Characterization of Reduced Graphene Oxide (rGO)-Loaded SnO2 Nanocomposite and Applications in C2H2 Gas Detection
Abstract
:1. Introduction
2. Experimental Method
2.1. Materials
2.1.1. SnO2 Nanoparticle Synthesis
2.1.2. SnO2–rGO Nanocomposite Preparation
2.1.3. Characterization
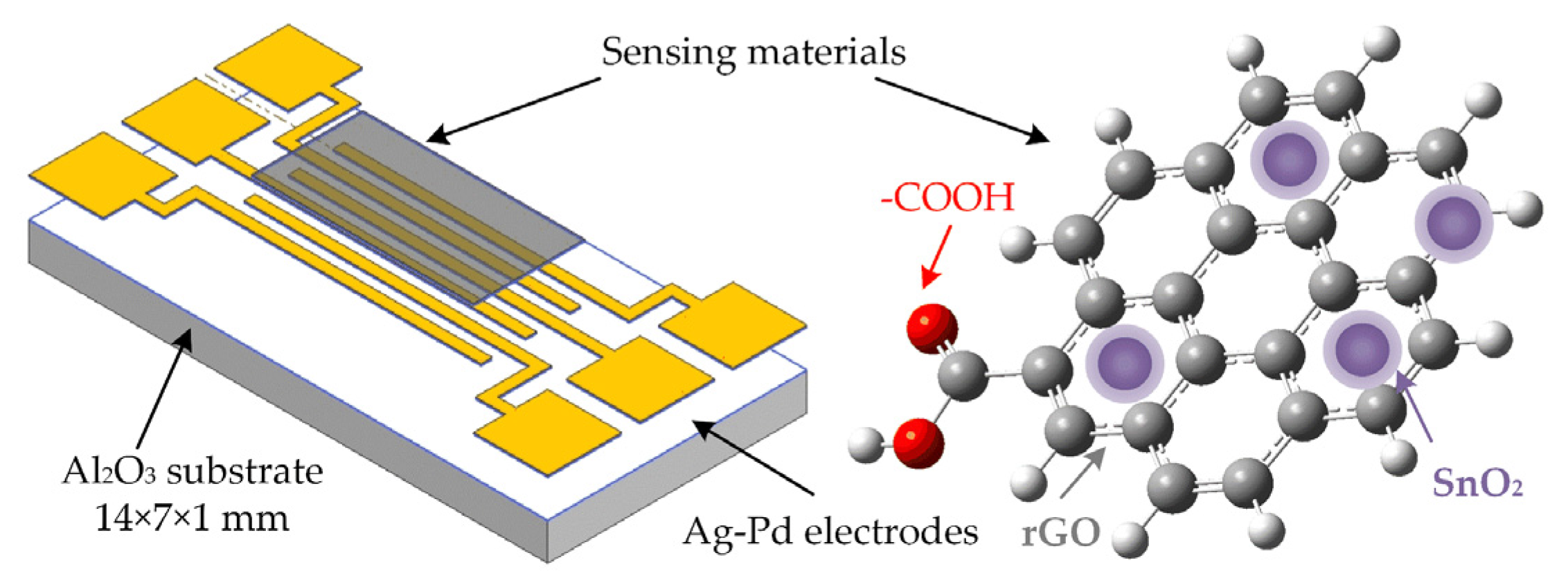
2.2. Fabrication of Planar Gas Sensor
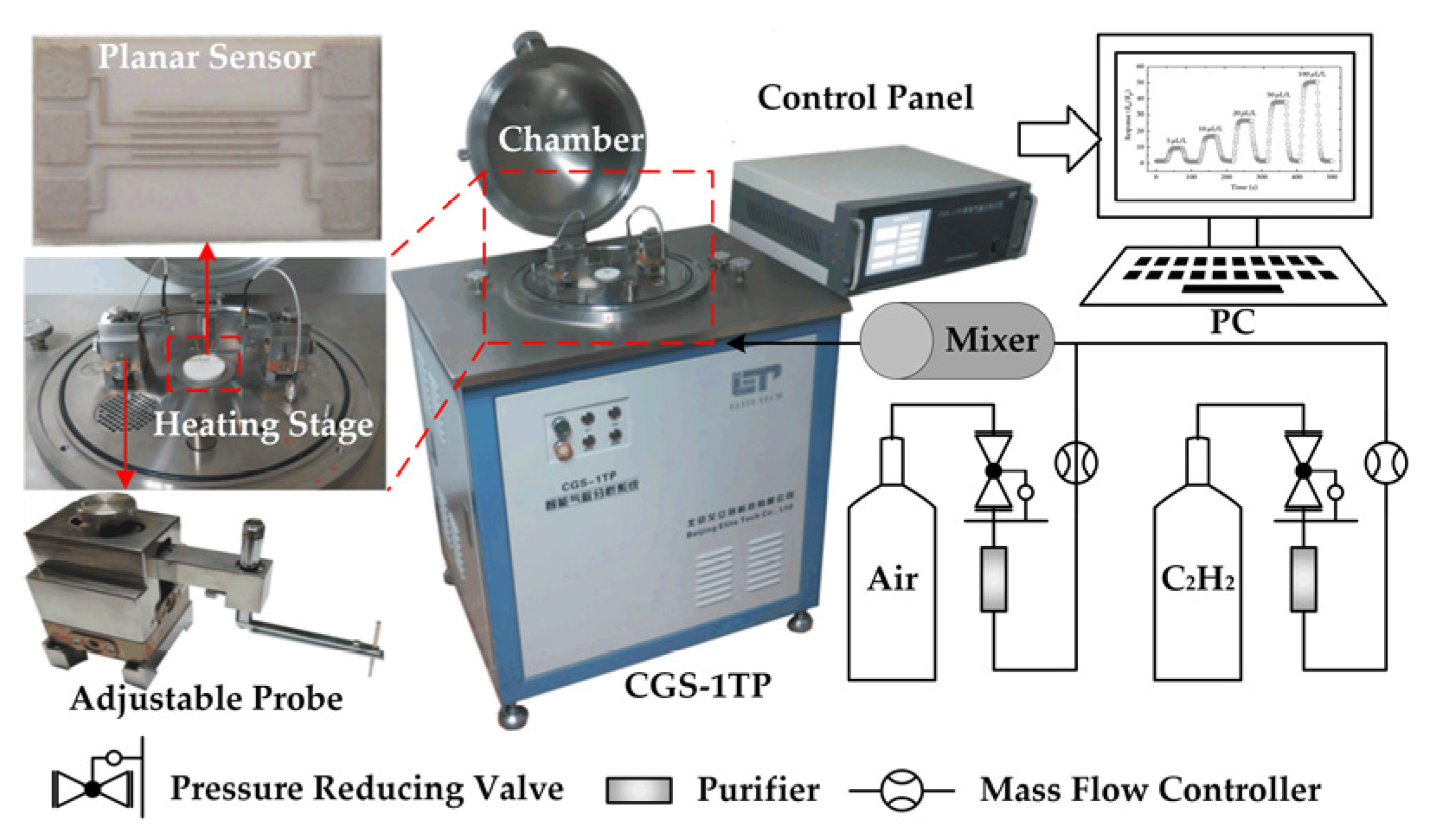
2.3. Gas Sensing Measurements
3. Results and Discussion
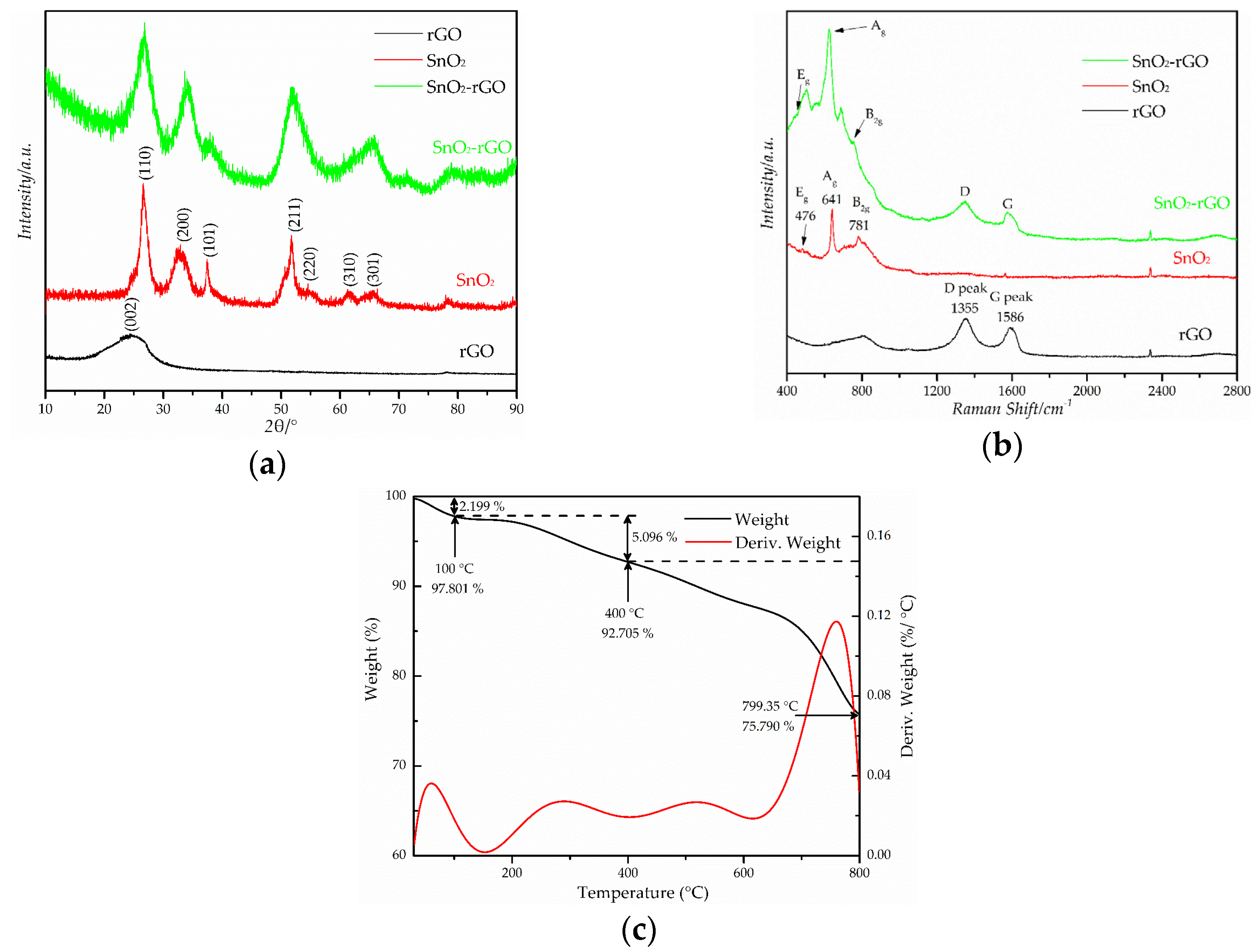
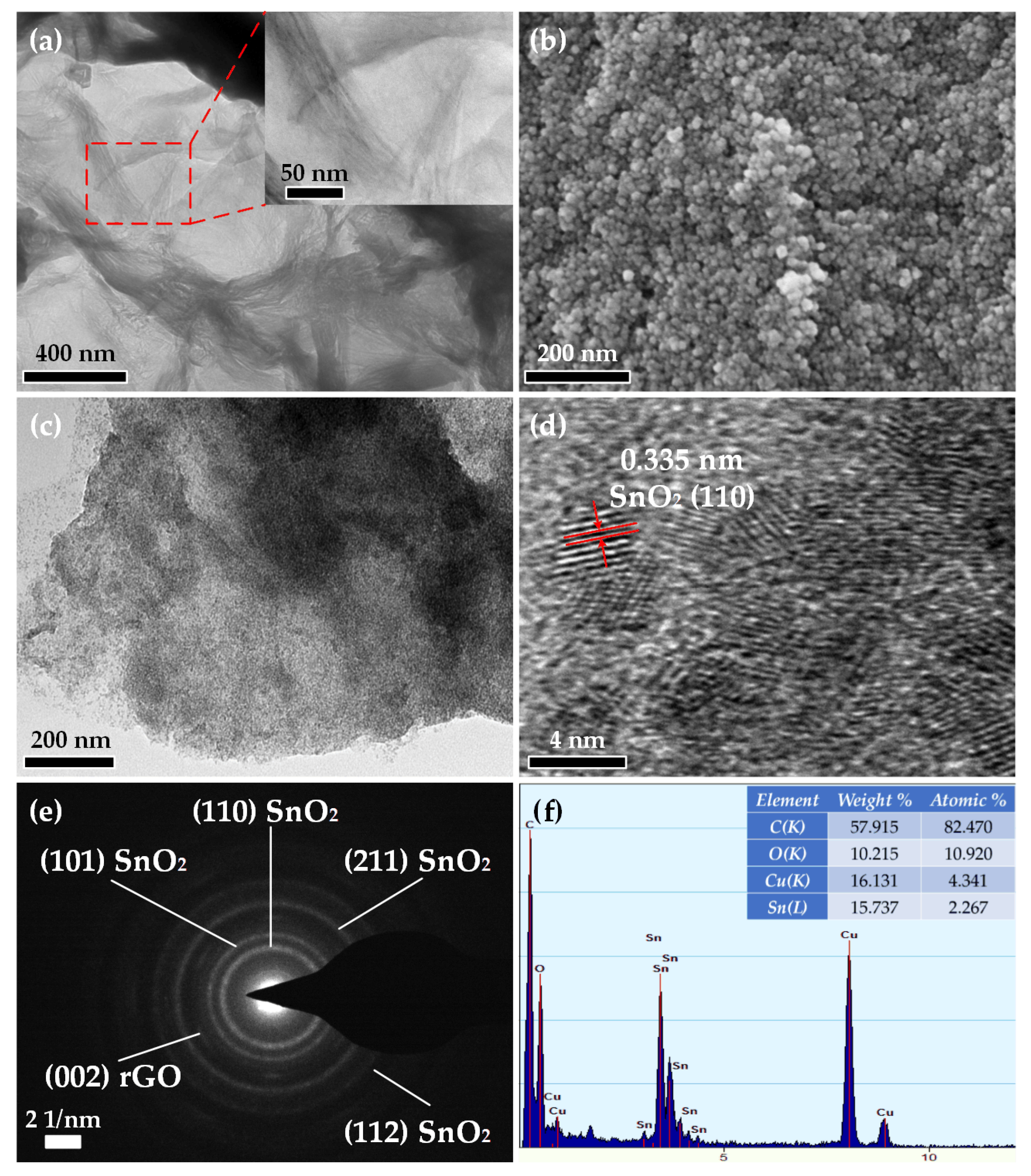
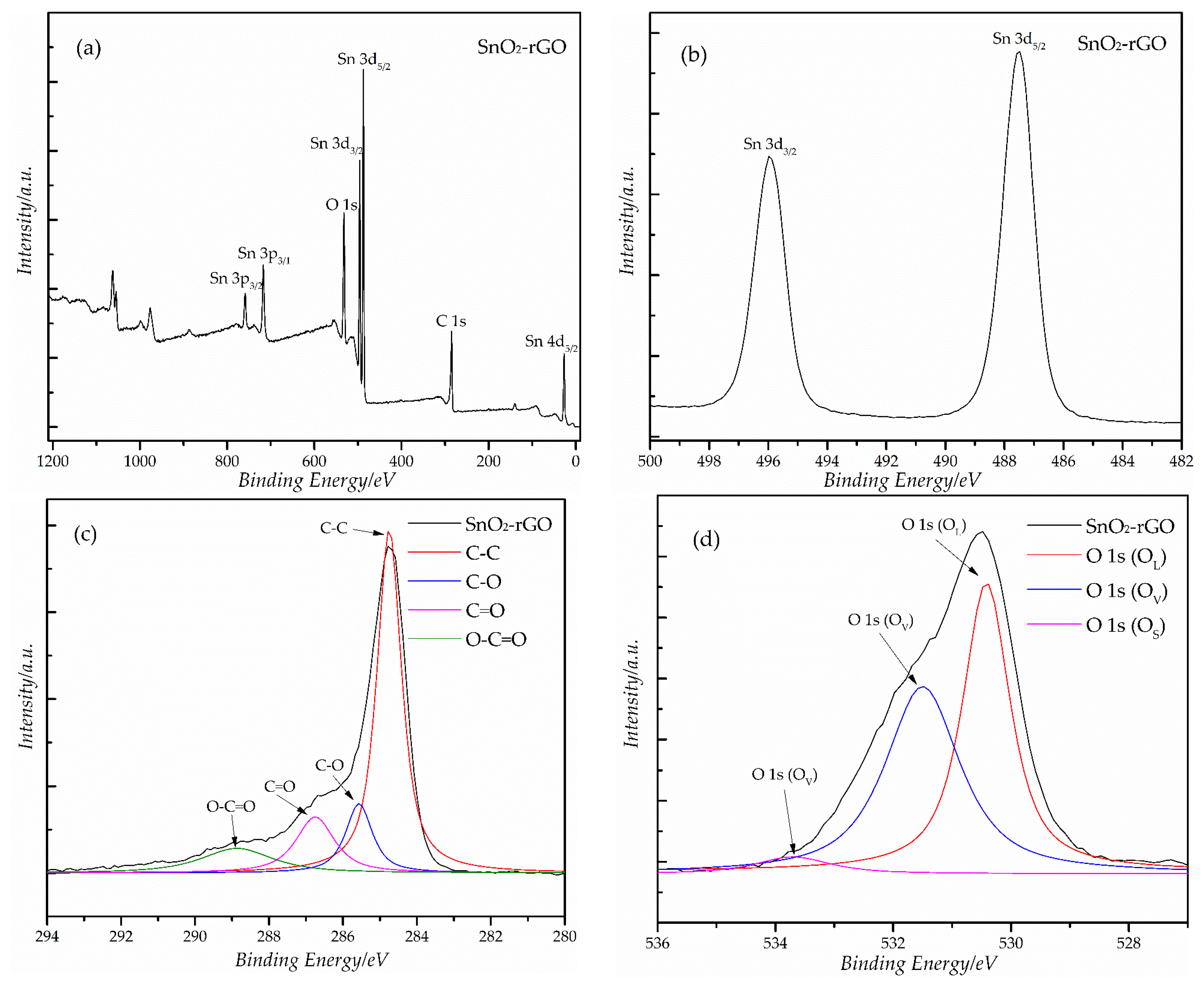
3.1. Structural Characterization
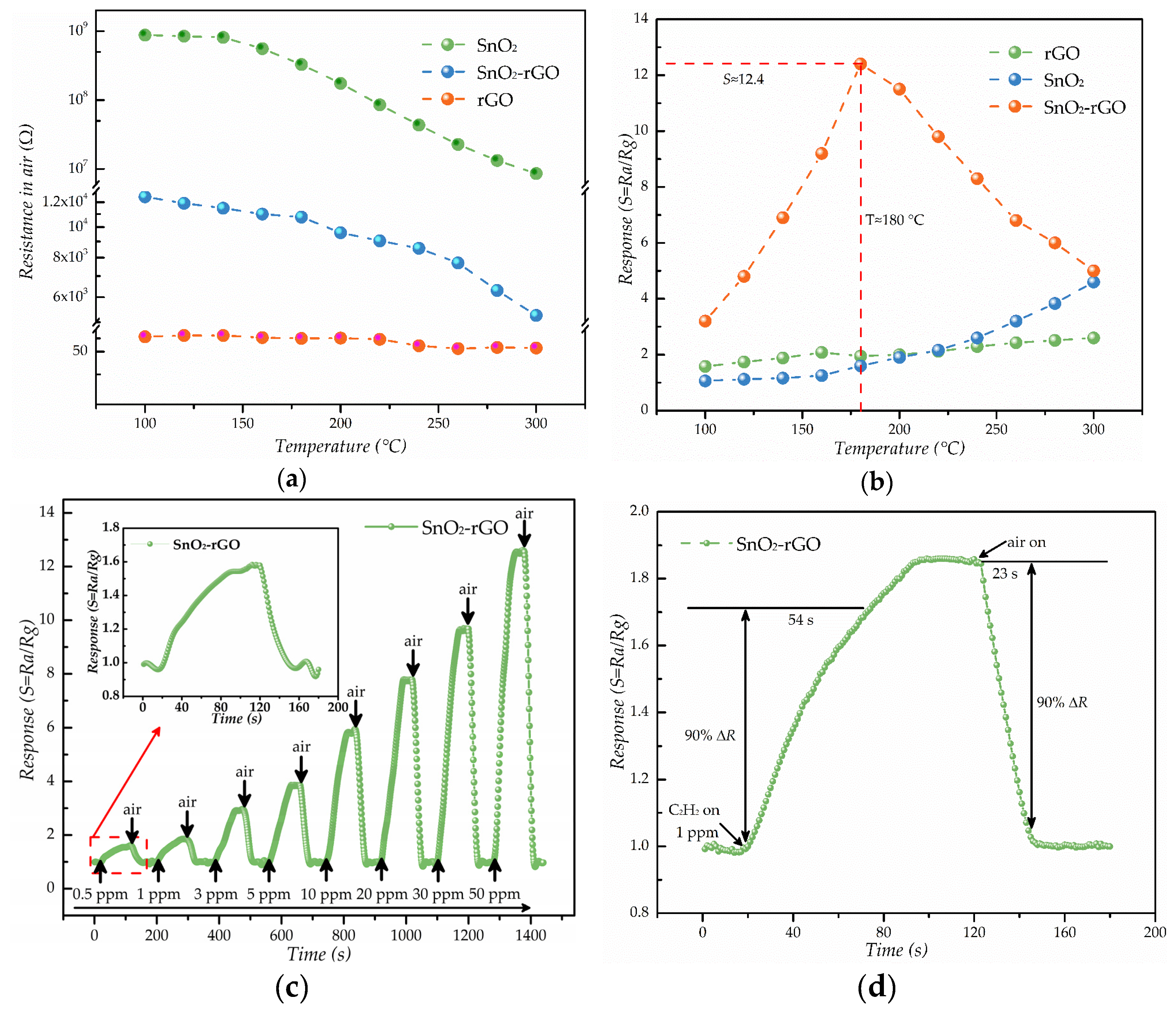
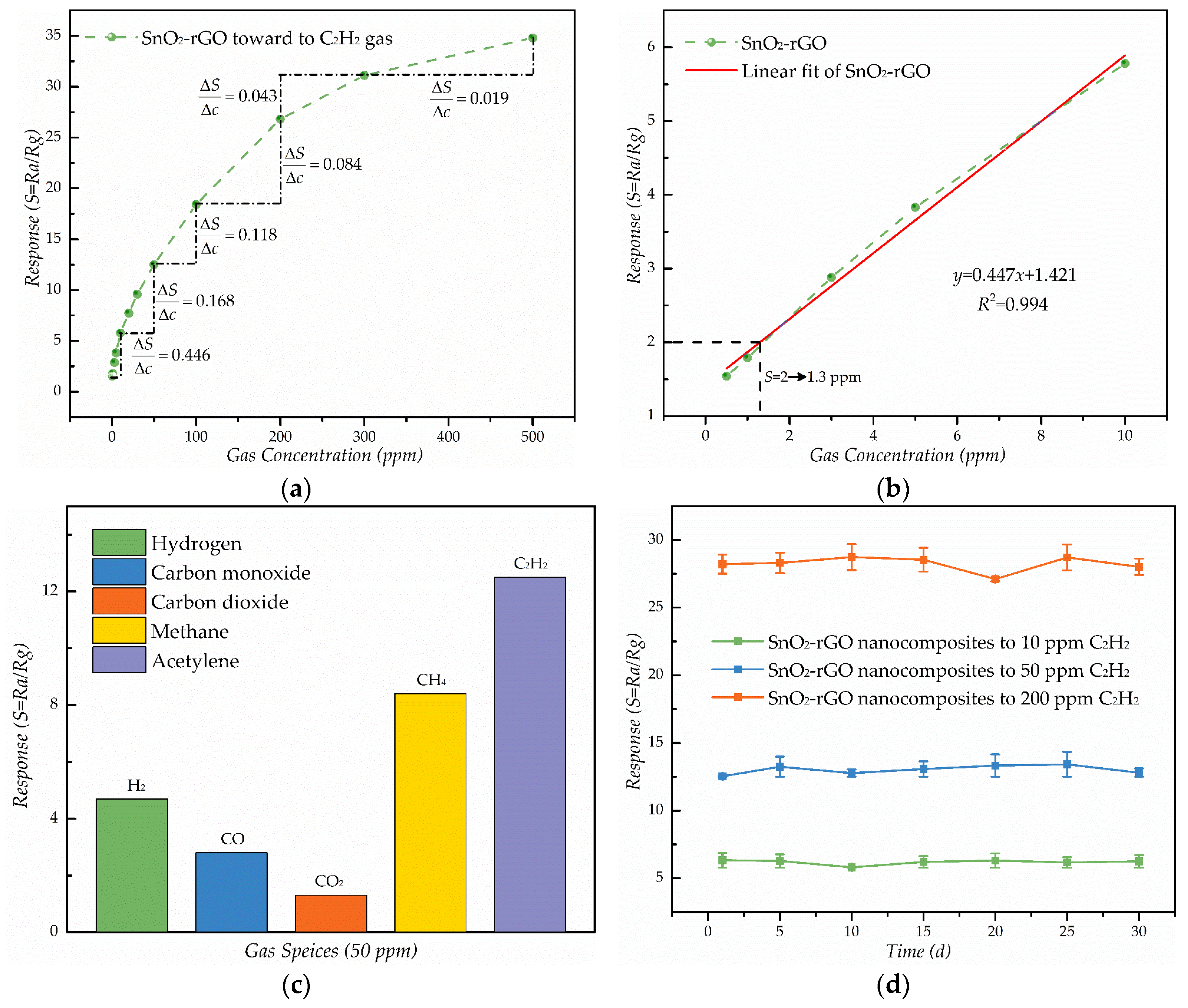
3.2. C2H2 Gas Sensing Properties
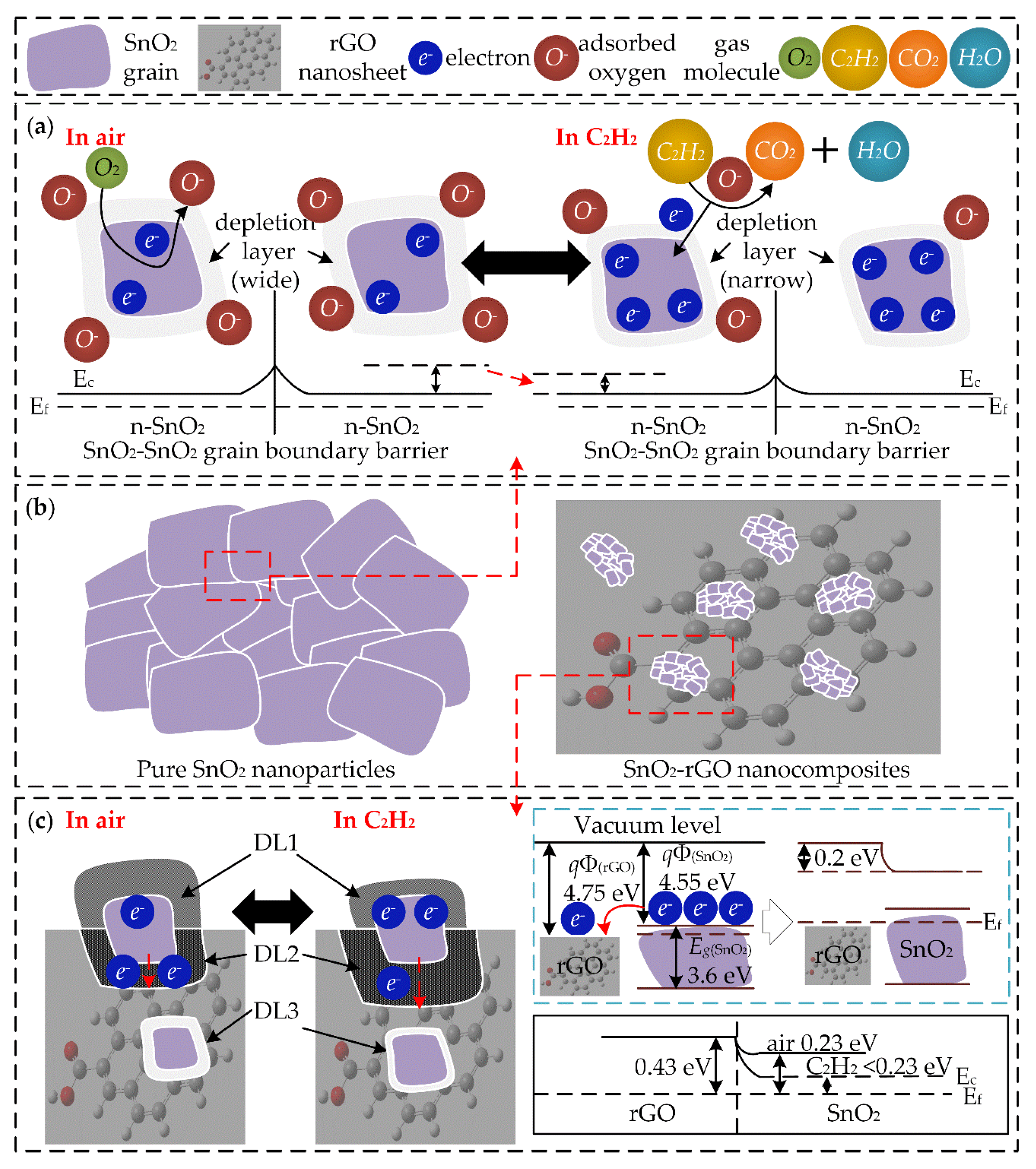
3.3. Gas Sensing Mechanism
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Saha, T.K. Review of modern diagnostic techniques for assessing insulation condition in aged transformers. IEEE Trans. Dielectr. Electr. Insul. 2003, 10, 903–917. [Google Scholar] [CrossRef]
- Arshad, M.; Islam, S.M. Significance of cellulose power transformer condition assessment. IEEE Trans. Dielectr. Electr. Insul. 2011, 18, 1591–1598. [Google Scholar] [CrossRef]
- Duval, M. A review of faults detectable by gas-in-oil analysis in transformers. IEEE Electr. Insul. Mag. 2002, 18, 8–17. [Google Scholar] [CrossRef]
- Wang, M.; Vandermaar, A.J.; Srivastava, K.D. Review of condition assessment of power transformers in service. IEEE Electr. Insul. Mag. 2002, 18, 12–25. [Google Scholar] [CrossRef]
