Cutaneous Fibrous Histiocytomas, Ossifying-Variant with Osteoclast-like Giant Cells and Granular Cell-Variant: A Series of Two Unusual Cases with Emphasis on the Differential Diagnosis
Abstract
:1. Introduction
2. Materials and Methods
3. Results
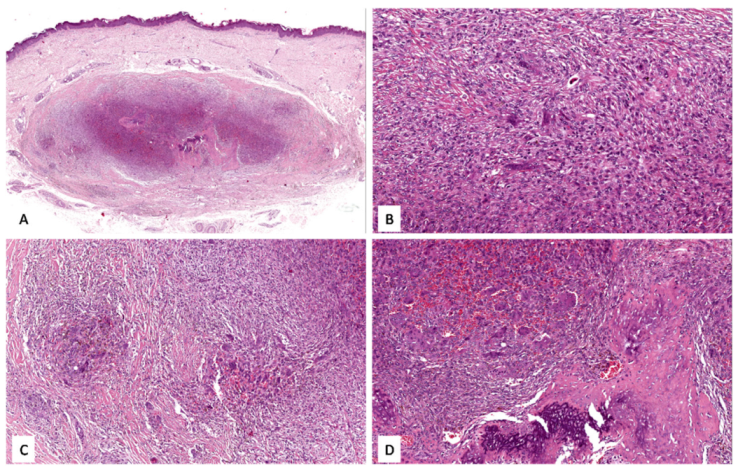
3.1. Case No. 1
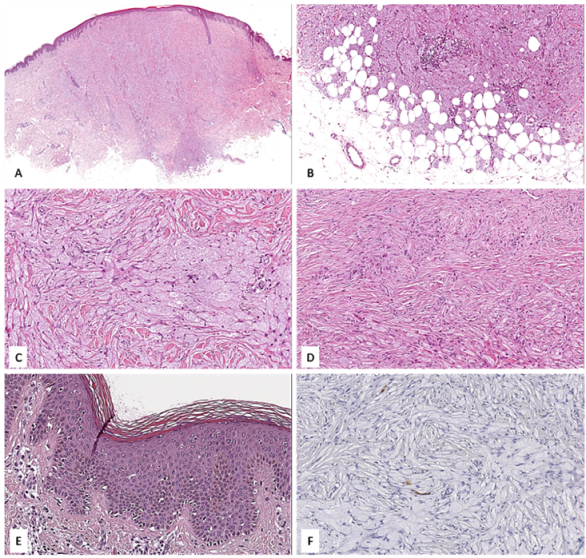
3.2. Case No. 2
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Papalas, J.A.; Balmer, N.N.; Wallace, C.; Sangüeza, O.P. Ossifying Dermatofibroma with Osteoclast-like Giant Cells: Report of a Case and Literature Review. Am. J. Dermatopathol. 2009, 31, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Abramovits, W.; Graham, G.; Har-Shai, Y.; Strumia, R. Dermatological Cryosurgery and Cryotherapy; Springer: London, UK, 2016; pp. 365–366. ISBN 978-1-4471-6765-5. [Google Scholar]
- Zaballos, P.; Puig, S.; Llambrich, A.; Malvehy, J. Dermoscopy of Dermatofibromas: A Prospective Morphological Study of 412 Cases. Arch. Dermatol. 2008, 144, 75–83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zelger, B.; Zelger, B.G.; Burgdorf, W.H. Dermatofibroma-a Critical Evaluation. Int. J. Surg. Pathol. 2004, 12, 333–344. [Google Scholar] [CrossRef] [PubMed]
- Hamie, L.; Abbas, O.; Kurban, M.; Bhawan, J. Osteoclast-Like Giant Cells: Focus on Entities Relevant to Dermatopathology and Underlying Pathogenesis. Am. J. Dermatopathol. 2021, 43, 163–173. [Google Scholar] [CrossRef]
- Urdiales-Viedma, M.; Santos Sánchez, E.; López-Urdiales, R.; Martos-Padilla, S. Metaplasia Ósea En Dermatofibroma Cutáneo Osseous Metaplasia in Cutaneous Dermatofibroma. Med. Cutánea Ibero-Lat.-Am. 2007, 35, 233–235. [Google Scholar]
- Kuo, T.-T.; Chan, H.L. Ossifying Dermatofibroma with Osteoclast-like Giant Cells. Am. J. Dermatopathol. 1994, 16, 193–195. [Google Scholar] [CrossRef]
- Kutchemeshgi, M.; Barr, R.J.; Henderson, C.D. Dermatofibroma with Osteoclast-like Giant Cells. Am. J. Dermatopathol. 1992, 14, 397–401. [Google Scholar] [CrossRef]
- Buselmeier, T.J.; Uecker, J.H. Invasive Basal Cell Carcinoma with Metaplastic Bone Formation Associated with a Long-Standing Dermatofibroma. J. Cutan. Pathol. 1979, 6, 496–500. [Google Scholar] [CrossRef]
- González-Vilas, D.; García-Gavín, J.; Ginarte, M.; Rodríguez-Blanco, I.; Toribio, J. Ulcerated Dermatofibroma with Osteoclast-like Giant Cells. J. Cutan. Pathol. 2009, 36 (Suppl. 1), 16–19. [Google Scholar] [CrossRef]
