Formation of a Colloidal CdSe and ZnSe Quantum Dots via a Gamma Radiolytic Technique
Abstract
:1. Introduction
2. Materials and Methods
3. Characterization
4. Results and Discussion
4.1. Formation Mechanism of CdSe and ZnSe Nanoparticles
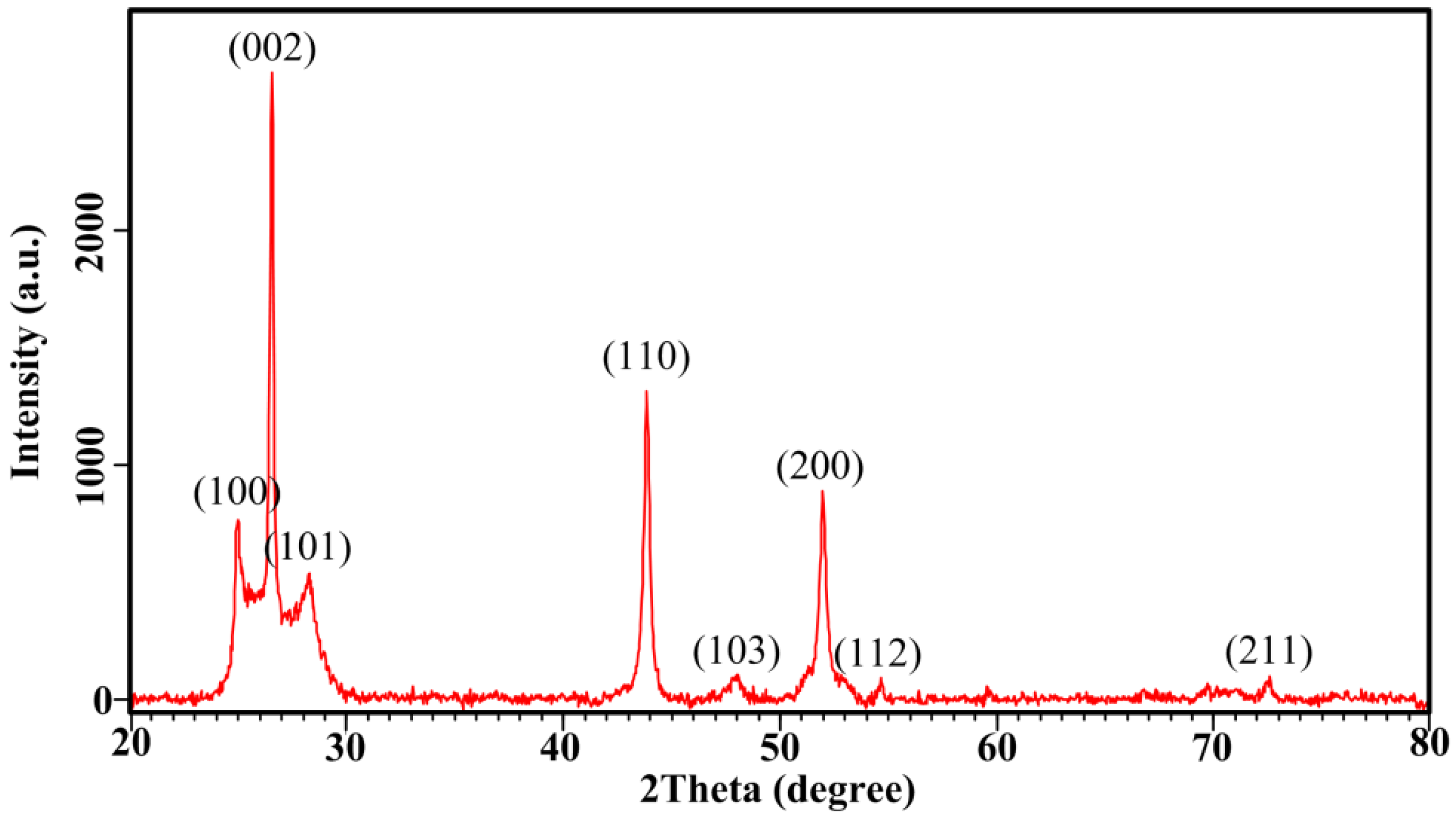
4.2. Structural Analysis (XRD)
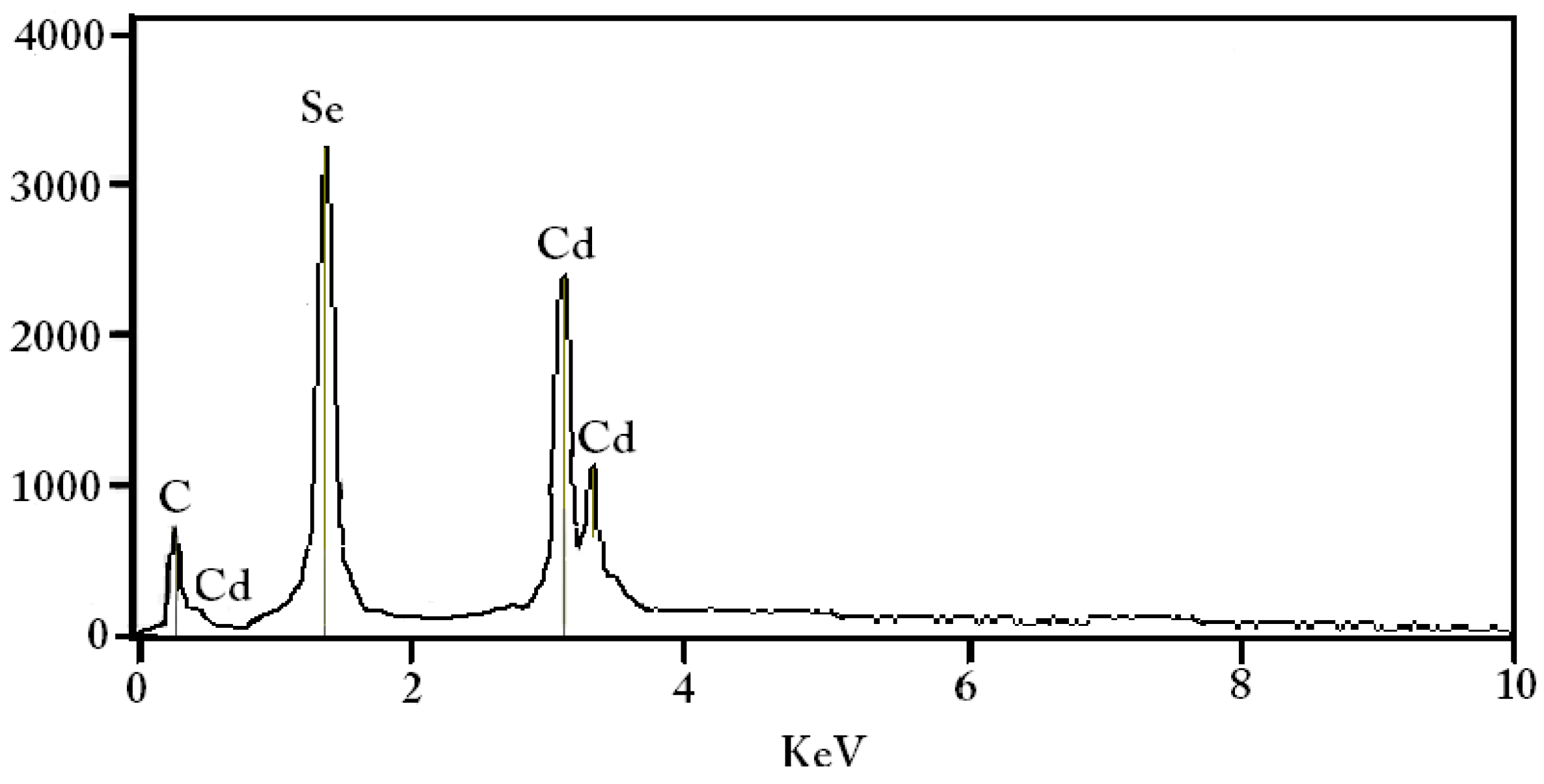
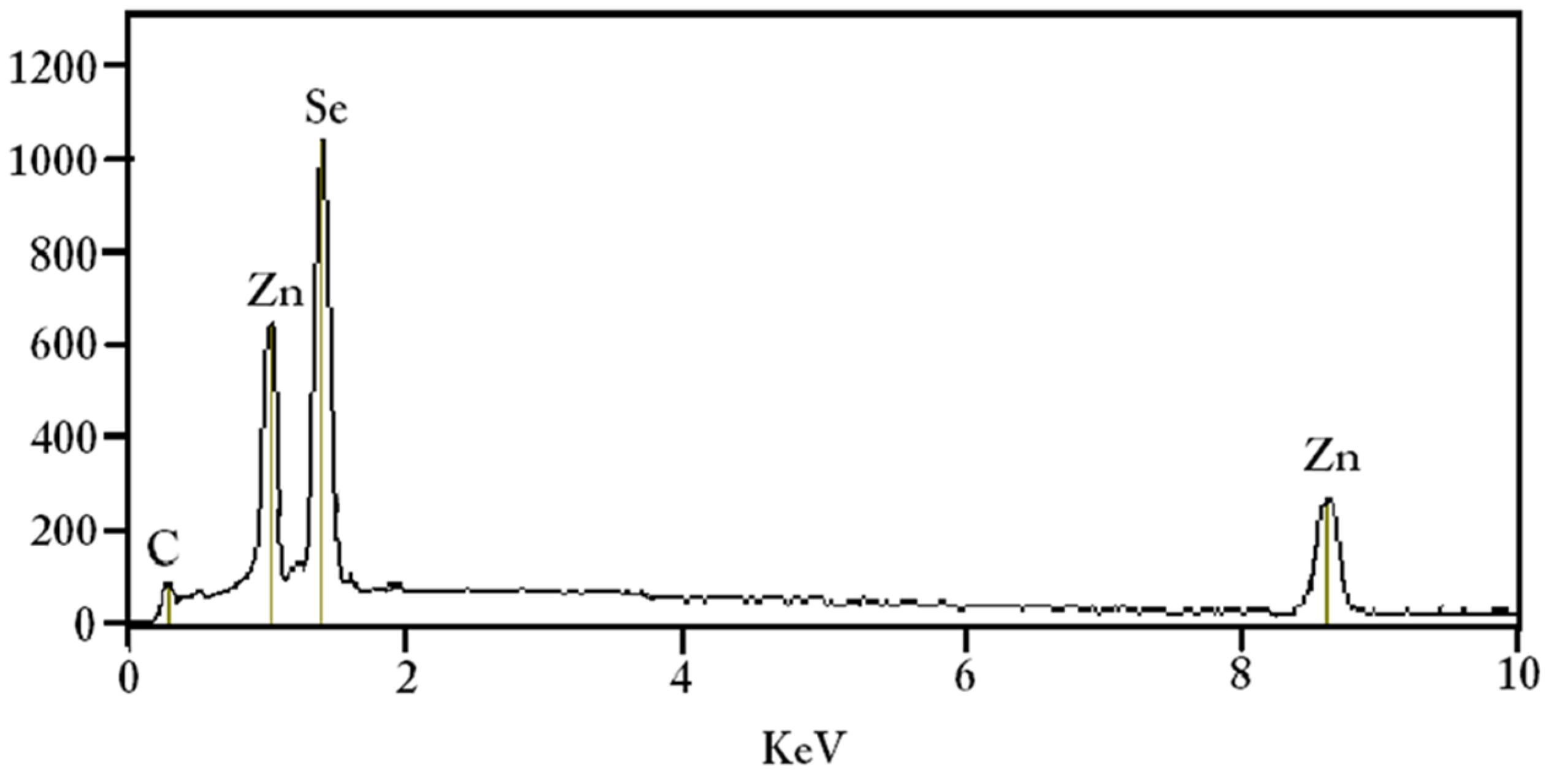
4.3. Elemental Composition Analysis (EDX)
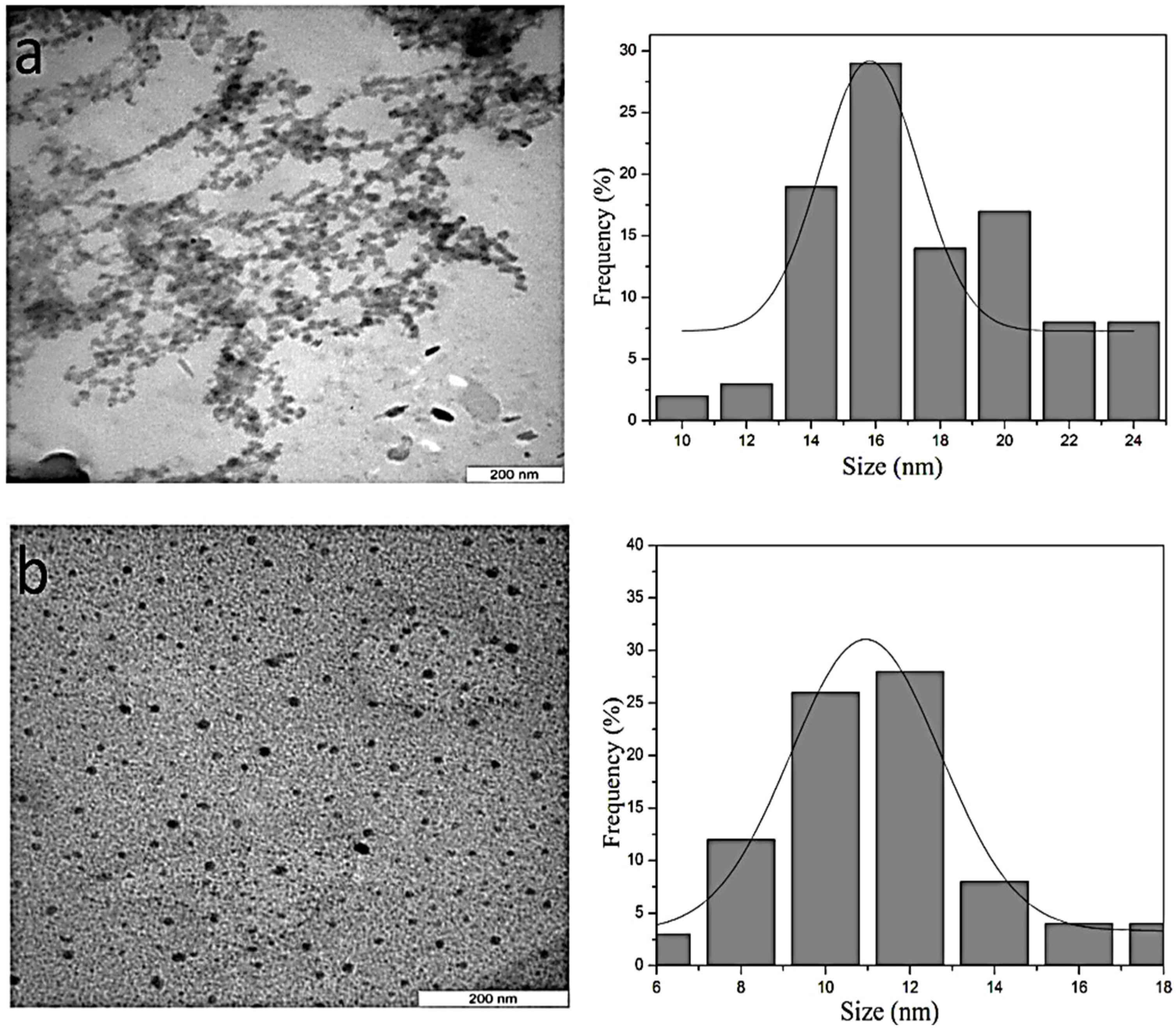
4.4. Morphology and Particle Size
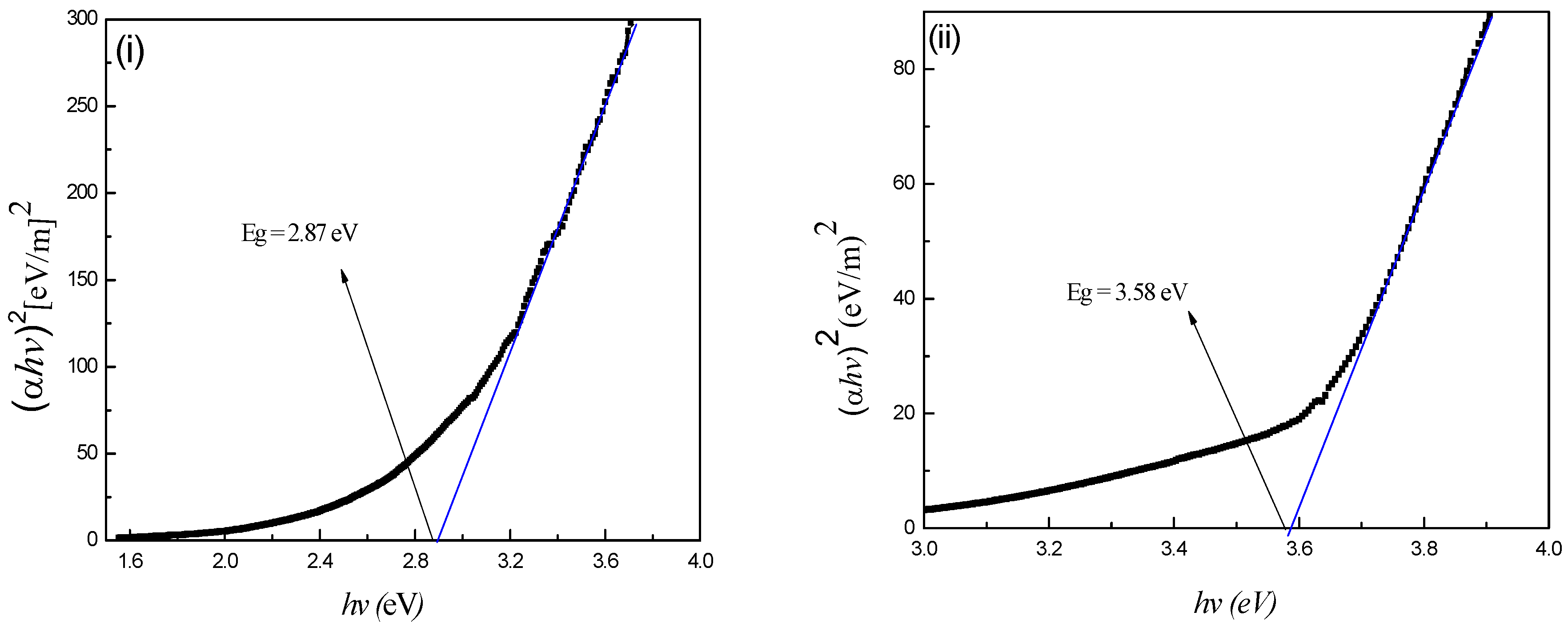
4.5. Optical Properties
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Talapin, D.V.; Lee, J.-S.; Kovalenko, M.V.; Shevchenko, E.V. Prospects of colloidal nanocrystals for electronic and optoelectronic applications. Chem. Rev. 2009, 110, 389–458. [Google Scholar] [CrossRef] [PubMed]
- Suresh, S. Semiconductor nanomaterials, methods and applications: A review. Nanosci. Nanotechnol. 2013, 3, 62–74. [Google Scholar]
- Boles, M.A.; Ling, D.; Hyeon, T.; Talapin, D.V. The surface science of nanocrystals. Nat. Mater. 2016, 15, 141–153. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.-S.; Hsu, C.-K.; Hong, H.-Y. Ingan-cdse-znse quantum dots white LEDs. IEEE Photonics Technol. Lett. 2006, 18, 193–195. [Google Scholar] [CrossRef]
