Combustion and Explosion Characteristics of Pulverised Wood, Valorized with Mild Pyrolysis in Pilot Scale Installation, Using the Modified ISO 1 m3 Dust Explosion Vessel
Abstract
Featured Application
Abstract
1. Introduction
2. Materials and Methods
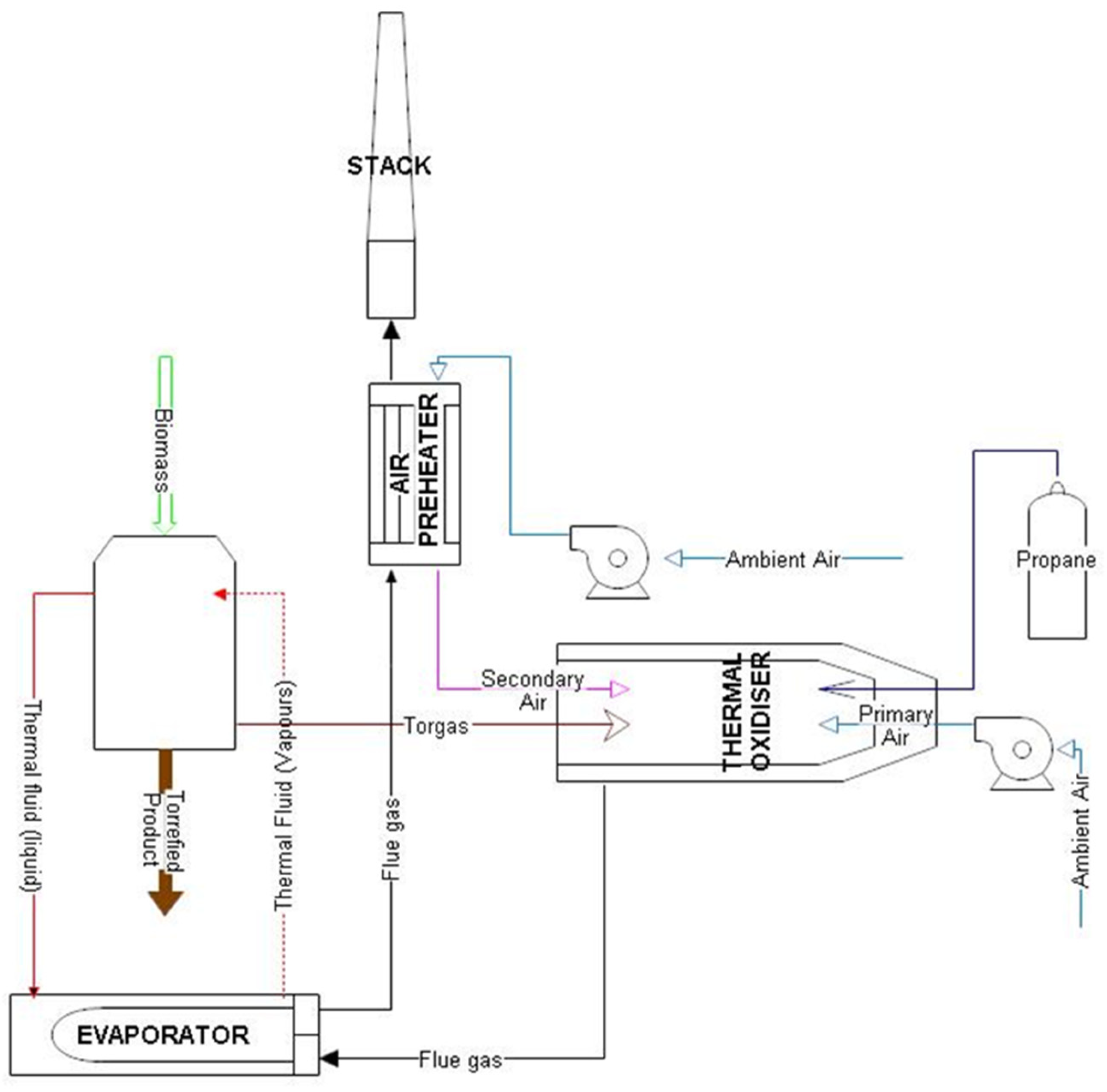
2.1. Mild Pyrolysis Process
2.2. Characterization of Feedstock and Product
2.3. Particle Size Distribution
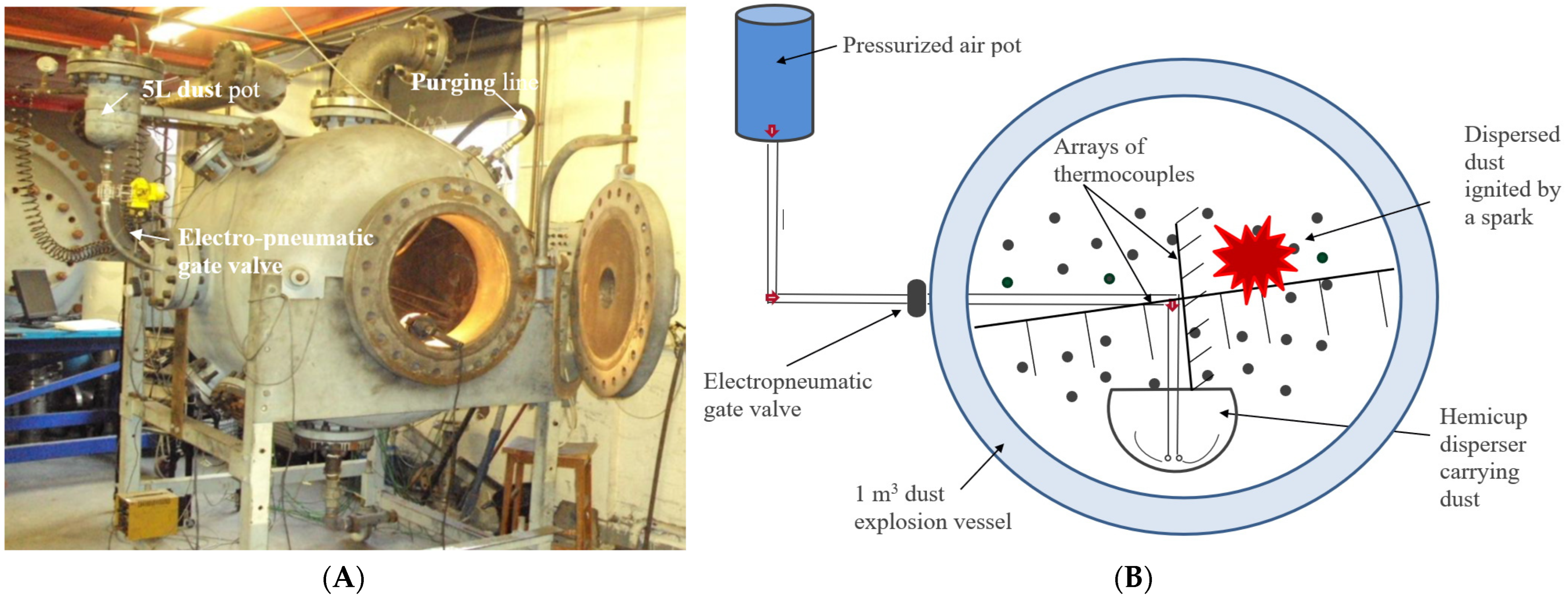
2.4. The Modified ISO 1 m3 Dust Explosion Vessel for Coarse Biomass Powders
2.5. The Measurement of Spherical Flame Speed and Burning Velocity
2.6. The Explosion Residue and Implications for the Flame Front Equivalence Ratio, Ø
3. Results and Discussion
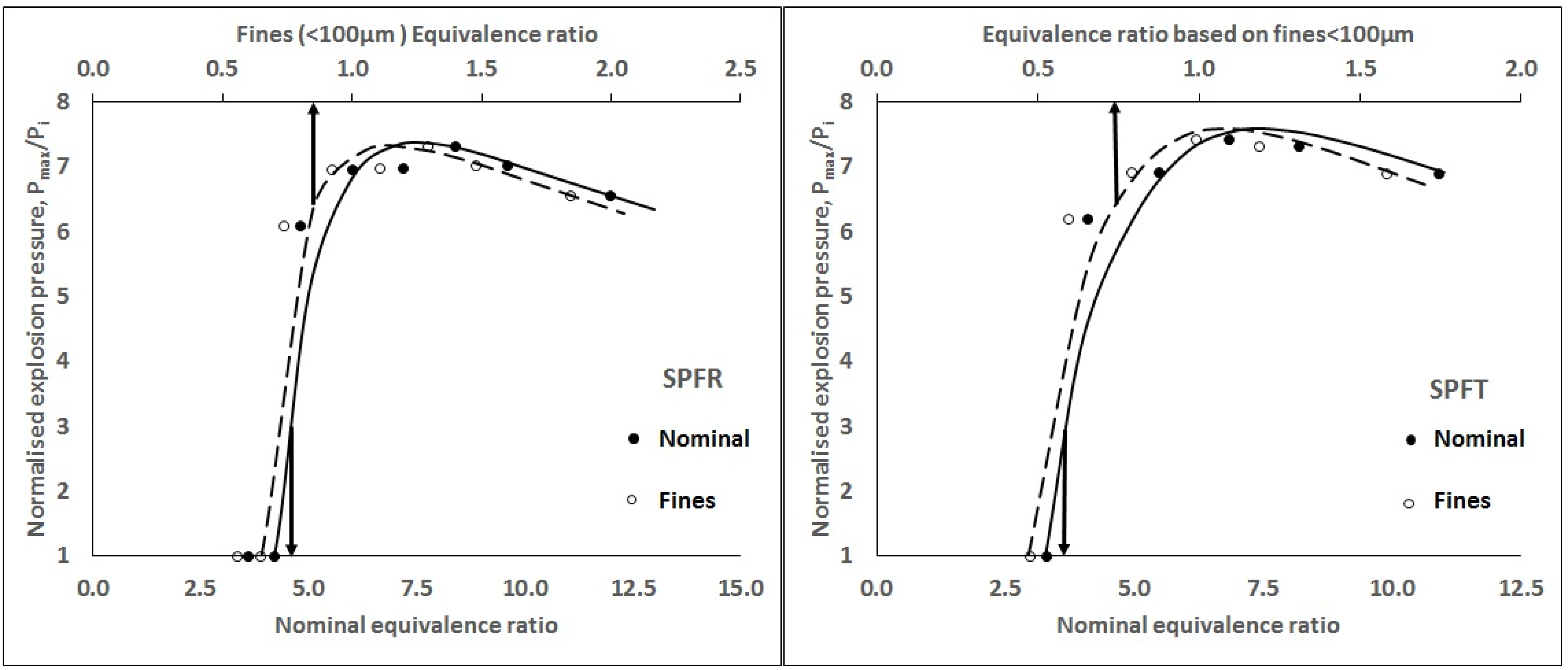
3.1. Fraction of Original Mass That Burns in the Explosion
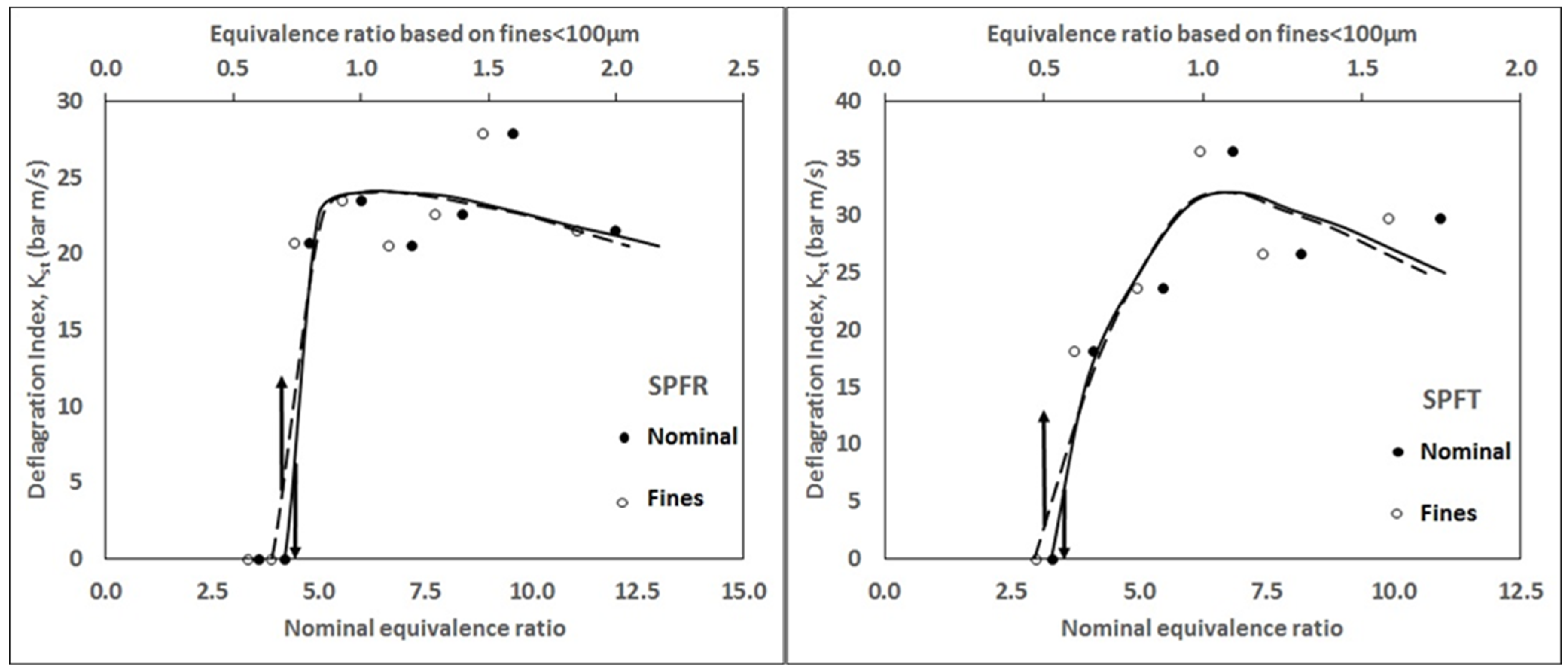
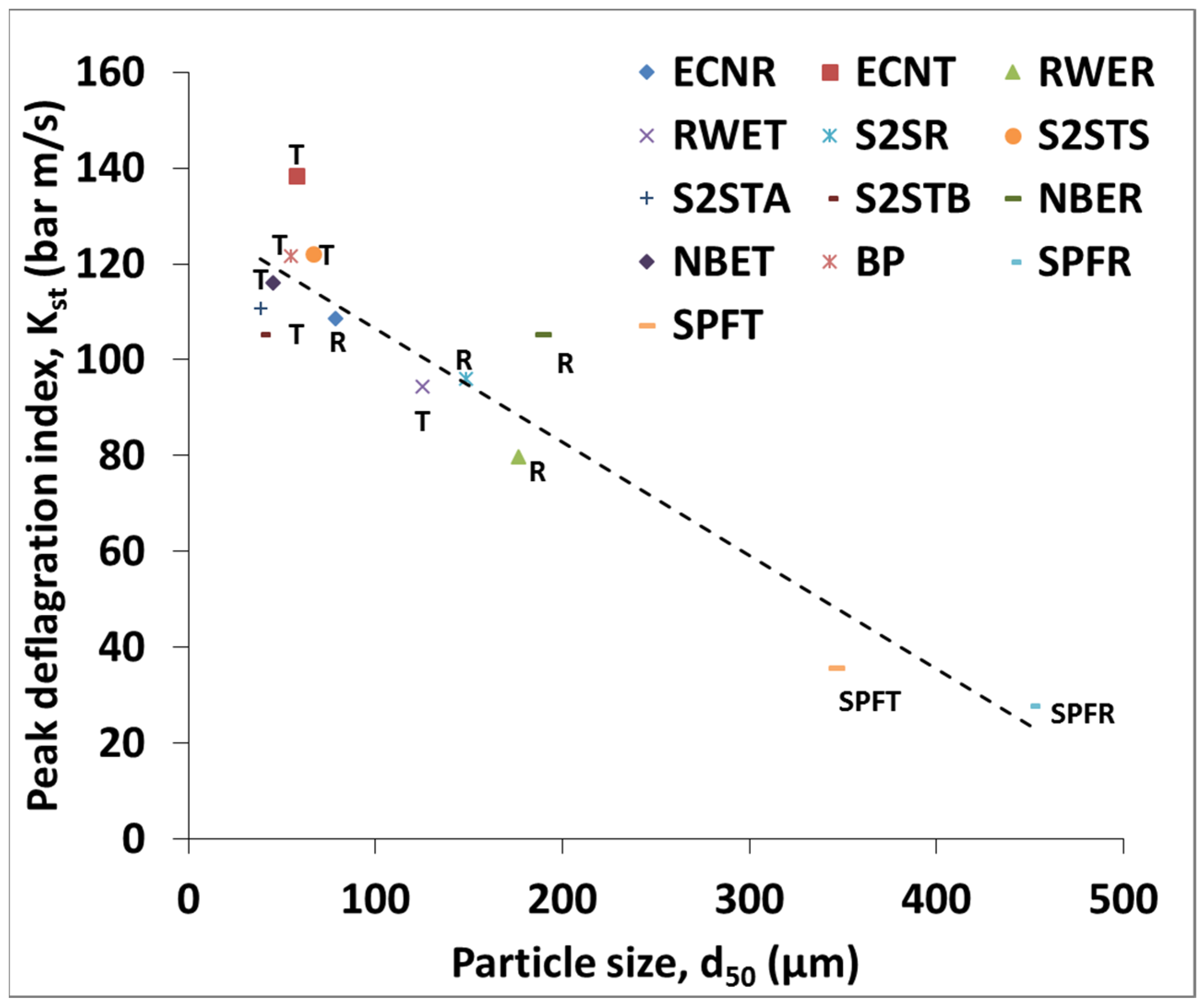
3.2. Deflagration Index
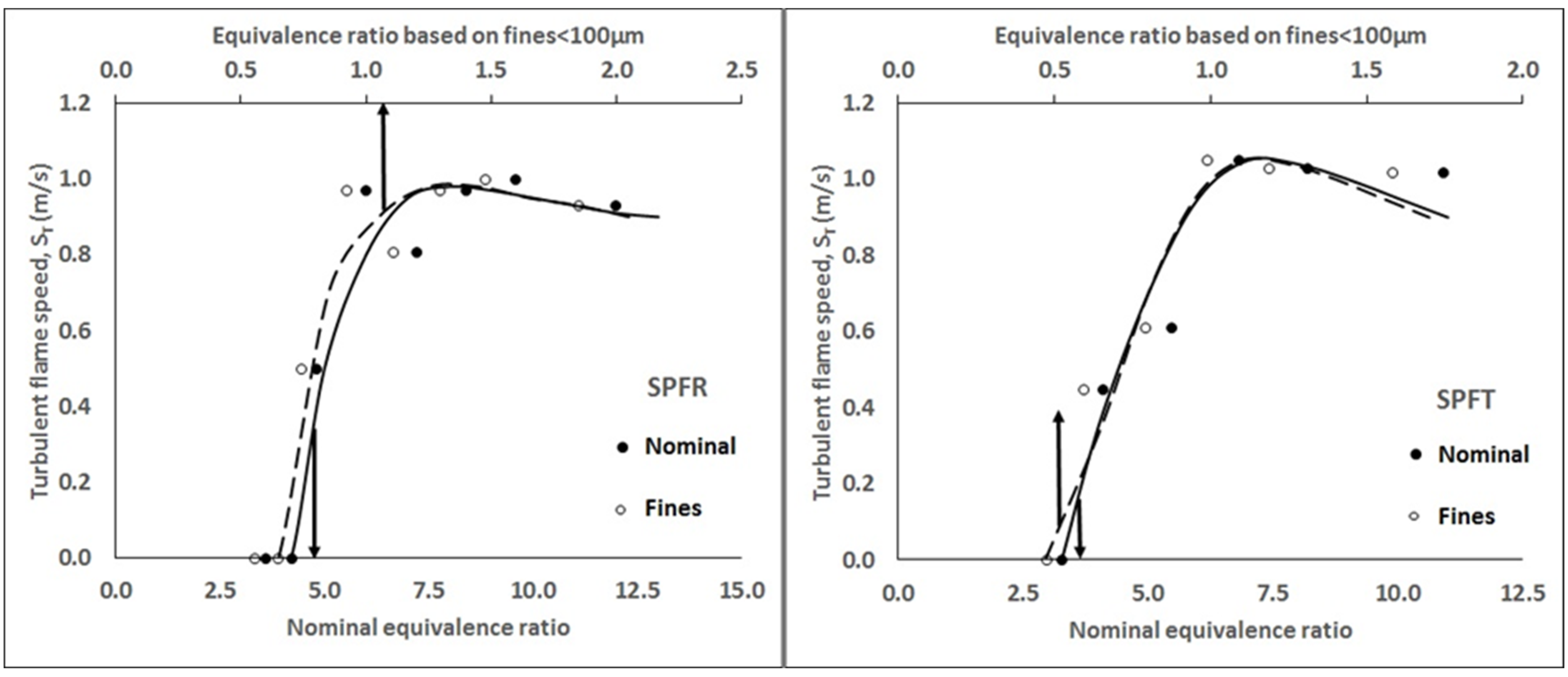
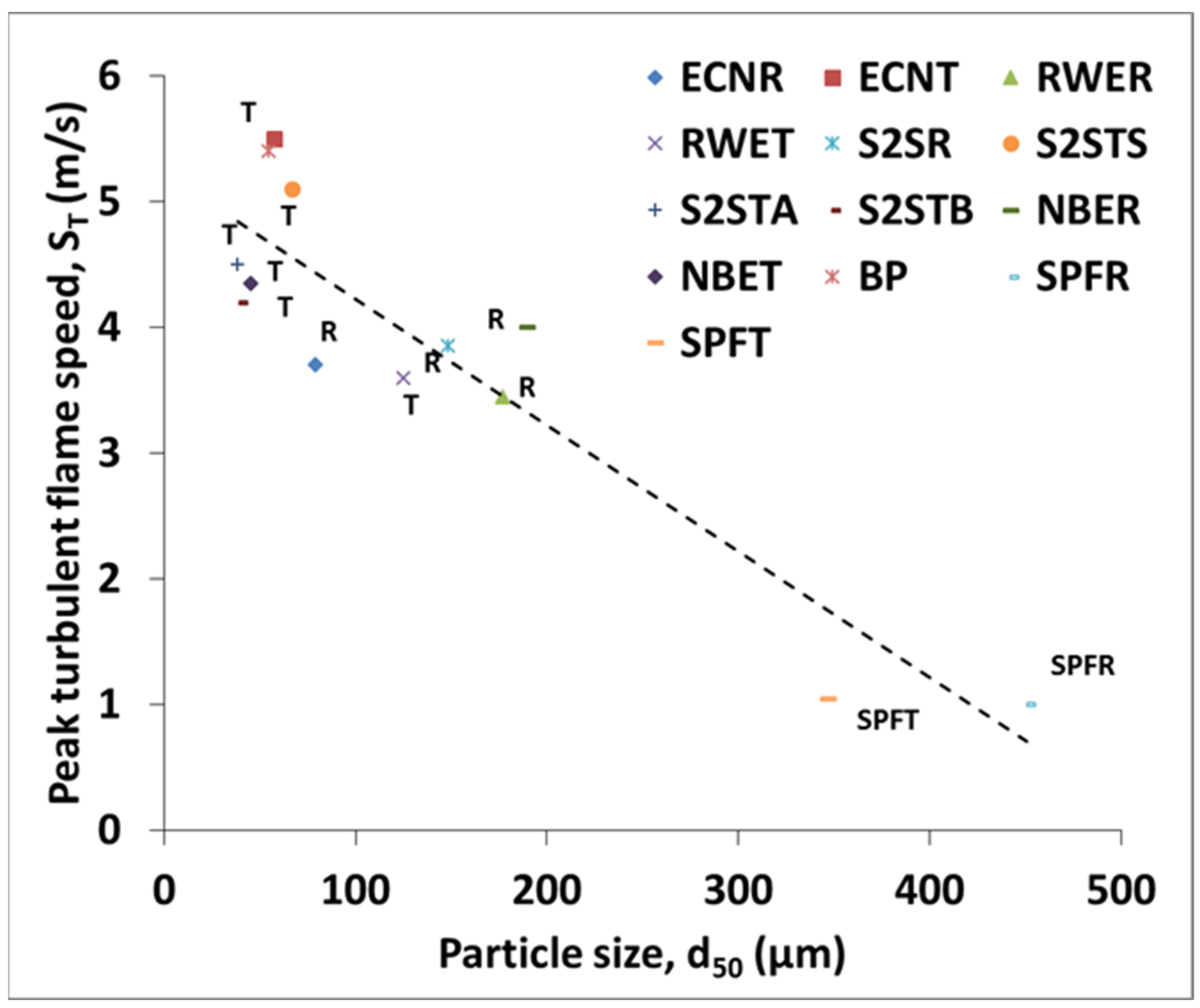
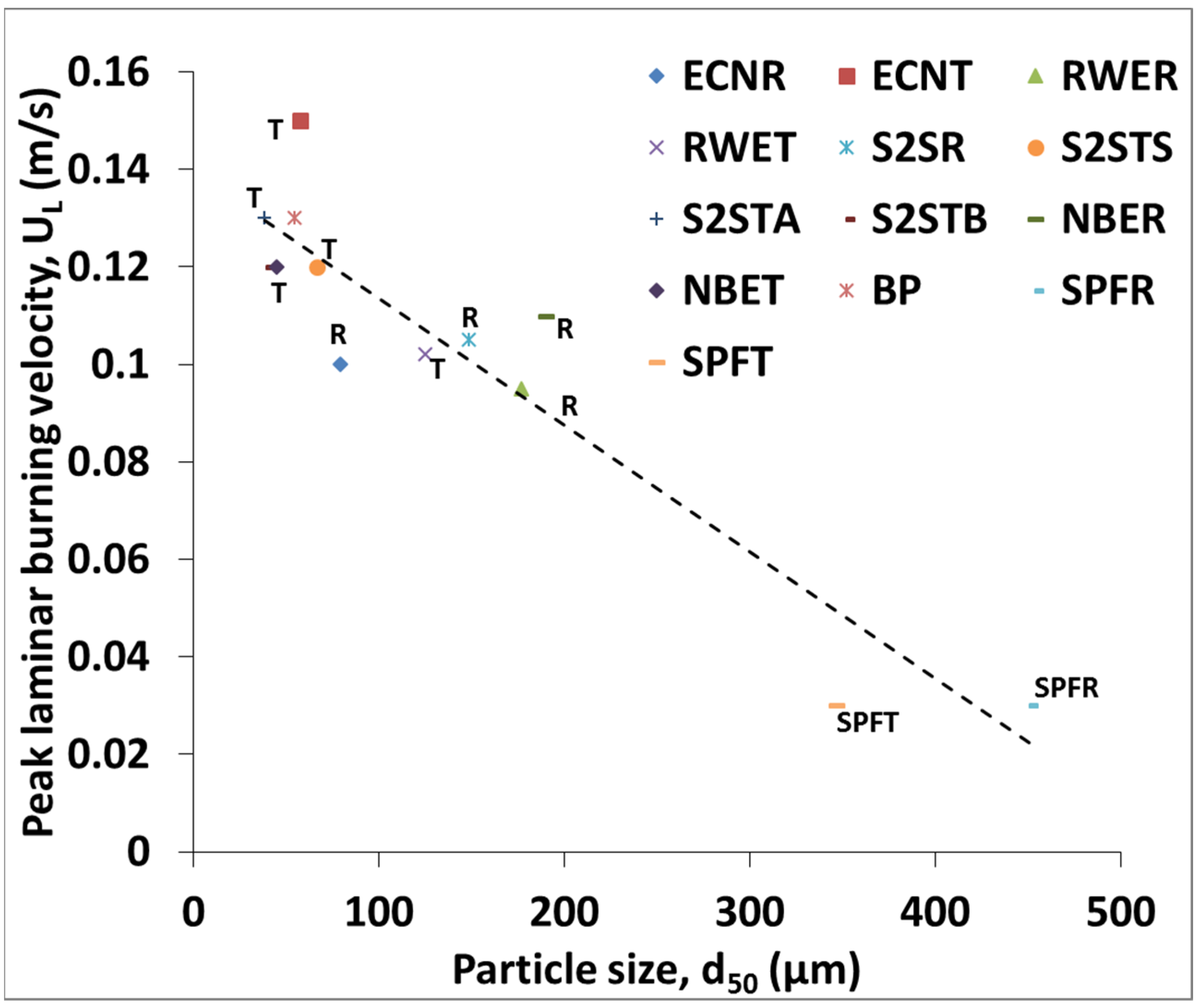
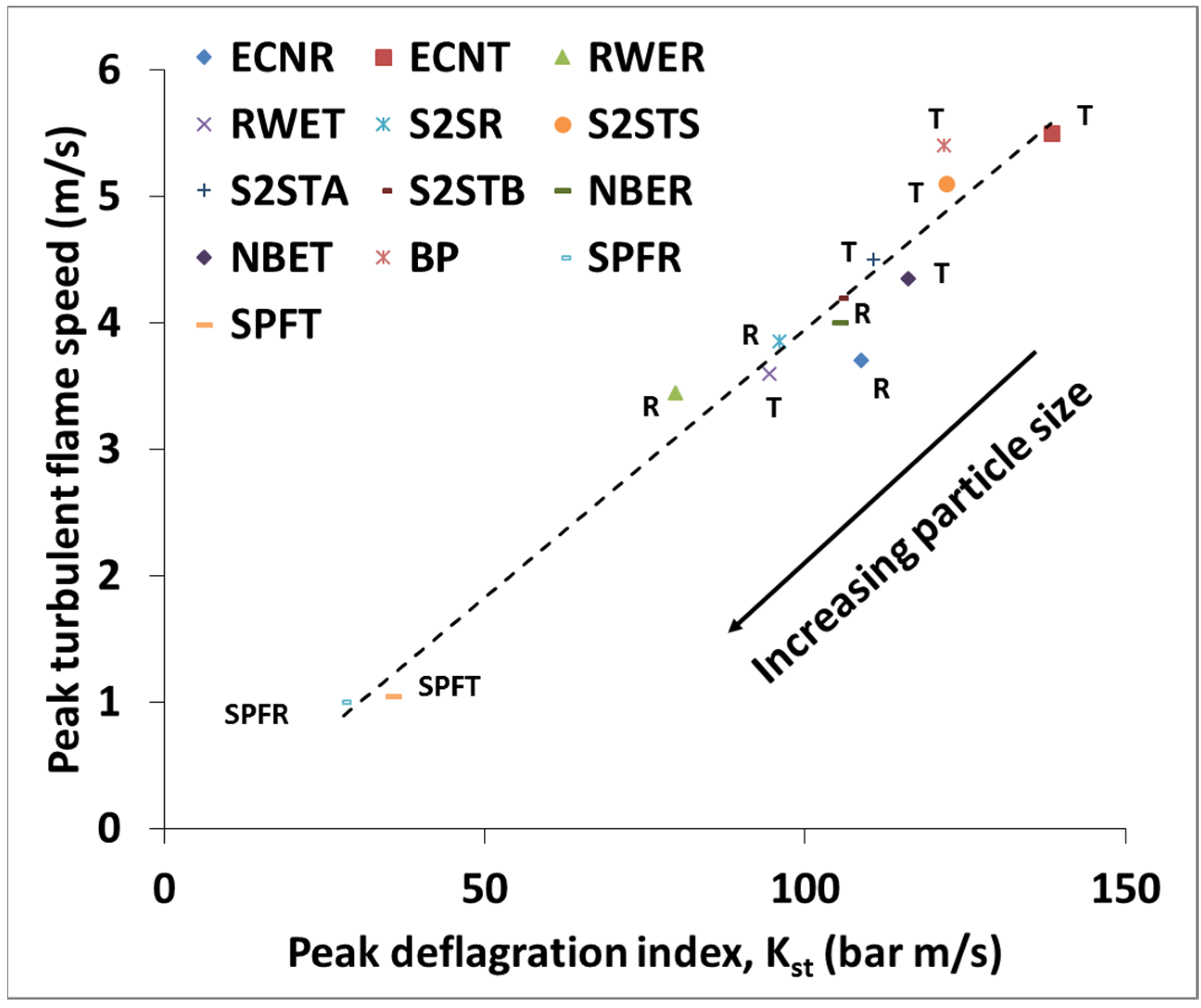
3.3. Flame Speed and Burning Velocity Measurements
3.4. Minimum Explosible Concentration, MEC
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tzelepi, V.; Zeneli, M.; Kourkoumpas, D.S.; Karampinis, E.; Gypakis, A.; Nikolopoulos, N.; Grammelis, P. Biomass Availability in Europe as an Alternative Fuel for Full Conversion of Lignite Power Plants: A Critical Review. Energies 2020, 13, 3390. [Google Scholar] [CrossRef]
- Mračková, E.; Schmidtová, J.; Marková, I.; Jad’ud’ová, J.; Tureková, I.; Hitka, M. Fire Parameters of Spruce (Picea Abies Karst. (L.)) Dust Layer from Different Wood Technologies Slovak Case Study. Appl. Sci. 2022, 12, 548. [Google Scholar] [CrossRef]
- Portarapillo, M.; Danzi, E.; Sanchirico, R.; Marmo, L.; Di Benedetto, A. Energy Recovery from Vinery Waste: Dust Explosion Issues. Appl. Sci. 2021, 11, 11188. [Google Scholar] [CrossRef]
- Tureková, I.; Marková, I. Ignition of Deposited Wood Dust Layer by Selected Sources. Appl. Sci. 2020, 10, 5779. [Google Scholar] [CrossRef]
- Eckhoff, R.K.; Li, G. Industrial Dust Explosions. A Brief Review. Appl. Sci. 2021, 11, 1669. [Google Scholar] [CrossRef]
