Study of Temperature Effect on Cascade Characteristics of Nanofluidic Energy Absorption System
Abstract
1. Introduction
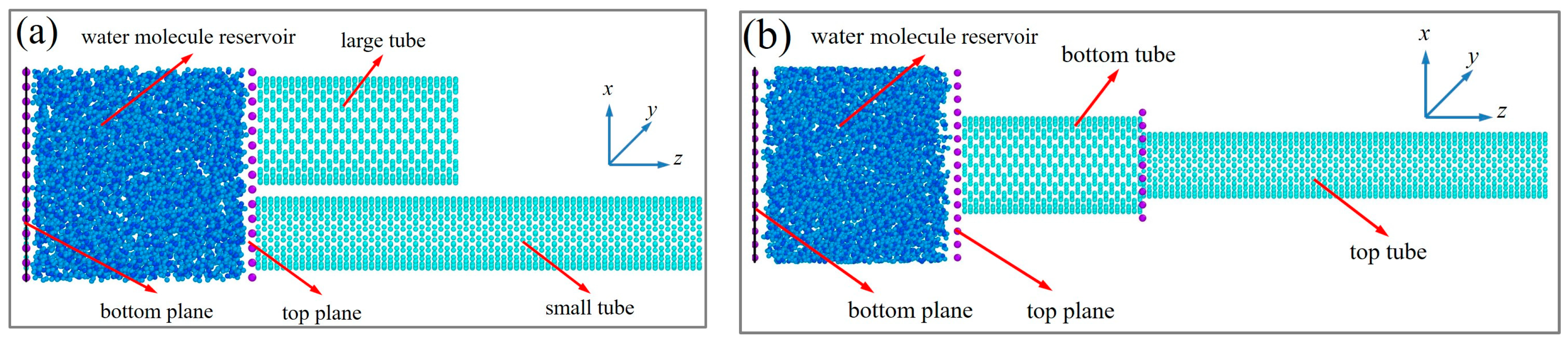
2. Establishment of Molecular Dynamics Model
3. Results and Analysis
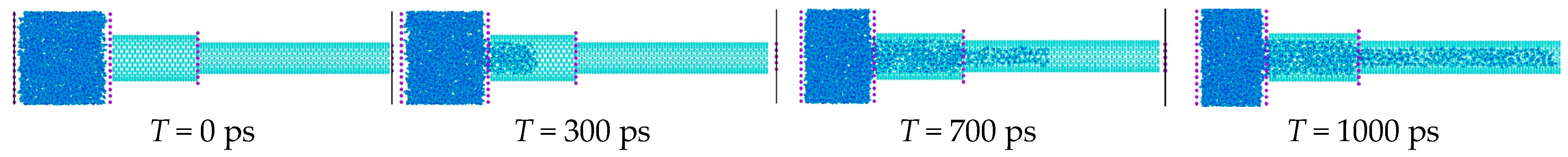
3.1. Cascade Infiltration Process of Water Molecules into CNTs
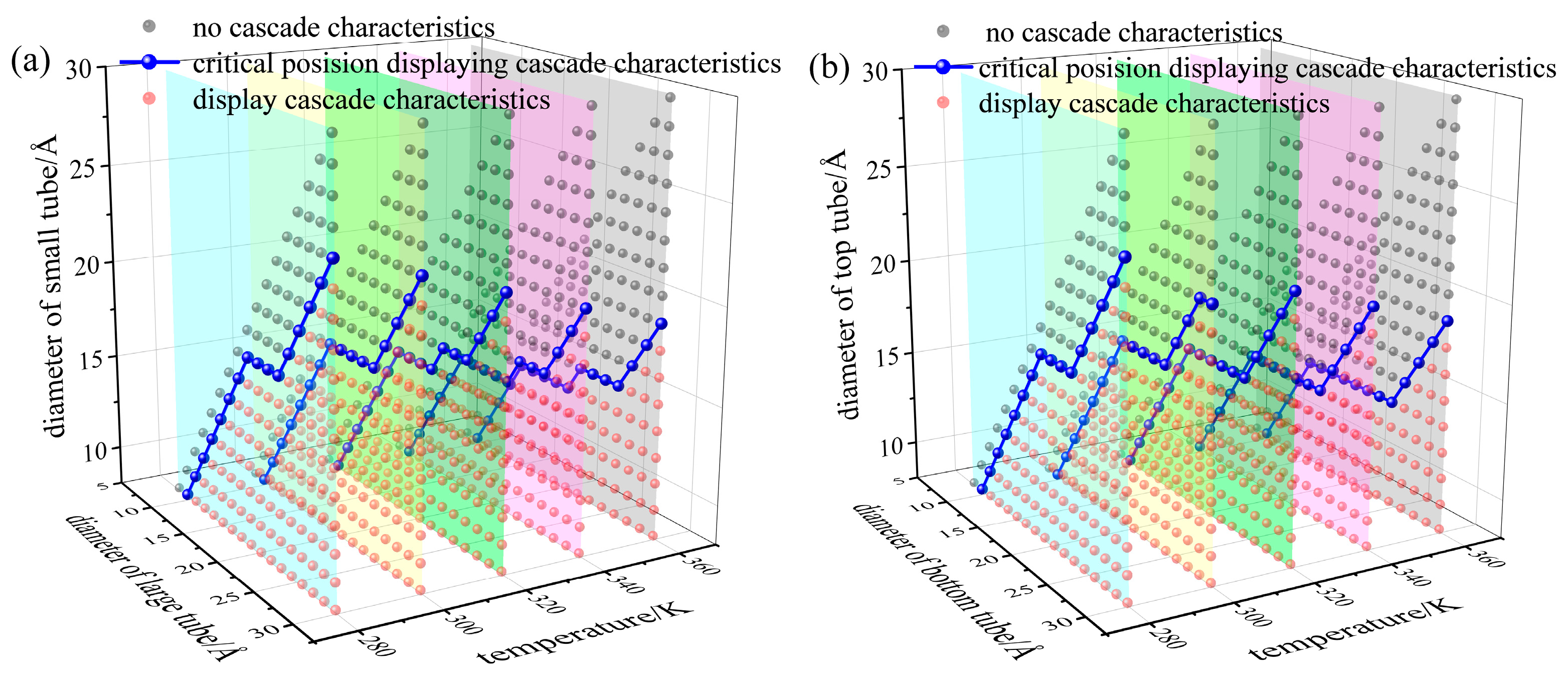
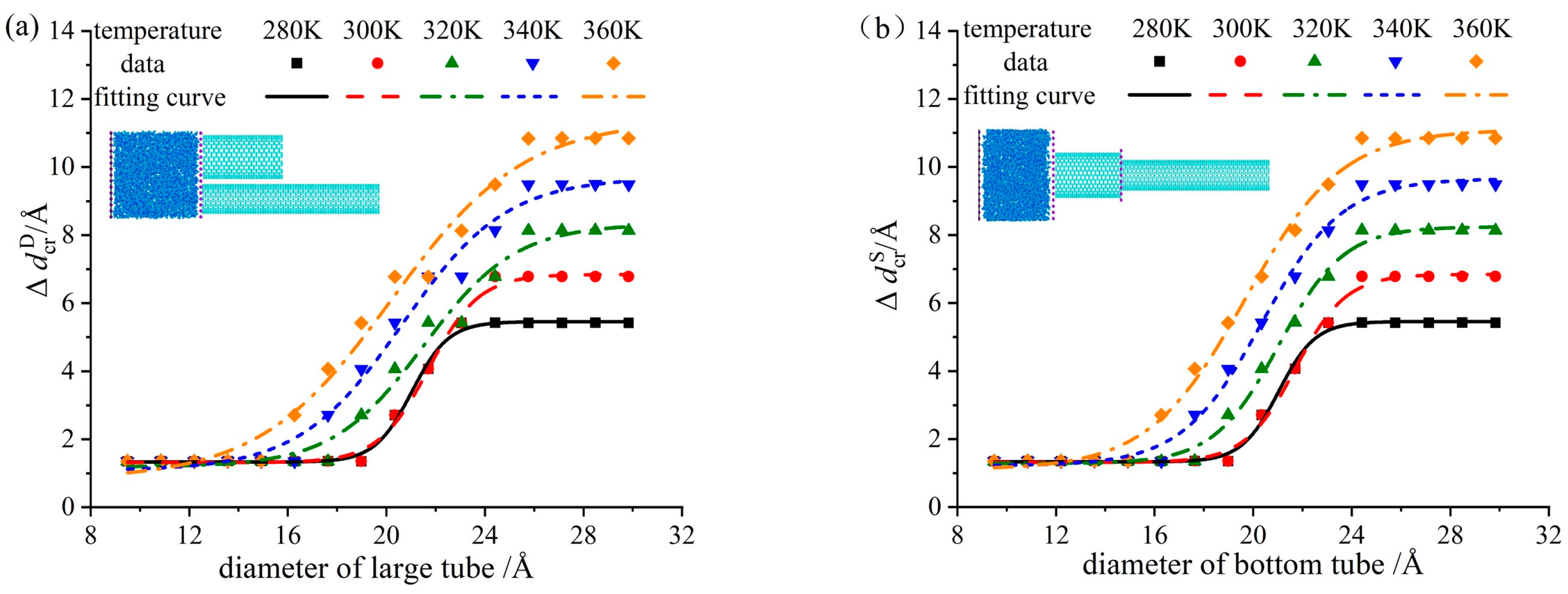
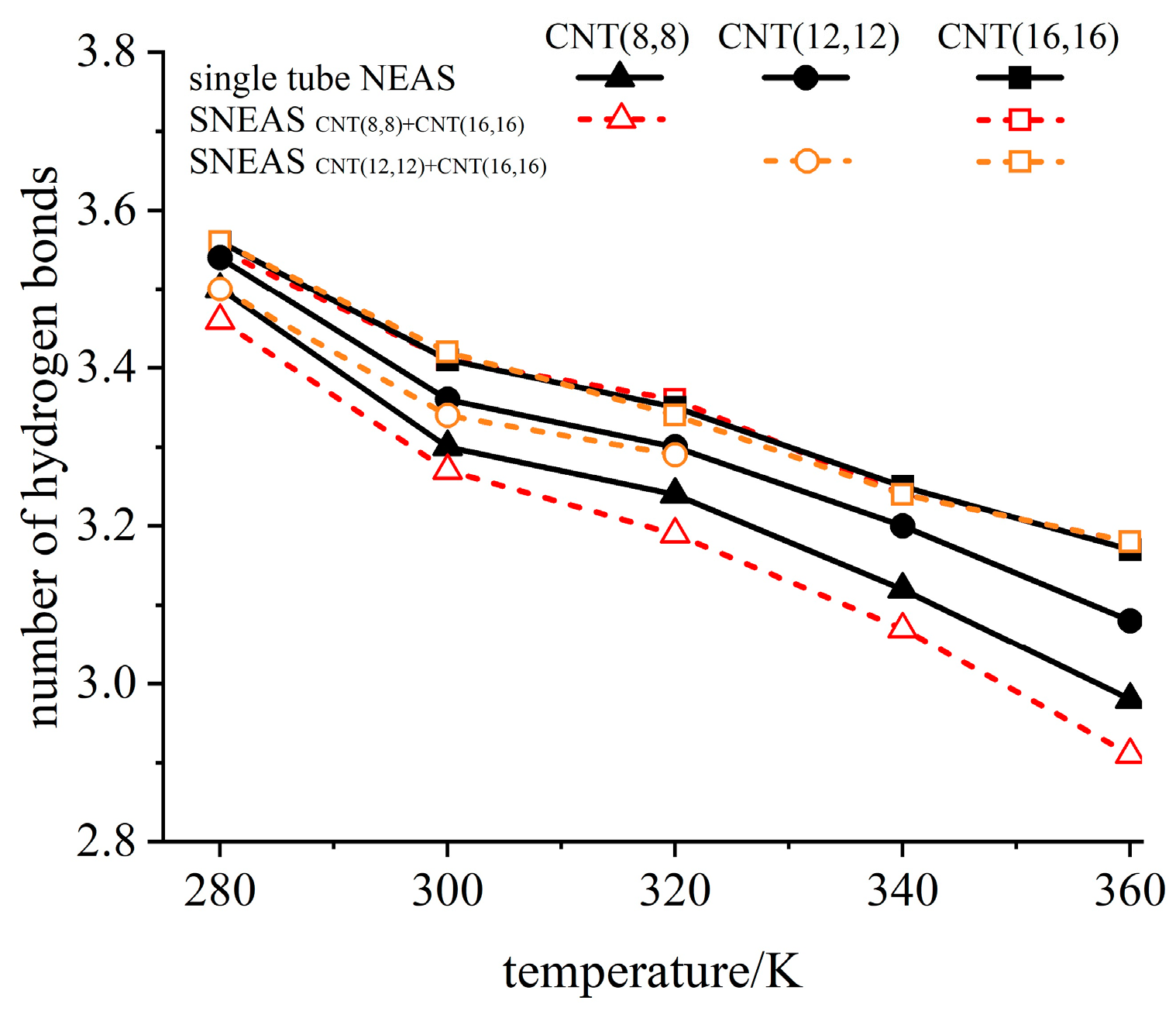
3.2. Analysis of Critical Pore Difference
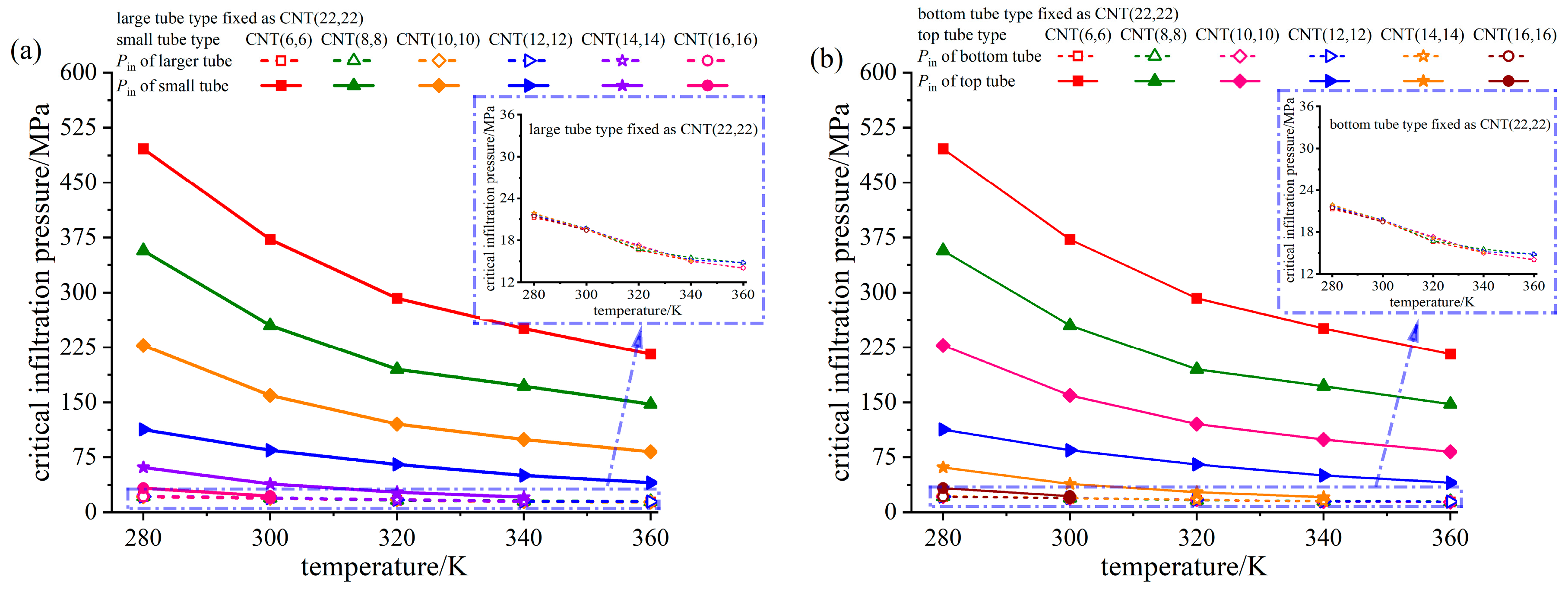
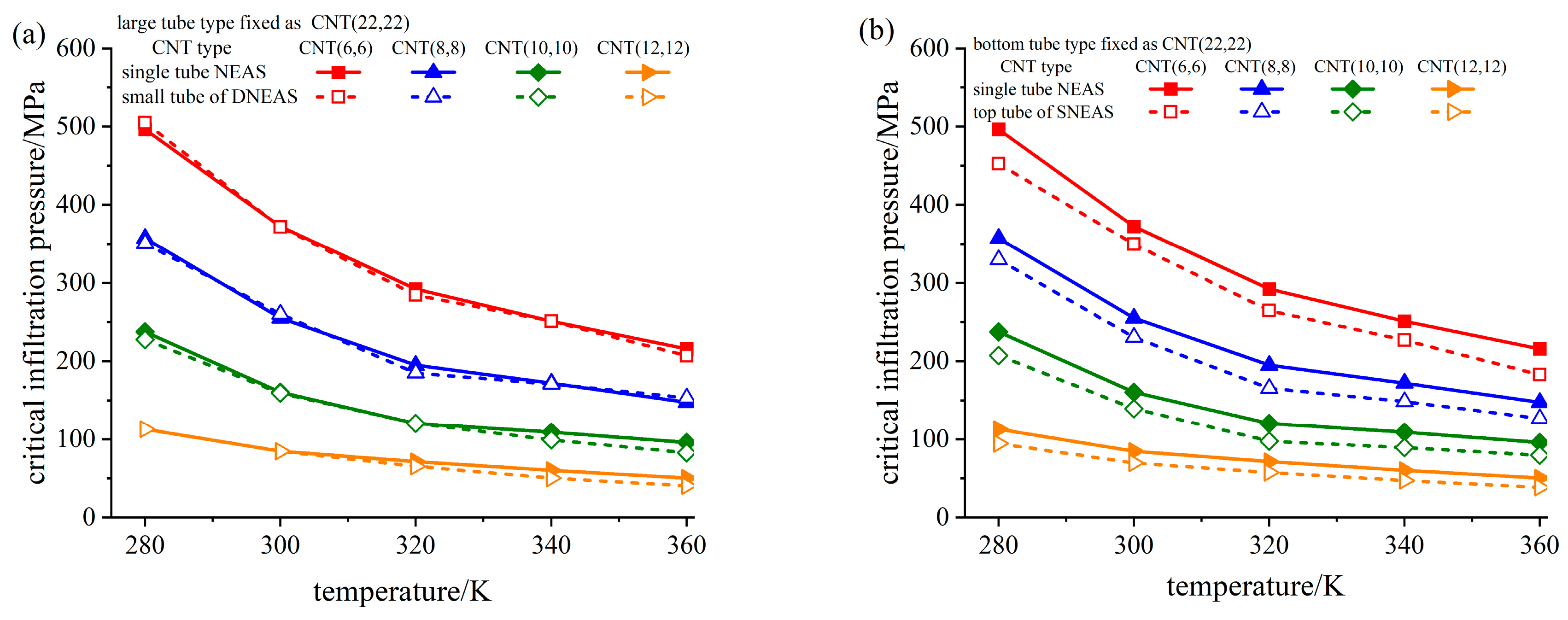
3.3. Analysis of Critical Infiltration Pressure
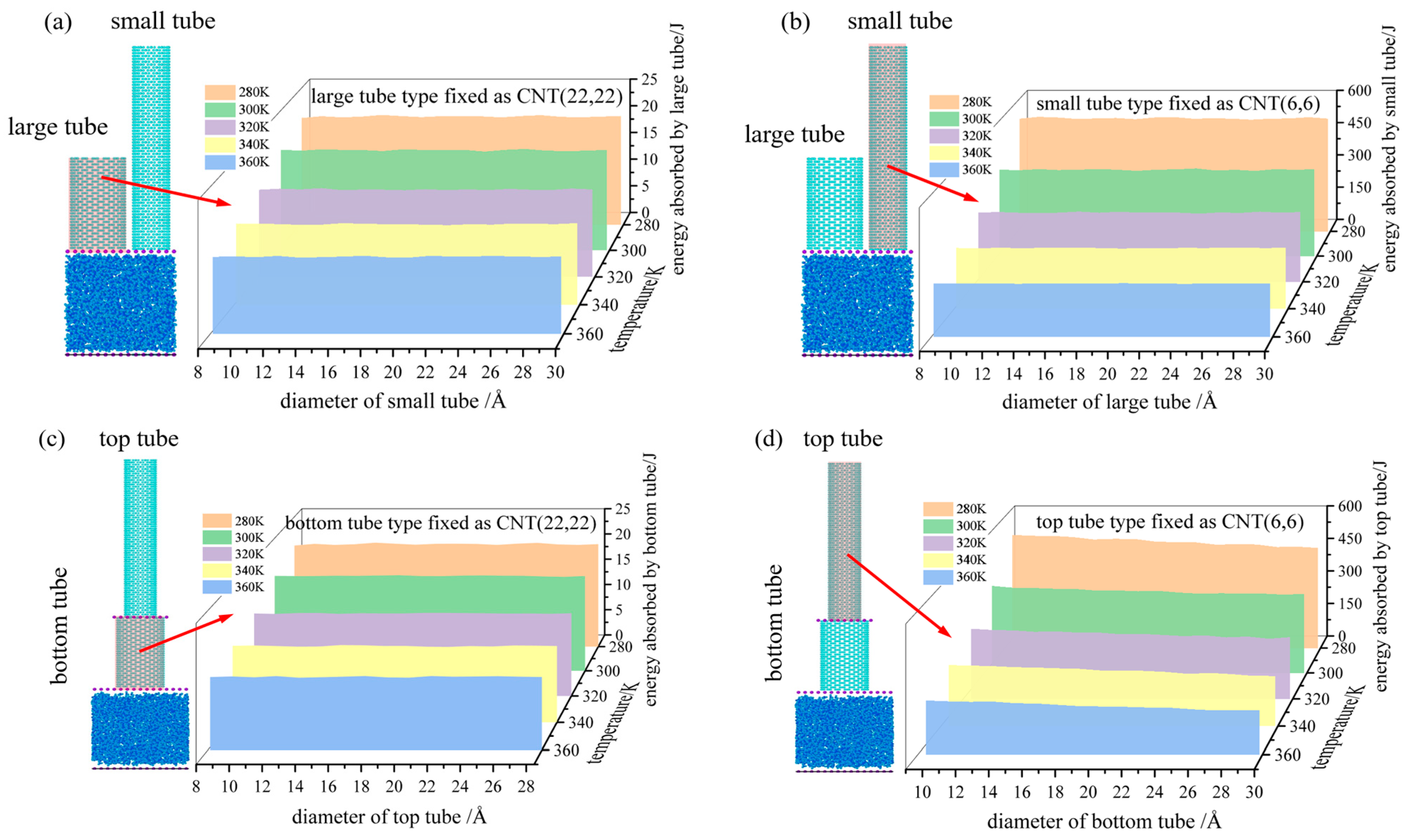
3.4. Analysis of System Energy Absorption
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chen, X.; Qiao, Y. Science and prospects of using nanoporous materials for energy absorption. MRS Online Proc. Libr. 2007, 205, 1041. [Google Scholar] [CrossRef]
- Liu, Z.; Huang, X.; Liu, R. Voltage generation induced by thermo-driven ion solution flow in CNTs for low-grade thermal energy harvesting. Int. J. Heat. Mass. Transfer. 2023, 202, 123751. [Google Scholar] [CrossRef]
- Fraux, G.; Coudert, F.X.; Boutin, A.; Fuchs, A.H. Forced intrusion of water and aqueous solutions in microporous materials: From fundamental thermodynamics to energy storage devices. Chem. Soc. Rev. 2017, 46, 7421. [Google Scholar] [CrossRef] [PubMed]
- Giacomello, A.; Casciola, C.; Grosu, Y.; Meloni, S. Liquid intrusion in and extrusion from non-wettable nanopores for technological applications. Eur. Phys. J. B 2021, 94, 163. [Google Scholar] [CrossRef]