- Bengtsson, C. Status and trends in transformer monitoring. IEEE Trans. Power Deliv. 1996, 11, 1379–1384. [Google Scholar] [CrossRef]
- Mor, G.K.; Carvalho, M.A.; Varghese, O.K.; Pishko, M.V.; Grimes, C.A. A room-temperature TiO2-nanotube hydrogen sensor able to self-clean photoactively from environmental contamination. J. Mater. Res. 2004, 19, 628–634. [Google Scholar] [CrossRef]
- Wang, L.W.; Kang, Y.F.; Liu, X.H.; Zhang, S.M.; Huang, W.P.; Wang, S.R. ZnO nanorod gas sensor for ethanol detection. Sens. Actuators B-Chem. 2012, 162, 237–243. [Google Scholar] [CrossRef]
- Batzill, M.; Diebold, U. The surface and materials science of tin oxide. Progress Surf. Sci. 2005, 79, 47–154. [Google Scholar] [CrossRef]
- Myung, N.V.; Mubeen, S.; Mulchandani, A.; Deshusses, M.A. Metal and Metal Oxide Co-Functionalized Single-Walled Carbon Nanotubes for High Performance Gas Sensors. US Patent 15/090,281, 4 April 2016. [Google Scholar]
- Cui, Y.; Wei, Q.Q.; Park, H.K.; Lieber, C.M. Nanowire nanosensors for highly sensitive and selective detection of biological and chemical species. Science 2001, 293, 1289–1292. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.X.; Yin, L.W.; Zhang, L.Y.; Xiang, D.; Gao, R. Metal oxide gas sensors: Sensitivity and influencing factors. Sensors 2010, 10, 2088–2106. [Google Scholar] [CrossRef] [PubMed]