- Garrido-Ceca, I.; Puig-Gamero, M.; Ramírez-Gómez, Á. Influence of Bends in the Functionality of Passive Explosion Isolation Valves. Appl. Sci. 2022, 12, 11654. [Google Scholar] [CrossRef]
- Knapczyk, A.; Francik, S.; Jewiarz, M.; Zawiślak, A.; Francik, R. Thermal Treatment of Biomass: A Bibliometric Analysis—The Torrefaction Case. Energies 2020, 14, 162. [Google Scholar] [CrossRef]
- Kantorek, M.; Jesionek, K.; Polesek-Karczewska, S.; Ziółkowski, P.; Stajnke, M.; Badur, J. Thermal Utilization of Meat-and-Bone Meal Using the Rotary Kiln Pyrolyzer and the Fluidized Bed Boiler—The Performance of Pilot-Scale Installation. Renew. Energy 2021, 164, 1447–1456. [Google Scholar] [CrossRef]
- Krochmalny, K.; Niedzwiecki, L.; Pelińska-Olko, E.; Wnukowski, M.; Czajka, K.; Tkaczuk-Serafin, M.; Pawlak-Kruczek, H. Determination of the Marker for Automation of Torrefaction and Slow Pyrolysis Processes—A Case Study of Spherical Wood Particles. Renew. Energy 2020, 161, 350–360. [Google Scholar] [CrossRef]
- Kerdsuwan, S.; Laohalidanond, K.; Gupta Ashwani, K. Upgrading Refuse-Derived Fuel Properties From Reclaimed Landfill Using Torrefaction. J. Energy Resour. Technol. 2021, 143, 021302. [Google Scholar] [CrossRef]
- Rasheed, T.; Anwar, M.T.; Ahmad, N.; Sher, F.; Khan, S.U.-D.; Ahmad, A.; Khan, R.; Wazeer, I. Valorisation and emerging perspective of biomass based waste-to-energy technologies and their socio-environmental impact: A review. J. Environ. Manag. 2021, 287, 112257. [Google Scholar] [CrossRef]
- Nunes, L.J.R.; Loureiro, L.M.E.F.; Sá, L.C.R.; Silva, H.F.C. Sugarcane Industry Waste Recovery: A Case Study Using Thermochemical Conversion Technologies to Increase Sustainability. Appl. Sci. 2020, 10, 6481. [Google Scholar] [CrossRef]
- Szwaja, S.; Magdziarz, A.; Zajemska, M.; Poskart, A. A Torrefaction of Sida Hermaphrodita to Improve Fuel Properties. Advanced Analysis of Torrefied Products. Renew. Energy 2019, 141, 894–902. [Google Scholar] [CrossRef]
- Sh, L.; Lee, B.H.; Lee, Y.J.; Jeon, C.H. Comparing the Physicochemical Properties of Upgraded Biomass Fuel by Torrefaction and the Ashless Technique. Appl. Sci. 2019, 9, 5519. [Google Scholar] [CrossRef]
- Luo, H.; Lu, Z.; Jensen, P.A.; Glarborg, P.; Lin, W.; Dam-Johansen, K.; Wu, H. Experimental and Modelling Study on the Influence of Wood Type, Density, Water Content, and Temperature on Wood Devolatilization. Fuel 2020, 260, 116410. [Google Scholar] [CrossRef]