- Grosu, Y.; Mierzwa, M.; Eroshenko, V.A.; Pawlus, S.; Chorażewski, M.; Nedelec, J.M.; Grolier, J.P.E. Mechanical, thermal, and electrical energy storage in a single working body: Electrification and thermal effects upon pressure-induced water intrusion-extrusion in nanoporous solids. ACS. Appl. Mater. Interfaces 2017, 9, 7044–7049. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Xu, Y.; Tong, Y.; Hu, J. The investigation of nanofluidic energy absorption system based on high porosity aerogel nano-materials. Microporous Mesoporous Mater. 2019, 277, 217–218. [Google Scholar] [CrossRef]
- Xu, B.; Wang, B.; Park, T.; Qiao, Y.; Zhou, Q.; Chen, X. Temperature dependence of fluid transport in nanopores. J. Chem. Phys. 2012, 136, 184701. [Google Scholar] [CrossRef]
- Ashok, D.; Bahubalendruni, M.; Mertens, A.J. A novel nature inspired 3D open lattice structure for specific energy absorption. Proc. Inst. Mech. Eng. E J. Process. Mech. Eng. 2022, 263, 2434–2440. [Google Scholar] [CrossRef]
- Pinna, A.; Pia, G.; Casula, M.F.; Delogu, F.; Sogne, E.; Falqui, A.; Pilia, L. Fabrication of nanoporous Al by vapor-phase dealloying: Morphology features, mechanical properties and model predictions. Appl. Sci. 2021, 11, 6639. [Google Scholar] [CrossRef]
- Jee, H.; Jeon, K.; Park, M.J.; Lee, J. Fabrication of large area, ordered nanoporous structures on various substrates for potential electro-optic applications. Appl. Sci. 2021, 11, 12136. [Google Scholar] [CrossRef]
- Li, M.; Xu, L.; Lu, W. Nanopore size effect on critical infiltration depth of liquid nanofoam as a reusable energy absorber. J. Appl. Phys. 2019, 125, 044303. [Google Scholar] [CrossRef]
- Wu, K.; Chen, Z.; Li, J.; Xu, J.; Wang, K.; Li, R.; Wang, S.; Dong, X. Ultrahigh water flow enhancement by optimizing nanopore chemistry and geometry. Langmuir 2019, 35, 8867–8873. [Google Scholar] [CrossRef] [PubMed]
- Maziar, F.T.; Xu, W.; Marshall, B.D.; Ravikovitch, P.I.; Müller, E.A. Significant effect of rugosity on transport of hydrocarbon liquids in carbonaceous nanopores. Energy Fuels 2022, 36, 10026–10042. [Google Scholar]
