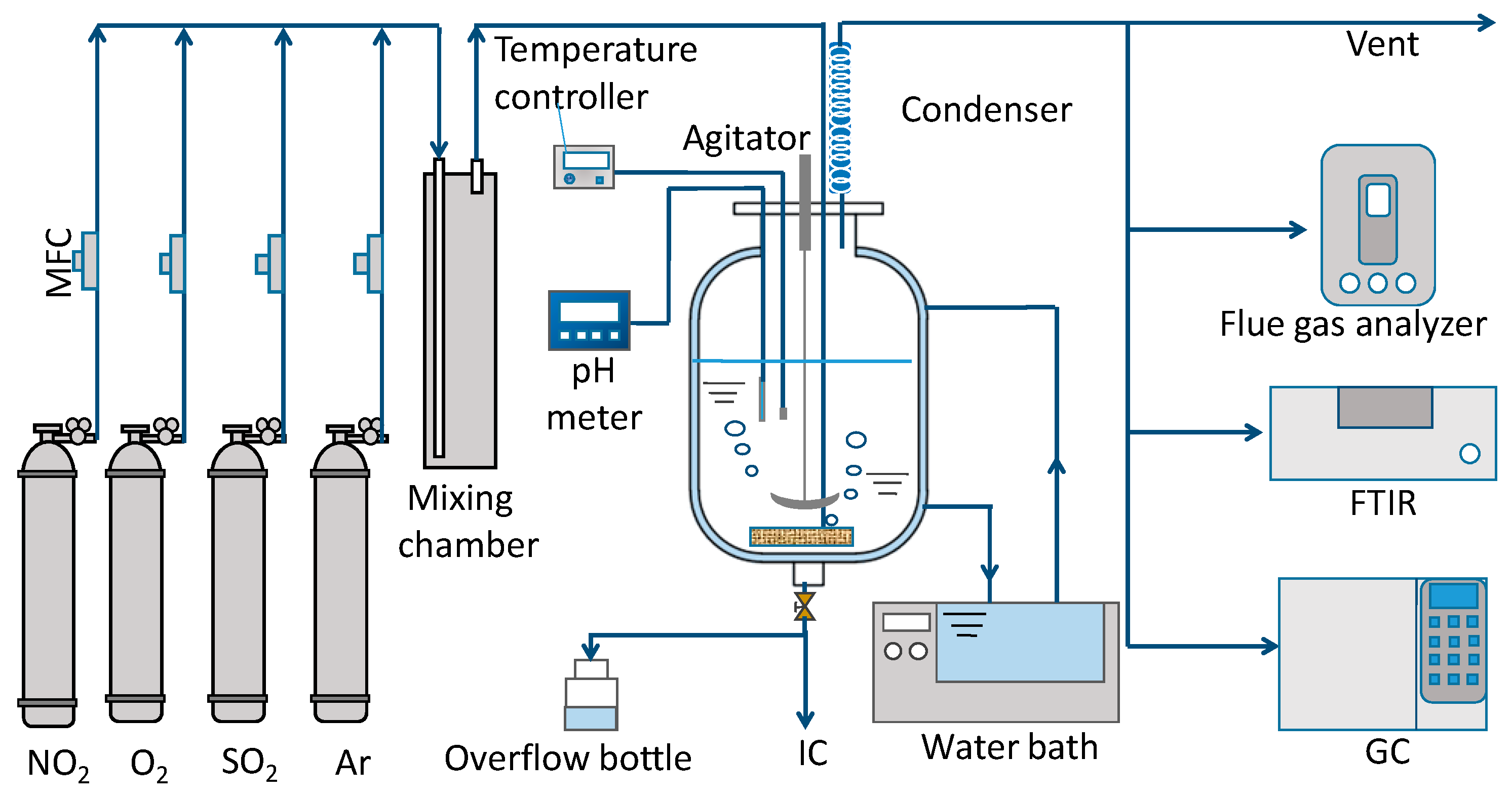
2.1. Experimental Setup
2.2. Experimental Methods
3. Results and Discussion
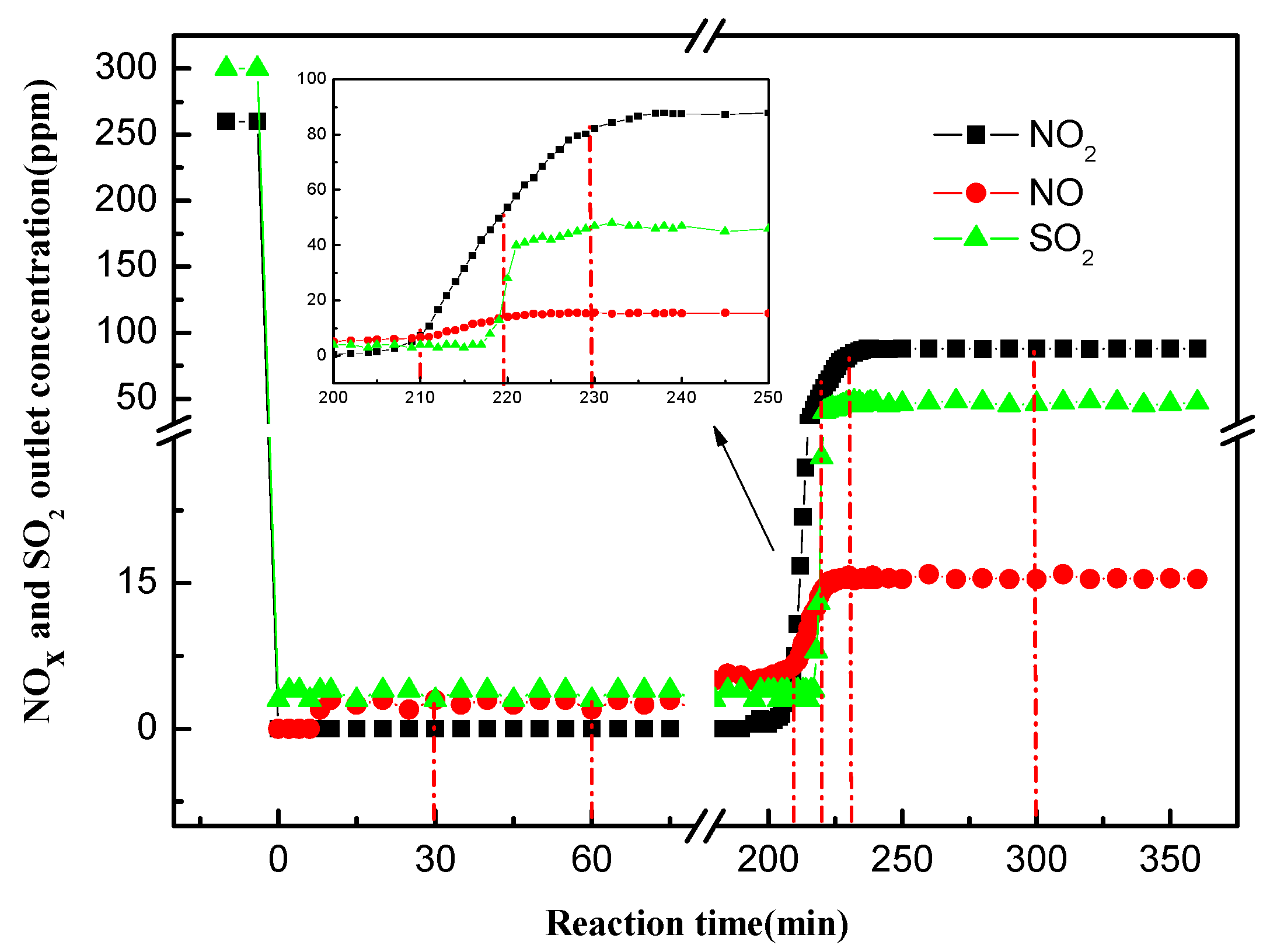
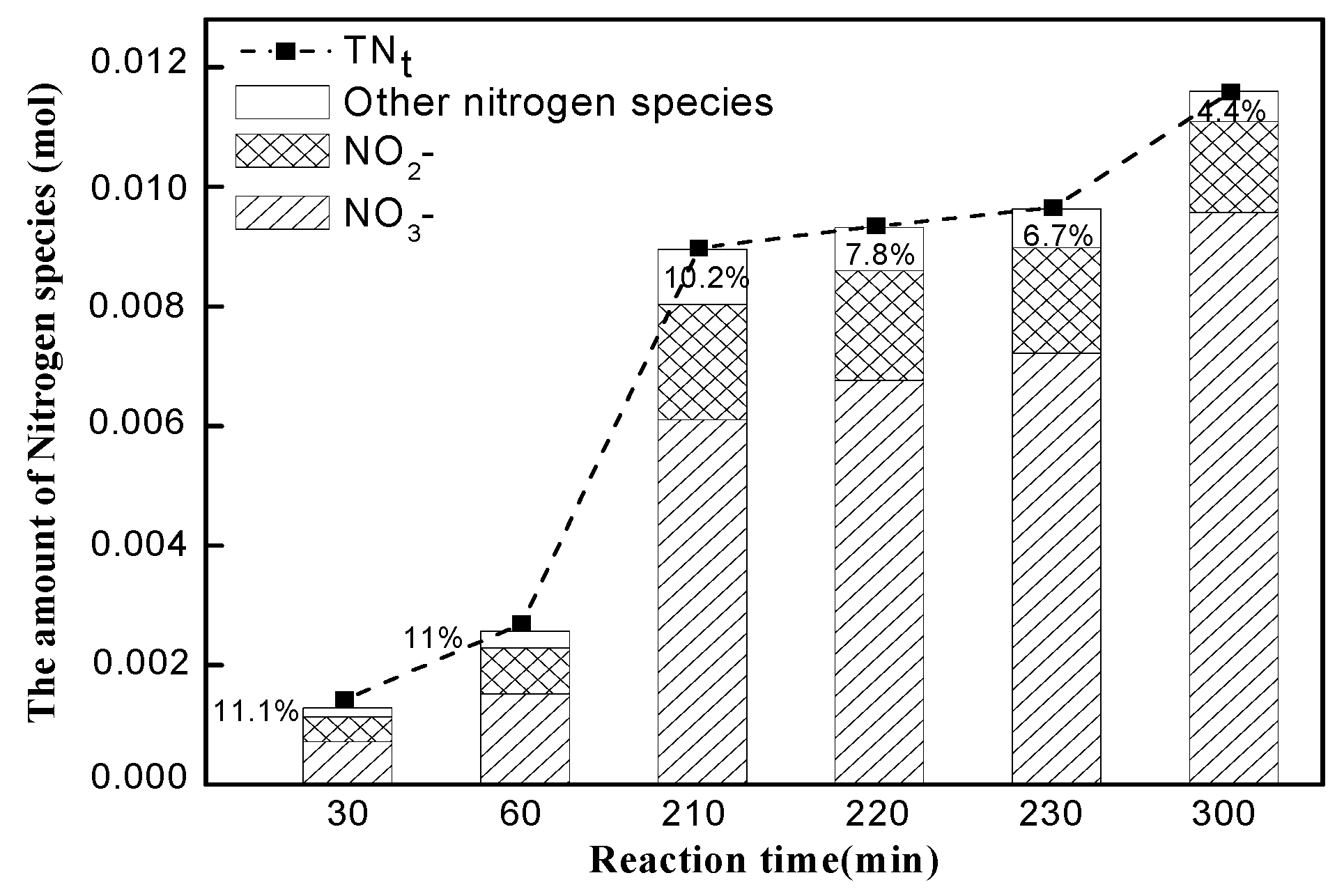
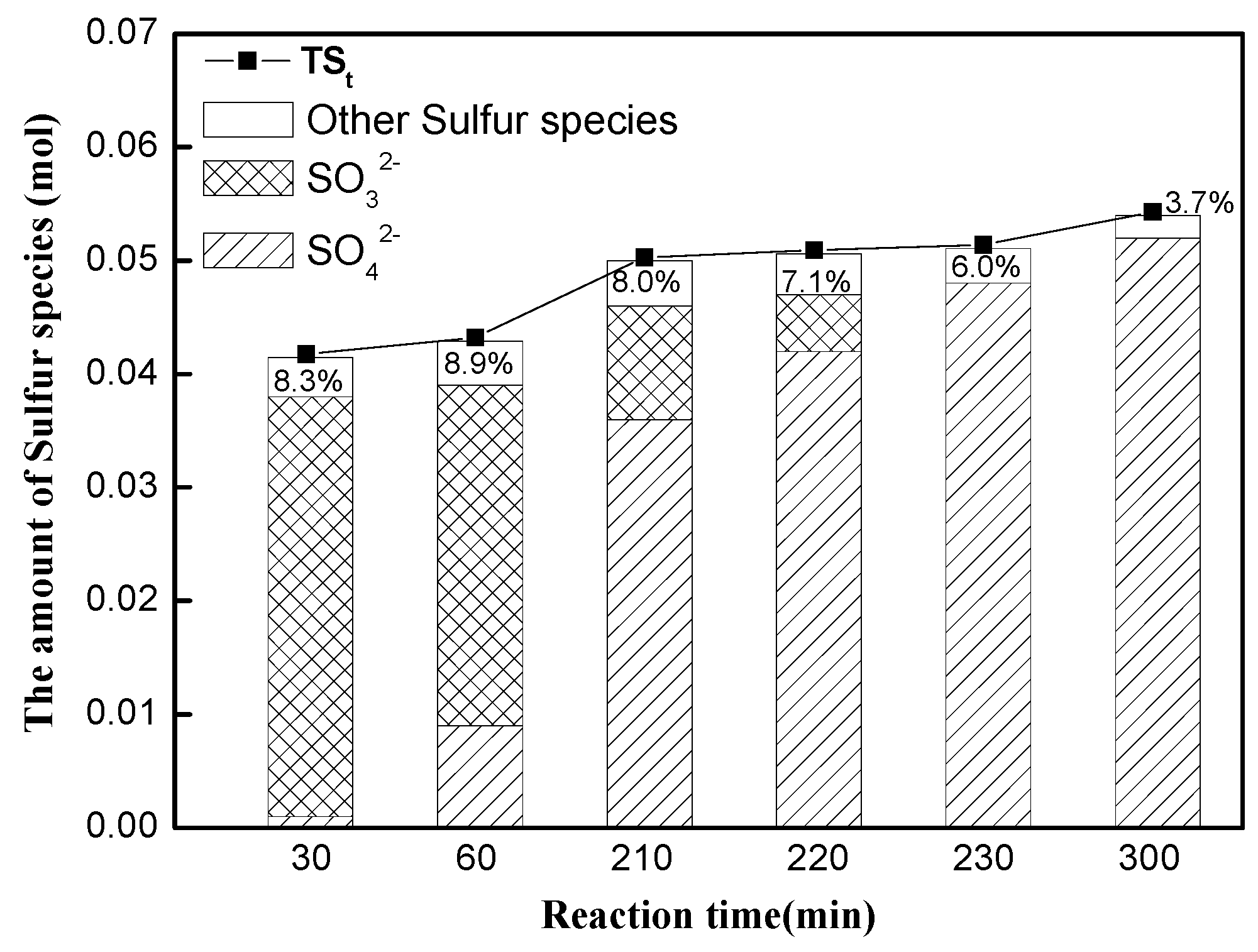
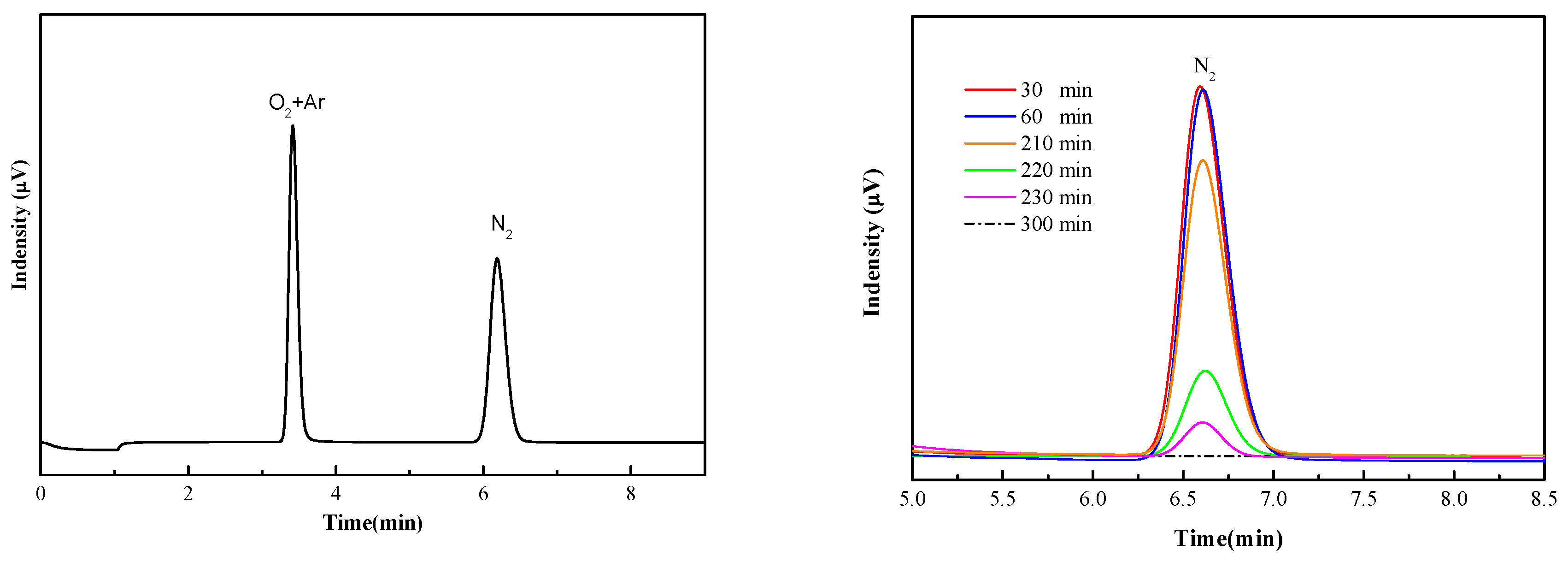
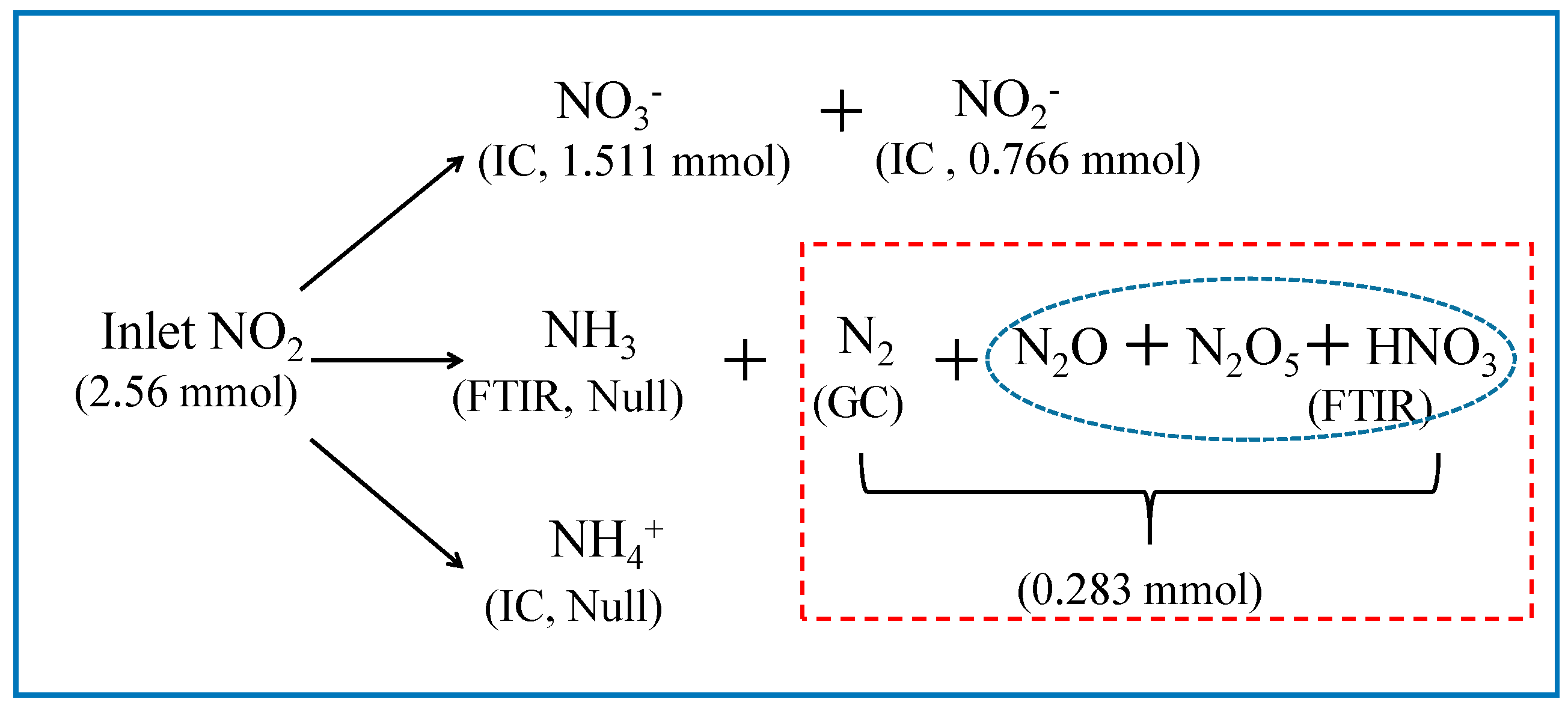
3.1. Quantitative Analysis of N and S Compositions in Gaseous and Liquid Phases
3.2. Equilibrium Analysis of Nand S Compositions
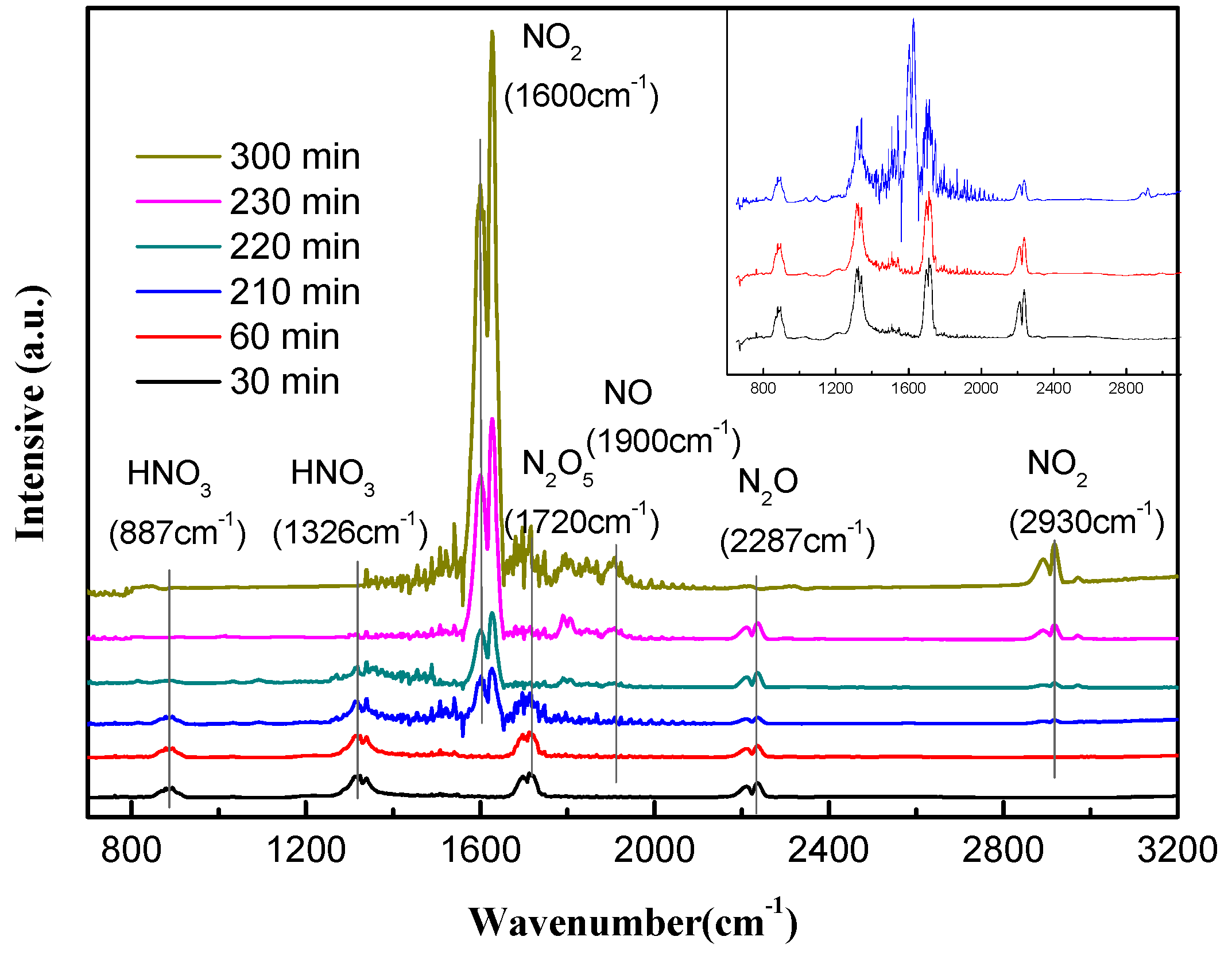
3.3. Qualitative Analysis of N Compositions
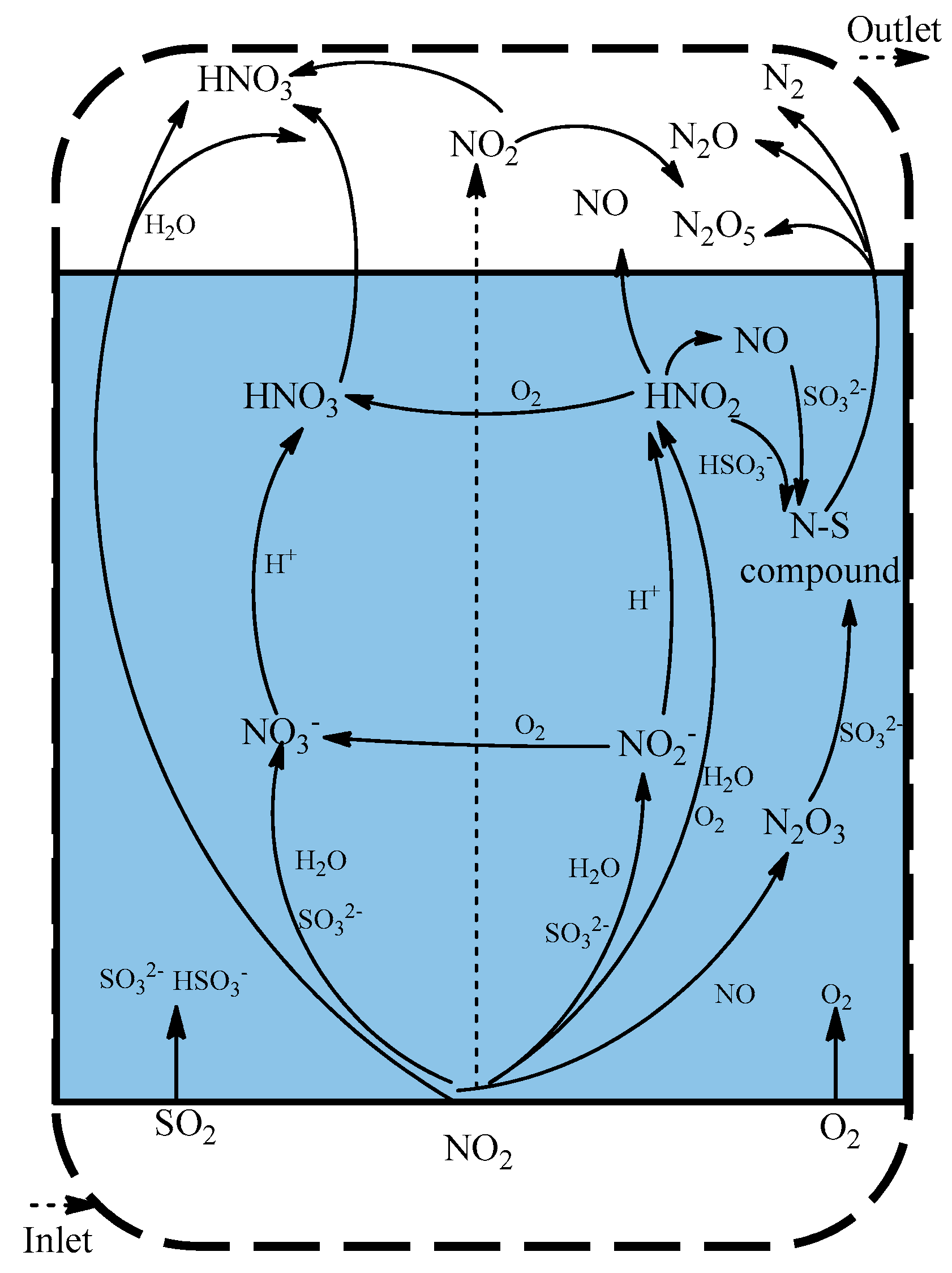
3.4. NO2 Absorption Pathway
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Adewuyi, Y.G.; Sakyi, N.Y. Simultaneous Absorption and Oxidation of Nitric Oxide and Sulfur Dioxide by Aqueous Solutions of Sodium Persulfate Activated by Temperature. Ind. Eng. Chem. Res. 2013, 52, 11702–11711. [Google Scholar] [CrossRef]
- Guo, R.T.; Hao, J.K.; Pan, W.G.; Yu, Y.L. Liquid Phase Oxidation and Absorption of NO from Flue Gas: A Review. Sep. Sci. Technol. 2015, 50, 310–321. [Google Scholar] [CrossRef]