- Žídek, K.; Abdellah, M.; Zheng, K.; Pullerits, T. Electron relaxation in the CdSe quantum dot-ZnO composite: Prospects for photovoltaic applications. Sci. Rep. 2014, 4. [Google Scholar] [CrossRef] [PubMed]
- Gronin, S.; Zverev, M.; Sorokin, S.; Sedova, I.; Gamov, N.; Peregoudov, D.; Studionov, V.; Kop’ev, P.; Ivanov, S. Effective green semiconductor lasers with multiple CdSe/ZnSe QD active region for electron beam pumping. Acta Phys. Pol. A 2008, 114, 1115–1122. [Google Scholar] [CrossRef]
- Saidov, A.; Amonov, K.; Kutlimurotov, B. Direct solar conversion to electricity nanoscale effects in pSi–n(Si2)1–x(ZnSe)x (0 ≤ x ≤ 0.01) of solar cells. Appl. Sol. Energy 2016, 52, 1–4. [Google Scholar] [CrossRef]
- Jiang, F.; Muscat, A.J. Ligand-controlled growth of ZnSe quantum dots in water during ostwald ripening. Langmuir 2012, 28, 12931–12940. [Google Scholar] [CrossRef] [PubMed]
- William, W.Y.; Chang, E.; Drezek, R.; Colvin, V.L. Water-soluble quantum dots for biomedical applications. Biochem. Biophys. Res. Commun. 2006, 348, 781–786. [Google Scholar]
