Impact of Preceded Tumor Therapeutic Irradiation on the Microtensile Bond Strength of Universal Adhesives Applied in Self-Etch Mode to Human Dentin In Vitro
Abstract
1. Introduction
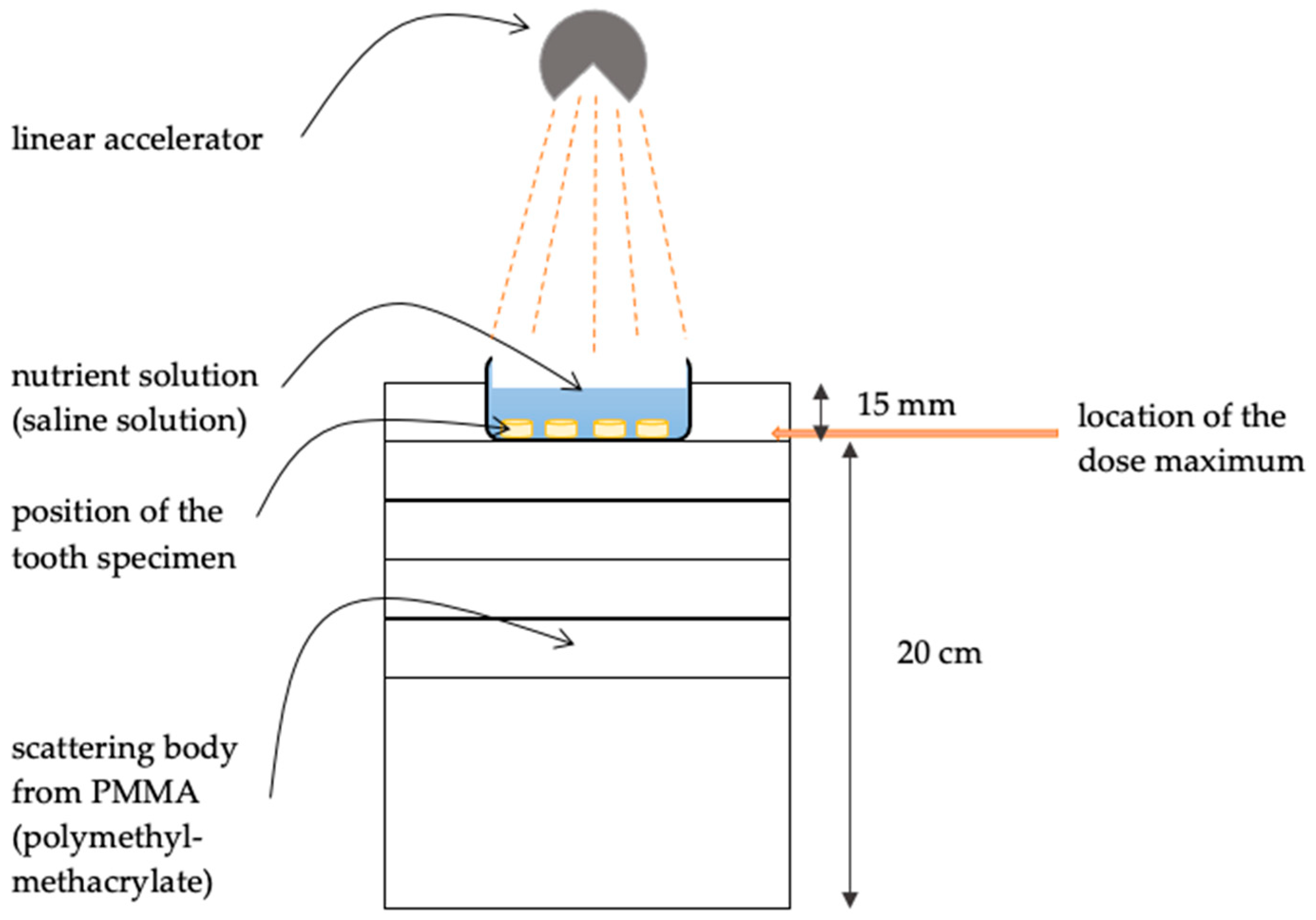
2. Materials and Methods
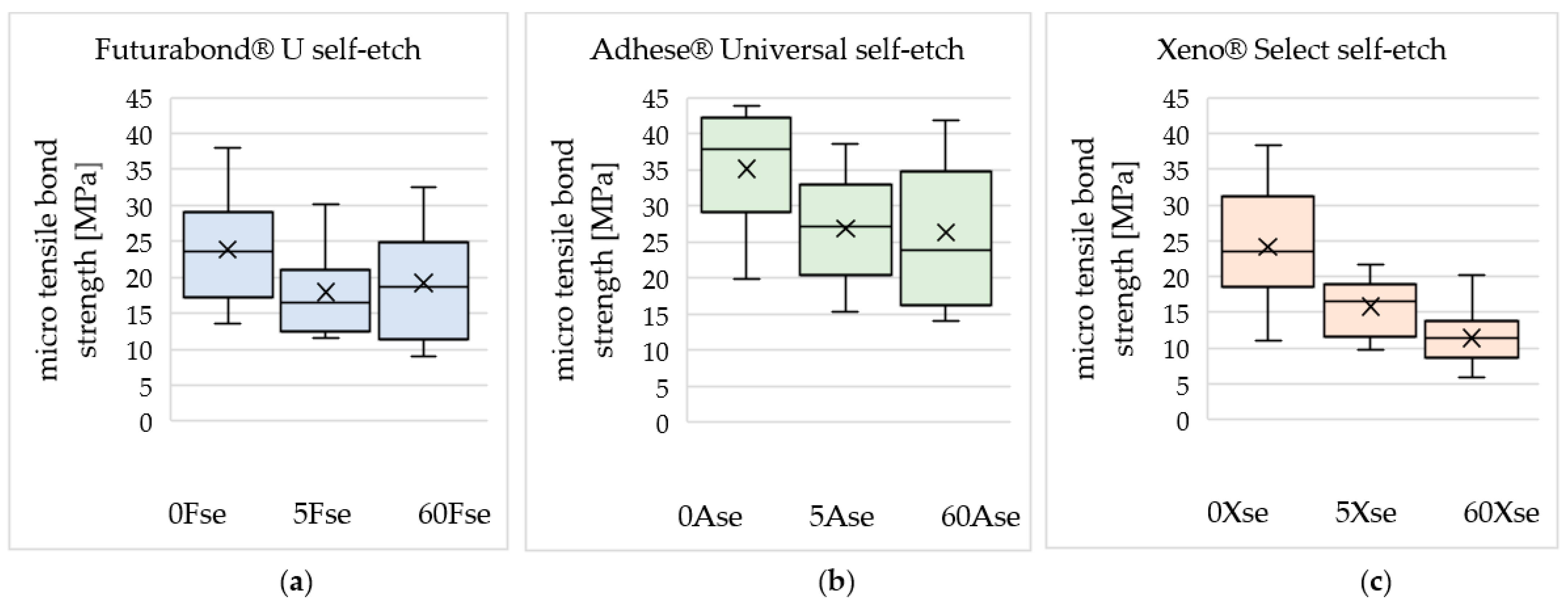
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Global Cancer Observatory. Cancer Today. Cancer Fact Sheets. Available online: https://gco.iarc.fr/today/fact-sheets-cancers (accessed on 28 March 2023).
- Robert Koch Institute; Association of Population-based Cancer Registries in Germany. Cancer in Germany 2017/2018, 13th ed.; Robert Koch Institute: Berlin, Germany, 2022; pp. 32–33. [Google Scholar] [CrossRef]
- Argiris, A.; Karamouzis, M.V.; Raben, D.; Ferris, R.L. Head and neck cancer. Lancet 2008, 371, 1695–1709. [Google Scholar] [CrossRef]
- Bourhis, J.; Etessami, A.; Lusinchi, A. New trends in radiotherapy for head and neck cancer. Ann. Oncol. 2005, 16 (Suppl. S2), ii255–ii257. [Google Scholar] [CrossRef] [PubMed]
- Sciubba, J.J.; Goldenberg, D. Oral complications of radiotherapy. Lancet Oncol. 2006, 7, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Kielbassa, A.M.; Hinkelbein, W.; Hellwig, E.; Meyer-Lückel, H. Radiation-related damage to dentition. Lancet Oncol. 2006, 7, 326–335. [Google Scholar] [CrossRef] [PubMed]
- Mesia, R.; Iglesias, L.; Lambea, J.; Martinez-Trufero, J.; Soria, A.; Taberna, M.; Trigo, J.; Chaves, M.; Garcia-Castano, A.; Cruz, J. SEOM clinical guidelines for the treatment of head and neck cancer (2020). Clin. Transl. Oncol. 2021, 23, 913–921. [Google Scholar] [CrossRef]
- Ray-Chaudhuri, A.; Shah, K.; Porter, R.J. The oral management of patients who have received radiotherapy to the head and neck region. Br. Dent. J. 2013, 214, 387–393. [Google Scholar] [CrossRef] [PubMed]
- Gernhardt, C.R.; Koravu, T.; Gerlach, R.; Schaller, H.G. The influence of dentin adhesives on the demineralization of irradiated and non-irradiated human root dentin. Oper. Dent. 2004, 29, 454–461. [Google Scholar]
- Hey, J.; Seidel, J.; Schweyen, R.; Paelecke-Habermann, Y.; Vordermark, D.; Gernhardt, C.; Kuhnt, T. The influence of parotid gland sparing on radiation damages of dental hard tissues. Clin. Oral Investig. 2013, 17, 1619–1625. [Google Scholar] [CrossRef]
- Kuhnt, T.; Jirsak, N.; Muller, A.C.; Pelz, T.; Gernhardt, C.; Schaller, H.G.; Janich, M.; Gerlach, R.; Dunst, J. Quantitative and qualitative investigations of salivary gland function in dependence on irradiation dose and volume for reduction of xerostomia in patients with head-and-neck cancer. Strahlenther. Onkol. 2005, 181, 520–528. [Google Scholar] [CrossRef]