- Lucas, D.; Tazelaar, H.; Unni, K.; Wold, L.; Okada, K.; Dimarzio, D.; Rolfe, B. Osteogenic melanoma. A rare variant of malignant melanoma. Am. J. Surg. Pathol. 1993, 17, 400–409. [Google Scholar] [CrossRef]
- Kuo, T. Primary Osteosarcoma of the Skin. J. Cutan. Pathol. 1992, 19, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Soukup, J.; Hadzi-Nikolov, D.; Ryska, A. Dermatofibroma-like Granular Cell Tumour: A Potential Diagnostic Pitfall. Pol. J. Pathol. 2016, 67, 291–294. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perret, R.E.; Jullie, M.L.; Vergier, B.; Coindre, J.M.; Le Loarer, F. A Subset of So-Called Dermal Non-Neural Granular Cell Tumours Are Underlined by ALK Fusions, Further Supporting the Idea That They Represent a Variant of Epithelioid Fibrous Histiocytoma. Histopathology 2018, 73, 532–534. [Google Scholar] [CrossRef] [PubMed]
- Cazzato, G.; Colagrande, A.; Cimmino, A.; Marrone, M.; Stellacci, A.; Arezzo, F.; Lettini, T.; Resta, L.; Ingravallo, G. Granular Cell Dermatofibroma: When Morphology Still Matters. Dermatopathology (Basel Switz.) 2021, 8, 371–375. [Google Scholar] [CrossRef]
- Aloi, F.; Albertazzi, D.; Pippione, M. Dermatofibroma with Granular Cells: A Report of Two Cases. Dermatology 1999, 199, 54–56. [Google Scholar] [CrossRef]
- Val-Bernal, J.F.; Mira, C. Dermatofibroma with Granular Cells. J. Cutan. Pathol. 1996, 23, 562–565. [Google Scholar] [CrossRef]
- Soyer, H.P.; Metze, D.; Kerl, H. Granular Cell Dermatofibroma. Am. J. Dermatopathol. 1997, 19, 168–173. [Google Scholar] [CrossRef]
- Zelger, B.G.; Steiner, H.; Kutzner, H.; Rütten, A.; Zelger, B. Granular Cell Dermatofibroma. Histopathology 1997, 31, 258–262. [Google Scholar] [CrossRef]
- Sanz-Trelles, A.; Weil-Lara, B.; Acedo-Rodriguez, C. Dermatofibroma with Granular Cells. Histopathology 1997, 30, 495–497. [Google Scholar] [CrossRef]
- Helm, T.N.; Kalb, R.E.; Helm, K.F. Granular Cell Dermatofibroma: A Benign Tumor That Can Simulate Malignancy. Dermatol. Surg. 1997, 23, 976–978. [Google Scholar] [CrossRef]
- Hong, S.B.; Yang, M.H.; Lee, M.H.; Haw, C.R. Dermatofibroma-like Atypical Granular Cell Tumour. Acta Derm. Venereol. 2005, 85, 179–180. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, S.D.; Usmani, A.S.; DeYoung, B.R.; Ly, M.; Pellegrini, A.E. Dermatofibroma-like Granular Cell Tumor. J. Cutan. Pathol. 2001, 28, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Zelger, B.G.; Sidoroff, A.; Zelger, B. Combined dermatofibroma: Co-existence of two or more variant patterns in a single lesion. Histopathology 2000, 36, 529–539. [Google Scholar] [CrossRef] [PubMed]