- Gurlo, A. Nanosensors: Towards morphological control of gas sensing activity. SnO2, In2O3, ZnO and WO3 case studies. Nanoscale 2011, 3, 154–165. [Google Scholar] [CrossRef] [PubMed]
- Yamazoe, N. New approaches for improving semiconductor gas sensors. Sens. Actuators B-Chem. 1991, 5, 7–19. [Google Scholar] [CrossRef]
- Schedin, F.; Geim, A.K.; Morozov, S.V.; Hill, E.W.; Blake, P.; Katsnelson, M.I.; Novoselov, K.S. Detection of individual gas molecules adsorbed on graphene. Nat. Mater. 2007, 6, 652–655. [Google Scholar] [CrossRef] [PubMed]
- Yuan, W.J.; Shi, G.Q. Graphene-based gas sensors. J. Mater. Chem. A 2013, 1, 10078–10091. [Google Scholar] [CrossRef]
- Zhang, D.Z.; Chang, H.Y.; Li, P.; Liu, R.H. Characterization of nickel oxide decorated-reduced graphene oxide nanocomposite and its sensing properties toward methane gas detection. J. Mater. Sci.-Mater. Electron. 2016, 27, 3723–3730. [Google Scholar] [CrossRef]
- Abideen, Z.U.; Katoch, A.; Kim, J.H.; Kwon, Y.J.; Kim, H.W.; Kim, S.S. Excellent gas detection of zno nanofibers by loading with reduced graphene oxide nanosheets. Sens. Actuators B-Chem. 2015, 221, 1499–1507. [Google Scholar] [CrossRef]
- Xu, S.P.; Sun, F.Q.; Pan, Z.Z.; Huang, C.W.; Yang, S.M.; Long, J.F.; Chen, Y. Reduced graphene oxide-based ordered macroporous films on a curved surface: General fabrication and application in gas sensors. ACS Appl. Mater. Interfaces 2016, 8, 3428–3437. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhao, Y.; Wang, X.; Wang, J.; Gaskov, A.M.; Akbar, S. Reduced graphene oxide (rGO) decorated TiO2 microspheres for selective room-temperature gas sensors. Sens. Actuators B Chem. 2016, 230, 330–336. [Google Scholar] [CrossRef]