- Fränzel, W.; Gerlach, R.; Hein, H.J.; Schaller, H.G. Effect of tumor therapeutic irradiation on the mechanical properties of teeth tissue. Z. Med. Phys. 2006, 16, 148–154. [Google Scholar] [CrossRef]
- Gonçalves, L.M.; Palma-Dibb, R.G.; Paula-Silva, F.W.; Oliveira, H.F.; Nelson-Filho, P.; Silva, L.A.; Queiroz, A.M. Radiation therapy alters microhardness and microstructure of enamel and dentin of permanent human teeth. J. Dent. 2014, 42, 986–992. [Google Scholar] [CrossRef] [PubMed]
- Reed, R.; Xu, C.; Liu, Y.; Gorski, J.P.; Wang, Y.; Walker, M.P. Radiotherapy effect on nano-mechanical properties and chemical composition of enamel and dentine. Arch. Oral Biol. 2015, 60, 690–697. [Google Scholar] [CrossRef] [PubMed]
- Nakabayashi, N.; Kojima, K.; Masuhara, E. The promotion of adhesion by the infiltration of monomers into tooth substrates. J. Biomed. Mater. Res. 1982, 16, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Hardan, L.; Daood, U.; Bourgi, R.; Cuevas-Suarez, C.E.; Devoto, W.; Zarow, M.; Jakubowicz, N.; Zamarripa-Calderon, J.E.; Radwanski, M.; Orsini, G.; et al. Effect of Collagen Crosslinkers on Dentin Bond Strength of Adhesive Systems: A Systematic Review and Meta-Analysis. Cells 2022, 11, 2417. [Google Scholar] [CrossRef] [PubMed]
- Eusufzai, S.Z.; Barman, A.; Jamayet, N.B.; Ahmad, W.; Mahdi, S.S.; Sheikh, Z.; Daood, U. Effects of Riboflavin Collagen Crosslinker on Dentin Adhesive Bonding Efficiency: A Systematic Review and Meta-Analysis. Materials 2023, 16, 1701. [Google Scholar] [CrossRef]
- Yadav, S.; Yadav, H. Ionizing irradiation affects the microtensile resin dentin bond strength under simulated clinical conditions. J. Conserv. Dent. 2013, 16, 148–151. [Google Scholar] [CrossRef]
- Gernhardt, C.R.; Nguyen, A.D.; Michaelis, M.; Puetz, N. Clinical Outcome of Class I and II Restorations with and without an Intermediary Layer of a Flowable Composite after 24 Months: A Prospective, Randomized, Split-Mouth-Designed, Controlled and Single-Blinded Clinical Trial. Appl. Sci. 2023, 13, 4224. [Google Scholar] [CrossRef]
- Badr, C.; Spagnuolo, G.; Amenta, F.; Khairallah, C.; Mahdi, S.S.; Daher, E.; Battineni, G.; Baba, N.Z.; Zogheib, T.; Qasim, S.S.B.; et al. A Two-Year Comparative Evaluation of Clinical Performance of a Nanohybrid Composite Resin to a Flowable Composite Resin. J. Funct. Biomater. 2021, 12, 51. [Google Scholar] [CrossRef]
- De Moor, R.J.G.; Stassen, I.G.; van ’t Veldt, Y.; Torbeyns, D.; Hommez, G.M.G. Two-year clinical performance of glass ionomer and resin composite restorations in xerostomic head- and neck-irradiated cancer patients. Clin. Oral Investig. 2011, 15, 31–38. [Google Scholar] [CrossRef]