- Lu, Z.; Jian, J.; Jensen, P.A.; Wu, H.; Glarborg, P. Influence of Torrefaction on Single Particle Combustion of Wood. Energy Fuels 2016, 30, 5772–5778. [Google Scholar] [CrossRef]
- Luo, H.; Lu, Z.; Jensen, P.A.; Glarborg, P.; Lin, W.; Dam-Johansen, K.; Wu, H. Effect of Gasification Reactions on Biomass Char Conversion under Pulverized Fuel Combustion Conditions. Proc. Combust. Inst. 2020, 38, 3919–3928. [Google Scholar] [CrossRef]
- Jaworski, T.J.; Kajda-Szcześniak, M. Study on the Similarity of the Parameters of Biomass and Solid Waste Fuel Combustion for the Needs of Thermal Power Engineering. Sustainability 2020, 12, 7894. [Google Scholar] [CrossRef]
- Pedroso, D.T.; Machin, E.B.; Cabrera-Barjas, G.; Flores, M.; Urra, H.G.; De Carvalho, F.S.; Silva Dos Santos, M.I.; Machín, A.B.; Canettieri, E.V.; Pérez, N.P.; et al. Sugarcane Bagasse Torrefaction for Fluidized Bed Gasification. Appl. Sci. 2021, 11, 6105. [Google Scholar] [CrossRef]
- Lucantonio, S.; Di Giuliano, A.; Gallucci, K. Influences of the Pretreatments of Residual Biomass on Gasification Processes: Experimental Devolatilizations Study in a Fluidized Bed. Appl. Sci. 2021, 11, 5722. [Google Scholar] [CrossRef]
- Čespiva, J.; Niedzwiecki, L.; Wnukowski, M.; Krochmalny, K.; Mularski, J.; Ochodek, T.; Pawlak-Kruczek, H. Torrefaction and Gasification of Biomass for Polygeneration: Production of Biochar and Producer Gas at Low Load Conditions. Energy Rep. 2022, 8, 134–144. [Google Scholar] [CrossRef]
- Sher, F.; Iqbal, S.Z.; Liu, H.; Imran, M.; Snape, C.E. Thermal and Kinetic Analysis of Diverse Biomass Fuels under Different Reaction Environment: A Way Forward to Renewable Energy Sources. Energy Convers. Manag. 2020, 203, 112266. [Google Scholar] [CrossRef]
- Wang, T.; Tang, L.; Raheem, A.; Chen, X.; Wang, F. Study on CO2 Gasification Characteristics of Pyrolysis Char from Pinewood Block and Pellet. Biomass Convers. Biorefin. 2021. [Google Scholar] [CrossRef]
- Zhang, S.; Su, Y.; Xiong, Y.; Zhang, H. Physicochemical Structure and Reactivity of Char from Torrefied Rice Husk: Effects of Inorganic Species and Torrefaction Temperature. Fuel 2020, 262, 116667. [Google Scholar] [CrossRef]
