The Less Known Cyclins—Uncovered
Abstract
1. Introduction
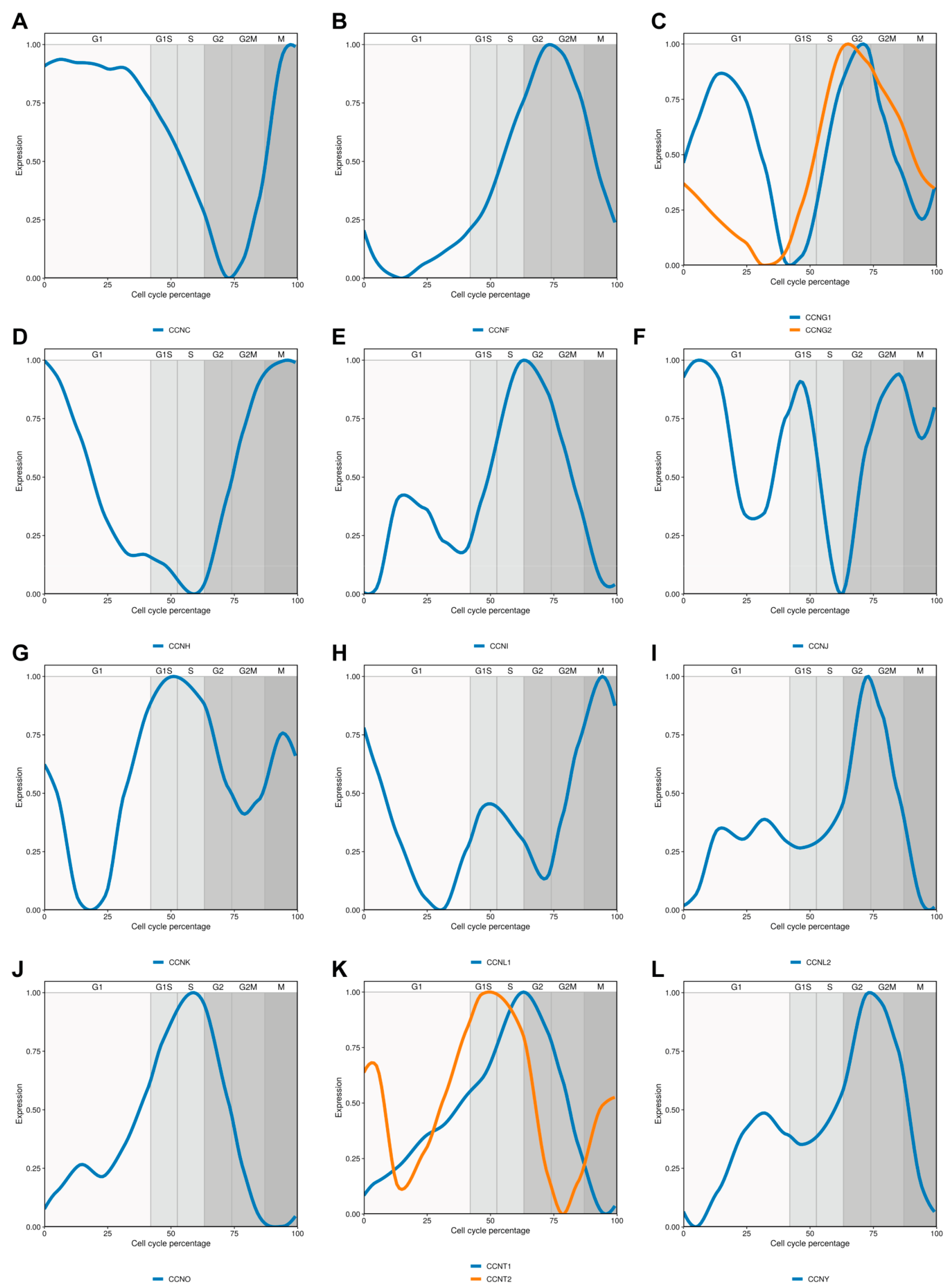
2. Cyclin C
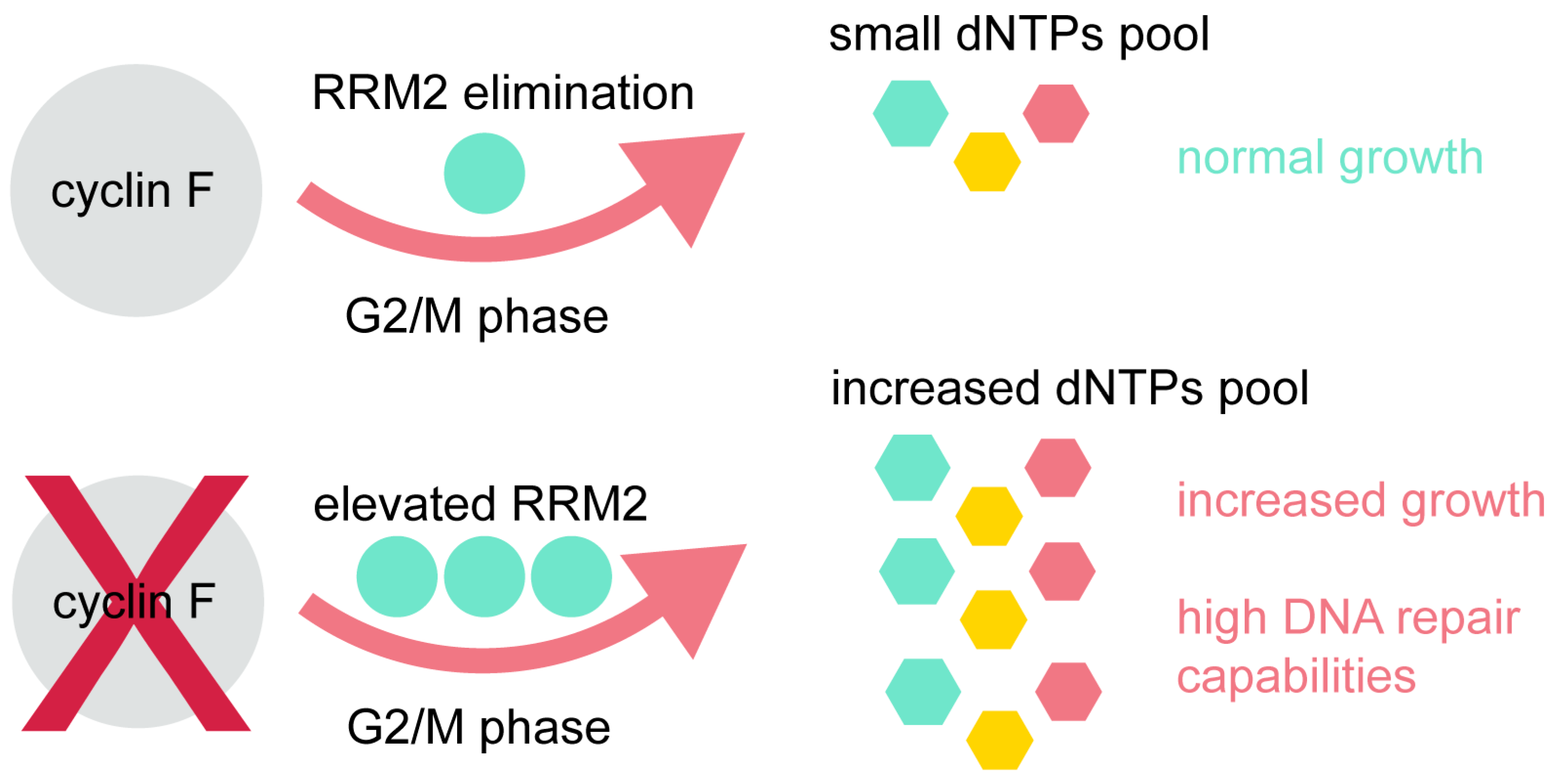
3. Cyclin F
4. Cyclin G
5. Cyclin H
6. Cyclin I
7. Cyclin J
8. Cyclin K
9. Cyclin L
10. Cyclin M
11. Cyclin O
12. Cyclin T
13. Cyclin Y
14. Summary
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Schafer, K.A. The Cell Cycle: A Review. Vet. Pathol. 1998, 35, 461–478. [Google Scholar] [CrossRef]
- Lim, S.; Kaldis, P. Cdks, Cyclins and CKIs: Roles beyond Cell Cycle Regulation. Development 2013, 140, 3079–3093. [Google Scholar] [CrossRef] [PubMed]
- D’Andrilli, G.; Kumar, C.; Scambia, G.; Giordano, A. Cell Cycle Genes in Ovarian Cancer: Steps toward Earlier Diagnosis and Novel Therapies. Clin. Cancer Res. 2004, 10, 8132–8141. [Google Scholar] [CrossRef] [PubMed]
- Park, M.-T.; Lee, S.-J. Cell Cycle and Cancer. J. Biochem. Mol. Biol. 2003, 36, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Lew, D.J.; Dulić, V.; Reed, S.I. Isolation of Three Novel Human Cyclins by Rescue of G1 Cyclin (Cln) Function in Yeast. Cell 1991, 66, 1197–1206. [Google Scholar] [CrossRef]
- Li, H.; Lahti, J.M.; Valentine, M.; Saito, M.; Reed, S.I.; Look, A.T.; Kidd, V.J. Molecular Cloning and Chromosomal Localization of the Human Cyclin C (CCNC) and Cyclin E (CCNE) Genes: Deletion of the CCNC Gene in Human Tumors. Genomics 1996, 32, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Ježek, J.; Smethurst, D.G.J.; Stieg, D.C.; Kiss, Z.A.C.; Hanley, S.E.; Ganesan, V.; Chang, K.-T.; Cooper, K.F.; Strich, R. Cyclin C: The Story of a Non-Cycling Cyclin. Biology (Basel) 2019, 8. [Google Scholar] [CrossRef]
- Akoulitchev, S.; Chuikov, S.; Reinberg, D. TFIIH Is Negatively Regulated by Cdk8-Containing Mediator Complexes. Nature 2000, 407, 102–106. [Google Scholar] [CrossRef]
- Cooper, K.F.; Scarnati, M.S.; Krasley, E.; Mallory, M.J.; Jin, C.; Law, M.J.; Strich, R. Oxidative-Stress-Induced Nuclear to Cytoplasmic Relocalization Is Required for Not4-Dependent Cyclin C Destruction. J. Cell. Sci. 2012, 125, 1015–1026. [Google Scholar] [CrossRef]
- Ganesan, V.; Willis, S.D.; Chang, K.-T.; Beluch, S.; Cooper, K.F.; Strich, R. Cyclin C Directly Stimulates Drp1 GTP Affinity to Mediate Stress-Induced Mitochondrial Hyperfission. Mol. Biol. Cell 2019, 30, 302–311. [Google Scholar] [CrossRef]
- Wang, K.; Yan, R.; Cooper, K.F.; Strich, R. Cyclin C Mediates Stress-Induced Mitochondrial Fission and Apoptosis. Mol. Biol. Cell 2015, 26, 1030–1043. [Google Scholar] [CrossRef]
- Ren, S.; Rollins, B.J. Cyclin C/Cdk3 Promotes Rb-Dependent G0 Exit. Cell 2004, 117, 239–251. [Google Scholar] [CrossRef]
- Miyata, Y.; Liu, Y.; Jankovic, V.; Sashida, G.; Lee, J.M.; Shieh, J.-H.; Naoe, T.; Moore, M.; Nimer, S.D. Cyclin C Regulates Human Hematopoietic Stem/Progenitor Cell Quiescence. Stem Cells 2010, 28, 308–317. [Google Scholar] [CrossRef]
- Liu, Z.-J.; Ueda, T.; Miyazaki, T.; Tanaka, N.; Mine, S.; Tanaka, Y.; Taniguchi, T.; Yamamura, H.; Minami, Y. A Critical Role for Cyclin C in Promotion of the Hematopoietic Cell Cycle by Cooperation with C-Myc. Mol. Cell. Biol. 1998, 18, 3445–3454. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Fassl, A.; Chick, J.; Inuzuka, H.; Li, X.; Mansour, M.R.; Liu, L.; Wang, H.; King, B.; Shaik, S.; et al. Cyclin C Is a Haploinsufficient Tumour Suppressor. Nat. Cell Biol. 2014, 16, 1080–1091. [Google Scholar] [CrossRef] [PubMed]
- Ohata, N.; Ito, S.; Yoshida, A.; Kunisada, T.; Numoto, K.; Jitsumori, Y.; Kanzaki, H.; Ozaki, T.; Shimizu, K.; Ouchida, M. Highly Frequent Allelic Loss of Chromosome 6q16-23 in Osteosarcoma: Involvement of Cyclin C in Osteosarcoma. Int. J. Mol. Med. 2006, 18, 1153–1158. [Google Scholar] [CrossRef]
- Bondi, J.; Husdal, A.; Bukholm, G.; Nesland, J.M.; Bakka, A.; Bukholm, I.R.K. Expression and Gene Amplification of Primary (A, B1, D1, D3, and E) and Secondary (C and H) Cyclins in Colon Adenocarcinomas and Correlation with Patient Outcome. J. Clin. Pathol. 2005, 58, 509–514. [Google Scholar] [CrossRef]
