Selective Use of Corn Crop Residues: Energy Viability
Abstract
1. Introduction
2. Materials and Methods
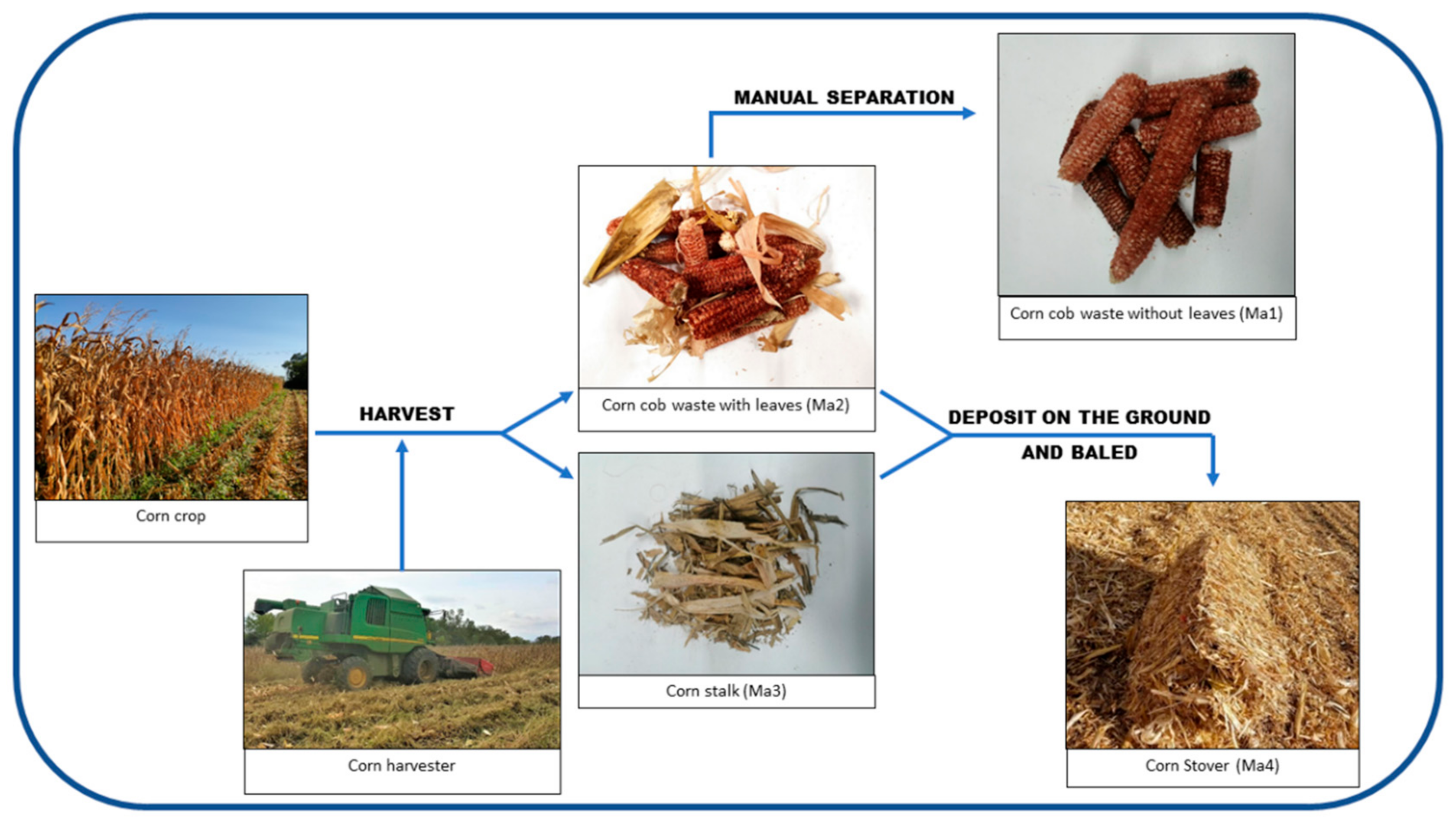
2.1. Sample Collection and Preparation
2.1.1. Sample Collection Process
2.1.2. Sample Preparation
2.2. Proximate and Ultimate Analysis
2.3. Higher Heating Value
2.4. Ash Composition
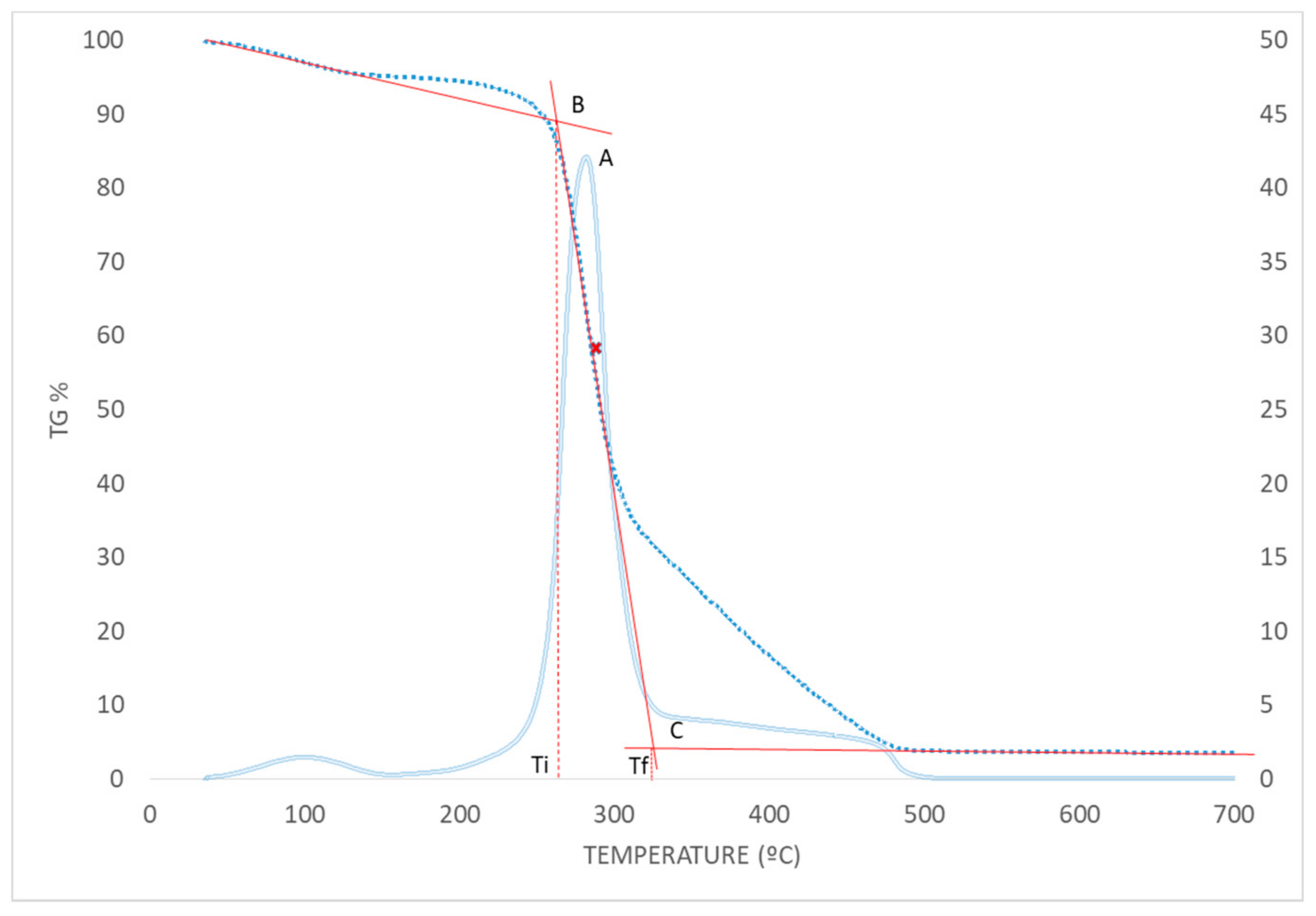
2.5. TG Analysis
3. Results and Discussion
3.1. Proximate and Ultimate Analysis and Higher Heating Value
3.2. Ash Composition
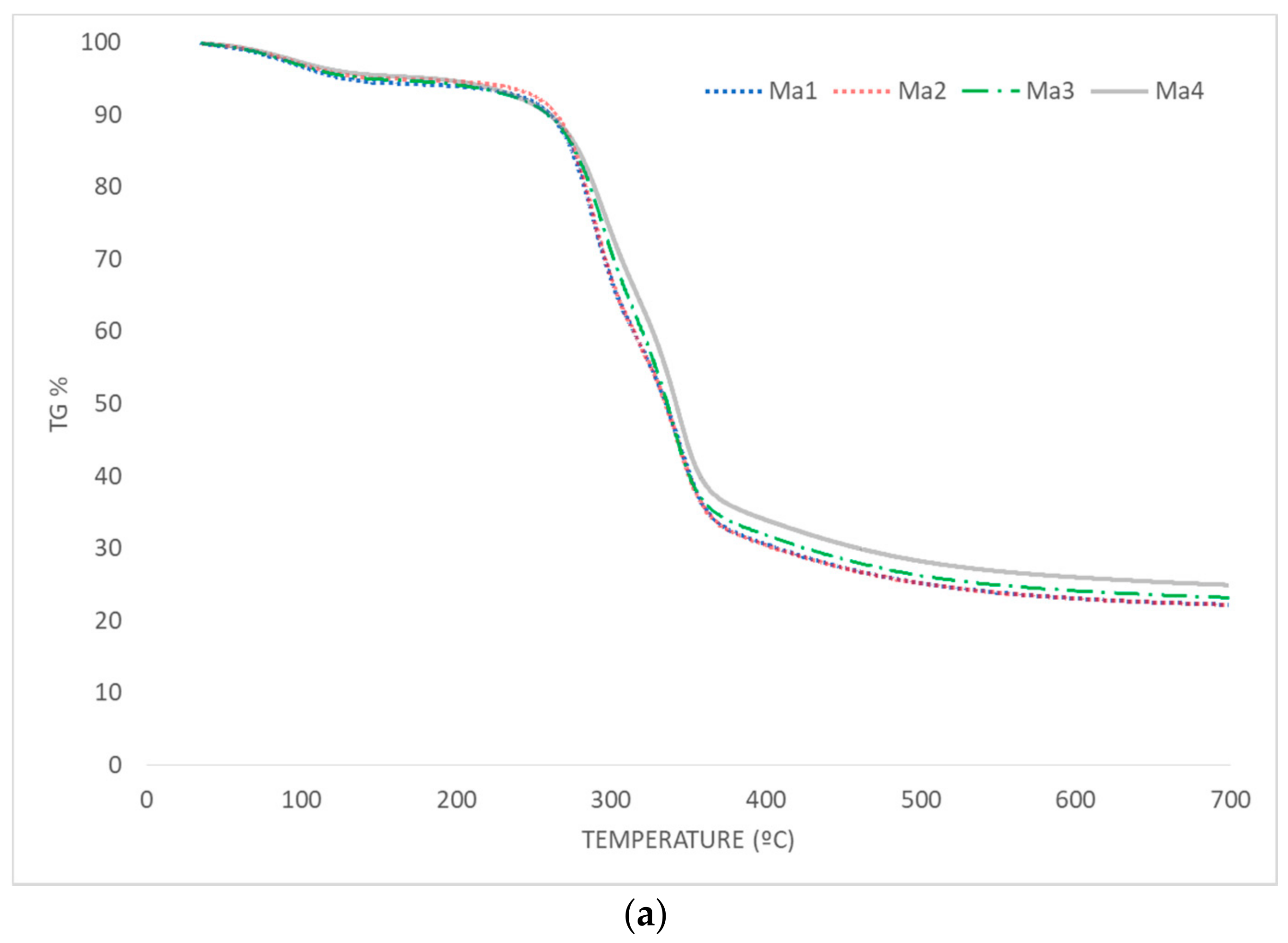