- Li, L.; Huang, Y.; Zhang, D.; Zheng, A.; Zhao, Z.; Xia, M.; Li, H. Uncovering Structure-Reactivity Relationships in Pyrolysis and Gasification of Biomass with Varying Severity of Torrefaction. ACS Sustain. Chem. Eng. 2018, 6, 6008–6017. [Google Scholar] [CrossRef]
- Junga, R.; Pospolita, J.; Niemiec, P. Combustion and Grindability Characteristics of Palm Kernel Shells Torrefied in a Pilot-Scale Installation. Renew. Energy 2020, 147, 1239–1250. [Google Scholar] [CrossRef]
- Sher, F.; Yaqoob, A.; Saeed, F.; Zhang, S.; Jahan, Z.; Klemeš, J.J. Torrefied Biomass Fuels as a Renewable Alternative to Coal in Co-Firing for Power Generation. Energy 2020, 209, 118444. [Google Scholar] [CrossRef]
- Khalsa, J.H.A.; Leistner, D.; Weller, N.; Darvell, L.I.; Dooley, B. Torrefied Biomass Pellets—Comparing Grindability in Different Laboratory Mills. Energies 2016, 9, 794. [Google Scholar] [CrossRef]
- Castells, B.; Amez, I.; Medic, L.; Torrent, J.G. Particle Size Influence on the Transport Classification Labels and Other Flammability Characteristics of Powders. Appl. Sci. 2020, 10, 8601. [Google Scholar] [CrossRef]
- Saeed, M.A.; Medina, C.H.; Andrews, G.E.; Phylaktou, H.N.; Slatter, D.; Gibbs, B.M. Agricultural Waste Pulverised Biomass: MEC and Flame Speeds. J. Loss Prev. Process Ind. 2014, 36, 308–317. [Google Scholar] [CrossRef]
- Saeed, M.A.; Anez, N.F.; Andrews, G.E.; Phylaktou, H.N.; Gibbs, B.M. Steam Exploded Pine Wood Burning Properties with Particle Size Dependence. Fuel 2017, 194, 527–532. [Google Scholar] [CrossRef]
- Szabová, Z.; Kuracina, R.; Sahul, M.; Mynarz, M.; Lepík, P.; Kosár, L. Influence of the Pyrotechnic Igniter Composition Aging on Explosion Parameters of Dispersed Dusts. Appl. Sci. 2021, 11, 10728. [Google Scholar] [CrossRef]
- Jagodzińska, K.; Czerep, M.; Kudlek, E.; Wnukowski, M.; Yang, W. Torrefaction of Wheat-Barley Straw: Composition and Toxicity of Torrefaction Condensates. Biomass Bioenergy 2019, 129, 105335. [Google Scholar] [CrossRef]
- Hardy, T.; Arora, A.; Pawlak-Kruczek, H.; Rafajłowicz, W.; Wietrzych, J.; Niedźwiecki, Ł.; Vishwajeet; Mościcki, K. Non-Destructive Diagnostic Methods for Fire-Side Corrosion Risk Assessment of Industrial Scale Boilers, Burning Low Quality Solid Biofuels—A Mini Review. Energies 2021, 14, 7132. [Google Scholar] [CrossRef]