- Firestein, R.; Bass, A.J.; Kim, S.Y.; Dunn, I.F.; Silver, S.J.; Guney, I.; Freed, E.; Ligon, A.H.; Vena, N.; Ogino, S.; et al. CDK8 Is a Colorectal Cancer Oncogene That Regulates Beta-Catenin Activity. Nature 2008, 455, 547–551. [Google Scholar] [CrossRef]
- Xu, W.; Ji, J.-Y. Dysregulation of CDK8 and Cyclin C in Tumorigenesis. J. Genet. Genomics 2011, 38, 439–452. [Google Scholar] [CrossRef]
- Bai, C.; Richman, R.; Elledge, S.J. Human Cyclin F. EMBO J. 1994, 13, 6087–6098. [Google Scholar] [CrossRef] [PubMed]
- Kraus, B.; Pohlschmidt, M.; Leung, A.L.; Germino, G.G.; Snarey, A.; Schneider, M.C.; Reeders, S.T.; Frischauf, A.M. A Novel Cyclin Gene (CCNF) in the Region of the Polycystic Kidney Disease Gene (PKD1). Genomics 1994, 24, 27–33. [Google Scholar] [CrossRef] [PubMed]
- D’Angiolella, V.; Esencay, M.; Pagano, M. A Cyclin without Cyclin-Dependent Kinases: Cyclin F Controls Genome Stability through Ubiquitin-Mediated Proteolysis. Trends Cell Biol. 2013, 23, 135–140. [Google Scholar] [CrossRef]
- Casimiro, M.C.; Crosariol, M.; Loro, E.; Li, Z.; Pestell, R.G. Cyclins and Cell Cycle Control in Cancer and Disease. Genes Cancer 2012, 3, 649–657. [Google Scholar] [CrossRef]
- D’Angiolella, V.; Donato, V.; Vijayakumar, S.; Saraf, A.; Florens, L.; Washburn, M.P.; Dynlacht, B.; Pagano, M. SCF(Cyclin F) Controls Centrosome Homeostasis and Mitotic Fidelity through CP110 Degradation. Nature 2010, 466, 138–142. [Google Scholar] [CrossRef]
- D’Angiolella, V.; Donato, V.; Forrester, F.M.; Jeong, Y.-T.; Pellacani, C.; Kudo, Y.; Saraf, A.; Florens, L.; Washburn, M.P.; Pagano, M. Cyclin F-Mediated Degradation of Ribonucleotide Reductase M2 Controls Genome Integrity and DNA Repair. Cell 2012, 149, 1023–1034. [Google Scholar] [CrossRef] [PubMed]
- Ribbeck, K.; Groen, A.C.; Santarella, R.; Bohnsack, M.T.; Raemaekers, T.; Köcher, T.; Gentzel, M.; Görlich, D.; Wilm, M.; Carmeliet, G.; et al. NuSAP, a Mitotic RanGTP Target That Stabilizes and Cross-Links Microtubules. Mol. Biol. Cell 2006, 17, 2646–2660. [Google Scholar] [CrossRef] [PubMed]
- Klein, D.K.; Hoffmann, S.; Ahlskog, J.K.; O’Hanlon, K.; Quaas, M.; Larsen, B.D.; Rolland, B.; Rösner, H.I.; Walter, D.; Kousholt, A.N.; et al. Cyclin F Suppresses B-Myb Activity to Promote Cell Cycle Checkpoint Control. Nat. Commun. 2015, 6, 5800. [Google Scholar] [CrossRef] [PubMed]
- Tetzlaff, M.T.; Bai, C.; Finegold, M.; Wilson, J.; Harper, J.W.; Mahon, K.A.; Elledge, S.J. Cyclin F Disruption Compromises Placental Development and Affects Normal Cell Cycle Execution. Mol. Cell. Biol. 2004, 24, 2487–2498. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Qiu, H.; Cai, M.; Pan, Y.; Cao, Y.; Liu, L.; Yun, J.; Zhang, C.Z. Low Cyclin F Expression in Hepatocellular Carcinoma Associates with Poor Differentiation and Unfavorable Prognosis. Cancer Sci. 2013, 104, 508–515. [Google Scholar] [CrossRef]
- Jackman, M.; Kubota, Y.; den Elzen, N.; Hagting, A.; Pines, J. Cyclin A- and Cyclin E-Cdk Complexes Shuttle between the Nucleus and the Cytoplasm. MBoC 2002, 13, 1030–1045. [Google Scholar] [CrossRef]
- Weng, L.; Du, J.; Zhou, Q.; Cheng, B.; Li, J.; Zhang, D.; Ling, C. Identification of Cyclin B1 and Sec62 as Biomarkers for Recurrence in Patients with HBV-Related Hepatocellular Carcinoma after Surgical Resection. Mol. Cancer 2012, 11, 39. [Google Scholar] [CrossRef] [PubMed]
- Faradji, F.; Bloyer, S.; Dardalhon-Cuménal, D.; Randsholt, N.B.; Peronnet, F. Drosophila Melanogaster Cyclin G Coordinates Cell Growth and Cell Proliferation. Cell Cycle 2011, 10, 805–818. [Google Scholar] [CrossRef]
- Horne, M.C.; Goolsby, G.L.; Donaldson, K.L.; Tran, D.; Neubauer, M.; Wahl, A.F. Cyclin G1 and Cyclin G2 Comprise a New Family of Cyclins with Contrasting Tissue-Specific and Cell Cycle-Regulated Expression. J. Biol. Chem. 1996, 271, 6050–6061. [Google Scholar] [CrossRef]