- Aluru, N.R.; Aydin, F.; Bazant, M.Z.; Blankschtein, D.; Brozena, A.H.; de Souza, J.P.; Elimelech, M.; Faucher, S.; Fourkas, J.T.; Koman, V.B.; et al. Fluids and electrolytes under confinement in single-digit nanopores. Chem. Rev. 2023, 123, 2737–2831. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Wu, S.; Tian, S.; Huang, D.; Xiong, G.; Luo, T. Molecular-level understanding of the effect of water on oil transport in graphene nanochannels. J. Phys. Chem. 2023, 127, 3671–3681. [Google Scholar] [CrossRef]
- Leong, I.W.; Tsutsui, M.; Murayama, S.; Hayashida, T.; He, Y.; Taniguchi, M. Quasi-stable salt gradient and resistive switching in solid-state nanopores. ACS. Appl. Mater. Interfaces 2020, 12, 52175–52181. [Google Scholar] [CrossRef]
- Ma, P.; Zheng, J.; Zhao, D.; Zhang, W.; Lu, G.; Lin, L.; Zhao, Z.; Huang, Z.; Cao, L. The selective transport of ions in charged nanopore with combined multi-physics fields. Materials 2021, 14, 7012. [Google Scholar] [CrossRef]
- Liu, L.; Chen, X.; Lu, W.; Han, A.; Qiao, Y. Infiltration of electrolytes in molecular-sized nanopores. Phys. Rev. Lett. 2009, 102, 184501. [Google Scholar] [CrossRef]
- Qiao, Y.; Cao, G.; Chen, X. Effects of gas molecules on nanofluidic behaviors. J. Am. Chem. Soc. 2007, 129, 2355–2359. [Google Scholar] [CrossRef]
- Sun, Y.; Li, P.; Qiao, Y.; Li, Y. Time-dependent gas-liquid interaction in molecular-sized nanopores. Sci. Rep. 2014, 4, 6547. [Google Scholar] [CrossRef]
- Zhang, Y.; Luo, R.; Zhou, Q.; Chen, X.; Dou, Y. Effect of degassing on the stability and reversibility of glycerol/ZSM-5 zeolite system. Appl. Sci. 2018, 8, 1065. [Google Scholar] [CrossRef]
- Kong, X.; Qiao, Y. Thermal effects on pressure-induced infiltration of a nanoporous system. Philos. Mag. Lett. 2005, 85, 331–337. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, N.; Luo, R.; Zhang, Y.; Chen, X. Experimental study on thermal effect on infiltration mechanisms of glycerol into ZSM-5 zeolite under cyclic loadings. J. Phys. D 2015, 49, 025303. [Google Scholar] [CrossRef]