- McComb, D.; Erickson, R.L.; Maxymiw, W.G.; Wood, R.E. A clinical comparison of glass ionomer, resin-modified glass ionomer and resin composite restorations in the treatment of cervical caries in xerostomic head and neck radiation patients. Oper. Dent. 2002, 27, 430–437. [Google Scholar]
- Van Meerbeek, B.; De Munck, J.; Yoshida, Y.; Inoue, S.; Vargas, M.; Vijay, P.; Van Landuyt, K.; Lambrechts, P.; Vanherle, G. Buonocore memorial lecture. Adhesion to enamel and dentin: Current status and future challenges. Oper. Dent. 2003, 28, 215–235. [Google Scholar] [PubMed]
- Hanabusa, M.; Mine, A.; Kuboki, T.; Momoi, Y.; Van Ende, A.; Van Meerbeek, B.; De Munck, J. Bonding effectiveness of a new ‘multi-mode’ adhesive to enamel and dentine. J. Dent. 2012, 40, 475–484. [Google Scholar] [CrossRef] [PubMed]
- Perdigão, J.; Sezinando, A.; Monteiro, P.C. Laboratory bonding ability of a multi-purpose dentin adhesive. Am. J. Dent. 2012, 25, 153–158. [Google Scholar] [PubMed]
- Nagarkar, S.; Theis-Mahon, N.; Perdigão, J. Universal dental adhesives: Current status, laboratory testing, and clinical performance. J. Biomed. Mater. Res. B. Appl. Biomater. 2019, 107, 2121–2131. [Google Scholar] [CrossRef]
- Van Landuyt, K.L.; Yoshida, Y.; Hirata, I.; Snauwaert, J.; De Munck, J.; Okazaki, M.; Suzuki, K.; Lambrechts, P.; Van Meerbeek, B. Influence of the chemical structure of functional monomers on their adhesive performance. J. Dent. Res. 2008, 87, 757–761. [Google Scholar] [CrossRef]
- Zhang, Z.Y.; Tian, F.C.; Niu, L.N.; Ochala, K.; Chen, C.; Fu, B.P.; Wang, X.Y.; Pashley, D.H.; Tay, F.R. Defying ageing: An expectation for dentine bonding with universal adhesives? J. Dent. 2016, 45, 43–52. [Google Scholar] [CrossRef]
- Yoshida, Y.; Nagakane, K.; Fukuda, R.; Nakayama, Y.; Okazaki, M.; Shintani, H.; Inoue, S.; Tagawa, Y.; Suzuki, K.; De Munck, J.; et al. Comparative study on adhesive performance of functional monomers. J. Dent. Res. 2004, 83, 454–458. [Google Scholar] [CrossRef]
- Yoshihara, K.; Nagaoka, N.; Okihara, T.; Kuroboshi, M.; Hayakawa, S.; Maruo, Y.; Nishigawa, G.; De Munck, J.; Yoshida, Y.; Van Meerbeek, B. Functional monomer impurity affects adhesive performance. Dent. Mater. 2015, 31, 1493–1501. [Google Scholar] [CrossRef]
- Chen, C.; Niu, L.N.; Xie, H.; Zhang, Z.Y.; Zhou, L.Q.; Jiao, K.; Chen, J.H.; Pashley, D.H.; Tay, F.R. Bonding of universal adhesives to dentine--Old wine in new bottles? J. Dent. 2015, 43, 525–536. [Google Scholar] [CrossRef]
- Muñoz, M.A.; Luque, I.; Hass, V.; Reis, A.; Loguercio, A.D.; Bombarda, N.H. Immediate bonding properties of universal adhesives to dentine. J. Dent. 2013, 41, 404–411. [Google Scholar] [CrossRef]
- Wagner, A.; Wendler, M.; Petschelt, A.; Belli, R.; Lohbauer, U. Bonding performance of universal adhesives in different etching modes. J. Dent. 2014, 42, 800–807. [Google Scholar] [CrossRef] [PubMed]