- Soltani, N.; Saion, E.; Erfani, M.; Rezaee, K.; Bahmanrokh, G.; Drummen, G.P.; Bahrami, A.; Hussein, M.Z. Influence of the polyvinyl pyrrolidone concentration on particle size and dispersion of ZnS nanoparticles synthesized by microwave irradiation. Int. J. Mol. Sci. 2012, 13, 12412–12427. [Google Scholar] [CrossRef] [PubMed]
- Steigerwald, M.L.; Alivisatos, A.P.; Gibson, J.; Harris, T.; Kortan, R.; Muller, A.; Thayer, A.; Duncan, T.; Douglass, D.; Brus, L.E. Surface derivatization and isolation of semiconductor cluster molecules. J. Am. Chem. Soc. 1988, 110, 3046–3050. [Google Scholar] [CrossRef]
- Chen, H.-S.; Lo, B.; Hwang, J.-Y.; Chang, G.-Y.; Chen, C.-M.; Tasi, S.-J.; Wang, S.-J.J. Colloidal znse, ZnSe/ZnS, and ZnSe/ZnSeS quantum dots synthesized from zno. J. Phys. Chem. B 2004, 108, 17119–17123. [Google Scholar] [CrossRef]
- Aboulaich, A.; Geszke, M.; Balan, L.; Ghanbaja, J.; Medjahdi, G.; Schneider, R. Water-based route to colloidal mn-doped ZnSe and core/shell ZnSe/ZnS quantum dots. Inorg. Chem. 2010, 49, 10940–10948. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Liao, X.; Zhao, X.; Wang, J. Photochemical synthesis and characterization of CdSe nanoparticles. Mater. Lett. 2001, 47, 339–343. [Google Scholar] [CrossRef]