- Smith, M.L.; Kontny, H.U.; Bortnick, R.; Fornace, A.J. The P53-Regulated Cyclin G Gene Promotes Cell Growth: P53 Downstream Effectors Cyclin G and Gadd45 Exert Different Effects on Cisplatin Chemosensitivity. Exp. Cell Res. 1997, 230, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Kimura, S.H.; Nojima, H. Cyclin G1 Associates with MDM2 and Regulates Accumulation and Degradation of P53 Protein. Genes Cells 2002, 7, 869–880. [Google Scholar] [CrossRef] [PubMed]
- Ohtsuka, T.; Jensen, M.R.; Kim, H.G.; Kim, K.-T.; Lee, S.W. The Negative Role of Cyclin G in ATM-Dependent P53 Activation. Oncogene 2004, 23, 5405–5408. [Google Scholar] [CrossRef] [PubMed]
- Chen, X. Cyclin G: A Regulator of the P53-Mdm2 Network. Dev. Cell 2002, 2, 518–519. [Google Scholar] [CrossRef]
- Perez, R.; Wu, N.; Klipfel, A.A.; Beart, R.W. A Better Cell Cycle Target for Gene Therapy of Colorectal Cancer: Cyclin G. J. Gastrointest. Surg. 2003, 7, 884–889. [Google Scholar] [CrossRef] [PubMed]
- Reimer, C.L.; Borras, A.M.; Kurdistani, S.K.; Garreau, J.R.; Chung, M.; Aaronson, S.A.; Lee, S.W. Altered Regulation of Cyclin G in Human Breast Cancer and Its Specific Localization at Replication Foci in Response to DNA Damage in P53+/+ Cells. J. Biol. Chem. 1999, 274, 11022–11029. [Google Scholar] [CrossRef]
- Wu, L.; Liu, L.; Yee, A.; Carbonarohall, D.; Tolo, V.; Hall, F. Molecular-Cloning of the Human Cycg1 Gene Encoding a g-Type Cyclin - Overexpression in Human Osteosarcoma Cells. Oncol. Rep. 1994, 1, 705–711. [Google Scholar] [CrossRef]
- Skotzko, M.; Wu, L.; Anderson, W.F.; Gordon, E.M.; Hall, F.L. Retroviral Vector-Mediated Gene Transfer of Antisense Cyclin G1 (CYCG1) Inhibits Proliferation of Human Osteogenic Sarcoma Cells. Cancer Res. 1995, 55, 5493–5498. [Google Scholar]
- Okamoto, K.; Prives, C. A Role of Cyclin G in the Process of Apoptosis. Oncogene 1999, 18, 4606–4615. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Gao, X.; Yu, H.; Yuan, D.; Zhang, J.; He, Y.; Yue, L. Effects of Expression of Exogenous Cyclin G1 on Proliferation of Human Endometrial Carcinoma Cells. Chin. J. Physiol. 2013, 56, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Gao, X.; Yu, H.; Yuan, D.; Zhang, J.; He, Y.; Yue, L. The Role of Progesterone and Its Receptor on Cyclin G1 Expression in Endometrial Carcinoma Cells. Reprod. Sci. 2012, 19, 1205–1210. [Google Scholar] [CrossRef] [PubMed]
- Schneider, E.; Kartarius, S.; Schuster, N.; Montenarh, M. The Cyclin H/Cdk7/Mat1 Kinase Activity Is Regulated by CK2 Phosphorylation of Cyclin H. Oncogene 2002, 21, 5031–5037. [Google Scholar] [CrossRef] [PubMed]
- Eki, T.; Okumura, K.; Abe, M.; Kagotani, K.; Taguchi, H.; Murakami, Y.; Pan, Z.Q.; Hanaoka, F. Mapping of the Human Genes Encoding Cyclin H (CCNH) and the CDK-Activating Kinase (CAK) Assembly Factor MAT1 (MNAT1) to Chromosome Bands 5q13.3-Q14 and 14q23, Respectively. Genomics 1998, 47, 115–120. [Google Scholar] [CrossRef]
- Lolli, G.; Johnson, L.N. CAK-Cyclin-Dependent Activating Kinase: A Key Kinase in Cell Cycle Control and a Target for Drugs? Cell Cycle 2005, 4, 572–577. [Google Scholar] [CrossRef]
- Andersen, G. The Structure of Cyclin H: Common Mode of Kinase Activation and Specific Features. EMBO J. 1997, 16, 958–967. [Google Scholar] [CrossRef]
- Patel, S.A.; Simon, M.C. Functional Analysis of the Cdk7.Cyclin H.Mat1 Complex in Mouse Embryonic Stem Cells and Embryos. J. Biol. Chem. 2010, 285, 15587–15598. [Google Scholar] [CrossRef]
- Dorn, J.; Spatz, H.; Schmieder, M.; Barth, T.F.; Blatz, A.; Henne-Bruns, D.; Knippschild, U.; Kramer, K. Cyclin H Expression Is Increased in GIST with Very-High Risk of Malignancy. BMC Cancer 2010, 10, 350. [Google Scholar] [CrossRef] [PubMed]
- Bavi, P.; Abubaker, J.; Hussain, A.; Sultana, M.; Al-Dayel, F.; Uddin, S.; Al-Kuraya, K.S. Reduced or Absent Cyclin H Expression Is an Independent Prognostic Marker for Poor Outcome in Diffuse Large B-Cell Lymphoma. Hum. Pathol. 2008, 39, 885–894. [Google Scholar] [CrossRef] [PubMed]