- Bernard, C.; Villat, C.; Abouelleil, H.; Gustin, M.P.; Grosgogeat, B. Tensile Bond Strengths of Two Adhesives on Irradiated and Nonirradiated Human Dentin. Biomed. Res. Int. 2015, 2015, 798972. [Google Scholar] [CrossRef] [PubMed]
- Galetti, R.; Santos-Silva, A.R.; Antunes, A.N.; Alves Fde, A.; Lopes, M.A.; de Goes, M.F. Radiotherapy does not impair dentin adhesive properties in head and neck cancer patients. Clin. Oral Investig. 2014, 18, 1771–1778. [Google Scholar] [CrossRef] [PubMed]
- Gernhardt, C.R.; Kielbassa, A.M.; Hahn, P.; Schaller, H.G. Tensile bond strengths of four different dentin adhesives on irradiated and non-irradiated human dentin in vitro. J. Oral Rehabil. 2001, 28, 814–820. [Google Scholar] [CrossRef]
- Rodrigues, R.B.; Soares, C.J.; Junior, P.C.S.; Lara, V.C.; Arana-Chavez, V.E.; Novais, V.R. Influence of radiotherapy on the dentin properties and bond strength. Clin. Oral Investig. 2018, 22, 875–883. [Google Scholar] [CrossRef] [PubMed]
- Kobler, A.; Schaller, H.G.; Gernhardt, C.R. Effects of the desensitizing agents Gluma and Hyposen on the tensile bond strength of dentin adhesives. Am. J. Dent. 2008, 21, 388–392. [Google Scholar]
- Awwad, H.K.; Lotayef, M.; Shouman, T.; Begg, A.C.; Wilson, G.; Bentzen, S.M.; Abd El-Moneim, H.; Eissa, S. Accelerated hyperfractionation (AHF) compared to conventional fractionation (CF) in the postoperative radiotherapy of locally advanced head and neck cancer: Influence of proliferation. Br. J. Cancer 2002, 86, 517–523. [Google Scholar] [CrossRef]
- Kielbassa, A.M.; Beetz, I.; Schendera, A.; Hellwig, E. Irradiation effects on microhardness of fluoridated and non-fluoridated bovine dentin. Eur. J. Oral Sci. 1997, 105, 444–447. [Google Scholar] [CrossRef]
- McLean, D.E.; Meyers, E.J.; Guillory, V.L.; Vandewalle, K.S. Enamel Bond Strength of New Universal Adhesive Bonding Agents. Oper. Dent. 2015, 40, 410–417. [Google Scholar] [CrossRef]
- Pouyanfar, H.; Tabaii, E.S.; Aghazadeh, S.; Nobari, S.; Imani, M.M. Microtensile Bond Strength of Composite to Enamel Using Universal Adhesive with/without Acid Etching Compared to Etch and Rinse and Self-Etch Bonding Agents. Open. Access. Maced. J. Med. Sci. 2018, 6, 2186–2192. [Google Scholar] [CrossRef]
- Elkaffas, A.A.; Hamama, H.H.H.; Mahmoud, S.H. Do universal adhesives promote bonding to dentin? A systematic review and meta-analysis. Restor. Dent. Endod. 2018, 43, e29. [Google Scholar] [CrossRef]
- Yoshida, Y.; Yoshihara, K.; Nagaoka, N.; Hayakawa, S.; Torii, Y.; Ogawa, T.; Osaka, A.; Meerbeek, B.V. Self-assembled Nano-layering at the Adhesive interface. J. Dent. Res. 2012, 91, 376–381. [Google Scholar] [CrossRef]