- Sun, Y.X.; Zwolinska, E.; Chmielewski, A.G. Abatement Technologies for High Concentrations of NOx and SO2 Removal from Exhaust Gases: A Review. Crit. Rev. Environ. Sci. Technol. 2016, 46, 119–142. [Google Scholar] [CrossRef]
- Zhao, Y.; Hao, R.L.; Qi, M. Integrative Process of Preoxidation and Absorption for Simultaneous Removal of SO2, NO and Hg0. Chem. Eng. J. 2015, 269, 159–167. [Google Scholar] [CrossRef]
- Deshwal, B.R.; Lee, S.H.; Jung, J.H.; Shon, B.H.; Lee, H.K. Study on the Removal of NOx from Simulated Flue Gas Using Acidic NaClO2 Solution. J. Environ. Sci. China 2008, 20, 33–38. [Google Scholar] [CrossRef]
- Jin, D.-S.; Deshwal, B.-R.; Park, Y.-S.; Lee, H.-K. Simultaneous Removal of SO2 and NO by Wet Scrubbing Using Aqueous Chlorine Dioxide Solution. J. Hazard. Mater. 2006, 135, 412–417. [Google Scholar] [CrossRef] [PubMed]
- Fang, P.; Cen, C.-P.; Wang, X.-M.; Tang, Z.-J.; Tang, Z.-X.; Chen, D.-S. Simultaneous Removal of SO2, NO and Hg0 by Wet Scrubbing Using Urea + KMnO4 Solution. Fuel Process. Technol. 