- Pawlak-Kruczek, H.; Arora, A.; Mościcki, K.; Krochmalny, K.; Sharma, S.; Niedzwiecki, L. A Transition of a Domestic Boiler from Coal to Biomass—Emissions from Combustion of Raw and Torrefied Palm Kernel Shells (PKS). Fuel 2020, 263, 116718. [Google Scholar] [CrossRef]
- Szufa, S.; Piersa, P.; Junga, R.; Błaszczuk, A.; Modliński, N.; Sobek, S.; Marczak-Grzesik, M.; Adrian, Ł.; Dzikuć, M. Numerical Modeling of the Co-Firing Process of an in Situ Steam-Torrefied Biomass with Coal in a 230 MW Industrial-Scale Boiler. Energy 2023, 263, 125918. [Google Scholar] [CrossRef]
- Li, J.; Zhang, X.; Pawlak-Kruczek, H.; Yang, W.; Kruczek, P.; Blasiak, W. Process Simulation of Co-Firing Torrefied Biomass in a 220 MWe Coal-Fired Power Plant. Energy Convers. Manag. 2014, 84, 503–511. [Google Scholar] [CrossRef]
- Huéscar Medina, C.; Sattar, H.; Phylaktou, H.N.; Andrews, G.E.; Gibbs, B.M. Explosion Reactivity Characterisation of Pulverised Torrefied Spruce Wood. J. Loss Prev. Process Ind. 2015, 36, 287–295. [Google Scholar] [CrossRef]
- Saeed, M.A.; Farooq, M.; Anwar, A.; Abbas, M.M.; Soudagar, M.E.M.; Siddiqui, F.A.; Shakir, M.A.; Nizami, A.S.; Chaudhry, I.A.; Pettinau, A.; et al. Flame propagation and burning characteristics of pulverized biomass for sustainable biofuel. Biomass Convers. Biorefin. 2021, 11, 409–417. [Google Scholar] [CrossRef]
- Abelha, P.; Carbo, M.; Cieplik, M. Explosivity Properties of Dusts from Torrefied Biomass Pellets. Chem. Eng. Trans. 2016, 48, 403–408. [Google Scholar] [CrossRef]
- Saeed, M.A.; Slatter, D.M.; Andrews, G.E.; Phylaktou, H.N.; Gibbs, B.M. The Burning Velocity of Pulverised Biomass: The Influence of Particle Size. Chem. Eng. Trans. 2016, 53, 31–36. [Google Scholar] [CrossRef]
- Saeed, M.A.; Andrews, G.E.; Phylaktou, H.N.; Gibbs, B.M. Flame Speed and Kst Reactivity Data for Pulverised Corn Cobs and Peanut Shells. J. Loss Prev. Process Ind. 2017, 49, 880–887. [Google Scholar] [CrossRef]
- Huéscar Medina, C.; Maccoitir, B.; Sattar, H.; Slatter, D.J.F.; Phylaktou, H.N.; Andrews, G.E.; Gibbs, B.M. Comparison of the Explosion Characteristics and Flame Speeds of Pulverised Coals and Biomass in the ISO Standard 1 M3 Dust Explosion Equipment. Fuel 2015, 151, 91–101. [Google Scholar] [CrossRef]