- Entezari, M.H.; Ghows, N. Micro-emulsion under ultrasound facilitates the fast synthesis of quantum dots of cds at low temperature. Ultrason. Sonochem. 2011, 18, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Senthilkumar, K.; Kalaivani, T.; Kanagesan, S.; Balasubramanin, V. Low temperature method for synthesis of starch-capped ZnSe nanoparticles and its characterization studies. J. Appl. Phys. 2012, 112. [Google Scholar] [CrossRef]
- Gaeeni, M.R.; Tohidian, M.; Majles-Ara, M. Green synthesis of CdSe colloidal nanocrystals with strong green emission by the sol–gel method. Ind. Eng. Chem. Res. 2014, 53, 7598–7603. [Google Scholar] [CrossRef]
- Guleria, A.; Singh, S.; Rath, M.; Singh, A.; Adhikari, S.; Sarkar, S. Tuning of photoluminescence in cadmium selenide nanoparticles grown in CTAB based quaternary water-in-oil microemulsions. J. Lumin. 2012, 132, 652–658. [Google Scholar] [CrossRef]
- Amiri, G.R.; Fatahian, S.; Mahmoudi, S. Preparation and optical properties assessment of CdSe quantum dots. Mater. Sci. Appl. 2013, 4, 134–137. [Google Scholar] [CrossRef]
- Klauke, K.; Hahn, B.; Schütte, K.; Barthel, J.; Janiak, C. Bis ((dialkylamino) alkylselenolato) metal complexes as precursors for microwave-assisted synthesis of semiconductor metal selenide nanoparticles of zinc and cadmium in the ionic liquid [bMIm][BF4]. Nano-Struct. Nano-Objects 2015, 1, 24–31. [Google Scholar] [CrossRef]