- Salman, S.; Zhao, Y.; Zhang, X.; Su, J. Effect of temperature on the coupling transport of water and ions through a carbon nanotube in an electric field. J. Chem. Phys. 2020, 153, 184503. [Google Scholar] [CrossRef]
- Fraux, G.; Boutin, A.; Fuchs, A.; Coudert, F.X. Structure, dynamics and thermodynamics of intruded electrolytes in ZIF-8. J. Phys. Chem. C 2019, 25, 123. [Google Scholar] [CrossRef]
- Surani, F.B.; Kong, X.; Panchal, D.; Qiao, Y. Energy absorbed of a nanoporous system subjected to dynamic loadings. Appl. Phys. Lett. 2005, 87, 163111. [Google Scholar] [CrossRef]
- Sun, Y.; Rogge, S.M.; Lamaire, A.; Vandenbrande, S.; Wieme, J.; Siviour, C.R.; Van Speybroeck, V.; Tan, J.C. High-rate nanofluidic energy absorption in porous zeolitic frameworks. Nat. Mater. 2021, 20, 1015–1023. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Qiao, Y.; Li, Y.; Zhou, Q.; Chen, X. An electroactuation system based on nanofluids. Appl. Phys. Lett. 2011, 98, 221909. [Google Scholar] [CrossRef]
- Ritos, K.; Borg, M.K.; Mottram, N.J.; Reese, J.M. Electric fields can control the transport of water in carbon nanotubes. Philos. Trans. R. Soc. A 2016, 374, 20150025. [Google Scholar] [CrossRef]
- Russell, W.S.; Lin, C.Y.; Siwy, Z.S. Gating with charge inversion to control ionic transport in nanopores. ACS. Appl. Nano Mater. 2022, 5, 17682–17692. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, Y.; Su, J. Promoting Electroosmotic water flow through a carbon nanotube by weakening the competition between cations and anions in a lateral electric field. Langmuir 2022, 38, 3530–3539. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, J.; Luo, R.; Dou, Y. Experimental study on the effects of applied electric field on liquid infiltration into hydrophobic zeolite. Energies 2023, 16, 5065. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhou, Q.; Chen, X.; Dou, Y. Nanofluidic Energy Absorption/Conversion System and Its Application, 1st ed.; Xi ’an Jiaotong University Press: Xi ’an, China, 2019; pp. 67–72. [Google Scholar]
- Sun, Y. Study on Energy Dissipation Mechanism of Liquid System of Nanometer Porous Material for Vehicle. Doctor’s Thesis, Tsinghua University, Beijing, China, 2015. [Google Scholar]
- Gu, H.; Marth, J.D.; Orban, P.C.; Mossmann, H.; Rajewsky, K. Deletion of a DNA polymerase β gene segment in T cells using cell type-specific gene targeting. Science 1994, 265, 100–103. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wu, K.; Chen, Z.; Li, J.; Yu, X.; Yang, S.; Hui, G.; Yang, M. Quasi-continuum water flow under nanoconfined conditions: Coupling the effective viscosity and the slip length. Ind. Eng. Chem. Res. 2020, 59, 20504–20514. [Google Scholar] [CrossRef]
- Vaitheeswaran, S.; Rasaiah, J.C.; Hummer, G. Electric field and temperature effects on water in the narrow nonpolar pores of carbon nanotubes. J. Chem. Phys. 2004, 121, 7955–7965. [Google Scholar] [CrossRef] [PubMed]
- Thiemann, F.L.; Schran, C.; Rowe, P.; Müller, E.A.; Michaelides, A. Water flow in single-wall nanotubes: Oxygen makes It slip, hydrogen makes it stick. ACS. Nano 2022, 16, 10775–10782. [Google Scholar] [CrossRef]
- Alghamdi, M.; Memon, A.A.; Muhammad, T.; Ali, M.R. A numerical investigation of a photovoltaic thermal system contained a trapezoidal channel with transport of silver and titanium oxide using the water as base fluids. Case Stud. Therm. Eng. 2023, 47, 103056. [Google Scholar] [CrossRef]
- Usman, M.A.A.; Alghamdi, M. A forced convection of water aluminum oxide nanofluid flow and heat transfer study for a three dimensional annular with inner rotated cylinder. Sci. Rep. 2022, 12, 16735. [Google Scholar] [CrossRef]