2013, 106, 645–653. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, J.; Sheng, C.; Zhang, Y.; Zhao, L. Simultaneous Removal of NO and SO2 from Coal-fired Flue Gas by UV/H2O2 Advanced Oxidation Process. Chem. Eng. J. 2010, 162, 1006–1011. [Google Scholar] [CrossRef]
- Yoon, H.J.; Park, H.W.; Park, D.W. Simultaneous Oxidation and Absorption of NOx and SO2 in an Integrated O3 Oxidation/Wet Atomizing System. Energy Fuel 2016, 30, 3289–3297. [Google Scholar] [CrossRef]
- Wang, M.; Sun, Y.; Zhu, T. Removal of NOx, SO2, and Hg From Simulated Flue Gas by Plasma-Absorption Hybrid System. IEEE Trans. Plasma Sci. 2013, 41, 312–318. [Google Scholar] [CrossRef]
- Van Eynde, E.; Lenaerts, B.; Tytgat, T.; Blust, R.; Lenaerts, S. Valorization of Flue Gas by Combining Photocatalytic Gas Pretreatment with Microalgae Production. Environ. Sci. Technol. 2016, 50, 2538–2545. [Google Scholar] [CrossRef] [PubMed]
- Adewuyi, Y.G.; Khan, M.A. Nitric Oxide Removal by Combined Persulfate and Ferrous-Edta Reaction Systems. Chem. Eng. J. 2015, 281, 575–587. [Google Scholar] [CrossRef]