- Xi, L.; Lam, Y.M.; Xu, Y.P.; Li, L.-J. Synthesis and characterization of one-dimensional cdse by a novel reverse micelle assisted hydrothermal method. J. Colloid Interface Sci. 2008, 320, 491–500. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Liu, L.; Xiao, D.; Zhu, J. Preparation and characterization of ZnSe nanocrystals by a microemulsion-mediated method. Mater. Lett. 2012, 72, 113–115. [Google Scholar] [CrossRef]
- Deshpande, M.; Chaki, S.; Patel, N.; Bhatt, S.; Soni, B. Study on nanoparticles of ZnSe synthesized by chemical method and their characterization. J. Nano Electron. Phys. 2011, 3, 193–202. [Google Scholar]
- Zeng, Q.; Xue, S.; Wu, S.; Gan, K.; Xu, L.; Han, J.; Zhou, W.; Shi, Y.; Zou, R. Synthesis, field emission and optical properties of znse nanobelts, nanorods and nanocones by hydrothermal method. Mater. Sci. Semicond. Process. 2015, 31, 189–194. [Google Scholar] [CrossRef]
- Naghavi, K.; Saion, E.; Rezaee, K.; Yunus, W.M.M. Influence of dose on particle size of colloidal silver nanoparticles synthesized by gamma radiation. Radiat. Phys. Chem. 2010, 79, 1203–1208. [Google Scholar] [CrossRef]