- Patel, H.; Abduljabbar, R.; Lai, C.-F.; Periyasamy, M.; Harrod, A.; Gemma, C.; Steel, J.H.; Patel, N.; Busonero, C.; Jerjees, D.; et al. Expression of CDK7, Cyclin H, and MAT1 Is Elevated in Breast Cancer and Is Prognostic in Estrogen Receptor-Positive Breast Cancer. Clin. Cancer Res. 2016, 22, 5929–5938. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Sanokawa, R.; Sasaki, Y.F.; Ayusawa, D.; Oishi, M.; Mori, N. Cyclin I: A New Cyclin Encoded by a Gene Isolated from Human Brain. Exp. Cell Res. 1995, 221, 534–542. [Google Scholar] [CrossRef] [PubMed]
- Nagano, T.; Hashimoto, T.; Nakashima, A.; Hisanaga, S.; Kikkawa, U.; Kamada, S. Cyclin I Is Involved in the Regulation of Cell Cycle Progression. Cell Cycle 2013, 12, 2617–2624. [Google Scholar] [CrossRef] [PubMed]
- Griffin, S.V.; Olivier, J.P.; Pippin, J.W.; Roberts, J.M.; Shankland, S.J. Cyclin I Protects Podocytes from Apoptosis. J. Biol. Chem. 2006, 281, 28048–28057. [Google Scholar] [CrossRef]
- Liu, Y.; Tang, M.K.; Cai, D.Q.; Li, M.; Wong, W.M.; Chow, P.H.; Lee, K.K.H. Cyclin I and P53 Are Differentially Expressed during the Terminal Differentiation of the Postnatal Mouse Heart. Proteomics 2007, 7, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Liu, G.-Z.; Luo, S.-Y.; Chen, R.; Zhang, J.-X. Cyclin I Promotes Cisplatin Resistance via Cdk5 Activation in Cervical Cancer. Eur. Rev. Med. Pharmacol. Sci. 2015, 19, 4533–4541. [Google Scholar] [PubMed]
- Cybulski, M.; Jarosz, B.; Nowakowski, A.; Jeleniewicz, W.; Seroczyński, P.; Mazurek-Kociubowska, M. Cyclin I Correlates with VEGFR-2 and Cell Proliferation in Human Epithelial Ovarian Cancer. Gynecol. Oncol. 2012, 127, 217–222. [Google Scholar] [CrossRef]
- Finley, R.L.; Thomas, B.J.; Zipursky, S.L.; Brent, R. Isolation of Drosophila Cyclin D, a Protein Expressed in the Morphogenetic Furrow before Entry into S Phase. Proc. Natl. Acad. Sci. USA 1996, 93, 3011–3015. [Google Scholar] [CrossRef] [PubMed]
- Kolonin, M.G.; Finley, R.L. A Role for Cyclin J in the Rapid Nuclear Division Cycles of Early Drosophila Embryogenesis. Dev. Biol. 2000, 227, 661–672. [Google Scholar] [CrossRef]
- Atikukke, G.; Albosta, P.; Zhang, H.; Finley, R.L. A Role for Drosophila Cyclin J in Oogenesis Revealed by Genetic Interactions with the PiRNA Pathway. Mech. Dev. 2014, 133, 64–76. [Google Scholar] [CrossRef] [PubMed]
- Venturutti, L.; Cordo Russo, R.I.; Rivas, M.A.; Mercogliano, M.F.; Izzo, F.; Oakley, R.H.; Pereyra, M.G.; De Martino, M.; Proietti, C.J.; Yankilevich, P.; et al. MiR-16 Mediates Trastuzumab and Lapatinib Response in ErbB-2-Positive Breast and Gastric Cancer via Its Novel Targets CCNJ and FUBP1. Oncogene 2016, 35, 6189–6202. [Google Scholar] [CrossRef] [PubMed]
- Takano, N.; Hishida, M.; Inokawa, Y.; Hayashi, M.; Kanda, M.; Nishikawa, Y.; Iwata, N.; Kobayashi, D.; Tanaka, C.; Yamada, S.; et al. CCNJ Detected by Triple Combination Array Analysis as a Tumor-Related Gene of Hepatocellular Carcinoma. Int. J. Oncol. 2015, 46, 1963–1970. [Google Scholar] [CrossRef] [PubMed]
- Harvey, R.C.; Mullighan, C.G.; Wang, X.; Dobbin, K.K.; Davidson, G.S.; Bedrick, E.J.; Chen, I.-M.; Atlas, S.R.; Kang, H.; Ar, K.; et al. Identification of Novel Cluster Groups in Pediatric High-Risk B-Precursor Acute Lymphoblastic Leukemia with Gene Expression Profiling: Correlation with Genome-Wide DNA Copy Number Alterations, Clinical Characteristics, and Outcome. Blood 2010, 116, 4874–4884. [Google Scholar] [CrossRef]
- Shi, L.; Xu, Z.; Wu, G.; Chen, X.; Huang, Y.; Wang, Y.; Jiang, W.; Ke, B. Up-Regulation of MiR-146a Increases the Sensitivity of Non-Small Cell Lung Cancer to DDP by Downregulating Cyclin J. BMC Cancer 2017, 17, 138. [Google Scholar] [CrossRef]