- Krzyzynska, R.; Hutson, N.D. Effect of Solution pH on SO2, NOx, and Hg Removal from Simulated Coal Combustion Flue Gas in an Oxidant-enhanced Wet Scrubber. J. Air Waste Manag. Assoc. 2012, 62, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Komiyama, H.; Inoue, H. Absorption of Nitrogen Oxides into Water. Chem. Eng. Sci. 1980, 35, 154–161. [Google Scholar] [CrossRef]
- Thomas, D.; Vanderschuren, J. Analysis and Prediction of the Liquid Phase Composition for the Absorption of Nitrogen Oxides into Aqueous Solutions. Sep. Purif. Technol. 1999, 18, 37–45. [Google Scholar] [CrossRef]
- Hüpen, B.; Kenig, E.Y. Rigorous Modelling of Absorption in Tray and Packed Columns. Chem. Eng. Sci. 2005, 60, 6462–6471. [Google Scholar] [CrossRef]
- Hu, G.; Sun, Z.; Gao, H. Novel Process of Simultaneous Removal of SO2 and NO2 by Sodium Humate Solution. Environ. Sci. Technol. 2010, 44, 6712–6717. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.-Y.; Wang, Q.-Y.; Ding, S.-L.; Su, S.-J.; Jiang, W.-J.; Zhu, E.-G. Reaction Mechanism of NOx Removal from Flue Gas with Pyrolusite Slurry. Sep. Purif. Technol. 2013, 118, 576–582. [Google Scholar]