- Yoshihara, K.; Yoshida, Y.; Hayakawa, S.; Nagaoka, N.; Irie, M.; Ogawa, T.; Van Landuyt, K.L.; Osaka, A.; Suzuki, K.; Minagi, S.; et al. Nanolayering of phosphoric acid ester monomer on enamel and dentin. Acta Biomater. 2011, 7, 3187–3195. [Google Scholar] [CrossRef]
- Yoshida, Y.; Van Meerbeek, B.; Nakayama, Y.; Yoshioka, M.; Snauwaert, J.; Abe, Y.; Lambrechts, P.; Vanherle, G.; Okazaki, M. Adhesion to and decalcification of hydroxyapatite by carboxylic acids. J. Dent. Res. 2001, 80, 1565–1569. [Google Scholar] [CrossRef] [PubMed]
- Tian, F.; Zhou, L.; Zhang, Z.; Niu, L.; Zhang, L.; Chen, C.; Zhou, J.; Yang, H.; Wang, X.; Fu, B.; et al. Paucity of Nanolayering in Resin-Dentin Interfaces of MDP-based Adhesives. J. Dent. Res. 2016, 95, 380–387. [Google Scholar] [CrossRef] [PubMed]
- Torres, C.R.; Zanatta, R.F.; Silva, T.J.; Huhtala, M.F.; Borges, A.B. Influence of previous acid etching on bond strength of universal adhesives to enamel and dentin. Gen. Dent. 2017, 65, e17–e21. [Google Scholar]
- Carrilho, E.; Cardoso, M.; Marques Ferreira, M.; Marto, C.M.; Paula, A.; Coelho, A.S. 10-MDP Based Dental Adhesives: Adhesive Interface Characterization and Adhesive Stability-A Systematic Review. Materials 2019, 12, 790. [Google Scholar] [CrossRef]
- Steel, G.G.; McMillan, T.J.; Peacock, J.H. The 5Rs of radiobiology. Int. J. Radiat. Biol. 1989, 56, 1045–1048. [Google Scholar] [CrossRef] [PubMed]
- Withers, H. The four R’s of radiotherapy. Adv. Radiat. Biol. 1975, 5, 261. [Google Scholar]
- Bourhis, J.; Overgaard, J.; Audry, H.; Ang, K.K.; Saunders, M.; Bernier, J.; Horiot, J.C.; Le Maître, A.; Pajak, T.F.; Poulsen, M.G.; et al. Hyperfractionated or accelerated radiotherapy in head and neck cancer: A meta-analysis. Lancet 2006, 368, 843–854. [Google Scholar] [CrossRef]
- Fu, K.K.; Pajak, T.F.; Trotti, A.; Jones, C.U.; Spencer, S.A.; Phillips, T.L.; Garden, A.S.; Ridge, J.A.; Cooper, J.S.; Ang, K.K. A Radiation Therapy Oncology Group (RTOG) phase III randomized study to compare hyperfractionation and two variants of accelerated fractionation to standard fractionation radiotherapy for head and neck squamous cell carcinomas: First report of RTOG 9003. Int. J. Radiat. Oncol. Biol. Phys. 2000, 48, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Mallick, S.; Benson, R.; Julka, P.K.; Rath, G.K. Altered fractionation radiotherapy in head and neck squamous cell carcinoma. J. Egypt. Natl. Cancer Inst. 2016, 28, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Bortfeld, T. IMRT: A review and preview. Phys. Med. Biol. 2006, 51, R363–R379. [Google Scholar] [CrossRef]