- Pogorzelska-Antkowiak, A.; Wcisło-Dziadecka, D.; Brzezińska-Wcisło, L.; Pawlicki, K.; Antkowiak, R.; Corneli, P. Features of dermatofibroma in reflectance confocal microscopy. Int. J. Dermatol. 2020, 59, 951–954. [Google Scholar] [CrossRef] [PubMed]
- Mazzella, C.; Villani, A.; Scalvenzi, M.; Fabbrocini, G.; Cantelli, M.; Costa, C. How reflectance confocal microscopy can help diagnose an enigmatic variant of dermatofibroma. Dermatol. Ther. 2019, 32, e12857. [Google Scholar] [CrossRef] [PubMed]
- Lacarrubba, F.; Verzì, A.E.; Caltabiano, R.; Broggi, G.; Di Natale, A.; Micali, G. Discoid Lupus Erythematosus: Reflectance Confocal Microscopy Features Correlate with Horizontal Histopathological Sections. Skin Res. Technol. 2019, 25, 242–244. [Google Scholar] [CrossRef]
- Caccavale, S.; Vitiello, P.; Agozzino, M.; Mascolo, M.; Ciancia, G.; Argenziano, G. Dermoscopy and reflectance confocal microscopy of osteoclastic deep benign fibrous histiocytoma. G. Ital. Dermatol. Venereol. 2019, 154, 363–364. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Farina, J.; Broggi, G.; Caltabiano, R. Cutaneous Fibrous Histiocytomas, Ossifying-Variant with Osteoclast-like Giant Cells and Granular Cell-Variant: A Series of Two Unusual Cases with Emphasis on the Differential Diagnosis. Appl. Sci. 2022, 12, 2977. https://doi.org/10.3390/app12062977
Farina J, Broggi G, Caltabiano R. Cutaneous Fibrous Histiocytomas, Ossifying-Variant with Osteoclast-like Giant Cells and Granular Cell-Variant: A Series of Two Unusual Cases with Emphasis on the Differential Diagnosis. Applied Sciences. 2022; 12(6):2977. https://doi.org/10.3390/app12062977
Chicago/Turabian StyleFarina, Jessica, Giuseppe Broggi, and Rosario Caltabiano. 2022. "Cutaneous Fibrous Histiocytomas, Ossifying-Variant with Osteoclast-like Giant Cells and Granular Cell-Variant: A Series of Two Unusual Cases with Emphasis on the Differential Diagnosis" Applied Sciences 12, no. 6: 2977. https://doi.org/10.3390/app12062977
APA StyleFarina, J., Broggi, G., & Caltabiano, R. (2022). Cutaneous Fibrous Histiocytomas, Ossifying-Variant with Osteoclast-like Giant Cells and Granular Cell-Variant: A Series of Two Unusual Cases with Emphasis on the Differential Diagnosis. Applied Sciences, 12(6), 2977. https://doi.org/10.3390/app12062977