- Guo, Q.; Sun, T.; Wang, Y.; He, Y.; Jia, J. Spray Absorption and Electrochemical Reduction of Nitrogen Oxides from Flue Gas. Environ. Sci. Technol. 2013, 47, 9514–9522. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Guo, R.-T.; Ding, H.-L.; Luo, Z.-Y.; Cen, K.-F. Absorption of NO2 into Na2S Solution in a Stirred Tank Reactor. J. Zhejiang Univ. Sci. A 2009, 10, 434–438. [Google Scholar] [CrossRef]
- Guo, Q.; He, Y.; Sun, T.; Wang, Y.; Jia, J. Simultaneous Removal of NOx and SO2 from Flue Gas Using Combined Na2SO3 Assisted Electrochemical Reduction and Direct Electrochemical Reduction. J. Hazard. Mater. 2014, 276, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Du, Z.; Ding, H.-L.; Wu, Z.-L.; Lu, H.; Luo, Z.-Y.; Cen, K.-F. Effect of Gas-liquid Phase Compositions on NO2 and NO Absorption into Ammonium-sulfite and Bisulfite Solutions. Fuel Process. Technol. 2011, 92, 1506–1512. [Google Scholar] [CrossRef]
- Roy, S.; Rochelle, G. Simultaneous Absorption of Chlorine and Mercury in Sulfite Solutions. Chem. Eng. Sci. 2004, 59, 1309–1323. [Google Scholar] [CrossRef]
- Rochelle, G.T. Chlorine Absorption in Sulfite Solutions. Sep. Sci. Technol. 2005, 39, 3057–3077. [Google Scholar]
- Shibukawa, T.; Ohira, Y.; Obata, E. Absorption of Nitrogen Dioxide by Sodium Sulfite Solution. Kagaku Kogaku Ronbun 2008, 34, 438–443. [Google Scholar] [CrossRef]
- Gao, X.; Du, Z.; Ding, H.-L.; Wu, Z.-L.; Lu, H.; Luo, Z.-Y.; Cen, K.-F. Kinetics of NOx Absorption into (NH4)2SO3 Solution in an Ammonia-Based Wet Flue Gas Desulfurization Process. Energy Fuel 2010, 24, 5876–5882. [Google Scholar] [CrossRef]
- Zheng, C.; Xu, C.; Zhang, Y.; Zhang, J.; Gao, X.; Luo, Z.; Cen, K. Nitrogen Oxide Absorption and Nitrite/nitrate Formation in Limestone Slurry for WFGD System. Appl. Energy 2014, 129, 187–194. [Google Scholar] [CrossRef]
- Shen, C.H.; Rochelle, G.T. Nitrogen Dioxide Absorption and Sulfite Oxidation in Aqueous Sulfite. Environ. Sci. Technol. 1998, 32, 1994–2003. [Google Scholar] [CrossRef]