- Nutting, C.M.; Morden, J.P.; Harrington, K.J.; Urbano, T.G.; Bhide, S.A.; Clark, C.; Miles, E.A.; Miah, A.B.; Newbold, K.; Tanay, M.; et al. Parotid-sparing intensity modulated versus conventional radiotherapy in head and neck cancer (PARSPORT): A phase 3 multicentre randomised controlled trial. Lancet Oncol. 2011, 12, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Arid, J.; Palma-Dibb, R.G.; de Oliveira, H.F.; Nelson-Filho, P.; de Carvalho, F.K.; da Silva, L.A.B.; de Siqueira Mellara, T.; da Silva, R.A.B.; Faraoni, J.J.; de Queiroz, A.M. Radiotherapy impairs adhesive bonding in permanent teeth. Support. Care Cancer 2020, 28, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Grötz, K.; Duschner, H.; Kutzner, J.; Thelen, M.; Wagner, W. Neue Erkenntnisse zur Ätiologie der sogenannten Strahlenkaries. Nachweis direkter radiogener Veränderungen an der Schmelz-Dentin-Grenze. Strahlenther. Onkol. 1997, 173, 668–676. (In German) [Google Scholar] [CrossRef]
- Lu, H.; Zhao, Q.; Guo, J.; Zeng, B.; Yu, X.; Yu, D.; Zhao, W. Direct radiation-induced effects on dental hard tissue. Radiat. Oncol. 2019, 14, 5. [Google Scholar] [CrossRef]
- Fonseca, J.M.; Troconis, C.C.; Palmier, N.R.; Gomes-Silva, W.; Paglioni, M.D.; Araújo, A.L.; Arboleda, L.P.; Filho, A.J.; González-Arriagada, W.A.; Goes, M.F.; et al. The impact of head and neck radiotherapy on the dentine-enamel junction: A systematic review. Med. Oral Patol. Oral Cir. Bucal 2020, 25, e96–e105. [Google Scholar] [CrossRef]
- Pioch, T.; Golfels, D.; Staehle, H.J. An experimental study of the stability of irradiated teeth in the region of the dentinoenamel junction. Endod. Dent. Traumatol. 1992, 8, 241–244. [Google Scholar] [CrossRef]
- al-Nawas, B.; Grötz, K.A.; Rose, E.; Duschner, H.; Kann, P.; Wagner, W. Using ultrasound transmission velocity to analyse the mechanical properties of teeth after in vitro, in situ, and in vivo irradiation. Clin. Oral Investig. 2000, 4, 168–172. [Google Scholar] [CrossRef]
- Seyedmahmoud, R.; Wang, Y.; Thiagarajan, G.; Gorski, J.P.; Reed Edwards, R.; McGuire, J.D.; Walker, M.P. Oral cancer radiotherapy affects enamel microhardness and associated indentation pattern morphology. Clin. Oral Investig. 2018, 22, 1795–1803. [Google Scholar] [CrossRef] [PubMed]
- Lieshout, H.F.; Bots, C.P. The effect of radiotherapy on dental hard tissue--a systematic review. Clin. Oral Investig. 2014, 18, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Schweyen, R.; Hey, J.; Fränzel, W.; Vordermark, D.; Hildebrandt, G.; Kuhnt, T. Radiation-related caries: Etiology and possible preventive strategies. What should the radiotherapist know? Strahlenther. Onkol. 2012, 188, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Kielbassa, A.M.; Wrbas, K.T.; Schulte-Mönting, J.; Hellwig, E. Correlation of transversal microradiography and microhardness on in situ-induced demineralization in irradiated and nonirradiated human dental enamel. Arch. Oral Biol. 1999, 44, 243–251. [Google Scholar] [CrossRef]