- Saion, E.; Gharibshahi, E.; Naghavi, K. Size-controlled and optical properties of monodispersed silver nanoparticles synthesized by the radiolytic reduction method. Int. J. Mol. Sci. 2013, 14, 7880–7896. [Google Scholar] [CrossRef] [PubMed]
- Gharibshahi, E.; Saion, E. Influence of dose on particle size and optical properties of colloidal platinum nanoparticles. Int. J. Mol. Sci. 2012, 13, 14723–14741. [Google Scholar] [CrossRef] [PubMed]
- Abedini, A.; Larki, F.; Saion, E.; Zakaria, A.; Hussein, M.Z. Influence of dose and ion concentration on formation of binary al–ni alloy nanoclusters. Radiat. Phys. Chem. 2012, 81, 1653–1658. [Google Scholar] [CrossRef]
- Abedini, A.; Larki, F.; Saion, E.; Noroozi, M. Effect of Cu2+/Al3+ mole ratio on structure of Cu–Al bimetallic nanoparticles prepared by radiation induced method. Kerntechnik 2013, 78, 214–219. [Google Scholar] [CrossRef]
- Abedini, A.; Daud, A.R.; Hamid, M.A.A.; Othman, N.K.; Saion, E. A review on radiation-induced nucleation and growth of colloidal metallic nanoparticles. Nanoscale Res. Lett. 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Kharazmi, A.; Saion, E.; Faraji, N.; Soltani, N.; Dehzangi, A. Optical properties of CdS/PVA nanocomposite films synthesized using the gamma-irradiation-induced method. Chin. Phys. Lett. 2013, 30, 1–5. [Google Scholar] [CrossRef]