- Latif, U.; Dickert, F.L. Graphene hybrid materials in gas sensing applications. Sensors 2015, 15, 30504–30524. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.F.; Chen, W.G.; Peng, S.Y.; Zhou, Q. Study on gas sensing properties and mechanism of Ag-doped SnO2 gas sensor to H2. In Proceedings of the 2015 IEEE Electrical Insulation Conference (EIC), Seattle, WA, USA, 7–10 June 2015; pp. 407–410.
- Li, L.; Yang, H.; Zhao, H.; Yu, J.; Ma, J.; An, L.; Wang, X. Hydrothermal synthesis and gas sensing properties of single-crystalline ultralong ZnO nanowires. Appl. Phys. A 2010, 98, 635–641. [Google Scholar] [CrossRef]
- Qi, Q.; Zhang, T.; Yu, Q.; Wang, R.; Zeng, Y.; Liu, L.; Yang, H. Properties of humidity sensing ZnO nanorods-base sensor fabricated by screen-printing. Sens. Actuators B Chem. 2008, 133, 638–643. [Google Scholar] [CrossRef]
- Vincenzi, D.; Butturi, M.; Guidi, V.; Carotta, M.; Martinelli, G.; Guarnieri, V.; Brida, S.; Margesin, B.; Giacomozzi, F.; Zen, M. Development of a low-power thick-film gas sensor deposited by screen-printing technique onto a micromachined hotplate. Sens. Actuators B Chem. 2001, 77, 95–99. [Google Scholar] [CrossRef]
- Ehsani, M.; Hamidon, M.N.; Toudeshki, A.; Abadi, M.S.; Rezaeian, S. CO2 gas sensing properties of screen-printed La2O3/SnO2 thick film. IEEE Sens. J. 2016, 16, 6839–6845. [Google Scholar] [CrossRef]