- Springer, I.N.; Niehoff, P.; Warnke, P.H.; Böcek, G.; Kovács, G.; Suhr, M.; Wiltfang, J.; Açil, Y. Radiation caries--radiogenic destruction of dental collagen. Oral Oncol. 2005, 41, 723–728. [Google Scholar] [CrossRef]
- Frank, R.M.; Herdly, J.; Philippe, E. Acquired dental defects and salivary gland lesions after irradiation for carcinoma. J. Am. Dent. Assoc. 1965, 70, 868–883. [Google Scholar] [CrossRef]
- Hong, C.H.L.; Hu, S.; Haverman, T.; Stokman, M.; Napeñas, J.J.; Braber, J.B.; Gerber, E.; Geuke, M.; Vardas, E.; Waltimo, T.; et al. A systematic review of dental disease management in cancer patients. Support. Care Cancer 2018, 26, 155–174. [Google Scholar] [CrossRef]
- Clinical Guideline “Evidence-Based Management Strategies for Oral Complication from Cancer Treatment”. Available online: http://www.isoo.world/continuing-education.html#guidelines (accessed on 22 September 2020).
- Haveman, C.W.; Summitt, J.B.; Burgess, J.O.; Carlson, K. Three restorative materials and topical fluoride gel used in xerostomic patients: A clinical comparison. J. Am. Dent. Assoc. 2003, 134, 177–184. [Google Scholar] [CrossRef]
- Thornton, J.B.; Retief, D.H.; Bradley, E.L. Fluoride release from and tensile bond strength of Ketac-Fil and Ketac-Silver to enamel and dentin. Dent. Mater. 1986, 2, 241–245. [Google Scholar] [CrossRef]
- Hicks, J.; Garcia-Godoy, F.; Donly, K.; Flaitz, C. Fluoride-releasing restorative materials and secondary caries. Dent. Clin. N. Am. 2002, 46, 247–276. [Google Scholar] [CrossRef]
- Wiegand, A.; Buchalla, W.; Attin, T. Review on fluoride-releasing restorative materials—Fluoride release and uptake characteristics, antibacterial activity and influence on caries formation. Dent. Mater. 2007, 23, 343–362. [Google Scholar] [CrossRef] [PubMed]
- Krämer, N.; Schmidt, M.; Lücker, S.; Domann, E.; Frankenberger, R. Glass ionomer cement inhibits secondary caries in an in vitro biofilm model. Clin. Oral Investig. 2018, 22, 1019–1031. [Google Scholar] [CrossRef] [PubMed]
- Horiot, J.C.; Schraub, S.; Bone, M.C.; Bain, Y.; Ramadier, J.; Chaplain, G.; Nabid, N.; Thevenot, B.; Bransfield, D. Dental preservation in patients irradiated for head and neck tumours: A 10-year experience with topical fluoride and a randomized trial between two fluoridation methods. Radiother. Oncol. 1983, 1, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Mjör, I.A. The reasons for replacement and the age of failed restorations in general dental practice. Acta Odontol. Scand. 1997, 55, 58–63. [Google Scholar] [CrossRef]