- Piersa, P.; Unyay, H.; Szufa, S.; Lewandowska, W.; Modrzewski, R.; Ślężak, R.; Ledakowicz, S. An Extensive Review and Comparison of Modern Biomass Torrefaction Reactors vs. Biomass Pyrolysis—Part 1. Energies 2022, 15, 2227. [Google Scholar] [CrossRef]
- Tic, W.; Guziałowska-Tic, J.; Pawlak-Kruczek, H.; Woźnikowski, E.; Zadorożny, A.; Niedźwiecki, Ł.; Wnukowski, M.; Krochmalny, K.; Czerep, M.; Ostrycharczyk, M.; et al. Novel Concept of an Installation for Sustainable Thermal Utilization of Sewage Sludge. Energies 2018, 11, 748. [Google Scholar] [CrossRef]
- Nunes, L.J.R. A Case Study about Biomass Torrefaction on an Industrial Scale: Solutions to Problems Related to Self-Heating, Difficulties in Pelletizing, and Excessive Wear of Production Equipment. Appl. Sci. 2020, 10, 2546. [Google Scholar] [CrossRef]
- Nanou, P.; Carbo, M.C.; Kiel, J.H.A. Detailed Mapping of the Mass and Energy Balance of a Continuous Biomass Torrefaction Plant. Biomass Bioenergy 2016, 89, 67–77. [Google Scholar] [CrossRef]
- Margaritis, N.; Grammelis, P.; Karampinis, E.; Kanaveli, I.-P. Impact of Torrefaction on Vine Pruning’s Fuel Characteristics. J. Energy Eng. 2020, 146, 04020006. [Google Scholar] [CrossRef]
- Jagodzińska, K.; Czerep, M.; Kudlek, E.; Wnukowski, M.; Pronobis, M.; Yang, W. Torrefaction of Agricultural Residues: Effect of Temperature and Residence Time on the Process Products Properties. J. Energy Resour. Technol. 2020, 142, 070912. [Google Scholar] [CrossRef]
- Piersa, P.; Szufa, S.; Czerwińska, J.; Ünyay, H.; Adrian, Ł.; Wielgosinski, G.; Obraniak, A.; Lewandowska, W.; Marczak-Grzesik, M.; Dzikuć, M.; et al. Pine Wood and Sewage Sludge Torrefaction Process for Production Renewable Solid Biofuels and Biochar as Carbon Carrier for Fertilizers. Energies 2021, 14, 8176. [Google Scholar] [CrossRef]
- Szufa, S.; Piersa, P.; Adrian, Ł.; Sielski, J.; Grzesik, M.; Romanowska-Duda, Z.; Piotrowski, K.; Lewandowska, W. Acquisition of Torrefied Biomass from Jerusalem Artichoke Grown in a Closed Circular System Using Biogas Plant Waste. Molecules 2020, 25, 3862. [Google Scholar] [CrossRef]
- Saeed, M.A.; Andrews, G.E.; Phylaktou, H.N.; Gibbs, B.M. Raw and Steam Exploded Pine Wood: Possible Enhanced Reactivity with Gasification Hydrogen. Int. J. Hydrogen Energy 2016, 41, 16566–16576. [Google Scholar] [CrossRef]