- DiGiulio, M.; Micocci, G.; Serra, A.; Tepore, A.; Rella, R.; Siciliano, R. Characteristics of reactively sputtered Pt-SnO2 thin films for CO gas sensors. J. Vac. Sci. Technol. a-Vac. Surf. Films 1996, 14, 2215–2219. [Google Scholar] [CrossRef]
- Zhang, D.Z.; Liu, J.J.; Chang, H.Y.; Liu, A.M.; Xia, B.K. Characterization of a hybrid composite of SnO2 nanocrystal-decorated reduced graphene oxide for ppm-level ethanol gas sensing application. RSC Adv. 2015, 5, 18666–18672. [Google Scholar] [CrossRef]
- Dieguez, A.; Romano-Rodriguez, A.; Vila, A.; Morante, J.R. The complete Raman spectrum of nanometric SnO2 particles. J. Appl. Phys. 2001, 90, 1550–1557. [Google Scholar] [CrossRef]
- Zhang, H.; Feng, J.C.; Fei, T.; Liu, S.; Zhang, T. SnO2 nanoparticles-reduced graphene oxide nanocomposites for NO2 sensing at low operating temperature. Sens. Actuators B-Chem. 2014, 190, 472–478. [Google Scholar] [CrossRef]
- Li, W.Q.; Ma, S.Y.; Li, Y.F.; Yang, G.J.; Mao, Y.Z.; Luo, J.; Gengzang, D.J.; Xu, X.L.; Yan, S.H. Enhanced ethanol sensing performance of hollow ZnO-SnO2 core-shell nanofibers. Sens. Actuators B-Chem. 2015, 211, 392–402. [Google Scholar] [CrossRef]
- Lu, G.; Ocola, L.E.; Chen, J. Reduced graphene oxide for room-temperature gas sensors. Nanotechnology 2009, 20. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Zhou, Q.; Gao, T.; Su, X.; Wan, F. Pd-doped SnO2-based sensor detecting characteristic fault hydrocarbon gases in transformer oil. J. Nanomater. 2013, 2013. [Google Scholar] [CrossRef]