- Kharazmi, A.; Faraji, N.; Hussin, R.M.; Saion, E.; Yunus, W.M.M.; Behzad, K. Structural, optical, opto-thermal and thermal properties of ZnS–PVA nanofluids synthesized through a radiolytic approach. Beilstein J. Nanotechnol. 2015, 6, 529–536. [Google Scholar] [CrossRef] [PubMed]
- Khampieng, T.; Brikshavana, P.; Supaphol, P. Silver nanoparticle-embedded poly (vinyl pyrrolidone) hydrogel dressing: Gamma-ray synthesis and biological evaluation. J. Biomater. Sci. Polym. Ed. 2014, 25, 826–842. [Google Scholar] [CrossRef] [PubMed]
- Cullity, B.D. Elements of X-ray Diffraction, 2nd ed.; Addison-Wesley: London, UK, 1978. [Google Scholar]
- Simmons, B.A.; Li, S.; John, V.T.; McPherson, G.L.; Bose, A.; Zhou, W.; He, J. Morphology of CdS nanocrystals synthesized in a mixed surfactant system. Nano Lett. 2002, 2, 263–268. [Google Scholar] [CrossRef]

| Sample | Element | Wt/% |
|---|---|---|
| CdSe | Se | 34.27 |
| Cd | 50.47 | |
| C | 15.26 | |
| ZnSe | Se | 46.13 |
| Zn | 33.65 | |
| C | 20.22 |
| Sample | Crystal Structure | Crystallite Size/nm | Particle Size/nm | Bandgap /eV |