- Laouira, H.; Mebarek-Oudina, F.; Hussein, A.K.; Kolsi, L.; Merah, A.; Younis, O. Heat transfer inside a horizontal channel with an open trapezoidal enclosure subjected to a heat source of different lengths. Heat. Transfer. 2020, 49, 406–423. [Google Scholar] [CrossRef]
- Bui, A.T.; Thiemann, F.L.; Michaelides, A.; Cox, S.J. Classical quantum friction at water–carbon interfaces. Nano Lett. 2023, 23, 580–587. [Google Scholar] [CrossRef]
- Zhao, J.; Liu, L.; Culligan, P.J.; Chen, X. Thermal effect on the dynamic infiltration of water into single-walled carbon nanotubes. Phys. Rev. E Stat. Nonlin. Soft. Matter. Phys. 2009, 80, 061206. [Google Scholar] [CrossRef]
- Faucher, S.; Aluru, N.; Bazant, M.Z.; Blankschtein, D.; Brozena, A.H.; Cumings, J.; Pedro de Souza, J.; Elimelech, M.; Epsztein, R.; Fourkas, J.T.; et al. Critical knowledge gaps in mass transport through single-digit nanopores: A review and perspective. J. Phys. Chem. C 2019, 123, 21309–21326. [Google Scholar] [CrossRef]
- Alessio, A.; Stavros, K. Molecular simulation of water in carbon nanotubes. Chem. Rev. 2008, 108, 5014–5034. [Google Scholar]
- Cao, G. Working mechanism of nanoporous energy absorption system under high speed loading. J. Phys. Chem. C 2012, 116, 8278–8286. [Google Scholar] [CrossRef]
- Cao, G. Nanofluidic energy damper: Modeling, simulation and analysis. Mol. Phys. 2022, 7, 237–259. [Google Scholar] [CrossRef]
- Cox, B.J.; Hill, J.M. Carbon nanocones with curvature effects close to the vertex. Nanomaterials 2018, 8, 624. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Cao, G.; Han, A.; Punyamurtula, V.K.; Liu, L.; Culligan, P.J.; Kim, T.; Qiao, Y. Nanoscale fluid transport: Size and rate effects. Nano Lett. 2008, 8, 2988–2992. [Google Scholar] [CrossRef]