- Szufa, S.; Wielgosiński, G.; Piersa, P.; Czerwińska, J.; Dzikuć, M.; Adrian, Ł.; Lewandowska, W.; Marczak, M. Torrefaction of Straw from Oats and Maize for Use as a Fuel and Additive to Organic Fertilizers—TGA Analysis, Kinetics as Products for Agricultural Purposes. Energies 2020, 13, 2064. [Google Scholar] [CrossRef]
- Horák, J.; Kuboňová, L.; Hopan, F.; Kremer, J.; Dej, M.; Tomšej, T.; Krpec, K.; Ryšavý, J.; Molchanov, O.; Garba, M.; et al. Influence of Co-Combustion of Unsuitable Fuels with Standardized Fuels in Households on CO, OGC, PM, and PAH Emissions. Environ. Sci. Pollut. Res. 2022, 29, 44297–44307. [Google Scholar] [CrossRef]
- Pawlak-Kruczek, H.; Arora, A.; Gupta, A.; Saeed, M.A.; Niedzwiecki, L.; Andrews, G.; Phylaktou, H.; Gibbs, B.; Newlaczyl, A.; Livesey, P.M. Biocoal—Quality Control and Assurance. Biomass Bioenergy 2020, 135, 105509. [Google Scholar] [CrossRef]
- Szufa, S.; Dzikuć, M.; Adrian, Ł.; Piersa, P.; Romanowska-Duda, Z.; Lewandowska, W.; Marcza, M.; Błaszczuk, A.; Piwowar, A. Torrefaction of Oat Straw to Use as Solid Biofuel, an Additive to Organic Fertilizers for Agriculture Purposes and Activated Carbon—TGA Analysis, Kinetics. E3S Web Conf. 2020, 154, 02004. [Google Scholar] [CrossRef]
- Horák, J.; Hopan, F.; Kremer, J.; Kuboňová, L.; Polcar, L.; Molchanov, O.; Ryšavý, J.; Krpec, K.; Kubesa, P.; Dej, M.; et al. Real Measurement of Carbon Monoxide, Total Suspended Particulate, and Thermal Efficiency in Modern Biomass Household Boilers. Biomass Convers. Biorefin. 2022, 12, 4463–4472. [Google Scholar] [CrossRef]
- Sattar, H.; Andrews, G.E.; Phylaktou, H.N.; Gibbs, B.M. Turbulent Flames Speeds and Laminar Burning Velocities of Dusts Using the ISO 1 M3 Dust Explosion Method. Chem. Eng. Trans. 2014, 36, 157–162. [Google Scholar] [CrossRef]
- Saeed, M.A.; Slatter, D.J.F.; Andrews, G.E.; Phylaktou, H.N.; Gibbs, B.M. Combustion of Pulverized Biomass Crop Residues and Their Explosion Characteristics. Combust. Sci. Technol. 2016, 188, 2200–2216. [Google Scholar] [CrossRef]
- Saeed, M.A.; Andrews, G.E.; Phylaktou, H.N.; Gibbs, B.M. Global Kinetics of the Rate of Volatile Release from Biomasses in Comparison to Coal. Fuel 2016, 181, 347–357. [Google Scholar] [CrossRef]