- Althoff, F.; Viktorinová, I.; Kastl, J.; Lehner, C.F. Drosophila Cyclin J Is a Mitotically Stable Cdk1 Partner without Essential Functions. Dev. Biol. 2009, 333, 263–272. [Google Scholar] [CrossRef][Green Version]
- Lin, X.; Taube, R.; Fujinaga, K.; Peterlin, B.M. P-TEFb Containing Cyclin K and Cdk9 Can Activate Transcription via RNA. J. Biol. Chem. 2002, 277, 16873–16878. [Google Scholar] [CrossRef]
- Edwards, M.C.; Wong, C.; Elledge, S.J. Human Cyclin K, a Novel RNA Polymerase II-Associated Cyclin Possessing Both Carboxy-Terminal Domain Kinase and Cdk-Activating Kinase Activity. Mol. Cell. Biol. 1998, 18, 4291–4300. [Google Scholar] [CrossRef]
- Baek, K.; Brown, R.S.; Birrane, G.; Ladias, J.A.A. Crystal Structure of Human Cyclin K, a Positive Regulator of Cyclin-Dependent Kinase 9. J. Mol. Biol. 2007, 366, 563–573. [Google Scholar] [CrossRef]
- Kohoutek, J.; Blazek, D. Cyclin K Goes with Cdk12 and Cdk13. Cell Div. 2012, 7, 12. [Google Scholar] [CrossRef]
- Mori, T.; Anazawa, Y.; Matsui, K.; Fukuda, S.; Nakamura, Y.; Arakawa, H. Cyclin K as a Direct Transcriptional Target of the P53 Tumor Suppressor. Neoplasia 2002, 4, 268–274. [Google Scholar] [CrossRef]
- Blazek, D.; Kohoutek, J.; Bartholomeeusen, K.; Johansen, E.; Hulinkova, P.; Luo, Z.; Cimermancic, P.; Ule, J.; Peterlin, B.M. The Cyclin K/Cdk12 Complex Maintains Genomic Stability via Regulation of Expression of DNA Damage Response Genes. Genes Dev. 2011, 25, 2158–2172. [Google Scholar] [CrossRef]
- Blazek, D. The Cyclin K/Cdk12 Complex: An Emerging New Player in the Maintenance of Genome Stability. Cell Cycle 2012, 11, 1049–1050. [Google Scholar] [CrossRef] [PubMed]
- Schecher, S.; Walter, B.; Falkenstein, M.; Macher-Goeppinger, S.; Stenzel, P.; Krümpelmann, K.; Hadaschik, B.; Perner, S.; Kristiansen, G.; Duensing, S.; et al. Cyclin K Dependent Regulation of Aurora B Affects Apoptosis and Proliferation by Induction of Mitotic Catastrophe in Prostate Cancer. Int. J. Cancer 2017, 141, 1643–1653. [Google Scholar] [CrossRef] [PubMed]
- Loyer, P.; Trembley, J.H.; Grenet, J.A.; Busson, A.; Corlu, A.; Zhao, W.; Kocak, M.; Kidd, V.J.; Lahti, J.M. Characterization of Cyclin L1 and L2 Interactions with CDK11 and Splicing Factors: Influence of Cyclin L Isoforms on Splice Site Selection. J. Biol. Chem. 2008, 283, 7721–7732. [Google Scholar] [CrossRef]
- Yang, L.; Li, N.; Wang, C.; Yu, Y.; Yuan, L.; Zhang, M.; Cao, X. Cyclin L2, a Novel RNA Polymerase II-Associated Cyclin, Is Involved in Pre-MRNA Splicing and Induces Apoptosis of Human Hepatocellular Carcinoma Cells. J. Biol. Chem. 2004, 279, 11639–11648. [Google Scholar] [CrossRef]
- Dickinson, L.A.; Edgar, A.J.; Ehley, J.; Gottesfeld, J.M. Cyclin L Is an RS Domain Protein Involved in Pre-MRNA Splicing. J. Biol. Chem. 2002, 277, 25465–25473. [Google Scholar] [CrossRef]
- Zhou, Y.; Shen, J.K.; Hornicek, F.J.; Kan, Q.; Duan, Z. The Emerging Roles and Therapeutic Potential of Cyclin-Dependent Kinase 11 (CDK11) in Human Cancer. Oncotarget 2016, 7, 40846–40859. [Google Scholar] [CrossRef]
- Muller, D.; Millon, R.; Théobald, S.; Hussenet, T.; Wasylyk, B.; du Manoir, S.; Abecassis, J. Cyclin L1 (CCNL1) Gene Alterations in Human Head and Neck Squamous Cell Carcinoma. Br. J. Cancer 2006, 94, 1041–1044. [Google Scholar] [CrossRef] [PubMed]
- Redon, R.; Hussenet, T.; Bour, G.; Caulee, K.; Jost, B.; Muller, D.; Abecassis, J.; du Manoir, S. Amplicon Mapping and Transcriptional Analysis Pinpoint Cyclin L as a Candidate Oncogene in Head and Neck Cancer. Cancer Res. 2002, 62, 6211–6217. [Google Scholar]
- Sticht, C.; Hofele, C.; Flechtenmacher, C.; Bosch, F.X.; Freier, K.; Lichter, P.; Joos, S. Amplification of Cyclin L1 Is Associated with Lymph Node Metastases in Head and Neck Squamous Cell Carcinoma (HNSCC). Br. J. Cancer 2005, 92, 770–774. [Google Scholar] [CrossRef]
- Loyer, P.; Trembley, J.; Katona, R.; Kidd, V.; Lahti, J. Role of CDK/Cyclin Complexes in Transcription and RNA Splicing. Cell. Signal. 2005, 17, 1033–1051. [Google Scholar] [CrossRef]