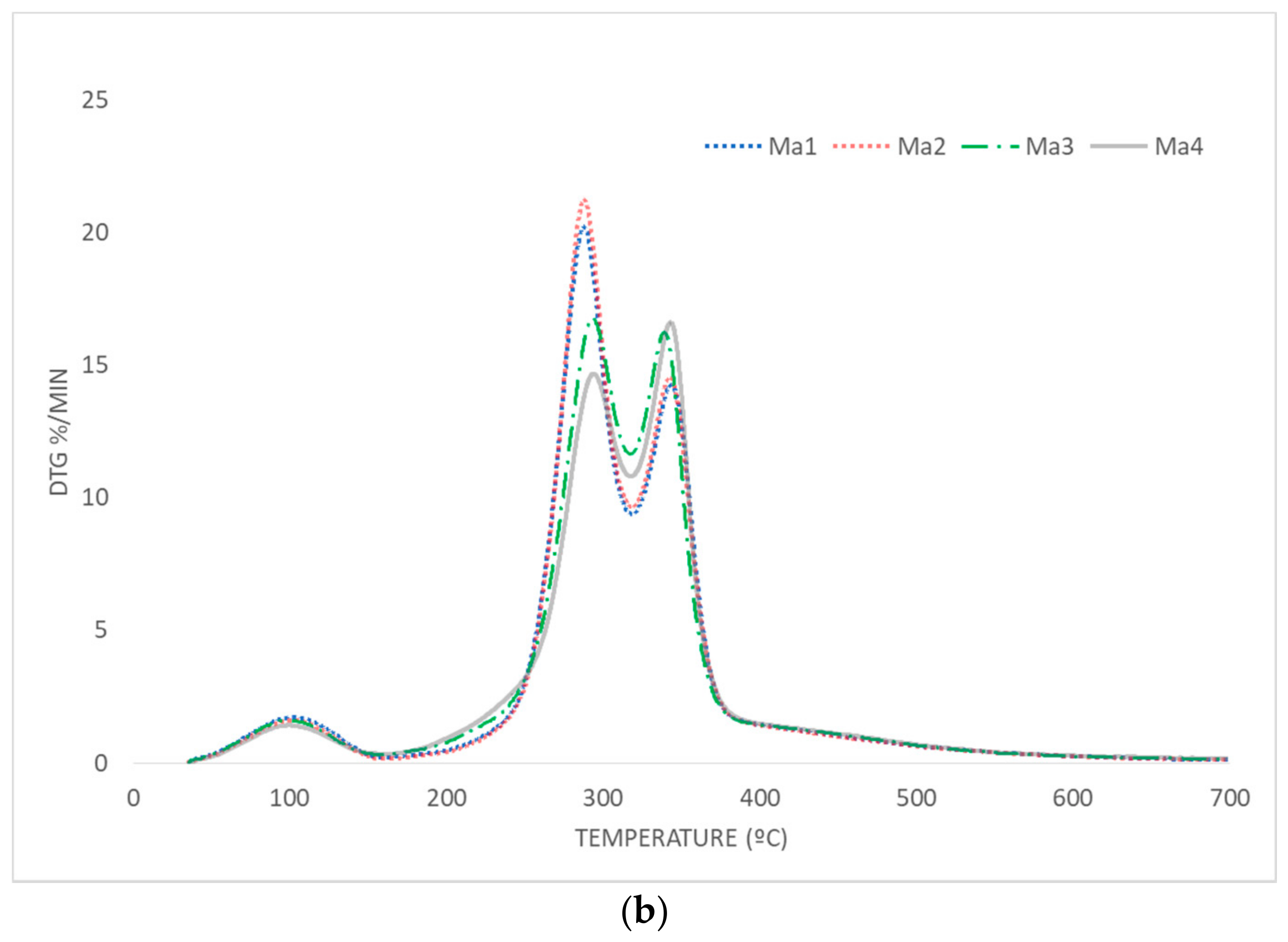
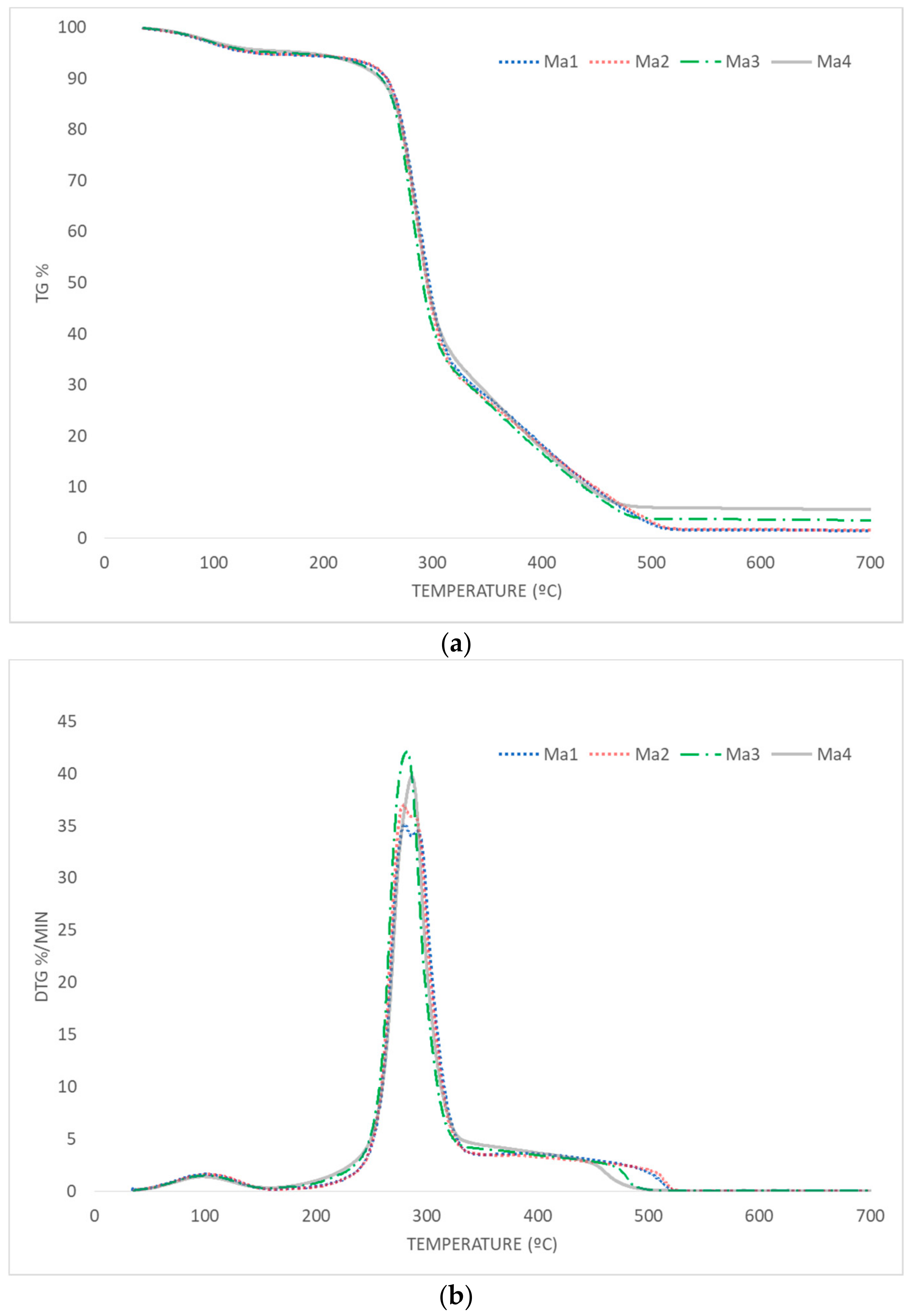
3.3. Thermal Behavior
3.3.1. Pyrolysis
3.3.2. Combustion
4. Conclusions
- It can be inferred, from the characterization of the residues, that the moisture content was suitable for subsequent pelletizing without any previous drying process (less than 15%).
- Regarding ash content, all the residues were suitable for pellet production (A class) except for Ma4 residue, which was collected on the soil and showed a high ash content (9.68%). This fact pointed out that the contact with the soil had a negative effect on the subsequent valorization of the waste. Therefore, direct collection by harvesting machines is desirable.
- The heating values are within the optimum range established by the corresponding quality standard for pelletizing (more than 16 MJ/kg, dry basis).
- Concerning ash composition, SiO2 and K2O values imply a possible low ash melting point, which would be interesting to check through combustion tests of the residues.
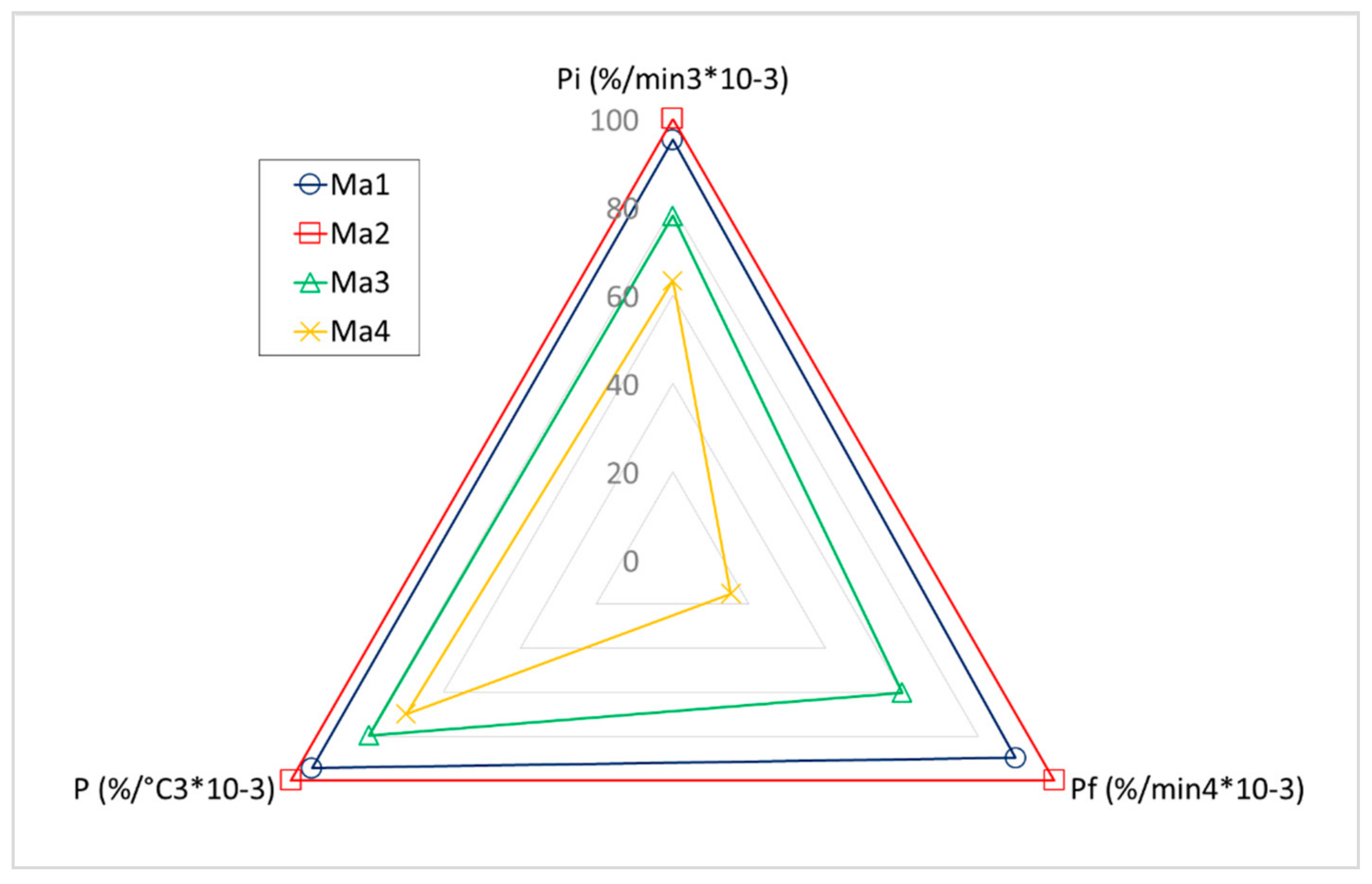
- With regard to the pyrolysis process, optimum characteristics were observed for corncob waste or samples with the majority of its content containing this residue (Ma1 and Ma2); no significant difference was found in the presence of leaves. In this case, the selective collection of residues was highly recommended. Moreover, the direct collection by harvesting machines (without any contact between residues and soil) was advisable, as the subsequent degradation produced unfavorable results in pyrolysis.