- D’Amico, A.; Di Natale, C.; Sarro, P.M. Ingredients for sensors science. Sens. Actuators B Chem. 2015, 207, 1060–1068. [Google Scholar] [CrossRef]
- Gurav, K.; Deshmukh, P.; Lokhande, C. LPG sensing properties of Pd-sensitized vertically aligned zno nanorods. Sens. Actuators B Chem. 2011, 151, 365–369. [Google Scholar] [CrossRef]
- Uddin, A.I.; Lee, K.-W.; Chung, G.-S. Acetylene gas sensing properties of an Ag-loaded hierarchical zno nanostructure-decorated reduced graphene oxide hybrid. Sens. Actuators B Chem. 2015, 216, 33–40. [Google Scholar] [CrossRef]
- Hosseini, Z.S.; Mortezaali, A. Room temperature H2S gas sensor based on rather aligned ZnO nanorods with flower-like structures. Sens. Actuators B Chem. 2015, 207, 865–871. [Google Scholar] [CrossRef]
- Kim, H.-J.; Lee, J.-H. Highly sensitive and selective gas sensors using p-type oxide semiconductors: Overview. Sens. Actuators B Chem. 2014, 192, 607–627. [Google Scholar] [CrossRef]
- Liewhiran, C.; Tamaekong, N.; Wisitsoraat, A.; Phanichphant, S. Highly selective environmental sensors based on flame-spray-made SnO2 nanoparticles. Sens. Actuators B Chem. 2012, 163, 51–60. [Google Scholar] [CrossRef]
- Qi, Q.; Zhang, T.; Zheng, X.J.; Fan, H.T.; Liu, L.; Wang, R.; Zeng, Y. Electrical response of Sm2O3-doped SnO2 to C2H2 and effect of humidity interference. Sens. Actuators B-Chem. 2008, 134, 36–42. [Google Scholar] [CrossRef]