- Guen, V.J.; Gamble, C.; Lees, J.A.; Colas, P. The Awakening of the CDK10/Cyclin M Protein Kinase. Oncotarget 2017, 8, 50174–50186. [Google Scholar] [CrossRef]
- Guen, V.J.; Gamble, C.; Perez, D.E.; Bourassa, S.; Zappel, H.; Gärtner, J.; Lees, J.A.; Colas, P. STAR Syndrome-Associated CDK10/Cyclin M Regulates Actin Network Architecture and Ciliogenesis. Cell Cycle 2016, 15, 678–688. [Google Scholar] [CrossRef]
- Dwyer, J.M.; Liu, J.-P. Ets2 Transcription Factor, Telomerase Activity and Breast Cancer. Clin. Exp. Pharmacol. Physiol. 2010, 37, 83–87. [Google Scholar] [CrossRef]
- Raouf, A.; Seth, A. Ets Transcription Factors and Targets in Osteogenesis. Oncogene 2000, 19, 6455–6463. [Google Scholar] [CrossRef] [PubMed]
- Guen, V.J.; Gamble, C.; Flajolet, M.; Unger, S.; Thollet, A.; Ferandin, Y.; Superti-Furga, A.; Cohen, P.A.; Meijer, L.; Colas, P. CDK10/Cyclin M Is a Protein Kinase That Controls ETS2 Degradation and Is Deficient in STAR Syndrome. Proc. Natl. Acad. Sci. USA 2013, 110, 19525–19530. [Google Scholar] [CrossRef] [PubMed]
- Unger, S.; Böhm, D.; Kaiser, F.J.; Kaulfuss, S.; Borozdin, W.; Buiting, K.; Burfeind, P.; Böhm, J.; Barrionuevo, F.; Craig, A.; et al. Mutations in the Cyclin Family Member FAM58A Cause an X-Linked Dominant Disorder Characterized by Syndactyly, Telecanthus and Anogenital and Renal Malformations. Nat. Genet. 2008, 40, 287–289. [Google Scholar] [CrossRef]
- Yu, J.-H.; Zhong, X.-Y.; Zhang, W.-G.; Wang, Z.-D.; Dong, Q.; Tai, S.; Li, H.; Cui, Y.-F. CDK10 Functions as a Tumor Suppressor Gene and Regulates Survivability of Biliary Tract Cancer Cells. Oncol. Rep. 2012, 27, 1266–1276. [Google Scholar] [CrossRef] [PubMed]
- Iorns, E.; Turner, N.C.; Elliott, R.; Syed, N.; Garrone, O.; Gasco, M.; Tutt, A.N.J.; Crook, T.; Lord, C.J.; Ashworth, A. Identification of CDK10 as an Important Determinant of Resistance to Endocrine Therapy for Breast Cancer. Cancer Cell 2008, 13, 91–104. [Google Scholar] [CrossRef]
- You, Y.; Bai, F.; Ye, Z.; Zhang, N.; Yao, L.; Tang, Y.; Li, X. Downregulated CDK10 Expression in Gastric Cancer: Association with Tumor Progression and Poor Prognosis. Mol. Med. Rep. 2018, 17, 6812–6818. [Google Scholar] [CrossRef] [PubMed]
- Weiswald, L.-B.; Hasan, M.R.; Wong, J.C.T.; Pasiliao, C.C.; Rahman, M.; Ren, J.; Yin, Y.; Gusscott, S.; Vacher, S.; Weng, A.P.; et al. Inactivation of the Kinase Domain of CDK10 Prevents Tumor Growth in a Preclinical Model of Colorectal Cancer, and Is Accompanied by Downregulation of Bcl-2. Mol. Cancer Ther. 2017, 16, 2292–2303. [Google Scholar] [CrossRef]
- Roig, M.B.; Roset, R.; Ortet, L.; Balsiger, N.A.; Anfosso, A.; Cabellos, L.; Garrido, M.; Alameda, F.; Brady, H.J.M.; Gil-Gómez, G. Identification of a Novel Cyclin Required for the Intrinsic Apoptosis Pathway in Lymphoid Cells. Cell Death Differ. 2009, 16, 230–243. [Google Scholar] [CrossRef] [PubMed]
- Funk, M.C.; Bera, A.N.; Menchen, T.; Kuales, G.; Thriene, K.; Lienkamp, S.S.; Dengjel, J.; Omran, H.; Frank, M.; Arnold, S.J. Cyclin O (Ccno) Functions during Deuterosome-Mediated Centriole Amplification of Multiciliated Cells. EMBO J. 2015, 34, 1078–1089. [Google Scholar] [CrossRef]
- Ma, J.-Y.; Ou-Yang, Y.-C.; Luo, Y.-B.; Wang, Z.-B.; Hou, Y.; Han, Z.-M.; Liu, Z.; Schatten, H.; Sun, Q.-Y. Cyclin O Regulates Germinal Vesicle Breakdown in Mouse Oocytes. Biol. Reprod. 2013, 88, 110. [Google Scholar] [CrossRef] [PubMed]
- Núnez-Ollé, M.; Jung, C.; Terré, B.; Balsiger, N.A.; Plata, C.; Roset, R.; Pardo-Pastor, C.; Garrido, M.; Rojas, S.; Alameda, F.; et al. Constitutive Cyclin O Deficiency Results in Penetrant Hydrocephalus, Impaired Growth and Infertility. Oncotarget 2017, 8, 99261–99273. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Cao, Y.; Zhou, H.; Li, Y.; He, B.; Zhou, X.; Nie, Z.; Liang, L.; Liu, Y.; Ye, L. Knockdown of CCNO Decreases the Tumorigenicity of Gastric Cancer by Inducing Apoptosis. OTT 2018, Volume 11, 7471–7481. [Google Scholar] [CrossRef]