- Tang, N.; Liu, Y.; Wang, H.; Xiao, L.; Wu, Z. Enhanced Absorption Process of NO2 in CaSO3 Slurry by the Addition of MgSO4. Chem. Eng. J. 2010, 160, 145–149. [Google Scholar] [CrossRef]
- Siddiqi, M.A.; Petersen, J.; Lucas, K. A Study of the Effect of Nitrogen Dioxide on the Absorption of Sulfur Dioxide in Wet Flue Gas Cleaning Processes. Ind. Eng. Chem. Res. 2001, 40, 2116–2127. [Google Scholar] [CrossRef]
- Yan, B.; Yang, J.H.; Guo, M.; Zhu, S.J.; Yu, W.J.; Ma, S.C. Experimental Study on Fe(II)Cit Enhanced Absorption of NO in (NH4)2SO3 Solution. J. Ind. Eng. Chem. 2015, 21, 476–482. [Google Scholar] [CrossRef]
- Chen, L.; Lin, J.W.; Yang, C.L. Absorption of NO2 in a Packed Tower with Na2SO3 Aqueous Solution. Environ. Prog. 2002, 21, 225–230. [Google Scholar] [CrossRef]
- Zhuang, Z.K.; Sun, C.L.; Zhao, N.; Wang, H.Q.; Wu, Z.B. Numerical Simulation of NO2 Absorption Using Sodium Sulfite in a Spray Tower. J. Chem. Technol. Boitechnol. 2016, 91, 994–1003. [Google Scholar] [CrossRef]