- Tarnaekong, N.; Liewhiran, C.; Wisitsoraat, A.; Phanichphant, S. Acetylene sensor based on Pt/ZnO thick films as prepared by flame spray pyrolysis. Sens. Actuators B Chem. 2011, 152, 155–161. [Google Scholar] [CrossRef]
- Uddin, A.; Chung, G.S. Synthesis of highly dispersed ZnO nanoparticles on graphene surface and their acetylene sensing properties. Sens. Actuators B-Chem. 2014, 205, 338–344. [Google Scholar] [CrossRef]
- Hotovy, I.; Huran, J.; Siciliano, P.; Capone, S.; Spiess, L.; Rehacek, V. Enhancement of H2 sensing properties of nio-based thin films with a pt surface modification. Sens. Actuators B Chem. 2004, 103, 300–311. [Google Scholar] [CrossRef]
- Singkammo, S.; Wisitsoraat, A.; Sriprachuabwong, C.; Tuantranont, A.; Phanichphant, S.; Liewhiran, C. Electrolytically exfoliated graphene-loaded flame-made ni-doped SnO2 composite film for acetone sensing. ACS Appl. Mater. Interfaces 2015, 7, 3077–3092. [Google Scholar] [CrossRef] [PubMed]
- Russo, P.A.; Donato, N.; Leonardi, S.G.; Baek, S.; Conte, D.E.; Neri, G.; Pinna, N. Room-temperature hydrogen sensing with heteronanostructures based on reduced graphene oxide and tin oxide. Angew. Chemie Int. Ed. 2012, 51, 11053–11057. [Google Scholar] [CrossRef] [PubMed]
- Lin, Q.Q.; Li, Y.; Yang, M.J. Tin oxide/graphene composite fabricated via a hydrothermal method for gas sensors working at room temperature. Sens. Actuators B-Chem. 2012, 173, 139–147. [Google Scholar] [CrossRef]