- Napolitano, G.; Licciardo, P.; Gallo, P.; Majello, B.; Giordano, A.; Lania, L. The CDK9-Associated Cyclins T1 and T2 Exert Opposite Effects on HIV-1 Tat Activity. AIDS 1999, 13, 1453–1459. [Google Scholar] [CrossRef][Green Version]
- De Luca, A.; De Falco, M.; Baldi, A.; Paggi, M.G. Cyclin T: Three Forms for Different Roles in Physiological and Pathological Functions. J. Cell. Physiol. 2003, 194, 101–107. [Google Scholar] [CrossRef]
- De Luca, A.; Russo, P.; Severino, A.; Baldi, A.; Battista, T.; Cavallotti, I.; De Luca, L.; Baldi, F.; Giordano, A.; Paggi, M.G. Pattern of Expression of Cyclin T1 in Human Tissues. J. Histochem. Cytochem. 2001, 49, 685–692. [Google Scholar] [CrossRef]
- De Luca, A.; Tosolini, A.; Russo, P.; Severino, A.; Baldi, A.; De Luca, L.; Cavallotti, I.; Baldi, F.; Giordano, A.; Testa, J.R.; et al. Cyclin T2a Gene Maps on Human Chromosome 2q21. J. Histochem. Cytochem. 2001, 49, 693–698. [Google Scholar] [CrossRef]
- Peng, J.; Zhu, Y.; Milton, J.T.; Price, D.H. Identification of Multiple Cyclin Subunits of Human P-TEFb. Genes Dev. 1998, 12, 755–762. [Google Scholar] [CrossRef]
- Leucci, E.; De Falco, G.; Onnis, A.; Cerino, G.; Cocco, M.; Luzzi, A.; Crupi, D.; Tigli, C.; Bellan, C.; Tosi, P.; et al. The Role of the Cdk9/Cyclin T1 Complex in T Cell Differentiation. J. Cell. Physiol. 2007, 212, 411–415. [Google Scholar] [CrossRef]
- De Falco, G.; Leucci, E.; Onnis, A.; Bellan, C.; Tigli, C.; Wirths, S.; Cerino, G.; Cocco, M.; Crupi, D.; De Luca, A.; et al. Cdk9/Cyclin T1 Complex: A Key Player during the Activation/Differentiation Process of Normal Lymphoid B Cells. J. Cell. Physiol. 2008, 215, 276–282. [Google Scholar] [CrossRef]
- Simone, C.; Bagella, L.; Bellan, C.; Giordano, A. Physical Interaction between PRb and Cdk9/CyclinT2 Complex. Oncogene 2002, 21, 4158–4165. [Google Scholar] [CrossRef] [PubMed]
- Bellan, C.; De Falco, G.; Lazzi, S.; Micheli, P.; Vicidomini, S.; Schürfeld, K.; Amato, T.; Palumbo, A.; Bagella, L.; Sabattini, E.; et al. CDK9/CYCLIN T1 Expression during Normal Lymphoid Differentiation and Malignant Transformation. J. Pathol. 2004, 203, 946–952. [Google Scholar] [CrossRef]
- Liu, D.; Guest, S.; Finley, R.L. Why Cyclin Y? A Highly Conserved Cyclin with Essential Functions. Fly (Austin) 2010, 4, 278–282. [Google Scholar] [CrossRef]
- Mikolcevic, P.; Sigl, R.; Rauch, V.; Hess, M.W.; Pfaller, K.; Barisic, M.; Pelliniemi, L.J.; Boesl, M.; Geley, S. Cyclin-Dependent Kinase 16/PCTAIRE Kinase 1 Is Activated by Cyclin Y and Is Essential for Spermatogenesis. Mol. Cell. Biol. 2012, 32, 868–879. [Google Scholar] [CrossRef] [PubMed]
- Yue, W.; Zhao, X.; Zhang, L.; Xu, S.; Liu, Z.; Ma, L.; Jia, W.; Qian, Z.; Zhang, C.; Wang, Y.; et al. Cell Cycle Protein Cyclin Y Is Associated with Human Non-Small-Cell Lung Cancer Proliferation and Tumorigenesis. Clin. Lung Cancer 2011, 12, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Davidson, G.; Shen, J.; Huang, Y.-L.; Su, Y.; Karaulanov, E.; Bartscherer, K.; Hassler, C.; Stannek, P.; Boutros, M.; Niehrs, C. Cell Cycle Control of Wnt Receptor Activation. Dev. Cell 2009, 17, 788–799. [Google Scholar] [CrossRef] [PubMed]
- Yue, W.; Zhao, X.; Zhang, L.; Liu, Z.; Ma, L.; Jia, W.; Qian, Z.; Zhang, C.; Wang, Y.; Yang, X.; et al. Overexpression of Cyclin Y in Non-Small Cell Lung Cancer Is Associated with Cancer Cell Proliferation. Sci. China Life Sci. 2010, 53, 511–516. [Google Scholar] [CrossRef]
- Liu, H.; Shi, H.; Fan, Q.; Sun, X. Cyclin Y Regulates the Proliferation, Migration, and Invasion of Ovarian Cancer Cells via Wnt Signaling Pathway. Tumour Biol. 2016, 37, 10161–10175. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Wang, Z.; Wang, J.; Li, J.; Wang, H.; Yue, W. Lentivirus-Mediated Knockdown of Cyclin Y (CCNY) Inhibits Glioma Cell Proliferation. Oncol. Res. Featur. Preclin. Clin. Cancer Ther. 2009, 18, 359–364. [Google Scholar] [CrossRef] [PubMed]