| Biomass | %C | %H | %N | %O | %H2O | %VM | %FC | %Ash | HHV MJ/kg | LHV MJ/kg | Stoich A/F g/g | Stoich. F/A g/m3 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dry Ash-Free (daf) | As Received (ar) | Dry | ar | daf | Actual | |||||||
| SPFR | 50.4 | 6.9 | 1.2 | 41.4 | 7.8 | 73.4 | 16.2 | 2.6 | 19.9 | 17.8 | 6.4 | 187 |
| SPFR residue | 51.1 | 6.3 | 1.2 | 41.4 | 6.8 | 72.2 | 17.5 | 3.5 | 19.8 | 17.8 | 6.3 | 212 |
| SPFT | 54.7 | 6.9 | 1.1 | 37.4 | 4 | 74.6 | 18.1 | 3.2 | 21.7 | 20.1 | 7.05 | 183 |
| SPFT residue | 57.9 | 6.2 | 1.4 | 34.5 | 4.2 | 65.2 | 22.7 | 7.8 | 21.3 | 18.7 | 7.3 | 187 |
| Biomass | d10, µm | d50, µm | d90, µm | dsmd or d32, µm | Fines (Particles < 100 µm), % |
|---|---|---|---|---|---|
| SPFR | 91 | 451 | 866 | 184 | 15.4 |
| Post-explosion SPFR residue | 69 | 288 | 747 | 124 | 17 |
| SPFT | 73 | 347 | 785 | 151 | 14.5 |
| Post-explosion SPFT residue | 78 | 343 | 781 | 164 | 14 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saeed, M.A.; Niedzwiecki, L.; Arshad, M.Y.; Skrinsky, J.; Andrews, G.E.; Phylaktou, H.N. Combustion and Explosion Characteristics of Pulverised Wood, Valorized with Mild Pyrolysis in Pilot Scale Installation, Using the Modified ISO 1 m3 Dust Explosion Vessel. Appl. Sci. 2022, 12, 12928. https://doi.org/10.3390/app122412928
Saeed MA, Niedzwiecki L, Arshad MY, Skrinsky J, Andrews GE, Phylaktou HN. Combustion and Explosion Characteristics of Pulverised Wood, Valorized with Mild Pyrolysis in Pilot Scale Installation, Using the Modified ISO 1 m3 Dust Explosion Vessel. Applied Sciences. 2022; 12(24):12928. https://doi.org/10.3390/app122412928
Chicago/Turabian StyleSaeed, Muhammad Azam, Lukasz Niedzwiecki, Muhammad Yousaf Arshad, Jan Skrinsky, Gordon E. Andrews, and Herodotos N. Phylaktou. 2022. "Combustion and Explosion Characteristics of Pulverised Wood, Valorized with Mild Pyrolysis in Pilot Scale Installation, Using the Modified ISO 1 m3 Dust Explosion Vessel" Applied Sciences 12, no. 24: 12928. https://doi.org/10.3390/app122412928
APA StyleSaeed, M. A., Niedzwiecki, L., Arshad, M. Y., Skrinsky, J., Andrews, G. E., & Phylaktou, H. N. (2022). Combustion and Explosion Characteristics of Pulverised Wood, Valorized with Mild Pyrolysis in Pilot Scale Installation, Using the Modified ISO 1 m3 Dust Explosion Vessel. Applied Sciences, 12(24), 12928. https://doi.org/10.3390/app122412928