|---|---|---|---|---|
| CdSe | Hexagonal | 16.3 | 17.3 | 2.87 |
| ZnSe | Hexagonal | 10.7 | 11.2 | 3.58 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salem, A.; Saion, E.; Al-Hada, N.M.; Shaari, A.H.; Kamari, H.M.; Soltani, N.; Radiman, S. Formation of a Colloidal CdSe and ZnSe Quantum Dots via a Gamma Radiolytic Technique. Appl. Sci. 2016, 6, 278. https://doi.org/10.3390/app6100278
Salem A, Saion E, Al-Hada NM, Shaari AH, Kamari HM, Soltani N, Radiman S. Formation of a Colloidal CdSe and ZnSe Quantum Dots via a Gamma Radiolytic Technique. Applied Sciences. 2016; 6(10):278. https://doi.org/10.3390/app6100278
Chicago/Turabian StyleSalem, Aeshah, Elias Saion, Naif Mohammed Al-Hada, Abdul Halim Shaari, Halimah Mohamed Kamari, Nayereh Soltani, and Shahidan Radiman. 2016. "Formation of a Colloidal CdSe and ZnSe Quantum Dots via a Gamma Radiolytic Technique" Applied Sciences 6, no. 10: 278. https://doi.org/10.3390/app6100278
APA StyleSalem, A., Saion, E., Al-Hada, N. M., Shaari, A. H., Kamari, H. M., Soltani, N., & Radiman, S. (2016). Formation of a Colloidal CdSe and ZnSe Quantum Dots via a Gamma Radiolytic Technique. Applied Sciences, 6(10), 278. https://doi.org/10.3390/app6100278