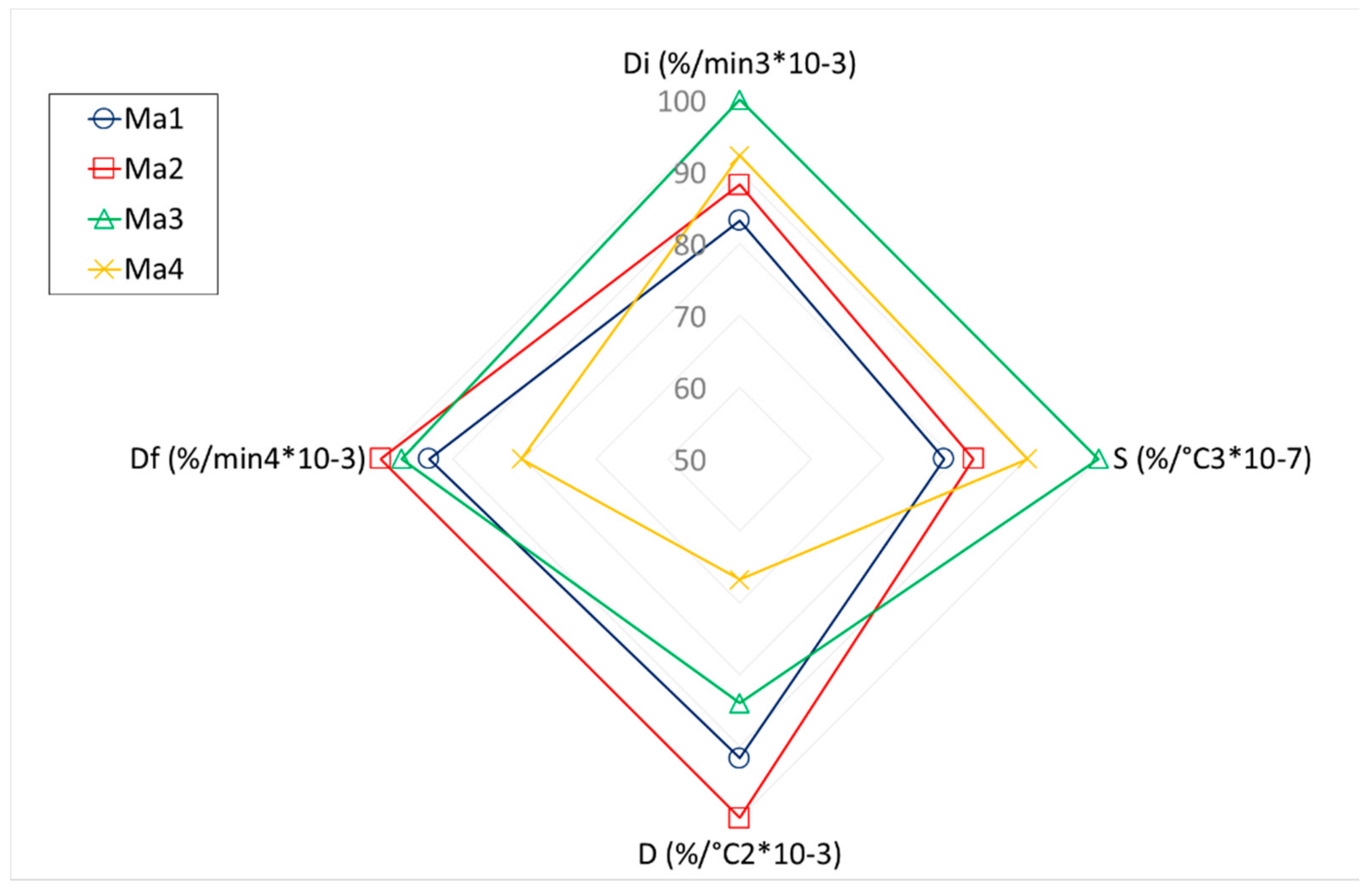
- Concerning the study of combustion process, better characteristics were observed when corncob waste and corn stalk were combined. On one hand, the wastes with a majority of its content containing corncob waste obtained lower ash content. On the other hand, the wastes with corn stalk had improved combustibility. In any case, the negative effect of the degradation of the waste collected on the soil can be observed.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Food and Agriculture Organitation of the United Nations. FAOSTAT. 2020. Available online: http://fao.org/faostat/en/#data (accessed on 1 September 2020).