| Ref. | Material | Temperature (°C) | Measuring Range (ppm) | Response (S = Ra/Rg) | τResponse (s) | τRecovery (s) |
|---|---|---|---|---|---|---|
| [38] | Pure SnO2 | 300 | 200–10,000 | 6.3 (10,000 ppm) | 34 | >10 min |
| [39] | Sm2O3/SnO2 | 180 | 1–5000 | 63.8 (1000 ppm) | 3 | 17 |
| [40] | Pt/ZnO | 300 | 50–10,000 | 43 (1000 ppm) | 6 | 65 |
| [41] | ZnO/rGO 1 | 250 | 30–1000 | 18.2 (100 ppm) | 100 | 28 |
| Our | SnO2–rGO | 180 | 0.5–500 | 12.4 (50 ppm) | 54 | 23 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jin, L.; Chen, W.; Zhang, H.; Xiao, G.; Yu, C.; Zhou, Q. Characterization of Reduced Graphene Oxide (rGO)-Loaded SnO2 Nanocomposite and Applications in C2H2 Gas Detection. Appl. Sci. 2017, 7, 19. https://doi.org/10.3390/app7010019
Jin L, Chen W, Zhang H, Xiao G, Yu C, Zhou Q. Characterization of Reduced Graphene Oxide (rGO)-Loaded SnO2 Nanocomposite and Applications in C2H2 Gas Detection. Applied Sciences. 2017; 7(1):19. https://doi.org/10.3390/app7010019
Chicago/Turabian StyleJin, Lingfeng, Weigen Chen, He Zhang, Gongwei Xiao, Chutian Yu, and Qu Zhou. 2017. "Characterization of Reduced Graphene Oxide (rGO)-Loaded SnO2 Nanocomposite and Applications in C2H2 Gas Detection" Applied Sciences 7, no. 1: 19. https://doi.org/10.3390/app7010019
APA StyleJin, L., Chen, W., Zhang, H., Xiao, G., Yu, C., & Zhou, Q. (2017). Characterization of Reduced Graphene Oxide (rGO)-Loaded SnO2 Nanocomposite and Applications in C2H2 Gas Detection. Applied Sciences, 7(1), 19. https://doi.org/10.3390/app7010019