- da Cunha, S.R.; Ramos, P.A.; Haddad, C.M.; da Silva, J.L.; Fregnani, E.R.; Aranha, A.C. Effects of Different Radiation Doses on the Bond Strengths of Two Different Adhesive Systems to Enamel and Dentin. J. Adhes. Dent. 2016, 18, 151–156. [Google Scholar] [CrossRef]
- Ugurlu, M. Effect of the double application of universal adhesives on the dentine bond strength after radiotherapy. Aust. Dent. J. 2020, 65, 181–188. [Google Scholar] [CrossRef]

| 0 Gy | 5 Gy | 60 Gy | |
|---|---|---|---|
| Futurabond® U | 0 Fse 1 | 5 Fse | 60 Fse |
| AdheSE® Universal | 0 Ase | 5 Ase | 60 Ase |
| Xeno® Select | 0 Xse | 5 Xse | 60 Xse |
| Adhesive | Batch Number | pH | Main Components * | |
|---|---|---|---|---|
| F | Futurabond® U, Voco | LOT 1527116 | 2.3 | BIS-GMA 1 HEMA 2 HEDMA 3 Acidic adhesive monomer Urethanedimethacrylate Catalyst |
| A | AdheSE® Universal, Ivoclar Vivadent | LOT U54013 | 2.5–3 | HEMA 2 BIS-GMA 1 Ethanol D3MA 4 MDP 5 MCAP 6 Camphorquinone |
| X | Xeno® Select, Dentsply Sirona | LOT 1602000694 | <2 | Bifunctional acrylates Tert-butyl alcohol Functionalized phosphoric acid ester (ethyl 2-[5-dihydrogen phosphoryl-5,2-dioxapentyl]acrylate) Acidic acrylates 4-dimetylaminobenzonitril |
| Futurabond® U | AdheSE® Universal | Xeno® Select | |
|---|---|---|---|
| 0 Gy | 23.87 | 35.10 | 24.17 |
| ±7.49 | ±8.41 | ±8.36 | |
| 5 Gy | 17.95 | 26.86 | 15.78 |
| ±5.95 | ±7.41 | ±3.89 | |
| 60 Gy | 19.21 | 26.30 | 11.42 |
| ±7.34 | ±10.07 | ±3.86 | |
| Decrease in mean microtensile bond strength after irradiation with 60 Gy compared to 0 Gy | −19.5% | −25.1% | −52.8% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Broscheit, S.; Vordermark, D.; Gerlach, R.; Gernhardt, C.R. Impact of Preceded Tumor Therapeutic Irradiation on the Microtensile Bond Strength of Universal Adhesives Applied in Self-Etch Mode to Human Dentin In Vitro. Appl. Sci. 2023, 13, 7873. https://doi.org/10.3390/app13137873
Broscheit S, Vordermark D, Gerlach R, Gernhardt CR. Impact of Preceded Tumor Therapeutic Irradiation on the Microtensile Bond Strength of Universal Adhesives Applied in Self-Etch Mode to Human Dentin In Vitro. Applied Sciences. 2023; 13(13):7873. https://doi.org/10.3390/app13137873
Chicago/Turabian StyleBroscheit, Sina, Dirk Vordermark, Reinhard Gerlach, and Christian Ralf Gernhardt. 2023. "Impact of Preceded Tumor Therapeutic Irradiation on the Microtensile Bond Strength of Universal Adhesives Applied in Self-Etch Mode to Human Dentin In Vitro" Applied Sciences 13, no. 13: 7873. https://doi.org/10.3390/app13137873
APA StyleBroscheit, S., Vordermark, D., Gerlach, R., & Gernhardt, C. R. (2023). Impact of Preceded Tumor Therapeutic Irradiation on the Microtensile Bond Strength of Universal Adhesives Applied in Self-Etch Mode to Human Dentin In Vitro. Applied Sciences, 13(13), 7873. https://doi.org/10.3390/app13137873