- Azuara, M.; Baguer, B.; Villacampa, J.; Hedin, I.; Manyà, J.J. Influence of pressure and temperature on key physico-chemical properties of corn stover-derived biochar. Fuel 2016, 186, 525–533. [Google Scholar] [CrossRef]
- Zheng, J.; Choo, K.; Bradt, C.; Lehoux, R.; Rehmann, L. Enzymatic hydrolysis of steam exploded corncob residues after pretreatment in a twin-screw extruder. Biotechnol. Rep. 2014, 3, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Brutti, L.; Beltran, M. García de Salamone, Biorremediación de los Recursos Naturales; INTA Ediciones: Buenos Aires, Argentina, 2018. [Google Scholar]
- Anwar, Z.; Gulfraz, M.; Irshad, M. Agro-industrial lignocellulosic biomass a key to unlock the future bio-energy: A brief review. J. Radiat. Res. Appl. Sci. 2014, 7, 163–173. [Google Scholar] [CrossRef]
- Prasad, S.; Singh, A.; Joshi, H. Ethanol as an alternative fuel from agricultural, industrial and urban residues. Resour. Conserv. Recycl. 2007, 50, 1–39. [Google Scholar] [CrossRef]
- Chen, Q.; Yang, R.; Zhao, B.; Li, Y.; Wang, S.; Wu, H.; Zhuo, Y.; Chen, C. Investigation of heat of biomass pyrolysis and secondary reactions by simultaneous thermogravimetry and differential scanning calorimetry. Fuel 2014, 134, 467–476. [Google Scholar] [CrossRef]
- Shen, J.; Igathinathane, C.; Yu, M.; Pothula, A.K.; Cannayen, I. Biomass pyrolysis and combustion integral and differential reaction heats with temperatures using thermogravimetric analysis/differential scanning calorimetry. Bioresour. Technol. 2015, 185, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Berndes, G.; Abt, B.; Asikainen, A.; Cowie, A.; Dale, V.; Egnell, G.; Lindner, M.; Marelli, L.; Paré, D.; Pingoud, K.; et al. Forest biomass, carbon neutrality and climate change mitigation. In From Science to Policy 3; European Forest Institute: Joensuu, Finland, 2016. [Google Scholar] [CrossRef]
- Arranz, J.I.; Miranda, M.T.; Montero, I.; Sepulveda, F.J.; Rojas, C.V. Characterization and combustion behaviour of commercial and experimental wood pellets in South West Europe. Fuel 2015, 142, 199–207. [Google Scholar] [CrossRef]
- Zhang, G.; Sun, Y.; Shi, Y.; Jia, Y.; Xu, Y.; Zhao, P.; Zhang, Y. Characteristic and kinetics of corn stalk pyrolysis in a high pressure reactor and steam gasification of its char. J. Anal. Appl. Pyrolysis 2016, 122, 249–257. [Google Scholar] [CrossRef]
- Aboyade, A.O.; Hugo, T.J.; Carrier, M.; Meyer, E.L.; Stahl, R.; Knoetze, J.H.; Görgens, J.F. Non-isothermal kinetic analysis of the devolatilization of corn cobs and sugar cane bagasse in an inert atmosphere. Thermochim. Acta 2011, 517, 81–89. [Google Scholar] [CrossRef]
- Yang, X.; Zeng, Y.; Ma, F.; Zhang, X.; Yu, H. Effect of biopretreatment on thermogravimetric and chemical characteristics of corn stover by different white-rot fungi. Bioresour. Technol. 2010, 101, 5475–5479. [Google Scholar] [CrossRef]
- Maddi, B.; Viamajala, S.; Varanasi, S.; Yuan, T.; Tahmasebi, A.; Yu, J. Comparative study of pyrolysis of algal biomass from natural lake blooms with lignocellulosic biomass. Bioresour. Technol. 2011, 102, 11018–11026. [Google Scholar] [CrossRef]
- Gai, C.; Dong, Y.; Zhang, T. The kinetic analysis of the pyrolysis of agricultural residue under non-isothermal condi-tions. Bioresour. Technol. 2013, 127, 298–305. [Google Scholar] [CrossRef]
- Kumar, A.; Wang, L.; Dzenis, A.; Jones, D.; Hanna, M.A. Thermogravimetric characterization of corn stover as gasification and pyrolysis feedstock. Biomass Bioenergy 2008, 32, 460–467. [Google Scholar] [CrossRef]
- Li, Z.; Zhao, W.; Meng, B.; Liu, C.; Zhu, Q.; Zhao, G. Kinetic study of corn straw pyrolysis: Comparison of two different three-pseudocomponent models. Bioresour. Technol. 2008, 99, 7616–7622. [Google Scholar] [CrossRef]
- Álvarez, A.; Pizarro, C.; García, R.; Bueno, J.L.; Lavín, A.G. Determination of kinetic parameters for biomass combus-tion. Bioresour. Technol. 2016, 216, 36–43. [Google Scholar] [CrossRef]
- ISO. ISO 17225-6:2014. In Solid Biofuels. Fuel Specifications and Classes. Part 6: Graded Non-Woody Pellets; ISO: Geneva, Switzerland, 2014. [Google Scholar]