- Hummer, G.; Rasalah, J.G.; Noworyta, J.P. Water conduction through the hydrophobic channel of a carbon nanotube. Nature 2001, 414, 188–190. [Google Scholar] [CrossRef]
- Majumder, M.; Chopra, N.; Andrews, R.; Hinds, B.J. Nanoscale hydrodynamics: Enhanced flow in carbon nanotubes. Nature 2005, 44, 438. [Google Scholar] [CrossRef]
- Liu, L.; Zhao, J.; Culligan, P.J.; Qiao, Y.; Chen, X. Thermally responsive fluid behaviors in hydrophobic nanopores. Langmuir 2009, 25, 11862–11868. [Google Scholar] [CrossRef]
- Hoover, W.G. Canonical dynamics: Equilibrium phase-space distributions. Phys. Rev. A 1985, 31, 1695–1697. [Google Scholar] [CrossRef] [PubMed]
- Kumar, G.; Mishra, R.R. Introduction to molecular dynamics simulations. Lect. Notes Appl. Comput. 2022, 99, 1–19. [Google Scholar]
- Sun, H. Compass: An ab initio force-field optimized for condensed-phase applications—Overview with details on alkane and benzene compounds. J. Phys. Chem. B 1998, 102, 7338–7364. [Google Scholar] [CrossRef]
- Berendsen, H.J.C.; Grigera, J.R. The missing term in effective pair potentials. J. Phys. Chem. 1987, 91, 6269–6271. [Google Scholar] [CrossRef]
- Werder, T.; Walther, J.H. On the water− carbon interaction for use in molecular dynamics simulations of graphite and carbon nanotubes. J. Phys. Chem. B 2003, 107, 1345–1352. [Google Scholar] [CrossRef]
- Joseph, S.; Aluru, N.R. Why are carbon nanotubes fast transporters of water. Nano Lett. 2008, 8, 452–458. [Google Scholar] [CrossRef] [PubMed]
- Ryckaert, J.P.; Ciccotti, G. Numerical integration of the cartesian equations of motion of a system with constraints: Molecular dynamics of n-alkanes. J. Comput. Phys. 1977, 23, 327–341. [Google Scholar] [CrossRef]
- Hockney, R.W.; Eastwood, J.W. Computer Simulation Using Particles, 1st ed.; CRC Press: Boca Raton, FL, USA, 1988; pp. 266–304. [Google Scholar]