- Sun, C.; Zhao, N.; Zhuang, Z.; Wang, H.; Liu, Y.; Weng, X.; Wu, Z. Mechanisms and Reaction Pathways for Simultaneous Oxidation of NOx and SO2 by Ozone Determined by in situ IR Measurements. J. Hazard. Mater. 2014, 274, 376–383. [Google Scholar] [CrossRef] [PubMed]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, Y.; Hong, X.; Zhu, T.; Guo, X.; Xie, D. The Chemical Behaviors of Nitrogen Dioxide Absorption in Sulfite Solution. Appl. Sci. 2017, 7, 377. https://doi.org/10.3390/app7040377
Sun Y, Hong X, Zhu T, Guo X, Xie D. The Chemical Behaviors of Nitrogen Dioxide Absorption in Sulfite Solution. Applied Sciences. 2017; 7(4):377. https://doi.org/10.3390/app7040377
Chicago/Turabian StyleSun, Ye, Xiaowei Hong, Tianle Zhu, Xiaoyan Guo, and Deyuan Xie. 2017. "The Chemical Behaviors of Nitrogen Dioxide Absorption in Sulfite Solution" Applied Sciences 7, no. 4: 377. https://doi.org/10.3390/app7040377
APA StyleSun, Y., Hong, X., Zhu, T., Guo, X., & Xie, D. (2017). The Chemical Behaviors of Nitrogen Dioxide Absorption in Sulfite Solution. Applied Sciences, 7(4), 377. https://doi.org/10.3390/app7040377