- ISO. ISO 18134-2:2017. In Solid Biofuels. Determination of Moisture Content. Oven Dry Method. Part 2: Total Moisture. Simplified Method; ISO: Geneva, Switzerland, 2017. [Google Scholar]
- ISO. ISO 17828:2015. In Solid Biofuels. Determination of Bulk Density; ISO: Geneva, Switzerland, 2015. [Google Scholar]
- ISO. ISO 16948:2015. In Solid Biofuels. Determination of Total Content of Carbon, Hydrogen and Nitrogen; ISO: Geneva, Switzerland, 2015. [Google Scholar]
- ISO. ISO 16994:2016. In Solid Biofuels. Determination of Content of Sulfur and Chlorine; ISO: Geneva, Switzerland, 2017. [Google Scholar]
- ISO. ISO 18123:2016. In Solid Biofuels. Determination of the Content of Volatile Matter; ISO: Geneva, Switzerland, 2015. [Google Scholar]
- ISO. ISO 18122:2015. In Solid Biofuels. Determination of Ash Content; ISO: Geneva, Switzerland, 2015. [Google Scholar]
- ISO. ISO 18125:2017. In Solid Biofuels. Determination of Calorific Value; ISO: Geneva, Switzerland, 2017. [Google Scholar]
- Song, H.; Liu, G.; Zhang, J.; Wu, J. Pyrolysis characteristics and kinetics of low rank coals by TG-FTIR method. Fuel Process. Technol. 2017, 156, 454–460. [Google Scholar] [CrossRef]
- Mureddu, M.; Dessì, F.; Orsini, D.; Ferrara, F.; Pettinau, A. Air- and oxygen-blown characterization of coal and biomass by thermogravimetric analysis. Fuel 2018, 212, 626–637. [Google Scholar] [CrossRef]
- Moon, C.; Sung, Y.; Ahn, S.; Kim, T.; Choi, G.; Kim, D. Effect of blending ratio on combustion performance in blends of biomass and coals of different ranks. Exp. Therm. Fluid Sci. 2013, 47, 232–240. [Google Scholar] [CrossRef]
- Lu, J.-J.; Chen, W.-H. Investigation on the ignition and burnout temperatures of bamboo and sugarcane bagasse by thermogravimetric analysis. Appl. Energy 2015, 160, 49–57. [Google Scholar] [CrossRef]
- Paniagua, S.; Escudero, L.; Coimbra, R.N.; Escapa, C.; Otero, M.; Calvo, L.F. Effect of Applying Organic Amendments on the Pyrolytic Behavior of a Poplar Energy Crop. Waste Biomass Valoriz. 2018, 9, 1435–1449. [Google Scholar] [CrossRef]
- Paniagua, S.; García-Pérez, A.I.; Calvo, L.F. Biofuel consisting of wheat straw–poplar wood blends: Thermogravimetric studies and combustion characteristic indexes estimation. Biomass Convers. Biorefin. 2018, 9, 433–443. [Google Scholar] [CrossRef]
- Zhang, Y.; Guo, Y.; Cheng, F.; Yan, K.; Cao, Y. Investigation of combustion characteristics and kinetics of coal gangue with different feedstock properties by thermogravimetric analysis. Thermochim. Acta 2015, 614, 137–148. [Google Scholar]
- Zhang, Y.; Ghaly, A.E.; Li, B. Physical Properties of Corn Residues. Am. J. Biochem. Biotechnol. 2012, 8, 44–53. [Google Scholar] [CrossRef]
- Rony, A.H. Kinetics, thermodynamics, and physical characterization of corn stover (Zea mays) for solar biomass py-rolysis potential analysis. Bioresour. Technol. 2019, 284, 466–473. [Google Scholar] [CrossRef]
- Wang, C.; Wang, F.; Yang, Q.; Liang, R. Thermogravimetric studies of the behavior of wheat straw with added coal dur-ing combustion. Biomass Bioenergy 2009, 33, 50–56. [Google Scholar] [CrossRef]
- Vassilev, S.V.; Baxter, D.; Andersen, L.K.; Vassileva, C.G. An overview of the composition and application of biomass ash. Part 1. Phase–mineral and chemical composition and classification. Fuel 2013, 105, 40–76. [Google Scholar] [CrossRef]
- Vassilev, S.V.; Baxter, D.; Andersen, L.K.; Vassileva, C.G. An overview of the composition and application of biomass ash. Fuel 2013, 105, 19–39. [Google Scholar] [CrossRef]
- Rodríguez, J.L.; Álvarez, X.; Valero, E.; Ortiz, L.; De La Torre-Rodríguez, N.; Acuña-Alonso, C. Influence of ashes in the use of forest biomass as source of energy. Fuel 2021, 283, 119256. [Google Scholar] [CrossRef]
- Diblasi, C. Modeling chemical and physical processes of wood and biomass pyrolysis. Prog. Energy Combust. Sci. 2008, 34, 47–90. [Google Scholar] [CrossRef]
- Yang, Z. Combustion behaviours of tobacco stem in a thermogravimetric analyser and a pilot-scale fluidized bed reac-tor. Bioresour. Technol. 2012, 110, 595–602. [Google Scholar] [CrossRef] [PubMed]
- Parshetti, G.K.; Quek, A.; Betha, R.; Balasubramanian, R. TGA–FTIR investigation of co-combustion characteristics of blends of hydrothermally carbonized oil palm biomass (EFB) and coal. Fuel Process. Technol. 2014, 118, 228–234. [Google Scholar] [CrossRef]