- Aidan, P.T.; Steven, J.P.; Mattson, W. General formulation of pressure and stress tensor for arbitrary many-body interaction potentials under periodic boundary conditions. Chem. Phys. 2009, 131, 154107. [Google Scholar]
- Zhang, Y.; Xiao, H. Research on the influence of pore size on the characteristics of cascade nanofluidic energy absorption systems. J. Mol. Sci. 2023; accepted. [Google Scholar]
- Chakraborty, S.; Kumar, H.; Dasgupta, C.; Maiti, P.K. Confined water: Structure, dynamics, and thermodynamics. Acc. Chem. Res. 2017, 50, 2139–2146. [Google Scholar] [CrossRef]
- Fecko, C.J. Ultrafast hydrogen-bond dynamics in the infrared spectroscopy of water. Science 2003, 301, 1698–1702. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.M.; Jiang, Q. Size-dependent surface tension and tolman’s length of droplets. Langmuir 2005, 21, 779–781. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Wang, H.; Xiao, H.; Liu, S.; Wei, W.; Dou, Y. Study of Temperature Effect on Cascade Characteristics of Nanofluidic Energy Absorption System. Appl. Sci. 2023, 13, 8150. https://doi.org/10.3390/app13148150
Zhang Y, Wang H, Xiao H, Liu S, Wei W, Dou Y. Study of Temperature Effect on Cascade Characteristics of Nanofluidic Energy Absorption System. Applied Sciences. 2023; 13(14):8150. https://doi.org/10.3390/app13148150
Chicago/Turabian StyleZhang, Yafei, Haitao Wang, Hongjiu Xiao, Shenlei Liu, Wenlan Wei, and Yihua Dou. 2023. "Study of Temperature Effect on Cascade Characteristics of Nanofluidic Energy Absorption System" Applied Sciences 13, no. 14: 8150. https://doi.org/10.3390/app13148150
APA StyleZhang, Y., Wang, H., Xiao, H., Liu, S., Wei, W., & Dou, Y. (2023). Study of Temperature Effect on Cascade Characteristics of Nanofluidic Energy Absorption System. Applied Sciences, 13(14), 8150. https://doi.org/10.3390/app13148150