- Tai, J.; Li, A.-D.; Rao, Y.-S.; Huang, Y.-B.; Huang, Z.-G.; Yu, Z.-K.; Chen, X.-H.; Zhou, W.-G.; Xiao, X.; Wang, S.; et al. [Influence on cell proliferation by small interfering RNA of Cyclin Y expression in laryngeal cancer cells]. Zhonghua Er Bi Yan Hou Tou Jing Wai Ke Za Zhi 2013, 48, 761–764. [Google Scholar] [PubMed]
- Shi, K.; Ru, Q.; Zhang, C.; Huang, J. Cyclin Y Modulates the Proliferation, Invasion, and Metastasis of Hepatocellular Carcinoma Cells. Med. Sci. Monit. 2018, 24, 1642–1653. [Google Scholar] [CrossRef]
- Yan, F.; Wang, X.; Zhu, M.; Hu, X. RNAi-Mediated Downregulation of Cyclin Y to Attenuate Human Breast Cancer Cell Growth. Oncol. Rep. 2016, 36, 2793–2799. [Google Scholar] [CrossRef][Green Version]


| Protein | Function in Normal Cells | References |
|---|---|---|
| Cyclin C | RNAPII transcription in complex with Cdk8 Transmission of the oxidative stress Cell’s exit from the G0 phase through phosphorylation of the retinoblastoma protein (Rb) in complex with Cdk3 Regulation of G1 and G2 phases | Ježek et al., 2019 [7] Ganesan et al., 2019 [10] Ren et al., 2004 [12] Liu cet al., 1998 [14] |
| Cyclin F | Synthesis, stability and repair of DNA Transcription Cell proliferation Embryogenesis | D’Angiolella et al., 2010 [24] D’Angiolella et al., 2012 [25] Klein et al., 2015 [27] Klein et al., 2015 [27] Tetzlaff et al., 2004 [28] |
| Cyclin G | Transcription The p53-Mdm2 pathway | Ohtsuka et al., 2004 [36] Chen et al., 2002 [37] |
| Cyclin H | Transcription in the CAK complex | Patel et al., 2010 [49] |
| Cyclin I | Cell cycle Protection of podocytes from apoptosis after mitosis Final arrest of growth of cardiomyocytes | Nagano et al., 2013 [54] Griffin et al., 2006[55] Liu et al., 2007 [56] |
| Cyclin J | Early embryogenesis in complex with Cdk2 Normal development of the ovaries | Kolonin et al., 2000 [60] Atikukke et al., 2014 [61] |
| Cyclin K | RNAPII transcription in complex with Cdk9, Cdk12 or Cdk13 DNA repair and genome stability in complex with Cdk12 | Mori et al., 2002 [71] Kohoutek et al., 2012 [70] Blazek et al., 2011 [72] |
| Cyclin L | Regulation of pre-mRNA splicing in complex with Cdk11 | Loyer et al., 2008 [75] |
| Cyclin M | Ciliogenesis proces in complex with Cdk10 Regulation of the protein C-ets-2 (ETS2) transcription factor in complex with Cdk10 | Guen et al., 2017[83] Guen et al., 2013 [87] |
| Cyclin O | Process of central cell amplification Development of oocytes Normal development of the body | Funk et al., 2015 [94] Ma et al., 2013 [95] Núnez-Ollé et al., 2017 [96] |
| Cyclin T | RNAPII transcription in complex with Cdk9 Differentiation of muscle cells in complex with Cdk9 Presentation and processing of antigen in complex with Cdk9 Cell proliferation in complex with Cdk9 Differentiation and activation B-cell in complex with Cdk9 | De Luca et al., 2001 [100] Peng et al., 1998 [102] De Luca et al., 2001 [101] De Falco et al., 2008 [104] |
| Cyclin Y | Wnt-B-catenin pathway in complex with Cdk14 Spermatogenesis in complex with Cdk16 | Davidson et al., 2009 [110] Mikolcevic et al., 2012 [108] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Żuryń, A.; Opacka, A.; Krajewski, A.; Zielińska, W.; Grzanka, A. The Less Known Cyclins—Uncovered. Appl. Sci. 2021, 11, 2320. https://doi.org/10.3390/app11052320
Żuryń A, Opacka A, Krajewski A, Zielińska W, Grzanka A. The Less Known Cyclins—Uncovered. Applied Sciences. 2021; 11(5):2320. https://doi.org/10.3390/app11052320
Chicago/Turabian StyleŻuryń, Agnieszka, Aleksandra Opacka, Adrian Krajewski, Wioletta Zielińska, and Alina Grzanka. 2021. "The Less Known Cyclins—Uncovered" Applied Sciences 11, no. 5: 2320. https://doi.org/10.3390/app11052320
APA StyleŻuryń, A., Opacka, A., Krajewski, A., Zielińska, W., & Grzanka, A. (2021). The Less Known Cyclins—Uncovered. Applied Sciences, 11(5), 2320. https://doi.org/10.3390/app11052320