| Moisture % wb | Density kg/m3 wb | Ultimate Analysis (% db) | Proximate Analysis (% db) | HHV MJ/kg db | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| C | H | N | S | O | Ash | Volatile Matter | Fixed Carbon | ||||
| Ma 1 | 11.30 | 220.02 | 41.57 | 6.48 | 0.35 | 0.04 | 51.56 | 1.92 | 85.79 | 12.29 | 16.54 |
| Ma 2 | 10.37 | 204.80 | 42.20 | 6.35 | 0.34 | 0.04 | 51.07 | 1.98 | 86.72 | 11.30 | 16.72 |
| Ma 3 | 9.23 | 121.80 | 41.44 | 6.37 | 0.54 | 0.07 | 51.58 | 4.41 | 80.72 | 14.87 | 16.52 |
| Ma 4 | 7.15 | 96.86 | 39.98 | 5.87 | 0.50 | 0.08 | 53.57 | 9.68 | 79.75 | 10.57 | 16.37 |
| Ash Composition (% db) | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| K | Cl | Si | P | Mg | Ca | S | K2O | SiO2 | P2O5 | CaO | MgO | SO3 | Al2O3 | Fe2O3 | ZnO | |
| Ma 1 | 48.20 | 9.53 | 6.27 | 3.59 | 2.48 | 1.73 | 1.23 | 58.11 | 13.40 | 8.23 | 2.42 | 4.12 | 3.08 | 0.12 | 0.32 | 0.24 |
| Ma 2 | 46.45 | 9.65 | 7.94 | 2.83 | 2.35 | 2.13 | 1.12 | 55.96 | 17.00 | 6.48 | 2.98 | 3.90 | 2.80 | 0.20 | 0.40 | 0.13 |
| Ma 3 | 29.07 | 7.33 | 13.42 | 2.67 | 4.87 | 6.68 | 1.26 | 35.02 | 28.72 | 6.12 | 9.35 | 8.07 | 3.14 | 0.78 | 0.59 | 0.07 |
| Ma 4 | 18.93 | 5.98 | 17.57 | 1.55 | 7.05 | 8.04 | 1.24 | 23.40 | 37.58 | 3.56 | 11.26 | 11.70 | 3.09 | 1.51 | 1.06 | 0.05 |
| Main Pyrolysis Parameters | ||||
|---|---|---|---|---|
| Ma1 | Ma2 | Ma3 | Ma4 | |
| Fast Pyrolysis initial Tm (°C) | 272.5 | 272.3 | 269.9 | 284.6 |
| Peak temperature Tp (°C) | 287.6 | 287.8 | 293.2 | 342.9 |
| Shoulder peak Ts (°C) | 343.5 | 342.4 | 339.0 | 293.7 |
| Pyrolysis finish Tf (°C) | 374.5 | 373.6 | 368.9 | 371.3 |
| DTG peak max (%/min) | 20.23 | 21.24 | 16.76 | 16.62 |
| DTG shoulder (%/min) | 14.25 | 14.53 | 16.26 | 14.68 |
| DTG mean (%/min) | 2.08 | 2.09 | 2.05 | 2.00 |
| Fast Pyrolysis initial time tm (min) | 12.40 | 12.39 | 12.31 | 12.88 |
| Peak time tp (min) | 12.97 | 12.98 | 13.21 | 15.42 |
| Pyrolysis finish time tf (min) | 16.97 | 16.93 | 16.69 | 16.81 |
| Δt1/2(min) | 0.72 | 0.68 | 0.89 | 2.95 |
| Pi (%/min3 × 10−3) | 125.79 | 132.07 | 103.07 | 83.68 |
| Pf (%/min4 × 10−3) | 127.66 | 142.14 | 85.41 | 21.73 |
| P (%/°C3 × 10−3) | 16.13 | 17.08 | 13.58 | 11.92 |
| Residual Mass (%) | 21.30 | 21.34 | 22.22 | 23.96 |
| Main Combustion Parameters | ||||
|---|---|---|---|---|
| Ma1 | Ma2 | Ma3 | Ma4 | |
| T for 5% weigh loss (°C) | 135.0 | 137.2 | 164.5 | 183.1 |
| Ignition temperature Ti (°C) | 266.6 | 266.0 | 264.6 | 266.6 |
| Peak temperature Tp (°C) | 279.6 | 278.6 | 281.5 | 286.3 |
| Burnout temperature Tf (°C) | 506.8 | 511.4 | 485.8 | 486.6 |
| DTG max (%/min) | 35.12 | 36.99 | 42.10 | 39.72 |
| DTG mean (%/min) | 2.73 | 2.74 | 2.74 | 2.66 |
| Ignition time ti (min) | 12.18 | 12.15 | 12.10 | 12.18 |
| Peak time tp (min) | 12.64 | 12.59 | 12.69 | 12.89 |
| Burnout time tf (min) | 23.96 | 24.12 | 22.9 | 23.00 |
| Δt1/2 (min) | 14.0 | 13.8 | 17.0 | 18.6 |
| Di (%/min3 × 10−3) | 228.12 | 241.81 | 274.18 | 252.99 |
| S (%/°C3 × 10−7) | 26.62 | 28.01 | 33.92 | 30.55 |
| D (%/°C2 × 10−3) | 9.66 | 10.54 | 8.85 | 7.04 |
| Df (%/min4 × 10−3) | 231.93 | 248.59 | 241.45 | 199.96 |
| Residual mass (%) | 1.22 | 1.24 | 3.10 | 5.23 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miranda, M.T.; García-Mateos, R.; Arranz, J.I.; Sepúlveda, F.J.; Romero, P.; Botet-Jiménez, A. Selective Use of Corn Crop Residues: Energy Viability. Appl. Sci. 2021, 11, 3284. https://doi.org/10.3390/app11073284
Miranda MT, García-Mateos R, Arranz JI, Sepúlveda FJ, Romero P, Botet-Jiménez A. Selective Use of Corn Crop Residues: Energy Viability. Applied Sciences. 2021; 11(7):3284. https://doi.org/10.3390/app11073284
Chicago/Turabian StyleMiranda, M. T., R. García-Mateos, J. I. Arranz, F. J. Sepúlveda, P. Romero, and A. Botet-Jiménez. 2021. "Selective Use of Corn Crop Residues: Energy Viability" Applied Sciences 11, no. 7: 3284. https://doi.org/10.3390/app11073284
APA StyleMiranda, M. T., García-Mateos, R., Arranz, J. I., Sepúlveda, F. J., Romero, P., & Botet-Jiménez, A. (2021). Selective Use of Corn Crop Residues: Energy Viability. Applied Sciences, 11(7), 3284. https://doi.org/10.3390/app11073284






